Abstract
Sjögren's syndrome (SS) is an autoimmune disease characterized by clonal B cell attack of the exocrine glands and dysregulated expression of B cell-activating factor (BAFF). Based upon the current data of increased rates of lymphoid malignancy, as non-Hodgkin's lymphoma (NHL) is associated with SS, we propose the detection of clonal rearrangements of immunoglobulin heavy chain (IgH) gene in those patients as a predictor of malignant clonal expansion. To test our proposal, we examined the IgH clonal rearrangements in SS patients (60) and healthy control subjects (42) having chronic non-specific sialadenitis, to determine the presence of clonal B cells in minor labial salivary glands (MSG) of SS patients. Clonal B cell expansion was assessed by two polymerase chain reaction (PCR) assays: (i) semi-nested PCR, against sequences encoding framework regions FR3, FR2 and FR1c of the variable chain IgH gene in B cells present in the MSG infiltrate; and (ii) the PCR–enzyme-linked immunosorbent assay (ELISA) technique, against the major and minor breakpoint regions of the Bcl-2 oncogene coupled with a variable segment of the IgH to assess the Bcl-2/JH translocation. When FR3, FR2 and FR1c primers were employed, we detected B cell monoclonality in 87% of the SS patients and 19% of the control subjects. The association between inflammation severity of the MSG pattern and the presence of B cell clonality was found to be statistically significant (P < 0·01). We concluded that the presence of B cell clonality in MSG can be used as a index of an altered microenvironment favouring the development of lymphoma in SS patients.
Keywords: B cell clonality, chronic sialadenitis, immunoglobulin heavy chain gene, PCR
Introduction
Sjögren's syndrome (SS) is a chronic autoimmune disease characterized by lymphocyte infiltration of the exocrine glands, the symptoms of which are eye and mouth dryness [1–3]. A key event occurring at the onset of SS development is polyclonal B cell activation leading to local production of cytokines and to increased titres of multiple circulating autoantibodies [2]. Recent studies have shown significant enhancement of B cell survival after the increase of the B cell activating factor (BAFF) levels – a family member of the tumour necrosis factor (TNF) – on the progression of SS [4].
Infiltrated glands are frequently the site of B cell oligoclonal and monoclonal expansion, an undesirable condition leading to lymphoid malignancy in >14% of SS cases [5,6]. In fact, a large number of SS patients develop B cell non-Hodgkin's lymphoma (NHL), associated mainly with mucosa-associated lymphoid tissue (MALT) lymphomas of primary gland origin, according to a concept introduced by Dong et al.[5], Tonami et al.[7] and Isaacson et al.[8]. Elevated serum levels of BAFF have been also found in patients with NHL [9]. Current studies have suggested a relationship between the detection rate of the immunoglobulin heavy chain gene (IgH) clonal rearrangement and the cellular origin of the lymphomas [10]. A high detection rate of clonal IgH gene rearrangement by polymerase chain reaction (PCR) is achieved in tumoral cells derived from naive lymphocytes – also known as pre-germinal centre (pre-GC) naive B cells – expressing the unmutated variable chain (VH) region [11]. Examples of this category are B lymphoblastic leukaemia, chronic lymphocytic leukaemia and mantle cell lymphoma [9,10]. Tumoral cells harbouring somatic mutations, derived from memory B cells generated in the germinal centres, show a low detection rate of clonality by PCR [10,11]. Examples of the last group are the majority of NHL, MALT lymphoma, multiple myeloma and Burkitt's lymphoma [12,13].
The detection of IgH gene rearrangements has been applied successfully to investigate the clonality and cell lineage of several other lymphoid malignancies and some autoimmune diseases, rheumatoid arthritis being a prominent example [5,13,14]. In these studies, the relatively conserved framework regions FR3, FR2 and FR1c – within the variable segment of IgH genes – have been targeted by PCR as useful markers for clonality of lymphoid malignancies of B cell lineage, with detection rates ranging from 50% to almost 99% [5,11,15–18].
We propose the detection of clonal rearrangements of the IgH gene as a predictor of malignant clonal expansion in SS patients.
In this paper we describe the development of a methodology to detect of IgH gene rearrangements in SS patients, and its further application in the prediction of malignant clonal expansion. To this end, clonal B cell expansion in minor labial salivary glands (MSG) infiltrates of SS patients was evaluated using a semi-nested PCR method [17,18]. To assess the suitability of this approach, the results were compared with those obtained from control patients exhibiting different degrees of chronic non-specific sialadenitis (CS), a non-specific inflammatory process of the salivary glands, characterized by diffuse lymphocytic infiltration [19,20]. Finally, we determined the risk of these patients in developing NHL through detection of the t(14;18) translocation by PCR [21,22].
Materials and methods
Patients and control subjects
All patients in the study were diagnosed according to the American European Consensus Group Criteria for SS [23]. The SS patients were divided into two groups; the first group comprised 48 primary SS patients (pSS), with different degrees of disease severity. Criteria included severity of keratoconjunctivitis sicca, xerostomia and the presence of autoantibodies, anti-Ro and anti-La antibodies. The second group comprised 12 secondary SS patients (sSS) positive for rheumatoid factor, anti-nuclear antibodies, as shown in Table 1. MSG biopsies were obtained from 102 patients in the study (five glands for each subject), using the technique described by Daniels [20]. The MSGs were classified according to histopathological detection of focal lymphocytic sialadenitis (FLS), as described by Daniels and Whitcher [20,24]. The biopsies were considered positive for disease if the focus score ≥ 1, defined as the number of lymphocytic foci per 4 mm2 of glandular tissue [24]. To preserve MSG before clonality analysis, biopsy samples were snap-frozen in liquid nitrogen and stored at −80°C (two glands for each subject).
Table 1.
Clinical laboratory and histological features of Sjögren's syndrome (SS) patients and controls subjects analysed in this study.
| Healthy control subjects (n = 2) | Control subjects with chronic non-specific sialadenitis* (n = 40) | Primary SS (n = 48) | Secondary SS (n = 12) | |
|---|---|---|---|---|
| Sex (male/female) | 0/2 | 1/40 | 2/46 | 2/10 |
| Age, mean (range) years | 36 (27–45) | 43 (16–67) | 46 (18–73) | 48·5 (38–60) |
| Focal lymphocytic sialadenitis (FS)† | ||||
| FS ≤ 1 (n = 16) | – | – | 10 | 6 |
| FS > 1 (n = 15) | – | – | 12 | 3 |
| FS 2–3 (n = 14) | – | – | 12 | 2 |
| FS ≥ 4 (n = 14) | – | – | 14 | 0 |
| Anti-nuclear antibodies | 0/2 | 0/40 | 40/46 | 6 |
| Ro/La antibodies | 0 | 0 | 28/20 | 0/0 |
| Rheumatoid factor | 0 | 0 | 6 | 12 |
Chronic non-specific sialadenitis.
Biopsy focus score (number of lymphocytic foci/4 mm2): abnormal biopsy (positive result), FS ≥ 1 and normal biopsy, FS < 1.
The control group (42 subjects) was diagnosed with non-specific chronic sialadenitis (not fulfilling the classification criteria for pSS), and was divided into three according to the inflammation pattern: (i) with normal biopsy (n = 2); (ii) with mild presence of diffuse infiltration lymphoid on lip biopsy (n = 20); or (iii) had moderate or severe sialadenitis defined as the presence of non-focal lymphoid infiltration (grade 2 according to the Chisholm and Mason scale [19]). All patients signed their informed consent before undergoing MSG biopsy. The study protocol was approved by the Indisa Clinic Ethics Committee.
DNA extraction
Genomic DNA from whole frozen MSG or NHL cells (clone CRL-2261; American Type Culture Collection, Manassas, VA, USA) were extracted using guanidine-detergent lysing solution (DNAzol® Reagents, Invitrogen, Carlsbad, CA, USA), according to the manufacturer's instructions. The NHL cells were used as a positive control to translocation t(14:18). The integrity of the extracted DNA was tested by amplification of the glyceraldehyde-3-phosphate dehydrogenase (GAPDH) human gene (Table 2).
Table 2.
Sequences of oligonucleotidic primers used in this study.
| Primer | Sequence |
|---|---|
| GDPH | |
| FW | 5′-GTC AAC GGA TTT GGT CTG TAT T-3′ |
| RV | 5′-AGT CTT CTG GGT GGC AGT GAT-3′ |
| V-region | |
| FR3 | 5′-ACA CGG C(C/T)(G/C) TGT ATT ACT GT-3′ |
| FR2 | 5′-TGG (A/G)TC CG(A/C) CAG SCY YCN GG-3′ |
| FR1c | 5′-AGG TGC AGC TG(C/G) (A/T)G(C/G) AGT C(A/T/G)GG-3′ |
| JH-region | |
| VLJH (outer) | 5′-TGA GGA GAC GGT GAC C-3′ |
| VLJH (inner) | 5′-GTG ACC A(G/T)G GT(C/G/T) CCT TGG CCC CAG-3′ |
| JH1245 | 5′-ACC TGA GGA GAC GGT GAC CAG GGT |
| JH3 | 5′-TAC CTG AAG AGA CGG TGA CCA TTG T |
| JH6 | 5′-ACC TGA GGA GAC GGT GAC CGT GGT |
| Bcl-2 region | |
| mbr | 5′-TTA GAC AGT TGC TTT ACG TGG CCT-3′ |
| mcr | 5′-GAC TCC TTT ACG TGC TGG TAC C-3′ |
Mbr, major breakpoint region; mcr, minor breakpoint region.
Detection of clonality and Bcl-2/JH translocation
VHDJH rearrangements were detected using a modified semi-nested PCR procedure on each sample to increase the assay sensitivity, using FR2/LJH-VLJH and conventional PCR to FR1c/JH1–6 primers [17,25,26]. All primers used in this study are listed in Table 2. All PCR runs were performed in an iCycler thermalcycler (Bio-Rad, Hercules, CA, USA), using platinum Taq polymerase (Invitrogen). In order to amplify using FR2/LJH primers, in the first PCR 50 ng genomic DNA were used and the reaction mix contained 1× PCR buffer, 200 µM 2′-deoxynucleosides 5′-triphosphate (dNTPs), 2 µM primers, 2 mM MgCl2, 0·001% gelatin and 1·5 U Taq DNA polymerase. The PCR conditions were initial denaturation at 95°C for 7 min followed by 40 cycles of the following parameters: denaturation, 94°C for 45 s; annealing, 50°C for 30 s; and extension, 72°C for 45 s. For the second round the reaction mixture contained 1 µl of the first PCR product and primers FR2 and VLJH. The cycling protocols to FR3/LJH were the same as FR2, with the exception of the annealing temperature (56°C). To amplify the Fr1c/JH1–6 primers, we employed the same reaction mix described above without gelatin and supplemented with 10% dimethylsulphoxide (DMSO), 1·25 U of Taq DNA polymerase and 50 ng of genomic DNA. The PCR conditions were the same as FR2, with the exception of 35 cycles and annealing temperature of 60°C. Samples in which DNA amplification was not clear were reamplified using the following specific primers: one directed to the FR1 region and the other to the JH region. PCR to amplify the GAPDH gene was performed under standard conditions, with the exception of an annealing temperature of 55°C. The specific primers are indicated in Table 2 and the samples were amplified as described above.
Bcl-2/JH translocation was analysed by a modified PCR–enzyme-linked immunosorbent assay (ELISA) technique (PharmaGen, Madrid, Spain), using primers directed to the major breakpoint region (mbr) and minor breakpoint region (mcr) of the bcl-2 oncogene coupled with LJH primer as indicated in Table 2[21]. Briefly, the PCR reactions were performed in similar conditions as described above, using 2′-deoxyuridine 5′-triphosphate (dUTP) digoxygenin instead of thymidine triphosphate (dTTP) and 100 ng of genomic DNA at an annealing temperature of 60°C. The amplified product was hybridized to a biotin-labelled probe and quantified by ELISA, according to the manufacturer's instructions. The PCR reaction was performed under standard conditions, as described above, under the following amplification conditions: initial denaturation at 95°C for 7 min followed by 30 cycles using the following parameters: denaturation, 94°C for 45 s; annealing, 56°C for 45 s; and extension, 72°C for 110 s. The PCR products were analysed on 3% agarose gels using the FR1c/JH1–6 or FR2/LJH-VLJH amplification protocol or 8% polyacrylamide gels using the FR3/LJH amplification protocol. Gels were photographed under ultraviolet light after staining with ethidium bromide or silver nitrate staining.
Sensitivity of IgH PCR
To determine the sensitivity of our IgH PCR method, we prepared serial 10-fold dilutions of the LM cell line (lymphoblastic lymphoma) in normal peripheral blood mononuclear cells (PBMC). For this purpose, 100–105 clonal B lymphocytes from the LM cell line were diluted with 105 PBMC. DNA obtained from these cells was purified and subjected to amplification using consensus primers directed to FR1c, FR2 and FR3 of the VH and JH regions (Fig. 2).
Fig. 2.
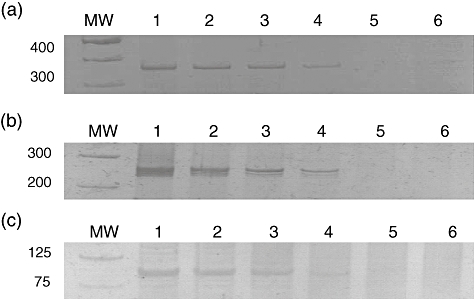
Sensitivity of polymerase chain reaction (PCR) technique for V–D–J rearrangement determination; 1, 10, 102, 103, 104 and 105 clonal B cells from lymphoblastic lymphoma (LM) cell lines were mixed with 105 normal peripheral blood mononuclear cells (PBMC). Lanes 1–6 correspond to the following clonal B cell/PBMC ratios: 1:1, 1:10, 1:1000, 1:10 000, 1:104 and 1:105 Sensitivity determinations using FR1c/JH1–6 primers (a); FR2/VLJH primers (b) and FR3/LJH primers (c).
Statistical method
We used χ2 tests or, if appropriate, Fisher's exact test to compare differences between groups with and without SS [27]. P-values < 0·01 were considered significant, with a confidence interval (CI) of 99%. Statistical analyses were performed using SigmaStat program version 1·02 (Systat Software Inc., Richmond, CA, USA).
Results
In this paper we propose that the detection of IgH gene rearrangements in MSG of SS patients is a predictor of malignant clonal expansion. To test our hypothesis, using PCR we analysed 102 DNA samples from whole MSG biopsies of SS patients and control subjects using FR2/LJH-VLJH, FR3/LJH and FR1c/JH1–6 primers (Table 2).
The results obtained in the clonality assay by PCR using different primers are shown in Table 3, where the clonal IgH gene rearrangement was found in 28 of 48 (58%) patients with pSS using FR3/LJH primers; one band of amplification was observed in the gel. The remaining 20 cases presented a polyclonal rearrangement and were observed as a smear in the gel (Fig. 1a). When FR2/LJH-VLJH primers were used, the clonal rearrangement was found in 79% of the pSS patients (Fig. 1b). Similar results were obtained in the sSS cases (Table 3 and Fig. 1c). Therefore, this analysis shows that patients with SS contained clonal B cell infiltrates in their MSG. When a polyclonal background was observed as a smear in the gel, the co-existence of polyclonal and monoclonal B cell populations was hypothesized to explain the results (Fig. 1).
Table 3.
Clonality frequency in labial salivary glands from Sjögren's syndrome (SS) and chronic non-specific sialadenitis (CS) patients; positivity obtained for immunoglobulin heavy chain (IgH) rearrangement and t(14;18) by polymerase chain reaction (PCR).
| pSS cases |
sSS cases |
Control cases |
||
|---|---|---|---|---|
| n | 48 | 12 | 42 | P-value* |
| FR3 (%) | 28/48 (58) | 6/12 (50) | 5/42 (14) | <0·01 |
| FR2 (%) | 38/48 (79) | 10/12 (75) | 7/42 (17) | <0·01 |
| FR1c (%) | 40/48 (83·4) | 12/12 (100) | 8/42 (19) | <0·01 |
| FR1c–FR2–FR3 (%) | 40/48 (83·4) | 12/12 (100) | 8/42 (19) | <0·01 |
| mbr (%) | 4/48 (8·3%) | n.d. | 0 (0) | |
| mcr (%) | 0/48 (0%) | n.d. | 0 (0) |
P-values were assessed using χ2 test (comparing proportions) between patients versus subjects control. Mbr, major breakpoint region; mcr, minor breakpoint region; n.d., not determined; pSS, primary; sSS, secondary SS.
Fig. 1.
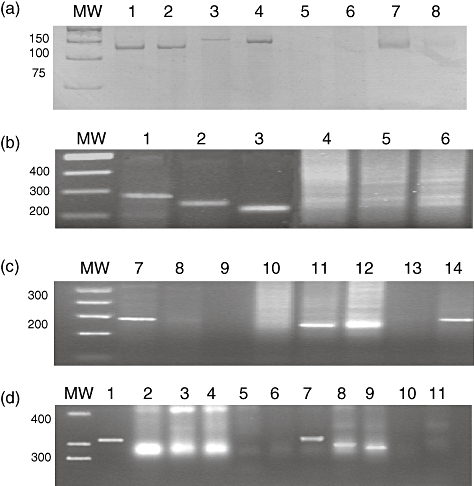
Semi-nested polymerase chain reaction (PCR) amplifications of DNA from labial salivary glands biopsies of Sjögren's syndrome (SS) patients and subject controls. (a) FR3-VLJH2 protocol: one band indicating monoclonal immunoglobulin heavy chain (IgH) rearrangement from pSS (lanes 1–4, 7 and 8) and a smear of bands corresponding to polyclonal IgH rearrangement from CS (lanes 5, 6); (b,c) FR2-VLJH protocol: one or two bands indicating monoclonal IgH rearrangement from SS (b, lanes 1–3; c, lanes 7, 8 and 12) and a smear of bands corresponding to polyclonal B cell expansion (b, lanes 4–6) or containing polyclonal and clonal IgH rearrangement from CS (c, lanes 9–11). Negative control in lane 13 and positive control, lane 14; (d) FR1c/JH1–6 protocol: one or two bands indicating monoclonal IgH rearrangement from primary SS (pSS) (lanes 2–4) and from secondary SS (sSS) (lanes 7–9) and weak smear or bands corresponding to polyclonal B and clonal B cell expansion from chronic non-specific sialadenitis (CS) (lanes 5, 6 and 11). Positive control in lane 1 and negative control in lane 10. MW: molecular weight marker.
The FR2/LJH-VLJH primers amplified successfully a higher proportion of cases with SS than FR3/LJH primers, as shown in Table 3. To assess the false negative results, all the cases were analysed with FR1c/JH1–6 primers (Table 3 and Fig. 1d). We should point out that after use of the three sets of primers, the clonality detection rate reached 86·7% in SS patients (pSS and sSS), as indicated in Table 3.
Nineteen per cent of the control subjects exhibited oligo-monoclonal bands with similar PCR amplification and histopathological analysis of the gland exhibited different degrees of CS. A strong polyclonal cell background was observed in the eight PCR-positive cases. The level of amplification was notably lower than in all the cases with SS (Fig. 1). The number of positive cases for the presence of clonal expansion in MSG from SS patients was very high compared with the control cases without SS (86·7 versus 19%, P < 0·01; χ2 test). Translocation t(14;18) was observed in 8·3% of the cases with pSS (Table 3).
In addition, we demonstrated that our IgH PCR method was highly sensitive to detected clonal cells. This PCR method was able to detect 102 clonal cells in 105 PBMC, using the three consensus regions (Fig. 2).
In addition, we wanted to analyse the relationship between the degree of FLS in SS patients and different degrees of CS in control subjects with the presence of clonal B cells in MSG. Histopathological analysis of the MSG biopsies from 48 patients with pSS showed a different degree of FLS, defined as focus score, for those with normal biopsy, or abnormal, as indicated in Table 1. Histopathological analysis of labial biopsies of 40 control subjects show different degrees of CS, as shown in Table 4. We observed that 40% of pSS patients with FLS < 1 showed clonal IgH rearrangements compared with patients who had an abnormal biopsy (FLS ≥ 1), in some cases reaching 100%, as shown in Table 4. This difference was statistically significant (P < 0·01; χ2 test, 99% CI). In addition, we determined that 83·4% of the cases with pSS presented an oligo–monoclonal IgH rearrangement compared with 19% of the cases diagnosed with CS. There was a high correlation in control cases between the severity of CS and the presence of B cell clonality (Table 4). Seven cases with severe CS showed B cell oligo–monoclonality compared with those diagnosed with mild to intermediate CS (87·5 versus 3·1%; P < 0·01; χ2 test). The biopsy was completely normal in only two cases and we did not detect a clonal IgH gene rearrangement by PCR (Table 4).
Table 4.
Relationship between histopathological analysis and B cell clonality in focal lymphocytic sialadenitis (primary Sjögren's syndrome) and chronic non-specific sialadenitis.
| Histopathology diagnosis | Monoclonal IgH gene rearrangement (%) |
|---|---|
| (a) Normal (n = 2) | 0/2(0) |
| (b) Chronic non-specific sialadenitis*(n = 40) | |
| Mild (n = 20) | 0/20(0) |
| Moderate (n = 12) | 1/12 (8·3) |
| Mild + intermediate (n = 32) | 1/32 (3·1)† |
| Severe (n = 8) | 7/8 (87·5)† |
| Total cases | 8/42 (19)† |
| (c) Focal lymphocytic sialadenitis‡ (n = 48) | |
| Focus score <1 (n = 10) | 4/10 (40) |
| Focus score ≥1 (n = 12) | 10/12 (83) |
| Focus score 2–3 (n = 12) | 12/12 (100) |
| Focus score ≥4 (n = 14) | 14/14 (100) |
| Total cases | 40/48 (83·3) |
No lymphocytic infiltrate.
P < 0·01.
Biopsy focus score (FS) (number of lymphocytic foci/4 mm2): abnormal biopsy (positive result), FS ≥ 1 and normal biopsy, FS < 1. IgH, immunoglobulin heavy chain.
Discussion
Our results showed 58% and 79% of B cell clonality or oligoclonality, respectively, in the MSG of SS patients using FR3/LJH and FR2/LJH-VLJH primers. Similar results have been reported in the literature, where 77% of cases with NHL were PCR-positive, arguing that the low detection of clonal B cells is due to partial rearrangements, inversions, somatic mutations or deletions that can be missed by PCR [26]. The addition of FR1c-LJH primers to our PCR analysis allowed a higher detection rate of SS cases, as reported previously by Aubin and co-workers [17]. Therefore, the use of the three sets of primers diminished the false negative results and improved the detection rate in 86·7% of the SS patients (Table 3). Also, we observed that the addition of FR3 did not increase the number of positive cases, therefore the failure of FR3 or FR2 to detect clonality in some cases could be the acquisition of somatic mutations in the primer target sequences, due to mispriming during the PCR [11,12,15,26]. Another possibility is that the IgH gene rearrangement is related closely to the cellular origin involved in the lymphoid pathology. In these cases, absence of clonality for the FR3-VLJH primers would indicate the presence of post-GC B cells or memory B cells in the salivary glands, characterized by cells bearing somatically hypermutated VH genes, as has been found in a series of studies in NHL and MALT [10,28,29].
It has been determined that patients with SS have a 16-fold increased risk of developing lymphoma [5,30]. Several studies have suggested that lympho-epithelial lesions in SS patients show a high presence of clonal expansion of B cells, as determined by molecular analysis of the IgH rearrangement, morphological or immunophenotypic determination. The possibility that detection of clonal B cells by PCR may help to distinguish lymphoma from benign lympho-epithelial lesions (BLEL) remains controversial. In a previous study, 100% of labial salivary gland (LSG) specimens of SS patients exhibited monoclonal IgH gene rearrangements by PCR, and only one patient with lymphoma displayed a different IgH gene rearrangement in the tumour and LSG [28,31–33]. Conversely, it was reported that clonality was evident in 15% of MSG specimens detected by PCR in pSS patients: four of 11 patients developed extrasalivary lymphoma and in all the cases the rearranged bands in the biopsy and the lymphoma were the same size [33]. In this context, it is now established that the risk of lymphoma progression is high if the same B cell clone is detected in different tissues at different times [33]. In a recent study, Dong et al.[5] analysed B cell clonality over the CDR3 region of IgH by sequence analysis in SS patients; they observed the presence of expansion of the same B cell clones in different sites (lacrymal glands and MSG) during the course of SS. It has been suggested that monoclonal B cell populations could spread from one site to another during the progression of the disease [5]. One possible explanation for this phenomenon is the enhancement of monoclonal B cell proliferation in the microenvironment of lacrymal gland, MSG or lymph nodes in SS patients, because the same clone has been identified in different tissues during the course of disease [5]. Moreover, some researchers have suggested that these B cell clones, present in BLEL, evolved to malignant lymphoma probably because of additional genetic events on the basis of chronic antigen stimulation [34–36]. It is possible that the intense proliferation of B cell lymphocytes in the ectopic GC microenvironment in salivary glands of SS patients precludes the recombination of the variable gene region, and therefore are responsible for the B cell monoclonal expansion of hypermutated B cells. All the above events could play a key role in neoplastic transformation [10]. Their role in tumorigenesis is less clear [12,36,37]. Recent findings suggest that ectopic lymphoid neogenesis in the CG in SS with dense B cell aggregates in salivary glands may indicate subsequent neoplastic transformation, as well as other factors related to BAFF-expression dysregulation [4]. In our cohort, we detected a clonal rearrangement by PCR in 52 patients with SS, where two patients developed a salivary gland MALT lymphoma determined by pathological diagnosis after of 5 years of disease duration; one t(14;18)-positive patient developed benign IgG-k class monoclonal gammopathy and showed some clinical signs, such as swollen salivary glands and low levels of C3 and C4, described as laboratory predictors [30]. The remaining patients have not developed clinical lymphoma, even 8 years from the first reported symptoms of the disease. However, it is unknown if patients containing clonal cells in MSG may develop lymphoma in the future. The diagnostician should interpret the t(14:18) aberration carefully, because it is also detected at a low level in healthy individuals, suggesting that factors such as age, smoking habits or dioxin exposure are associated with that translocation [38].
Using DNA-cytometric analysis, Ihrler et al.'s [37] study described the presence of chromosomal alterations in salivary gland MALT lymphoma in SS. Regarding the key role of BAFF in SS proposed by some authors [4,39], the assessment of BAFF levels in serum is an exciting field for future research.
Our study showed a high prevalence (86·7%) of B cell clonality in patients with SS and a direct relationship with the degree of focal lymphocytic infiltrates. In healthy control groups, we observed a direct correlation between the degree of CS and the presence of oligo- or monoclonal bands. Therefore, this study supported the hypothesis that an increasing number of patients with different degrees of CS may result in clonal B infiltration of the gland, showing an association between the severity of the MSG inflammation pattern and the presence of clonality. The finding of clonality in samples from this group of individuals is interesting, and possible explanations of these results are: (i) the development of reactive clonal population, distributed widely in the salivary glands, as has been reported in other studies [33,34]; and (ii) PCR is a very sensitive technique, and could detect a few cells among a normal cellular background.
According to our results, we show in this paper that the detection of B cell clonality by PCR in MSG of SS patients is a predictor of clonal expansion. Clonal expansion during chronic gland inflammation of B cell mutations takes place regularly, accompanied by mutations of tumour-suppressor genes, p53 mutations and a high level of BAFF expression. Together, these alterations constitute a risk factor for the development of lymphoma in SS patients [4–6,29,30,34,40]. We conclude that the presence of B cell clonality in MSG can be used as an index of an altered microenvironment, which could enable the development of lymphoma in SS patients.
Acknowledgments
This research was supported by funds of the Public Institute of Health from Chile, Bagó Laboratory and Chile Laboratory.
Disclosure
All authors declare no conflicts of interest.
References
- 1.Fox RI, Howell FV, Bone RC, Michelson P. Primary Sjögren syndrome: clinical and immunopathologic features. Semin Arthritis Rheum. 1984;14:77–105. doi: 10.1016/0049-0172(84)90001-5. [DOI] [PubMed] [Google Scholar]
- 2.Anaya J-M, Vega P, Correa PA, et al. Síndrome de Sjögren. In: Anaya JM, Shoenfeld Y, Correa PA, García-Carrasco M, Cervera R, editors. Autoinmunidad y Enfermedad Autoinmune [Autoimmunity and Autoimmune Disease] Medellin: Biological Research Corporation; 2005. pp. 295–315. [Google Scholar]
- 3.Martin T, Weber JC, Levallois H, et al. Salivary gland lymphomas in patients with Sjögren's syndrome may frequently develop from rheumatoid factor B cells. Arthritis Rheum. 2000;43:908–16. doi: 10.1002/1529-0131(200004)43:4<908::AID-ANR24>3.0.CO;2-K. [DOI] [PubMed] [Google Scholar]
- 4.Szodoray P, Jonsson R. The BAFF/APRIL system in systemic autoimmune diseases with a special emphasis on Sjögren's syndrome. Scand J Immunol. 2005;62:421–8. doi: 10.1111/j.1365-3083.2005.01688.x. [DOI] [PubMed] [Google Scholar]
- 5.Dong L, Masaki Y, Takegami T, et al. Clonality analysis of lymphoproliferative disorders in patients with Sjögren's syndrome. Clin Exp Immunol. 2007;150:279–84. doi: 10.1111/j.1365-2249.2007.03486.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kassan SS, Thomas TL, Moutsopoulos HM, et al. Increased risk of lymphoma in sicca syndrome. Ann Intern Med. 1978;89:888–92. doi: 10.7326/0003-4819-89-6-888. [DOI] [PubMed] [Google Scholar]
- 7.Tonami H, Matoba M, Yokota H, Higashi K, Yamamoto I, Sugai S. Mucosa-associated lymphoid tissue lymphoma in Sjögren's syndrome: initial and follow-up imaging features. Am J Roentgenol. 2002;179:485–9. doi: 10.2214/ajr.179.2.1790485. [DOI] [PubMed] [Google Scholar]
- 8.Isaacson P, Wright DH. Malignant lymphoma of mucosa-associated lymphoid tissue: a distinctive type of B-cell lymphoma. Cancer. 1983;52:1410–16. doi: 10.1002/1097-0142(19831015)52:8<1410::aid-cncr2820520813>3.0.co;2-3. [DOI] [PubMed] [Google Scholar]
- 9.Briones J, Timmerman JM, Hilbert DM, Levy R. BLyS and BLyS receptor expression in non-Hodgkin's lymphoma. Exp Hematol. 2002;30:135–41. doi: 10.1016/s0301-472x(01)00774-3. [DOI] [PubMed] [Google Scholar]
- 10.Küppers R, Klein U, Hansmann ML, Rajewsky K. Cellular origin of human B-cell lymphomas. N Engl J Med. 1999;341:1520–9. doi: 10.1056/NEJM199911113412007. [DOI] [PubMed] [Google Scholar]
- 11.Thériault C, Galoin S, Valmary S, et al. PCR analysis of immunoglobulin heavy chain (IgH) and TCR-g chain gene rearrangements in the diagnosis of lymphoproliferative disorders: results of a study of 525 cases. Mod Pathol. 2000;13:1269–79. doi: 10.1038/modpathol.3880232. [DOI] [PubMed] [Google Scholar]
- 12.Fais F, Ghiotto F, Hashimoto S, et al. Chronic lymphocytic leukemia B cells express restricted sets of mutated and unmutated antigen receptors. J Clin Invest. 1998;102:1515–25. doi: 10.1172/JCI3009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Itoh K, Patki V, Furie RA, et al. Clonal expression is a characteristic feature of the B-cell repertoire of patients with rheumatoid arthritis. Arthritis Res. 2000;2:50–8. doi: 10.1186/ar68. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Retamales E, Rodriguez L, Guzmán L, et al. Analytical detection of immunoglobulin heavy chain gene rearrangements in gastric lymphoid infiltrates by peak area analysis of the melting curve in the LightCycler System. J Mol Diagn. 2007;9:351–7. doi: 10.2353/jmoldx.2007.050129. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Arnold A, Cossman J, Bakhshi A, Jaffe ES, Waldmann TA, Korsmeyer SJ. Immunoglobulin–gene rearrangements as unique clonal markers in human lymphoid neoplasms. N Engl J Med. 1983;309:1593–9. doi: 10.1056/NEJM198312293092601. [DOI] [PubMed] [Google Scholar]
- 16.Segal GH, Jorgensen T, Masih AS, Braylan RC. Optimal primer selection for clonality assessment by polymerase chain reaction analysis: I. Low grade B-cell lymphoproliferative disorders of nonfollicular center cell type. Hum Pathol. 1994;25:1269–75. doi: 10.1016/0046-8177(94)90084-1. [DOI] [PubMed] [Google Scholar]
- 17.Aubin J, Davi F, Nguyen-Salomon F, et al. Description of a novel FR1 IgH PCR strategy and its comparison with three other strategies for the detection of clonality in B cell malignancies. Leukemia. 1995;9:471–9. [PubMed] [Google Scholar]
- 18.Uchiyama M, Maesawa C, Yashima A, et al. Development of consensus fluorogenically labeled probes of the immunoglobulin heavy-chain gene for detecting minimal residual disease in B-cell non-Hodgkin lymphomas. Cancer Sci. 2003;94:877–85. doi: 10.1111/j.1349-7006.2003.tb01370.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Chisholm DM, Mason DK. Labial salivary gland biopsy in Sjögren's disease. J Clin Pathol. 1968;21:656–60. doi: 10.1136/jcp.21.5.656. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Daniels TE, Whitcher JP. Association of patterns of labial salivary gland inflammation with keratoconjunctivitis sicca. Analysis of 618 patients with suspected Sjögren's syndrome. Arthritis Rheum. 1994;37:869–77. doi: 10.1002/art.1780370615. [DOI] [PubMed] [Google Scholar]
- 21.Crescenzi M, Seto M, Herzig GP, Weiss PD, Griffith RC, Korsmeyer SJ. Thermostable DNA polymerase chain amplification of t(14;18) chromosome breakpoints and detection of minimal residual disease. Proc Natl Acad Sci USA. 1988;85:4869–73. doi: 10.1073/pnas.85.13.4869. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Ngan BY, Nourse J, Cleary ML. Detection of chromosomal translocation t(14;18) within the minor cluster region of bcl-2 by polymerase chain reaction and direct genomic sequencing of the enzymatically amplified DNA in follicular lymphomas. Blood. 1989;73:1759–62. [PubMed] [Google Scholar]
- 23.Vitali C, Bombardieri S, Jonsson R, et al. Classification criteria for Sjögren's syndrome: a revised version of the European criteria proposed by the American–European Consensus Group. Ann Rheum Dis. 2002;61:554–8. doi: 10.1136/ard.61.6.554. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Daniel TE. Labial salivary gland biopsy in Sjögren's syndrome. Assessment as a diagnostic criterion in 362 suspected cases. Arthritis Rheum. 1984;27:147–56. doi: 10.1002/art.1780270205. [DOI] [PubMed] [Google Scholar]
- 25.Diss TC, Pan L, Wotherspoon AC, Isaacson PG. Sources of DNA for detecting B cell monoclonality using PCR. J Clin Pathol. 1994;47:493–6. doi: 10.1136/jcp.47.6.493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.De Vita S, Ferraccioli G, De Re V, et al. The polymerase chain reaction detects B cell clonalities in patients with Sjögren's syndrome and suspected malignant lymphoma. J Rheumatol. 1994;21:1497–501. [PubMed] [Google Scholar]
- 27.Fisher RA. On the interpretation of χ2 from contingency tables, and the calculation of P. J R Stat Soc. 1922;85:87–94. [Google Scholar]
- 28.Bahler DW, Miklos JA, Swerdlow SH. Ongoing Ig gene hypermutation in salivary gland mucosa-associated lymphoid tissue-type lymphomas. Blood. 1997;89:3335–44. [PubMed] [Google Scholar]
- 29.Kato K, Ohshima K, Shiokawa S, Shibata T, Suzumiya J, Kikuchi M. Rearrangement of immunoglobulin heavy and light chains and VH family in thyroid and salivary gland lymphomas. Pathol Int. 2002;52:747–54. doi: 10.1046/j.1440-1827.2002.01418.x. [DOI] [PubMed] [Google Scholar]
- 30.Theander E, Henriksson G, Ljungberg O, Mandl T, Manthorpe R, Jacobsson LT. Lymphoma and other malignancies in primary Sjögren's syndrome: a cohort study on cancer incidence and lymphoma predictors. Ann Rheum Dis. 2006;65:796–803. doi: 10.1136/ard.2005.041186. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Trainor KJ, Brisco MJ, Story CJ, Morley AA. Monoclonality in B-lymphoproliferative disorders detected at the DNA level. Blood. 1990;75:2220–2. [PubMed] [Google Scholar]
- 32.Pablos JL, Carreira PE, Morillas L, Montalvo G, Ballestin C, Gómez-Reino JJ. Clonally expanded lymphocytes in the minor salivary glands of Sjögren's syndrome patients without lymphoproliferative disease. Arthritis Rheum. 1994;37:1441–4. doi: 10.1002/art.1780371006. [DOI] [PubMed] [Google Scholar]
- 33.Jordan RC, Odell EW, Speight PM. B-cell monoclonality in salivary lymphoepithelial lesions. Eur J Cancer B Oral Oncol. 1996;32B:38–44. doi: 10.1016/0964-1955(95)00050-x. [DOI] [PubMed] [Google Scholar]
- 34.Mariette X. Lymphomas complicating Sjögren's syndrome and hepatitis C virus infection may share a common pathogenesis: chronic stimulation of rheumatoid factor B cells. Ann Rheum Dis. 2001;60:1007–10. doi: 10.1136/ard.60.11.1007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Fishleder A, Tubbs R, Hesse B, Levine H. Uniform detection of immunoglobulin-gene rearrangement in benign lymphoepithelial lesions. N Engl J Med. 1987;316:1118–21. doi: 10.1056/NEJM198704303161803. [DOI] [PubMed] [Google Scholar]
- 36.Knowles DM, Athan E, Ubriaco A, et al. Extranodal noncutaneous lymphoid hyperplasias represent a continuous spectrum of B-cell neoplasia: demonstration by molecular genetic analysis. Blood. 1989;73:1635–45. [PubMed] [Google Scholar]
- 37.Ihrler S, Baretton GB, Menauer F, Blasenbreu-Vogt S, Löhrs U. Sjögren's syndrome and MALT lymphomas of salivary glands: a DNA-cytometric and interphase-cytogenetic study. Mod Pathol. 2000;13:4–12. doi: 10.1038/modpathol.3880002. [DOI] [PubMed] [Google Scholar]
- 38.Schüler F, Hirt C, Dölken G. Chromosomal translocation t(14;18) in healthy individuals. Semin Cancer Biol. 2003;13:203–9. doi: 10.1016/s1044-579x(03)00016-6. [DOI] [PubMed] [Google Scholar]
- 39.Jonsson MV, Szodoray P, Jellestad S, et al. Association between circulating levels of the novel TNF family members APRIL and BAFF and lymphoid organization in primary Sjögren's syndrome. J Clin Immunol. 2005;25:189–201. doi: 10.1007/s10875-005-4091-5. [DOI] [PubMed] [Google Scholar]
- 40.Tapinos NI, Polihronis M, Moutsopoulos HM. Lymphoma development in Sjögren's syndrome. Novel p53 mutations. Arthritis Rheum. 1999;42:1466–72. doi: 10.1002/1529-0131(199907)42:7<1466::AID-ANR21>3.0.CO;2-L. [DOI] [PubMed] [Google Scholar]


