Abstract
Several T cell abnormalities have been described in common variable immunodeficiency (CVID), a B cell disorder of mainly unknown origin. A subset of CVID patients suffers from frequent reactivations of herpes viruses. We studied T cell function in CVID [and in a subset of paediatric patients with specific antibody deficiency (SAD)] by measuring T cell proliferation and cytokine production in response to herpes virus-antigens in paediatric CVID patients (n = 9) and paediatric SAD patients (n = 5), in adult CVID patients (n = 14) and in healthy controls. Paediatric CVID patients, but not SAD patients, displayed moderately increased CD8+ T cell proliferation in response to cytomegalovirus, human herpes virus type 6B (HHV6-B) and herpes simplex virus compared to controls. CD8+ T cell responses in adult CVID patients tended to be increased in response to cytomegalovirus and herpes simplex virus. In response to stimulation with herpes virus antigens, the proinflammatory cytokines interleukin (IL)-1β, IL-6, tumour necrosis factor (TNF)-α and interferon inducible protein (IP)-10 were produced. Overall, no major differences were detected in cytokine production upon stimulation between patients and controls, although higher IL-10 and IL-12 production was detected in paediatric patients. In conclusion, cellular immunity against herpes virus antigens appears undisturbed in CVID patients, although defects in subpopulations of CVID patients cannot be excluded.
Keywords: herpes virus antigens, paediatric and adult CVID patients, T cell function
Introduction
Common variable immunodeficiency (CVID) is a heterogeneous group of primary immunodeficiencies characterized by low serum immunoglobulin concentrations, decreased production of specific antibodies upon vaccinations, increased susceptibility to infections, autoimmunity and malignancies [1,2]. Four monogenic defects associated with CVID have been identified in the genes encoding inducible T cell co-stimulator (ICOS), transmembrane activator and CAML interactor (TACI), B cell (lymphocyte) activating factor receptor (BAFF-R) and CD19 [3,4]. Recently, deficiencies in the genes encoding CD20 and CD81 have been reported [5,6]. Deficiencies in these molecules disrupt B cell differentiation and B cell function at different stages. However, no genetic defect has been found in the majority of patients.
Although the phenotypic defect in CVID is a failure of B cell function, it has been shown that B cells in some CVID patients can proliferate and secrete normal amounts of immunoglobulins if stimulated appropriately in vitro[7–11]. Therefore, T cell abnormalities resulting in decreased or absent contact-mediated B cell activation may be of importance for the failure of B cells to differentiate into immunoglobulin-secreting plasma cells. Approximately half the CVID patients show laboratory abnormalities in the T cell compartment [12,13]. These abnormalities include decreased lymphocyte proliferation [14–17], impaired cytokine production to mitogens and recall-antigens [10,18–21], changes in CD4/CD8 ratios [22,23] and reduced expression of CD40L on activated T cells [7,24]. Several studies describe defects in the T cell receptor (TCR). In patients with T cell dysfunction, tumour necrosis factor (TNF)-receptor 2 co-stimulation was impaired [25]. Also, defective recruitment and activation of protein ZAP70 [26] and a defect in Vav-expression [27] have been identified. All these factors are important for intact signalling after binding to the TCR. Recently, low numbers of regulatory T cells have been found to be associated with chronic inflammation in CVID [28,29].
Most CVID patients are susceptible to recurrent bacterial infections due to diminished specific antibody production. However, in patients with concurrent disturbances in T lymphocytes, an increased number of viral infections could also be detected [8]. In particular, viral infections and reactivations with herpes viruses have been described, including Epstein–Barr virus (EBV) and cytomegalovirus (CMV) infections [30,31]. One study described the EBV- and CMV-specific T cell responses in CVID patients and showed that about half the patients have increased numbers of CMV- (and to a lesser extent EBV-) specific CD8+ T cells. An association was found between a high percentage of CD8+CD57+CD27– T cells containing perforin, a low CD4/CD8 ratio and CMV infection [32]. Also, strong correlations have been found in healthy subjects between raised numbers of CD8+highCD57+ lymphocytes and asymptomatic CMV infection [33]. These results may support the hypothesis that CMV plays an important role in the origination of T cell abnormalities associated with CVID, at least in a subset of patients. Human herpes virus type 8 may play a role in the existence of lymphoproliferative disorders in CVID patients [34], and abnormal courses of infections with herpes simplex virus (HSV) have also been described [35,36]. Besides the limited data on T cell responses to EBV and CMV, little is known about T cell-mediated immunity to viral antigens in CVID patients.
We hypothesized that specific T cell abnormalities play an important role in the frequently observed herpes virus infections and reactivations in CVID, and that these T cell abnormalities may also play a role in the aetiology of the disease. Therefore, we studied T cell function in CVID patients and in age-matched controls by measuring T cell proliferation and cytokine production in response to a wide array of herpes viruses. The immune response to human adenovirus (HAdV), a widespread respiratory non-herpes virus in the common population, was determined as a control. Because the higher infection pressure in children may influence the in vitro immune response, we measured T cell responses in both adult and paediatric CVID patients and in paediatric patients with specific antibody deficiency (SAD). As the underlying defect of CVID is unknown, this study could improve our knowledge of the pathophysiology of the disease.
Materials and methods
Study population
Nine children with CVID and five children with SAD (see below) and 14 adults with CVID, all being treated in the University Medical Centre of Utrecht, the Netherlands, were included in this study after the patients or legal representatives had provided written informed consent. Diagnosis of CVID was made according to standard international criteria, including impaired specific antibody production upon vaccination with conjugate or polysaccharide vaccines [1,2]. Recurrent infections, mainly in the respiratory and gastrointestinal system, were the hallmark of disease in all CVID patients; SAD patients suffered mainly from recurrent airway infections. Furthermore, patients were selected for the presence of clues for impaired immunity against herpes viruses, including recurrent HSV reactivations, or a clinical diagnosis of recurrent viral (airway) infections. All CVID and SAD patients had defective specific antibody production (defective production of specific antibodies upon vaccination). All SAD patients also suffered from decreased values of at least one of the following immunoglobulin (Ig)G subclasses: IgG1, IgG2 or IgG3. Clinically, these patients suffered from recurrent airway infections in the same severity as did patients with a diagnosis of CVID. Patients receiving immunosuppressive therapy were not included in the study. All patients (adults and children) received intravenous or subcutaneous immunoglobulin replacement therapy. To minimize the risk of potential immunomodulatory effects by exogenously administered immunoglobulins, study samples were drawn just before immunoglobulin administration.
As controls we included 14 children, matched for age, undergoing an elective orthopaedic, plastic surgical or ophthalmological operation. Blood was also taken from 14 healthy adult volunteers. Controls did not suffer from any known immunological disorder. Both patients and controls did not suffer from infections in the 3-month period preceding the study. This study was approved by the Medical Ethics Committee of the University Medical Centre of Utrecht.
Viral diagnostics
Because all patients received immunoglobulin replacement therapy and had defective antibody production, previous exposure to EBV, CMV and HSV was screened in saliva with a quantitative real-time–polymerase chain reaction (RT–PCR) assay, as described previously [37]. In the controls, prior exposure against the above-described viruses, except for human herpes virus type 6B (HHV6-B) and adenovirus (HAdV), was determined serologically using standard procedures. Previous infection with HHV6-B and HAdV was assumed positive, as previous studies have shown that nearly all children have acquired HHV6-B by 2 years of age, and that the incidence of previous HAdV infection surpasses 80 and 95% by the ages of 5 and 18 years, respectively [38–40].
Antigen-specific T cell proliferation
Peripheral blood mononuclear cells (PBMC) were isolated by Ficoll-Paque density gradient centrifugation (Amersham Pharmacia, Uppsala, Sweden). PBMC were pelleted and washed with phosphate-buffered saline (PBS) at a final concentration of maximal 20 × 106 PBMC/ml. Cells were labelled with 5,6-carboxyfluorescein diacetate succinimidyl ester (CFSE) (Molecular Probes, Invitrogen, Breda, the Netherlands) according to the manufacturer's protocol. Subsequently, 1–2 × 106 PBMC were cultured in RPMI-1640 (Gibco, Life Technologies, Breda, the Netherlands) supplemented with 1% penicillin/streptomycin and 10% human pooled serum stimulated with medium alone (negative control), different viral antigens (as described below) or with 2·5 µg/ml concanavalin A (ConA; Calbiochem, La Jolla, CA, USA) as a positive control. The following viral antigens were used: HSV type 1 antigen (5 µl/ml; Microbix Biosystems Inc., Toronto, Canada), HHV-6B lysate (1 µl/ml; Advanced Biotechnologies Incorporated, Columbia, MD, USA), overlapping peptide pools consisting of 15-mer peptides with 11 amino acid overlap of the entire pp65 or IE1 protein of CMV (Pepmix, 138 and 120 peptides, respectively; 1 µg/ml of each peptide) or the immunogenic C-terminal region of EBNA1 or the entire BZLF1 protein derived from EBV (57 and 59 peptides, respectively; 1 mg/ml of each peptide) (JPT Peptide Technologies GmbH, Berlin, Germany). As a control, complete HAdV serotype 5 with a multiplicity of infection of 10 [American Type Culture Collection (ATCC) no: VR1516], inactivated by methylene blue plus visible light, as described earlier [41], and a pool of 5 15-mer HAdV-peptides (2 µg/ml each), derived from HAdV5 as described earlier [42], was used. Cells were cultured for 5 days at 37°C, 5% CO2.
After 5 days PBMC were stained with CD3 peridinin chlorophyll (PerCP), CD8 allophycocyanin (APC) [Becton Dickinson (BD), San José, CA, USA] and CD4 Alexa Fluor 750 (eBioscience, San Diego, CA, USA). Cells were washed and 200 000 cells were acquired by the LSRII flow cytometer (BD). Data were analysed by BD FACSDiva software. Stimulation indices (SI) were calculated by dividing the percentage proliferation after stimulation with antigen by the percentage of proliferating cells after stimulation with medium.
Cytokine production
PBMCs from CVID patients and controls were cultured with medium alone, the viral antigens of interest or ConA, as described above. After 72 h cell supernatants were collected and stored at −80°C until further analysis. Cytokine production was measured with multiplex immunoassay (MIA). MIA combines the principle of a sandwich immunoassay with luminex fluorescent-bead-based technology (Lab-MAP) [43]. MIA is a validated method for the measurement of cytokines and chemokines in culture supernatants of human PBMCs. Comparing MIA with the regular ELISA technique values appear comparable in sensitivity, accuracy and reproducibility and is less time-consuming [44,45]. The concentrations of the following nine soluble mediators, all involved in inflammation or clearance of infection, were measured: interleukin (IL)-1β, IL-4, IL-6, IL-10, IL-12, IL-17, tumour necrosis factor (TNF)-α, interferon (IFN)-γ and interferon inducible protein [(IP-10) = CXCL-10].
Statistics
The Mann–Whitney U-test was used to describe the differences in the lymphocyte proliferative responses between adults with CVID, children with CVID and their controls. Differences in cytokine production between different groups were determined by the Mann–Whitney U-test (two-tailed). P-values < 0·05 were considered significant. Data analysis of detected soluble mediators was performed with Bio-Plex Manager 3.0 (Bio-Rad Laboratories, Hercules CA, USA) using a five parametric-curve fitting. Statistics were computed by spss version 15.0 for Windows (SPSS, Inc., Chicago, IL, USA).
Results
Study population
Nine children with CVID [seven male, two female; mean age 10·1 years (±3·8)], five patients with SAD [two male, three female; mean age 11·6 years (±3·5)] and 14 adults with CVID [six male, eight female; mean age 42·7 years (±14·5)] were included. As controls, we included 14 children [nine male, five female, matched for age [mean age 10·9 years (±3·6)] and 14 healthy adult volunteers [eight male, six female; mean age 42·3 (±6·8) years]. The mean age of onset of symptoms in paediatric patients was 1·5 years (±0·8), both in CVID patients and in patients with SAD. In patients with an age of onset < 4 years, the diagnostic criterion of defective antibody production was reconfirmed after the age of 4 years. In adults the mean age of onset of symptoms was 16·6 years (±17).
Recently, Chapel et al. [46] developed a classification to define distinct clinical phenotypes based on the presence of specific complications in CVID patients. Five clinical phenotypes were described: no complications, autoimmunity, polyclonal lymphocytic infiltration, enteropathy and lymphoid malignancy. Almost half of our patients (46%) did not suffer from any illness-related complication. Twenty-one per cent of the patients had an autoimmune disease. Polyclonal lymphocytic infiltration was seen in 39% of the patients, and enteropathy in 7%. Patient characteristics are described in Table 1.
Table 1.
Patient characteristics.
| Patient no. | Sex | Age (years) | Age onset of symptoms (years) | IgA level (g/l) | IgG level (g/l) | IgM level (g/l) | IgA + IgG-positive switched memory (% of total B cells) | CD4/CD8 T cell ratio | Chapel classification† | Autoimmune disease |
|---|---|---|---|---|---|---|---|---|---|---|
| 1 | F | 16·3 | 2·0 | 0·07 | 3·4 | 2·5 | Unknown | 1·4 | 1, 2 | AIHA, ITP |
| 2 | M | 6·0 | 0·8 | 0·2 | 4·1 | 0·5 | 13 | 1·4 | 1 | JIA |
| 3 | M | 8·0 | 2·5 | 0·43 | 4·81 | 1 | 4 | 1·4 | 5 | |
| 4 | M | 11·1 | 1·0 | 0·6 | 4 | 0·9 | 5·2 | 1·6 | 2 | |
| 5 | M | 13·5 | 3·0 | 0·15 | 4·7 | 1·2 | 9 | 2·9 | 1 | Crohn |
| 6 | M | 6·7 | 2·0 | 0·48 | 4·3 | 0·32 | 1·4 | 1·6 | 5 | |
| 7 | M | 5·3 | 0·5 | 0·21 | 5·1 | 0·24 | 3 | 1·7 | 2 | |
| 8 | F | 11·5 | 1·0 | 0·4 | 5·3 | 0·8 | 0·3 | 1·7 | 5 | |
| 9 | M | 12·7 | 1·0 | 0·3 | 5·9 | 0·2 | 1·3 | 0·7 | 5 | |
| 10‡ | F | 13·9 | 2·0 | 0·8 | 10·6 | 1·4 | 0·7 | 1·3 | 2 | |
| 11‡ | F | 16·3 | 2·0 | 0·9 | 8·6 | 0·7 | 1·7 | 1·8 | 5 | |
| 12‡ | M | 10·6 | 0·5 | 0·2 | 9·8 | 0·6 | 1·3 | 1·2 | 1 | AIT |
| 13‡ | M | 10·4 | 2·0 | 1·72 | 7·9 | 0·24 | 3 | 1·6 | 5 | |
| 14‡ | F | 7·1 | 1·0 | < 0·07 | 9·7 | 0·8 | 5·6 | 1·9 | 2 | |
| 15 | M | 26·8 | 20·0 | 0·08 | 3·21 | 0·2 | 17 | 1·8 | 5 | |
| 16 | M | 42·3 | 31·0 | 0·38 | 4·6 | 0·31 | 12 | 3·7 | 5 | |
| 17 | F | 35·9 | 16·0 | < 0·1 | 2·8 | 0·4 | 3 | 6·3 | 2 | |
| 18 | F | 34·7 | 12·0 | < 0·1 | 1·3 | n.d. | 8 | 3·2 | 5 | |
| 19 | F | 58·2 | 3·0 | 0·85 | 5·2 | 0·96 | 1·7 | 4·7 | 5 | |
| 20 | F | 45·7 | 2·0 | 0·04 | 0·52 | < 0·07 | 2·8 | 2·2 | 1, 2, 4 | AA |
| 21 | F | 35·1 | 4·0 | 1·05 | 4·7 | 0·96 | 36 | 0·9 | 2 | |
| 22 | M | 26·6 | 2·0 | ND | 8·2 | 0·25 | 5 | 1·5 | 2 | |
| 23 | M | 60·2 | 54·0 | 0·1 | 1·3 | 0·1 | Unknown | 2·1 | 5 | RA/DM II |
| 24 | F | 39·8 | 4·0 | < 0·01 | 4·5 | 0·5 | 3 | 1·9 | 4 | |
| 25 | F | 55·1 | 27·0 | 0·6 | 5·1 | 0·8 | 19 | 4·2 | 5 | |
| 26 | M | 42·8 | 12·0 | < 0·1 | < 0·1 | 2·4 | Unknown | 0·5 | 5 | |
| 27 | M | 22·0 | 0·7 | 0·85 | 4·8 | 0·6 | 6 | 1·1 | 2 | |
| 28 | F | 72·6 | 45·0 | 0·03 | 2·8 | < 0·1 | Unknown | 0·5 | 1, 2 |
Chapel classification: 1: autoimmune disease, 2: polyclonal lymphocytic infiltration, 3: lymphoid malignancy, 4: unexplained enteropathy, 5: no disease-related complications.
Paediatric patients with specific antibody deficiency (SAD). AIHA, autoimmune haemolytic anaemia; AIT, autoimmune thyroiditis; DMII, diabetes mellitus, type II; ITP, ideopathic trombocytopenia; JIA, juvenile ideopathic arthritis; RA, rheumatoid arthritis; Ig, immunoglobulin; F, female; M, male.
Presence of herpes viruses in the study population
In paediatric patients, previous exposure to the herpes viruses of interest (except for HHV-6B) was determined by RT–PCR. Overall, a positive PCR for EBV could be detected in five of the 14 patients (two CVID patients and three patients with SAD), and one SAD patient had a positive PCR for HSV. None of the patients had a positive PCR for CMV. In adult CVID patients, we detected a positive PCR for EBV in three patients, and none of the patients had a positive PCR for CMV or HSV. Although RT–PCR for the detection of herpes viruses has a high sensitivity [37,47], in clinical practice the frequency of reactivation with herpes viruses is low and reliable determination of prior exposition is not possible. Therefore, a negative PCR does not exclude that patients had contact with the herpes viruses of interest.
In paediatric controls, similar virology status could be detected as in paediatric CVID patients, determined by serology: seven of the 14 controls had a positive serology for EBV, only one control had a positive CMV serology and two of the 14 controls showed a positive serology for HSV. In adult controls, a positive serology could be found against EBV in 13 controls, against CMV in six controls and HSV in five controls.
Virus-specific T cell proliferation
To investigate the ability of CVID patients to mount an adequate T cell response to different herpes viral antigens, we measured the proliferative capacity of both CD4+ and CD8+ T cells in paediatric and adult CVID patients and age-matched controls using CFSE dilution assays. In Fig. 1a representative FACS plots are shown of CFSE dilution of CD8+ T cells in response to medium alone, pp65- and IE1-overlapping peptide pools derived from CMV, HSV-type 1 antigen and ConA of a paediatric CVID patient and a control child. Stimulation indices were calculated to compare the proliferative capacities of CD8+ T cells of CVID patients and controls. CD8+ T cell proliferation in response to pp65, IE1 and HHV6-B in paediatric CVID patients (n = 9) was increased significantly compared to age-matched controls (P < 0·05; Fig. 1b). Although not significant (P = 0·058) the CD8+ response to HSV was also increased in paediatric CVID patients compared to controls. CD4+ T cell proliferation was not different between paediatric CVID patients and controls. No clear differences could be detected in proliferative capacities in response to the herpes viruses and HAdV between the paediatric SAD patients and paediatric controls (Fig. 1c). Together these data suggest that paediatric CVID patients have increased CD8+ T cell responses to CMV, HHV6B and HSV.
Fig. 1.
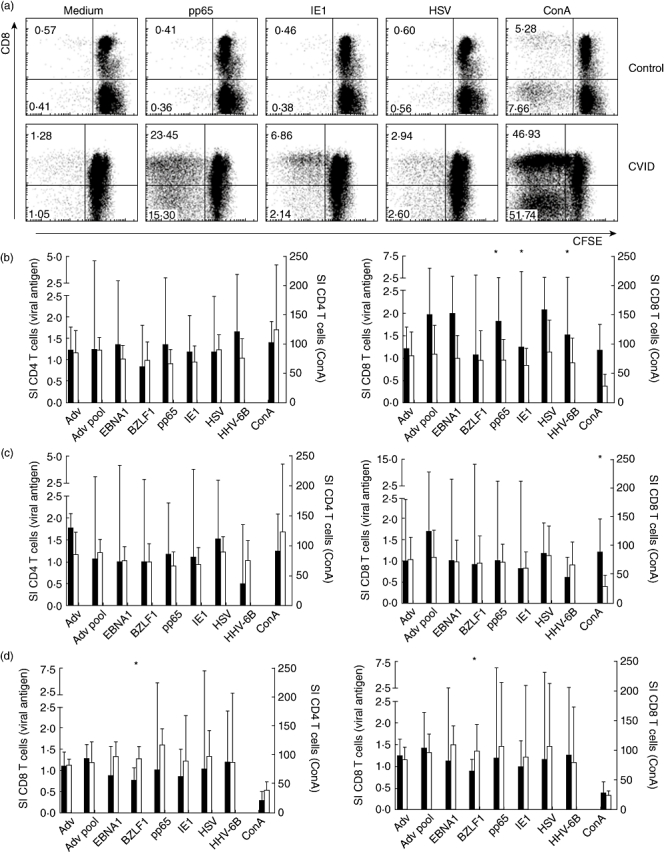
Virus-specific CD4+ and CD8+ T cell proliferation. (a) Representative fluorescence activated cell sorter (FACS) plots of antigen-specific T cell proliferation of a control (upper panel) and a common variable immunodeficiency (CVID) patient (lower panel). Cells were gated in lymphocyte morphology based on forward- and side-scatter and the expression of CD3. Percentages of proliferating CD8+ T cells are shown in the upper left corners, whereas percentages of proliferating CD4+ T cells are presented in the lower left corners. Shown are responses to medium alone, the cytomegalovirus (CMV) matrix protein pp65, the CMV immediate early antigen (IE)1, HSV type I antigen and concanavalin A. Bars indicate the median (with interquartile range) of the stimulation index (SI) of paediatric CVID patients compared to age-matched controls (b), of pediatric SAD patients compared to age-matched controls (c) and of adult CVID patients compared to adult controls (d). Black: CVID; white: controls. P-values < 0·05 are considered significant (Mann–Whitney U-test) and indicated with an asterisk.
In adults, we found only subtle differences between CVID patients and controls. CD4+ and CD8+ T cell proliferation was decreased in response to BZLF1, an immediate early protein derived from EBV (P < 0·05; Fig. 1d). Furthermore, in adult CVID patients, more variation in the stimulation indices and a tendency towards higher values was observed in the CD8+ T cell proliferation in response to pp65 and HSV compared to other viral antigens.
Cytokine production in response to viral antigens
After 3 days of stimulation with viral antigen, cytokine production was measured in supernatants of a total of eight adult CVID patients, five paediatric CVID patients, three paediatric SAD patients and eight controls (four adults and four children) by MIA. In Table 2 the median [standard error of the mean (s.e.m.)] cytokine production in response to the different viral antigens minus the response to medium is shown.
Table 2.
Cytokine response to viral antigens.
| Cytokine | EBNA | BZLF1 | Pp65 | IE1 | HSV | HHV6B | HAdV | HAdV pool | |
|---|---|---|---|---|---|---|---|---|---|
| Adult CVID (n = 8) | IL-1β | 12·1 (25·6) | 10·6 (7·6) | 34·1 (53·0) | 34·5 (31·4)* | 126 (1051) | 0·01 (5·3) | 3·1 (6·4) | 0·11 (18·1) |
| IL-4 | 0·01 (0·0) | 0·01 (0·01) | 0·01 (0·01) | 0·01 (0·0) | 0·01 (0·07) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·0) | |
| IL-6 | 159 (622) | 478 (377) | 985 (785) | 576 (671) | 1149 (578) | 5·5 (431) | 51 (158) | 136 (712) | |
| IL-10 | 0·8 (1·97) | 0·22 (1·38) | 0·85 (0·98) | 0·09 (0·48) | 2·97 (10·4) | 0·01 (0·31) | 0·01 (0·0) | 0·57 (1·36) | |
| IL-12 | 0·01 (0·33)* | 0·01 (1·7) | 0·73 (1·52) | 0·01 (0·0) | 0·01 (0·0)* | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·32)* | |
| IL-17 | 0·01 (0·11) | 0·06 (0·18) | 0·09 (0·07) | 0·01 (0·61) | 0·01 (0·86) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·76) | |
| TNF-α | 0·01 (18·1) | 0·71 (25·5) | 0·01 (21·6) | 0·01 (2·0) | 138 (78·7) | 0·32 (16·8) | 12·8 (39·4) | 4·86 (31·6) | |
| IFN-γ | 0·13 (0·63) | 0·18 (0·43) | 0·33 (7·0) | 0·01 (0·0) | 0·01 (23·0) | 0·01 (0·02) | 0·01 (0·0) | 0·01 (0·15) | |
| IP-10 | 0·01 (23·4) | 30·7 (90·7)* | 12·5 (110) | 0·01 (11·8) | 93·4 (554) | 16·1 (101) | 4·8 (40·1) | 58·6 (31·8) | |
| Adult controls (n = 4) | IL-1β | 10·8 (13·5) | 3·6 (4·4) | 79 (1848) | 36·8 (37·5) | 116 (1819) | 0·01 (0·6) | 3·4 (8·1) | 0·01 (31·1) |
| IL-4 | 0·01 (0·0) | 0·02 (0·01) | 0·02 (0·01) | 0·01 (0·0) | 0·02 (0·11) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·0) | |
| IL-6 | 233 (400) | 321 (333) | 2184 (999) | 1057 (1076) | 1795 (674) | 12·64 (82·5) | 273 (234) | 424 (645) | |
| IL-10 | 0·32 (0·35) | 0·11 (0·07) | 1·15 (1·09) | 0·15 (0·60) | 4·96 (5·77) | 0·01 (0·0) | 0·01 (0·0) | 0·61 (1·31) | |
| IL-12 | 0·01 (0·6) | 0·81 (0·83) | 2·9 (1·96) | 0·01 (1·22) | 0·01 (1·22) | 0·01 (0·0) | 0·01 (12·2) | 0·01 (31) | |
| IL-17 | 0·01 (0·0) | 0·01 (0·00) | 0·01 (0·01) | 0·01 (0·05) | 0·66 (1·07) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (1·13) | |
| TNF-α | 0·01 (0·0) | 0·01 (31·1) | 0·13 (34·8) | 0·01 (3·08) | 153·7 (101) | 0·45 (8·42) | 30·75 (53·9) | 8·9 (16·01) | |
| IFN-γ | 0·04 (0·37) | 0·29 (0·21) | 0·15 (11·2) | 0·01 (0·0) | 14·8 (17·8) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·15) | |
| IP-10 | 0·01 (1·03) | 54·7 (83) | 1·54 (149) | 0·01 (1·87) | 757 (772) | 10·1 (62) | 7·83 (52) | 63 (25) | |
| Paediatric CVID (n = 5) | IL-1β | 0·98 (31·6) | 0·82 (38·2) | 3·61 (53) | 0·01 (6·11)* | 203 (2366) | 0·01 (3·21) | 1·84 (1·25) | 3·95 (1580) |
| IL-4 | 0·01 (0·01) | 0·01 (0·02) | 0·03 (0·35) | 0·03 (0·12) | 0·01 (0·02) | 0·01 (0·0) | 0·01 (0·02) | 0·03 (0·02) | |
| IL-6 | 14·7 (957) | 24 (2097) | 910 (2319) | 0·01 (227) | 3568 (3406) | 4·32 (55·7) | 0·01 (34·3) | 998 (3260) | |
| IL-10 | 0·10 (0·61) | 0·74 (3·18) | 2·94 (1·85) | 0·57 (0·70) | 14·86 (8·27) | 0·01 (0·0) | 0·01 (0·43) | 1·75 (7·51) | |
| IL-12 | 1·61 (11)* | 1·61 (14·4) | 4·08 (15·2) | 4·92 (11·8) | 1·61 (10·9)*/** | 0·01 (0·0) | 0·01 (10·01) | 1·61 (4·12)* | |
| IL-17 | 0·01 (0·32) | 0·01 (0·14) | 0·12 (0·22) | 0·01 (0·15) | 0·01 (0·01) | 0·01 (0·00) | 0·01 (0·00) | 0·01 (0·07) | |
| TNF-α | 0·01 (1·90) | 0·01 (15·43) | 0·01 (35·03) | 0·01 (8·42) | 102·0 (70·6) | 1·76 (8·13) | 10·75 (13·4) | 33·7 (39·44) | |
| IFN-γ | 0·05 (1·91) | 0·11 (3·89) | 0·12 (62·4) | 1·10 (4·57) | 0·01 (3·26) | 0·01 (0·00) | 0·01 (2·33) | 1·34 (13·66) | |
| IP-10 | 0·01 (122·4) | 0·01 (0·27)*/** | 0·01 (675) | 2·29 (794) | 53·67 (85·6) | 8·81 (445) | 0·01 (6·03) | 57·7 (594·7) | |
| Paediatric SAD (n = 3) | IL-1β | 1·32 (24·6) | 0·01 (21·5) | 0·83 (6·1) | 0·05 (11·92) | 4958 (2207) | 0·01 (2·4) | 0·05 (15·6) | 0·01 (33·04) |
| IL-4 | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·0) | 0·05 (0·02) | 0·04 (0·02) | 0·01 (0·09) | 0·01 (0·36) | |
| IL-6 | 33·3 (453) | 0·08 (2427) | 139 (243) | 2118 (1955) | 4033 (1849) | 4033 (1849) | 0·08 (4·52) | 0·08 (1912) | |
| IL-10 | 0·26 (1·71) | 0·31 (1·50) | 0·94 (2·94) | 1·27 (0·63) | 27·48 (11·2) | 0·41 (0·34) | 0·01 (0·37) | 0·17 (2·59) | |
| IL-12 | 1·68 (1·09)* | 0·01 (1·44) | 0·01 (0·71) | 0·01 (1·44) | 0·01 (0·71) | 1·61 (1·15) | 0·01 (0·0) | 0·01 (2·24) | |
| IL-17 | 0·01 (0·52) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·54) | 2·02 (3·98) | 0·01 (0·00) | 0·01 (6·28) | 0·01 (0·0) | |
| TNF-α | 0·01 (14·87) | 0·06 (4·22) | 0·48 (40·9) | 0·06 (23·26) | 17·4 (37·98) | 0·01 (0·04) | 2·88 (58·61) | 1·73 (68·7) | |
| IFN-γ | 0·33 (0·49) | 0·12 (0·18) | 0·01 (0·46) | 0·12 (0·44) | 0·02 (68·32) | 0·01 (0·11) | 0·01 (0·23) | 0·33 (4·7) | |
| IP-10 | 40·31 (963) | 244 (54) | 0·01 (0·0) | 0·01 (35·96) | 1681 (760) | 1019 (2046) | 69·1 (2516) | 216 (1245) | |
| Paediatric controls (n = 4) | IL-1β | 25·4 (28·7) | 13·6 (8·06) | 14·3 (17·2) | 15·2 (18·3) | 72 (1844) | 0·01 (9·3) | 4·8 (6·3) | 3·31 (6·8) |
| IL-4 | 0·01 (0·0) | 0·01 (0·01) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·02) | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·01) | |
| IL-6 | 1127 (1133) | 541 (426) | 199 (340) | 588 (648) | 961 (1433) | 85 (813) | 278 (365) | 258 (654) | |
| IL-10 | 2·13 (1·65) | 0·09 (1·93) | 0·07 (0·44) | 0·01 (0·17) | 1·03 (11·7) | 0·01 (0·2) | 1·03 (11·7) | 0·01 (0·23) | |
| IL-12 | 0·01 (1·89) | 0·84 (2·4) | 0·01 (0·62) | 0·01 (1·25) | 0·01 (0·0)** | 0·01 (0·0) | 0·01 (0·0) | 0·01 (0·62) | |
| IL-17 | 0·01 (0·03) | 0·09 (0·15) | 0·14 (0·14) | 0·01 (1·06) | 0·01 (0·38) | 0·01 (0·0) | 0·01 (0·0) | 0·02 (0·7) | |
| TNF-α | 0·11 (25·5) | 6·36 (18·7) | 0·01 (0·76) | 0·01 (0·0) | 98·0 (63·6) | 0·11 (23·6) | 15·8 (10·8) | 34·3 (42) | |
| IFN-γ | 1·07 (1·2) | 0·01 (0·6) | 0·21 (0·33) | 0·14 (0·07) | 0·01 (28·5) | 0·01 (0·0) | 0·01 (0·0) | 0·2 (0·23) | |
| IP-10 | 7·24 (39) | 29·9 (88)** | 33·2 (87) | 0·86 (19·8) | 85·4 (342) | 33·4 (131) | 3·9 (17·2) | 49·4 (34·2) |
Cytokine responses were calculated by subtracting cytokine levels in unstimulated samples from values measured in samples stimulated with the antigen of interest. All values [median ± standard error of the mean (s.e.m.)] are expressed in pg/ml.
Significant difference between adult common variable immunodeficiency (CVID) and paediatric CVID/specific antibody deficiency (SAD) patients, P < 0·05.
Significant difference between CVID/SAD patients and controls, P < 0·05. IFN, interferon; IL, interleukin; IP-10, interferon inducible protein (CXCL-10); TNF, tumour necrosis factor.
In all groups, the proinflammatory cytokines IL-1β, IL-6, TNF-α and IP-10 were produced predominantly. The production of IFN-γ was low in all groups, which could be due to timing of the sampling. A large variation was observed in cytokine production in individual subjects within all groups. In general, only minor differences in cytokine production could be detected between CVID patients, paediatric SAD patients and controls. In paediatric CVID patients, significantly more IL-12 production could be found in response to EBNA1 as well in response to HSV compared to adult CVID patients and healthy controls. Finally, an overall trend towards higher production of IL-10 could be seen in paediatric patients with CVID and SAD, compared with both adult CVID patients and with age-matched controls (Fig. 2).
Fig. 2.
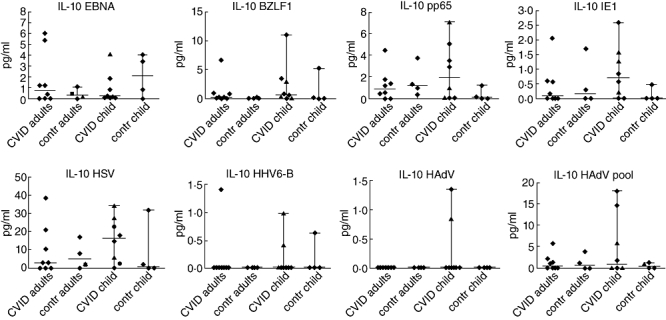
Cytokine production in response to the different viral antigens. Interleukin (IL)-10 production in response to different viral antigens in common variable immunodeficiency (CVID) patients and children with specific antibody deficiency (SAD) (depicted by triangles) and control groups (median with range). P-values < 0·05 are considered significant (Mann–Whitney U-test).
Association between viral exposure, proliferation and cytokine production
Comparing the cytokine production of healthy controls with a positive serostatus and patients with a detectable DNA viral load or a proliferative T cell response to viral antigen, no changes in cytokine profiles were observed (data not shown). In addition, no relation could be found between a positive proliferative T cell response and cytokine production.
Remarkably, one individual paediatric patient with SAD had recurrent cutaneous HSV reactivations (patient no. 10; Table 1) without having reactivations of other herpes viruses. This patient had almost undetectable proliferation and cytokine production towards HSV and low but detectable responses to the other herpes viral antigens.
Discussion
This study was undertaken to investigate further the T cell function in CVID patients, focusing upon T cell function against herpes viridae. Previous studies have focused upon T cell proliferation to mitogens and recall antigens, including EBV- and CMV-derived epitopes [12,14–17,32]. We studied T cell proliferation and cytokine responsiveness to a number of herpes viruses in adults and children with CVID and paediatric patients with SAD, suffering from recurrent viral infections or recurrent herpes virus reactivations, and compared these values with age-matched controls. In antigen-specific CD4+ and CD8+ T cell proliferation, no major differences could be detected between paediatric and adult CVID patients and controls. We observed that paediatric CVID patients had higher proliferative CD8+ T cell responses to the CMV-derived antigens pp65 and IE1, to HSV and to HHV6-B, when compared to age-matched controls. T cell responses to EBV were not increased compared to controls. No significant differences were found in antigen-specific T cell responses between paediatric SAD patients and controls. Although these findings were observed in small patient groups, they may reflect differences in humoral and cellular immunity between CVID and SAD patients.
Significant differences in T cell proliferation were not observed between adult CVID patients and controls; however, we noticed large variations in T cell responses to pp65 and HSV in adult CVID patients compared to controls. This finding may suggest that subgroups of patients have disturbed immunity against EBV (pp65) and HSV. These results are in line with data observed by Reiszadeh et al. [32], who reported increased CMV-specific T cell responses in CVID patients. CMV- and HSV-specific CD8+ T cell responses may be increased in response to relatively diminished viral control because of aberrant antibody production. Variation in CD8+ T cell responses may be related to time since exposure to the viral antigens; however, as there seems no reason for CVID patients to contract primo infection with herpes viruses earlier than other patients, we do not consider this a likely explanation for wide variations in T cell responses in some CVID patients.
In all study groups, production of the proinflammatory cytokines IL-1β, IL-6, TNF-α and IP-10 could be detected, compatible with a normal immune response upon contact with viral pathogens. Between CVID patients and controls, only small differences in cytokine production could be observed. However, children with CVID and SAD had a higher overall production of proinflammatory cytokines compared to adult CVID patients, with the highest increase in cytokine production being observed for IL-12. Remarkably, the production of the anti-inflammatory cytokine IL-10 was also increased in paediatric patients, both compared with adult patients and with age-matched controls. Earlier studies in adult CVID patients reported deficient production of IL-12 by dendritic cells [48] and impaired IL-10 production by T lymphocytes [21]. Giovannetti et al. [23] showed increased IFN-γ production and decreased IL-10 production in a subset of CVID patients. Deficient IL-12 production could lead to attenuated T cell activation and defective immunization [9], and although IL-10 production is important for B cell function, high levels of IL-10 can also result in a less efficient anti-viral immune response [49]. Because cytokine production in this study was measured in PBMCs, future work could focus upon measuring intracellular cytokine production to delineate cytokine profiles by the different cell populations and to further detect differences between patients and controls.
Although subtle differences were observed in T cell proliferation in response to several of the herpes viruses, such as increased T cell proliferation in response to CMV and HSV in CVID patients, no clear general patterns in T cell responses could be seen that would indicate a general T cell defect towards herpes viruses in CVID patients. Recurrent herpes virus infections and reactivations in a subset of CVID patients may be due to aberrant antibody production or a disturbed interaction between T and B cells, rather than to T cell defects alone. Furthermore, disturbed T cell-mediated immunity against herpes viruses in individual patients cannot be excluded.
This study was limited by the fact that it is not possible to determine reliably previous contact with herpes viruses in patients with antibody deficiency syndromes, despite the use of RT–PCR. Nevertheless, our data showed that proliferation in response particularly to CMV and HSV was increased slightly in CVID patients with recurrent viral infections, whereas the response to HAdV and HHV6-B was comparable between patients and controls, compatible with a normal immune response to those viruses. It is possible that differences between subgroups will be more prominent by increasing the study population or by including only patients with recurrent herpes virus infections. Future research should also focus on patients with defective proliferative responses and deviant cytokine production.
In conclusion, the overall findings of T cell proliferation and cytokine production upon stimulation with herpes viruses and HAdV in CVID patients and SAD patients were comparable to values found in healthy controls. In paediatric CVID, but not in SAD, we found moderately increased proliferation upon stimulation with CMV, HSV and HHV6-B antigens. Because the underlying defects in CVID patients may be extremely heterogeneous and it was not possible to determine reliably previous exposure to the studied viruses, these findings do not exclude the possibility of T cell abnormalities in subsets of CVID patients. The present study, however, suggests that CVID patients have normal T cell responses to viral antigens.
Disclosure
The authors have no financial conflict of interest.
References
- 1.Conley ME, Notarangelo LD, Etzioni A. Diagnostic criteria for primary immunodeficiencies. Clin Immunol. 1999;93:190–7. doi: 10.1006/clim.1999.4799. [DOI] [PubMed] [Google Scholar]
- 2.Notarangelo LD, Casanova JL, Conley ME, et al. Primary immunodeficiency diseases: an update from the international Union of Immunological Societies Primary Immunodeficiency Diseases Classification Committee Meeting in Budapest, 2005. J Allergy Clin Immunol. 2006;117:883–96. doi: 10.1016/j.jaci.2005.12.1347. [DOI] [PubMed] [Google Scholar]
- 3.Goldacker S, Warnatz K. Tackling the heterogeneity of CVID. Curr Opin Allergy Clin Immunol. 2005;5:504–9. doi: 10.1097/01.all.0000191888.97397.b3. [DOI] [PubMed] [Google Scholar]
- 4.Bacchelli C, Buckridge S, Trasher AJ, Gaspar HB. Translational mini-review series on immunodeficiency: molecular defects in common variable immunodeficiency. Clin Exp Immunol. 2007;149:401–9. doi: 10.1111/j.1365-2249.2007.03461.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.van Zelm M. Antibody deficiency and acute nefritis syndrome in a patient with homozygous dysruption of the CD81 gene. Oral presentation at the 13th Meeting of the European Society for Immunodeficiencies, November 2008, Den Bosch, the Netherlands.
- 6.Kuijpers TW. Human CD20 deficiency impairs the ability to mount T-cell dependent antibody responses. Oral presentation at the 13th Meeting of the European Society for Immunodeficiencies, November 2008, Den Bosch, the Netherlands.
- 7.Farrington M, Grosmaire LS, Nonoyama S, et al. CD40 ligand expression is defective in a subset of patients with common variable immunodeficiency. Proc Natl Acad Sci USA. 1994;91:1099–103. doi: 10.1073/pnas.91.3.1099. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Farrant J, Spickett G, Matamoros N, et al. Study of B and T cell phenotypes in blood from patients with common variable immunodeficiency (CVID) Immunodeficiency. 1994;5:159–69. [PubMed] [Google Scholar]
- 9.Cunningham-Rundles C, Radigan L. Deficient IL-12 and dendritic cell function in common variable immune deficiency. Clin Immunol. 2005;115:147–53. doi: 10.1016/j.clim.2004.12.007. [DOI] [PubMed] [Google Scholar]
- 10.Kruger G, Welte K, Ciobanu N, et al. Interleukin-2 correction of defective in vitro T-cell mitogenesis in patients with common varied immunodeficiency. J Clin Immunol. 1984;4:295–303. doi: 10.1007/BF00915297. [DOI] [PubMed] [Google Scholar]
- 11.Nonoyama S, Farrington ML, Ochs HD. Effect of IL-2 on immunoglobulin production by anti-CD40-activated human B cells: synergistic effect with IL-10 and antagonistic effect with IL-4. Clin Immunol Immunopathol. 1994;72:373–9. doi: 10.1006/clin.1994.1155. [DOI] [PubMed] [Google Scholar]
- 12.Cunningham-Rundles C, Bodian C. Common variable immunodeficiency: clinical and immunological features of 248 patients. Clin Immunol. 1999;92:34–48. doi: 10.1006/clim.1999.4725. [DOI] [PubMed] [Google Scholar]
- 13.Di Renzo M, Zhou Z, George I, Becker K, Cunningham-Rundles C. Enhanced apoptosis of T cells in common variable immunodeficiency (CVID): role of defective CD28 co-stimulation. Clin Exp Immunol. 2000;120:503–11. doi: 10.1046/j.1365-2249.2000.01239.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.North ME, Spickett GP, Allsop J, Webster AD, Farrant J. Defective DNA synthesis by T cells in acquired ‘common-variable’ hypogammaglobulinaemia on stimulation with mitogens. Clin Exp Immunol. 1989;76:19–23. [PMC free article] [PubMed] [Google Scholar]
- 15.Kondratenko I, Amlot PL, Webster AD, Farrant J. Lack of specific antibody response in common variable immunodeficiency (CVID) associated with failure in production of antigen-specific memory T cells. MRC Immunodeficiency Group. Clin Exp Immunol. 1997;108:9–13. doi: 10.1046/j.1365-2249.1997.d01-993.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Stagg AJ, Funauchi M, Knight SC, Webster AD, Farrant J. Failure in antigen responses by T cells from patients with common variable immunodeficiency (CVID) Clin Exp Immunol. 1994;96:48–53. doi: 10.1111/j.1365-2249.1994.tb06228.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Fischer MB, Hauber I, Eggenbauer H, et al. Activation of CVID patients' T cells with conventional antigens and superantigens. Immunodeficiency. 1993;4:15–16. [PubMed] [Google Scholar]
- 18.Sneller MC, Strober W. Abnormalities of lymphokine gene expression in patients with common variable immunodeficiency. J Immunol. 1990;144:3762–9. [PubMed] [Google Scholar]
- 19.Aukrust P, Müller F, Frøland SS. Elevated serum levels of interleukin-4 and interleukin-6 in patients with common variable immunodeficiency (CVI) are associated with chronic immune activation and low numbers of CD4+ lymphocytes. Clin Immunol Immunopathol. 1994;70:217–24. doi: 10.1006/clin.1994.1032. [DOI] [PubMed] [Google Scholar]
- 20.Holm AM, Aukrust P, Damas JK, Muller F, Halvorsen B, Froland SS. Abnormal interleukin-7 function in common variable immunodeficiency. Blood. 2005;105:2887–90. doi: 10.1182/blood-2004-06-2423. [DOI] [PubMed] [Google Scholar]
- 21.Holm AM, Aukrust P, Aandahl EM, Muller F, Tasken K, Froland SS. Impaired secretion of IL-10 by T cells from patients with common variable immunodeficiency – involvement of protein kinase A type I. J Immunol. 2003;170:5772–7. doi: 10.4049/jimmunol.170.11.5772. [DOI] [PubMed] [Google Scholar]
- 22.Wright JJ, Wagner DK, Blaese RM, Hagengruber C, Waldmann TA, Fleisher TA. Characterization of common variable immunodeficiency: identification of a subset of patients with distinctive immunophenotypic and clinical features. Blood. 1990;76:2046–51. [PubMed] [Google Scholar]
- 23.Giovannetti A, Pierdominici M, Mazzetta F, et al. Unravelling the complexity of T cell abnormalities in common variable immunodeficiency. J Immunol. 2007;178:3932–43. doi: 10.4049/jimmunol.178.6.3932. [DOI] [PubMed] [Google Scholar]
- 24.Oliva A, Scala E, Quinti I, et al. IL-10 production and CD40L expression in patients with common variable immunodeficiency. Scand J Immunol. 1997;46:86–90. doi: 10.1046/j.1365-3083.1997.d01-95.x. [DOI] [PubMed] [Google Scholar]
- 25.Aspalter RM, Eibl MM, Wolf HM. Defective T-cell activation caused by impairment of the TNF receptor 2 costimulatory pathway in common variable immunodeficiency. J Allergy Clin Immunol. 2007;120:1193–2000. doi: 10.1016/j.jaci.2007.07.004. [DOI] [PubMed] [Google Scholar]
- 26.Boncristiano M, Majolini MB, D'Elios MM, et al. Defective recruitment and activation of ZAP-70 in common variable immunodeficiency patients with T cell defects. Eur J Immunol. 2000;30:2632–8. doi: 10.1002/1521-4141(200009)30:9<2632::AID-IMMU2632>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- 27.Paccani SR, Boncristiano M, Patrussi L, et al. Defective Vav expression and impaired F-actin reorganization in a subset of patients with common variable immunodeficiency characterized by T-cell defects. Blood. 2005;106:626–34. doi: 10.1182/blood-2004-05-2051. [DOI] [PubMed] [Google Scholar]
- 28.Fevang B, Yndestad A, Sandberg WJ, et al. Low numbers of regulatory T cells in common variable immunodeficiency: association with chronic inflammation in vivo. Clin Exp Immunol. 2007;147:521–5. doi: 10.1111/j.1365-2249.2006.03314.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Genre J, Errante PR, Kokron CM, Toledo-Barros M, Câmara NO, Rizzo LV. Reduced frequency of CD4(+)CD25(HIGH)FOXP3(+) cells and diminished FOXP3 expression in patients with common variable immunodeficiency: a link to autoimmunity? Clin Immunol. 2009;132:215–21. doi: 10.1016/j.clim.2009.03.519. [DOI] [PubMed] [Google Scholar]
- 30.Witte T, Werwitzke S, Schmidt RE. CMV complications in common variable immunodeficiency. Immunobiology. 2000;202:194–208. doi: 10.1016/S0171-2985(00)80066-2. [DOI] [PubMed] [Google Scholar]
- 31.Webster AD. Virus infections in primary immunodeficiency. J Clin Pathol. 1994;47:965–7. doi: 10.1136/jcp.47.11.965. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Raeiszadeh M, Kopycinski J, Paston SJ, et al. The T cell response to persistent herpes virus infections in common variable immunodeficiency. Clin Exp Immunol. 2006;146:234–42. doi: 10.1111/j.1365-2249.2006.03209.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Wang EC, Moss PA, Frodsham P, Lehner PJ, Bell JI, Borysiewicz LK. CD8highCD57+ T lymphocytes in normal, healthy individuals are oligoclonal and respond to human cytomegalovirus. J Immunol. 1995;155:5046–56. [PubMed] [Google Scholar]
- 34.Wheat WH, Cool CD, Morimoto Y, et al. Possible role of human herpesvirus 8 in the lymphoproliferative disorders in common variable immunodeficiency. J Exp Med. 2005;202:479–84. doi: 10.1084/jem.20050381. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Dray X, Treton X, Mazeron MC, et al. Herpes simplex virus type 1 colitis in a patient with common variable immunodeficiency syndrome. Eur J Gastroenterol Hepatol. 2006;18:541–4. doi: 10.1097/00042737-200605000-00015. [DOI] [PubMed] [Google Scholar]
- 36.Witt MD, Torno MS, Sun N, Stein T. Herpes simplex virus lymphadenitis: case report and review of the literature. Clin Infect Dis. 2002;34:1–6. doi: 10.1086/323400. [DOI] [PubMed] [Google Scholar]
- 37.van Doornum GJ, Guldemeester J, Osterhaus AD, Niesters HG. Diagnosing herpesvirus infections by real-time amplification and rapid culture. J Clin Microbiol. 2003;41:576–80. doi: 10.1128/JCM.41.2.576-580.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Levine P. Human herpesvirus-6 and human herpesvirus-7. In: Evans AS, Kaslow RA, editors. Viral infections of humans: epidemiology and control. 4th edn. New York: Springer; 1997. pp. 455–71. [Google Scholar]
- 39.Schmitz H, Wigand R, Heinrich W. Worldwide epidemiology of human adenovirus infections. Am J Epidemiol. 1983;117:455–66. doi: 10.1093/oxfordjournals.aje.a113563. [DOI] [PubMed] [Google Scholar]
- 40.Bauer U, Flunker G, Bruss K, et al. Detection of antibodies against adenovirus protein IX, fiber, and hexon in human sera by immunoblot assay. J Clin Microbiol. 2005;43:4426–33. doi: 10.1128/JCM.43.9.4426-4433.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Schagen FH, Moor AC, Cheong SC, et al. Photodynamic treatment of adenoviral vectors with visible light: an easy and convenient method for viral inactivation. Gene Ther. 1999;6:873–81. doi: 10.1038/sj.gt.3300897. [DOI] [PubMed] [Google Scholar]
- 42.Haveman LM, Bierings M, Legger E, et al. Novel pan-DR-binding T cell epitopes of adenovirus induce pro-inflammatory cytokines and chemokines in healthy donors. Int Immunol. 2006;18:1521–9. doi: 10.1093/intimm/dxl085. [DOI] [PubMed] [Google Scholar]
- 43.de Jager W, te Velthuis H, Prakken BJ, Kuis W, Rijkers GT. Simultaneous detection of 15 human cytokines in a single sample of stimulated peripheral blood mononuclear cells. Clin Diagn Lab Immunol. 2003;10:133–9. doi: 10.1128/CDLI.10.1.133-139.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Vignali DA. Multiplexed particle-based flow cytometric assays. J Immunol Methods. 2000;243:243–55. doi: 10.1016/s0022-1759(00)00238-6. [DOI] [PubMed] [Google Scholar]
- 45.Khan SS, Smith MS, Reda D, Suffredini AF, McCoy JP., Jr Multiplex bead array assays for detection of soluble cytokines: comparisons of sensitivity and quantitative values among kits from multiple manufacturers. Cytometry. 2004;61:35–9. doi: 10.1002/cyto.b.20021. [DOI] [PubMed] [Google Scholar]
- 46.Chapel H, Lucas M, Lee M, et al. Common variable immunodeficiency disorders: division into distinct clinical phenotypes. Blood. 2008;112:277–86. doi: 10.1182/blood-2007-11-124545. [DOI] [PubMed] [Google Scholar]
- 47.Stránská R, Schuurman R, de Vos M, van Loon AM. Routine use of a highly automated and internally controlled real-time PCR assay for the diagnosis of herpes simplex and varicella-zoster virus infections. J Clin Virol. 2004;30:39–44. doi: 10.1016/j.jcv.2003.08.006. [DOI] [PubMed] [Google Scholar]
- 48.Bayry J, Lacroix-Desmazes S, Kazatchkine MD, et al. Common variable immunodeficiency is associated with defective functions of dendritic cells. Blood. 2004;104:2441–3. doi: 10.1182/blood-2004-04-1325. [DOI] [PubMed] [Google Scholar]
- 49.Brooks DG, Ha SJ, Elsaesser H, Sharpe AH, Freeman GJ, Oldstone MB. IL-10 and PD-L1 operate through distinct pathways to suppress T-cell activity during persistent viral infection. Proc Natl Acad Sci USA. 2008;105:20428–320433. doi: 10.1073/pnas.0811139106. [DOI] [PMC free article] [PubMed] [Google Scholar]


