Abstract
KRAS mutations are found in nearly 90% of human pancreatic ductal adenocarcinomas (PDACs). However, mice genetically engineered to express KrasG12D from its endogenous locus develop PDACs only after a prolonged latency, indicating that other genetic events or pathway alterations are necessary for PDAC progression. The PTEN controlled PI3K/AKT signaling axis is known to be dysregulated in later stages of PDAC. To better elucidate the role of PTEN/PI3K/AKT signaling in KrasG12D-induced PDAC development, we crossed Pten conditional knockout mice (Ptenlox/lox) to mice with conditional activation of KrasG12D. The resulting compound heterozygous mutant mice showed significantly accelerated development of acinar-to-ductal metaplasia (ADM), mPanINs, and PDAC within a year. Moreover, all mice with KrasG12D activation and Pten homozygous deletion succumbed to cancer by three weeks of age. Our data supports a dosage-dependent role for PTEN, and the resulting dysregulation of the PI3K/AKT signaling axis, in both PDAC initiation and progression and sheds additional light on the signaling mechanisms that lead to the development of ADM and subsequent PanIN and pancreatic cancer.
Keywords: K-RAS, PTEN, pancreatic cancer, acinar to ductal metaplasia
Introduction
Pancreatic ductal adenocarcinoma (PDAC) is one of the deadliest of all cancers with a 5-year survival rate of less than 5%. Genetically engineered mouse models that recapitulate the human disease (1) have become essential tools in the dissection of the cellular and molecular pathogenesis of PDAC and the pre-clinical assessment of novel biomarkers and therapeutics. The most studied model to date has been the Pdx1-Cre-mediated expression of a knock-in mutant allele of Kras with a glycine to aspartic acid substitution (Pdx1-Cre;KrasG12D/+) from its endogenous locus. These mice slowly develop pre-malignant pancreatic intraepithelial neoplasias (mPanINs) with prolonged latencies and incomplete penetrance for the development of PDAC (2). However, PDAC developed far more rapidly when Pdx1-Cre;KrasG12D/+ mice were crossed with mice deficient in one of the genes commonly associated with PDAC, such as Ink4a/Arf(3), TGF-β receptor type 2 (Tgfbr2)(4), Smad4 (5), or carrying a mutation in p53 (6), although these molecular alterations alone do not lead to the development of pancreatic cancer in the absence of oncogenic Kras. These data indicate that additional genetic events beyond Kras mutation are responsible for pancreatic cancer progression. To date, the function of phosphatase and tensin homolog (PTEN) tumor suppressor which is mutated in PDAC far less frequently (7, 8), has yet to be examined in the context of mutant KrasG12D activation. In this study we examined the effects of concomitant conditional Pten deletion and KrasG12D activation and observed an accelerated and accentuated phenotype of acinar-to-ductal metaplasia (ADM), leading mPanIN and malignant progression. Our findings indicate that concurrent dysregulation of the PTEN/PI3K/AKT and RAS/RAF/MAPK pathways act synergistically to promote pancreatic cancer initiation and progression. This model provides us with a means to explore the role of PTEN and the PI3K-AKT signaling axis in pancreatic tumor development and treatment.
Materials and Methods
Mouse strains
To generate Pdx1-Cre+;Ptenlox/lox;KrasG12D/+ and Pdx1-Cre+;Ptenlox/+;KrasG12D/+ mice, we backcrossed the KrasG12D/+ line (2) (on a C57 background) to the Ptenlox/lox line (on a 129/BALB/c background) twice to generate KrasG12D/+;Ptenlox/lox mice. We then crossed KrasG12D/+;Ptenlox/loxp mice to Pdx1-Cre+;Ptenlox/+ mice we had previously generated (9) to produce experimental animals on a mixed C57/BL/6,129/BALB/c background. All studies were performed under the regulation of the Division of Laboratory Animal Medicine at the University of California at Los Angeles. KrasG12D primers, designed to recognize the KrasG12D mutant allele (10), and Pten primers, which detect the Pten floxed allele (11), were utilized to confirm the genotypes of mice from the breeding crosses.
Histology and immunohistochemistry
Immunohistochemical analysis was performed on formalin-fixed, paraffin-embedded tissue. Antigen retrieval was performed by heating the slides at 95°C in citrate buffer (pH 6.0) for 15 minutes prior to staining. The following primary antibodies were used: rabbit anti-phospho-AKT(Ser473) (Cell Signaling; 1:50), guinea pig anti-Pdx1 (a gift from Chris Wright; 1:5000), rabbit anti-ERK1/2(p44/p42) (Sigma; 1:100), rabbit anti-Ki67 (VP-RM04 )(Vector Labs; 1:1000), rabbit anti-Amylase (A8273)(Sigma; 1:500), mouse anti-Cytokeratin 19 (ab15463)(Abcam; 1:100), mouse anti-pan Cytokeratin (7H8C4)(Abcam; 1:500), mouse anti-Phospho-S6(Ser240/244) (Cell Signaling; 1:100), rat anti-mouse/human CD44 (14-0441)(eBiosciences; 1:50), and mouse anti-Smooth Muscle Actin α (2547) (Sigma; 1:1000), rabbit anti-PDGFR-β (28E1) (Cell Signaling; 1:100), rabbit anti-Cyclin D1 (92G2) (Cell Signaling; 1:25). Biotinylated DBA lectin (Vector) was used at 1:250 in HEPES/NaCl.
The collection of all human tumor samples used for this study was approved by the UCLA Institutional Review Board (IRB). The UCLA Pathology databases were used to identify patients with PDAC. Slides from the selected cases were reviewed. Anonymously labeled sections for immunohistochemistry studies, as detailed above, were prepared by the UCLA Department of Pathology Translational Pathology Laboratory Core facility. Additionally, an H&E stained slide was made from each block in order to confirm the diagnosis.
Proliferation index and statistic evaluation
The proliferation index for ADMs and mPanINs was determined by averaging the percentage of Ki67 positive cells per field for 10 fields at 400X magnification (n=4). Each field selected contained only ADMs or only mPanINs. The percentage of Ki67 positive cells was determined as the number of Ki67 positive cells per field (excluding positive cells in the stroma or untransformed acinar cells)/total cells per field (excluding cells in the stroma). Differences between mice in each cohort were evaluated by Student’s t-test. p < 0.05 was considered of statistical significance.
Histopathological scoring of murine pancreatic lesions
H&E-stained sections were reviewed by R.H. and H.W. and two pathologists with extensive experience in murine and human pancreas pathology (D.D. and S.D). Histopathologic lesions in mouse pancreata were scored in blinded fashion using serial step sections (20μm apart, 4 sections per mouse) and consensus criteria established at the 2004 Penn Workshop (12).
Laser-Capture Micro-dissection and LOH Analysis
Laser-capture microdissection (LCM) of H&E-stained pancreatic sections was performed using an Arcturus PixCell IIe laser capture microdissection system. Approximately fifty cells from mPanINs or PDACs in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice were collected on CapSure® HS LCM caps (Molecular Devices). DNA was extracted by sixteen hour incubation (55°C) in the ExtracSure sample extraction devices (Molecular Devices) with lysis buffer (10 mM Tris-HCl [pH 8.0], 1% Tween 20,100 μg Proteinase K). Following incubation, Proteinase K was inactivated by incubation at 99°C for 10 min, and 1μl aliquots of DNA were used for PCR analysis. PCR was performed for detection of wild-type, floxed, and recombined alleles of Pten using previously described primers (11).
Results
PTEN dosage controls pancreatic cancer progression in the Pdx1-Cre+;KrasG12D/+ mouse model
The prolonged latencies and incomplete penetrance for the development of PDAC observed in the Pdx1-Cre+;KrasG12D/+ mouse model prompted us to determine whether alteration of the PTEN/PI3K pathway could promote accelerated PDAC initiation and progression. For this, we crossed the Pdx1-Cre+;KrasG12D/+ model (2) with the Pten conditional knockout line we had previously generated (9). We found that Pten haploinsufficiency significantly shortened the life span of Pdx1-Cre+;KrasG12D/+ mice to a median survival of approximately 3.5 months (green line in Figure 1A; Pdx1-Cre+;KrasG12D/+;Ptenlox/+; n=45). Most Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice exhibited abdominal distension due to a combination of ascites fluid and pancreatic enlargement, at which time the animals were sacrificed. Nearly 100% of the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice succumbed to cancer burden by 12 months as compared to 90% survival of the Pdx1-Cre+;KrasG12D/+ model (blue line in Figure 1A; n=25). We examined sections from the pancreata of each transgenic model to score the presence of different grades of mPanIN or PDAC lesions which were defined by the consensus criteria of the pancreatic mouse modeling community (13). Histological examination showed evidence of accelerated PDAC development in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice compared to Pdx1-Cre+;KrasG12D/+ littermate controls. While Pdx1-Cre+;KrasG12D/+ mice exhibited limited early stage mPanIN lesions within an otherwise normal pancreas parenchyma at 3 months, Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed an almost complete loss of normal gland architecture and were involved by invasive ductal tumors histologically similar to human PDACs (Figure 1A, right and Figure 1B, bottom). Invasive cancers were defined as the presence of malignant ductal epithelial cells, identifiable by Cytokeratin-19 (CK19) staining, that had penetrated through the basement membrane into the surrounding smooth muscle actin (SMA) positive stroma (Figure 1B). Twenty one out of twenty five of Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice alive past two months of age developed well-to-moderately differentiated invasive ductal cancers. The spleen, liver, and lungs of tumor-bearing mice were examined, with only rare microscopic metastasis to the lungs noted (n=2, data not shown). The metastatic lung lesions histologically resembled the primary pancreatic tumor. The infrequent rate of metastasis indicates that either the mice rapidly succumb to their primary cancers prior to the time needed to develop metastatic lesions and/or that other genetic events are necessary for metastasis.
Figure 1. Mice with KrasG12D activation and Pten heterozygosity have decreased survival.
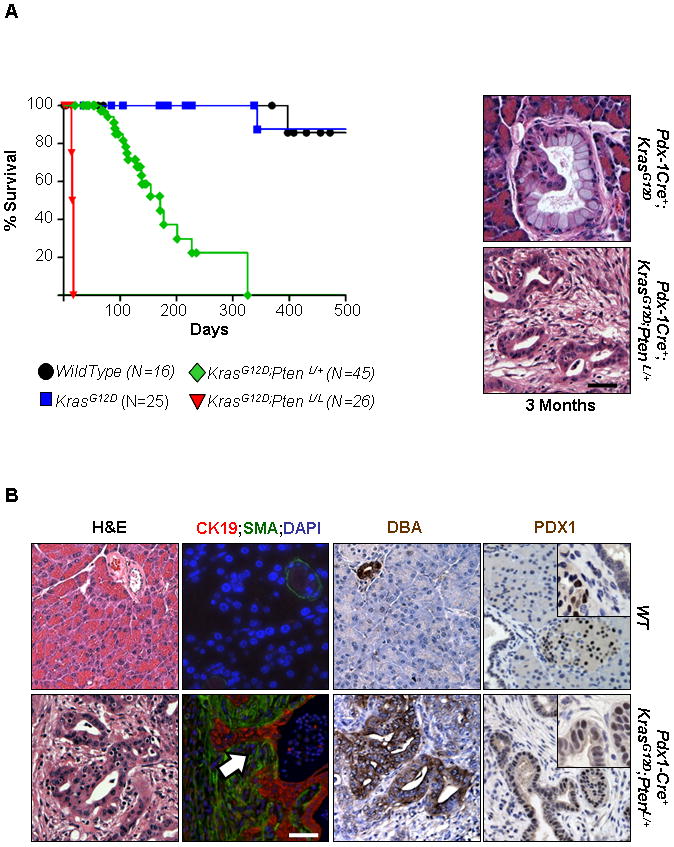
A Kaplan-Meier analysis reveal a median survival of approximately 17 days in Pdx1-Cre+;KrasG12D/+;Ptenlox/lox mice (n=26), significantly less than wild-type mice (n=16). Pten heterozygosity decreases the survival of mice with an activated KrasG12D mutation (n=45) which survive a significantly shorter time than mice with only an activated KrasG12D mutation alone (n=25) (p < 0.001, log-rank test, for each pair-wise combination). Low grade mPanIN lesion in a 3 month old Pdx1-Cre+;KrasG12D/+ mouse (right side, top) and a PDAC in a Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mouse of the same age (200x). B: Histological, immunofluorescence, and immunohistochemical analysis of 3 month old Pdx1-Cre+;KrasG12D/+;Ptenlox/+ (bottom row) and wild type mice (top row). H&E staining of representative histology of WT and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice shows PDAC lesions disrupting the normal glandular architecture found in WT mice (200x). Cells which display nuclear atypia, a high nuclear–to- cytoplasmic ratio, and loss of normal cell polarity can be observed in well-differentiated cancers in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice. These lesions exhibit invasive behavior as determined by the presence of ductal epithelium (Cytokeratin-19; red; white arrow) which is found invading into the surrounding desmoplastic stromal compartment (identified by smooth muscle actin (SMA) staining, green) (200x). The cells of these invasive cancers demonstrate the presence of lectin Dolichos biflorus agglutinin (DBA) which also marks normal ducts of WT mice (200x). Tumors also showed positive PDX-1 staining (200x; inset 400x), which stained islet cells but not ductal cells in WT mice (200x; inset 400x). Scale bars, 50μm.
Mice with KrasG12D activation and homozygous Pten deletion had an even earlier onset of lethal PDAC. Pdx1-Cre+;KrasG12D/+;Ptenlox/lox mice were moribund and either naturally succumbed or had to be euthanized prior to weaning (red line in Figure 1A; n=26). Histopathological examination of the pancreata of the Pdx1-Cre+;KrasG12D/+;Ptenlox/lox mice from postnatal day 1 to 17 (P1–P17) showed pancreatic enlargement (Supplemental Figure 1A, left) and severe exocrine pancreatic atrophy and edema. In addition, the presence of mPanINs, metaplasias, ducto-insular complexes (endocrine cells that contain aberrantly proliferating ductules (13)) and cancer formation was observed (Supplemental Figure 1B). Taken together, these data suggest that PTEN and PTEN controlled signaling pathways may serve as an important “brake” for KrasG12D-initiated PDAC progression. The rapid onset and lethality observed in the Pdx1-Cre+;KrasG12D/+;Ptenlox/lox mice precluded any detailed mechanistic analysis on the initiation and progression of PDAC, which led us to focus our attention on the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice.
Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice recapitulate the pathologic features of human PDACs
The hallmarks of human pancreatic cancers are an abundance of neoplastic cells expressing ductal markers, such as cytokeratin 19 (CK19) and Dolichos biflorus agglutinin (DBA) lectin (14), as well as their invasive behavior (12). Similar to human pancreatic cancers, the PDAC lesions in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ pancreata were positive for DBA and PDX-1, a transcription factor associated with early pancreatic development and known to be upregulated in human PDAC (Figure 1B) (15, 16). Using double immuno-fluorescence (IF) analysis, we observed that the majority of tumor cells in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ pancreata are CK19 positive and invasive, penetrating through the basement membrane into the stroma (Figure 1B, white arrow in the lower right panel), consistent with phenotypes observed in human PDAC. Taken together, these findings show that the tumors in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ PDAC mice resemble human PDAC on the histological level and can be used to study the molecular and genetic events leading to human PDAC initiation and progression.
PTEN haploinsufficiency determines the onset of mPanIN but complete PTEN loss is required for PDAC progression
Of the many different types of lesions thought to be the precursors to PDAC, PanIN lesions have been the best characterized (12). For this reason, we decided to first determine if Pten haploinsufficiency could accelerate mPanIN development by following three cohorts of mice (Pdx1-Cre+;Ptenloxp/+, Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+) from 1 to 6 months. We examined multiple sections from each pancreata (see methods) and scored each animal for the presence of mPanIN lesions of various grades or PDAC (Supplemental Figure 2A). At one month of age, neither Pdx1-Cre+;Ptenloxp/+ nor Pdx1-Cre+;KrasG12D/+ mice (Figure 2A, open or gray bar, respectively) showed mPanIN development (n=5 each cohort) while 2/6 Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (Figure 2A, black bar) showed mPanIN lesions. At three months of age all Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice examined (n=12) had mPanIN lesions while only 1/7 Pdx1-Cre+;KrasG12D/+ had mPanINs.
Figure 2. Mutant mice with KrasG12D activation and Pten heterozygosity have an earlier onset of mPanIN and PDAC.
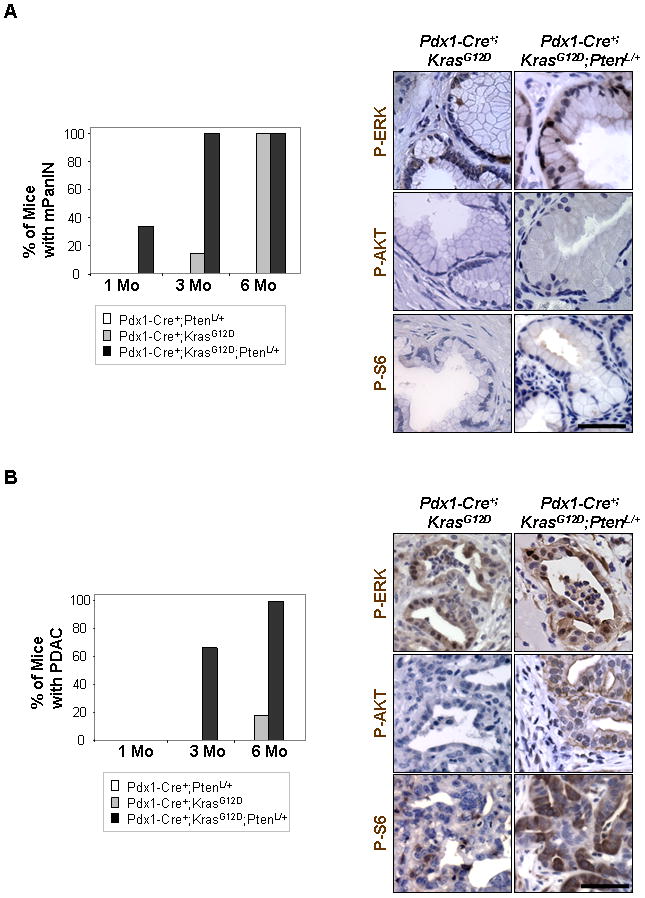
A Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (black bar) show earlier mPanIN development at 1 month of age and more frequent mPanIN development at 3 months of age compared to Pdx1-Cre+;PtenL/+ (white bar) or Pdx1-Cre+;KrasG12D/+ mice (gray bar). The mPanIN lesions that develop in both Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (age 3 months) and Pdx1-Cre+;KrasG12D/+ mice (age 3–6 months) show up-regulation of phosphorylated ERK but not phosphorylated AKT or S6 as demonstrated by IHC on sections containing mPanINs (right columns) (400x). B: Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice show earlier and more frequent onset of PDAC compared to mice with KrasG12D activation alone. Neoplastic ducts in PDACs of Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (age 6 months) show up-regulation of membrane bound phosphorylated AKT, ERK, and S6 compared to the neoplastic ducts in PDACs of Pdx1-Cre+;KrasG12D mice (age 6 months) which show only up-regulation of phosphorylated ERK (400x). A minimum of 4 non consecutive H&E sections from each mouse were examined for lesion scoring for each genotype. Scale bars, 50μm.
Having established that Pten haploinsufficiency led to earlier development of mPanIN lesions, we next examined if PDAC development was accelerated as well. At three months of age 8/12 of Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed the development of PDACs (Figure 2B, black bar) while Pdx1-Cre+;Ptenloxp/+ or Pdx1-Cre+;KrasG12D mice of the same age (n=5; n=7, respectively) show no evidence of PDAC development (Figure 2B, open or gray bar, respectively). Ultimately, all Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice that we examined at the six month time point (n=6) showed evidence of PDAC (Figure 2B, black bar), as compared to 2/11 of Pdx1-Cre+;KrasG12D/+ mice (Figure 2B, gray bar). Therefore, the PTEN/PI3K/AKT and RAS/RAF/MAPK pathways synergize at both initiation and progression steps of PDAC development.
We next determined if the synergy between the PTEN/PI3K/AKT and RAS/RAF/MAPK pathways in PDAC development requires complete loss of PTEN function or loss of heterozygosity (LOH). For this, we first evaluated the status of phosphorylated p44/42 MAPK (P-ERK), AKT (P-AKT) and S6 (P-S6), surrogate markers for dysregulated RAS/RAF/MAPK and PTEN/PI3K/AKT pathways, respectively, in the mPanINs and PDACs of Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice. IHC showed positive staining of P-ERK, but not P-AKT or P-S6, in the stage matched mPanINs lesions of both Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (Figure 2A, right panel), indicating that it was unlikely that LOH had happened at this stage in lesion development. On the other hand, PDACs in six month old Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed positive staining for P-ERK, P-AKT, and P-S6, strongly indicating that Pten LOH may have occurred in these lesions (Figure 2B, right panels).
To confirm Pten LOH indeed happened during PDAC progression, we used laser capture micro-dissection (LCM) to separate PDAC lesions from adjacent mPanINs in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice(n=5) (Supplemental 2B, left panels) and performed PCR analysis using DNA isolated from the micro-dissected tissues. As shown in right panel of Supplemental 2B, all of the PDAC lesions showed the presence of a band corresponding to the Cre-mediated Pten deletion product while 4/5 showed absence of the WT Pten allele, indicating Pten LOH had occurred in these samples (the WT band in sample #3 could have come from stromal cell contamination). In contrast, all adjacent mPanINs lesions showed the presence of the WT allele (Supplemental Figure 2B, right-lower panel, outlined in red). Taken together, these findings suggest that while Pten LOH contributes to PDAC development in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ model, Pten haploinsufficiency, as observed in other systems (17), is sufficient to cause accelerated mPanIN initiation.
ADM development precedes mPanIN formation in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice
When we examined the pathology of the Pdx1-Cre+;KrasG12D;Ptenlox/+ mice between one and three months of age, we observed, in addition to traditional mPanIN lesions, a large number of ADMs (Figure 3A, upper left panel; white arrow), similar to the metaplasias (12) we had observed in the Pdx1-Cre+;Ptenlox/lox model (9). These ADMs were concentrated in the “transition zone” between morphologically normal pancreatic structures and mPanIN and PDAC lesions and could be easily identified on H&E stained sections where ductal, tubular structures, often with mucinous cytoplasm, emerged from acinar cells. These structures can be numerous, replacing the normal acinar parenchyma and are thought to possibly arise from either acinar (18) or centroacinar cells (9). H&E stained sections from resected human tumors revealed similar pathology in areas adjacent to PDAC (Figure 3A, white arrow). Double IF confirmed that these lesions in fact contained duct-like cells (pan-cytokeratin-labeled; green) within acinar structures (amylase+; red) (Figure 3A, white arrow). ADMs were also observed in the Pdx1-Cre+;KrasG12D/+ mice but were not observed in wild-type controls (Figure 3B).
Figure 3. Mutant KrasG12D activation and Pten heterzygozity leads to earlier onset of ADM.
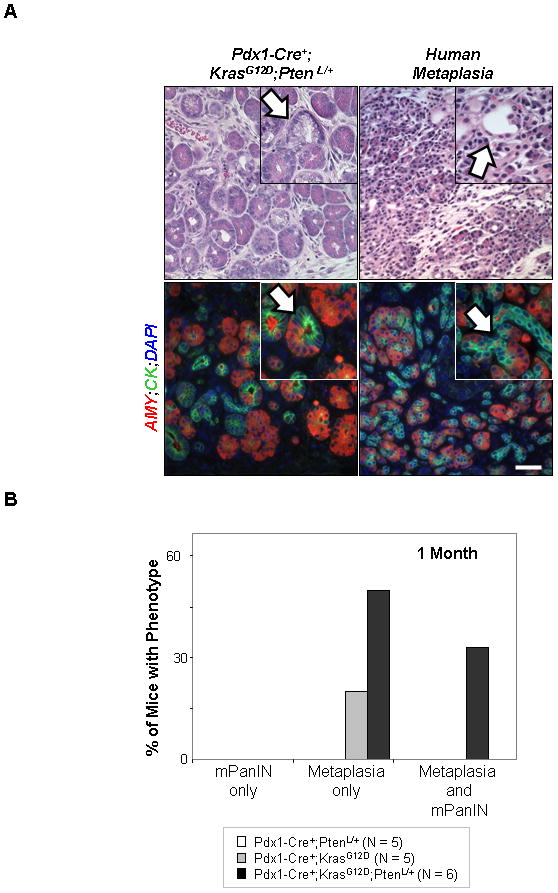
A: H&E staining shows the morphology of a zone of transition where diffuse metaplasia of acinar tissue to a ductal phenotype is observed in a 3 month old Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mouse (left column, white arrow) (400x; inset 600x). Immunofluorescence on a serial section shows that these ADMs contain duct-like cells (pan-cytokeratin-labeled; green; inset 60x; white arrow) and acinar cells (amylase+; red) (400x). H&E staining of slides with sections taken from areas adjacent to PDACs from human patients show similar transition zones where ADMs can be observed (right column, red arrow) (400x; inset 600x). Likewise, immunofluorescence on a serial section confirms the presence of structures containing both ductal (pan-cytokeratin-labeled; green; white arrow) and acinar cells (amylase+; red) (400x; inset 600x). B: Quantification of metaplasia and mPanIN presence in early lesion development. A minimum of five mice per genotype were analyzed with 4 non-consecutive sections examined per mouse. Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (black bar, n=6) show a higher number of mice with only ADMs compared to Pdx1-Cre+;KrasG12D/+ mice (gray bar, n=5) at 1 month of age. Scale bars, 50μm.
To decipher the relationship between ADM and mPanINs, we first investigated the chronological appearance of the two lesions. Although no mPanIN lesions were detected in the pancreata of Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;Ptenlox/+ mice at 1 month of age, 1/5 Pdx1-Cre+;KrasG12D/+ mice and 3/6 Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed ADM alone, and another 2/6 Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed both metaplasias and mPanIN lesions (Figure 3B). Interestingly, none of the animals developed mPanIN lesions only, suggesting that 1) metaplasias are the earliest pathological lesions identified in the Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice, 2) KRAS and PTEN/PI3K pathways collaborate to promote metaplasia development, and 3) ADMs likely serve as the precursors of mPanINs or parallel with mPanIN in pancreatic cancer development.
ADM may serve as the precursor of PDAC in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mouse model and human cancers
In order to investigate the functional significance of metaplasias in pancreatic cancer development, we quantified the proliferation of metaplasia and mPanIN lesions on stage-matched Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (see Materials and Methods) (Figure 4; n=4). The proliferation index of metaplasia lesions was significantly higher than that of the mPanINs lesions in both the Pdx1-Cre+;KrasG12D/+ (7.4% versus 2.4%) and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ cohorts (11.4% versus 6.2%) (Figure 4). Moreover, there was a significant increase in the proliferation rates of both types of lesions in Pdx-1Cre+;KrasG12D/+;Ptenlox/+ mice compared to Pdx-1Cre+;KrasG12D/+ mice. This shows that ADMs not only precede the development of mPanINs, but that Pten haploinsufficiency makes the already more rapidly dividing ADMs even more proliferative, which could contribute to accelerated disease initiation and progression.
Figure 4. Mutant KrasG12D activation and Pten heterzygozity leads to aggressive ductal metaplasia.
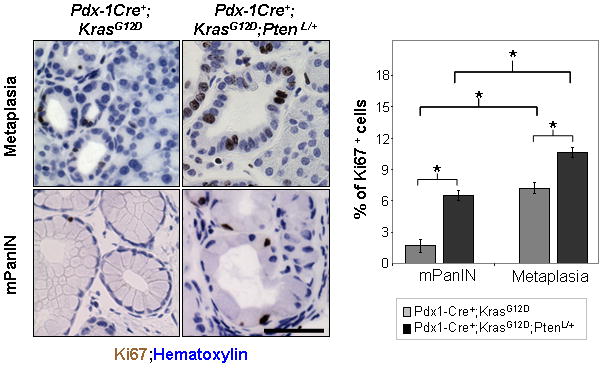
Quantification of proliferation in mPanINs and metaplasias. Left, sections from Pdx1-Cre+;KrasG12D/+ mice (n=4, age 3–6 months) and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (n=4, age 3 months) were stained with an antibody against the proliferation marker Ki67. The percentage of Ki67 positive cells for each type of lesion was determined by averaging the percentage of Ki67 positive cells per field (10 total which each contained only ADMs or only mPanINs). ADMs and mPanIN lesions in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (right column) show a higher percentage of cells positive for Ki67 than similar lesions in Pdx1-Cre+;KrasG12D/+ mice (left column) (400x); right, quantification. *p<.05. Scale bars, 50μm.
Increase in CD44 positive cells in murine and human metaplastic lesions
CD44 is a cell surface glycoprotein known to be up-regulated in human PDACs (19) and has been used as a marker to identify a stem-like sub-population in the pancreas(20). CD44 positive cells were present in the early ADMs in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ model as well as in human samples (Figure 5A, upper row; Supplemental Figure 3B) while almost absent in the nearby mPanINs (Figure 5A, lower row) or in normal acinar and ductal cells (Supplemental Figure 3A). Interestingly, we observed high CD44 expression in the invasive PDAC lesions in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (Figure 5B, upper row), which matches the CD44 staining pattern observed in human PDAC samples (Figure 5B, lower row). The level of CD44 staining was much less intense in the PDAC lesions in Pdx1-Cre+;KrasG12D/+ mice (Supplemental Figure 3A). Taken together, these data suggest that PTEN/PI3K and KRAS pathways collaborate in accelerating ADM development and in turn promote CD44 positive PDAC formation.
Figure 5. Mice with Pten heterozygosity and mutant KrasG12D activation show an increase in CD44 positive cells.
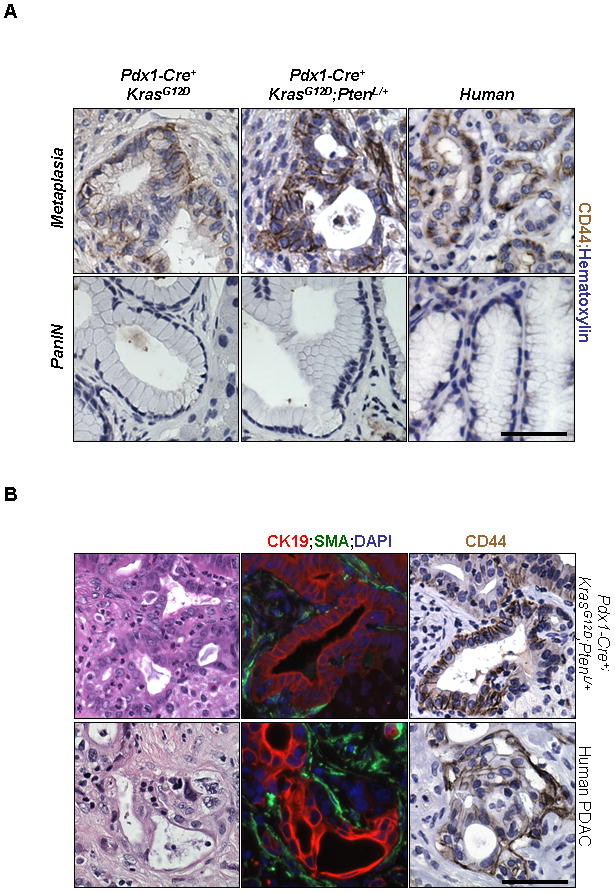
A Immunohistochemistry on Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice at 3 months of age showed that the cells of metaplasias (top row), but not mPanINs (bottom row), showed expression of CD44. This pattern of staining was also observed in areas adjacent to PDACs from human tumors which contained metaplasias and mPanINs (400x). B: H&E staining shows the morphology of a PDAC (400×) in a 6 month old Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mouse (top row). Immunofluorescence on a serial section confirms the presence of invading ductal epithelium (right panel, Cytokeratin-19; red/SMA staining, green). Immunohistochemistry shows that these invasive tumors show CD44 staining, matching the expression pattern of CD44 seen in human PDAC samples (bottom panel) (400x). Scale bars, 50μm.
Discussion
PTEN and pancreatic cancer development
Even with improvements in surgical techniques, the overall survival rate for patients with PDAC remains dismal. There is still much to be learned about the origin and development of PDAC, but unfortunately most of our knowledge about this disease comes from biopsies of patients with late stage tumors making it difficult to determine which cell types, and more importantly which molecular changes, are important for disease onset and progression. Animal models that faithfully recapitulate both the histology and molecular profile of the human disease have been an indispensable tool which has allowed us to dissect out the role various genetic events play in the initiation and progression of PDAC. Mutations in KRAS resulting in its constitutive activation occur in over 90% of PDACs (21). However, mice expressing KrasG12D from its endogenous locus progress to invasive cancers only after a prolonged latency (2) unless coupled with other genetic alterations commonly found in human PDAC (21). Previous studies using the KrasG12D mouse model, where median survival is 15 months (2), have shown that homozygous deletion of both p16Ink4A and p19Arf loci decreased survival to 8 weeks (3). Likewise, KrasG12D mice with either p53, type II TGF-β receptor gene (Tgfbr2), or Smad4 inactivation show accelerated development of PDACs with median survival times of 5, 4, or 2 months, respectively (4–6). Our study demonstrates that loss of PTEN function causes accelerated cancer development and decreased survival in the KrasG12D mouse model to either 3.5 months in the case of heterozygous Pten or 3 weeks with homozygous Pten deletion (Figure 1 and 2). Although point mutations or LOH in PTEN rarely occur in human PDAC (7, 8), there is evidence to support that loss of PTEN function or alteration of PTEN controlled signaling pathways can occur through alternative mechanisms, including promoter methylation (22), increased expression of AKT2 (23), or reduction of mRNA levels (24). In addition, the expression of several signaling pathways linked to PDAC development, including RAS (25), TGF-β (26, 27), and IGF-1 (28), have been shown to down regulate the expression of PTEN or inactivate PTEN’s catalytic activity, leading to reduced apoptosis (25), increased cell motility (26, 27), increased cell proliferation and invasion (28) in pancreatic cancer cells. Therefore, although PTEN itself is not mutated frequently in the human disease, PTEN and PTEN controlled pathways are being modulated in ways that are critical for disease development.
Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice recapitulate human PDAC
Histological and immunohistochemical analysis verified that the cancers in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice recapitulated the pathologic features of human PDACs (Figure 1B). Despite their rapid tumor onset, which is similar in latency to mice with p16Ink4A and p19Arf deletion, Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice don’t show the development of a large percentage of sarcomatoid tumors, a tumor type that makes up only a small portion of the human disease, but is prevalent in the Pdx1-Cre+; KrasG12D/+;Ink4a/Arflox/lox model (3). In this respect the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice are similar to the Ptf1a-Cre+;KrasG12D/+;Tgfbr2flox/flox mice which developed well-differentiated ductal adenocarcinomas with no evidence of sarcomatoid features (4). The drastic acceleration of PDAC development and early lethality seen in these two models may explain why metastases are only seen in a few cases when animals live well beyond the median survival age. The two metastases found in our Pdx1-Cre+;KrasG12D/+;Ptenlox/+ model occurred in mice at 20 and 24 weeks of age (data not shown). This is very close to the age of metastases development in the Ptf1a-Cre+;KrasG12D/+;Tgfbr2flox/flox model where metastases are discovered around 25 weeks (4). The limited number of metastases seen in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice compared to the highly metastatic Pdx1-Cre+;KrasG12D/+;Trp53R172H/+ and Pdx1-Cre+; KrasG12D/+;Ink4a/Arflox/lox models could indicate that either the mice succumb to their primary cancers prior to the time needed to develop metastatic lesions and/or that other genetic events found in late stage tumors (such as p16Ink4A, p19Arf, p53, or Smad4 deletion) (21) are necessary for metastases development.
Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice drive tumor progression through non-cell autonomous mechanisms?
The Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice provide a valuable resource for studying the alterations that are required to push an invasive cancer to one with metastatic potential. Even without evidence of prominent metastases, invasive tumors in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice show significant changes in not only the epithelial cell compartment but in the surrounding desmoplastic stroma as well, again recapitulating the nature of the human disease. Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed a significant stromal component (as marked by SMA-positive staining) surrounding both mPanINs and PDACs (Figure 1B). While investigating the nature of the desmoplastic stroma seen in the tumors of Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice we observed increased staining for platelet derived growth factor receptor beta (PDGFRβ) in the stroma of Pdx-1Cre+;KrasG12D;Ptenlox/+ mice compared to that of Pdx-1Cre+;KrasG12D alone. This staining pattern matched what was observed in the human samples (Supplemental Figure 3A), as human PDACs are characterized by induction of PDGFRβ expression in the stromal compartment (29). The presence of cells positive for PDGFRβ has been found to aid tumor angiogenesis through pericyte recruitment (30) and it is believed that targeting VEGFR and PDGFRβ may be capable of preventing the growth of some types of pancreatic cancers (31). This strongly suggests that combined alteration of the PTEN/PI3K and KRAS pathways in the epithelial cells can elicit a pro-tumorigenic response from the surrounding microenvironment through non-cell autonomous mechanisms.
KrasG12D activation and Pten haploinsufficiency is sufficient for accelerated lesion development
Our results show that Pten LOH corresponded only with PDAC development in this mouse model (Supplemental Figure 2B). This raises the question of what is driving earlier mPanIN or ADM development in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice since we cannot detect Pten LOH in either of these precursor lesions (Supplemental Figure 2B and 3C). This earlier onset could be brought about by the increased cell proliferation which results from the synergy between the KRAS and PI3K/AKT pathway alterations. Both ADMs and mPanINs in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed an increased rate of proliferation compared to their Pdx1-Cre+;KrasG12D/+ littermates (Figure 4). In addition, these early ADMs and PDACs in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice showed intense staining of Cyclin D1 (Supplemental Figure 3B), which has been shown to be regulated by PTEN levels (32, 33). This selective pressure to acquire additional genetic alteration brought about by this accelerated proliferation is likely what contributes to the Pten LOH seen in the PDACs in this model.
Moreover, PTEN is capable of regulating gene expression via hypoxia inducible factor 1 (HIF-1) (34). A study which identified genes regulated by Pten haploinsufficiency identified several known HIF-1-regulated genes including VEGF (35). Emerging evidence has linked loss of PTEN function with increased PDGFR-β expression in malignant tumors (36) so it is possible that in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice, Pten haploinsufficiency acts to induce HIF-1 which in turn could induce VEGFR and PDGFR-β expression. Thus, the PTEN pathways could play a critical role in controlling the angiogenesis of invasive PDACs. The Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice will provide a good resource to study how angiogenesis affects tumor development and whether targeting tumors with compounds such as Sunitinib (alone or in combination with other drugs) will yield therapeutic results.
Acinar Cell of Origin?
Recent work has shown that KrasG12D mice with alterations in SMAD4 (5) and TGF-β signaling (4) have an increase in ADM formation accompanying tumor acceleration. Likewise, expression of the KrasG12V/+ oncogene in the acinar/centroacinar lineage results in ADMs preceding the development of mPanINs and invasive PDAC (18). In addition, even though mice with KrasG12D activation and NOTCH gain of function did not progress to PDAC, a significant acceleration of ADM reprogramming leading to mPanIN formation was observed (37). Our analysis indicates that Pten haploinsufficiency in KrasG12D mice is also sufficient to accelerate the development of ADM lesions (Figure 2 and 3) adding to the growing lines of evidence (18, 37, 41) that shows that these ADMs are the earliest precursor lesions seen in the KrasG12D mouse models. The development of metaplasias precedes mPanINs in both the Pdx1-Cre+;KrasG12D/+ and Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice (Figure 3B) suggesting that while PTEN dosage plays a critical role in promoting metaplasia development, ADMs are likely the actual precursor lesions which give rise to the neoplastic ducts seen in PDAC regardless of PTEN function. Our hypothesis, which will require vigorous experimental testing utilizing more sophisticated methodologies such as lineage tracing, is that ADMs, independent of mPanIN development, are capable of giving rise to PDAC. We have also observed mPanIN/ADM hybrid lesions, as reported by other studies, which have linked metaplasia with mPanIN development (18, 41). Taken together, these observations indicate that some lesions traditionally identified as mPanINs most likely developed from the pre-existing metaplasias. The fact that we see more proliferation in ADMs than mPanINs (Figure 4) could indicate that mPanINs may be a more quiescent, intermediate state between ADM and PDAC (Figure 6). Alternatively, it is possible, as others have suggested (37), that mPanINs may represent a “dead-end” stage for highly proliferative precursor lesions that would have otherwise immediately progressed to PDAC. Given their earlier appearance and hyperproliferative features (Figure 4) it is possible that most ADMs generated in this model due to Pten haploinsufficiency could develop directly into PDACs, bypassing the mPanIN stage. Interestingly, no evidence of P-AKT was detectable by IHC in early metaplasias (Supplemental Figure 3C). Likewise, our data showed that similar to mPanINs, ADMs showed no evidence of Pten LOH (data not shown). Further studies utilizing isolated lesion populations will have to be performed to uncover the exact mechanism through which Pten haploinsufficiency leads to earlier ADM.
Figure 6. KrasG12D activation and PTEN loss collaborate to promote ADM, mPanIN, and pancreatic cancer development.
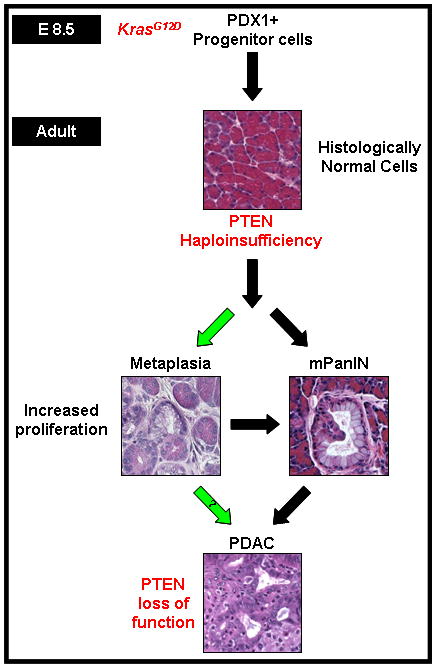
In the context of an existing activated KrasG12D mutation, PTEN haploinsufficiency drives ADM and mPanIN development with complete PTEN loss of function promoting PDAC development.
Regulation of Cancer Stem Cells?
In our study, we also observed that the ADMs contained more CD44-positive cells than mPanINs and PanINs (Figure 5A). Expression of CD44 has recently been utilized as a marker for isolating CSCs in many tumors (42, 43) and its expression has been linked to poor prognosis and gemcitabine resistance in pancreatic cancer (44). Moreover, metaplastic, highly aggressive, chemoresistant tumors in the breast (45) have been shown to have an increase in CD44+ cell populations as well as a high frequency of mutation, amplification, and activation of PI3-K/AKT pathway components(46). It will be interesting to test whether 1) metaplasias contain stem cell-like properties, 2) PTEN loss leads to the enrichment of tumor-initiating/stem cells, and 3) CD44 up-regulation found upon lesion initiation may aid tumor survival through PI3-K/AKT dependent pathways.
Previous work with mice with only Pten deficiency in the pancreas (9) showed that loss of PTEN function in Pdx1-Cre+;Ptenlox/lox mice led to centroacinar cell expansion which produced metaplasias similar to what is seen in Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice. However, while both models exhibit metaplasia, the tumors in Pdx1-Cre+;Ptenlox/lox mice are more papillary in nature while Pdx1-Cre+;KrasG12D/+;Ptenlox/+ tumors appear identical to Pdx1-Cre+;KrasG12D mice. This could indicate that the phenotype in the Pdx1-Cre+;Ptenlox/lox mice might reflect the consequence of Pten loss in the centroacinar cell compartment while the phenotype in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice might represent the combined effect of KRAS and PTEN signaling alteration in the acinar cells. Similar to results seen in our model, rapid ADM and increased mPanIN development was observed when KrasG12D and NOTCH were targeted directly to the acinar cells using the Elastase promoter (37). Interestingly, lesion initiation did not occur when either NOTCH activation or Pten deletion were targeted to the acinar cells alone (9). It will take further studies utilizing lineage targeting to elucidate whether it is the acinar, centroacinar, or non-islet associated Pdx-1+ cells (47) that are most sensitive to the effects of concurrent alteration of the KRAS and PTEN pathways.
Overall, our findings show that the histological and immunohistochemical profile of the tumors in the Pdx1-Cre+;KrasG12D/+;Ptenlox/+ mice mimic that of the human disease. This model provides a valuable resource for elucidating how loss of PTEN function aids tumor development and for determining whether targeting PI3-K/AKT signaling is a viable therapeutic strategy.
Supplementary Material
Acknowledgments
This work is supported, in part, by Damon Runyon Cancer Foundation and USHHS Ruth L. Kirschstein Institutional National Research Service Award T32 CA009056 (to R.H), (to J.H.C) UCLA Jonsson Comprehensive Cancer Center Seed Grant (to H.W) and NIH grants (UO1 CA84128 and RO1 CA107166 to H.W).
We thank the UCLA Tissue Procurement Core Laboratory for their technical help; Dr. Bangyan Stiles for sharing protocols; Antreas Hindoyan for helpful discussions. This work is supported, in part, by Damon Runyon Cancer Foundation and USHHS Ruth L. Kirschstein Institutional National Research Service Award T32 CA009056 (to R.H and J.H.C), UCLA Jonsson Comprehensive Cancer Center Seed Grant and NIH grants (UO1 CA84128 and RO1 CA107166 to H.W).
Footnotes
No potential conflicts of interest.
References
- 1.Bardeesy N, Sharpless NE, DePinho RA, Merlino G. The genetics of pancreatic adenocarcinoma: a roadmap for a mouse model. Semin Cancer Biol. 2001;11(3):201–18. doi: 10.1006/scbi.2000.0371. [DOI] [PubMed] [Google Scholar]
- 2.Hingorani SR, Petricoin EF, Maitra A, et al. Preinvasive and invasive ductal pancreatic cancer and its early detection in the mouse. Cancer Cell. 2003;4(6):437–50. doi: 10.1016/s1535-6108(03)00309-x. [DOI] [PubMed] [Google Scholar]
- 3.Aguirre AJ, Bardeesy N, Sinha M, et al. Activated Kras and Ink4a/Arf deficiency cooperate to produce metastatic pancreatic ductal adenocarcinoma. Genes Dev. 2003;17(24):3112–26. doi: 10.1101/gad.1158703. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Ijichi H, Chytil A, Gorska AE, et al. Aggressive pancreatic ductal adenocarcinoma in mice caused by pancreas-specific blockade of transforming growth factor-beta signaling in cooperation with active Kras expression. Genes Dev. 2006;20(22):3147–60. doi: 10.1101/gad.1475506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Kojima K, Vickers SM, Adsay NV, et al. Inactivation of Smad4 accelerates Kras(G12D)-mediated pancreatic neoplasia. Cancer Res. 2007;67(17):8121–30. doi: 10.1158/0008-5472.CAN-06-4167. [DOI] [PubMed] [Google Scholar]
- 6.Hingorani SR, Wang L, Multani AS, et al. Trp53R172H and KrasG12D cooperate to promote chromosomal instability and widely metastatic pancreatic ductal adenocarcinoma in mice. Cancer Cell. 2005;7(5):469–83. doi: 10.1016/j.ccr.2005.04.023. [DOI] [PubMed] [Google Scholar]
- 7.Sakurada A, Suzuki A, Sato M, et al. Infrequent genetic alterations of the PTEN/MMAC1 gene in Japanese patients with primary cancers of the breast, lung, pancreas, kidney, and ovary. Jpn J Cancer Res. 1997;88(11):1025–8. doi: 10.1111/j.1349-7006.1997.tb00324.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Okami K, Wu L, Riggins G, et al. Analysis of PTEN/MMAC1 alterations in aerodigestive tract tumors. Cancer Res. 1998;58(3):509–11. [PubMed] [Google Scholar]
- 9.Stanger BZ, Stiles B, Lauwers GY, et al. Pten constrains centroacinar cell expansion and malignant transformation in the pancreas. Cancer Cell. 2005;8(3):185–95. doi: 10.1016/j.ccr.2005.07.015. [DOI] [PubMed] [Google Scholar]
- 10.Jackson EL, Willis N, Mercer K, et al. Analysis of lung tumor initiation and progression using conditional expression of oncogenic K-ras. Genes Dev. 2001;15(24):3243–8. doi: 10.1101/gad.943001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Wang S, Gao J, Lei Q, et al. Prostate-specific deletion of the murine Pten tumor suppressor gene leads to metastatic prostate cancer. Cancer Cell. 2003;4(3):209–21. doi: 10.1016/s1535-6108(03)00215-0. [DOI] [PubMed] [Google Scholar]
- 12.Hruban RH, Rustgi AK, Brentnall TA, Tempero MA, Wright CV, Tuveson DA. Pancreatic cancer in mice and man: the Penn Workshop 2004. Cancer Res. 2006;66(1):14–7. doi: 10.1158/0008-5472.CAN-05-3914. [DOI] [PubMed] [Google Scholar]
- 13.Hruban RH, Adsay NV, Albores-Saavedra J, et al. Pathology of genetically engineered mouse models of pancreatic exocrine cancer: consensus report and recommendations. Cancer Res. 2006;66(1):95–106. doi: 10.1158/0008-5472.CAN-05-2168. [DOI] [PubMed] [Google Scholar]
- 14.Murtaugh LC, Stanger BZ, Kwan KM, Melton DA. Notch signaling controls multiple steps of pancreatic differentiation. Proc Natl Acad Sci U S A. 2003;100(25):14920–5. doi: 10.1073/pnas.2436557100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Offield MF, Jetton TL, Labosky PA, et al. PDX-1 is required for pancreatic outgrowth and differentiation of the rostral duodenum. Development. 1996;122(3):983–95. doi: 10.1242/dev.122.3.983. [DOI] [PubMed] [Google Scholar]
- 16.Gu G, Dubauskaite J, Melton DA. Direct evidence for the pancreatic lineage: NGN3+ cells are islet progenitors and are distinct from duct progenitors. Development. 2002;129(10):2447–57. doi: 10.1242/dev.129.10.2447. [DOI] [PubMed] [Google Scholar]
- 17.Trotman LC, Niki M, Dotan ZA, et al. Pten dose dictates cancer progression in the prostate. PLoS Biol. 2003;1(3):E59. doi: 10.1371/journal.pbio.0000059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Guerra C, Schuhmacher AJ, Canamero M, et al. Chronic pancreatitis is essential for induction of pancreatic ductal adenocarcinoma by K-Ras oncogenes in adult mice. Cancer Cell. 2007;11(3):291–302. doi: 10.1016/j.ccr.2007.01.012. [DOI] [PubMed] [Google Scholar]
- 19.Castella EM, Ariza A, Ojanguren I, et al. Differential expression of CD44v6 in adenocarcinoma of the pancreas: an immunohistochemical study. Virchows Arch. 1996;429(4–5):191–5. doi: 10.1007/BF00198333. [DOI] [PubMed] [Google Scholar]
- 20.Li C, Heidt DG, Dalerba P, et al. Identification of pancreatic cancer stem cells. Cancer Res. 2007;67(3):1030–7. doi: 10.1158/0008-5472.CAN-06-2030. [DOI] [PubMed] [Google Scholar]
- 21.Hruban RH, Goggins M, Parsons J, Kern SE. Progression model for pancreatic cancer. Clin Cancer Res. 2000;6(8):2969–72. [PubMed] [Google Scholar]
- 22.Asano T, Yao Y, Zhu J, Li D, Abbruzzese JL, Reddy SA. The PI 3-kinase/Akt signaling pathway is activated due to aberrant Pten expression and targets transcription factors NF-kappaB and c-Myc in pancreatic cancer cells. Oncogene. 2004;23(53):8571–80. doi: 10.1038/sj.onc.1207902. [DOI] [PubMed] [Google Scholar]
- 23.Altomare DA, Tanno S, De Rienzo A, et al. Frequent activation of AKT2 kinase in human pancreatic carcinomas. J Cell Biochem. 2002;87(4):470–6. doi: 10.1002/jcb.10287. [DOI] [PubMed] [Google Scholar]
- 24.Ebert MP, Fei G, Schandl L, et al. Reduced PTEN expression in the pancreas overexpressing transforming growth factor-beta 1. Br J Cancer. 2002;86(2):257–62. doi: 10.1038/sj.bjc.6600031. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Vasudevan KM, Burikhanov R, Goswami A, Rangnekar VM. Suppression of PTEN expression is essential for antiapoptosis and cellular transformation by oncogenic Ras. Cancer Res. 2007;67(21):10343–50. doi: 10.1158/0008-5472.CAN-07-1827. [DOI] [PubMed] [Google Scholar]
- 26.Chow JY, Quach KT, Cabrera BL, Cabral JA, Beck SE, Carethers JM. RAS/ERK modulates TGFbeta-regulated PTEN expression in human pancreatic adenocarcinoma cells. Carcinogenesis. 2007;28(11):2321–7. doi: 10.1093/carcin/bgm159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Chow JY, Dong H, Quach KT, Van Nguyen PN, Chen K, Carethers JM. TGF-beta mediates PTEN suppression and cell motility through calcium-dependent PKC-alpha activation in pancreatic cancer cells. Am J Physiol Gastrointest Liver Physiol. 2008;294(4):G899–905. doi: 10.1152/ajpgi.00411.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Ma J, Sawai H, Ochi N, et al. PTEN regulates angiogenesis through PI3K/Akt/VEGF signaling pathway in human pancreatic cancer cells. Mol Cell Biochem. 2009;331(1–2):161–71. doi: 10.1007/s11010-009-0154-x. [DOI] [PubMed] [Google Scholar]
- 29.Fjallskog ML, Hessman O, Eriksson B, Janson ET. Upregulated expression of PDGF receptor beta in endocrine pancreatic tumors and metastases compared to normal endocrine pancreas. Acta Oncol. 2007;46(6):741–6. doi: 10.1080/02841860601048388. [DOI] [PubMed] [Google Scholar]
- 30.Furuhashi M, Sjoblom T, Abramsson A, et al. Platelet-derived growth factor production by B16 melanoma cells leads to increased pericyte abundance in tumors and an associated increase in tumor growth rate. Cancer Res. 2004;64(8):2725–33. doi: 10.1158/0008-5472.can-03-1489. [DOI] [PubMed] [Google Scholar]
- 31.Bergers G, Song S, Meyer-Morse N, Bergsland E, Hanahan D. Benefits of targeting both pericytes and endothelial cells in the tumor vasculature with kinase inhibitors. J Clin Invest. 2003;111(9):1287–95. doi: 10.1172/JCI17929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Radu A, Neubauer V, Akagi T, Hanafusa H, Georgescu MM. PTEN induces cell cycle arrest by decreasing the level and nuclear localization of cyclin D1. Mol Cell Biol. 2003;23(17):6139–49. doi: 10.1128/MCB.23.17.6139-6149.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Rodriguez OC, Lai EW, Vissapragada S, et al. A reduction in Pten tumor suppressor activity promotes ErbB-2-induced mouse prostate adenocarcinoma formation through the activation of signaling cascades downstream of PDK1. Am J Pathol. 2009;174(6):2051–60. doi: 10.2353/ajpath.2009.080859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Zhong H, Chiles K, Feldser D, et al. Modulation of hypoxia-inducible factor 1alpha expression by the epidermal growth factor/phosphatidylinositol 3-kinase/PTEN/AKT/FRAP pathway in human prostate cancer cells: implications for tumor angiogenesis and therapeutics. Cancer Res. 2000;60(6):1541–5. [PubMed] [Google Scholar]
- 35.Li L, He F, Litofsky NS, Recht LD, Ross AH. Profiling of genes expressed by PTEN haploinsufficient neural precursor cells. Mol Cell Neurosci. 2003;24(4):1051–61. doi: 10.1016/j.mcn.2003.08.010. [DOI] [PubMed] [Google Scholar]
- 36.Thorarinsdottir HK, Santi M, McCarter R, et al. Protein expression of platelet-derived growth factor receptor correlates with malignant histology and PTEN with survival in childhood gliomas. Clin Cancer Res. 2008;14(11):3386–94. doi: 10.1158/1078-0432.CCR-07-1616. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.De La OJ, Emerson LL, Goodman JL, et al. Notch and Kras reprogram pancreatic acinar cells to ductal intraepithelial neoplasia. Proc Natl Acad Sci U S A. 2008;105(48):18907–12. doi: 10.1073/pnas.0810111105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Brembeck FH, Schreiber FS, Deramaudt TB, et al. The mutant K-ras oncogene causes pancreatic periductal lymphocytic infiltration and gastric mucous neck cell hyperplasia in transgenic mice. Cancer Res. 2003;63(9):2005–9. [PubMed] [Google Scholar]
- 39.Grippo PJ, Nowlin PS, Demeure MJ, Longnecker DS, Sandgren EP. Preinvasive pancreatic neoplasia of ductal phenotype induced by acinar cell targeting of mutant Kras in transgenic mice. Cancer Res. 2003;63(9):2016–9. [PubMed] [Google Scholar]
- 40.Tuveson DA, Zhu L, Gopinathan A, et al. Mist1-KrasG12D knock-in mice develop mixed differentiation metastatic exocrine pancreatic carcinoma and hepatocellular carcinoma. Cancer Res. 2006;66(1):242–7. doi: 10.1158/0008-5472.CAN-05-2305. [DOI] [PubMed] [Google Scholar]
- 41.Zhu L, Shi G, Schmidt CM, Hruban RH, Konieczny SF. Acinar cells contribute to the molecular heterogeneity of pancreatic intraepithelial neoplasia. Am J Pathol. 2007;171(1):263–73. doi: 10.2353/ajpath.2007.061176. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Tanaka H, Nakamura M, Kameda C, et al. The Hedgehog signaling pathway plays an essential role in maintaining the CD44+CD24-/low subpopulation and the side population of breast cancer cells. Anticancer Res. 2009;29(6):2147–57. [PubMed] [Google Scholar]
- 43.Snyder EL, Bailey D, Shipitsin M, Polyak K, Loda M. Identification of CD44v6(+)/CD24- breast carcinoma cells in primary human tumors by quantum dot-conjugated antibodies. Lab Invest. 2009;89(8):857–66. doi: 10.1038/labinvest.2009.54. [DOI] [PubMed] [Google Scholar]
- 44.Hong SP, Wen J, Bang S, Park S, Song SY. CD44-positive cells are responsible for gemcitabine resistance in pancreatic cancer cells. Int J Cancer. 2009;125(10):2323–31. doi: 10.1002/ijc.24573. [DOI] [PubMed] [Google Scholar]
- 45.Liu R, Wang X, Chen GY, et al. The prognostic role of a gene signature from tumorigenic breast-cancer cells. N Engl J Med. 2007;356(3):217–26. doi: 10.1056/NEJMoa063994. [DOI] [PubMed] [Google Scholar]
- 46.Hennessy BT, Gonzalez-Angulo AM, Stemke-Hale K, et al. Characterization of a naturally occurring breast cancer subset enriched in epithelial-to-mesenchymal transition and stem cell characteristics. Cancer Res. 2009;69(10):4116–24. doi: 10.1158/0008-5472.CAN-08-3441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Gidekel Friedlander SY, Chu GC, Snyder EL, et al. Context-dependent transformation of adult pancreatic cells by oncogenic K-Ras. Cancer Cell. 2009;16(5):379–89. doi: 10.1016/j.ccr.2009.09.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


