Abstract
Tumor progression in vitro has traditionally been studied in the context of two-dimensional (2D) environments. However, it is now well accepted that 2D substrates are unnaturally rigid compared to the physiological substrate known as extracellular matrix (ECM) that is in direct contact with both normal and tumorigenic cells in vivo. Hence, the patterns of interactions, as well as the strategies used by cells in order to penetrate the ECM, and migrate through a three-dimensional (3D) environment are notoriously different than those observed in 2D. Several substrates, such as collagen I, laminin, or complex mixtures of ECM components have been used as surrogates of native 3D ECM to more accurately study cancer cell behaviors. In addition, 3D matrices developed from normal or tumor-associated fibroblasts have been produced to recapitulate the mesenchymal 3D environment that assorted cells encounter in vivo. Some of these substrates are being used to evaluate physico-mechanical effects on tumor cell behavior. Physiological 3D ECMs exhibit a wide range of rigidities amongst different tissues while the degree of stromal stiffness is known to change during tumorigenesis. In this review we describe some of the physico-mechanical characteristics of tumor-associated ECMs believed to play important roles in regulating epithelial tumorigenic behaviors.
Keywords: Tumor-associated stroma, extracellular matrix, matrix architecture, stromal stiffness, 3D matrices, mechanobiology and cell-derived matrices
Tumorigenesis occurs within dynamically changing environments; cancer cells reside within a plethora of distinct biological locations as they advance through stages during tumor initiation, progression and invasion [1]. Cancer cells reside within a primary site where they are first transformed into a malignant phenotype (i.e. acquisition of hyper proliferate behavior in an uncontrolled manner). As the tumor progresses, the basement membrane, that physically separates the epithelial from the connective (mesenchymal) tissue, is degraded thus facilitating a direct interaction between cancer cells and the tumor-associated mesenchymal stroma [2]. In addition, during their invasive stage, cancer cells spend some time suspended within fluids that serve as cell transportation means such as lymph and blood and then they extravasate from the hematogenous compartment into distant sites where cancer cells will colonize and eventually metastasize [3]. Interestingly, many of these compartments where cancer cells reside are changed and affected by the tumorigenic process itself (Figure 1) and, in turn, these altered microenvironments are believed to facilitate tumor progression [1, 3–5]. Epithelial cells are believed to induce or suppress a different set of genes in order to accomplish the changing physiological activities needed during the different steps of tumor progression [6–9]. In this context, the classical paradigm of epithelial tumorigenesis (e.g., tumor development and cell invasion to the underlying stroma) has been typically described as an exclusively epithelial-centered mechanism. In this review, we emphasize the physico-mechanical microenvironmental changes that are observed in the tumor-associated stromal compartments during tumorigenesis, which are considered to be important promoters of tumor development, progression and metastasis.
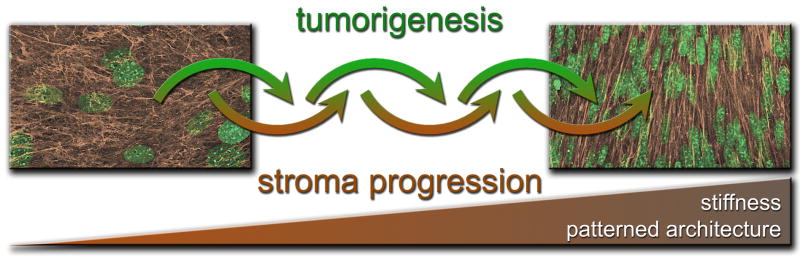
Figure 1.
Composite describing how tumorigenesis and mesenchymal stroma progression are two processes which affect and incite each other. The photographs represent confocal 3D reconstructions of fibroblast-derived ECMs (in brown) and cell nuclei (in green) where the disorganized in vitro stroma is shown in the left panel while the parallel patterned architecture of tumor-associated stromal matrix is evident in the right panel. The gradient progression bar in brown, at the bottom of the composite, represents the increased stiffness and architectural patterning of the stroma during these joint progression processes. The two confocal images are a variation from images published in Amatangelo, Bassi et al. 2005 [20].
The re-consideration of the stroma as a key player in development and tumorigenesis came back after decades of studies focused almost entirely in the epithelial counterpart of these processes. Probably, one of the reasons of this lag has been due to the complex constitution of the stroma, as a highly structured compartment. The cancerous stroma contains fibroblasts, immune cells, adipocytes (e.g., in breast cancer), and myofibroblasts, in addition to increased levels of selected extracellular matrix (ECM) proteins, such as collagen I [7]. The function that these altered components play in the tumorigenic process is relatively poorly understood, but extensive studies are currently undertaken in order to characterize these structures, their relationship, and the spatial and temporal organization of the stroma, as well as the role that tumor-stroma plays, initially preventing and later on, promoting tumor progression (Figure 1). The interaction between cancer cells and their microenvironment, promotes tumor growth and also protects them from innate immune response [7]. It has been suggested that the functional association of cancer cells with their altered tissue of origin forms a new and dynamic ‘organ-like tissue’ that changes as malignancy progresses [10]. Investigation of this process might provide new insights into the mechanisms of tumorigenesis, and could also lead to the development of new therapeutic targets.
1-Stroma and 3D matrices
Not only is the ECM a mere scaffold used by developing and/or cancerous cells, but it also plays a major role during these processes [11]. Despite the rather complex organization of the stromal ECM, several three dimensional (3D) systems that aim to recapitulate various aspects of the in vivo microenvironmental settings have been developed [12]. Many of these 3D systems have focused on the use of collagen I [13], which constitutes one of the main components that are altered and over-expressed during tumorigenesis at the mesenchymal stromal compartment [14]. In addition, many aspects of tissue development and tumorigenesis have been effectively studied using a basement membrane material rich in laminin [15], which has also been shown to reconstitute many aspects of the microenvironmental settings needed to induce in vivo-like epithelial cell behaviors [16, 17]. More recently, sophisticated in vitro systems such as epithelial-mesenchymal organotypic constructs [18, 19] and fibroblasts-derived 3D matrices provided an alternative view to assist in decreasing the gap between in vitro and in vivo systems [5, 12]. To this end, it has been shown that primary fibroblasts produce mesenchymal 3D matrices which effectively mimic the ECMs corresponding to their original mesenchymal in vivo counterparts [20–22]. In fact, the composition of these matrices proved to be more complex than traditional 3D collagen or laminin, reflecting more accurately the makeup and architecture of the in vivo mesenchymal ECMs [12, 23, 24].
1.1 Collagen 3D gels
Collagen I, member of the fibrillar collagen family, is one of the most abundant structural proteins of the interstitial ECM [25]. Since collagen I can spontaneously polymerize in vitro, extracted (using acidic conditions) collagen I from mammalian tendons can be used to produce, after pH neutralization, allogenic 3D gels which are believed to mimic many aspects of the mesenchymal mammalian microenvironment. These 3D gels, though weaker than the parental natural ECM, recapitulate many aspects of the biological in vivo mesenchymal ECMs [26]. In this context, 3D collagen gels have been used to study fibroblast contraction and migration [27], angiogenesis [28], as well as tumor cell migration [29–31]. Historically, collagen I has received special attention in breast development and carcinogenesis since its altered organization is known to drive breast cancer initiation and tumorigenesis in animal models [32, 33]. Moreover, there is a very well established correlation between increased collagen I density and greater risk for breast cancer development [34, 35]. There are also strong indications that cells could “sense” different degrees of collagen stiffness and respond to it [36, 37]. Collagen I 3D gels can be studied as floating or attached substrates. Cells grown in collagen gels attached to a surface encounter a more stressed and loaded environment that those grown in floating collagen matrices. More than three decades ago Emerman and Pitelka observed that cells cultured on floating collagen matrices formed alveolar structures, and maintained a long lasting differentiated phenotype [38]. However, these same cells grown on collagen gels that remained attached to the Petri dishes, lose their secretory and differentiation abilities [38]. These distinct 3D architectures also elicit completely different epithelial or epithelial-stromal cell arrangements. The normal yet immortalized cell line MCF10A, grown on floating collagen gels effectively mimics in vitro, breast gland development in vivo [39]. In this context, the cells produce in vivo-like acini and tubular structures that will later develop into ducts. Strikingly, attached gels fail to produce these mature structures, yielding instead sheets of cells arranged in patterns that are parallel to the gel periphery [39]. Similar to this set of experiments, normal and transformed mammary epithelial cells such as MCF10A, and T47D produced tubule or duct-like structures. However, when grown in high density collagen matrices, these cells present larger morphologies, greater cell densities and their luminal space is filled up with cells. This differential behavior is attributed to the increased stiffness of the denser matrices, pointing to a crucial effect of substrate rigidity and resistance to matrix contraction on epithelial cell behavior [40]. Increased substrate stiffness results in activation of the non-receptor focal adhesion kinase (FAK) and small GTP binding protein (RhoGTPase) pathways, leading to increased cell proliferation and invasive phenotype changes including changes in gene expression [40].
1.2 Laminin
Laminin is one of the main basement membrane components believed to be responsible for many of the ECM-regulated activities observed on epithelial cells while inducing and supporting tumor initiation and early tumor development [41]. The role of laminin and its role in matrix elasticity has been linked to the acquisition and/or maintenance of epithelial cell polarity [42] and to the formation of acinar and tubular structures in mammary (and other) epithelial cells [43]. In fact, laminin 1 is believed to be responsible for both the ‘softness’ of the breast stroma, and its signaling, believed to be transmitted though β1-integrin and to be necessary for the expression of β-casein in mammary epithelial cells [41]. Moreover, it was shown that blocking the activity of β1-integrin in the breast epithelial cell line SCp2 resulted in abrogation of cell elasticity. This observation suggested a unique role for this integrin on the process of “sensing” the physical variations imparted upon cells by the altered ECM cues [41]. Cellular elasticity is believed to be largely the result of the contribution of the actin-myosin cytoskeleton [41]. In fact, blockage of actin polymerization, induction of myosin II kinase or Rho kinase activities, resulted in decreased cell stiffness, and consequently, in increased cell spreading when SCp2 cells where cultured under classic rigid 2D conditions. Cells cultured on laminin-rich 3D matrices display flexibility and elasticity. However, these cells were not sensitive to the actin-myosin inhibitors, suggesting that cells may exhibit differences in the responses to drugs depending on their underlying 3D substrate [41].
1.3 Fibroblast-derived 3D matrices
Three-dimensional matrices derived from fibroblasts in vitro have been shown to impart in vivo-like responses onto cells cultured using these matrices as substrates [21, 23, 44]. Stromal fiber organization, fibroblast morphology, and gene expression patterns varied not only within the stroma of different normal tissues, but also between normal and tumor-associated stroma [20, 45, 46]. These features suggest that the mesenchymal stromal ECM actively participates in tumor progression and metastasis, thus prompting investigators to consider an approach where both stroma and epithelial cells are integrated. As tumor progresses, epithelial and stromal cells influence each other (Figure 1), reflecting progressively malignant patho-physiological stages [4, 5]. It is believed that the normal stroma is restrictive of tumorigenesis [47]. Indeed, fibroblasts of the normal stroma exert a protective barrier against hyper-proliferation and invasion of epithelial cells [48] while in an opposite way, at later stages, the stroma becomes more permissive to epithelial cell proliferation and invasion [49]. Although this process is believed to be reversible, fibroblasts may co-evolve with the malignant epithelial cells and progress to an irreversible state of progressive tumor and stroma [5, 50, 51]. Tumor-associated fibroblasts (TAFs) acquire a myofibroblastic (e.g., desmoplastic) structural phenotype reflected by the expression of α-smooth muscle actin together with additional stromal markers such as fibroblast activating protein, desmin and others [14, 52]. In fact, TAFs have been shown to organize themselves in characteristic parallel patterns (Figure 1) that may support tumor migration, invasion, and proliferation, and also to favor the spread of tumor cells into distant organs [20, 32, 53]. The fact that fibroblasts differ between normal and tumor-associated stroma [45, 46, 54, 55] and that matrices derived from these distinct fibroblasts effectively recapitulate the in vivo ECM differences and specific normal and tumor-associated architectural characteristics [20–22], make these cell-derived mesenchymal 3D systems to be attractive means for the study of ECM effects on cancer cell behaviors. This system has been used to study fibronectin fibrillogenesis [56], physical aspects of fibrillar fibronectin [57], matrix-regulated signal transduction [21, 53, 58, 59], cell invasion [44, 60, 61], cell adhesion and dynamics [62], matrix induced drug responses [63], as well as effective drug screening [64].
2. Mechanobiology: role of the matrix architectural organization
Epithelial cell behavior can be modulated according to intrinsic characteristics of the epithelial cells, i.e. patterns of gene expression due to differentiation stage and the presence of somatic mutations, in case of malignant cells [65, 66]. In addition, external cues such as extrinsic soluble factors, availability of substrate ligands, direct effects imparted by neighboring cells and the physical properties that the ECM exerts onto cells can epigenetically affect epithelial cell behaviors [67]. Therefore, it is well accepted that the mechanical properties of the stroma, its topography and compliance are related to the biological influences imparted upon epithelial cells and together they dictate cellular behaviors [68]. In relatively soft tissues, a compliant or flexible extracellular matrix favors the development of normal structures and acts as a barrier against tumor growth and invasion. The notion that physical features of the environment control natural cell behavior implies that traditional two-dimensional (2D) cell culturing conditions that provide a flat and rigid environment are unnatural as they elicit cell responses to extreme stiffness (linked to some tumorigenic processes) as opposed to more compliant (natural) 3D settings that are typical for supporting normal cell growth [69]. The forces that a cell “senses” are rather different when the cell growths onto a 2D substrate or within a 3D microenvironmental setting. In the first situation, restricted surfaces of the cell (i.e., ventral or basal surface) are attached to the provided substrate, allowing only for basal mechanical-generated signals [70]. However, cells immersed within 3D scaffolds interact with this microenvironment in a way that the influence of traction forces can be imparted in every direction [70, 71]. As a matter of fact, it has been shown that cues “sensed” by cells under 3D conditions are transmitted through the cell body and affect the nucleus dynamics in a way that they even regulate the expression of specific genes [72]. A resent work using micropatterned wells demonstrated that three-dimensionality and changes in stiffness can influence single cell physiology and cytoskeletal organization [73]. ECM stiffness is often quantified by calculating the Young modulus (E values) usually in Pascal units (Pa). For instance, soft tissues exhibit a low E value (0.5–2 kPa). Conversely, the rigidity of the trabecular bone has a one hundred times higher E value (2.5 GPa). Normal mammary tissues are quite soft (0.15 kPa), but this low stiffness is greatly modified during the process of tumorigenesis. Indeed, an advanced invasive mammary tumor microenvironment goes through a 10 to 20 fold increase in its rigidity, reaching a staggering E value of ~4 kPa. This increase in tautness is believed to greatly influence cell behavior and facilitate tumor progression [1]. Interestingly, investigators have shown that factors related with ECM remodeling, such as TGF-β can be exposed and become activated during mechano-environmental changes such as stretching of the ECM fibers [74]. Thus, physico-mechanical factors of the microenvironment are belived to play a crucial role in cellular responses.
2.1 Matrix stiffness regulates cell behavior: natural collagen cross-linking
As a major component of the ECM, collagen I constitutes an ideal candidate to study the changes in the biomechanical properties of the cellular microenvironment during tumor progression [1]. Increased collagen deposition has been associated to augmented risk for breast cancer [35]. Similarly, increased collagen cross-linking has been shown to lead to enhanced acquisition of malignant features [37]. Elevated expression of lysil-oxidase (LOX), an enzyme necessary for natural collagen I cross-linking [75], has been linked in premalignant cells, to increased fibrillar collagen deposition and linearization, inducing integrin clustering and phosphorylation of integrin-regulated effectors, such as non-receptor tyrosine kinases like focal adhesion kinase (FAK) and p130Crk-associated substrate (p130Cas), leading to the promotion of invasive behaviors [1, 37]. Levental et. al., recently showed that ECM stiffness increases from normal to premalignant to tumor. Strikingly, the stiffness of the tumor adjacent stroma was shown to be elevated in conjunction with the above-mentioned increases in levels of LOX, and linearization of collagen [37]. Conversely, blockage of LOX activity propagated non tumorigenic latency and lowered the incidence of tumor formation in animals injected with breast cancer cells. Moreover, in vivo LOX inhibition resulted in the development of pre-malignant and/or low-grade neoplasias [37]. These studies suggest that matrix stiffness plays a decisive role in the alteration of biochemical pathways that lead to cell transformation. The same group of investigators demonstrated that mammary epithelial cells grown within 3D matrices at physiological Young modulus (E values of 160 to 170 Pa) form small growth-arrested colonies with polarized β4-integrin and apical-lateral cortical actin, which are all features found in normal mammary epithelium [76]. Strikingly, a small increase in stiffness (400 Pa) promoted the formation of double-sized colonies while further increases in matrix stiffness, closer to those exhibit by tumor-associated ECM, stimulated the formation of greater colonies with atypical (tumorigenic) acini structures and altered integrin and actin polarization [76]. The study concluded that increased matrix stiffness generates the tension necessary to cluster α5β1-integrin, increasing the length of cell-matrix adhesions thus facilitating cell migration and invasion. In fact, the study demonstrated that integrin clustering stimulated FAK phosphorylation, RhoA activity and cytoskeleton contractility, all factors known to enhance cell migration and spreading [76]. It is believed that increased ECM stiffness also exerts increased invasive effects on tumor cells since it allows for greater traction forces that can be used by the cells to migrate to areas of nutrients availability, promoting survival. This migration also requires integrin signaling transmitted through Rho GTPases, which results in augmented actomyosin cytoskeleton contractility [77, 78]. Lauffenburger and Horwits have identified four stages during invasion; protrusion, attachment, localized ECM degradation, and rear end detachment [79]. During the first two stages, cells may need a firm and stiff substrate, such as collagen I cross-linked matrices, to exert propulsive traction forces within the leading migration edge, sometimes referred to anterior traction zones [80]. For the other two stages, where cells need to detach via proteolytic mechanisms, matrices’ mechanochemical properties may display quite different characteristics [80].
2.2. Migration and invasion are altered by the mechanical properties of ECMs
During the process of intravasation, cells migrate through the stroma and display an invasive behavior characterized by the formation of a plethora of different projections known as lamellipodia, pseudopodia, and invadopodia [81–83]. In vivo cell migration through mesenchymal compartments is believed to be accompanied by active proteolysis [80]. Membrane type 1 matrix metalloproteinase (MT1-MMP) has been identified as one of the main ECM-degrading protease used by migrating fibroblasts and in epithelial to mesenchymal transduced invasive cancer cells (which behave similarly to fibroblasts), at the major surface in contact with the ECM [84, 85]. Migratory proteolysis has been associated to integrin positive structures localized nearby the cell’s leading edge (at the lamelopodium) under classic 2D conditions [86, 87]. In contrast, in 3D environments, numerous steric constrains, as well as matrix factors, such as matrix density, pore size, stiffness, and susceptibility to proteolytic degradation predict a more complex situation. The migratory patterns of cells within 3D environments have recently been mapped with the aid of diverse microscopic techniques, such as scanning electron microscopy [80, 88, 89]. Interestingly, when cells are cultured within 3D ECMs, matrix degradation occurs at diverse regions of the invading cell as opposed to only at the front edge. These regions include, the leading edge, compression zones at the mid-body region, and the trailing edge [80]. The assorted regions exhibit distinct morphologies and are apparently exposed to variation of ECM stiffness [80, 90, 91]. The leading edge of cells migrating through 3D substrates develops actin-rich, thin and cylindrical pseudopodia organized in a manner that facilitates “pulling” the cell forward [92]. In gliomas [93] and in smooth muscle cells [94], migration ids believed to be promoted by pseudopodia formation and to be favored by increased ECM stiffness, which is “sensed” in an integrin-dependent manner and is regulated through Rho-A dependent cytoskeletal contractions. In this context, it is believed that cells tend to migrate towards areas of increased stromal stiffness [76, 95]. After the initial attachment of pseudopodia to stiff ECMs, the bulk of the cell located posterior to the traction front may not be able to effectively penetrate the elastic and contracting ECM, therefore necessitating an additional prolonged and active focus of pericellular proteolytic activity. In order to prevent getting trapped due to steric hindrance, long-lived foci of MT1-MMP localized immediately in the rear back of the leading edge degrading the immediately adjacent ECM and therefore eliminating this physical obstacle [80]. In this context, traction and proteolysis are localized in close, albeit distinct, regions of the cell and are believed to be regulated in response to physically distinct microenvironmental cues [80]. The cell’s mid-body diameter increases as the cell moves forward and areas of pericellular proteolysis that contain MT1-MMP, as well as β1-integrin positive structures and F-actin, form a distinct structure that is similar to characteristic 3D-matrix adhesions [21, 23], suggesting a coupling between proteolysis and movement [96]. In summary, it is hypothesized that the proteolytic front of the invasive cell carves the pathway for the bulkier nucleus containing central zone of the cell; the proteolytic processes facilitated by MT1-MMP appears to be localized lateral to invadopodia-like spikes. Interestingly, the retracting rear edge of the migrating cell contains zones of MMP-2 and MT1-MMP. However, the pattern of proteolytic areas in the rear end is diffuse and appears less localized. Nevertheless, proteolytic activity in this zone generates fragments of fibronectin, collagen, and laminin, which compete with the non degraded ECM for cellular adhesion sites, facilitating cell detachment [80, 97, 98]. The altered ECM composition results in the facilitated release of growth and chemotactic factors, allowing additional cells to incorporate into this altered microenvironmental compartment [80, 99, 100] Also, this alteration of the ECM leads to areas of least resistance, and lower than normal stiffness, pointing to an unusual soft matrix, and decreased cellular attachments which induce cell rounding [92]. As long as cells advance in their migratory path, proteolytic processed collagen appears to realign forming low resistance “microtracks,” favoring migration.
Interestingly, investigators have found that different extracellular factors can regulate both migratory and contractile features of cells thus modifying their morphologies [101]. In fact, Grinnell and colleagues demonstrated that during fibroblast-collagen matrix interactions, traction forces exerted by the cells can cause cells to migrate. Nonetheless, if the matrix cannot resist the cellular traction force, then the matrix will tend to “move” therefore preventing migration [102]. Recent studies shed new light on the implications of the physical aspects of cell-matrix interactions in cancer cell behaviors [103, 104]. In fact, the authors suggested that integrins, which are the main receptors regulating cell-matrix interactions, are to be studied as the sensors or regulators that transmit biochemical information into cells in response to physical and topographical variations of the microenvironment [104]. Moreover, Zaman et. al., suggested that highest migratory speeds lie at regions where intermediate stiffness and relatively low force adhesion structures are formed as well as regions of relatively high adhesion and low stiffness when integrin binding is blocked [105]. In other words, migratory characteristics are governed by both integrin availability and changing of the environmental stiffness at hand.
3. Role of matrices produced by fibroblasts
In physiological conditions, during homeostasis, the ECM is maintained within a pre-existing micro environmental ‘status quo.’ Local matrix-producing fibroblasts, embedded within mesenchymal (connective tissue) environments “sense” the mechanical properties of the homeostatic matrix and respond accordingly to maintain this status. For example, cells can impart intrinsic forces onto cell-derived fibronectin elastic fibers to keep the naturally unfolded molecules stretched [106]. In fact, it has been suggested that cellular contractility may be necessary for the assembly of fibronectin fibers [107]. Nevertheless, under special circumstances such as wound healing, developmental processes or disease (e.g. cancer) mechanical changes that occur locally are “sensed” by resident cells, such as fibroblasts, which in turn respond to these changes by altering the ECM and transforming the environment into one that differentially regulates the activity and behavior of both resident and newly recruited cells [5, 36].
Stretching of fibronectin exposes cryptic sites on one of the molecule’s globular domains, FnIII, [108, 109]. As a consequence, the binding of newly synthesized soluble fibronectin occurs, directly regulating fibrillogenesis [57] and favoring de-novo deposition of matrix. In sharp contrast to the native gels, when fibroblasts are seeded onto artificially cross-linked matrices, the newly deposited fibronectin appears to be highly stretched, with similar proportion of unfolded fibronectin than in the non cross linked matrices. In addition, these recently deposited fibers do not necessarily co-localize with the pre-existing fibers [57]. In fact, studies performed using isolated fibronectin fibers [57, 110] showed that to strain fibronectin fibers within a cross-linked matrix, a force of 5.5 μN is required as opposed only 1.7 μN needed for the non-cross linked matrix. In this context, cytoskeleton-generated tension may not be sufficient to stretch cross-linked matrices, decreasing the unfolding of fibronectin fibers and impairing new deposition of fibronectin fibers [57]. Taken together, these studies suggest that altered matrix deposition that promotes increased tensional forces, as observed during tumorigenesis and wound healing, could play decisive roles in these pathologies.
4. Matrix topography and specific architectural composition
It has been suggested that the topographical organization of substrates can greatly affect cell responses [61]. Physical interactions within the ECM strongly depend on fiber orientation, and not only on its stiffness. However, ECM architecture can be also characterized by the shapes that epithelial or mesenchymal cells encounter in the process of migration and invasion. The shape and the area where a cell resides may determine its behavior. For example, Killian et al [111] studied the effect of assorted ECM shapes, using patterned substrates, on adherent mesenchymal cell differentiation. This study demonstrated that mesenchymal stem cells can differentiate into adipocytes or osteoblasts, depending on the geometric cues of the provided substrate. Similarly, others have shown that cells grown in sharp-edge surfaces express higher amounts of proteins involved in osteogenic programs of differentiation, such as RhoA, Rac, and Cdc42, ROCK kinase [112]. Indeed, patterned substrates have been used to study force induced proliferation due to local mechanical stress [113]. Moreover, magnetic microposts organized in assorted architectural topographies have been suggested as new tools to mimic and study the mechanical forces that are imparted by ECMs on cell behaviors [114]. Patterned substrates have also been used to induce variations in the polarization state of cells. Different substrate shapes, which are believed to induce altered sub cellular curvatures, affect the cytoskeleton response and thus trigger different cell responses [115].
Looking at 3D substrates, additional studies have shown that cancer cell invasion strategies are directly affected by the architecture of ECMs [116]. Interestingly, in vitro assorted fibroblast-derived 3D matrices effectively reproduce the parallel vs. disorganized patterned characteristics (Figure 1) of tumor-associated and normal ECM stroma in vivo [20, 22]. This observation suggests that these systems could be used for in vitro studies on which to assess matrix effects on cancer cell behaviors. Altogether these findings indicate that the architecture of the niches where cancer cells reside may be critical for their tumorigenic behavior and therefore, drugs targeted at impairing specific architectural features of the microenvironment should be looked at as possible novel therapies to inhibit invasive progression.
5. Conclusion
There is an increasing body of evidences that point to the ECM as a crucial aspect of tumorigenic progression, wound healing, and differentiation. Although the ECM is structurally complex, different mechano-physical and architectural elements have been considered as contributing to tumor progression. As new techniques are being developed, the interactions of one cell with just one fiber [117], the production of matrices spanning a wide range of stiffness [37, 76, 91], and the utilization of patterned substrates [113, 116] are now being determined. The biological significance of these parameters is rapidly emerging.
Drug development largely takes into account cellular targets, focusing in the alterations occurring to cells during disease progression, such as, genomic changes, variations in gene expression, and distorted signaling networks. However, it is increasingly recognized that the role of the environment, in particular the stroma, may decisively affect the outcome of therapies [118, 119]. Interestingly, increased matrix stiffness results in higher efficiency of gene expression [120]. Dosages and therapeutic regimens may be affected by matrix stiffness; thus, the degree of stiffness of the matrix should be determined prior to deciding on a particular protocol treatment.
Predictive medicine seeks to analyze large amounts of data such as patterns of gene expression, cell-cell signaling, and microenvironment cues to perform computational modeling, and provide models outcomes of disease. Network biology takes into account the multiple signals a cell receives and how the processing of all these signals may produce a response [121]. Needless to say that among the vast array of signals perceived by cells, the physical characteristics of the environment are sure to play a prominent role.
Acknowledgments
In this review we intended to present an overview of the vast body of literature. Therefore, we have inadvertently omitted specific studies. Nevertheless, we would like to state that their omission does not diminish their significance.
The authors thank Drs. J. Franco-Barraza and R. López de Cicco for their helpful suggestions and comments, as well as Mrs. A. Carson for the assertive proofreading. This work is supported by a NIH/NCI grants CA113451 (EC) and CA06927, as well as by the Fox Chase cancer Center’s Kidney Keystone Initiative.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- 1.Butcher DT, Alliston T, Weaver VM. A tense situation: forcing tumour progression. Nat Rev Cancer. 2009;9(2):108–122. doi: 10.1038/nrc2544. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Kunz-Schughart LA, Knuechel R. Tumor-associated fibroblasts (Part II): functional impact on tumor tissue. Histol Histopathol. 2002;17(2):623–637. doi: 10.14670/HH-17.623. [DOI] [PubMed] [Google Scholar]
- 3.Geho DH, Bandle RW, Clair T, Liotta LA. Physiological mechanisms of tumor-cell invasion and migration. Physiology (Bethesda) 2005;20:194–200. doi: 10.1152/physiol.00009.2005. [DOI] [PubMed] [Google Scholar]
- 4.Geiger TR, Peeper DS. Metastasis mechanisms. Biochim Biophys Acta. 2009;1796(2):293–308. doi: 10.1016/j.bbcan.2009.07.006. [DOI] [PubMed] [Google Scholar]
- 5.Beacham DA, Cukierman E. Stromagenesis: the changing face of fibroblastic microenvironments during tumor progression. Semin Cancer Biol. 2005;15(5):329–341. doi: 10.1016/j.semcancer.2005.05.003. [DOI] [PubMed] [Google Scholar]
- 6.Nowell PC. Tumor progression: a brief historical perspective. Semin Cancer Biol. 2002;12(4):261–266. doi: 10.1016/s1044-579x(02)00012-3. [DOI] [PubMed] [Google Scholar]
- 7.Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57–70. doi: 10.1016/s0092-8674(00)81683-9. [DOI] [PubMed] [Google Scholar]
- 8.Michor F, Iwasa Y, Nowak MA. Dynamics of cancer progression. Nat Rev Cancer. 2004;4(3):197–205. doi: 10.1038/nrc1295. [DOI] [PubMed] [Google Scholar]
- 9.Fusenig NE, Boukamp P. Multiple stages and genetic alterations in immortalization, malignant transformation, and tumor progression of human skin keratinocytes. Mol Carcinog. 1998;23(3):144–158. doi: 10.1002/(sici)1098-2744(199811)23:3<144::aid-mc3>3.0.co;2-u. [DOI] [PubMed] [Google Scholar]
- 10.Fidler IJ. The pathogenesis of cancer metastasis: The ‘seed and soil’ hypothesis revisited. Nature Reviews Cancer. 2003;3(6):453–458. doi: 10.1038/nrc1098. [DOI] [PubMed] [Google Scholar]
- 11.Bissell MJ, Radisky D. Putting tumours in context. Nat Rev Cancer. 2001;1(1):46–54. doi: 10.1038/35094059. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Yamada KM, Cukierman E. Modeling tissue morphogenesis and cancer in 3D. Cell. 2007;130(4):601–610. doi: 10.1016/j.cell.2007.08.006. [DOI] [PubMed] [Google Scholar]
- 13.Grinnell F. Fibroblast biology in three-dimensional collagen matrices. Trends Cell Biol. 2003;13(5):264–269. doi: 10.1016/s0962-8924(03)00057-6. [DOI] [PubMed] [Google Scholar]
- 14.Desmouliere A, Guyot C, Gabbiani G. The stroma reaction myofibroblast: a key player in the control of tumor cell behavior. Int J Dev Biol. 2004;48(5–6):509–517. doi: 10.1387/ijdb.041802ad. [DOI] [PubMed] [Google Scholar]
- 15.Kleinman HK, McGarvey ML, Hassell JR, Star VL, Cannon FB, Laurie GW, Martin GR. Basement membrane complexes with biological activity. Biochemistry. 1986;25(2):312–318. doi: 10.1021/bi00350a005. [DOI] [PubMed] [Google Scholar]
- 16.Vukicevic S, Somogyi L, Martinovic I, Zic R, Kleinman HK, Marusic M. Reconstituted basement membrane (Matrigel) promotes the survival and influences the growth of murine tumors. Int J Cancer. 1992;50(5):791–795. doi: 10.1002/ijc.2910500521. [DOI] [PubMed] [Google Scholar]
- 17.Weaver VM, Howlett AR, Langton-Webster B, Petersen OW, Bissell MJ. The development of a functionally relevant cell culture model of progressive human breast cancer. Semin Cancer Biol. 1995;6(3):175–184. doi: 10.1006/scbi.1995.0021. [DOI] [PubMed] [Google Scholar]
- 18.Chioni AM, Grose R. Organotypic modelling as a means of investigating epithelial-stromal interactions during tumourigenesis. Fibrogenesis Tissue Repair. 2008;1(1):8. doi: 10.1186/1755-1536-1-8. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Smalley KS, Lioni M, Herlyn M. Life isn’t flat: taking cancer biology to the next dimension. In Vitro Cell Dev Biol Anim. 2006;42(8–9):242–247. doi: 10.1290/0604027.1. [DOI] [PubMed] [Google Scholar]
- 20.Amatangelo MD, Bassi DE, Klein-Szanto AJ, Cukierman E. Stroma-derived three-dimensional matrices are necessary and sufficient to promote desmoplastic differentiation of normal fibroblasts. Am J Pathol. 2005;167(2):475–488. doi: 10.1016/S0002-9440(10)62991-4. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Cukierman E, Pankov R, Stevens DR, Yamada KM. Taking cell-matrix adhesions to the third dimension. Science. 2001;294(5547):1708–1712. doi: 10.1126/science.1064829. [DOI] [PubMed] [Google Scholar]
- 22.Quiros RM, Valianou M, Kwon Y, Brown KM, Godwin AK, Cukierman E. Ovarian normal and tumor-associated fibroblasts retain in vivo stromal characteristics in a 3-D matrix-dependent manner. Gynecologic Oncology. 2008;110(1):99–109. doi: 10.1016/j.ygyno.2008.03.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Cukierman E, Pankov R, Yamada KM. Cell interactions with three- dimensional matrices. Curr Opin Cell Biol. 2002;14(5):633–639. doi: 10.1016/s0955-0674(02)00364-2. [DOI] [PubMed] [Google Scholar]
- 24.Xiao Z, Blonder J, Zhou M, Veenstra TD. Proteomic analysis of extracellular matrix and vesicles. J Proteomics. 2009;72(1):34–45. doi: 10.1016/j.jprot.2008.11.011. [DOI] [PubMed] [Google Scholar]
- 25.Gelse K, Poschl E, Aigner T. Collagens--structure, function, and biosynthesis. Adv Drug Deliv Rev. 2003;55(12):1531–1546. doi: 10.1016/j.addr.2003.08.002. [DOI] [PubMed] [Google Scholar]
- 26.Pedersen JA, Swartz MA. Mechanobiology in the third dimension. Ann Biomed Eng. 2005;33(11):1469–1490. doi: 10.1007/s10439-005-8159-4. [DOI] [PubMed] [Google Scholar]
- 27.Brown RA, Prajapati R, McGrouther DA, Yannas IV, Eastwood M. Tensional homeostasis in dermal fibroblasts: mechanical responses to mechanical loading in three-dimensional substrates. J Cell Physiol. 1998;175(3):323–332. doi: 10.1002/(SICI)1097-4652(199806)175:3<323::AID-JCP10>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 28.Yang S, Graham J, Kahn JW, Schwartz EA, Gerritsen ME. Functional roles for PECAM-1 (CD31) and VE-cadherin (CD144) in tube assembly and lumen formation in three-dimensional collagen gels. Am J Pathol. 1999;155(3):887–895. doi: 10.1016/S0002-9440(10)65188-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Friedl P, Zanker KS, Brocker EB. Cell migration strategies in 3-D extracellular matrix: differences in morphology, cell matrix interactions, and integrin function. Microsc Res Tech. 1998;43(5):369–378. doi: 10.1002/(SICI)1097-0029(19981201)43:5<369::AID-JEMT3>3.0.CO;2-6. [DOI] [PubMed] [Google Scholar]
- 30.Maaser K, Wolf K, Klein CE, Niggemann B, Zanker KS, Brocker EB, Friedl P. Functional hierarchy of simultaneously expressed adhesion receptors: integrin alpha2beta1 but not CD44 mediates MV3 melanoma cell migration and matrix reorganization within three-dimensional hyaluronan-containing collagen matrices. Mol Biol Cell. 1999;10(10):3067–3079. doi: 10.1091/mbc.10.10.3067. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Schor SL, Allen TD, Harrison CJ. Cell migration through three-dimensional gels of native collagen fibres: collagenolytic activity is not required for the migration of two permanent cell lines. J Cell Sci. 1980;46:171–186. doi: 10.1242/jcs.46.1.171. [DOI] [PubMed] [Google Scholar]
- 32.Provenzano PP, Eliceiri KW, Campbell JM, Inman DR, White JG, Keely PJ. Collagen reorganization at the tumor-stromal interface facilitates local invasion. BMC Med. 2006;4(1):38. doi: 10.1186/1741-7015-4-38. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Provenzano PP, Inman DR, Eliceiri KW, Knittel JG, Yan L, Rueden CT, White JG, Keely PJ. Collagen density promotes mammary tumor initiation and progression. BMC Med. 2008;6:11. doi: 10.1186/1741-7015-6-11. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Alowami S, Troup S, Al-Haddad S, Kirkpatrick I, Watson PH. Mammographic density is related to stroma and stromal proteoglycan expression. Breast Cancer Res. 2003;5(5):R129–135. doi: 10.1186/bcr622. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Boyd NF, Lockwood GA, Byng JW, Tritchler DL, Yaffe MJ. Mammographic densities and breast cancer risk. Cancer Epidemiol Biomarkers Prev. 1998;7(12):1133–1144. [PubMed] [Google Scholar]
- 36.Discher DE, Janmey P, Wang YL. Tissue cells feel and respond to the stiffness of their substrate. Science. 2005;310(5751):1139–1143. doi: 10.1126/science.1116995. [DOI] [PubMed] [Google Scholar]
- 37.Levental KR, Yu H, Kass L, Lakins JN, Egeblad M, Erler JT, Fong SF, Csiszar K, Giaccia A, Weninger W, et al. Matrix crosslinking forces tumor progression by enhancing integrin signaling. Cell. 2009;139(5):891–906. doi: 10.1016/j.cell.2009.10.027. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Emerman JT, Pitelka DR. Maintenance and induction of morphological differentiation in dissociated mammary epithelium on floating collagen membranes. In Vitro. 1977;13(5):316–328. doi: 10.1007/BF02616178. [DOI] [PubMed] [Google Scholar]
- 39.Dhimolea E, Maffini MV, Soto AM, Sonnenschein C. The role of collagen reorganization on mammary epithelial morphogenesis in a 3D culture model. Biomaterials. 2010;31(13):3622–3630. doi: 10.1016/j.biomaterials.2010.01.077. [DOI] [PubMed] [Google Scholar]
- 40.Provenzano PP, Inman DR, Eliceiri KW, Keely PJ. Matrix density-induced mechanoregulation of breast cell phenotype, signaling and gene expression through a FAK-ERK linkage. Oncogene. 2009;28(49):4326–4343. doi: 10.1038/onc.2009.299. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Alcaraz J, Xu R, Mori H, Nelson CM, Mroue R, Spencer VA, Brownfield D, Radisky DC, Bustamante C, Bissell MJ. Laminin and biomimetic extracellular elasticity enhance functional differentiation in mammary epithelia. EMBO J. 2008;27(21):2829–2838. doi: 10.1038/emboj.2008.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Gudjonsson T, Ronnov-Jessen L, Villadsen R, Rank F, Bissell MJ, Petersen OW. Normal and tumor-derived myoepithelial cells differ in their ability to interact with luminal breast epithelial cells for polarity and basement membrane deposition. J Cell Sci. 2002;115(Pt 1):39–50. doi: 10.1242/jcs.115.1.39. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 43.Xu R, Spencer VA, Bissell MJ. Extracellular matrix-regulated gene expression requires cooperation of SWI/SNF and transcription factors. J Biol Chem. 2007;282(20):14992–14999. doi: 10.1074/jbc.M610316200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 44.Even-Ram S, Yamada KM. Cell migration in 3D matrix. Curr Opin Cell Biol. 2005;17(5):524–532. doi: 10.1016/j.ceb.2005.08.015. [DOI] [PubMed] [Google Scholar]
- 45.Kalluri R, Zeisberg M. Fibroblasts in cancer. Nat Rev Cancer. 2006;6(5):392–401. doi: 10.1038/nrc1877. [DOI] [PubMed] [Google Scholar]
- 46.Singer CF, Gschwantler-Kaulich D, Fink-Retter A, Haas C, Hudelist G, Czerwenka K, Kubista E. Differential gene expression profile in breast cancer- derived stromal fibroblasts. Breast Cancer Res Treat. 2008;110(2):273–281. doi: 10.1007/s10549-007-9725-2. [DOI] [PubMed] [Google Scholar]
- 47.Mintz B, Illmensee K. Normal genetically mosaic mice produced from malignant teratocarcinoma cells. Proc Natl Acad Sci U S A. 1975;72(9):3585–3589. doi: 10.1073/pnas.72.9.3585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 48.Tuxhorn JA, Ayala GE, Rowley DR. Reactive stroma in prostate cancer progression. J Urol. 2001;166(6):2472–2483. [PubMed] [Google Scholar]
- 49.Liotta LA, Kohn EC. The microenvironment of the tumour-host interface. Nature. 2001;411(6835):375–379. doi: 10.1038/35077241. [DOI] [PubMed] [Google Scholar]
- 50.De Wever O, Mareel M. Role of tissue stroma in cancer cell invasion. J Pathol. 2003;200(4):429–447. doi: 10.1002/path.1398. [DOI] [PubMed] [Google Scholar]
- 51.Hsu MY, Meier F, Herlyn M. Melanoma development and progression: a conspiracy between tumor and host. Differentiation. 2002;70(9–10):522–536. doi: 10.1046/j.1432-0436.2002.700906.x. [DOI] [PubMed] [Google Scholar]
- 52.Kunz-Schughart LA, Knuechel R. Tumor-associated fibroblasts (part I): Active stromal participants in tumor development and progression? Histol Histopathol. 2002;17(2):599–621. doi: 10.14670/HH-17.599. [DOI] [PubMed] [Google Scholar]
- 53.Pankov R, Endo Y, Even-Ram S, Araki M, Clark K, Cukierman E, Matsumoto K, Yamada KM. A Rac switch regulates random versus directionally persistent cell migration. J Cell Biol. 2005;170(5):793–802. doi: 10.1083/jcb.200503152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 54.Paulitschke V, Kunstfeld R, Mohr T, Slany A, Micksche M, Drach J, Zielinski C, Pehamberger H, Gerner C. Entering a new era of rational biomarker discovery for early detection of melanoma metastases: secretome analysis of associated stroma cells. J Proteome Res. 2009;8(5):2501–2510. doi: 10.1021/pr8010827. [DOI] [PubMed] [Google Scholar]
- 55.Sund M, Kalluri R. Tumor stroma derived biomarkers in cancer. Cancer Metastasis Rev. 2009;28(1–2):177–183. doi: 10.1007/s10555-008-9175-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 56.Mao Y, Schwarzbauer JE. Fibronectin fibrillogenesis, a cell-mediated matrix assembly process. Matrix Biology. 2005;24(6):389–399. doi: 10.1016/j.matbio.2005.06.008. [DOI] [PubMed] [Google Scholar]
- 57.Kubow KE, Klotzsch E, Smith ML, Gourdon D, Little WC, Vogel V. Crosslinking of cell-derived 3D scaffolds up-regulates the stretching and unfolding of new extracellular matrix assembled by reseeded cells. Integr Biol (Camb) 2009;1(11–12):635–648. doi: 10.1039/b914996a. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 58.Damianova R, Stefanova N, Cukierman E, Momchilova A, Pankov R. Three-dimensional matrix induces sustained activation of ERK1/2 via Src/Ras/Raf signaling pathway. Cell Biol Int. 2008;32(2):229–234. doi: 10.1016/j.cellbi.2007.08.029. [DOI] [PubMed] [Google Scholar]
- 59.Pankov R, Cukierman E, Clark K, Matsumoto K, Hahn C, Poulin B, Yamada KM. Specific beta 1 Integrin Site Selectively Regulates Akt/Protein Kinase B Signaling via Local Activation of Protein Phosphatase 2A. J Biol Chem. 2003;278(20):18671–18681. doi: 10.1074/jbc.M300879200. [DOI] [PubMed] [Google Scholar]
- 60.Castelló-Cros R, Khan DR, Simons J, Valianou M, Cukierman E. Staged stromal extracellular 3D matrices differentially regulate breast cancer cell responses through PI3K and beta1-integrins. BMC Cancer. 2009;9(1):94. doi: 10.1186/1471-2407-9-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 61.Petrie RJ, Doyle AD, Yamada KM. Random versus directionally persistent cell migration. Nat Rev Mol Cell Biol. 2009;10(8):538–549. doi: 10.1038/nrm2729. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 62.Engler AJ, Chan M, Boettiger D, Schwarzbauer JE. A novel mode of cell detachment from fibrillar fibronectin matrix under shear. J Cell Sci. 2009:jcs.040824. doi: 10.1242/jcs.040824. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 63.Serebriiskii I, Castello-Cros R, Lamb A, Golemis EA, Cukierman E. Fibroblast-derived 3D matrix differentially regulates the growth and drug-responsiveness of human cancer cells. Matrix Biology. 2008;27(6):573–585. doi: 10.1016/j.matbio.2008.02.008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 64.Chen CZ, Peng YX, Wang ZB, Fish PV, Kaar JL, Koepsel RR, Russell AJ, Lareu RR, Raghunath M. The Scar-in-a-Jar: studying potential antifibrotic compounds from the epigenetic to extracellular level in a single well. Br J Pharmacol. 2009 doi: 10.1111/j.1476-5381.2009.00387.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 65.Van Dyke T, Jacks T. Cancer modeling in the modern era: progress and challenges. Cell. 2002;108(2):135–144. doi: 10.1016/s0092-8674(02)00621-9. [DOI] [PubMed] [Google Scholar]
- 66.Felsher DW. Oncogene addiction versus oncogene amnesia: perhaps more than just a bad habit? Cancer Res. 2008;68(9):3081–3086. doi: 10.1158/0008-5472.CAN-07-5832. discussion 3086. [DOI] [PubMed] [Google Scholar]
- 67.LaBarge MA, Nelson CM, Villadsen R, Fridriksdottir A, Ruth JR, Stampfer MR, Petersen OW, Bissell MJ. Human mammary progenitor cell fate decisions are products of interactions with combinatorial microenvironments. Integr Biol (Camb) 2009;1(1):70–79. doi: 10.1039/b816472j. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 68.Weigelt B, Bissell MJ. Unraveling the microenvironmental influences on the normal mammary gland and breast cancer. Semin Cancer Biol. 2008;18 (5):311–321. doi: 10.1016/j.semcancer.2008.03.013. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 69.Petersen OW, Ronnov-Jessen L, Howlett AR, Bissell MJ. Interaction with basement membrane serves to rapidly distinguish growth and differentiation pattern of normal and malignant human breast epithelial cells. Proc Natl Acad Sci U S A. 1992;89(19):9064–9068. doi: 10.1073/pnas.89.19.9064. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 70.Larsen M, Artym VV, Green JA, Yamada KM. The matrix reorganized: extracellular matrix remodeling and integrin signaling. Current Opinion in Cell Biology. 2006;18(5):463–471. doi: 10.1016/j.ceb.2006.08.009. [DOI] [PubMed] [Google Scholar]
- 71.Freytes DO, Wan LQ, Vunjak-Novakovic G. Geometry and force control of cell function. J Cell Biochem. 2009;108(5):1047–1058. doi: 10.1002/jcb.22355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 72.Spencer VA, Xu R, Bissell MJ. Gene expression in the third dimension: the ECM-nucleus connection. J Mammary Gland Biol Neoplasia. 2010;15(1):65–71. doi: 10.1007/s10911-010-9163-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 73.Ochsner M, Textor M, Vogel V, Smith ML. Dimensionality Controls Cytoskeleton Assembly and Metabolism of Fibroblast Cells in Response to Rigidity and Shape. PLoS ONE. 2010;5(3):e9445. doi: 10.1371/journal.pone.0009445. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 74.Wipff P-J, Rifkin DB, Meister J-J, Hinz B. Myofibroblast contraction activates latent TGF-beta1 from the extracellular matrix. J Cell Biol. 2007;179(6):1311–1323. doi: 10.1083/jcb.200704042. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 75.Pfeiffer BJ, Franklin CL, Hsieh FH, Bank RA, Phillips CL. Alpha 2(I) collagen deficient oim mice have altered biomechanical integrity, collagen content, and collagen crosslinking of their thoracic aorta. Matrix Biol. 2005;24(7):451–458. doi: 10.1016/j.matbio.2005.07.001. [DOI] [PubMed] [Google Scholar]
- 76.Paszek MJ, Zahir N, Johnson KR, Lakins JN, Rozenberg GI, Gefen A, Reinhart-King CA, Margulies SS, Dembo M, Boettiger D, et al. Tensional homeostasis and the malignant phenotype. Cancer Cell. 2005;8(3):241–254. doi: 10.1016/j.ccr.2005.08.010. [DOI] [PubMed] [Google Scholar]
- 77.O’Brien LE, Jou TS, Pollack AL, Zhang Q, Hansen SH, Yurchenco P, Mostov KE. Rac1 orientates epithelial apical polarity through effects on basolateral laminin assembly. Nat Cell Biol. 2001;3(9):831–838. doi: 10.1038/ncb0901-831. [DOI] [PubMed] [Google Scholar]
- 78.Wang F, Weaver VM, Petersen OW, Larabell CA, Dedhar S, Briand P, Lupu R, Bissell MJ. Reciprocal interactions between beta1-integrin and epidermal growth factor receptor in three-dimensional basement membrane breast cultures: a different perspective in epithelial biology. Proc Natl Acad Sci U S A. 1998;95(25):14821–14826. doi: 10.1073/pnas.95.25.14821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 79.Lauffenburger DA, Horwitz AF. Cell migration: a physically integrated molecular process. Cell. 1996;84(3):359–369. doi: 10.1016/s0092-8674(00)81280-5. [DOI] [PubMed] [Google Scholar]
- 80.Wolf K, Friedl P. Mapping proteolytic cancer cell-extracellular matrix interfaces. Clin Exp Metastasis. 2009;26(4):289–298. doi: 10.1007/s10585-008-9190-2. [DOI] [PubMed] [Google Scholar]
- 81.Oser M, Condeelis J. The cofilin activity cycle in lamellipodia and invadopodia. J Cell Biochem. 2009;108(6):1252–1262. doi: 10.1002/jcb.22372. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Machesky LM. Lamellipodia and filopodia in metastasis and invasion. FEBS Lett. 2008;582(14):2102–2111. doi: 10.1016/j.febslet.2008.03.039. [DOI] [PubMed] [Google Scholar]
- 83.Matsumoto K, Ziober BL, Yao CC, Kramer RH. Growth factor regulation of integrin-mediated cell motility. Cancer Metastasis Rev. 1995;14(3):205–217. doi: 10.1007/BF00690292. [DOI] [PubMed] [Google Scholar]
- 84.Sabeh F, Ota I, Holmbeck K, Birkedal-Hansen H, Soloway P, Balbin M, Lopez-Otin C, Shapiro S, Inada M, Krane S, et al. Tumor cell traffic through the extracellular matrix is controlled by the membrane-anchored collagenase MT1-MMP. J Cell Biol. 2004;167(4):769–781. doi: 10.1083/jcb.200408028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 85.Sabeh F, Ota I, Holmbeck K, Birkedal-Hansen H, Soloway P, Balbin M, Lopez-Otin C, Shapiro S, Inada M, Krane S, et al. Tumor cell traffic through the extracellular matrix is controlled by the membrane-anchored collagenase MT1-MMP. J Cell Biol. 2004;167(4):769–781. doi: 10.1083/jcb.200408028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 86.Takino T, Saeki H, Miyamori H, Kudo T, Sato H. Inhibition of membrane-type 1 matrix metalloproteinase at cell-matrix adhesions. Cancer Res. 2007;67 (24):11621–11629. doi: 10.1158/0008-5472.CAN-07-5251. [DOI] [PubMed] [Google Scholar]
- 87.Galvez BG, Matias-Roman S, Yanez-Mo M, Sanchez-Madrid F, Arroyo AG. ECM regulates MT1-MMP localization with beta1 or alphavbeta3 integrins at distinct cell compartments modulating its internalization and activity on human endothelial cells. J Cell Biol. 2002;159(3):509–521. doi: 10.1083/jcb.200205026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 88.Sahai E, Wyckoff J, Philippar U, Segall JE, Gertler F, Condeelis J. Simultaneous imaging of GFP, CFP and collagen in tumors in vivo using multiphoton microscopy. BMC Biotechnol. 2005;5:14. doi: 10.1186/1472-6750-5-14. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 89.Sidani M, Wyckoff J, Xue C, Segall JE, Condeelis J. Probing the microenvironment of mammary tumors using multiphoton microscopy. J Mammary Gland Biol Neoplasia. 2006;11(2):151–163. doi: 10.1007/s10911-006-9021-5. [DOI] [PubMed] [Google Scholar]
- 90.Parekh A, Weaver AM. Regulation of cancer invasiveness by the physical extracellular matrix environment. Cell Adh Migr. 2009;3(3):288–292. doi: 10.4161/cam.3.3.8888. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 91.Engler AJ, Sen S, Sweeney HL, Discher DE. Matrix elasticity directs stem cell lineage specification. Cell. 2006;126(4):677–689. doi: 10.1016/j.cell.2006.06.044. [DOI] [PubMed] [Google Scholar]
- 92.Friedl P, Wolf K. Plasticity of cell migration: a multiscale tuning model. J Cell Biol. 2010;188(1):11–19. doi: 10.1083/jcb.200909003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Ulrich TA, de Juan Pardo EM, Kumar S. The Mechanical Rigidity of the Extracellular Matrix Regulates the Structure, Motility, and Proliferation of Glioma Cells. Cancer Res. 2009;69(10):4167–4174. doi: 10.1158/0008-5472.CAN-08-4859. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 94.Peyton SR, Kim PD, Ghajar CM, Seliktar D, Putnam AJ. The effects of matrix stiffness and RhoA on the phenotypic plasticity of smooth muscle cells in a 3-D biosynthetic hydrogel system. Biomaterials. 2008;29(17):2597–2607. doi: 10.1016/j.biomaterials.2008.02.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 95.Isenberg BC, Dimilla PA, Walker M, Kim S, Wong JY. Vascular smooth muscle cell durotaxis depends on substrate stiffness gradient strength. Biophys J. 2009;97(5):1313–1322. doi: 10.1016/j.bpj.2009.06.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 96.Wolf K, Wu YI, Liu Y, Geiger J, Tam E, Overall C, Stack MS, Friedl P. Multistep pericellular proteolysis controls the transition from individual to collective cancer cell invasion. Nat Cell Biol. 2007;9(8):893–904. doi: 10.1038/ncb1616. [DOI] [PubMed] [Google Scholar]
- 97.Carragher NO, Levkau B, Ross R, Raines EW. Degraded collagen fragments promote rapid disassembly of smooth muscle focal adhesions that correlates with cleavage of pp125(FAK), paxillin, and talin. J Cell Biol. 1999;147(3):619–630. doi: 10.1083/jcb.147.3.619. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 98.Franco SJ, Huttenlocher A. Regulating cell migration: calpains make the cut. J Cell Sci. 2005;118(Pt 17):3829–3838. doi: 10.1242/jcs.02562. [DOI] [PubMed] [Google Scholar]
- 99.Quaranta V, Giannelli G. Cancer invasion: watch your neighbourhood! Tumori. 2003;89(4):343–348. doi: 10.1177/030089160308900401. [DOI] [PubMed] [Google Scholar]
- 100.Nabeshima K, Kataoka H, Koono M. Enhanced migration of tumor cells in response to collagen degradation products and tumor cell collagenolytic activity. Invasion Metastasis. 1986;6(5):270–286. [PubMed] [Google Scholar]
- 101.Rhee S, Ho C-H, Grinnell F. Promigratory and procontractile growth factor environments differentially regulate cell morphogenesis. Experimental Cell Research. 2010;316(2):232–244. doi: 10.1016/j.yexcr.2009.09.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Miron-Mendoza M, Seemann J, Grinnell F. Collagen fibril flow and tissue translocation coupled to fibroblast migration in 3D collagen matrices. Mol Biol Cell. 2008;19(5):2051–2058. doi: 10.1091/mbc.E07-09-0930. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 103.Baker EL, Bonnecaze RT, Zaman MH. Extracellular matrix stiffness and architecture govern intracellular rheology in cancer. Biophys J. 2009;97 (4):1013–1021. doi: 10.1016/j.bpj.2009.05.054. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 104.Baker EL, Zaman MH. The biomechanical integrin. J Biomech. 2009 doi: 10.1016/j.jbiomech.2009.09.007. in press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 105.Zaman MH, Trapani LM, Siemeski A, Mackellar D, Gong H, Kamm RD, Wells A, Lauffenburger DA, Matsudaira P. Migration of tumor cells in 3D matrices is governed by matrix stiffness along with cell-matrix adhesion and proteolysis. Proc Natl Acad Sci U S A. 2006;103(29):10889–10894. doi: 10.1073/pnas.0604460103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 106.Baneyx G, Baugh L, Vogel V. Fibronectin extension and unfolding within cell matrix fibrils controlled by cytoskeletal tension. Proc Natl Acad Sci U S A. 2002;99(8):5139–5143. doi: 10.1073/pnas.072650799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 107.Ohashi T, Kiehart DP, Erickson HP. Dynamics and elasticity of the fibronectin matrix in living cell culture visualized by fibronectin-green fluorescent protein. Proc Natl Acad Sci U S A. 1999;96(5):2153–2158. doi: 10.1073/pnas.96.5.2153. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 108.Zhong C, Chrzanowska-Wodnicka M, Brown J, Shaub A, Belkin AM, Burridge K. Rho-mediated contractility exposes a cryptic site in fibronectin and induces fibronectin matrix assembly. J Cell Biol. 1998;141(2):539–551. doi: 10.1083/jcb.141.2.539. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 109.Erickson HP. Reversible unfolding of fibronectin type III and immunoglobulin domains provides the structural basis for stretch and elasticity of titin and fibronectin. Proc Natl Acad Sci U S A. 1994;91 (21):10114–10118. doi: 10.1073/pnas.91.21.10114. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 110.Little WC, Smith ML, Ebneter U, Vogel V. Assay to mechanically tune and optically probe fibrillar fibronectin conformations from fully relaxed to breakage. Matrix Biol. 2008;27(5):451–461. doi: 10.1016/j.matbio.2008.02.003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Kilian KA, Bugarija B, Lahn BT, Mrksich M. Geometric cues for directing the differentiation of mesenchymal stem cells. Proc Natl Acad Sci U S A. 2010;107(11):4872–4877. doi: 10.1073/pnas.0903269107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 112.Hartmann C. A Wnt canon orchestrating osteoblastogenesis. Trends Cell Biol. 2006;16(3):151–158. doi: 10.1016/j.tcb.2006.01.001. [DOI] [PubMed] [Google Scholar]
- 113.Nelson CM, Jean RP, Tan JL, Liu WF, Sniadecki NJ, Spector AA, Chen CS. Emergent patterns of growth controlled by multicellular form and mechanics. Proc Natl Acad Sci U S A. 2005;102(33):11594–11599. doi: 10.1073/pnas.0502575102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 114.Sniadecki NJ, Anguelouch A, Yang MT, Lamb CM, Liu Z, Kirschner SB, Liu Y, Reich DH, Chen CS. Magnetic microposts as an approach to apply forces to living cells. Proc Natl Acad Sci U S A. 2007;104(37):14553–14558. doi: 10.1073/pnas.0611613104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 115.James J, Goluch ED, Hu H, Liu C, Mrksich M. Subcellular curvature at the perimeter of micropatterned cells influences lamellipodial distribution and cell polarity. Cell Motil Cytoskeleton. 2008;65(11):841–852. doi: 10.1002/cm.20305. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 116.Van Goethem E, Poincloux R, Gauffre F, Maridonneau-Parini I, Le Cabec V. Matrix Architecture Dictates Three-Dimensional Migration Modes of Human Macrophages: Differential Involvement of Proteases and Podosome-Like Structures. J Immunol. 2010;184(2):1049–1061. doi: 10.4049/jimmunol.0902223. [DOI] [PubMed] [Google Scholar]
- 117.Doyle AD, Wang FW, Matsumoto K, Yamada KM. One-dimensional topography underlies three-dimensional fibrillar cell migration. J Cell Biol. 2009;184(4):481–490. doi: 10.1083/jcb.200810041. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 118.Kong HJ, Mooney DJ. Microenvironmental regulation of biomacromolecular therapies. Nat Rev Drug Discov. 2007;6(6):455–463. doi: 10.1038/nrd2309. [DOI] [PubMed] [Google Scholar]
- 119.Netti PA, Berk DA, Swartz MA, Grodzinsky AJ, Jain RK. Role of extracellular matrix assembly in interstitial transport in solid tumors. Cancer Res. 2000;60 (9):2497–2503. [PubMed] [Google Scholar]
- 120.Kong HJ, Liu J, Riddle K, Matsumoto T, Leach K, Mooney DJ. Non-viral gene delivery regulated by stiffness of cell adhesion substrates. Nat Mater. 2005;4(6):460–464. doi: 10.1038/nmat1392. [DOI] [PubMed] [Google Scholar]
- 121.Erler JT, Linding R. Network-based drugs and biomarkers. J Pathol. 2010;220(2):290–296. doi: 10.1002/path.2646. [DOI] [PubMed] [Google Scholar]