Abstract
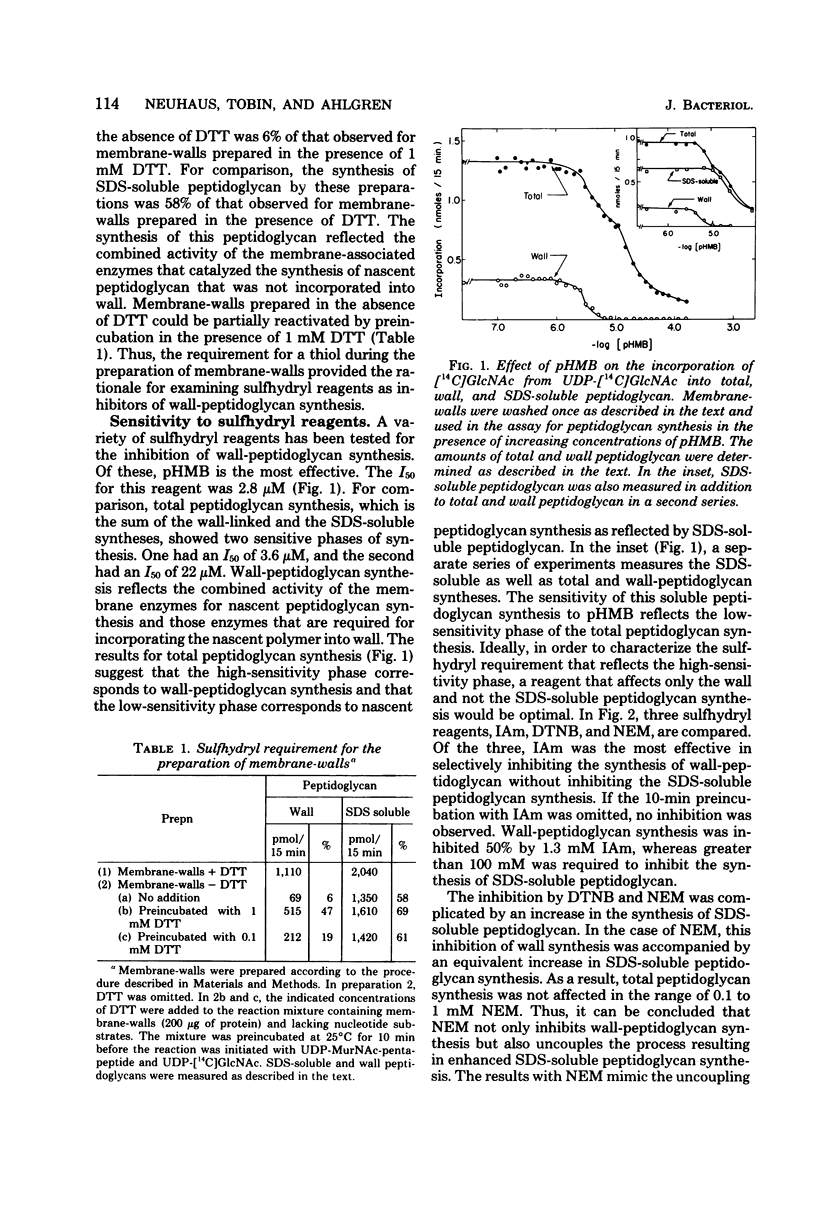
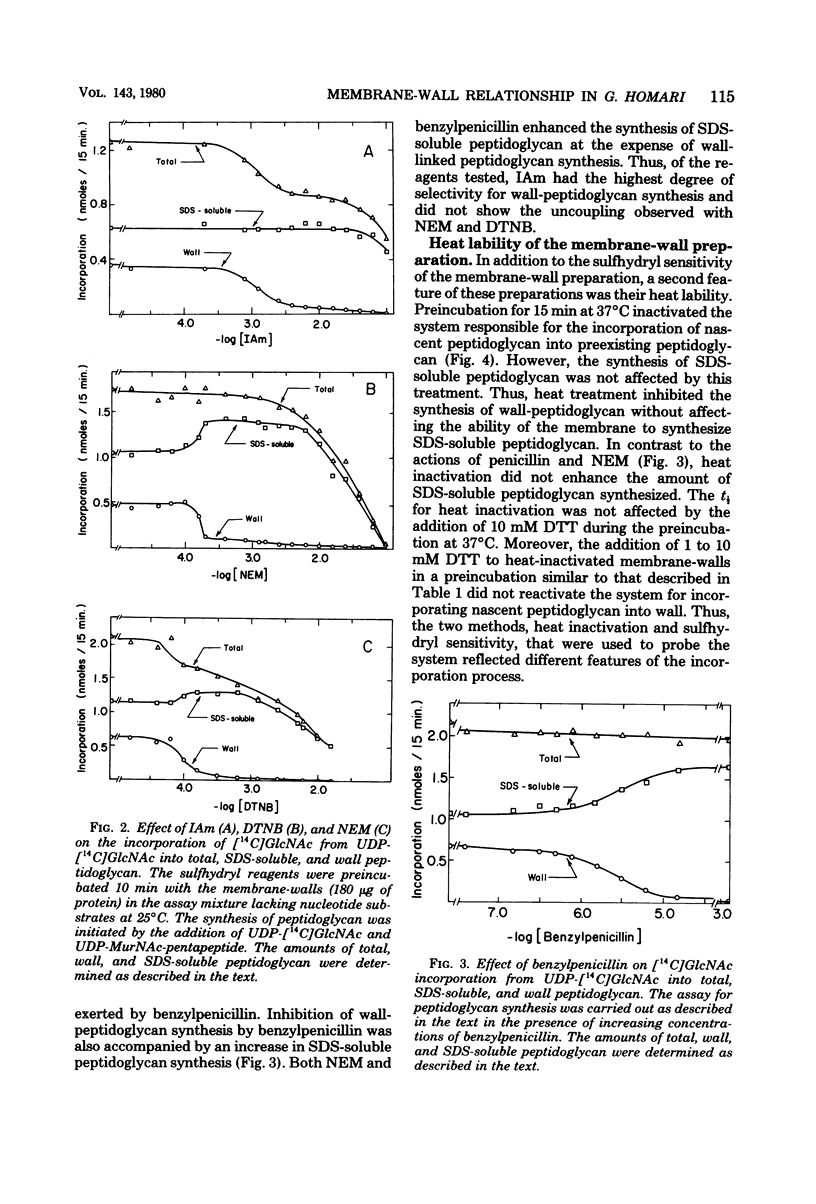
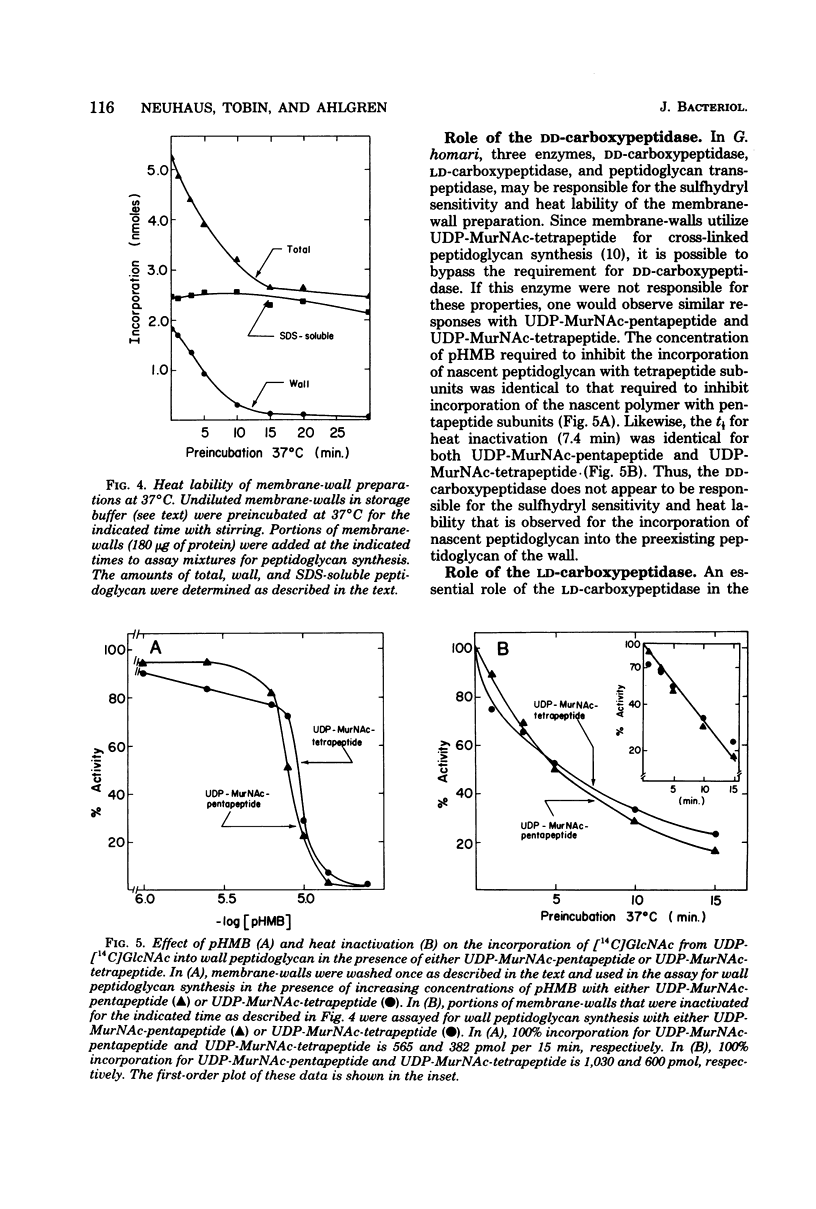
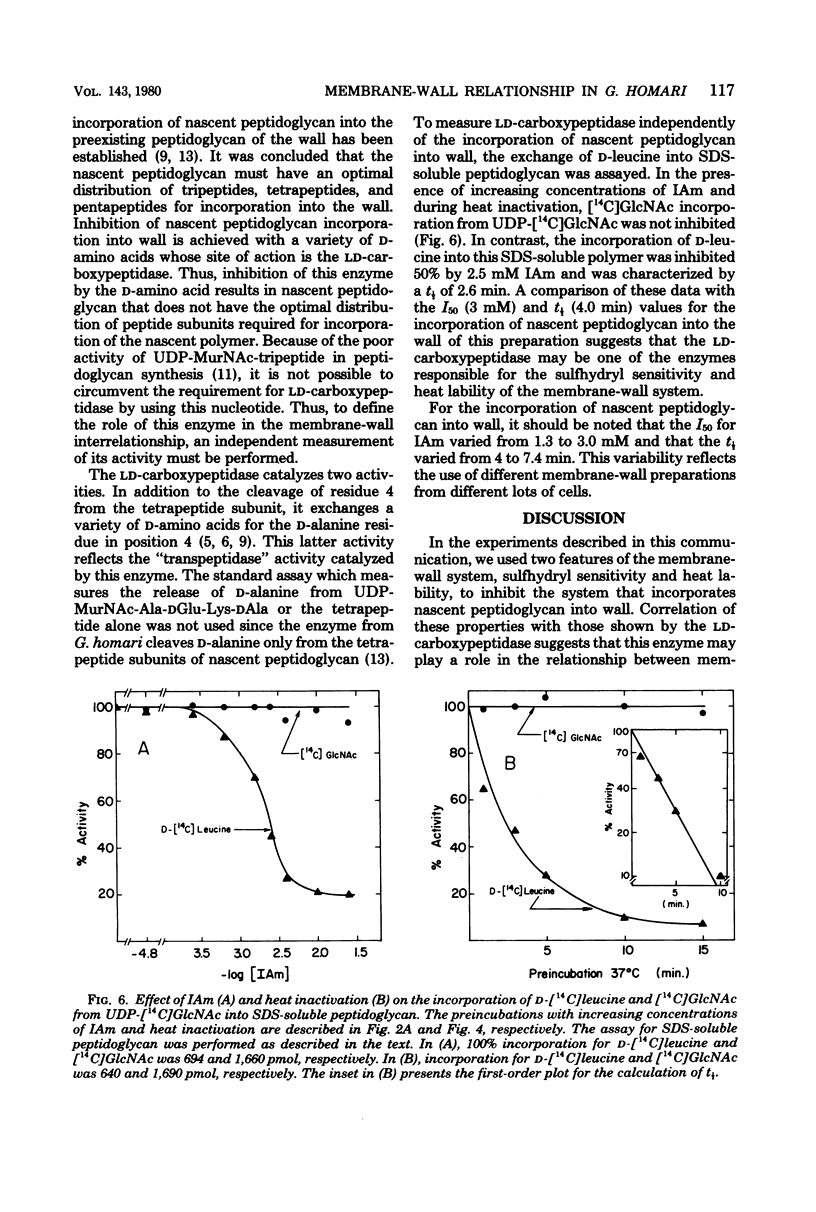
Membrane-walls from Gaffkya homari require a specific interrelationship between membrane and wall that functions in the incorporation of nascent peptidoglycan into the preexisting peptidoglycan of the wall. Two different methods were used to inhibit selectively this incorporation process: (i) sensitivity to sulfhydryl reagents and (ii) heat inactivation. Of the sulfhydryl reagents tested, 2.2 mM iodoacetamide inhibited the synthesis of wall peptidoglycan 50%, whereas greater than 100 mM was required to inhibit the synthesis of sodium dodecyl sulfate (SDS)-soluble peptidoglycan. Heat treatment at 37 degrees C (t 1/2 = 5.7 min) inhibited wall peptidoglycan synthesis without affecting SDS-soluble peptidoglycan synthesis. Inhibition of LD-carboxypeptidase by iodoacetamide and heat gave 50% inhibition and t 1/2 values similar to those observed for the incorporation process. Thus, it is suggested that the LD-carboxypeptidase may be one of the enzymes responsible for the sulfhydryl sensitivity and heat lability and that this enzyme may play a role in the relationship between membrane and wall in G. homari.
Full text
PDF



Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Bayer M. E. Areas of adhesion between wall and membrane of Escherichia coli. J Gen Microbiol. 1968 Oct;53(3):395–404. doi: 10.1099/00221287-53-3-395. [DOI] [PubMed] [Google Scholar]
- Beck B. D., Park J. T. Activity of three murein hydrolases during the cell division cycle of Escherichia coli K-12 as measured in toluene-treated cells. J Bacteriol. 1976 Jun;126(3):1250–1260. doi: 10.1128/jb.126.3.1250-1260.1976. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Carpenter C. V., Goyer S., Neuhaus F. C. Steric effects on penicillin-sensitive peptidoglycan synthesis in a membrane-wall system Gaffkya homari. Biochemistry. 1976 Jul 13;15(14):3146–3152. doi: 10.1021/bi00659a031. [DOI] [PubMed] [Google Scholar]
- Coyette J., Perkins H. R., Polacheck I., Shockman G. D., Ghuysen J. M. Membrane-bound DD-carboxypeptidase and LD-transpeptidase of Streptococcus faecalis ATCC 9790. Eur J Biochem. 1974 May 15;44(2):459–468. doi: 10.1111/j.1432-1033.1974.tb03504.x. [DOI] [PubMed] [Google Scholar]
- DasGupta H., Fan D. P. Purification and characterization of a carboxypeptidase-transpeptidase of Bacillus megaterium acting on the tetrapeptide moiety of the peptidoglycan. J Biol Chem. 1979 Jul 10;254(13):5672–5683. [PubMed] [Google Scholar]
- Giesbrecht P., Wecke J., Reinicke B. The demonstration of the existence of an interlayer between the cytoplasmic membrane and the cell wall proper of staphylococci. Arch Microbiol. 1977 Oct 24;115(1):25–35. doi: 10.1007/BF00427841. [DOI] [PubMed] [Google Scholar]
- Hammes W. P. Biosynthesis of peptidoglycan in Gaffkya homari. The mode of action of penicillin G and mecillinam. Eur J Biochem. 1976 Nov 1;70(1):107–113. doi: 10.1111/j.1432-1033.1976.tb10961.x. [DOI] [PubMed] [Google Scholar]
- Hammes W. P., Kandler O. Biosynthesis of peptidoglycan in Gaffkya homari. The incorporation of peptidoglycan into the cell wall and the direction of transpeptidation. Eur J Biochem. 1976 Nov 1;70(1):97–106. doi: 10.1111/j.1432-1033.1976.tb10960.x. [DOI] [PubMed] [Google Scholar]
- Hammes W. P., Neuhaus F. C. Biosynthesis of peptidoglycan in Gaffkya homari: role of the peptide subunit of uridine diphosphate-N-acetylmuramyl-pentapeptide. J Bacteriol. 1974 Oct;120(1):210–218. doi: 10.1128/jb.120.1.210-218.1974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hammes W. P., Neuhaus F. C. On the specificity of phospho-N-acetylmuramyl-pentapeptide translocase. The peptide subunit of uridine diphosphate-N-actylmuramyl-pentapeptide. J Biol Chem. 1974 May 25;249(10):3140–3150. [PubMed] [Google Scholar]
- Hammes W. P., Seidel H. The LD-carboxypeptidase activity in Gaffkya homari. The target of the action of certain beta-lactam antibiotics on the formation of wall-bound peptidoglycan. Eur J Biochem. 1978 Nov 15;91(2):509–515. doi: 10.1111/j.1432-1033.1978.tb12704.x. [DOI] [PubMed] [Google Scholar]
- Hammes W. P., Seidel H. The activities in vitro of DD-carboxypeptidase and LD-carboxypeptidase of Gaffkya homari during biosynthesis of peptidoglycan. Eur J Biochem. 1978 Mar;84(1):141–147. doi: 10.1111/j.1432-1033.1978.tb12150.x. [DOI] [PubMed] [Google Scholar]
- Hammes W. P. The LD-carboxypeptidase activity in Gaffkya homari. The target of the action of D-amino acids or glycine on the formation of wall-bound peptidoglycan. Eur J Biochem. 1978 Nov 15;91(2):501–507. doi: 10.1111/j.1432-1033.1978.tb12703.x. [DOI] [PubMed] [Google Scholar]
- Hungerer K. D., Tipper D. J. Cell wall polymers of Bacillus sphaericus 9602. I. Structure of the vegetative cell wall peptidoglycan. Biochemistry. 1969 Sep;8(9):3577–3587. doi: 10.1021/bi00837a013. [DOI] [PubMed] [Google Scholar]
- Hurst A., Stubbs J. M. Electron microscopic study of membranes and walls of bacteria and changes occurring during growth initiation. J Bacteriol. 1969 Mar;97(3):1466–1479. doi: 10.1128/jb.97.3.1466-1479.1969. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kato K., Strominger J. L., Kotani S. Structure of the cell wall of Corynebacterium diphtheriae. I. Mechanism of hydrolysis by the L-3 enzyme and the structure of the peptide. Biochemistry. 1968 Aug;7(8):2762–2773. doi: 10.1021/bi00848a010. [DOI] [PubMed] [Google Scholar]
- Kelly K. F., Evans J. B. Deoxyribonucleic acid homology among strains of the lobster pathogen 'Gaffkya homari' and Aerococcus viridans. J Gen Microbiol. 1974 Mar;81(1):257–260. doi: 10.1099/00221287-81-1-257. [DOI] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Mirelman D., Bracha R., Sharon N. Role of the penicillin-sensitive transpeptidation reaction in attachment of newly synthesized peptidoglycan to cell walls of Micrococcus luteus. Proc Natl Acad Sci U S A. 1972 Nov;69(11):3355–3359. doi: 10.1073/pnas.69.11.3355. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mirelman D., Bracha R., Sharon N. Studies on the elongation of bacterial cell wall peptidoglycan and its inhibition by penicillin. Ann N Y Acad Sci. 1974 May 10;235(0):326–347. doi: 10.1111/j.1749-6632.1974.tb43275.x. [DOI] [PubMed] [Google Scholar]
- Mirelman D., Sharon N. Biosynthesis of peptidoglycan by a cell wall preparation of Staphylococcus aureus and its inhibition by penicillin. Biochem Biophys Res Commun. 1972 Mar 10;46(5):1909–1917. doi: 10.1016/0006-291x(72)90069-1. [DOI] [PubMed] [Google Scholar]
- Nakel M., Ghuysen J. M., Kandler O. Wall peptidoglycan in Aerococcus viridans strains 201 Evans and ATCC 11563 and in Gaffkya homari strain ATCC 10400. Biochemistry. 1971 May 25;10(11):2170–2175. doi: 10.1021/bi00787a033. [DOI] [PubMed] [Google Scholar]
- Oka T. Mode of action of penicillins in vivo and in vitro in Bacillus megaterium. Antimicrob Agents Chemother. 1976 Oct;10(4):579–591. doi: 10.1128/aac.10.4.579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- PATTERSON M. S., GREENE R. C. MEASUREMENT OF LOW ENERGY BETA-EMITTERS IN AQUEOUS SOLUTION BY LIQUID SCINTILLATION COUNTING OF EMULSIONS. Anal Chem. 1965 Jun;37:854–857. doi: 10.1021/ac60226a017. [DOI] [PubMed] [Google Scholar]
- PELZER H. MUCOPEPTIDHYDROLASEN IN ESCHERICHIA COLI B. I. NACHWEIS UND WIRKUNGSSPEZIFITAET. Z Naturforsch B. 1963 Nov;18:950–956. [PubMed] [Google Scholar]
- Petit J. F., Munoz E., Ghuysen J. M. Peptide cross-links in bacterial cell wall peptidoglycans studied with specific endopeptidases from Streptomyces albus G. Biochemistry. 1966 Aug;5(8):2764–2776. doi: 10.1021/bi00872a037. [DOI] [PubMed] [Google Scholar]
- Rogers H. J. Bacterial growth and the cell envelope. Bacteriol Rev. 1970 Jun;34(2):194–214. doi: 10.1128/br.34.2.194-214.1970. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Van Heijenoort J., Elbaz L., Dezélée P., Petit J. F., Bricas E., Ghuysen J. M. Structure of the meso-diaminopimelic acid containing peptidoglycans in Escherichia coli B and Bacillus megaterium KM. Biochemistry. 1969 Jan;8(1):207–213. doi: 10.1021/bi00829a030. [DOI] [PubMed] [Google Scholar]
- Ward J. B., Perkins H. R. The direction of glycan synthesis in a bacterial peptidoglycan. Biochem J. 1973 Dec;135(4):721–728. doi: 10.1042/bj1350721. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ward J. B. The synthesis of peptidoglycan in an autolysin-deficient mutant of Bacillus licheniformis N.C.T.C. 6346 and the effect of beta-lactam antibiotics, bacitracin and vancomycin. Biochem J. 1974 Jul;141(1):227–241. doi: 10.1042/bj1410227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wietzerbin J., Das B. C., Petit J. F., Lederer E., Leyh-Bouille M., Ghuysen J. M. Occurrence of D-alanyl-(D)-meso-diaminopimelic acid and meso-diaminopimelyl-meso-diaminopimelic acid interpeptide linkages in the peptidoglycan of Mycobacteria. Biochemistry. 1974 Aug 13;13(17):3471–3476. doi: 10.1021/bi00714a008. [DOI] [PubMed] [Google Scholar]



