Abstract
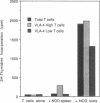
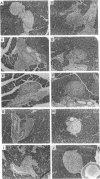
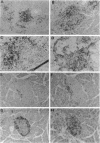
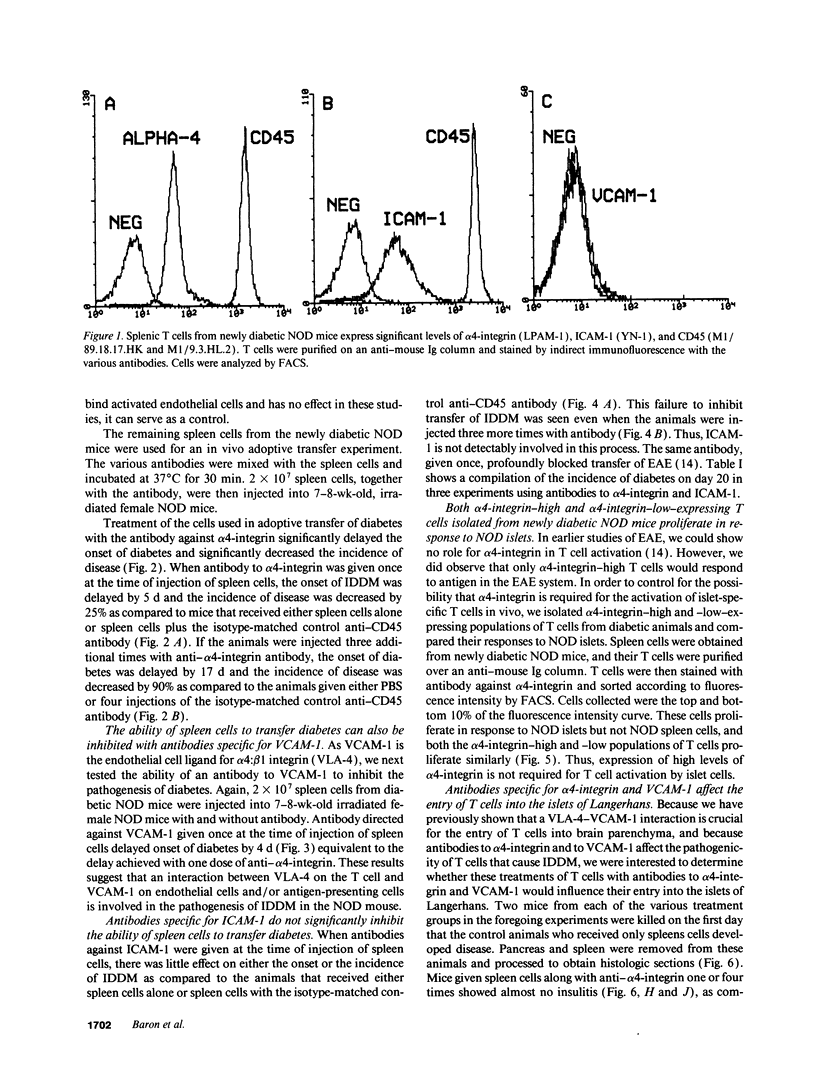
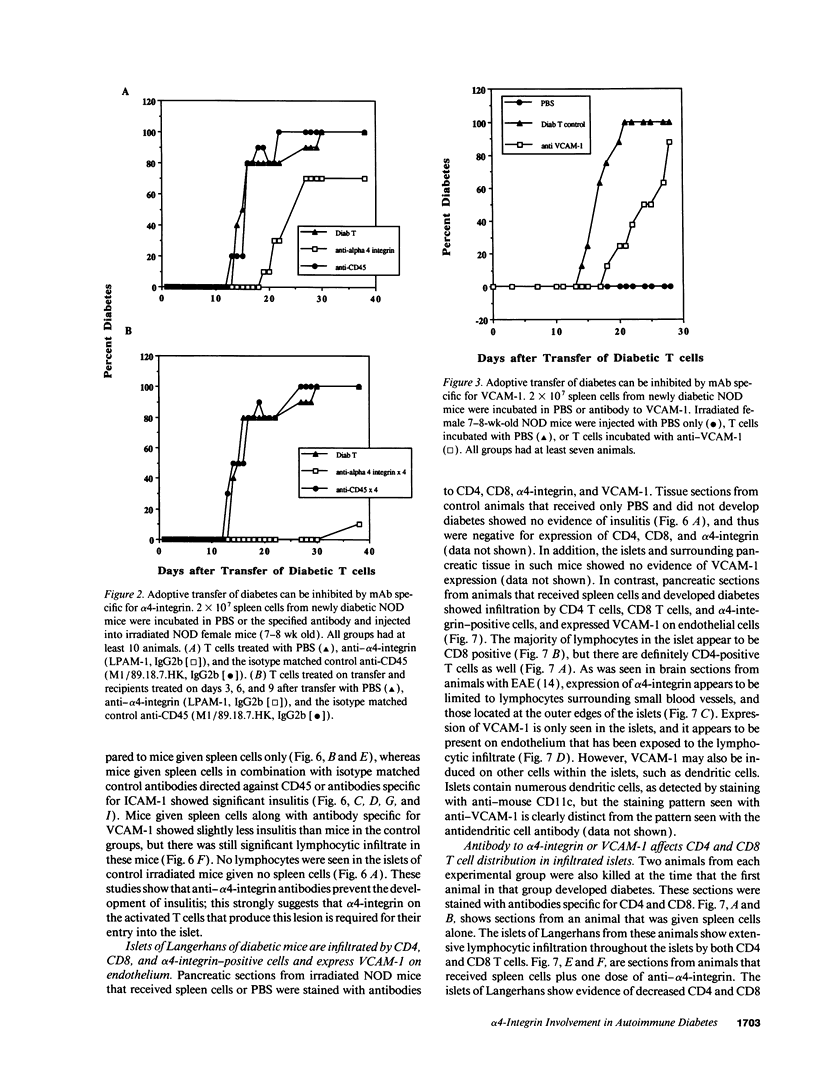
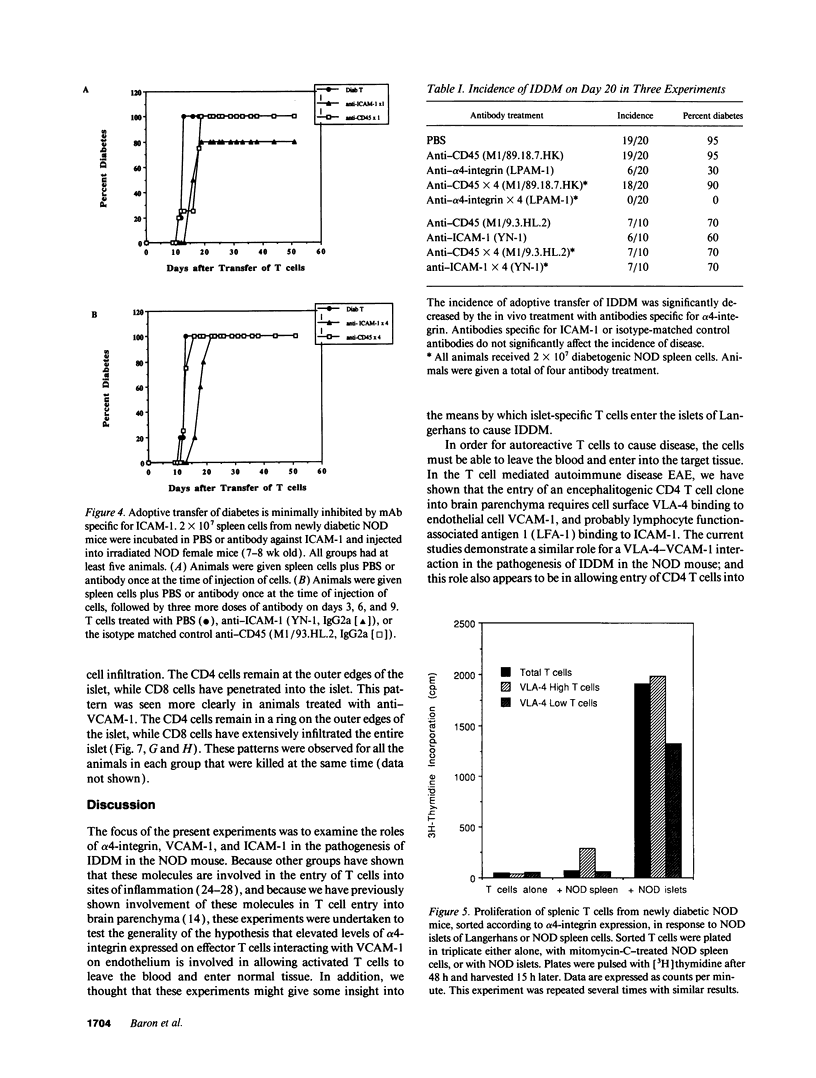
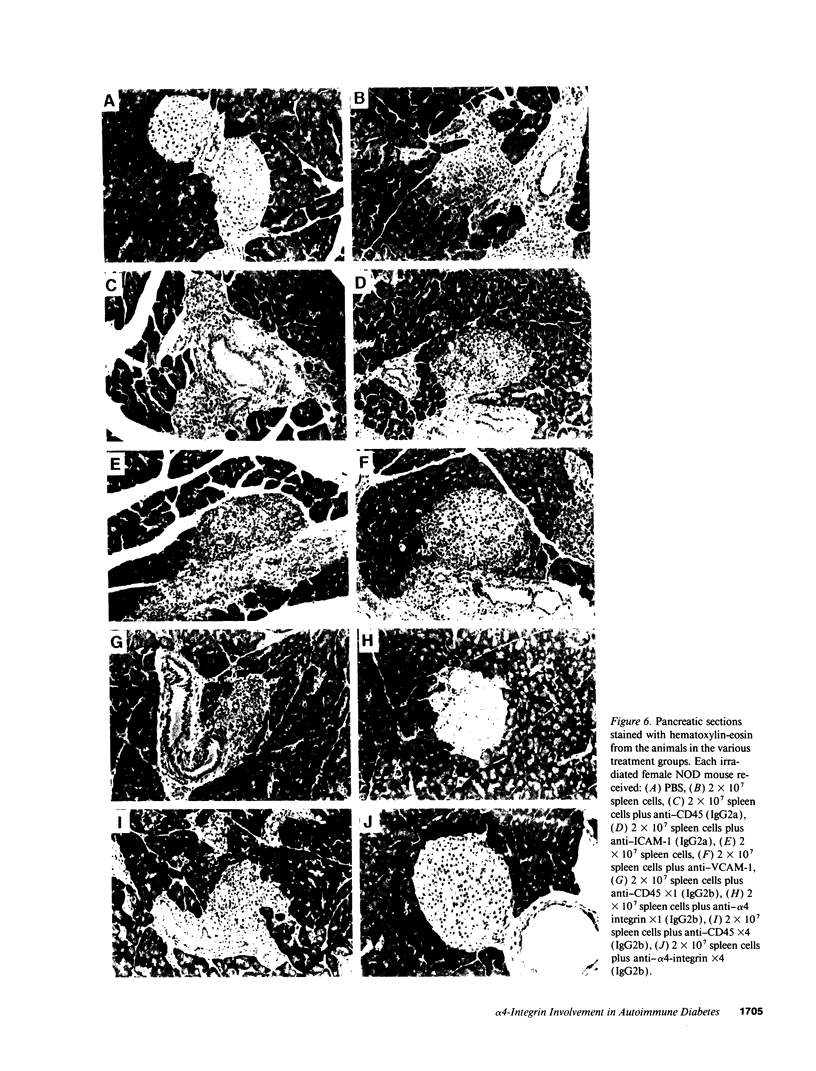
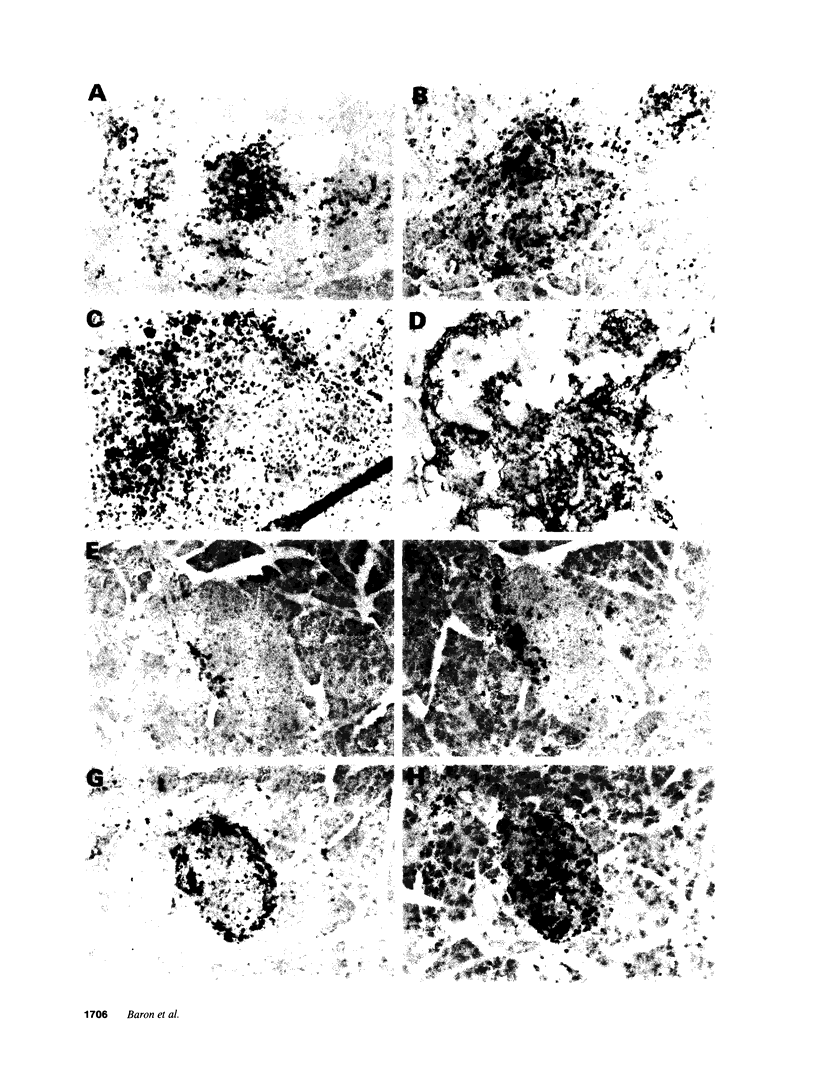
An adoptive transfer model of insulin-dependent diabetes mellitus (IDDM) in the nonobese diabetic mouse was used to examine the roles of alpha 4-integrin, vascular cell adhesion molecule 1 (VCAM-1); and intercellular adhesion molecule 1 (ICAM-1) in the pathogenesis of autoimmune diabetes. Antibodies specific for both alpha 4-integrin and one of its ligands, VCAM-1, were able to delay onset of diabetes and decrease the incidence of the disease in adoptive transfer studies. This blocking of disease was accompanied by a marked decrease in lymphocytic infiltration of the islets of Langerhans. Furthermore, these antibodies preferentially block entrance of CD4 T cells into the tissue. Antibodies specific for ICAM-1 had little effect on the onset or incidence of IDDM. Thus, we conclude that an alpha 4-integrin-VCAM-1 interaction is important in T cell entry into the islets of Langerhans and in the pathogenesis of IDDM. In addition, the cascade of events leading to T cell transit across endothelium may be different for CD4 and CD8 cells, and may differ depending on the endothelium involved. Our results support the more general conclusion that an alpha 4-integrin-VCAM-1 interaction may be crucial in allowing activated effector CD4T cells to leave the blood and enter tissue to clear infection.
Full text
PDF



Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Baron J. L., Madri J. A., Ruddle N. H., Hashim G., Janeway C. A., Jr Surface expression of alpha 4 integrin by CD4 T cells is required for their entry into brain parenchyma. J Exp Med. 1993 Jan 1;177(1):57–68. doi: 10.1084/jem.177.1.57. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Boitard C., Yasunami R., Dardenne M., Bach J. F. T cell-mediated inhibition of the transfer of autoimmune diabetes in NOD mice. J Exp Med. 1989 May 1;169(5):1669–1680. doi: 10.1084/jem.169.5.1669. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Butcher E. C. Leukocyte-endothelial cell recognition: three (or more) steps to specificity and diversity. Cell. 1991 Dec 20;67(6):1033–1036. doi: 10.1016/0092-8674(91)90279-8. [DOI] [PubMed] [Google Scholar]
- Dialynas D. P., Wilde D. B., Marrack P., Pierres A., Wall K. A., Havran W., Otten G., Loken M. R., Pierres M., Kappler J. Characterization of the murine antigenic determinant, designated L3T4a, recognized by monoclonal antibody GK1.5: expression of L3T4a by functional T cell clones appears to correlate primarily with class II MHC antigen-reactivity. Immunol Rev. 1983;74:29–56. doi: 10.1111/j.1600-065x.1983.tb01083.x. [DOI] [PubMed] [Google Scholar]
- Doukas J., Mordes J. P. T lymphocytes capable of activating endothelial cells in vitro are present in rats with autoimmune diabetes. J Immunol. 1993 Feb 1;150(3):1036–1046. [PubMed] [Google Scholar]
- Eisenbarth G. S. Type I diabetes mellitus. A chronic autoimmune disease. N Engl J Med. 1986 May 22;314(21):1360–1368. doi: 10.1056/NEJM198605223142106. [DOI] [PubMed] [Google Scholar]
- Hayward A. R., Cobbold S. P., Waldmann H., Cooke A., Simpson E. Delay in onset of insulitis in NOD mice following a single injection of CD 4 and CD 8 antibodies. J Autoimmun. 1988 Feb;1(1):91–96. doi: 10.1016/0896-8411(88)90080-7. [DOI] [PubMed] [Google Scholar]
- Holzmann B., McIntyre B. W., Weissman I. L. Identification of a murine Peyer's patch--specific lymphocyte homing receptor as an integrin molecule with an alpha chain homologous to human VLA-4 alpha. Cell. 1989 Jan 13;56(1):37–46. doi: 10.1016/0092-8674(89)90981-1. [DOI] [PubMed] [Google Scholar]
- Holzmann B., Weissman I. L. Peyer's patch-specific lymphocyte homing receptors consist of a VLA-4-like alpha chain associated with either of two integrin beta chains, one of which is novel. EMBO J. 1989 Jun;8(6):1735–1741. doi: 10.1002/j.1460-2075.1989.tb03566.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Horley K. J., Carpenito C., Baker B., Takei F. Molecular cloning of murine intercellular adhesion molecule (ICAM-1). EMBO J. 1989 Oct;8(10):2889–2896. doi: 10.1002/j.1460-2075.1989.tb08437.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hutchings P., Rosen H., O'Reilly L., Simpson E., Gordon S., Cooke A. Transfer of diabetes in mice prevented by blockade of adhesion-promoting receptor on macrophages. Nature. 1990 Dec 13;348(6302):639–642. doi: 10.1038/348639a0. [DOI] [PubMed] [Google Scholar]
- Issekutz T. B. Inhibition of in vivo lymphocyte migration to inflammation and homing to lymphoid tissues by the TA-2 monoclonal antibody. A likely role for VLA-4 in vivo. J Immunol. 1991 Dec 15;147(12):4178–4184. [PubMed] [Google Scholar]
- Issekutz T. B., Issekutz A. C. T lymphocyte migration to arthritic joints and dermal inflammation in the rat: differing migration patterns and the involvement of VLA-4. Clin Immunol Immunopathol. 1991 Dec;61(3):436–447. doi: 10.1016/s0090-1229(05)80014-5. [DOI] [PubMed] [Google Scholar]
- Ledbetter J. A., Herzenberg L. A. Xenogeneic monoclonal antibodies to mouse lymphoid differentiation antigens. Immunol Rev. 1979;47:63–90. doi: 10.1111/j.1600-065x.1979.tb00289.x. [DOI] [PubMed] [Google Scholar]
- Lee K. U., Amano K., Yoon J. W. Evidence for initial involvement of macrophage in development of insulitis in NOD mice. Diabetes. 1988 Jul;37(7):989–991. doi: 10.2337/diab.37.7.989. [DOI] [PubMed] [Google Scholar]
- Leiter E. H., Serreze D. V. Antigen presenting cells and the immunogenetics of autoimmune diabetes in NOD mice. Reg Immunol. 1992 Sep-Oct;4(5):263–273. [PubMed] [Google Scholar]
- Leiter E. H., Serreze D. V., Prochazka M. The genetics and epidemiology of diabetes in NOD mice. Immunol Today. 1990 May;11(5):147–149. doi: 10.1016/0167-5699(90)90057-g. [DOI] [PubMed] [Google Scholar]
- Makino S., Kunimoto K., Muraoka Y., Mizushima Y., Katagiri K., Tochino Y. Breeding of a non-obese, diabetic strain of mice. Jikken Dobutsu. 1980 Jan;29(1):1–13. doi: 10.1538/expanim1978.29.1_1. [DOI] [PubMed] [Google Scholar]
- Metlay J. P., Witmer-Pack M. D., Agger R., Crowley M. T., Lawless D., Steinman R. M. The distinct leukocyte integrins of mouse spleen dendritic cells as identified with new hamster monoclonal antibodies. J Exp Med. 1990 May 1;171(5):1753–1771. doi: 10.1084/jem.171.5.1753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miller B. J., Appel M. C., O'Neil J. J., Wicker L. S. Both the Lyt-2+ and L3T4+ T cell subsets are required for the transfer of diabetes in nonobese diabetic mice. J Immunol. 1988 Jan 1;140(1):52–58. [PubMed] [Google Scholar]
- O'Neill J. K., Butter C., Baker D., Gschmeissner S. E., Kraal G., Butcher E. C., Turk J. L. Expression of vascular addressins and ICAM-1 by endothelial cells in the spinal cord during chronic relapsing experimental allergic encephalomyelitis in the Biozzi AB/H mouse. Immunology. 1991 Apr;72(4):520–525. [PMC free article] [PubMed] [Google Scholar]
- Pardi R., Inverardi L., Bender J. R. Regulatory mechanisms in leukocyte adhesion: flexible receptors for sophisticated travelers. Immunol Today. 1992 Jun;13(6):224–230. doi: 10.1016/0167-5699(92)90159-5. [DOI] [PubMed] [Google Scholar]
- Pitzalis C., Kingsley G., Haskard D., Panayi G. The preferential accumulation of helper-inducer T lymphocytes in inflammatory lesions: evidence for regulation by selective endothelial and homotypic adhesion. Eur J Immunol. 1988 Sep;18(9):1397–1404. doi: 10.1002/eji.1830180915. [DOI] [PubMed] [Google Scholar]
- Portoles P., Rojo J., Golby A., Bonneville M., Gromkowski S., Greenbaum L., Janeway C. A., Jr, Murphy D. B., Bottomly K. Monoclonal antibodies to murine CD3 epsilon define distinct epitopes, one of which may interact with CD4 during T cell activation. J Immunol. 1989 Jun 15;142(12):4169–4175. [PubMed] [Google Scholar]
- Raine C. S., Cannella B., Duijvestijn A. M., Cross A. H. Homing to central nervous system vasculature by antigen-specific lymphocytes. II. Lymphocyte/endothelial cell adhesion during the initial stages of autoimmune demyelination. Lab Invest. 1990 Oct;63(4):476–489. [PubMed] [Google Scholar]
- Reich E. P., Sherwin R. S., Kanagawa O., Janeway C. A., Jr An explanation for the protective effect of the MHC class II I-E molecule in murine diabetes. Nature. 1989 Sep 28;341(6240):326–328. doi: 10.1038/341326a0. [DOI] [PubMed] [Google Scholar]
- Rossini A. A., Handler E. S., Greiner D. L., Mordes J. P. Insulin dependent diabetes mellitus hypothesis of autoimmunity. Autoimmunity. 1991;8(3):221–235. doi: 10.3109/08916939108997110. [DOI] [PubMed] [Google Scholar]
- Shimizu Y., Newman W., Gopal T. V., Horgan K. J., Graber N., Beall L. D., van Seventer G. A., Shaw S. Four molecular pathways of T cell adhesion to endothelial cells: roles of LFA-1, VCAM-1, and ELAM-1 and changes in pathway hierarchy under different activation conditions. J Cell Biol. 1991 Jun;113(5):1203–1212. doi: 10.1083/jcb.113.5.1203. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu Y., Newman W., Tanaka Y., Shaw S. Lymphocyte interactions with endothelial cells. Immunol Today. 1992 Mar;13(3):106–112. doi: 10.1016/0167-5699(92)90151-V. [DOI] [PubMed] [Google Scholar]
- Springer T., Galfrè G., Secher D. S., Milstein C. Monoclonal xenogeneic antibodies to murine cell surface antigens: identification of novel leukocyte differentiation antigens. Eur J Immunol. 1978 Aug;8(8):539–551. doi: 10.1002/eji.1830080802. [DOI] [PubMed] [Google Scholar]
- van Dinther-Janssen A. C., Horst E., Koopman G., Newmann W., Scheper R. J., Meijer C. J., Pals S. T. The VLA-4/VCAM-1 pathway is involved in lymphocyte adhesion to endothelium in rheumatoid synovium. J Immunol. 1991 Dec 15;147(12):4207–4210. [PubMed] [Google Scholar]