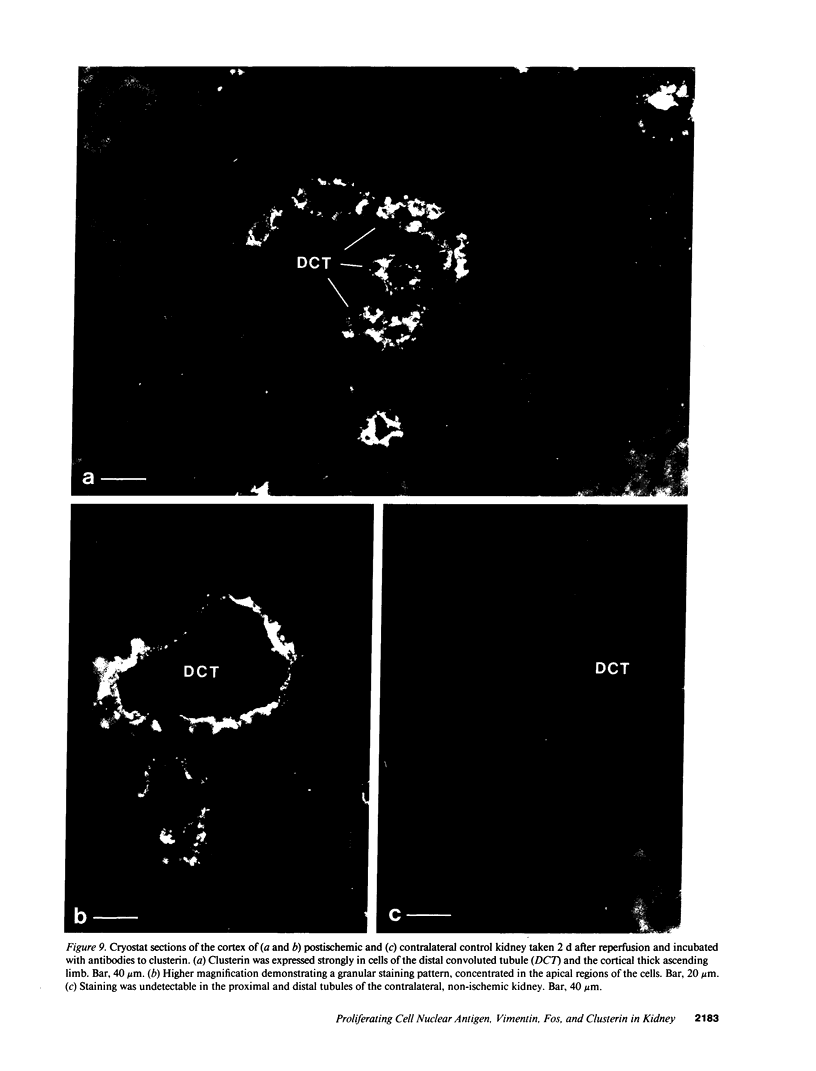
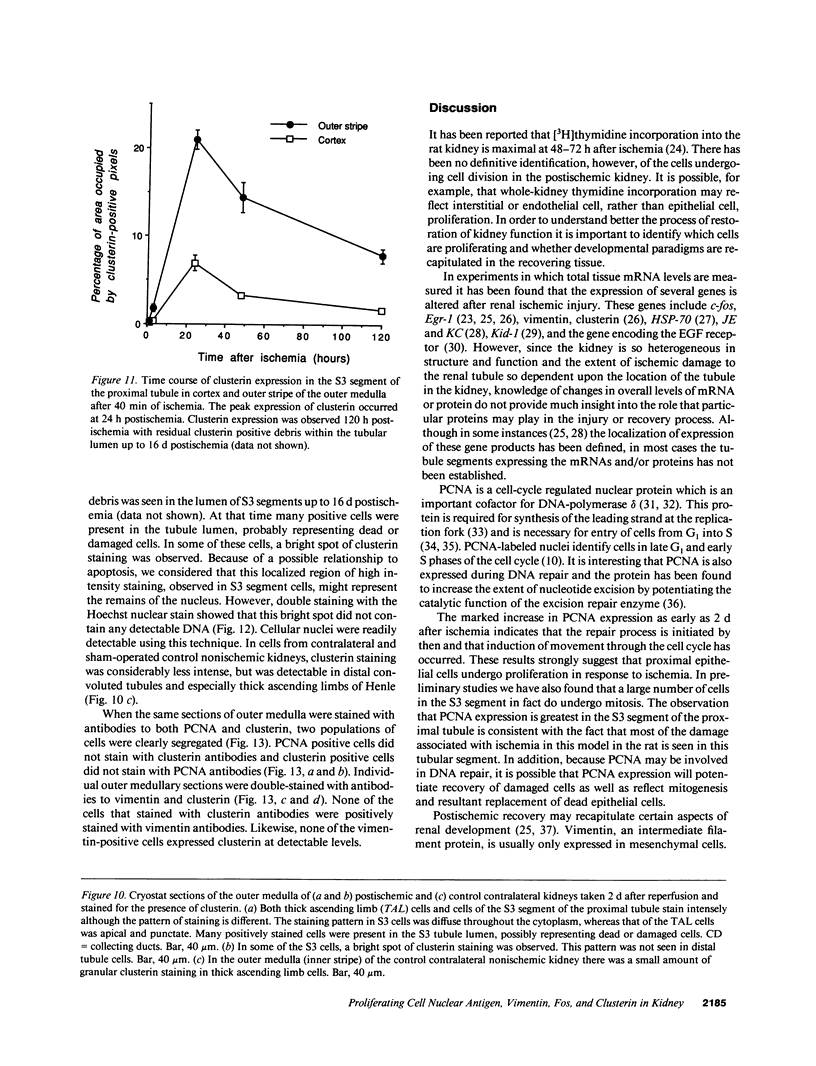
Abstract
The mechanisms leading to the recovery of the kidney after ischemic acute renal failure are poorly understood. To explore the role played by mitogenesis and dedifferentiation in this repair process and to identify whether the genetic response of the nephron segments reflects the level of susceptibility to injury, the temporal and nephron segment expressions of various proteins implicated in mitogenesis, differentiation, and injury were determined. Proliferating cell nuclear antigen (PCNA), a marker for the G1-S transition in the cell cycle and hence mitogenesis, was detected primarily in the S3 segment of the proximal tubule, with maximal expression at 2 d postischemia. Vimentin, normally present in mesenchymal cells but not epithelial cells, and hence a marker for the state of differentiation, was prominently expressed in the S3 segment 2-5 d postischemia. In the S3 segments in the outer stripe of the medulla cells that stained positively for PCNA also stained positively for vimentin. Clusterin, a marker for cell injury, was expressed primarily in the S3 segment and in the distal tubule with distinct staining patterns in each segment. None of the cells that stained with clusterin antibodies were positively stained with PCNA or vimentin antibodies. Likewise, none of the PCNA or vimentin-positive cells expressed clusterin at detectable levels. Thus, in the S3 segment, where there is significant ischemic injury, surviving cells express markers indicating that they undergo mitogenesis and dedifferentiate in the postischemic period. While there is some expression of c-Fos in the S3 segment, c-Fos was expressed predominantly, at 1 and 3 h postischemia, in the nuclei of the distal nephron, particularly in the thick ascending limb. The data support the view that the mature renal S3 segment epithelial cell can be a progenitor cell.
Full text
PDF










Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adamkiewicz J., Brüller H. J., Gausepohl H., Frank R., Müller R. Mapping of functional domains in Fos and Jun proteins using epitope-specific antibodies. Oncogene. 1990 Apr;5(4):525–533. [PubMed] [Google Scholar]
- Arendshorst W. J., Finn W. F., Gottschalk C. W. Pathogenesis of acute renal failure following temporary renal ischemia in the rat. Circ Res. 1975 Nov;37(5):558–568. doi: 10.1161/01.res.37.5.558. [DOI] [PubMed] [Google Scholar]
- Bacallao R., Fine L. G. Molecular events in the organization of renal tubular epithelium: from nephrogenesis to regeneration. Am J Physiol. 1989 Dec;257(6 Pt 2):F913–F924. doi: 10.1152/ajprenal.1989.257.6.F913. [DOI] [PubMed] [Google Scholar]
- Bayati A., Nygren K., Källskog O., Wolgast M. The long-term outcome of post-ischaemic acute renal failure in the rat. II. A histopathological study of the untreated kidney. Acta Physiol Scand. 1990 Jan;138(1):35–47. doi: 10.1111/j.1748-1716.1990.tb08810.x. [DOI] [PubMed] [Google Scholar]
- Bonventre J. V. Mechanisms of ischemic acute renal failure. Kidney Int. 1993 May;43(5):1160–1178. doi: 10.1038/ki.1993.163. [DOI] [PubMed] [Google Scholar]
- Bonventre J. V., Sukhatme V. P., Bamberger M., Ouellette A. J., Brown D. Localization of the protein product of the immediate early growth response gene, Egr-1, in the kidney after ischemia and reperfusion. Cell Regul. 1991 Mar;2(3):251–260. doi: 10.1091/mbc.2.3.251. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bravo R., Frank R., Blundell P. A., Macdonald-Bravo H. Cyclin/PCNA is the auxiliary protein of DNA polymerase-delta. Nature. 1987 Apr 2;326(6112):515–517. doi: 10.1038/326515a0. [DOI] [PubMed] [Google Scholar]
- Brown D., Kunz A., Wohlwend A., Vassalli J. D., Orci L. Détection ultrastructurale de l'hétérogénéité du glycocalyx dans les tubes contournés et droits proximaux du rein de Rat par la technique de la lectine couplée à l'or [1]. C R Seances Acad Sci III. 1983;297(10):501–506. [PubMed] [Google Scholar]
- Brown D., Zhu X. L., Sly W. S. Localization of membrane-associated carbonic anhydrase type IV in kidney epithelial cells. Proc Natl Acad Sci U S A. 1990 Oct;87(19):7457–7461. doi: 10.1073/pnas.87.19.7457. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Connolly K. M., Bogdanffy M. S. Evaluation of proliferating cell nuclear antigen (PCNA) as an endogenous marker of cell proliferation in rat liver: a dual-stain comparison with 5-bromo-2'-deoxyuridine. J Histochem Cytochem. 1993 Jan;41(1):1–6. doi: 10.1177/41.1.7678022. [DOI] [PubMed] [Google Scholar]
- Dierick A. M., Praet M., Roels H., Verbeeck P., Robyns C., Oosterlinck W. Vimentin expression of renal cell carcinoma in relation to DNA content and histological grading: a combined light microscopic, immunocytochemical and cytophotometrical analysis. Histopathology. 1991 Apr;18(4):315–322. doi: 10.1111/j.1365-2559.1991.tb00852.x. [DOI] [PubMed] [Google Scholar]
- Grima J., Zwain I., Lockshin R. A., Bardin C. W., Cheng C. Y. Diverse secretory patterns of clusterin by epididymis and prostate/seminal vesicles undergoing cell regression after orchiectomy. Endocrinology. 1990 Jun;126(6):2989–2997. doi: 10.1210/endo-126-6-2989. [DOI] [PubMed] [Google Scholar]
- Holthöfer H., Miettinen A., Lehto V. P., Lehtonen E., Virtanen I. Expression of vimentin and cytokeratin types of intermediate filament proteins in developing and adult human kidneys. Lab Invest. 1984 May;50(5):552–559. [PubMed] [Google Scholar]
- Humes H. D., Cieslinski D. A., Coimbra T. M., Messana J. M., Galvao C. Epidermal growth factor enhances renal tubule cell regeneration and repair and accelerates the recovery of renal function in postischemic acute renal failure. J Clin Invest. 1989 Dec;84(6):1757–1761. doi: 10.1172/JCI114359. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jaskulski D., deRiel J. K., Mercer W. E., Calabretta B., Baserga R. Inhibition of cellular proliferation by antisense oligodeoxynucleotides to PCNA cyclin. Science. 1988 Jun 10;240(4858):1544–1546. doi: 10.1126/science.2897717. [DOI] [PubMed] [Google Scholar]
- Jenne D. E., Tschopp J. Molecular structure and functional characterization of a human complement cytolysis inhibitor found in blood and seminal plasma: identity to sulfated glycoprotein 2, a constituent of rat testis fluid. Proc Natl Acad Sci U S A. 1989 Sep;86(18):7123–7127. doi: 10.1073/pnas.86.18.7123. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kelly K. J., Williams W. W., Jr, Colvin R. B., Bonventre J. V. Antibody to intercellular adhesion molecule 1 protects the kidney against ischemic injury. Proc Natl Acad Sci U S A. 1994 Jan 18;91(2):812–816. doi: 10.1073/pnas.91.2.812. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu Y. C., Marraccino R. L., Keng P. C., Bambara R. A., Lord E. M., Chou W. G., Zain S. B. Requirement for proliferating cell nuclear antigen expression during stages of the Chinese hamster ovary cell cycle. Biochemistry. 1989 Apr 4;28(7):2967–2974. doi: 10.1021/bi00433a034. [DOI] [PubMed] [Google Scholar]
- Malis C. D., Cheung J. Y., Leaf A., Bonventre J. V. Effects of verapamil in models of ischemic acute renal failure in the rat. Am J Physiol. 1983 Dec;245(6):F735–F742. doi: 10.1152/ajprenal.1983.245.6.F735. [DOI] [PubMed] [Google Scholar]
- Mason J., Olbricht C., Takabatake T., Thurau K. The early phase of experimental acute renal failure. I. Intratubular pressure and obstruction. Pflugers Arch. 1977 Aug 29;370(2):155–163. doi: 10.1007/BF00581689. [DOI] [PubMed] [Google Scholar]
- Molitoris B. A., Falk S. A., Dahl R. H. Ischemia-induced loss of epithelial polarity. Role of the tight junction. J Clin Invest. 1989 Oct;84(4):1334–1339. doi: 10.1172/JCI114302. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molitoris B. A. Ischemia-induced loss of epithelial polarity: potential role of the actin cytoskeleton. Am J Physiol. 1991 Jun;260(6 Pt 2):F769–F778. doi: 10.1152/ajprenal.1991.260.6.F769. [DOI] [PubMed] [Google Scholar]
- Nichols A. F., Sancar A. Purification of PCNA as a nucleotide excision repair protein. Nucleic Acids Res. 1992 Jul 11;20(13):2441–2446. doi: 10.1093/nar/20.10.2441. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osborn M., Debus E., Weber K. Monoclonal antibodies specific for vimentin. Eur J Cell Biol. 1984 May;34(1):137–143. [PubMed] [Google Scholar]
- Ouellette A. J., Malt R. A., Sukhatme V. P., Bonventre J. V. Expression of two "immediate early" genes, Egr-1 and c-fos, in response to renal ischemia and during compensatory renal hypertrophy in mice. J Clin Invest. 1990 Mar;85(3):766–771. doi: 10.1172/JCI114502. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Palmer D. J., Christie D. L. The primary structure of glycoprotein III from bovine adrenal medullary chromaffin granules. Sequence similarity with human serum protein-40,40 and rat Sertoli cell glycoprotein. J Biol Chem. 1990 Apr 25;265(12):6617–6623. [PubMed] [Google Scholar]
- Polla B. S., Mili N., Donati Y. R., Bonventre J. V. Les protéines du choc thermique: quelles implications en néphrologie? Nephrologie. 1991;12(3):119–123. [PubMed] [Google Scholar]
- Prelich G., Stillman B. Coordinated leading and lagging strand synthesis during SV40 DNA replication in vitro requires PCNA. Cell. 1988 Apr 8;53(1):117–126. doi: 10.1016/0092-8674(88)90493-x. [DOI] [PubMed] [Google Scholar]
- Prelich G., Tan C. K., Kostura M., Mathews M. B., So A. G., Downey K. M., Stillman B. Functional identity of proliferating cell nuclear antigen and a DNA polymerase-delta auxiliary protein. Nature. 1987 Apr 2;326(6112):517–520. doi: 10.1038/326517a0. [DOI] [PubMed] [Google Scholar]
- Rosenberg M. E., Paller M. S. Differential gene expression in the recovery from ischemic renal injury. Kidney Int. 1991 Jun;39(6):1156–1161. doi: 10.1038/ki.1991.146. [DOI] [PubMed] [Google Scholar]
- Safirstein R., Megyesi J., Saggi S. J., Price P. M., Poon M., Rollins B. J., Taubman M. B. Expression of cytokine-like genes JE and KC is increased during renal ischemia. Am J Physiol. 1991 Dec;261(6 Pt 2):F1095–F1101. doi: 10.1152/ajprenal.1991.261.6.F1095. [DOI] [PubMed] [Google Scholar]
- Safirstein R., Price P. M., Saggi S. J., Harris R. C. Changes in gene expression after temporary renal ischemia. Kidney Int. 1990 Jun;37(6):1515–1521. doi: 10.1038/ki.1990.143. [DOI] [PubMed] [Google Scholar]
- Sheng M., Greenberg M. E. The regulation and function of c-fos and other immediate early genes in the nervous system. Neuron. 1990 Apr;4(4):477–485. doi: 10.1016/0896-6273(90)90106-p. [DOI] [PubMed] [Google Scholar]
- Thurau K., Boylan J. W. Acute renal success. The unexpected logic of oliguria in acute renal failure. Am J Med. 1976 Sep;61(3):308–315. doi: 10.1016/0002-9343(76)90365-x. [DOI] [PubMed] [Google Scholar]
- Tsuruta J. K., Wong K., Fritz I. B., Griswold M. D. Structural analysis of sulphated glycoprotein 2 from amino acid sequence. Relationship to clusterin and serum protein 40,40. Biochem J. 1990 Jun 15;268(3):571–578. doi: 10.1042/bj2680571. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wallin A., Zhang G., Jones T. W., Jaken S., Stevens J. L. Mechanism of the nephrogenic repair response. Studies on proliferation and vimentin expression after 35S-1,2-dichlorovinyl-L-cysteine nephrotoxicity in vivo and in cultured proximal tubule epithelial cells. Lab Invest. 1992 Apr;66(4):474–484. [PubMed] [Google Scholar]
- Waseem N. H., Lane D. P. Monoclonal antibody analysis of the proliferating cell nuclear antigen (PCNA). Structural conservation and the detection of a nucleolar form. J Cell Sci. 1990 May;96(Pt 1):121–129. doi: 10.1242/jcs.96.1.121. [DOI] [PubMed] [Google Scholar]
- Witzgall R., O'Leary E., Gessner R., Ouellette A. J., Bonventre J. V. Kid-1, a putative renal transcription factor: regulation during ontogeny and in response to ischemia and toxic injury. Mol Cell Biol. 1993 Mar;13(3):1933–1942. doi: 10.1128/mcb.13.3.1933. [DOI] [PMC free article] [PubMed] [Google Scholar]



















