Abstract
Mechanosensitive large-conductance Ca2+-activated K+ channels encoded by the Slo1 gene (BKCa channels) are expressed in podocytes. Here we show that BKCa channels reciprocally coimmunoprecipitate with synaptopodin (Synpo) in mouse glomeruli, in mouse podocytes, and in a heterologous expression system (HEK293T cells) in which these proteins are transiently expressed. Synpo and Slo1 colocalize along the surface of the glomerular basement membrane in mouse glomeruli. Synpo interacts with BKCa channels at COOH-terminal domains that overlap with an actin-binding domain on the channel molecule that is necessary for trafficking of BKCa channels to the cell surface. Moreover, addition of exogenous β-actin to mouse podocyte lysates reduces BKCa-Synpo interactions. Coexpression of Synpo increases steady-state surface expression of BKCa channels in HEK293T cells. However, Synpo does not affect the stability of cell surface BKCa channels, suggesting a primary effect on the rate of forward trafficking, and Synpo coexpression does not affect BKCa gating. Conversely, stable knockdown of Synpo expression in mouse podocyte cell lines reduces steady-state surface expression of BKCa channels but does not affect total expression of BKCa channels or their gating. The effects of Synpo on surface expression of BKCa are blocked by inhibition of Rho signaling in HEK293T cells and in podocytes. Functional cell surface BKCa channels in podocytes are also reduced by sustained (2 h) but not acute (15 min) depolymerization of actin with cytochalasin D. Synpo may regulate BKCa channels through its effects on actin dynamics and by modulating interactions between BKCa channels and regulatory proteins of the podocyte slit diaphragm.
Keywords: potassium channels, slit diaphragm, traffic, renal glomerulus, cytoskeleton
glomerular capillaries have an unusually high pressure compared with other capillary beds (7), and there is evidence that stretching of the glomerular capillary and fluid shear forces subject podocytes and slit diaphragms to substantial mechanical stress (12, 22). The contractile machinery of podocyte foot processes is generally believed to counteract these forces and thereby maintain the stability of the glomerular filter (8), but this requires mechanisms to transduce mechanical forces into biochemical signals that can lead to altered behavior in podocytes. Large-conductance Ca2+-activated K+ channels (BKCa channels) may contribute to mechanosensation in foot processes, as they are expressed in podocytes and have gating properties that are sensitive to mechanical stretch of the plasma membrane (24).
The pore-forming α-subunits of BKCa channels are encoded by the Slo1 gene (also known as KCNMA1 and KCa1.1). These channels are characterized by an unusually large unitary conductance, and therefore activation of a small number of them can produce large changes in membrane potential. Moreover, coordinated activation of BKCa channels, by maintaining a driving force for entry of divalent cations, would be expected to increase Ca2+ influx through canonical transient receptor potential (TRPC)6 channels or other cation channels with similar permeation properties (10, 16).
In previous studies, we have shown that BKCa channels interact with podocyte proteins, including nephrin (18), Neph1 (17), MAGI-1 (31), and TRPC6 (16) that are functionally connected to the foot process cytoskeleton. In the present study we examined an interaction between BKCa channels and synaptopodin (Synpo), a proline-rich actin-associated protein expressed in podocyte foot processes (25). Synpo is a key node in a protein interaction network that regulates podocyte actin dynamics (11) by competitively inhibiting binding of Cdc42 to insulin receptor substrate p53 (IRSp53):Mena complexes (36), thereby inhibiting formation of filopodia (32). Synpo also binds to the E3-ligase Smurf-1, which prevents ubiquitinylation and subsequent degradation of RhoA, thereby promoting formation of stress fibers (3). Synpo also associates with other actin-binding proteins including α-actinin-4 (2, 21), CD2AP (13), and MAGI-1 (29), which contribute to the structural links between the slit diaphragm and basal membrane domains and the underlying foot process cytoskeleton (11). Through these interactions, Synpo increases the stability of the glomerular filter and minimizes effacement caused by acute insults such as in vivo administration of protamine sulfate or lipopolysaccharide (2).
Synpo is itself regulated by several mechanisms. When Synpo is phosphorylated, for example by protein kinase A, it can bind to 14-3-3β, and this interaction prevents proteolytic degradation of Synpo by cathepsin-L (11). Moreover, Synpo can be dephosphorylated by calcineurin and is therefore connected to a potentially large number of signaling cascades, especially cascades coupled to Ca2+ influx or mobilization (11). We have previously shown that interactions with actin are essential for normal trafficking of BKCa channels to the cell surface (37) and that actin dynamics regulate BKCa trafficking in neurons (5). This raises the possibility that a protein like Synpo, which regulates actin dynamics in the slit diaphragm, could play a role in regulation of ion channel trafficking in podocytes. In the present study we tested this hypothesis and show that Synpo interacts with BKCa channels and regulates their normal steady-state expression in the podocyte plasma membrane. We also present data implicating a role for actin and Rho signaling the trafficking of these channels.
MATERIALS AND METHODS
Cell culture and transfection.
Procedures for growing HEK293T cells and mouse podocyte cell lines (obtained from Dr. Peter Mundel of the University of Miami Miller School of Medicine) have been described previously (16–18, 31). The Synpo KD podocyte cell line stably expresses a small interfering RNA that specifically ablates expression of all three Synpo splice variants (2). The podocyte cell lines were propagated at 33°C in the presence of mouse γ-interferon (10 U/ml) (Sigma, St. Louis, MO). Removal of γ-interferon and raising the temperature to 37°C for 2 wk induced differentiation and expression of podocyte markers. Transfection of full-length expression plasmids into HEK293T cells occurred with an efficiency of >90% with Lipofectamine as described previously (18–20). Cells were used for biochemistry or electrophysiology 48 h after transfection.
Glomerular isolation.
Mice were anesthetized and killed according to procedures approved by the University of Houston Animal Care and Use Committee, and kidneys were excised and placed in ice-cold PBS. The capsule and adhering fat were removed, and the cortex was carefully dissected and chopped into 1-mm3 pieces with a razor blade. A preparation enriched in glomeruli was made by a standard sequential sieving procedure using three stainless steel sieves with mesh sizes of 150 μm, 75 μm, and 53 μm. Glomeruli were then collected by brief centrifugation and used for immunoprecipitation.
Plasmid constructs, glutathione S-transferase fusion proteins, and antibodies.
An expression plasmid encoding an NH2-terminal (ectofacial) Myc-tagged Slo1VEDEC isoform of Slo1 was provided by Dr. Min Li of the Department of Neuroscience at Johns Hopkins University and has been described in detail previously (19, 20). A plasmid encoding FLAG-tagged Synpo-L was provided by Peter Mundel (2). Plasmids encoding glutathione S-transferase (GST)-Slo1 fusion proteins were described in detail previously (37). A number of antibodies were used for immunoblot, immunoprecipitation, or confocal microscopy, including mouse anti-Myc 9B11 (Cell Signaling, Danvers, MA), rabbit anti-Synpo (Santa Cruz Biotechnology), mouse anti-Synpo (American Research Products), anti-FLAG (Millipore), rabbit anti-Slo1 (Alomone), and a mouse monoclonal antibody against Slo1 (clone L6/60) obtained from the University of California Davis/National Institute of Neurological Disorders and Stroke/National Institute of Mental Health NeuroMab Facility, supported by National Institute of Neurological Disorders and Stroke Grant U24-NS-050606. In addition we utilized a rabbit anti-Slo1 that we had commercially prepared against COOH-terminal domains of Slo1. Properties of this antibody are presented in the Supplemental Data for this article.1
Coimmunoprecipitation, GST pull-down assays, and measurements of endocytosis.
Coimmunoprecipitation, GST pull-down assays, and measurements of endocytosis were done as described previously (6, 16–19, 31, 37, 38). For coimmunoprecipitation, HEK293T cell, podocyte, or glomerular lysates were cleared by centrifugation and the resulting extracts were incubated in the presence of anti-Myc, anti-FLAG, anti-Slo1, Anti-Synpo, or IgG for 4 h at 4°C. Protein A/G agarose (Santa Cruz Biotechnology) was added to the lysates and incubated for 12 h. Pellets were washed, and proteins were separated by SDS-PAGE on 10% gels and transferred to filters. Cell extracted protein (50–100 μg) was used as a positive control in each experiment. Blots were blocked, washed, incubated with the primary antibody overnight at 4°C, and washed again, and the membrane was incubated with horseradish peroxidase (HRP)-conjugated secondary antibody for 1 h at room temperature. The proteins were visualized with chemiluminescent substrates. GST pull-down assays were performed on podocyte lysates as described previously (31). Briefly, a series of purified GST-Slo1 COOH-terminal domain fusion proteins were bound to glutathione-Sepharose 4B beads (GE Healthcare Biosciences, Piscataway, NJ) in ice-cold PBS. Podocyte lysates were analyzed for interaction with the different fusion proteins in 0.5% Triton X-100 in PBS (PBST) at 4°C with gentle rotation overnight. In experiments to determine whether actin could compete for Synpo binding to GST-Slo1 fusion proteins, various concentrations of recombinant globular β-actin (Cytoskeleton, Denver, CO) were added to a podocyte lysate, and the samples were probed with the GST-Slo1-CT3 fusion proteins described below. Proteins that bound to GST-Slo1 fusion proteins were eluted into SDS sample buffer, separated on 10% SDS-PAGE gels, transferred to filters, and analyzed by immunoblot. GST pull-down assays to analyze Rho activation were carried out with reagents from Cytoskeleton according to the manufacturer's instructions. For endocytosis assays (6), HEK293T cells transiently expressing Myc-tagged Slo1VEDEC and Flag-Synpo or empty vector were placed at 4°C for 20 min to stop trafficking and degradation. Surface Slo1 was labeled with mouse anti-Myc in 4°C medium for 1 h. After extensive washing with cold medium, trafficking was allowed to resume by incubating cells at 37°C for different periods of time. Cells were then fixed in 4% paraformaldehyde without being permeabilized. Anti-Myc remaining on the cell surface was detected with HRP-conjugated anti-mouse IgG and then exposed to FAST OPD reagents (Sigma-Aldrich, St. Louis, MO) for 3 min with constant shaking, and the colorimetric reaction was then stopped by adding 3 N HCl for 10 min. The supernatant was collected, and the optical absorbance was measured at 492 nm with a Multiskan MCC microplate reader.
Cell surface biotinylation assays.
These were carried out as described in detail previously (6, 18–20). Briefly, HEK293T cells or podocytes were treated with a membrane-impermeant biotinylation reagent, sulfo-N-hydroxy-succinimidobiotin (Pierce Biotechnology, Rockford, IL; 1 mg/ml in PBS buffer) for 1 h. The reaction was stopped, cells were lysed, and biotinylated proteins from the cell surface were recovered from lysates by incubation with immobilized streptavidin-agarose beads (Pierce Biotechnology). A sample of the initial cell lysate was retained for analysis of total proteins. These samples were separated on SDS-PAGE, and proteins were quantified by immunoblot analysis using anti-Myc in the case of HEK293T cells or anti-Slo1 in the case of podocytes, followed by densitometric analysis of the main (monomeric) band of Slo1. All experiments were repeated at least three times.
Electrophysiology and data analysis.
All recordings were made at room temperature (22°C). Whole cell recordings of BKCa channels from HEK293T cells were made as described previously (18, 19). Briefly, plasmids encoding green fluorescent protein (GFP) were cotransfected with Myc-Slo1VEDEC and FLAG-Synpo-L constructs or empty vector. Cotransfection with GFP allowed identification of transfected cells by fluorescence microscopy during whole cell recordings. The bathing solution contained (in mM) 150 NaCl, 0.08 KCl, 0.8 MgCl2, 5.4 CaCl2, 10 glucose, and 10 HEPES, and the pH was adjusted to 7.4 with NaOH. The pipette solution contained (in mM) 145 NaCl, 2 KCl, 6.2 MgCl2, 10 HEPES, and 5 N-(2-hydroxyethyl)ethylenediamine-N,N′,N′-triacetic acid (HEDTA), pH 7.2. The free Ca2+ concentration in this solution was adjusted to 5 μM, which allowed for robust activation of BKCa channels by depolarizing voltage pulses from a holding potential of −60 mV. It should be noted that the reduced K+ concentrations in these solutions are designed to reduce macroscopic K+ current amplitudes to levels that can be observed without saturation of the patch-clamp amplifier. However, the K+ equilibrium potential under these conditions is physiological. For measurements of whole cell currents through endogenous BKCa channels in podocytes, the bath solution contained (in mM) 150 NaCl, 5.4 KCl, 0.8 MgCl2, 5.4 CaCl2, and 10 HEPES, pH 7.4. Pipette solutions contained (in mM) 10 NaCl, 125 KCl, 6.2 MgCl2, and 10 HEPES, pH 7.2, with 5 μM free Ca2+ buffered with 10 mM HEDTA, as determined with a calcium electrode. Currents were evoked by depolarizing voltage steps from a holding potential of −60 mV. We showed previously (18) that currents evoked by these protocols in podocytes are completely blocked by paxiline, a selective blocker of BKCa channels. In all of these experiments, data were digitized with a Digidata 1322A interface (Molecular Devices) and stored for off-line analysis with pCLAMP software (Molecular Devices).
Confocal microscopy.
For double-labeling experiments in podocytes, we used mouse anti-Slo1 (clone L6/60) and Santa Cruz Biotechnology rabbit anti-Synpo. Secondary antibodies were Alexa Fluor 488-conjugated anti-rabbit and Alexa Fluor 594-conjugated anti-mouse (both from Molecular Probes; 1:1,000 dilution). Actin was labeled with rhodamine-conjugated phalloidin (Molecular Probes). For experiments on paraffin sections of mouse kidney, we used our custom-made anti-Slo1, which gives good results in this application together with mouse anti-Synpo. Adult mice were anesthetized and perfused with saline followed by 4% paraformaldehyde. Kidneys were then excised and embedded in paraffin, and 7-μm microtome sections were prepared on glass slides and dried. After deparaffinization and rehydration, sections were incubated with rabbit anti-Slo1 and mouse anti-Synpo (Santa Cruz Biotechnology) at 25°C for 12 h. To improve signal-to-noise ratios in double-label immunofluorescence, the tyramide signal amplification method was used according to the protocol furnished by the manufacturer (TSA kits no. 22 and no. 25, Invitrogen, Carlsbad, CA). All images were collected on an Olympus FV-1000 inverted stage confocal microscope with a Plan Apo N ×60 1.42NA oil-immersion objective. Stacks of images from different Z planes were obtained with a step size of 200 nm, and fluorescent signals from different channels were scanned sequentially. Images that show colocalization of proteins are from the same optical section.
Statistics.
All quantitative data are presented as means ± SE, and bar graphs constructed from electrophysiological data comprise a minimum of 15 cells in each group. Data were analyzed by Student's t-test, with P < 0.05 regarded as significant, or by one-way analysis of variance followed by Tukey's post hoc test.
RESULTS
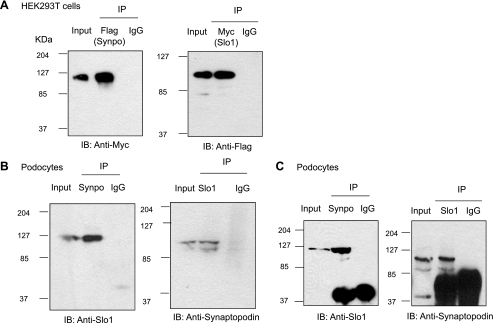
Interactions between BKCa channels and Synpo were readily detected by coimmunoprecipitation. Initial experiments were carried out in HEK293T cells transiently expressing Flag-tagged Synpo and an NH2-terminal (ectofacial) Myc-tagged VEDEC isoform of Slo1 (Slo1VEDEC) (19, 20). When immunoprecipitation was carried out with anti-Flag, we readily detected Myc-Slo1VEDEC as a component of the precipitate. Conversely, when initial immunoprecipitation was carried out with anti-Myc, we detected Synpo in the immunoprecipitate with anti-Flag. We did not see any signal when immunoprecipitation was carried out with IgG (Fig. 1A). A similar pattern was obtained when we examined interactions between endogenous Slo1 proteins and Synpo in differentiated cells of an immortalized mouse podocyte cell line. In these experiments, Slo1 was detected with either a custom-made anti-Slo1 (Fig. 1B, left) or anti-Slo1 (Fig. 1B, right) in precipitates prepared with an antibody against Synpo. Also, Synpo was detected in immunoprecipitates prepared with Alomone anti-Slo1 or the custom-made anti-Slo1 (Fig. 1B). Using the custom anti-Slo1, we could also observe colocalization between Synpo and Slo1 in paraffin sections of mouse kidney cortex (Fig. 2A). Colocalization could be seen along the areas adjacent to the glomerular basement membrane (GBM), which presumably represent podocyte foot processes. Slo1 expression could also be observed by itself in areas inside the GBM, which probably reflects Slo1 expression in mesangial cells. With the same antibody combination, it was also possible to demonstrate coimmunoprecipitation from mouse glomeruli prepared by sieving (Fig. 2B). Thus interactions between BKCa channels and Synpo can be detected in multiple cell systems by using multiple antibody pairs. The experiments also suggest that this interaction occurs in podocytes in vivo.
Fig. 1.
Interaction between pore-forming subunit of large-conductance Ca2+-activated K+ (BKCa) channels (Slo1) and synaptopodin (Synpo). A: representative example of reciprocal coimmunoprecipitation of Slo1 and Synpo transiently expressed in HEK293T cells. Immunoprecipitation (IP) and detection were carried out with antibodies against the Flag tag on Synpo and against the Myc tag on Slo1. In controls, immunoprecipitation was carried out with IgG. In this and subsequent figures, a sample of the cell lysate was used as a positive control, and these lanes are marked “Input.” IB, immunoblot. B: reciprocal coimmunoprecipitation of Slo1 and Synpo in differentiated cells of a mouse podocyte cell line using an antibody against Synpo carried and a custom-made anti-Slo1. C: reciprocal coimmunoprecipitation of Slo1 and Synpo in differentiated cells of a mouse podocyte cell line using anti-Synpo and a commercially available anti-Slo1 (see materials and methods).
Fig. 2.
Colocalization and interaction of Slo1 and Synpo in mouse glomeruli. A: from a paraffin section of mouse kidney cortex, confocal image of double-label immunofluorescence showing Slo1 (red), Synpo (green), and a merged image, as indicated. Note colocalization of Synpo and Slo1 along the surface of the glomerular basement membrane, indicating expression in podocyte foot processes. There also appears to be Slo1 expression in locations consistent with expression in mesangial cells. B: reciprocal coimmunoprecipitation of Slo1 and Synpo in isolated mouse glomeruli.
To further analyze this interaction, we carried out a series of GST pull-down assays on podocyte lysates, using fusion proteins prepared from COOH-terminal domains of Slo1 that we have described previously (20, 31, 37, 38). We first examined three different constructs derived from the cytosolic COOH terminus. We observed that only the most distal of these, Slo1 CT3, comprising GST fused to residues L985–Q1108 of Slo1, was able to pull Synpo out of the lysates (Fig. 3A). This construct contains an actin-binding domain that we have previously shown to be involved in Slo1 trafficking (37), but it is downstream of a noncanonical SH3-binding motif that can bind to cortactin and other actin-binding proteins (34); that motif is located inside our Slo1 CT1 construct. Synpo interactions are also distal to a GK-domain binding motif, which is located inside CT2 (37). In this and subsequent experiments, we observed that GST itself was unable to interact with Synpo in podocyte lysates. We subsequently divided the distal Slo1 CT3 construct into four smaller domains (CT3A, CT3B, CT3C, and CT3D) and used them to probe podocyte lysates for their ability to interact with Synpo (Fig. 3B). The actin-binding sequence that we described previously (37) is in Slo1 CT3B, which comprises residues F1015–T1049 of Slo1. We observed that this motif and the adjacent CT3A region (L985–R1019), as well as the more distal CT3D region (E1068–Q1108), could pull Synpo out of lysates, whereas the CT3C region (E1043–E1072) was ineffective. In other words, interactions with Synpo occur in domains that bracket and overlap a conserved domain on Slo1 that directly binds to actin and is essential for channel trafficking to the cell surface (37). Therefore, we examined whether addition of an excess of exogenous actin could suppress binding of Synpo to the Slo1 CT3 construct (Fig. 3C). We observed that addition of exogenous recombinant globular β-actin was able to reduce Synpo binding to CT3.
Fig. 3.
Interactions between Slo1 and Synpo mapped with glutathione S-transferase (GST) pull-down assays. A: Synpo was pulled out of podocyte lysates (arrow) with a GST fusion protein comprised of the most distal portion of the large cytosolic COOH terminal of Slo1 (CT3) but not by GST or the other GST-Slo1 fusion proteins described below. B: mapping of Synpo interactions using 4 smaller GST-Slo1 fusion proteins that span the distal portion of the COOH terminal. CT3B contains an actin-binding domain described previously (37). C: adding recombinant β-actin to podocyte lysates causes concentration-dependent reduction in interaction between Synpo and GST-Slo1 fusion protein.
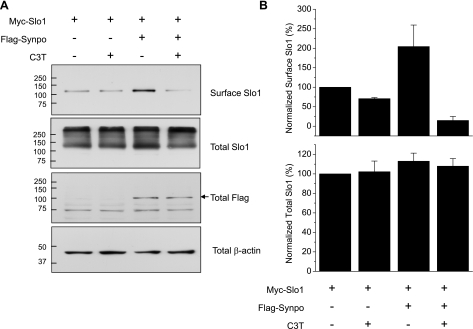
We next examined the effect of Synpo on the surface expression of Slo1 proteins transiently expressed in HEK293T cells. For this experiment, we again utilized an expression plasmid encoding Myc-Slo1VEDEC, as we have shown that this Slo1 isoform exhibits regulated trafficking in several cell types (19, 20). In addition, it is expressed in mouse podocytes and can exert dominant-negative effects on surface expression of other Slo1 splice variants (6, 23). We initially examined Myc-Slo1VEDEC surface expression 48 h after transfection by means of cell surface biotinylation assays using an antibody against the Myc tag. We observed that coexpression of Synpo increased steady-state expression of Myc-Slo1VEDEC on the HEK293T cell surface but had no effect on total expression of Myc-Slo1VEDEC (Fig. 4A). To make this more quantitative, we carried out quantitative densitometry on four replicates of this experiment (Fig. 4B), which confirmed the conclusion. This effect was also observed with whole cell recordings from transiently transfected HEK293T cells using recording pipettes filled with a solution containing 5 μM free Ca2+ to allow for robust activation of BKCa channels (Fig. 5). In these experiments, K+ concentrations in the recording pipette and external bathing solutions were reduced to allow whole cell currents to be recorded without saturating the patch-clamp amplifier (20). Mean currents and current densities measured at step potentials ranging between −40 and +80 mV were substantially and significantly higher (P < 0.050) in cells coexpressing Synpo compared with cells expressing Myc-Slo1VEDEC alone (Fig. 5, A and B). Synpo coexpression did not change the voltage dependence of BKCa channels (Fig. 5C). In addition, Synpo coexpression did not produce any significant effect on the kinetics of BKCa activation, which were quantified by using the time constant of single-exponential curves fitted to the rising phase of macroscopic currents evoked by steps to +40, +60, and +80 mV (Fig. 5D). The electrophysiological data are quantitatively consistent with the observation that Synpo coexpression increases the surface expression of Slo1 channels, at least in a heterologous expression system that does not express a complete complement of slit diaphragm proteins. Moreover, we observed that coexpression of Synpo did not affect the rate of Slo1VEDEC removal from the surface of HEK293T cells (Fig. 5E), which followed an approximately exponential time course with a time constant of 16–18 min whether or not Synpo was present. This suggests that the main effect of Synpo is on forward trafficking, rather than in regulating surface stability or endocytosis.
Fig. 4.
Coexpression of Synpo increases steady-state surface expression of Slo1 in HEK293T cells. A: representative cell surface biotinylation assay in HEK293T cells transiently expressing Myc-Slo1 and/or Flag-Synpo as indicated. Stimulatory effect of Synpo was blocked in cells treated with a membrane-permeant C3 transferase (C3T), a Rho inhibitor. Coexpression of Synpo did not affect total expression of Slo1 relative to actin. B: densitometric quantification of 4 repetitions of this experiment presented as means ± SE.
Fig. 5.
Coexpression of Synpo increases macroscopic currents through BKCa channels in HEK293T cells transiently expressing Slo1. A: representative traces showing families of whole cell currents evoked by depolarizing steps as indicated above the current traces. B: current-voltage diagram showing that currents were increased at all step potentials. In this and subsequent panels, numbers of cells in each group (n) are indicated. C: normalization of currents to the average evoked by steps to +80 mV (G/G80) reveals that coexpression of Synpo does not affect the voltage dependence of BKCa activation. D: Synpo coexpression does not affect BKCa activation kinetics. y-Axis shows mean time constant (τ) fitted to rising phase of macroscopic currents evoked by steps to +40, +60, and +80 mV. None of the differences is statistically significant (n.s.). E: time course of internalization of cell surface Slo1 proteins in HEK293T cells expressing Slo1 by itself (●) or coexpressing Synpo (■). Data points represent means ± SE of 4 repetitions of this experiment. OD492 is optical density at 492 nm. Superimposed curve shows exponential decay with a time constant of 16 h.
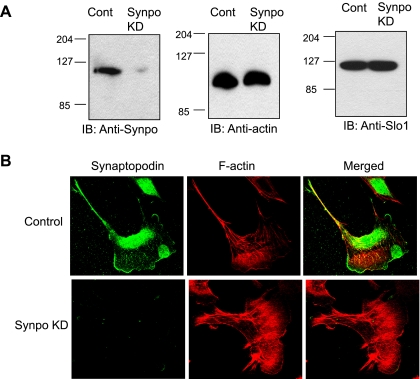
We next examined the role of Synpo in the steady-state expression of endogenous BKCa channels in mouse podocyte cell lines. To do this, we utilized an immortalized clonal podocyte cell line in which the Synpo gene was stably silenced (Synpo KD cells) (2). We confirmed the absence of Synpo expression in these cells by immunoblot analysis (Fig. 6A) and confocal microscopy (Fig. 6B). Total Slo1 expression in Synpo KD cells was indistinguishable from that in control cells (Fig. 6A). Under our growth conditions, staining for actin filaments revealed that stress fibers were present in both cell populations. The expression of functional cell surface BKCa channels was reduced in Synpo KD cells compared with control podocytes. This was ascertained by three different methods. We initially imaged surface expression by dual-label confocal microscopy using antibodies against Slo1 and Synpo in the two podocyte cell lines. In control podocytes (Fig. 7A) there was very close colocalization of Slo1 and Synpo, with especially strong signal concentrated in areas of membrane ruffling, as well as in perinuclear regions that include the endoplasmic reticulum. In Synpo KD cells Slo1 staining is very much less intense in the periphery of the cells, especially in areas of membrane ruffling, but is otherwise similar (Fig. 7B). Cell surface biotinylation assays confirmed that there is a marked reduction in cell surface Slo1 in Synpo KD podocytes compared with controls (Fig. 8A), which was confirmed by quantitative densitometric analyses of four repetitions of this experiment (Fig. 8B). However, total Slo1 was not different from that in controls. Finally, whole cell recordings indicated markedly reduced BKCa currents in Synpo KD podocytes (Fig. 8C), with mean current densities of about half those of controls (Fig. 8D). This effect was seen at all membrane potentials, and it is fully consistent with the reduction in surface expression seen by immunochemical techniques. Consistent with observations in HEK293T cells, Synpo knockdown had no effect on the kinetics of macroscopic BKCa channels in podocytes (data not shown).
Fig. 6.
Characteristics of podocyte cell lines. Controls are an immortalized mouse podocyte cell line, whereas Synpo KD is a similar line containing a stably incorporated small interfering RNA that targets Synpo expression. A: immunoblot analyses show that the Synpo KD line has markedly reduced expression of Synpo, but this does not affect expression of total actin or total Slo1. Cont, control. B: confocal images show that Synpo KD lines have almost complete loss of Synpo and that this is associated with a reduction in the number of stress fibers seen with rhodamine-phalloidin staining.
Fig. 7.
Synpo and Slo1 distribution in control and Synpo KD podocyte cell lines. A: in control cells, there is close colocalization between Slo1 and Synpo in intracellular compartments and around the periphery of the cells, especially in regions of membrane ruffling. B: Synpo cannot be detected in Synpo KD cell lines, and there is a marked reduction in Slo1 expression in the periphery of the cells.
Fig. 8.
Synpo knockdown reduces cell surface expression of podocyte BKCa channels. A: representative cell surface biotinylation assay showing marked reduction in surface Slo1 in Synpo KD cells. Con, control. B: densitometric quantification of 3 replications of the cell surface biotinylation assay. Bar graphs show means ± SE. C: representative whole cell recordings made with recording pipettes containing 5 μM free Ca2+. In previous studies we have shown that all of these currents are blocked by the BKCa inhibitor paxiline (18). Note lower-amplitude currents in the recording from the Synpo KD cell. D: means ± SE of currents evoked by depolarizing step to +60 mV in control and Synpo KD cells. Numbers of cells are indicated above bars. *P < 0.05 (Student's unpaired t-test).
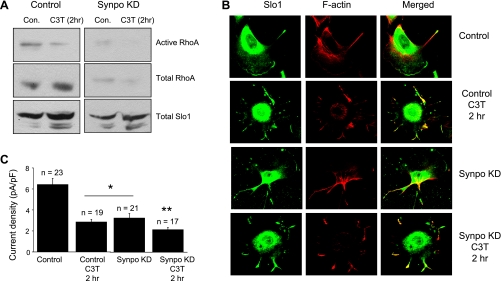
Synpo produces several distinct biochemical effects in podocytes that have consequences for regulation of cytoskeletal dynamics. One of these is to block ubiquitinylation and subsequent degradation of Rho GTPases in podocytes, thereby stabilizing stress fibers in these cells (3). Therefore we hypothesized that Rho stabilization underlies the effect of Synpo on cell surface expression of Slo1. This hypothesis predicts that Rho inhibition should produce an effect similar to Synpo on the functional surface expression of BKCa channels in podocytes. We tested this by treating differentiated podocytes with a commercially available cell-permeant C3 ADP-ribosyltransferase (C3T) for 2 h. This bacterial exoenzyme selectively modifies RhoA, RhoB, and RhoC at a conserved asparagine residue, thereby leading to inhibition of signaling through these pathways (1). We confirmed with a GST pull-down assay for Rho activation that treatment with C3T caused marked reduction in active Rho in control podocytes, and almost complete abolition of detectable active Rho in Synpo KD cells (Fig. 9A). In addition, treatment with C3T caused marked disruption of actin cytoskeleton that could be observed with phalloidin staining of podocytes (Fig. 9B). C3T also caused a >50% reduction in whole cell BKCa currents detected in podocytes (Fig. 9C). The effects of C3T on BKCa current, as with its effects on the amount of active Rho, appear additive with the effects of Synpo knockdown (Fig. 9C). We should note that 2 h of C3T treatment also reduced the stimulatory effect of Synpo on surface expression of BKCa channels in HEK293T cells (Fig. 4). To test whether this effect could be due to modulation of cytoskeleton, we observed that treatment with cytochalasin D (Cyto D) for 2 h caused a ∼50% in reduction in BKCa current density in podocytes whereas treatment with 15 min had no discernible effect on amplitude (Fig. 10A) or kinetics (not shown). As expected, Cyto D treatment for either 15 min or 2 h caused profound disruption of actin filaments as assessed by phalloidin staining (Fig. 10B).
Fig. 9.
Rho inhibition reduces surface expression of BKCa channels in podocytes. A: representative pull-down assay showing Rho activation in control and Synpo KD podocytes treated with vehicle (Con) or cell-permeant C3 transferase (C3T), an inhibitor of Rho. Note that CT3 causes reduction in total and active Rho in control and Synpo KD cells and results in almost complete loss of active Rho in Synpo KD cells. B: confocal images showing distribution of Slo1 and F-actin (rhodamine-phalloidin) in podocyte cell lines treated with vehicle or C3T. Note disruption of actin filaments and reduction in surface expression of Slo1 as a result of C3T. C: mean ± SE currents evoked by depolarizing steps to +60 mV in whole cell recordings from podocytes treated with vehicle or C3T for 2 h. As with analyses of active Rho, the effect of C3T on current amplitude is quantitatively similar to effect of Synpo knockdown. *P < 0.05 compared with control; **P < 0.05 compared with Synpo KD cells treated with vehicle (1-way ANOVA followed by Tukey's post hoc test).
Fig. 10.
Sustained disruption of actin filaments reduces macroscopic BKCa recorded from podocyte cell lines. A: mean ± SE currents in control podocytes and in podocytes treated with cytochalasin D (Cyto D) for 15 min or 2 h as indicated. Only the effect of 2 h of Cyto D is significantly different from control (1-way ANOVA followed by Tukey's post hoc test). B: Cyto D treatment for 15 min is sufficient to disrupt actin filament organization in podocytes.
DISCUSSION
In this study we have shown that Synpo interacts with endogenously expressed stretch-sensitive BKCa channels of podocytes and plays a role in regulating their steady-state expression on the cell surface. Synpo interactions do not appear to affect the gating properties of BKCa channels. Instead, Synpo regulates the trafficking of BKCa channels to the cell surface. Experiments in HEK293T cells indicate that Synpo does not require a complete complement of slit diaphragm-specific proteins in order to regulate BKCa trafficking, and they further suggest that at least part of the Synpo effect is caused by stabilization of Rho signaling systems. Indeed, the current density through BKCa channels in podocyte cell lines appears to be directly proportional to the amount of active Rho in the cells.
An important observation in this study is that an intact actin cytoskeleton is required for normal expression of BKCa channels on the podocyte cell surface, as disruption of actin filaments by Rho inhibition or Cyto D caused a marked reduction in the number of functional channels, at least after 2 h. However, a 15-min exposure to Cyto D, while sufficient to cause massive disruption of actin filaments, did not affect amplitudes of macroscopic currents through podocyte BKCa channels, which suggests that actin interactions are not a major regulator of BKCa gating in cultured podocytes. In several other cell types, depolymerization of actin filaments with Cyto D has been shown to facilitate the activation of cell surface BKCa channels (4, 9, 27, 28, 30, 34, 35) as a result of interactions between SH3-domain containing adaptor proteins such as Grb2 and cortactin and a proline-rich domain located in the flexible region between the two RCK domains (34, 35). These interactions with F-actin are required for stretch-sensitive gating in smooth muscle (30) and certain forms of neurohumoral modulation in BKCa channels (27, 28). Previous studies have shown that gating of BKCa channels in podocytes is also stretch sensitive (24). However, podocytes express a number of other proteins that could allow for outside-in signaling, and it is possible that stretch sensitivity arises as a result of interactions between BKCa channels and other potential mechanosensors such as nephrin (18) or podocin (14).
The role of F-actin in the regulation of BKCa trafficking in podocytes appears at first glance to be somewhat different from the role that it plays in neuronal systems. In parasympathetic neurons, we observed that depolymerization of F-actin with Cyto D leads to a rapid increase in surface expression of BKCa channels (5), whereas it initially has no effect, and over time suppresses surface expression of BKCa in podocytes. However, an important difference between neurons and cultured podocytes is that nearly all of the somatic F-actin in neurons lies in a cortical layer immediately subjacent to almost the entire plasma membrane, and stress fibers are not seen (5). By contrast, the cortical layer is much less developed in cultured podocytes and is only observed in a few patches of the surface, although a thin layer of highly branched filaments reminiscent of podosomes is present in normal podocyte foot processes in vivo (8, 11). Moreover, this layer becomes much more prominent in effaced podocytes (11). In neurons, cortical F-actin-bound BKCa represents a pool of intracellular channels that can be rapidly mobilized for translocation into the plasma membrane, much as is seen with GLUT4 transporters and other membrane proteins regulated by rapid changes in actin dynamics (15). By contrast, in podocytes, actin-bound pools of BKCa channels are more likely to be located farther from the cell surface, attached to stress fibers in cultured cells or to the highly ordered parallel actin fibers that run the length of foot processes in vivo. For this reason, disruption of actin filaments in podocytes is more likely to inhibit movement of BKCa channels over longer distances to the general vicinity of the plasma membrane, resulting in the loss of channels over a 2-h period that we observed here. In podocyte foot processes in vivo, actin-bound BKCa channels may be quite close to the cell surface, and that pool of channels may be regulated rather more like we have seen in neurons (5).
It is possible that Synpo regulates surface expression of BKCa channels by multiple mechanisms, especially its ability to protect Rho from ubiquitinylation and subsequent proteolytic degradation (3). However, Synpo also binds to an actin-binding site close to the COOH terminal that is essential for normal trafficking of BKCa channels to the cell surface (37), and addition of exogenous β-actin to a podocyte lysate reduces Synpo binding to BKCa channels. It is possible that Synpo plays a role in regulating binding and unbinding of intracellular pools of BKCa channels from actin, or at least certain pools of actin, during the process of trafficking. It is also possible that Synpo contributes to forward trafficking of BKCa channels in podocytes by sterically hindering access of regulatory proteins to the VEDEC motif in the COOH terminal that suppresses steady-state expression on the cell surface (6). Finally, it is possible that Synpo regulates interactions of BKCa channels with MAGI-1 (29), which we have previously shown to inhibit steady-state expression of BKCa channels on the podocyte cell surface (31).
In a previous study we demonstrated that BKCa channels interact with TRPC6 channels in podocytes (16), and it is possible that they traffic as a complex. In this regard, we have observed that Synpo also stimulates surface expression of TRPC6 channels in podocytes as part of a feedback loop that controls podocyte Ca2+ and actin dynamics in foot processes (unpublished data). The present data provide additional support that BKCa channels are a component of this same loop. TRPC6 channels are not ideal Ca2+ channels. Moderate membrane depolarization causes them to exclude divalent cations, although they continue to carry substantial current via monovalent cations (10). Instead, Ca2+ tends to block TRPC6 pores at more positive potentials (10, 33), and for TRPC6 channels to be an efficient source of Ca2+ influx, there may need to be a mechanism to limit the amount of depolarization that would otherwise occur as a result of their own activation (10). Coordinated activation of BKCa channels could play this role by maintaining the driving force for Ca2+ entry and preventing pore blockade by Ca2+, and by providing a mechanosensory element to the overall signaling dynamics in podocytes.
In summary, we have shown that mechanosensitive BKCa channels or podocytes are regulated by Synpo, Rho, and actin microfilaments, and these interactions may be part of a network that maintains the stability of the glomerular filtration barrier.
GRANTS
This work was supported by National Institute of Diabetes and Digestive and Kidney Diseases Grant 1RO1-DK-82529 to S. E. Dryer.
DISCLOSURES
No conflicts of interest, financial or otherwise, are declared by the author(s).
ACKNOWLEDGMENTS
We are grateful to Dr. Peter Mundel of the University of Miami for helpful suggestions on experimental designs and for supplying podocyte cell lines and Synpo constructs.
Footnotes
Supplemental Material for this article is available online at the Journal website.
REFERENCES
- 1.Aktories K, Wilde C, Vogelsgesang M. Rho-modifying C3-like ADP-ribosyltransferases. Rev Physiol Biochem Pharmacol 152: 1–22, 2004 [DOI] [PubMed] [Google Scholar]
- 2.Asanuma K, Kim K, Oh J, Giardino L, Chabanis S, Faul C, Reiser J, Mundel P. Synaptopodin regulates the actin-bundling activity of alpha-actinin in an isoform-specific manner. J Clin Invest 115: 1188–1198, 2005 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Asanuma K, Yanagida-Asanuma E, Faul C, Tomino Y, Kim K, Mundel P. Synaptopodin orchestrates actin organization and cell motility via regulation of RhoA signalling. Nat Cell Biol 8: 485–491, 2006 [DOI] [PubMed] [Google Scholar]
- 4.Brainard AM, Miller AJ, Martens JR, England SK. Maxi-K channels localize to caveolae in human myometrium: a role for an actin-channel-caveolin complex in the regulation of myometrial smooth muscle K+ current. Am J Physiol Cell Physiol 289: C49–C57, 2005 [DOI] [PubMed] [Google Scholar]
- 5.Chae KS, Dryer SE. The p38 mitogen-activated protein kinase pathway negatively regulates Ca2+-activated K+ channel trafficking in developing parasympathetic neurons. J Neurochem 94: 367–379, 2005 [DOI] [PubMed] [Google Scholar]
- 6.Chiu YH, Alvarez-Baron C, Kim EY, Dryer SE. Dominant-negative regulation of cell surface expression by a pentapeptide motif at the extreme COOH terminus of a Slo1 calcium-activated potassium channel splice variant. Mol Pharmacol 77: 497–507, 2010 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Cupples WA, Braam B. Assessment of renal autoregulation. Am J Physiol Renal Physiol 292: F1105–F1123, 2007 [DOI] [PubMed] [Google Scholar]
- 8.Drenckhahn D, Franke RP. Ultrastructural organization of contractile and cytoskeletal proteins in glomerular podocytes of chicken, rat, and man. Lab Invest 59: 673–682, 1988 [PubMed] [Google Scholar]
- 9.Ehrhardt AG, Frankish N, Isenberg G. A large-conductance K+ channel that is inhibited by the cytoskeleton in the smooth muscle cell line DDT1 MF-2. J Physiol 496: 663–676, 1996 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Estacion M, Sinkins WG, Jones SW, Applegate MA, Schilling WP. Human TRPC6 expressed in HEK 293 cells forms non-selective cation channels with limited Ca2+ permeability. J Physiol 572: 359–377, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Faul C, Asanuma K, Yanagida-Asanuma E, Kim K, Mundel P. Actin up: regulation of podocyte structure and function by components of the actin cytoskeleton. Trends Cell Biol 17: 428–437, 2007 [DOI] [PubMed] [Google Scholar]
- 12.Friedrich C, Endlich N, Kriz W, Endlich K. Podocytes are sensitive to fluid shear stress in vitro. Am J Physiol Renal Physiol 291: F856–F865, 2006 [DOI] [PubMed] [Google Scholar]
- 13.Huber TB, Kwoh C, Wu H, Asanuma K, Gödel M, Hartleben B, Blumer KJ, Miner JH, Mundel P, Shaw AS. Bigenic mouse models of focal segmental glomerulosclerosis involving pairwise interaction of CD2AP, Fyn, and synaptopodin. J Clin Invest 116: 1337–1345, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Huber TB, Schermer B, Müller RU, Höhne M, Bartram M, Calixto A, Hagmann H, Reinhardt C, Koos F, Kunzelmann K, Shirokova E, Krautwurst D, Harteneck C, Simons M, Pavenstädt H, Kerjaschki D, Thiele C, Walz G, Chalfie M, Benzing T. Podocin and MEC-2 bind cholesterol to regulate the activity of associated ion channels. Proc Natl Acad Sci USA 103: 17079–17086, 2006 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Kanzaki M, Pessin JE. Insulin-stimulated GLUT4 translocation in adipocytes is dependent upon cortical actin remodeling. J Biol Chem 276: 42436–42444, 2001 [DOI] [PubMed] [Google Scholar]
- 16.Kim EY, Alvarez-Baron CP, Dryer SE. Canonical transient receptor potential channel (TRPC)3 and TRPC6 associate with large-conductance Ca2+-activated K+ (BKCa) channels: role in BKCa trafficking to the surface of cultured podocytes. Mol Pharmacol 75: 466–477, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kim EY, Chiu YH, Dryer SE. Neph1 regulates steady-state surface expression of Slo1 Ca2+-activated K+ channels: different effects in embryonic neurons and podocytes. Am J Physiol Cell Physiol 297: C1379–C1388, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Kim EY, Choi KJ, Dryer SE. Nephrin binds to the COOH terminus of a large-conductance Ca2+-activated K+ channel isoform and regulates its expression on the cell surface. Am J Physiol Renal Physiol 295: F235–F246, 2008 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Kim EY, Ridgway LD, Zou S, Chiu YH, Dryer SE. Alternatively spliced C-terminal domains regulate the surface expression of large conductance calcium-activated potassium channels. Neuroscience 146: 1652–1661, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Kim EY, Zou S, Ridgway LD, Dryer SE. Beta1-subunits increase surface expression of a large-conductance Ca2+-activated K+ channel isoform. J Neurophysiol 97: 3508–3516, 2007 [DOI] [PubMed] [Google Scholar]
- 21.Kremerskothen J, Plaas C, Kindler S, Frotscher M, Barnekow A. Synaptopodin, a molecule involved in the formation of the dendritic spine apparatus, is a dual actin/alpha-actinin binding protein. J Neurochem 92: 597–606, 2005 [DOI] [PubMed] [Google Scholar]
- 22.Kriz W, Elger M, Mundel P, Lemley KV. Structure-stabilizing forces in the glomerular tuft. J Am Soc Nephrol 5: 1731–1739, 1995 [DOI] [PubMed] [Google Scholar]
- 23.Ma D, Nakata T, Zhang G, Hoshi T, Li M, Shikano S. Differential trafficking of carboxyl isoforms of Ca2+-gated (Slo1) potassium channels. FEBS Lett 581: 1000–1008, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Morton MJ, Hutchinson K, Mathieson PW, Witherden IR, Saleem MA, Hunter M. Human podocytes possess a stretch-sensitive, Ca2+-activated K+ channel: potential implications for the control of glomerular filtration. J Am Soc Nephrol 15: 2981–2987, 2004 [DOI] [PubMed] [Google Scholar]
- 25.Mundel P, Heid HW, Mundel TM, Krüger M, Reiser J, Kriz W. Synaptopodin: an actin-associated protein in telencephalic dendrites and renal podocytes. J Cell Biol 139: 193–204, 1997 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 26.Mundel P, Reiser J, Zúñiga Mejía Borja A, Pavenstädt H, Davidson GR, Kriz W, Zeller R. Rearrangements of the cytoskeleton and cell contacts induce process formation during differentiation of conditionally immortalized mouse podocyte cell lines. Exp Cell Res 236: 248–258, 1997 [DOI] [PubMed] [Google Scholar]
- 27.O'Malley D, Harvey J. MAPK-dependent actin cytoskeletal reorganization underlies BK channel activation by insulin. Eur J Neurosci 25: 673–682, 2007 [DOI] [PubMed] [Google Scholar]
- 28.O'Malley D, Irving AJ, Harvey J. Leptin-induced dynamic alterations in the actin cytoskeleton mediate the activation and synaptic clustering of BK channels. FASEB J 19: 1917–1919, 2005 [DOI] [PubMed] [Google Scholar]
- 29.Patrie KM, Drescher AJ, Welihinda A, Mundel P, Margolis B. Interaction of two actin-binding proteins, synaptopodin and alpha-actinin-4, with the tight junction protein MAGI-1. J Biol Chem 277: 30183–30190, 2002 [DOI] [PubMed] [Google Scholar]
- 30.Piao L, Ho WK, Earm YE. Actin filaments regulate the stretch sensitivity of large-conductance, Ca2+-activated K+ channels in coronary artery smooth muscle cells. Pflügers Arch 446: 523–528, 2003 [DOI] [PubMed] [Google Scholar]
- 31.Ridgway LD, Kim EY, Dryer SE. MAGI-1 interacts with Slo1 channel proteins and suppresses Slo1 expression on the cell surface. Am J Physiol Cell Physiol 297: C55–C65, 2009 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Scita G, Confalonieri S, Lappalainen P, Suetsugu S. IRSp53: crossing the road of membrane and actin dynamics in the formation of membrane protrusions. Trends Cell Biol 18: 52–60, 2008 [DOI] [PubMed] [Google Scholar]
- 33.Shi J, Mori E, Mori Y, Mori M, Li J, Ito Y, Inoue R. Multiple regulation by calcium of murine homologues of transient receptor potential proteins TRPC6 and TRPC7 expressed in HEK293 cells. J Physiol 561: 415–432, 2004 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Tian L, Chen L, McClafferty H, Sailer CA, Ruth P, Knaus HG, Shipston MJ. A noncanonical SH3 domain binding motif links BK channels to the actin cytoskeleton via the SH3 adapter cortactin. FASEB J 20: 2588–2590, 2006 [DOI] [PubMed] [Google Scholar]
- 35.Tian L, McClafferty H, Chen L, Shipston MJ. Reversible tyrosine protein phosphorylation regulates large conductance voltage- and calcium-activated potassium channels via cortactin. J Biol Chem 283: 3067–3076, 2008 [DOI] [PubMed] [Google Scholar]
- 36.Yanagida-Asanuma E, Asanuma K, Kim K, Donnelly M, Young Choi H, Hyung Chang J, Suetsugu S, Tomino Y, Takenawa T, Faul C, Mundel P. Synaptopodin protects against proteinuria by disrupting Cdc42:IRSp53:Mena signaling complexes in kidney podocytes. Am J Pathol 171: 415–427, 2007 [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Zou S, Jha S, Kim EY, Dryer SE. A novel actin-binding domain on Slo1 calcium-activated potassium channels is necessary for their expression in the plasma membrane. Mol Pharmacol 73: 359–368, 2008 [DOI] [PubMed] [Google Scholar]
- 38.Zou S, Jha S, Kim EY, Dryer SE. The β1 subunit of L-type voltage-gated Ca2+ channels independently binds to and inhibits the gating of large-conductance Ca2+-activated K+ channels. Mol Pharmacol 73: 369–378, 2008 [DOI] [PubMed] [Google Scholar]