Abstract
As part of a program to discover drug leads from plant biodiversity, the present investigation was undertaken to explore the anticancer potential of compounds derived from selected Latin American plants. Bioassay-guided fractionation of a crude extract of the aerial parts of Vassobia breviflora led to the isolation of the withanolide-type steroidal lactone, withaferin A (1). This compound was tested for antiproliferative activity against the head and neck squamous cell carcinoma (HNSCC) cell lines, MDA1986, JMAR, UM-SCC-2, and JHU011. The inhibitory concentrations to reduce cell viability to 50% (IC50) were determined by the MTS cytotoxicity assay and 1 reduced cell viability with IC50 values in the range 0.5–2.2 μM. A mechanistic study showed that 1 induces apoptosis and cell death in HNSCC cells as well as a cell-cycle shift from G0/G1 to G2/M. Cells treated with 1 exhibited inactivation of Akt and the reduction in total Akt concentration. This investigation constitutes the first report of the antiproliferative activity of withaferin A (1) against head and neck squamous carcinoma.
Head and neck squamous cell carcinoma (HNSCC) is the fifth most common cancer in humans and accounts for 90% of all head and neck malignancies, with over 780,000 new cases diagnosed yearly worldwide.1–3 Tumors in this disease may arise from any of the mucosal surfaces in the head and neck region including the lips and oral cavity, tongue, nasal cavity, oropharynx, larynx, and even the thyroid gland. Tumors typically will spread lymphatically to cervical lymph nodes in the neck. Platinum-based therapy is the most effective drug treatment for HNSCC; however, this treatment may lead to significant toxicity, including ototoxicity, peripheral neuropathy, and nephrotoxicity accounting for up to 15% of deaths.4 Despite advances in the diagnosis and staging of advanced HNSCC, survival has not significantly improved in the last 30 years,3 and even with aggressive therapy, the majority of such patients succumb to recurrent or metastatic disease.5–7 Many patients, especially the elderly, cannot complete chemo-radiation treatments and a significant number of deaths are due to treatment-related toxicity.4 Molecular-targeted therapies are being explored, but thus far have not shown the level of benefit in HNSCC that they produce in other forms of cancer.8 Novel drugs discovered for this disease, especially from natural product sources, would provide an exciting avenue for preclinical exploration in HNSCC.
Natural products have played a significant role in the discovery and development of new anticancer agents and they represent a rich source of biologically active compounds. More than 79.8% of anticancer drugs introduced from January 1981 to October 2008 were natural products, semi-synthetic analogues, or synthetic compounds based on natural-product pharmacophores.9 A library of 200 plant extracts from Latin America was evaluated for their cytotoxic activities against four HNSCC cell lines using the MTS assay. The species selected for this study included one of the most active samples from Argentina, Vassobia breviflora (Sendtn.) Hunz (Solanaceae), which is a widespread spiny shrub with small, purple, campanulate, glabrous flowers.10 Although significant antimicrobial activity of the methanol extract of V. breviflora was reported previously,11 there has been no chemical isolation work on this species reported previously. Cytotoxicity-guided purification led to the isolation and characterization of the active withanolide, withaferin A (1).
Withaferin A (1) is a part of the withanolide family of compounds. Withanolides are naturally occurring C28-steroidal lactones built on an intact or rearranged ergostane scaffold in which C-22 and C-26 are appropriately oxidized for forming a δ-lactone ring on the nine-carbon side chain.12 These polyhydroxy steroids are found in some species of the Solanaceae, and in particular, they are abundant and structurally diverse in the genera Acnistus, Datura, Dunalia, Lycium, Jaborosa, Physalis, and especially Withania.12,13 Withanolides are reported to possess various biological activities in vitro and in vivo including antimicrobial,14 antineoplastic and cytotoxic,15–21 anti-neurodegenerative,20 anti-inflammatory,22–25 radiosensitizing,26 as well as immunomodulating, antioxidant, anti-stress, cardiotonic, and insect antifeedant properties.13,26
Compound 1 was isolated earlier from Withania somnifera Dunal, a medicinal plant used in Ayurvedic medicine for the treatment of various ailments such as arthritis, asthma, hypertension, and rheumatism. Its structure was elucidated in 1965, and is characterized by an α,β-unsaturated ketone in ring A, an epoxide group in ring B, and an α,β-unsaturated lactone in its side chain.12 Withaferin A (1) has been the object of many studies and considered as a potential agent for use in cancer therapy because of its potential antitumor activity.13 This compound is reported to show cytotoxicity in a variety of tumor cells including breast,15 pancreatic,17 prostate,19 lung,27 and leukemia.28
Although recent studies have demonstrated that 1 suppresses the growth of human cancer cell lines in vitro and in vivo by causing apoptosis,15–17 the precise mechanism, including the signals and the pathways by which 1 induces cell death, is not fully understood. The present study was undertaken to assess for the first time the efficacy of 1 against several malignant head and neck squamous cell carcinoma cell lines and to broaden the understanding of its mechanism of action in these cells.
Results and Discussion
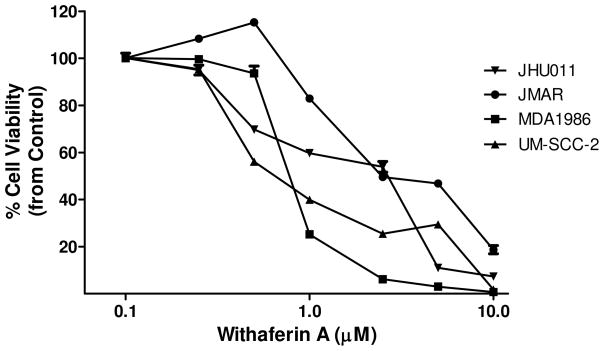
The isolation process of 1 from V. breviflora was guided by bioassay, and, at each stage of purification, fractions obtained were screened for their cytotoxic activities for JMAR, MDA1986, UM-SCC-2, and JHU011 human HNSCC cells using the MTS assay. The half-maximal inhibitory concentration (IC50) of the most active fraction at each step of the isolation is summarized in Table S1 (Supporting Information). The concentration-dependent cell viabilities for application of 1 to the head and neck squamous cell carcinoma are shown in Figure 1. Withaferin A reduced cell viability of HNSCC cell lines and inhibited their growth in a concentration-dependent manner. The IC50 values for UM-SCC-2, MDA1986, JMAR, and JHU011 cells were 0.5, 0.8, 2.0, and 2.2 μM, respectively. The results showed that these four cell lines are very sensitive to treatment with compound 1.
Figure 1.
Withaferin A (1) is an antiproliferative agent for the JHU011, JMAR, MDA1986, and UM-SCC-2 head and neck squamous cell lines. The MTS assay was used to study the effect of 1 on cell viability of head and neck squamous carcinoma cells. All experiments were performed in triplicate for each concentration.
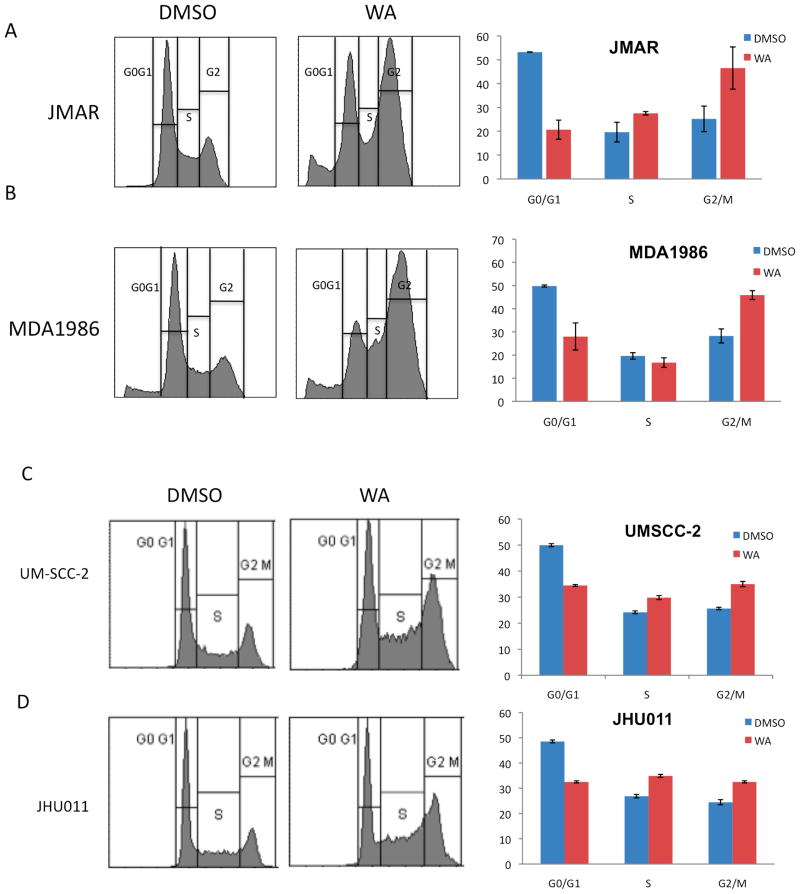
A series of experiments evaluating cell-cycle arrest and the apoptotic pathway was performed in order to investigate a possible mechanism for the antiproliferative effect of 1. As shown in Figure 2, treatment of MDA1986, JMAR, UM-SCC-2, and JHU011 HNSCC cells with 3.0 μM 1 for 24 h induced a significant cell cycle shift from G0/G1 to G2/M phase, from 27% to 42% in JMAR cells (Figure 2A) and from 26% to 49% in MDA1986 cells (Figure 2B; p <0.01). The G2/M phase shift for UM-SCC-2 cells was from 27% to 36% (Figure 2C) and for JHU011 cells, 25% to 34% (Figure 2D, p <0.01). Compound 1 additionally induced S phase arrest in JMAR, MDA1986, and UM-SCC-2 cells. Treatment with 1 also led to an increase in sub-G0 levels in both JMAR (from 2% to 12%; p <0.01) and MDA1986 cells (from 7% to 8%; not significant), suggesting that JMAR cells underwent DNA fragmentation, one of the early biochemical events leading to apoptosis.
Figure 2.
Compound 1 induced G2/M cell cycle arrest in HNSCC cells. JMAR (A) MDA1986 (B), UM-SCC-2 (C), and JHU011 (D) cells were treated with DMSO or 3.0 μM 1. Cells treated with vehicle (DMSO) were primarily at the G0/G1 phase, while cells treated with compound 1 showed shifts from the G0/G1 to the S and G2/M phases.
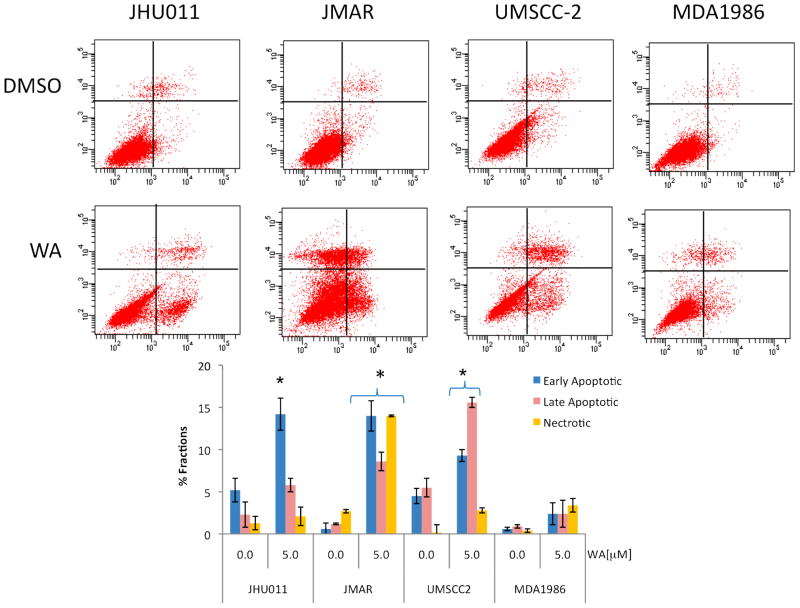
In order to confirm the apoptotic action induced by 1, cells were treated with 5.0 μM of 1 for 24 h and stained with annexin V/propidium iodide (PI). As shown in Figure 3, treatment of HNSCC cells with 1 increased the annexin V-positive cells. Annexin V only staining signified the early apoptotic events whereas dual annexin V and PI staining indicated the late phase of apoptosis. Treatment of JHU011 cells with 1 induced early apoptotic events (p <0.01) significantly from basal levels of 4.8% to 14.8%. There was a slight increase in the late phase of apoptosis induced by 1 in JHU011 cells, with very small PI only staining (necrosis) observed in JHU011 cells treated with 1. Treatment of JMAR and UM-SCC-2 cells with 1 induced significant increases in early and late apoptosis as well as necrosis (p <0.01). MDA1986 cells seemed less sensitive to treatment with 1, since withaferin A did not increase annexin V/PI staining herein.
Figure 3.
Withaferin A (1) induced apoptosis in head and neck squamous cell carcinoma (HNSCC) cells. HNSCC cells were treated with 5.0 μM of 1 for 24 h and then harvested and stained with annexin V and propidium iodide followed by analysis by flow cytometry. All experiments were performed in triplicate. Treatment with 1 exhibited an increase in annexin V and PI staining that indicated early as well as late apoptotic events. JMAR cells treated with 1 showed significant necrosis.
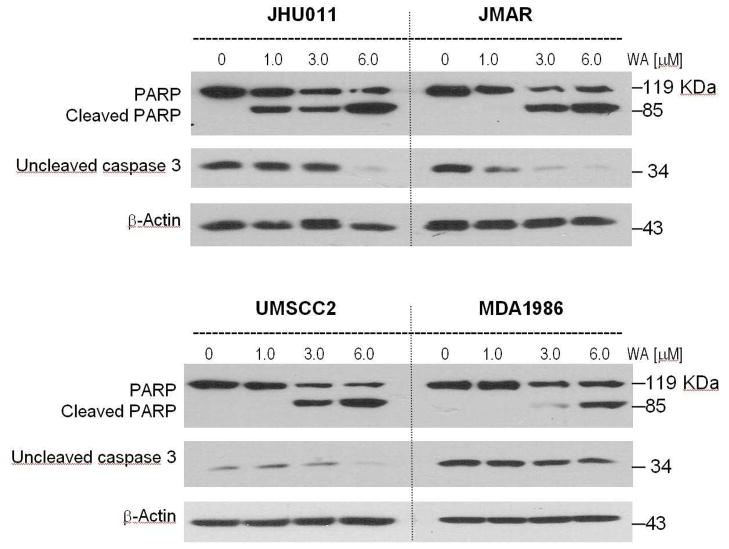
To confirm the annexin V studies on HNSCC cells, caspase 3 activation was investigated in HNSCC (Figure 4). Treatment of JHU011 cells induced activation of the caspase 3 enzyme, as signified by decreased procaspase 3 levels. In addition, the caspase 3 substrate PARP was cleaved upon treatment with 1, in a concentration-dependent manner (Figure 4). Treatment with compound 1 at 1.0 μM increased PARP cleavage in JHU011 cells. JMAR cells treated with 1 also showed similar decreases in procaspase 3 levels and cleavage of PARP protein in a concentration-dependent fashion. Similar results were also observed when UM-SCC-2 cells were treated with 1. Treatment of MDA1986 cells with withaferin A increased caspase 3 activation and PARP cleavage. However, in line with the annexin V/PI data, a slightly higher concentration of 1 was required to induce caspase 3 activation in MDA1986 cells (Figure 4). These results indicate that 1 induced cell-cycle arrest at the G2/M phase and apoptosis was confirmed by positive annexin V/PI staining as well as caspase 3 activation in the HNSCC cell lines investigated. At this point, it is not clear whether G2/M cell cycle arrest provides conditions conducive of caspase 3 activation.
Figure 4.
Caspase 3 was activated in head and neck squamous cell carcinoma cells treated with withaferin A (1). HNSCC cells was treated with different concentrations of 1 for 24 h. Levels of pro-caspase 3 (inactive form) was examined with the antibody against uncleaved caspase 3. In addition, activation of caspase 3 was examined with levels of the uncleaved and cleaved caspase 3 substrate, PARP. Compound 1 induced caspase 3 activation in all HNSCC cell lines in a concentration-dependent manner.
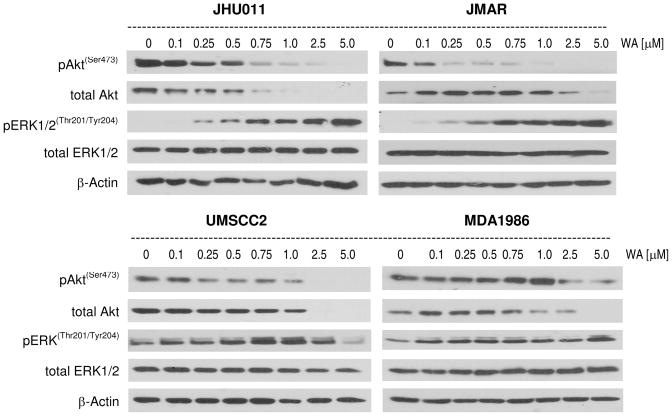
The effect of withaferin A (1) was examined on major prosurvival signaling pathways in HNSCC cells including PI3 kinase/Akt and ERK1/2 MAP kinase. These data showed that 1 reduced phosphorylation (ser473) as well as total Akt levels in all HNSCC cells in a concentration-dependent manner (Figure 5). Treatment of JHU011, JMAR, and UM-SCC-2 cells with as low as 100–250 nM concentrations of 1 reduced Akt phosphorylation (ser473). It is interesting to note that a higher concentration of 1 was required to inhibit Akt activity in MDA1986 cells (2.5 μM). In addition to the effect of 1 on the reduction of phosphorylated Akt, the total Akt levels were decreased significantly in all cell lines examined (Figure 5). These data show that 1 is a strong Akt inhibitor in HNSCC cells. The effect of 1 was examined on ERK1/2 activation in HNSCC cells. Withaferin A increased phosphorylation of ERK1/2 MAP kinase in JHU011, JMAR, and MDA1986 cells. Only at higher concentrations did 1 inhibit activation of ERK1/2 in UM-SCC-2 cells. Withaferin A had no effect on total ERK1/2 levels.
Figure 5.
Compound 1 modulated prosurvival signal transduction pathway. HNSCC cells were treated with increasing concentrations of 1 for 24 h. Total levels of phospho-Akt, total Akt, phospho-ERK and total ERK2 were determined by Western blotting. Antibody against β-actin was used as a protein loading control. Compound 1 reduced Akt activation as well as total cellular Akt, while ERK1/2 was activated in JHU011, JMAR, and MDA1986 cells while ERK1/2 activity decreased in UM-SCC-2 cells at a high concentration of 1. Total ERK levels remained unchanged when cells were treated with compound 1.
In the present study, a plant extract as well as purified 1 from V. breviflora potently decreased cell viability of head and neck squamous carcinoma cell lines MDA1986, JMAR, UM-SCC-2, and JHU011 cells using the MTS assay. The IC50 concentration of this inhibitor ranged from 0.5 to 2.2 μM among the HNSCC cell lines used, indicating a potent antiproliferative effect. Compound 1 reduced cell viability at sub-micromolar concentrations in UM-SCC-2, and MDA1986 cells. However, the head and neck squamous cell lines used displayed differential sensitivities to 1, with UM-SCC-2 cells demonstrating a significantly higher sensitivity to 1 than the other cell lines. Compound 1 induced a shift in cell cycle arrest from G0/G1 arrest to the S and G2/M phase. In addition, cell cycle arrest studies exhibited a significant increase in the subG0 fraction indicating a likely early-DNA fragmentation event due to treatment with 1. Moreover, flow cytometry analysis of annexin V/PI staining in treated cells in Figure 3 showed that 1 induced a significant level of apoptosis in these cells. Western blot analysis studies of activation caspase 3 confirmed the annexin V/PI data. Cell cycle arrest at mitosis seem to be greatest in JMAR cells with a correspondingly lower requirement for 1 for induction of caspase 3 activation and PARP cleavage, whereas MDA1986 cells exhibited a relatively smaller G2/M shift and showed relative resistance to caspase 3 activation. In addition, western blotting data demonstrated that the antiproliferative action of 1 could be in part explained through a decrease in activation of Akt as well as the degradation of total Akt levels. Compound 1 at sub-micromolar concentrations reduced Akt phosphorylation (ser473) in JMAR, JHU011, and UM-SCC-2 cells whereas 1 at slightly higher than 1.0 μM was required for inhibition of Akt phosphorylation in MDA1986 cells. Further study is required to elucidate the upstream effect of 1 in the PI3 kinase/Akt pathway as well as downstream events that may induce apoptosis upon Akt inhibition in HNSCC cells. In addition, Akt may phosphorylate and inhibit caspase activation and this may include initiator caspases (for example caspase -2, -8, -9, and -10) which in turn, proteolytically cleave and activate effector (executioner) caspases (for example, caspase -3, -6, and -7).29 Therefore, HNSCC cells (for example MDA1986) that are relatively resistant to withaferin A-mediated Akt inhibition may therefore require higher levels of 1 to inhibit Akt and in turn activate caspases.
In conclusion, this is the first report to date of the presence of the withaferin A in Vassobia breviflora and this species has been demonstrated as a new source for the production of 1. Extracts, as well as pure 1, reduced cell viability in all four head and neck squamous carcinoma cell lines in vitro. This mechanism of action is in part a result of G2/M cell cycle arrest and induction of apoptosis in HNSCC cells. Compound 1 inhibited activation of Akt and reduced total Akt levels. Additional studies will be required to evaluate the potential of application of the active compound 1 for future anticancer drug development in head and neck squamous carcinoma.
Experimental Section
General Experimental Procedures
HPLC was conducted on Agilent Technologies 1200 series system. IR data were obtained using a Thermo Nicolet Avatar 360 FT-IR instrument, NMR spectra were recorded on either a Bruker DRX-400 with a qnp probe or on a Bruder AV-500 with a cryoprobe. 1H and 13C spectra were recorded using the residual protonated signal in the CDCl3 solvent (δH 7.24) or the central peak of the CDCl3 triplet (δC 77.00) as the internal standard. High resolution mass spectrometry data were collected on a LCT Premier time of flight mass spectrometer (Waters Corp., Milford, MA). Normal phase TLC was performed on Sorbent Technologies Silica G TLC plates (200 μm, w/UV 254) using the solvent system DCM-EtOAc-MeOH (1:8:1), and reverse phase TLC was performed on Sorbent Technologies C18 TLC plates (150 μm, w/UV 254) using H2O-MeOH (1:1). Spots were visualized using UV light (254 nm) and spraying with vanillin-sulfuric acid reagent.
Plant Material
Aerial parts of V. breviflora were collected by R. Fortunato and A. Cabral along highway RN 86, 2 km northeast of Primavera, Department Patino, Province of Formosa, Argentina (lat. 25°14′0″S, long. 57°57′0″W) in November 1999. A voucher specimen (No. ARP 612) prepared by R. Fortunato was deposited in the Herbarium (BAB) of the Institute of Biological Resources, National Institute of Agricultural Technology (INTA), Castelar, Buenos Aires, Argentina. Intellectual Property Rights Agreements for plant collections and interdisciplinary research were executed between the collaborating institutions in the U.S. and Argentina.
Extraction and Isolation
Dried, ground biomass (100 g) was extracted at room temperature with CH2Cl2-MeOH (50:50) three times. The concentrated extract (9.8 g) was dissolved in MeOH-H2O (10:90) followed by partition with n-hexane, EtOAc, and n-butanol. The EtOAc extract (2.65 g) was subjected to silica gel column chromatography (32–60 μm, 5 × 60 cm) and eluted with gradient mixtures of CH2Cl2-MeOH (100:0, 98:2, 95:5, 93:7, 90:10, 80:20, 50:50 and 0:100), in order of increasing polarity. A total of 182 fractions were collected and fractions 49–54 (830.3 mg; CH2Cl2-MeOH 93:7) were combined and further purified by normal-phase SPE (20 g; 60 mL) using a step gradient of CH2Cl2-MeOH mixtures (90:10, 85:15, 80:20, 75:25, 70:30, 65:35, 60:40, 50:50, and 0:100). Among nine collected fractions, 50 mg withaferin A (1) was obtained in the most active fraction 5 (CH2Cl2-MeOH 70:30). The identity of withaferin A (1; 94.8% purity by HPLC) was determined by a combination of 1D and 2D NMR, HR-MS, IR, and X-ray diffraction data, which were in accordance with values reported in the literature,30,31 and co-chromatography done with an authentic sample purchased from Chromadex (Irvine, CA; see Supporting Information). Withaferin A (1) was soluble in chloroform but not in ethyl acetate. Colorless block-shape crystals of 1 were obtained from a mixture of chloroform and ethyl acetate (ratio 1:2). The purity of the final 1 was determined to be 95% by HPLC analysis according to method previously reported.13
Bioassay Materials
Culture media, fetal bovine serum (FBS), penicillin G, streptomycin, MEM-nonessential amino acids, ribonuclease A, and propidium iodide (PI) were obtained from Sigma-Aldrich (St. Louis, MO). MEM-vitamin solution was purchased from Life Technologies, Inc. (Grand Island, NY). MTS reagents were obtained from Promega Co (Madison, WI). Annexin V-FITC was from BD Bioscience (Bedford, MA). Primary antibodies against total ERK and β-actin and secondary antibodies against mouse and rabbit antibodies were purchased from Santa Cruz Biotechnology (Santa Cruz, CA). Primary antibodies against phospho-Akt, total-Akt, phospho-ERK, caspase 3 and PARP were obtained from Cell Signaling Technologies (Beverly, MA). BCA protein assay reagents were obtained from Pierce (Rockford, IL). Protease inhibitor mixture set II was obtained from Calbiochem (San Diego, CA),
Cell Culture and Media
The invasive oral squamous carcinoma cell lines, JMAR and MDA1986, were a gift from Dr. Jeffrey Myers (University of Texas, M.D. Anderson Cancer Center; Houston, TX). The head and neck squamous carcinoma cell line, UM-SCC-2, was a gift from Dr. Scott Weed (University of West Virginia, Morgantown, WV). They were grown in DMEM supplemented with 10% FBS, sodium pyruvate, nonessential amino acids, L-glutamine, a two-fold MEM-vitamin solution, and 1% penicillin/streptomycin. The laryngeal squamous carcinoma cell line, JHU-011, was kindly donated by Dr. Joseph Califano (Johns Hopkins University, Baltimore, MD). This was cultured in RPMI-1640, supplemented with 10% FBS and 1% penicillin/streptomycin. Adherent monolayer cultures were maintained on plastic plates and incubated at 37 °C with 5% CO2.
Cytotoxicity Assay
The experiment was performed by the MTS [3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H tetrazolium] assay according to the manufacturer’s instructions. In brief, 1 was dissolved in dimethylsulfoxide (DMSO) and then diluted with culture media to prepare serial concentrations while the controls received only DMSO (<0.1%, v/v). The cells were seeded in 96-well microtiter plates (2×103 cells/well) in 100 μL of growth medium. After incubation for 24 h, varying levels of plant extracts or 1 were added to each well, and incubated for 72h. Then, 20 μL MTS solution were added. After 3 h of incubation at 37 °C (5% CO2), absorbance was measured at 490 nm with a microplate reader. Percent viability was calculated as a fraction of the negative control (culture medium only), with data plotted as a function of % cell viability vs. concentration of 1. The half-maximal inhibitory concentration (IC50) was obtained from the MTS viability curves using GraphPad Prism 5. All studies were performed in triplicate. In parallel with wells of the test samples, negative control wells with predetermined cell numbers were used to normalize the absorbance readings and to provide internal confirmation that the assay was linear over the range of absorbance and cell numbers measured. Cisplatin at various concentrations was used as a positive control in the MTS cytotoxicity assay.
Western Blot Analysis
After treatment cells were lysed (0.5% Nonidet P-40, 100 mM, 10 mM Tris (pH 7.5), 1:500 protease inhibitor mixture set II, 1 mM NaF, and 1 mM sodium orthovanadate) and lysates were placed on ice for 20 min. Clear lysates were obtained by centrifugation (14,000 × g for 20 min). Equal amounts of protein were separated by SDS-PAGE and electro-transferred onto a Hybond nitrocellulose membrane (Amersham). The membranes were blocked and probed with the appropriate dilution of primary antibody overnight at 4 °C. The blots were washed three times in PBS-Tween-20 for 10 min, and then incubated in horseradish peroxidase-conjugated secondary antibody in PBS-Tween-20 at room temperature for 1 h. After washing in PBS-Tween-20, the proteins were visualized by enhanced chemiluminescence reagent (Amersham) and captured on Kodak XAR-5 film (Eastman Kodak, Rochester, NY). Where indicated, the blots were re-probed with antibody against β-actin to ensure equal loading and transfer of proteins.
Cell-Cycle Arrest Study
After treatment, cells were trypsinized and 1×106 cells were washed with 0.9% NaCl and fixed with 70% cold ethanol in 0.9% NaCl for 30 min at room temperature. After centrifugation (700 × g; 5 min) cells were stained with propidium iodide (50 μg/mL in PBS) for 30 min. Then, cells were treated with DNAse free RNAse (1 mg/mL) for 30 min and analyzed by a Becton Dickinson LSRII Flow Cytometer.
Apoptosis Analysis
In order to evaluate the induction of apoptosis, cells were co-stained with annexin V/PI and analyzed using flow cytometry. An analysis of phosphatidylserine on the outer leaflet of apoptotic cell membranes was performed using annexin V-FITC and PI to distinguish between apoptotic and necrotic cells. After treatment, cells (1×106 cells/mL) were stained with annexin-V and PI according to the manufacturer’s instruction (BD Pharmingen, San Diego, CA). To each tube, 400 μL of 1x annexin binding buffer was added and stained cells were analyzed using a flow cytometer (BD LSRII; Becton Dickinson, San Diego, CA).
Statistical Methods
Statistical analysis was carried out using the student t-test and the Fisher’s exact test. Means were calculated from three independent experiments.
Supplementary Material
Acknowledgments
The authors are thankful to the late Enrique Suarez (INTA) for the direction of the ICBG Project in Argentina (1995–2005); Renee Fortunato (INTA) for the collection and identification of the plant material, and Gemma O’Donnell for assistance with the compound isolation. This field work was supported by the ICBG “Bioactive Agents from Dryland Biodiversity in Latin America”, grant 5 U01 TW 00316-10, from the National Institutes of Health (NIH) Fogarty International Center (FIC) to B.N.T., whereas the chemical work was supported by KU 2506014-910/099 to B.N.T. The authors would also like to acknowledge the University of Kansas Center for Cancer Experimental Therapeutics NIH-COBRE P20 RR015563 (PI: B.N.T., project award PI: M.S.C.), for support of the in vitro experiments for this project. Additionally, the authors would like to thank Dr. Joyce Slusser (Flow Cytometry Core Laboratory, University of Kansas Medical Center) for assistance with flow cytometry experiments. The contents are solely the responsibility of the authors and do not necessarily represent the official views of the NIH.
Footnotes
Supporting Information Available: Spectroscopy data of withaferin A (1), and table of IC50 values. This information is available free-of-charge via Internet at http://pubs.acs.org.
References and Notes
- 1.Argiris A, Karamouzis MV, Raben D, Ferris RL. Lancet. 2008;371:1695–1709. doi: 10.1016/S0140-6736(08)60728-X. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Curado MP, Hashibe M. Curr Opin Oncol. 2009;21:194–200. doi: 10.1097/CCO.0b013e32832a68ca. [DOI] [PubMed] [Google Scholar]
- 3.Culliney B, Birhan A, Young AV, Choi W, Shulimovich M, Blum RH. Oncology. 2008;22:1152–1161. discussion 1162–1156, 1171–1152. [PubMed] [Google Scholar]
- 4.Argiris A, Li Y, Forastiere A. Cancer. 2004;101:2222–2229. doi: 10.1002/cncr.20640. [DOI] [PubMed] [Google Scholar]
- 5.Lemaire F, Millon R, Young J, Cromer A, Wasylyk C, Schultz I, Muller D, Marchal P, Zhao C, Melle D, Bracco L, Abecassis J, Wasylyk B. Br J Cancer. 2003;89:1940–1949. doi: 10.1038/sj.bjc.6601373. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Conley BA. J Clin Oncol. 2006;24:1023–1025. doi: 10.1200/JCO.2005.05.0682. [DOI] [PubMed] [Google Scholar]
- 7.Vokes EE, Weichselbaum RR, Lippman SM, Hong WK. N Engl J Med. 1993;328:184–194. doi: 10.1056/NEJM199301213280306. [DOI] [PubMed] [Google Scholar]
- 8.Burtness B, Goldwasser MA, Flood W, Mattar B, Forastiere AA Eastern Cooperative Oncology, G. J Clin Oncol. 2005;23:8646–8654. doi: 10.1200/JCO.2005.02.4646. [DOI] [PubMed] [Google Scholar]
- 9.Cragg GM, Grothaus PG, Newman DJ. Chem Rev. 2009;109:3012–3043. doi: 10.1021/cr900019j. [DOI] [PubMed] [Google Scholar]
- 10.Smith SD, Baum DA. Am J Bot. 2006;93:1140–1153. doi: 10.3732/ajb.93.8.1140. [DOI] [PubMed] [Google Scholar]
- 11.Salvat A, Antonnacci L, Fortunato RH, Suarez EY, Godoy HM. Lett Appl Microbiol. 2001;32:293–297. doi: 10.1046/j.1472-765x.2001.00923.x. [DOI] [PubMed] [Google Scholar]
- 12.Kirson I, Glotter E. J Nat Prod. 1981;44:633–647. [Google Scholar]
- 13.Glotter E. Nat Prod Rep. 1991;8:415–440. doi: 10.1039/np9910800415. [DOI] [PubMed] [Google Scholar]
- 14.Abou-Douh AM. Arch Pharm (Weinheim) 2002;335:267–276. doi: 10.1002/1521-4184(200208)335:6<267::AID-ARDP267>3.0.CO;2-E. [DOI] [PubMed] [Google Scholar]
- 15.Stan SD, Hahm ER, Warin R, Singh SV. Cancer Res. 2008;68:7661–7669. doi: 10.1158/0008-5472.CAN-08-1510. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Koduru S, Kumar R, Srinivasan S, Evers MB, Damodaran C. Mol Cancer Ther. 2010;9:202–210. doi: 10.1158/1535-7163.MCT-09-0771. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Yu Y, Hamza A, Zhang T, Gu M, Zou P, Newman B, Li Y, Gunatilaka AAL, Zhan CG, Sun D. Biochem Pharmacol. 2010;79:542–551. doi: 10.1016/j.bcp.2009.09.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Malik F, Singh J, Khajuria A, Suri KA, Satti NK, Singh S, Kaul MK, Kumar A, Bhatia A, Qazi GN. Life Sci. 2007;80:1525–1538. doi: 10.1016/j.lfs.2007.01.029. [DOI] [PubMed] [Google Scholar]
- 19.Srinivasan S, Ranga RS, Burikhanov R, Han SS, Chendil D. Cancer Res. 2007;67:246–253. doi: 10.1158/0008-5472.CAN-06-2430. [DOI] [PubMed] [Google Scholar]
- 20.Gunatilaka AAL, Wijeratne E, Mudiyanselage K, Xu YM, Whitesell L, Lindquist S. 2010/030395A2. Int Pat WO. 2010
- 21.Cohen MS, Samadi AK, Timmermann BN. 2010/030395 A2 20100514. Int Pat WO. 2010
- 22.Uma Devi P, Utsumiz H, Takata M, Takeda S. Indian J Exp Biol. 2008;46:437–442. [PubMed] [Google Scholar]
- 23.Oh JH, Lee TJ, Park JW, Kwon TK. Eur J Pharmacol. 2008;599:11–17. doi: 10.1016/j.ejphar.2008.09.017. [DOI] [PubMed] [Google Scholar]
- 24.Sabina EP, Chandal S, Rasool MK. J Pharm Pharm Sci. 2008;11:46–55. doi: 10.18433/j35k58. [DOI] [PubMed] [Google Scholar]
- 25.Kaileh M, Vanden Berghe W, Heyerick A, Horion J, Piette J, Libert C, De Keukeleire D, Essawi T, Haegeman G. J Biol Chem. 2007;282:4253–4264. doi: 10.1074/jbc.M606728200. [DOI] [PubMed] [Google Scholar]
- 26.Ray AB, Gupta M. Fortschr Chem Org Naturst. 1994;63:1–106. doi: 10.1007/978-3-7091-9281-8_1. [DOI] [PubMed] [Google Scholar]
- 27.Jayaprakasam B, Zhang Y, Seeram NP, Nair MG. Life Sci. 2003;74:125–132. doi: 10.1016/j.lfs.2003.07.007. [DOI] [PubMed] [Google Scholar]
- 28.Mandal C, Dutta A, Mallick A, Chandra S, Misra L, Sangwan RS. Apoptosis. 2008;13:1450–1464. doi: 10.1007/s10495-008-0271-0. [DOI] [PubMed] [Google Scholar]
- 29.Kurokawa M, Kornbluth S. Cell. 2009;138:838–854. doi: 10.1016/j.cell.2009.08.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Pelletier SW, Mody NV, Nowacki J, Bhattacharyya J. J Nat Prod. 1979;42:512–521. [Google Scholar]
- 31.Bandhoria P, Gupta V, Kumar P, Satti N, Dutt P, Suri KA. Anal Sci. 2006;22:89–90. [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.