Abstract
The present study examined the relationships among statin use, APOE genotype, and insulin resistance as measured by the homeostasis model assessment of insulin resistance (HOMA-IR) in healthy older adults. APOE ε4- (i.e., not having an ε4 allele) statin users had higher HOMA-IR values compared with ε4+/statin users (p = 0.0169), and with non-users who were ε4- (p = 0. 0003) or ε4+ (p = 0.0006). These results suggest that statin use may modulate insulin levels for individuals without an APOE ε4 allele.
Keywords: Statin, insulin, Alzheimer’s disease, dementia, diabetes
Introduction
Alzheimer’s disease (AD) has grown in prevalence and is reaching epidemic proportions. Two important risk factors for AD are the presence of the apolipoprotein E (APOE) ε4 allele and insulin resistance (the reduced ability of insulin to dispose of glucose in the periphery). APOE genotype is currently the strongest genetic risk factor for the development of AD[1]. Our work and the work of others has also implicated insulin resistance as a risk factor for AD[2]. Epidemiological studies show an increased risk for AD in people with diabetes or impaired glucose tolerance/insulin resistance [3–5]. Clinical studies also demonstrate increased signs of insulin resistance in patients with AD[6, 7]. Our work has demonstrated that patients with AD have lower glucose disposal rates, hyperinsulinemia, and lower cerebrospinal fluid (CSF) to plasma insulin ratios[5, 8], and that these characteristics are modulated by APOE genotype [9].
The relationship between insulin resistance and APOE genotype may be related in part to their effects on lipid metabolism. ApoE is a cholesterol transport molecule, and insulin is a primary regulator of lipid metabolism, stimulating lipogenesis and reducing lipolysis. In adipocytes, insulin resistance leads to accelerated lipolysis and increased free fatty acid (FFA) levels. In turn, excess FFA influx into the liver inhibits insulin suppression of hepatic very low-density lipoprotein (VLDL) secretion, an essential process for preventing post-prandial hyperlipidemia. Acute inhibitory effects of insulin on VLDL production are essential for rapid hepatic adaption to metabolic shifts between fasting and feeding in order to maintain plasma lipids within an optimal physiologic range [10]. Thus, insulin resistant adults have higher and more prolonged post-prandial excursions of VLDL and other deleterious lipids.
The effects of both insulin and APOE genotype in patients with AD may be, in part, lipid related. Hyperlipidemia is associated with increased AD pathology in a transgenic mouse model[11]. Epidemiological studies show increased lipid levels associated with AD[12]. Elevated cholesterol levels in mid-life can lead to a 2 to 3-fold increased risk of AD[13]. This pattern may explain in part the increase in risk associated with APOE ε4 carriage, given that ε4 carriers have higher total and LDL cholesterol levels than do adults without the ε4 allele[14]. Statin lipid-lowering agents (LLAs), HMG-CoA reductase inhibitors that are often used to treat high cholesterol, may reduce the risk of Alzheimer’s disease [15, 16]. Interestingly, statin use has been shown to affect insulin sensitivity, although the nature and direction of the effects has been inconsistent across studies[17]. Given the interrelationships among insulin, dyslipidemia, and APOE genotype, we sought to determine whether statin use and APOE genotype modulated insulin resistance as indexed by the homeostasis model assessment of insulin resistance (HOMA-IR) in healthy older adults.
Materials and Methods
All studies were approved by the University of Washington Institutional Review Board and the VA Puget Sound Research and Development Committee. All subjects gave written informed consent prior to study participation. Subjects were 251 cognitively intact and 87 memory impaired (30 with AD, 57 with amnestic mild cognitive impairment [MCI]) older adults in good general health who participated from 1997 to 2009 in our research studies exploring the relationship of insulin resistance and memory in aging adults. Exclusionary criteria included diabetes, neurodegenerative disorders other than Alzheimer’s disease, head trauma with loss of consciousness > 30 min., large vessel CVA, use of antipsychotic medications and medications known to affect cognition or glucoregulation. As previously described in our work, AD was diagnosed by NINCDS/ADRA criteria[18] and MCI, widely regarded as an AD prodrome, by Petersen’s criteria[19]. Subject characteristics are provided in Table 1.
Table 1.
Subject Characteristics.
| Statin Non-Users | Statin Users | ||||
|---|---|---|---|---|---|
| APOE ε4- (n = 201) |
APOE ε4+ (n = 81) |
APOE ε4- (n = 39) |
APOE 4+ (n = 17) |
p-value | |
| Gender (F/M) | 95/106 | 43/38 | 17/22 | 7/10 | 0.5950 |
| Mem imp (MI/intact) | 48/153 | 33/48 | 3/36 | 3/14 | 0.0007 |
| FPG (mmol/L) | 5.39±0.82 | 5.47±0.83 | 5.67±0.69 | 5.88±0.46 | 0.0058 |
| FPI (pmol/L) | 84.0±70/8 | 81.3±60.4 | 129.9±88.9 | 80.6±44.4 | 0.0018 |
| Age (yrs) | 73.1±7.1 | 72.5±7.0 | 74.9±5.8 | 73.1±7.5 | 0.3699 |
| BMI (kg/m2) | 26.3±4.7 | 25.9±4.4 | 28.7±4.9 | 28.2±4.2 | 0.0071 |
| DRS | 136.4±9.1 | 130.3±15.9 | 138.5±4.3 | 137.6±5.1 | 0.0001 |
| Education (yrs) | 15.0±2.7 | 14.7±2.5 | 14.7± 2.6 | 14.8±2.5 | 0.7626 |
Gender (F/M = female/male) and Mem imp (MI/intact = memory impaired/cognitively intact individuals) are expressed as sample sizes; p-values were derived from χ2 tests. FPG (fasting plasma glucose; sig. by statin use, p < .01), FPI (fasting plasma insulin; sig. by APOE and Statin use and APOE × Statin use, p < .05), Age, BMI (body mass index; sig. by statin use, p < .01), DRS (Mattis Dementia Rating Scale; sig. by APOE, p < .01 and Statin use, p < .05), and Education are expressed as mean±standard deviation; p-values were derived from two-way ANOVAs (statin use by APOE genotype) without covariates. The DRS is a measure of overall cognitive function; test ceiling = 144, suggested dementia cutoff = 123.
Venous fasting blood was acquired and assayed for insulin by RIA, as previously described[20] and for glucose by the glucose oxidase method (Beckman Instruments, Fullerton, CA) or the HemoCue system (Ängelholm, Sweden). The homeostasis model assessment of insulin resistance (HOMA-IR) was calculated as fasting plasma insulin (FPI) in µU/mL times fasting plasma glucose (FPG) in mM/L divided by 22.5 (HOMA-IR = [FPI * FPG] / 22.5)[21].
HOMA-IR was analyzed with analysis of covariance (ANCOVA), and pair-wise t-tests were conducted. ANCOVA factors were statin-use (no or yes), APOE genotype (ε4 carrier = ε4+ or ε4 non-carrier = ε4-), and memory diagnosis (AD/MCI or intact); covariates included in the model were age, body mass index (BMI), Mattis Dementia Rating Scale[22] (DRS; a continuous measure of cognitive status), and age. Pearson r correlations were calculated to examine the relationships among HOMA-IR, FPI, FPG, age, DRS, and BMI.
Results
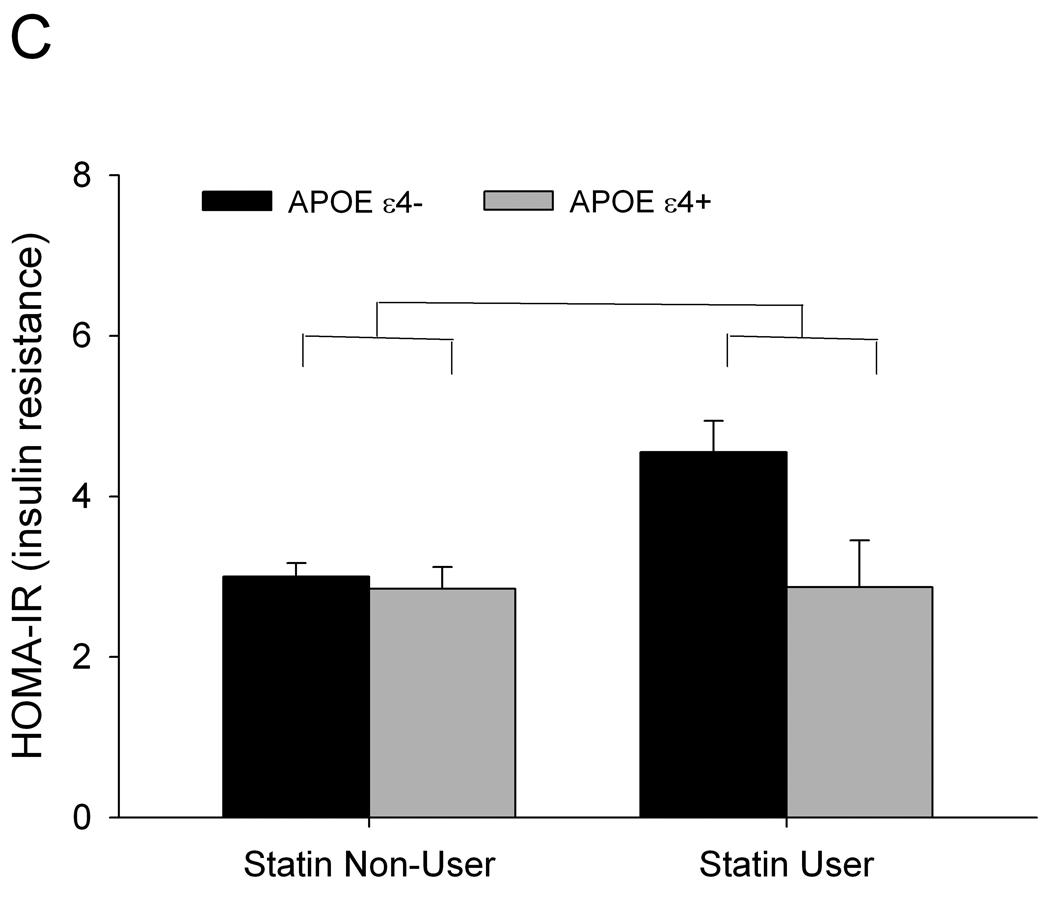
Memory diagnosis did not contribute significantly to the model and was removed from further analyses. For the remaining variables, the omnibus F-test was significant [F(7,331) = 8.86, p < 0.0001]. Statin users had significantly higher HOMA-IR values than did non-users, F(1,337) = 3.95, p = 0.0478 (Figure 1A). Carriers of an APOE ε4 allele (ε4+) had significantly lower HOMA-IR values than did non-carriers (ε4-), F(1,337) = 5.57, p = 0.0188 (Figure 1B). Statin use moderated the relationship between APOE genotype and HOMA-IR values, F(1, 337)] = 3.96, p = 0.0473 (Figure 1C). Post hoc comparisons revealed higher HOMA-IR values for APOE ε4- statin users compared with ε4+/statin users (p = 0.0169), and with non-users who were ε4- (p = 0. 0003) or ε4+ (p = 0.0006).
Figure 1.
Insulin resistance, statin use, and APOE genotype. HOMA-IR values were significantly higher (indicating more insulin resistance) (1A) for statin users than non-users, p = 0.0478, and (1B) non-narriers of an APOE ε4 allele (ε4-) than APOE ε4 carriers (ε4-), p = 0.0188. (1C).Statin use moderated the relationship between APOE genotype and fasting insulin levels, p = 0.0473, and (1D) exploratory analyses suggested an allele-dose effect, which did not to reach significance.
In exploratory analyses designed to characterize APOE allele-dose effects, we repeated the ANCOVA but substituted a three-level APOE factor (levels = 0, 1, 2 ε4 alleles). As shown in Figure 1D, the pattern of means support an allele-dose effect, but statistical significance was not reached due to insufficient numbers of APOE- ε4 homozygotes.
Across all subjects (n = 338), increases in HOMA-IR, indicating increased insulin resistance, were associated with expected increases in FPI (r = 0.9785, p < 0.0001), FPG (r = 0.2866, p < 0.0001), and BMI (r = 0.3011, p = 0.0006), and a higher BMI was associated with increased FPI (r = 0.2918, p = 0.0001) and statin use (r = 0.1832, p = 0.0007). Finally, BMI and DRS were not significantly related in the total sample or for ε4- subjects, but BMI rose as DRS increased in ε4+ subjects (n = 89, r = 0.2625, p = 0.093).
Discussion
This is the first study to suggest that statin users without the APOE- ε4 risk factor have greater insulin resistance as indexed by greater HOMA-IR values than do APOE- ε4 carriers or non-statin users. These results raise the possibility that cognitive benefits associated with statin use for APOE- ε4 carriers but not non-carriers in a recent AD trial[23] may be due in part to improved insulin function. The direction of this effect is interesting because as previously noted ε4 carriers have been found to have both higher total and LDL cholesterol levels than do adults without the ε4 allele and also to demonstrate increased AD pathology in a transgenic mouse model.[11, 14] As such, it would not have been surprising to see ε4 carriage as a risk factor that causes increased hyperlipidemia, inflammation, and reduced insulin function, at least as it pertains to a model that does not include statin medications. However, other studies have also shown this inconsistent effect direction when introducing statins into an APOE and insulin model[17].
A massing research body has supported the idea that statin use reduces the likelihood of developing or progressing to dementia. However, researchers have argued both for and against the hypothesized protective effect of statins on AD. For example, a large cohort study found no decreased risk of dementia for statin users[24] and statin users in the Religious Orders Study were also no more likely to develop incident AD nor a drop in cognitive scores.[25] Conversely, a re-examination of the ACT study in 2007 found that statin use at some point in participant’s lifetime left to significantly reduced neurofibrillary tangle burden at autopsy.[16]
This controversy may be due in part to the possibility that all statins may not be equal in their proposed neuroprotective effects on dementia. Riekse et al[15] found that simvastatin, a CNS-penatrant, reduced CSF levels of phoopho-tau-181 (a bio-marker of AD) in younger, cognitively normal participants. Conversely, Wolozin et al[26] established a lower prevalence of AD in those taking lovastatin and pravastatin, with no effects for simvastatin. However, a recent 2009 analysis of the Rotterdam Study which included 6992 participants found a reduced risk of AD for those taking statin medications, regardless of lipophilicity.[27] The current study did not have enough participants to determine relative effects of lipid soluble vs. non-lipid soluble statins. Additionally, the number of statin users in the current study was small; as such caution should be exercised in interpretation. However, the results are consistent with differential patterns of insulin function for APOE ε4 carriers. Further prospective studies may elucidate the mechanisms underlying the associations among statin use, insulin activity and APOE genotype.
Acknowledgements
This paper was supported by the Department of Veterans Affairs, NIH/NIA grants T32 AG000258 (BVF), R37AG010880 (SC) and K01AG23640 (GSW).
References
- 1.Corder EH, Saunders AM, Strittmatter WJ, Schmechel DE, Gaskell PC, Small GW, Roses AD, Haines JL, Pericak-Vance MA. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer's disease in late onset families. Science. 1993;261:921–923. doi: 10.1126/science.8346443. [DOI] [PubMed] [Google Scholar]
- 2.Craft S, Watson GS. Insulin and neurodegenerative disease: shared and specific mechanisms. Lancet Neurol. 2004;3:169–178. doi: 10.1016/S1474-4422(04)00681-7. [DOI] [PubMed] [Google Scholar]
- 3.Peila R, Rodriguez BL, Launer LJ. Type 2 diabetes, APOE gene, and the risk for dementia and related pathologies: The Honolulu-Asia Aging Study. Diabetes. 2002;51:1256–1262. doi: 10.2337/diabetes.51.4.1256. [DOI] [PubMed] [Google Scholar]
- 4.Leibson CL, Rocca WA, Hanson VA, Cha R, Kokmen E, O'Brien PC, Palumbo PJ. The risk of dementia among persons with diabetes mellitus: a population-based cohort study. Ann N Y Acad Sci. 1997;826:422–427. doi: 10.1111/j.1749-6632.1997.tb48496.x. [DOI] [PubMed] [Google Scholar]
- 5.Craft S, Asthana S, Schellenberg G, Cherrier M, Baker LD, Newcomer J, Plymate S, Latendresse S, Petrova A, Raskind M, Peskind E, Lofgreen C, Grimwood K. Insulin metabolism in Alzheimer's disease differs according to apolipoprotein E genotype and gender. Neuroendocrinology. 1999;70:146–152. doi: 10.1159/000054469. [DOI] [PubMed] [Google Scholar]
- 6.Razay G, Wilcock GK. Hyperinsulinaemia and Alzheimer's disease. Age Ageing. 1994;23:396–399. doi: 10.1093/ageing/23.5.396. [DOI] [PubMed] [Google Scholar]
- 7.Meneilly GS, Cheung E, Tessier D, Yakura C, Tuokko H. The effect of improved glycemic control on cognitive functions in the elderly patient with diabetes. J Gerontol. 1993;48:M117–M121. doi: 10.1093/geronj/48.4.m117. [DOI] [PubMed] [Google Scholar]
- 8.Craft S, Peskind E, Schwartz MW, Schellenberg GD, Raskind M, Porte D., Jr Cerebrospinal fluid and plasma insulin levels in Alzheimer's disease: relationship to severity of dementia and apolipoprotein E genotype. Neurology. 1998;50:164–168. doi: 10.1212/wnl.50.1.164. [DOI] [PubMed] [Google Scholar]
- 9.Craft S, Asthana S, Cook DG, Baker LD, Cherrier M, Purganan K, Wait C, Petrova A, Latendresse S, Watson GS, Newcomer JW, Schellenberg GD, Krohn AJ. Insulin dose-response effects on memory and plasma amyloid precursor protein in Alzheimer's disease: interactions with apolipoprotein E genotype. Psychoneuroendocrinology. 2003;28:809–822. doi: 10.1016/s0306-4530(02)00087-2. [DOI] [PubMed] [Google Scholar]
- 10.Kamagate A, Qu S, Perdomo G, Su D, Kim DH, Slusher S, Meseck M, Dong HH. FoxO1 mediates insulin-dependent regulation of hepatic VLDL production in mice. J Clin Invest. 2008;118:2347–2364. doi: 10.1172/JCI32914. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Refolo LM, Malester B, LaFrancois J, Bryant-Thomas T, Wang R, Tint GS, Sambamurti K, Duff K, Pappolla MA. Hypercholesterolemia accelerates the Alzheimer's amyloid pathology in a transgenic mouse model. Neurobiol Dis. 2000;7:321–331. doi: 10.1006/nbdi.2000.0304. [DOI] [PubMed] [Google Scholar]
- 12.Morris MC, Evans DA, Bienias JL, Tangney CC, Bennett DA, Aggarwal N, Schneider J, Wilson RS. Dietary fats and the risk of incident Alzheimer disease. Arch Neurol. 2003;60:194–200. doi: 10.1001/archneur.60.2.194. [DOI] [PubMed] [Google Scholar]
- 13.Kivipelto M, Helkala EL, Laakso MP, Hanninen T, Hallikainen M, Alhainen K, Soininen H, Tuomilehto J, Nissinen A. Midlife vascular risk factors and Alzheimer's disease in later life: longitudinal, population based study. BMJ. 2001;322:1447–1451. doi: 10.1136/bmj.322.7300.1447. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Reilly SL, Ferrell RE, Kottke BA, Kamboh MI, Sing CF. The gender-specific apolipoprotein E genotype influence on the distribution of lipids and apolipoproteins in the population of Rochester, MN. I. Pleiotropic effects on means and variances. Am J Hum Genet. 1991;49:1155–1166. [PMC free article] [PubMed] [Google Scholar]
- 15.Riekse RG, Li G, Petrie EC, Leverenz JB, Vavrek D, Vuletic S, Albers JJ, Montine TJ, Lee VM, Lee M, Seubert P, Galasko D, Schellenberg GD, Hazzard WR, Peskind ER. Effect of statins on Alzheimer's disease biomarkers in cerebrospinal fluid. J Alzheimers Dis. 2006;10:399–406. doi: 10.3233/jad-2006-10408. [DOI] [PubMed] [Google Scholar]
- 16.Silbert LC. Does statin use decrease the amount of Alzheimer disease pathology in the brain? Neurology. 2007;69:E8–E11. doi: 10.1212/01.wnl.0000280585.95661.1b. [DOI] [PubMed] [Google Scholar]
- 17.Auer J, Berent R, Weber T, Eber B. Clinical significance of pleiotropic effects of statins: lipid reduction and beyond. Curr Med Chem. 2002;9:1831–1850. doi: 10.2174/0929867023369024. [DOI] [PubMed] [Google Scholar]
- 18.McKhann G, Drachman D, Folstein M, Katzman R, Price D, Stadlan EM. Clinical diagnosis of Alzheimer's disease: report of the NINCDS-ADRDA Work Group under the auspices of Department of Health and Human Services Task Force on Alzheimer's Disease. Neurology. 1984;34:939–944. doi: 10.1212/wnl.34.7.939. [DOI] [PubMed] [Google Scholar]
- 19.Petersen RC. Mild cognitive impairment as a diagnostic entity. J Intern Med. 2004;256:183–194. doi: 10.1111/j.1365-2796.2004.01388.x. [DOI] [PubMed] [Google Scholar]
- 20.Kahn SE, Leonetti DL, Prigeon RL, Boyko EJ, Bergstrom RW, Fujimoto WY. Relationship of proinsulin and insulin with noninsulin-dependent diabetes mellitus and coronary heart disease in Japanese-American men: impact of obesity--clinical research center study. J Clin Endocrinol Metab. 1995;80:1399–1406. doi: 10.1210/jcem.80.4.7714116. [DOI] [PubMed] [Google Scholar]
- 21.Lansang MC, Williams GH, Carroll JS. Correlation between the glucose clamp technique and the homeostasis model assessment in hypertension. Am J Hypertens. 2001;14:51–53. doi: 10.1016/s0895-7061(00)01229-2. [DOI] [PubMed] [Google Scholar]
- 22.Mattis S. Dementia Rating Scale. Odessa, FL: Psychological Assessment Resources; 1988. [Google Scholar]
- 23.Sparks DL, Connor DJ, Sabbagh MN, Petersen RB, Lopez J, Browne P. Circulating cholesterol levels, apolipoprotein E genotype and dementia severity influence the benefit of atorvastatin treatment in Alzheimer's disease: results of the Alzheimer's Disease Cholesterol-Lowering Treatment (ADCLT) trial. Acta Neurol Scand Suppl. 2006;185:3–7. doi: 10.1111/j.1600-0404.2006.00690.x. [DOI] [PubMed] [Google Scholar]
- 24.Rea TD, Breitner JC, Psaty BM, Fitzpatrick AL, Lopez OL, Newman AB, Hazzard WR, Zandi PP, Burke GL, Lyketsos CG, Bernick C, Kuller LH. Statin use and the risk of incident dementia: the Cardiovascular Health Study. Arch Neurol. 2005;62:1047–1051. doi: 10.1001/archneur.62.7.1047. [DOI] [PubMed] [Google Scholar]
- 25.Arvanitakis Z, Schneider JA, Wilson RS, Bienias JL, Kelly JF, Evans DA, Bennett DA. Statins, incident Alzheimer disease, change in cognitive function, and neuropathology. Neurology. 2008;70:1795–1802. doi: 10.1212/01.wnl.0000288181.00826.63. [DOI] [PubMed] [Google Scholar]
- 26.Wolozin B, Kellman W, Ruosseau P, Celesia GG, Siegel G. Decreased prevalence of Alzheimer disease associated with 3-hydroxy-3-methyglutaryl coenzyme A reductase inhibitors. Arch Neurol. 2000;57:1439–1443. doi: 10.1001/archneur.57.10.1439. [DOI] [PubMed] [Google Scholar]
- 27.Haag MD, Hofman A, Koudstaal PJ, Stricker BH, Breteler MM. Statins are associated with a reduced risk of Alzheimer disease regardless of lipophilicity. The Rotterdam Study. J Neurol Neurosurg Psychiatry. 2009;80:13–17. doi: 10.1136/jnnp.2008.150433. [DOI] [PubMed] [Google Scholar]