Abstract
Patients with Kallmann syndrome (KS) have hypogonadotropic hypogonadism caused by a deficiency of gonadotropin-releasing hormone (GnRH) and a defective sense of smell related to olfactory bulb aplasia. Based on the findings in a fetus affected by the X chromosome–linked form of the disease, it has been suggested that hypogonadism in KS results from the failed embryonic migration of neuroendocrine GnRH1 cells from the nasal epithelium to the forebrain. We asked whether this singular observation might extend to other developmental disorders that also include arrhinencephaly. We therefore studied the location of GnRH1 cells in fetuses affected by different arrhinencephalic disorders, specifically X-linked KS, CHARGE syndrome, trisomy 13, and trisomy 18, using immunohistochemistry. Few or no neuroendocrine GnRH1 cells were detected in the preoptic and hypothalamic regions of all arrhinencephalic fetuses, whereas large numbers of these cells were present in control fetuses. In all arrhinencephalic fetuses, many GnRH1 cells were present in the frontonasal region, the first part of their migratory path, as were interrupted olfactory nerve fibers that formed bilateral neuromas. Our findings define a pathological sequence whereby a lack of migration of neuroendocrine GnRH cells stems from the primary embryonic failure of peripheral olfactory structures. This can occur either alone, as in isolated KS, or as part of a pleiotropic disease, such as CHARGE syndrome, trisomy 13, and trisomy 18.
Introduction
Gonadotropin-releasing hormone (GnRH) controls the pituitary-gonadal axis (1). Embryological studies in various vertebrate species have shown that neuroendocrine GnRH cells (i.e., GnRH1-synthesizing neurons) migrate from the medial part of the nasal epithelium up to the forebrain. In mammals, they migrate in close association with fibers of the vomeronasal and terminal nerves (2, 3). Upon reaching the base of the telencephalon, they penetrate the brain together with the central processes of the terminal nerves, just caudal to the olfactory bulb anlage, and make their way along the medial wall of the cerebral hemisphere to the preoptic/hypothalamic region, where they ultimately settle. Migration of these cells begins during the sixth embryonic week in humans (3). This is also when fibers of the olfactory and terminal nerves first come into contact with the brain, shortly before the emergence of the olfactory bulbs.
Kallmann syndrome (KS) is a developmental disorder that combines hypogonadotropic hypogonadism with anosmia/hyposmia, related to the aplasia/hypoplasia of the olfactory bulbs and tracts (4). It has been proposed that disrupted migration of neuroendocrine GnRH cells is responsible for the hypogonadism, based on the examination of a single 19-week-old male fetus carrying a chromosomal deletion at Xp22.3 that included KAL1 (5–7), the gene responsible for the X chromosome–linked form of the syndrome, KAL1. This fetus had no olfactory bulbs, and there was a complete absence of GnRH1 cells in the brain, whereas clusters of these cells were found in the nasal region and on the dorsal surface of the ethmoid bone cribriform plate, together with entangled fibers of the olfactory and terminal nerves that did not contact the forebrain (8).
Arrhinencephaly (i.e., absence of the olfactory bulbs) is observed in KS and some pleiotropic developmental diseases, including CHARGE syndrome (9), trisomy 13 (also known as Patau syndrome; ref. 10), and trisomy 18 (also known as Edwards syndrome; ref. 11). CHARGE refers to coloboma of the eye, heart defect, choanal atresia, retarded growth and development, genital hypoplasia, external ear anomalies and deafness (12); however, many individuals with CHARGE syndrome do not have all these clinical features, and other developmental anomalies can also be present (13). In particular, hypoplasia/aplasia of the inner ear semicircular canals has consistently been found in patients (9). Moreover, a recent study concluded that most, if not all, CHARGE patients have both olfactory bulb hypoplasia/aplasia and hypogonadotropic hypogonadism (14); that is, the KS-defining features. In approximately two-thirds of cases, CHARGE syndrome is caused by neomutations in CHD7 (15). Mutations in this gene have been identified in a few patients initially diagnosed with KS (16, 17), thus reinforcing the idea of a tight pathological relationship between the diseases. Therefore, we asked whether the hypogonadism in CHARGE syndrome could result from defective migration of neuroendocrine GnRH cells, as in the KAL1 fetus. We further asked whether defective migration of these cells is also associated with the absence of the olfactory bulbs found in some individuals with trisomy 13 or trisomy 18. Even though hypogonadotropic hypogonadism has not to our knowledge been reported in these genetic conditions (presumably because of short life expectancy), cryptorchidism and micropenis are common in affected males (10). We thus studied the location of GnRH1-immunoreactive cells in 4 different arrhinencephalic fetuses: another KAL1 male fetus, aged 25 weeks, carrying a nonsense mutation in KAL1; a male fetus, aged 23 weeks, with CHARGE syndrome due to a frame-shifting mutation in CHD7; a female fetus, aged 18 weeks, with trisomy 13 (47, XX+13 karyotype); and a male fetus, aged 35 weeks, with trisomy 18 (47, XY+18 karyotype) (see Supplemental Results and Supplemental Figure 1; supplemental material available online with this article; doi: 10.1172/JCI43699DS1).
Results and Discussion
Arrhinencephalic fetuses have few or no neuroendocrine GnRH cells in the preoptic and hypothalamic areas.
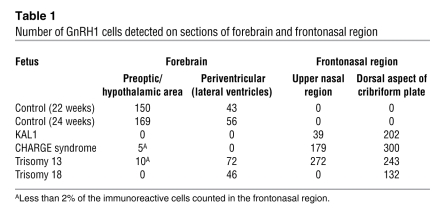
In 2 control fetuses aged 22 and 24 weeks, numerous GnRH1-immunoreactive cells were detected in the forebrain. They were found singly, in pairs, or, rarely, in small clusters. Most of these cells were neuroendocrine cells that were found in the medial preoptic area, the retrochiasmatic area, and the anterior and medial basal hypothalamus (Figure 1, Table 1, and ref. 8). In contrast, we detected few or no GnRH1 cells in the preoptic and hypothalamic areas of the 4 arrhinencephalic fetuses. In addition, in the control, trisomy 13, and trisomy 18 fetuses, scattered GnRH1 cells were found close to the lateral ventricles, at various emplacements from the dorsal thalamus to the fornix (Figure 1, Table 1, and ref. 18). These cells presumably represent misrouted GnRH1 neurons.
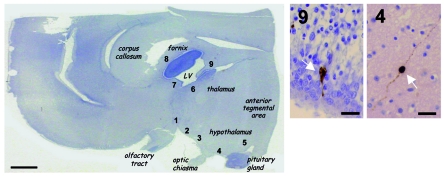
Figure 1. Distribution of GnRH1-immunoreactive cells in a normal fetal brain.
Left: Mayer hemalun staining of a sagittal section of the forebrain from a 24-week-old control fetus (rostral region at left). Arabic numerals denote the locations of GnRH1-immunoreactive cells: 1–5 are typical of neuroendocrine GnRH1 cells, whereas 6–9 presumably correspond to some misrouted GnRH1 neurons. Right: detailed microscopic views of regions 4 and 9, showing GnRH1 neurons (arrows). LV, lateral ventricle. Scale bars: 3 mm (left); 50 μm (right).
Table 1 .
Number of GnRH1 cells detected on sections of forebrain and frontonasal region
Arrhinencephalic fetuses, but not control fetuses, have many GnRH1 cells in the frontonasal region.
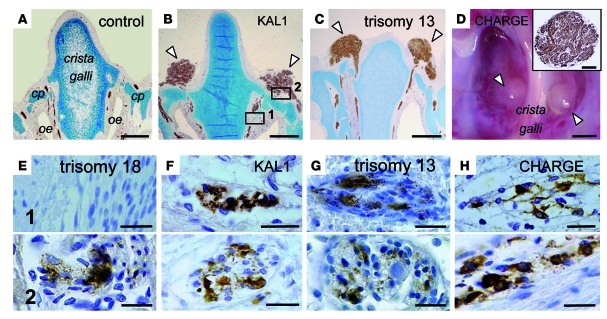
In all the arrhinencephalic fetuses, numerous GnRH1 cells were found in the upper nasal region and/or on the dorsal aspect of the ethmoid bone cribriform plate. These cells occasionally formed small clusters in ganglia of the terminal nerve (Figure 2, E–H). In contrast, we did not detect GnRH1 cells in the frontonasal region in any of the age-matched control fetuses. Notably, present on the dorsal aspect of the cribriform plate in the KAL1 and CHARGE fetuses were bilateral spherical structures consistent with neuromas, for they were composed of entangled nerve fibers, as shown by βIII-tubulin immunostaining. Such protrusive structures were not clearly identified in the 2 trisomic fetuses, but we did observe similar bulges on both sides of the crista galli in another arrhinencephalic trisomy 13 fetus, aged 23 weeks, whose brain could not be analyzed for GnRH1 immunoreactivity (Figure 2, A–D). The number of GnRH1-immunoreactive cells detected on brain sections and sections of the frontonasal region in the 4 arrhinencephalic fetuses and 2 control fetuses are presented in Table 1.
Figure 2. Neuromas and accumulation of GnRH1-immunoreactive cells in the frontonasal region of arrhinencephalic fetuses.
(A–C) Coronal sections of the frontonasal region in the KAL1 fetus and 23-week-old trisomy 13 and control fetuses, immunostained with an anti–βIII-tubulin antibody. The brains have been removed. In the KAL1 and trisomy 13 fetuses, bilateral spherical structures corresponding to neuromas (arrowheads) are visible on the dorsal aspect of the cribriform plate (cp) stained with Alcian blue. oe, olfactory epithelium. (D) Endocranial view of the cribriform plate region of the ethmoid bone in the CHARGE fetus. Abnormal spherical structures (arrowheads) were present on both sides of the crista galli. Inset: section from one of the spherical structures. The entangled nerve fibers that make up the neuroma were stained using an anti–βIII-tubulin antibody. (E–H) Coronal sections corresponding to the boxed regions in B from KAL1, CHARGE, trisomy 13, and trisomy 18 fetuses. Sections were immunostained using an anti-GnRH1 antibody. In all fetuses, GnRH1 cells were seen in the upper nasal region (region 1) and/or on the dorsal aspect of the cribriform plate (region 2). Note the presence of GnRH1 cells only on the dorsal aspect of the cribriform plate for the 35-week-old trisomy 18 fetus. Scale bars: 1 mm (A–D); 200 μm (D, inset); 30 μm (E–H).
The olfactogenital fetopathological sequence.
This study of human fetuses lacking olfactory bulbs showed in all cases an accumulation of GnRH1 cells in the first, extracerebral part of their migratory path. Although these cells had left the nasal epithelium, they seemed to be arrested in the frontonasal region, together with interrupted nerve fibers. Accordingly, few or no neuroendocrine GnRH cells were detected in the preoptic/hypothalamic regions of these fetuses (Figure 3). Our findings, combined with the previous study of a KAL1 fetus (8), thus delimit a pathological entity that can be defined clinically by the association of hypogonadotropic hypogonadism with anosmia (KS) and anatomically by the concomitance of olfactory bulb aplasia and accumulation of neuroendocrine GnRH cells outside the brain, together with the premature termination of olfactory and terminal nerve fibers. The failure of GnRH1 cell migration is likely to be the consequence of interrupted terminal nerve fibers, which normally serve as guidance cues for the penetration of these cells into the forebrain (2, 3). Our conclusions are primarily based on the study of 2 fetuses affected by the KAL1 genetic form (ref. 8 and the present study). To our knowledge, histopathological observations of human individuals affected by other genetic forms of KS, caused by loss-of-function mutations in FGFR1 (19), FGF8 (20), PROKR2, or PROK2 (21), are not yet available. These forms indeed do not involve life-threatening developmental anomalies that may lead to medical interruption of pregnancy and fetopathological examination. However, several mouse models of KS have been analyzed, which are useful to extend the pathological observations made in the KAL1 human fetuses to other genetic forms. Both Prokr2 and Prok2 knockout mice have a marked decrease in the number of GnRH1 cells present in the preoptic area of the brain (22, 23), which, in the Prok2-null mice, could be attributed to a defect in the embryonic migration of these cells. Indeed, in E13.5 mutant mice, GnRH1 cells, after crossing the cribriform plate, seemed to be trapped in a spherical-shaped abnormal structure (23), reminiscent of the neuromas we observed in the human fetuses. A similar structure was also reported in Prokr2-null mice as early as E12.5 (22), by the time axonal fibers of the olfactory and terminal nerves first come into contact with the rostral forebrain in a normal mouse. This is indicative of a primary failure of these axons to penetrate the brain. Mice deficient in Fgfr1 or Fgf8 because of hypomorphic mutations also have a markedly decreased number or a complete absence, respectively, of GnRH1 cells in the preoptic area (20, 24). In the Fgf8 hypomorphs, however, GnRH1 cells never emerge from the nasal epithelium (24), which suggests the existence of an earlier defect expected to mask the GnRH1 cell migration defect.
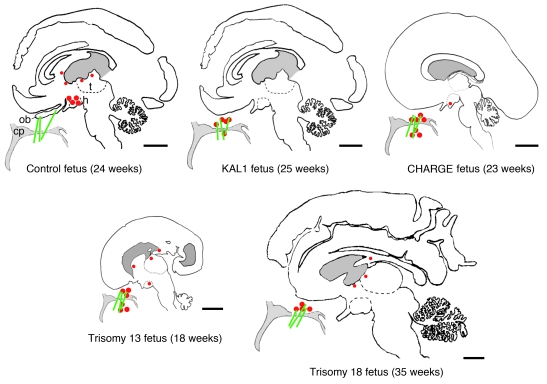
Figure 3. Distribution of GnRH1-immunoreactive cells in the forebrains and frontonasal regions of the 4 arrhinencephalic fetuses and a control fetus.
Red dots denote GnRH1 cells. Green lines indicate the path of the olfactory and terminal nerve fibers. The intracerebral fluid compartment is shaded gray. In the control fetus, GnRH1 cells were present in the preoptic and hypothalamic areas and were not detected in the frontonasal region. In addition, scattered, presumably misrouted GnRH1 neurons were found at several periventricular locations in the forebrain. In the arrhinencephalic fetuses, GnRH1 cells accumulated along the broken path of the olfactory and terminal nerve fibers that do not make contact with the forebrain. Few or no neuroendocrine cells were found in the preoptic/hypothalamic region. Scattered, periventricular GnRH1 neurons were also detected in the trisomy 13 and trisomy 18 fetuses. Scale bars: 1 cm. ob, olfactory bulb; h, hypothalamus; t, thalamus.
KS in pleiotropic developmental diseases.
From this and other studies, it appears that the olfactogenital pathological sequence defined herein can be either isolated or associated with various developmental anomalies. Some of these, including renal agenesis, cleft lip or palate, hypodontia, and bimanual synkinesia, were unambiguously ascribed to specific gene defects responsible for KS several years ago (4). While more recent clinical and genetic studies already supported the notion that CHARGE syndrome includes KS (14, 16, 17), our findings in the CHARGE fetus not only confirm this view, but also indicate that in the 2 pathological conditions, hypogonadism presumably results from the same fetopathological sequence. To our knowledge, a connection between the reported anomalies of the olfactory and reproductive systems in trisomy 13 and trisomy 18 had not previously been suspected. The study of the trisomic human fetuses was most informative, especially in the absence of animal models for these chromosomal anomalies. In addition, assuming that only 1 gene on each of the chromosomes 13 and 18 is responsible for the olfactogenital sequence in these fetuses, some patients with KS might carry gain-of-function mutations in these putative genes. Indeed, mutations in the genes already identified only account for approximately 30% of all KS cases (4).
In conclusion, we have extended the failure of neuroendocrine GnRH cell migration, initially reported in an individual with KAL1, to 3 different developmental diseases that share olfactory bulb aplasia: CHARGE syndrome, trisomy 13, and trisomy 18. Based on the connection between the peripheral olfactory system and the path of GnRH1 cells in embryonic life, as well as the olfactogenital pathological sequence defined herein, we surmise that such a GnRH1 cell migration defect occurs in most, if not all, arrhinencephalic conditions.
Methods
This study was approved by the national research ethics committee (Agence de Biomédecine, Paris, France).
Human tissues.
Fetuses were obtained from medically terminated pregnancies, with parents’ written informed consent. Termination of pregnancy was carried out by intracardiac injection of 25 U pentothal, followed by labor induction. Gestational ages of the fetuses were established by crown-rump length measurement during first-trimester ultrasonography examination, confirmed at autopsy by fetal biometry and evaluation of skeletal and organ maturation. The forebrains from 4 arrhinencephalic fetuses aged 18, 23, 25, and 35 weeks and 2 control fetuses aged 22 and 24 weeks were analyzed. Brains were removed and fixed in a 4% formaldehyde solution containing 154 mM NaCl and 16 mM ZnSO4 for 3–4 weeks. For frontonasal region analysis, all arrhinencephalic fetuses were compared with age-matched control fetuses. A fragment of about 1 cm3 of the frontonasal region was taken from each fetus, fixed in the formaldehyde solution for 1 week, and decalcified in 0.5 M EDTA for another week. The forebrains and frontonasal region fragments were embedded in paraffin and cut in sagittal and coronal slices, respectively. Serial sections (4 μm thick) were then taken on Superfrost Plus slides (Thermo Scientific) using a Microm HM340E microtome. 1 of 10 sections was stained with H&E for histological analysis.
Immunohistochemistry.
Immunochemistry analysis was conducted as previously described (8). Primary antibodies were rabbit anti-GnRH1 (1:2,000 dilution; catalog no. ab5617; Abcam) directed against the human GnRH1 decapeptide (cross-reaction with GnRH2, <0.1% by RIA using 125I GnRH1 as radio ligand; ref. 18) and mouse anti-human βIII-tubulin (1:50 dilution; Chemicon) for detection of GnRH1 cells and nerve fibers, respectively. Immunostainings used the UltraTech HRP kit (Immunotech). Sections were counterstained with Mayer hemalun. A few sections from the frontonasal region were counterstained with Alcian blue to reveal skeletal structures. Images were acquired using a DMR microscope (Leica) with a DFX-900 camera (Sony) and Matrox Inspector software (version 4.1; Matrox). We analyzed 144–173 consecutive brain sections and 45–100 consecutive frontonasal sections for each fetus. For quantification of GnRH1 cells, all sections were analyzed separately by 2 independent investigators, with common reevaluation when necessary. Special care was taken not to double-count GnRH cells on adjacent sections.
Supplementary Material
Acknowledgments
We thank Marie-Noelle Lotiquet, Béatrice Bartolt, Véronique Kyroudis, Rémi Pineau, Mélanie Dumand, Chrystel Leroy, and Corinne Fouveaut for technical help and Henryk Urbanski, Alon Chen, and Christine Petit for fruitful discussions. The work was supported by the Agence Nationale de la Recherche (ANR-09-GENO-017-01) and NIH grant HD-29186. L. Teixeira was supported by a fellowship from the Fondation pour la Recherche Médicale.
Footnotes
Conflict of interest: The authors have declared that no conflict of interest exists.
Citation for this article: J Clin Invest. 2010;120(10):3668–3672. doi:10.1172/JCI43699.
References
- 1.Plant TM. Hypothalamic control of the pituitary-gonadal axis in higher primates: key advances over the last two decades. J Neuroendocrinol. 2008;20(6):719–726. doi: 10.1111/j.1365-2826.2008.01708.x. [DOI] [PubMed] [Google Scholar]
- 2.Schwanzel-Fukuda M, Pfaff DW. Origin of luteinizing hormone-releasing hormone neurons. Nature. 1989;338(6211):161–164. doi: 10.1038/338161a0. [DOI] [PubMed] [Google Scholar]
- 3.Schwanzel-Fukuda M, Crossin KL, Pfaff DW, Bouloux PMG, Hardelin JP, Petit C. Migration of Luteinizing Hormone-Releasing Hormone (LHRH) neurons in early human embryos. J Comp Neurol. 1996;366(3):547–557. doi: 10.1002/(SICI)1096-9861(19960311)366:3<547::AID-CNE12>3.0.CO;2-M. [DOI] [PubMed] [Google Scholar]
- 4.Dodé C, Hardelin JP. Kallmann syndrome. Eur J Hum Genet. 2009;17(2):139–146. doi: 10.1038/ejhg.2008.206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Franco B, et al. A gene deleted in Kallmann’s syndrome shares homology with neural cell adhesion and axonal path-finding molecules. Nature. 1991;353(6344):529–536. doi: 10.1038/353529a0. [DOI] [PubMed] [Google Scholar]
- 6.Legouis R, et al. The candidate gene for the X-linked Kallmann syndrome encodes a protein related to adhesion molecules. Cell. 1991;67(2):423–435. doi: 10.1016/0092-8674(91)90193-3. [DOI] [PubMed] [Google Scholar]
- 7.Hardelin J-P, et al. X chromosome-linked Kallmann syndrome: stop mutations validate the candidate gene. Proc Natl Acad Sci U S A. 1992;89(17):8190–8194. doi: 10.1073/pnas.89.17.8190. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Schwanzel-Fukuda M, Bick D, Pfaff DW. Luteinizing hormone-releasing hormone (LHRH)-expressing cells do not migrate normally in an inherited hypogonadal (Kallmann) syndrome. Mol Brain Res. 1989;6(4):311–326. doi: 10.1016/0169-328X(89)90076-4. [DOI] [PubMed] [Google Scholar]
- 9.Sanlaville D, et al. Phenotypic spectrum of CHARGE syndrome in fetuses with CHD7 truncating mutations correlates with expression during human development. J Med Genet. 2006;43(3):211–317. doi: 10.1136/jmg.2005.036160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Taylor AI. Autosomal trisomy syndromes: a detailed study of 27 cases of Edwards’ syndrome and 27 cases of Patau’s syndrome. J Med Genet. 1968;5(3):227–252. doi: 10.1136/jmg.5.3.227. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Delezoide AL, Narcy F, Larroche JC. Cerebral midline developmental anomalies: spectrum and associated features. Genet Couns. 1991;1(3–4):197–210. [PubMed] [Google Scholar]
- 12.Pagon RA, Graham JM, Jr, Zonana J, Yong SL. Coloboma, congenital heart disease, and choanal atresia with multiple anomalies: CHARGE association. . J Pediatr. 1981;99(2):223–227. doi: 10.1016/s0022-3476(81)80454-4. [DOI] [PubMed] [Google Scholar]
- 13.Sanlaville D, Verloes A. CHARGE syndrome: an update. Eur J Hum Genet. 2007;15(4):389–399. doi: 10.1038/sj.ejhg.5201778. [DOI] [PubMed] [Google Scholar]
- 14.Pinto G, et al. CHARGE syndrome includes hypogonadotropic hypogonadism and abnormal olfactory bulb development. J Clin Endocrinol Metab. 2005;90(10):5621–5626. doi: 10.1210/jc.2004-2474. [DOI] [PubMed] [Google Scholar]
- 15.Vissers LE, et al. Mutations in a new member of the chromodomain gene family cause CHARGE syndrome. Nat Genet. 2004;36(9):955–957. doi: 10.1038/ng1407. [DOI] [PubMed] [Google Scholar]
- 16.Kim HG, et al. Mutations in CHD7, encoding a chromatin-remodeling protein, cause idiopathic hypogonadotropic hypogonadism and Kallmann syndrome. . Am J Hum Genet. 2008;83(4):511–519. doi: 10.1016/j.ajhg.2008.09.005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Jongmans MC, et al. CHD7 mutations in patients initially diagnosed with Kallmann syndrome - the clinical overlap with CHARGE syndrome. . Clin Genet. 2009;75(1):65–71. doi: 10.1111/j.1399-0004.2008.01107.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Quanbeck C, Sherwood NM, Millar RP, Terasawa E. Two populations of luteinizing hormone-releasing hormone neurons in the forebrain of the rhesus macaque during embryonic development. J Comp Neurol. 1997;380(3):293–309. doi: 10.1002/(SICI)1096-9861(19970414)380:3<293::AID-CNE1>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- 19.Dodé C, et al. Loss-of-function mutations in FGFR1 cause autosomal dominant Kallmann syndrome. . Nat Genet. 2003;33(4):463–465. doi: 10.1038/ng1122. [DOI] [PubMed] [Google Scholar]
- 20.Falardeau J, et al. Decreased FGF8 signaling causes deficiency of gonadotropin-releasing hormone in humans and mice. J Clin Invest. 2008;118(8):2822–2831. doi: 10.1172/JCI34538. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Dodé C, et al. Kallmann syndrome: mutations in the genes encoding prokineticin-2 and prokineticin receptor-2. PLoS Genet. 2006;2(10):1648–1652. doi: 10.1371/journal.pgen.0020175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Matsumoto S, et al. Abnormal development of the olfactory bulb and reproductive system in mice lacking prokineticin receptor PKR2. Proc Natl Acad Sci U S A. 2006;103(11):4140–4145. doi: 10.1073/pnas.0508881103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Pitteloud N, et al. Loss-of-function mutation in the prokineticin 2 gene causes Kallmann syndrome and normosmic idiopathic hypogonadotropic hypogonadism. Proc Natl Acad Sci U S A. 2007;104(44):17447–17452. doi: 10.1073/pnas.0707173104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Chung WC, Moyle SS, Tsai PS. Fibroblast growth factor 8 signaling through Fgf receptor 1 is required for the emergence of gonadotropin-releasing hormone neurons. Endocrinology. 2008;149(10):4997–5003. doi: 10.1210/en.2007-1634. [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.