Abstract
Objective
Bovine growth hormone (bGH) transgenic mice develop severe kidney damage. This damage may be due, at least in part, to changes in gene expression. Identification of genes with altered expression in the bGH kidney may identify mechanisms leading to damage in this system that may also be relevant to other models of kidney damage.
Design
cDNA subtraction libraries, northern blot analyses, microarray analyses and real-time reverse transcription polymerase chain reaction (RT/PCR) assays were used to identify and verify specific genes exhibiting differential RNA expression between kidneys of bGH mice and their non-transgenic (NT) littermates.
Results
Immunoglobulins were the vast majority of genes identified by the cDNA subtractions and the microarray analyses as being up-regulated in bGH. Several glycoprotein genes and inflammation-related genes also showed increased RNA expression in the bGH kidney. In contrast, only a few genes were identified as being significantly down-regulated in the bGH kidney. The most notable decrease in RNA expression was for the gene encoding kidney androgen-regulated protein.
Conclusions
A number of genes were identified as being differentially expressed in the bGH kidney. Inclusion of two groups, immunoglobulins and inflammation-related genes, suggests a role of the immune system in bGH kidney damage.
Keywords: Growth hormone transgenic mice, immunoglobulins, kidney, cDNA subtraction, microarray, real-time RT/PCR, kidney androgen-regulated protein, inflammation, chemokine (C-C motif) ligand 2/monocyte chemotactic protein-1
1. INTRODUCTION
A multitude of studies have led to the conclusion that GH plays a role in the progression of kidney damage, in the presence or absence of diabetes. Involvement of GH, either directly or through its effector molecule IGF-I, in the early stages of diabetic nephropathy was suggested in two cross-sectional studies in human type I diabetic patients. In one study, a positive correlation was found between serum IGF-I and glomerular filtration rate 1. In another study, a strong positive correlation between urinary IGF-I and kidney volume and between urinary IGF-I, urinary GH, and microalbuminuria was seen 2. Direct evidence for a role of GH in nephropathy came from studies of transgenic mice chronically expressing GH, growth hormone releasing factor (GHRF), or IGF-I 3,4. In the absence of diabetes, significant glomerular enlargement was seen in all three strains of mice. However, mesangial proliferation followed by progressive glomerulosclerosis was found in the GH and GHRF mice but not the IGF-I mice. The GH transgenic mice progressed to end stage renal disease, resulting in premature death 5. Progressive increases of mRNA levels of collagen IV α1, laminin B1, tenascin, transforming growth factor (TGF)-β1 and platelet-derived growth factor (PDGF)-B were also seen in the GH transgenic mice 6.
Additional studies suggested that suppression of the GH signaling pathway can reverse or prevent diabetic kidney complications. Initial evidence came from studies demonstrating the protective effect of hypopituitarism against glomerular basement membrane thickening and the normalization of glomerular basement membrane and mesangial changes caused by diabetes 7,8. Studies of transgenic mice that express a GH receptor antagonist demonstrated protection from glomerulosclerosis during streptozotocin (STZ) induced type I diabetes, even though blood glucose and glycated hemoglobin levels were high 9–11. A molecular analysis of these diabetic transgenic mice revealed normal mRNA and protein levels of glomerular type IV collagen and laminin B1, in contrast to the elevated levels seen in diabetic nontransgenic controls 11,12. It has been shown that STZ-diabetic as well as nonobese diabetic (NOD) mice have elevated levels of circulating GH and low levels of IGF-I, similar to the situation seen in human diabetic patients, suggesting that mice may serve as a good model for diabetes studies 13,14. In a test of therapeutic efficacy for the GH antagonist, administration of the GH antagonist to STZ-diabetic and nonobese diabetic (NOD) mice did not alter the already elevated serum GH levels and reduced serum IGF-I levels seen in the untreated diabetic control mice, but it did reduce the kidney weight, IGF-I accumulation, glomerular volume, and urinary albumin excretion (UAE), indicating a therapeutic role for GH antagonist 13,14. Finally, in a system where GH signaling is completely abolished due to disruption of the GH receptor gene, the kidneys of these mice also were protected against STZ-diabetes induced glomerular hypertrophy and glomerulosclerosis 15.
GH acts through its receptor to initiate a series of signaling cascades that ultimately lead to changes in gene expression 16. While a number of GH-regulated genes have been identified, genes relating GH to kidney damage are still largely unknown. Utilizing the fact that bGH mice develop progressive glomerulosclerosis with increasing age, we chose them as a model system for identifying genes involved in kidney damage. We created cDNA subtraction libraries from kidney RNA of bGH and NT littermates at different ages and validated differential expression of specific clones by northern blot analysis. We also performed microarray analyses comparing gene expression in the kidneys of bGH and NT littermates. Finally, we compared the levels of select gene expression by real-time RT/PCR analysis. Here we identify genes that appear to be differentially expressed in the kidneys of bGH mice as compared to their NT littermates. Consideration of the list of differentially expressed genes suggests involvement of the immune system in the bGH kidney damage.
2. MATERIALS AND METHODS
2.1. Animals
The mice used in this study express a bovine GH cDNA under the control of a mouse metallothionein transcriptional regulatory element. Most of the experiments used mice with a mixed C57Bl6J and SJL background as in previous studies 17–19. The mice used for the microarray study were a pure C57Bl6J genetic background 20. All transgenic mice were compared to NT controls of the same genetic background. Transgenic males were bred with non-transgenic female littermates in the mouse facility of Ohio University’s Edison Biotechnology Institute (EBI). Animals are only accepted into the facility from commercial specific-pathogen-free (SPF) suppliers or from other facilities that have a proven record of clean health. EBI facilities are serology tested for a standard panel of murine viruses four times a year. Necropsy, histology and parasitology are performed twice a year. Mice were housed in microisolator cages, maintained on a 14h light/10h dark cycle and allowed ad libitum access to water and standard rodent chow (Prolab RMH 3000, PMI Nutrition International, Inc., Brentwood, NJ). Genotypes of progeny were confirmed by PCR analysis of genomic DNA obtained by a tail clip using the same method as described for bGH antagonist (GHA) mice 21.
At 2 months, 5 months, or 12 months of age female NT and bGH transgenic mice (n=3 for each genotype and age) were weighed and urine collected for a 2 hour period and then stored at −80°C for albumin and creatinine analyses. The following day the mice were sacrificed by cervical dislocation and trunk blood collected following immediate decapitation. Whole blood was centrifuged at 7000 × g for 10 min at 4°C and serum stored at −80°C for IGF-I and IGFBP analyses. The right kidney was removed and snap-frozen in liquid nitrogen for later isolation of total RNA for cDNA subtraction library creation (n=1 for each genotype and age) and northern analyses. The left kidney was removed, weighed, and a transverse section of the kidney containing the papilla placed in fixative (0.1 M cacodylic acid, 1% glutaraldehyde, and 2% paraformaldehyde) for glomerular analyses by microscopy. Multiple tissues of two additional 12 month old NT females and one additional 12 month old bGH female were collected for subsequent RNA isolation and northern analyses. Kidneys from additional mice at each age also were collected for RNA isolation and northern analyses. Kidneys from 11 month old female bGH and NT mice were obtained for total RNA extraction and use in the microarray (n=3 for each genotype) and real-time RT/PCR analyses (n=5 for bGH and 6 for NT).
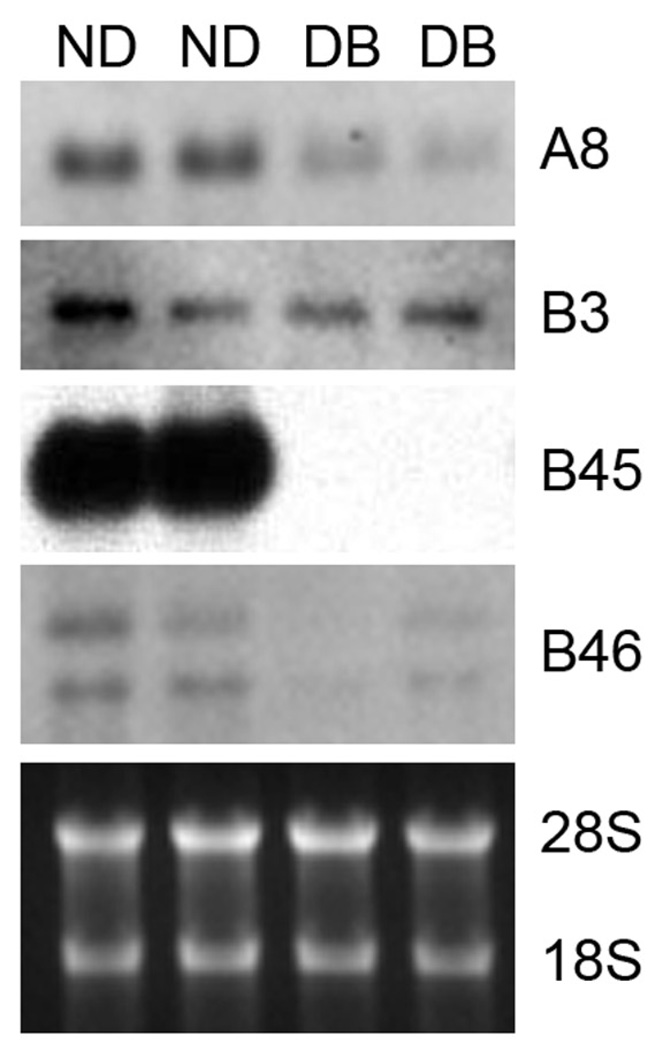
The RNA used for Fig. 6 was from an unrelated diabetes study using female NT mice. At two months of age, the mice received a single intraperitoneal injection of saline or 155 mg streptozotocin (STZ) / kg body weight. Diabetic mice and age-matched saline controls were sacrificed 10 weeks after the onset of diabetes (blood glucose > 300 mg/dl) and the right kidney removed and snap-frozen in liquid nitrogen for later isolation of total RNA. RNA samples from two saline-injected (nondiabetic; ND) and two STZ-induced diabetic (DB) mice were analyzed in the current study.
Fig. 6.
Northern blot analysis of expression of four clones (A8, B3, B45 and B46) in kidneys isolated from non-diabetic (ND) and STZ-induced diabetic (DB) mice. RNA was extracted from whole kidneys of two ND and two DB females 10 weeks after induction of diabetes and probed with DIG-labeled clones. Ethidium-bromide stained images of the 28S and 18S rRNA bands served as loading controls (bottom panel).
Protocols were approved by the Ohio University Institutional Animal Care and Use Committee and followed federal, state, and local guidelines.
2.2. Glomerular and mesangial measurements
Glomerular volume for the 2 month, 5 month and 12 month old mice (n=3 for each genotype and age) was measured as previously described 13. Fixed tissue was embedded in Technovit 7100 (Heracus Kulzer GMBH, Wehrheim, Germany), 2 µm thick sections were cut on a rotation microtome, and sections were stained with peroidic acid-Schiff (PAS) and hematoxylin. Profile areas were traced using a computer-assisted morphometric unit (Image Tool: University of Texas Health Science Center, San Antonio, TX). Glomerular cross-sectional area (AG) was determined as the average area of all of the glomeruli visualized in the section (a range of 33–56 glomeruli; tuft omitting the proximal tubular tissue within Bowman’s capsule). Glomerular tuft volume (VG) was calculated as VG=β/κ×(AG)3/2, where β=1.38, which is the shape coefficient for spheres (the idealized shape of glomeruli), and κ=1.1, which is a size distribution coefficient. The mesangial region was measured by point counting of the PAS stained area as a fraction of the thuft area by applying a grid where about 20 points fell on the PAS stained area in each of about 25 randomly selected glomerular profiles.
2.3. Urinary albumin excretion and serum IGF-I and IGFBP measurements
Urinary albumin concentrations for the 2 month, 5 month and 12 month old mice (n=3 for each genotype and age) were determined by radioimmunoassay using rat albumin antibody and standards as previously described 13. Semilog linearity of mouse urine and rat albumin (in the standard) was found at multiple dilutions, indicating antigen similarity between mouse albumin and rat albumin. Urinary volume output was normalized by urinary creatinine concentration measured using an automated technique adapted from the method of Jaffé 22.
Serum IGF-I levels were measured, after extraction with acid-ethanol, by radioimmunoassay (RIA) using a polyclonal rabbit antibody (Nichols Institute Diagnostics, San Capistrano, CA) and recombinant human IGF-I as standard (Amersham International) as previously described 13. Intra- and interassay coefficients of variation were <5% and <10%, respectively.
Serum IGFBP-1, -2, -3, and -4 levels were assessed by sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) and western ligand blotting (WLB) according to the method of Hossenlopp et al. 23 as described previously 21. Quantification of western ligand blots was done by densitometry using a Shimadzu CS-9001 PC dual-wavelength flying spot scanner (Shimadzu Europe GmbH, Duisburg, Germany). The relative densities of the bands are expressed as pixel intensity.
2.4. Identification of differentially expressed cDNA subtraction clones
Total RNA was isolated from the entire kidney of individual 2 month, 5 month and 12 month old female bGH transgenic mice and non-transgenic controls using RNA STAT60 Total RNA/mRNA Isolation Reagent (Tel-Test, Inc., Friendswood, TX) and small RNAs were removed using the RNeasy Mini Protocol for RNA clean-up (Qiagen Inc., Santa Clarita, CA). cDNA was generated from total RNA using the Smart PCR cDNA Synthesis kit (CLONTECH, Palo Alto, CA). The subtracted cDNA libraries were constructed using the PCR-Select cDNA Subtraction kit (CLONTECH) using RNA from individual mice. Two libraries were created at each time point, a forward subtracted library containing clones up-regulated in bGH mice (bGH library; A clones) as compared to NT mice and a reverse subtracted library containing clones up-regulated in the non-transgenic mice (NT library; B clones) as compared to bGH mice (RNA from a single mouse for each genotype and age were used).
PCR amplified inserts of randomly picked, individual clones from each library were spotted in duplicate on each of two separate positively-charged nylon membranes and hybridized with a random primed 32P labeled probe generated from each entire library using Clontech’s PCR-Select Differential Screening kit (CLONTECH). Potential differentially expressed clones were chosen based on difference in signal intensity upon exposure of the blots to autoradiography film.
Plasmid DNA from bacterial colonies carrying the differentially expressed cDNA insert was isolated using the QIAprep Spin Miniprep Kit according to the manufacturer’s instructions (Qiagen Inc.). Nucleotide sequences were determined by use of the ABI PRISM BigDye Terminator Cycle Sequencing Ready Reaction Kit with electrophoresis on the ABI PRISM 377 DNA Sequencer (PE Applied Biosystems, Foster City, CA). Oligonucleotides that hybridize in the polylinker region of the vector just beyond the insertion site were used to prime the reaction. Nucleotide sequences and predicted amino acid sequences were compared to public domain databases using the BLAST 2.1 and BLAST 2.2.21+ programs 24.
Differential gene expression was confirmed by northern blot analysis (n=2–5 for each genotype and age). 15µg of total kidney RNA was resolved by electrophoresis through a 1% agarose, 1% formaldehyde gel and transferred to a positively-charged nylon membrane. Membranes were hybridized in DIG Easy Hyb solution containing 50 ng/ml of a digoxigenin (DIG) labeled, asymmetric PCR amplified DNA probe synthesized from each clone of interest and detected following the manufacturer’s guidelines (The DIG System User’s Guide for Filter Hybridization, Roche Diagnostics Corp., Indianapolis, IN) and imaged with autoradiography film or a VersaDoc 3000 imaging system (Bio-Rad, Hercules, CA). Images of the ethidium-bromide stained ribosomal RNA bands visualized before transfer served as RNA loading controls.
2.5. Microarray analysis
Microarray analysis was performed essentially as previously described 25. GeneChip Mouse Expression 430 A arrays and hybridization protocols were obtained from Affymetrix (Santa Clara, CA). The array covered 14 484 full length defined genes and 3 771 identified but not full length genes. Six arrays were hybridized with probes made with RNA from kidneys of three individual 11 month old female bGH mice or three individual 11 month old female NT mice (one mouse sample per array with no pooling).
First-strand cDNA synthesis was performed using total RNA (10 µg), a T7-(dT)24 oligomer, and SuperScript II reverse transcriptase (Invitrogen, Carlsbad, CA). Second-strand synthesis converts the cDNA into the DNA template needed for in vitro transcription (IVT). Bacteriophage T7 RNA polymerase directs the synthesis of cRNA. Biotin-tagged NTPs are used in the IVT reaction to produce biotin-labeled cRNA for hybridization. Hybridization of gene chip arrays were performed at 45°C for 16 h in 0.1 M MES, pH 6.6, 1 M sodium chloride, 0.02 M EDTA, and 0.01% Tween 20. Array was washed using both nonstringent (1 M NaCl, 25°C) and stringent (1 M NaCl, 50°C) conditions before staining with phycoerythrin streptavidin. The GeneChips were then scanned with a Hewlett-Packard scanner and analyzed. The p values for detection of individual genes were calculated, and absent calls (detection p value >0.06) were removed. Further data analysis was performed using the Affymetrix EASI database (to assign gene descriptions to query probe sets) and the Spotfire Decision Site software system (Somerville, Mass.). Before comparison analysis, a global normalization method was used to correct for variations and normalize intensity levels. The comparitive analysis was performed by Wilcoxon signed rank test to examine the hybridization intensity data from one gene chip and compare that with another gene chip. Three comparisons were made, each between a bGH hybridized array and an NT hybridized array, and the results were filtered for genes with a greater than two-fold increase or decrease.
2.6. Real-time RT-PCR analysis
Total RNA was isolated from the entire kidney of individual 11 month old female bGH transgenic mice (n=5) and non-transgenic controls (n=6) using RNA STAT60 Total RNA/mRNA Isolation Reagent (Tel-Test, Inc.). RNA samples were treated with DNAse I to remove contaminating genomic DNA and repurified using the RNeasy Micro Kit (QIAGEN). RNA was quantified using the RiboGreen RNA Quantitation Reagent and Kit (Molecular Probes, Eugene, Oregon) and the Versafluor standard spectroflurometer (Bio-Rad, Hercules, CA). Synthesis of cDNA was performed using 1 µg of the isolated RNA and the iScript™ cDNA Synthesis Kit (Bio-Rad). Samples were analyzed for relative target RNA concentration via real-time RT-PCR analysis in duplicate using gene specific primers (see Tables 2 and 3 for target primer sequences) and the iQ Sybr Green Supermix Kit (Bio-Rad) in a MyiQ™ Single Color Real-Time PCR Detection System (Bio-Rad). Primer sequences were obtained from the literature, a primer database 26 or designed using the Primer3 program 27 and checked for specificity using BLAST analysis of the mouse nucleotide databases 24. Results were normalized using the geometric mean of expression of the two best control genes (HGPRT and γ-actin) out of six assessed (see Table 2) using the NormFinder application 28.
Table 2.
Validation of differential expression between bGH and NT kidney RNA of genes identified by cDNA subtraction or microarray analysis using real-time RT/PCR
| Clone name/ accession number |
Gene | Primer sequences (5’- 3’) | FC: RT-RT/PCR (relative to NT) † |
FC: Microarray (relative to NT) ‡ |
|---|---|---|---|---|
| A8 (XM_001476703.1) | Similar to Chain L, Structural Basis Of Antigen Mimicry In A Clinically Relevant Melanoma Antigen System, transcript variant 3 |
(+) GGCCACTCACAAGACATCAA (−) AAAGGGAGGAGGAGAAGGAG a |
4.79 ± 3.24 * | |
| M92849 | Acidic Epididymal Glycoprotein | (+) TGCATGTGGAGTTGCTGAAT (−) GGTGCATAGCCCATCTTCAC |
293.58 ± 224.21 * | 26.91 |
| X51547 | Lysozyme P | (+) CTCTGGGACTCCTCCTGCTT (−) CATAGTCGGTGCTTCGGTCT |
7.90 ± 2.92 * | 7.52 |
| X97227 | Cell Surface Glycoprotein CD53 | (+) TGGCAACATTCTGGTCATTG (−) CATTCAGACCCTCAGCCACT |
3.81 ± 1.32 * | 5.74 |
| AB006361 | Prostaglandin D Synthetase | (+) TCCGGGAGAAGAAAGCTGTA (−) ATAGTTGGCCTCCACCACTG |
2.13 ± 0.76 * | 5.54 |
| M55561 | CD80 antigen; also known as B7-1 | (+) CATCTCTCAAAGCTGCTCCA (−) GGATGAGGCCCCACTCTTTA |
2.30 ± 0.42 * | 4.79 |
| B3 (NT_039433.7) | No known transcript | (+) GGAGTTCATGCAAGGTGGAT (−) CAGCAGCATGTGATCCTCAG |
0.82 ± 0.21 | |
| B45 (NM_010594.2) | Kidney androgen-regulated protein (KAP) |
(+) ACAGTCTCCTCCGGCTTTCT (−) TTCTTTGCTGGGAGCATCTT |
0.20 ± 0.14 * | 0.08 |
| B46 (NM_027921.1) | Solute carrier family 16 member 14 | (+) CAGCCAATGTGCAGTCTCTC (−) CGCACAGGTATTTCAGCAAC |
0.42 ± 0.20 | |
| AJ000062 | DNAse I | (+) CCTTACCATAGCTGGGTGGA (−) GACTCAGGATCTGCGGGTAA |
0.31 ± 0.11 * | 0.09 |
| U31966 | Carbonyl Reductase | (+) CCACATTCAAGCAGAGGTGA (−) CAGCTCCTCCTCTGTGATGG |
0.66 ± 0.12 * | 0.34 |
| L42996 | Mitochondrial Acyltransferase | (+) TGTCCCATGATGAGCACACT (−) GAGGCAGTATGGCTCCTGTC |
0.70 ± 0.10 * | 0.41 |
| BC085275 | Glyceraldehyde-3-phosphate dehydrogenase (GAPDH) |
(+)TGTGTCCGTCGTGGATCTGA (−)CCTGCTTCACCACCTTCTTGA |
Control b | |
| BC021796 | γ-actin | (+) ACCAACAGCAGACTTCCAGGAT (−) AGACTGGCAAGAAGGAGTGGTAA |
Control c | |
| X03672 | β-actin | (+) AGCCATGTACGTAGCCATCCA (−) TCTCCGGAGTCCATCACAATG |
Control c | |
| BC083145 | Hypoxanthine guanine phosphoribosyl transferase (HGPRT) |
(+) CCTAAGATGAGCGCAAGTTGAA (−) CCACAGGACTAGAACACCTGCTAA |
Control c | |
| U63933 | TATA box binding protein (TBP) | (+) GGCCTCTCAGAAGCATCACTA (−) GATGGGAATTCCAGGAGTCA |
Control c | |
| AF285162 | Ubiquitin C (UBC) | (+) AGGTCAAACAGGAAGACAGACGTA (−) TCACACCCAAGAACAAGCACA |
Control c |
Fold change: Mean of bGH results normalized by geometric mean of HGPRT and γ-actin divided by mean of NT results normalized by geometric mean of HGPRT and γ-actin as determined by real-time RT/PCR; n = 5–6
Fold change: Mean of three independent pairwise comparisons between bGH and NT microarrays
P<0.05
(−) primer sequence based on M. spretus #M21796 which is missing three nucleotides in comparison to M. musculus sequence
Primer sequences from Fujimoto et al. 51
Primer sequences from RTPrimerDB 26
Table 3.
Assessment of differential expression between bGH and NT kidney RNA of inflammation-related genes using real-time RT/PCR
| Accession number |
Gene | Primer sequences (5’- 3’) | FC: Expression (relative to NT)† |
|---|---|---|---|
| NM_011333.3 | Chemokine (C-C motif) ligand 2 (CCL2); also known as monocyte chemotactic protein-1 (MCP-1) |
(+) CCCAATGAGTAGGCTGGAGA (−) AGACCTTAGGGCAGATGCAG |
7.52 ± 2.76 * |
| NM_008605.3 | Matrix metallopeptidase 12 (MMP-12); also known as macrophage elastase |
(+) TTTCTTCCATATGGCCAAGC (−) GGTCAAAGACAGCTGCATCA |
2.23 ± 0.58 * |
| NM_009853.1 | Cluster of differentiation 68 (CD68); also known as also known as macrosialin |
(+) CTTCCCACAGGCAGCACAG (−) AATGATGAGAGGCAGCAAGAGG |
2.05 ± 0.37 * |
| NM_013653.2 | Chemokine (C-C motif) ligand 5 (CCL5); also known as Regulated upon activation, normal T cell expressed and secreted (RANTES) |
(+) GTGCCCACGTCAAGGAGTAT (−) AGCAAGCAATGACAGGGAAG |
2.29 ± 0.75 |
| NM_011693.2 | Vascular cell adhesion molecule-1 (VCAM-1) | (+) ATTTTCTGGGGCAGGAAGTT (−) ACGTCAGAACAACCGAATCC |
2.10 ± 0.46 |
| X93328.1 | Antigen F4/80 | (+) CTTGGCTATGGGCTTCCAGTC (−) GCAAGGAGGACAGAGTTTATCGTG |
1.52 ± 0.37 |
| NM_009931.1 | Procollagen, type IV, alpha 1 | (+) CCTCCAGGTTTCCCTACTCC (−) TCCAGGTTGACACTCCACAA |
1.46 ± 0.48 |
| NM_010217.1 | Connective tissue growth factor (CTGF) | (+) TGGAGCGCCTGTTCTAAGAC (−) GCAGCCAGAAAGCTCAAACT |
1.38 ± 0.27 |
| NM_008689.2 | Nuclear factor kappa-light-chain-enhancer of activated B cells (NFκB) |
(+) CGGCAACTCACAGACAGAGA (−) ACGATTTTCAGGTTGGATGC |
1.28 ± 0.21 |
Fold change: Mean of bGH results normalized by geometric mean of HGPRT and γ-actin divided by mean of NT results normalized by geometric mean of HGPRT and γ-actin as determined by real-time RT/PCR; n = 5–6
P<0.05
2.7. Statistical analyses
All values are expressed as mean ± SEM. All parameters were statistically analyzed using analysis of variance (ANOVA) comparing bGH animals with age-matched littermate controls. Results were considered statistically significant at P < 0.05.
3. RESULTS
3.1. Mouse characteristics
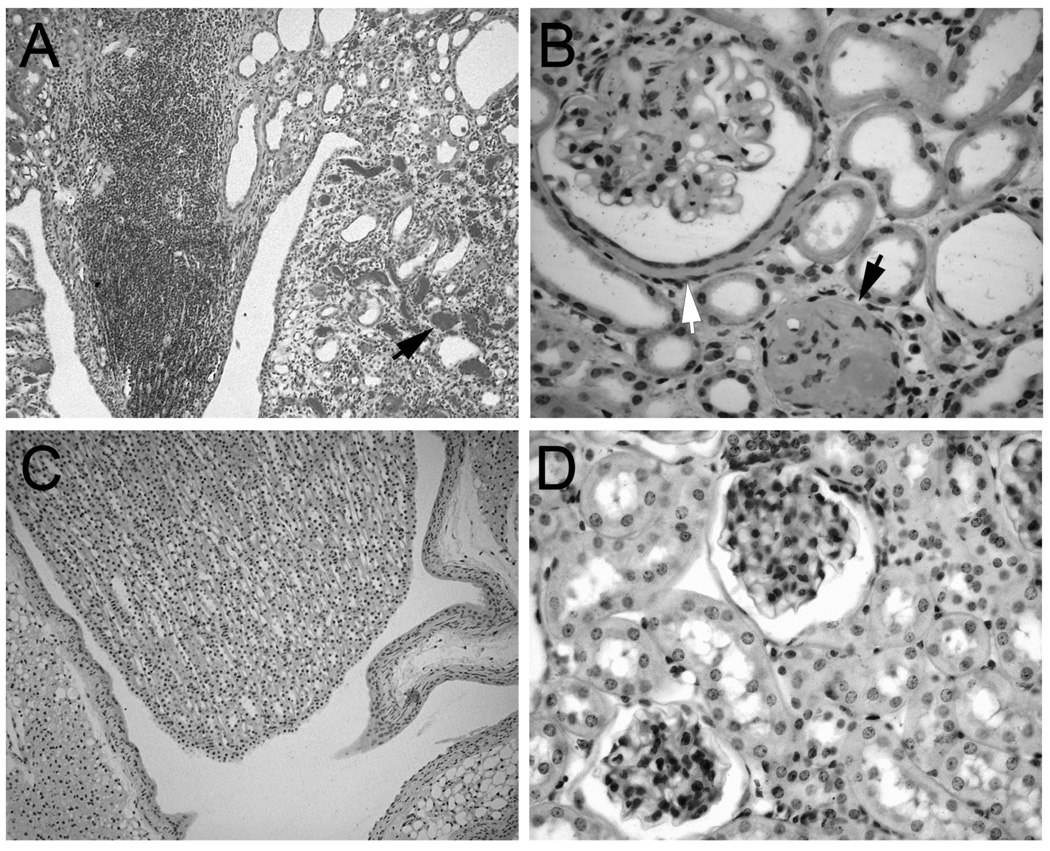
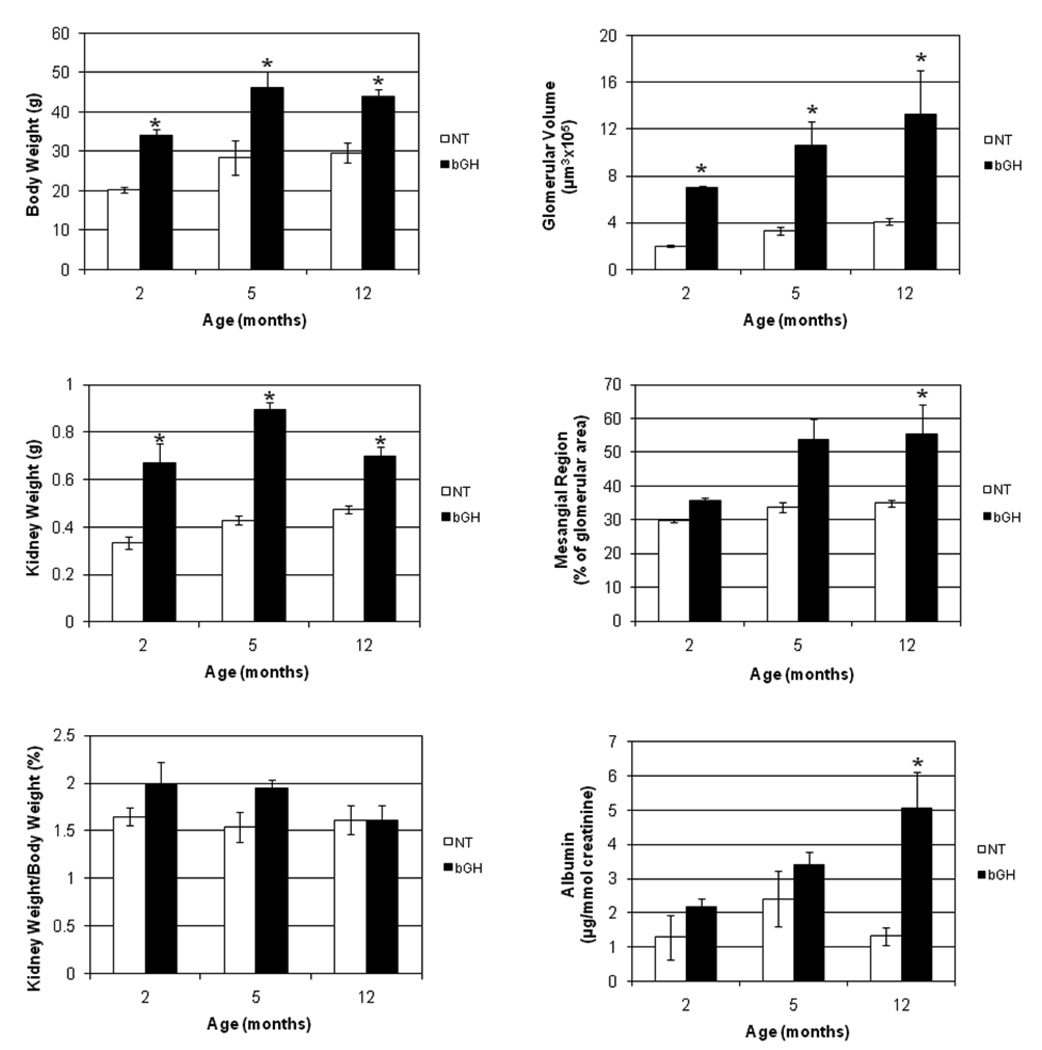
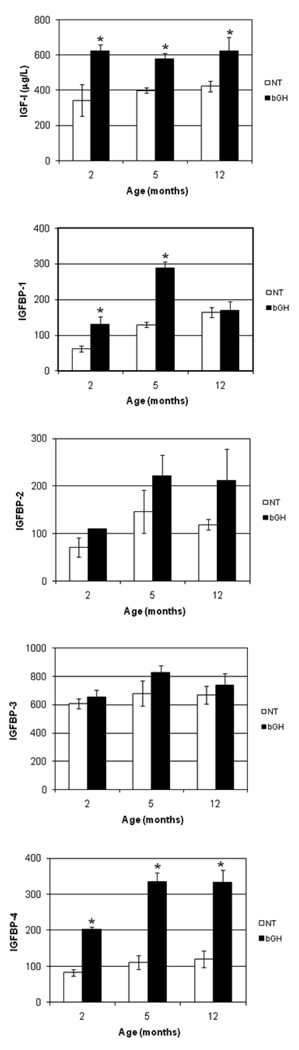
In an effort to identify genes involved in the progression of kidney damage, three ages (2, 5, and 12 months) were selected for the comparison of gene expression between NT and bGH female mice. Based on previous studies of these mice, it was anticipated that the bGH mice would exhibit increasing degrees of kidney damage in comparison to the NT mice at the three respective ages 4. Histopathological examination of PAS-stained sections supported this premise. Light microscopy revealed that, with increasing age, the bGH kidneys progressed from mild inflammation around the pelvis at 2 months of age to more diffuse inflammation at 5 months of age and finally to diffuse inflammation and mononuclear cell infiltration around the pelvis at 12 months of age (Fig. 1A). At higher magnification, the infiltrate looked like lymphocytes, plasma cells and macrophages, but immunohistochemistry was not performed to definitively identify the cell types. In parallel, dilated tubules were noted in some of the bGH mice at 2 months of age, large glomeruli were noted at 5 months of age, and thickened, PAS-positive Bowman’s capsules and protein in the tubules were noted at 12 months of age (Fig. 1B). In contrast, a very mild inflammation around the pelvis limited to the subepithelial region was noted in two of the 12 month NT mice, but all other NT kidneys appeared normal (Fig. 1C and D). A quantitative assessment of the mice and kidneys is shown in Fig. 2. The physical characteristics of absolute body and kidney weights were significantly greater for the bGH mice at all three ages. When normalized to body weights, kidney weights were not significantly different at any age. Absolute glomerular volume was significantly greater in bGH mice at all three ages (Fig. 2). This increase was significant only at 2 months of age when normalized to body weight or kidney weight (data not shown). Mesangial region and urinary albumin excretion were significantly increased in bGH mice only at 12 months (Fig. 2). IGF-I and IGF binding protein levels also were assessed in these same mice (Fig. 3). IGF-I and IGFBP-4 were significantly increased in bGH mice at all three ages. IGFBP-1 was significantly increased only at 2 and 5 months of age. IGFBP-2 and IGFBP-3 showed no significant differences at any age. Our results were similar to those obtained in an earlier study of these mice in terms of body weight, serum IGF-I levels, glomerular volume and urinary albumin excretion 4. In addition, the previous study reported marked mesangial sclerosis in the bGH mice at 2–3 months of age and severe mesangial sclerosis at 6–9 months of age.
Fig. 1.
Histopathology of bGH and NT kidneys (PAS-stained sections). A. Pelvic region of a kidney of a 12 month old bGH mouse showing a papilla with lymphocytes and plugged tubules (arrow). B. A section from the kidney of a 12 month old bGH mouse showing a glomerulus with a thickened Bowman’s capsule (white arrow) and a closed glomerulus (black arrow). C. Pelvic region of a kidney of a 2 month old NT mouse showing a normal papilla. D. A section from the kidney of a 12 month old NT mouse showing two normal glomeruli. A, C: low magnification (same for both); B, D: high magnification (same for both).
Fig. 2.
Growth, morphological, and physiological assessments of NT and bGH females at three different ages. The left panels compare body weight, kidney weight and kidney weight normalized to body weight (expressed as a percentage) and the right panels compare glomerular volume, mesangial region (expressed as percentage of glomerular area) and urinary albumin (normalized to creatinine concentration) for NT (□) and bGH (■) females at 2, 5, and 12 months of age. *, significantly different from corresponding NT (P < 0.05); error bars indicate SEM; n = 3.
Fig. 3.
Comparison of serum IGF-I concentration (determined by RIA) and IGFBP-1, -2, -3, and -4 pixel intensity (determined by WLB) for NT (□) and bGH (■) females at 2, 5, and 12 months of age. *, significantly different from corresponding NT (P < 0.05); error bars indicate SEM; n = 3.
3.2. Characterization of clones isolated from cDNA subtraction libraries
Two PCR-based cDNA subtraction libraries were created for each age with one library containing genes expressed at higher levels in the bGH kidney and the other containing genes expressed at higher levels in the NT kidney. Individual clones from the libraries were screened by differential hybridization using probes generated from each library. Of 192 clones screened from the 2 month libraries and 96 clones screened from the 5 month libraries, none could be confirmed as differentially expressed between NT and bGH mice. Of 96 clones screened from the 12 month libraries, seven were confirmed by northern blot analysis as differentially expressed between NT and bGH mice. Nucleotide sequencing and BLAST analysis suggested that the four clones up-regulated in bGH kidney (A clones) were immunoglobulin genes (Table 1). Of the three clones down-regulated in bGH mice (B clones), one did not correspond to any known transcript, one corresponded to the gene encoding kidney androgen regulated protein and the third corresponded to the gene encoding solute carrier family 16, member 14 (Table 1). Clone B45 appeared to be a full-length clone while the others appeared to be partial clones.
Table 1.
cDNA subtraction clone size and identity based on BLAST
| Clone | Insert (bp) | Gene Name of Best BLAST Match | Accession # |
|---|---|---|---|
| A8 | 334 | Similar to Chain L, Structural Basis Of Antigen Mimicry In A Clinically Relevant Melanoma Antigen System, transcript variant 3 |
XM_001476703.1 |
| A34 | 503 | Immunoglobulin heavy chain 6 (heavy chain of IgM) | NT_166318.1 |
| A39 | 482 | Immunoglobulin heavy chain (gamma polypeptide), transcript variant 1 (Ighg) |
XM_001001076.2 |
| A48 | 401 | Immunoglobulin heavy constant gamma 1 (G1m marker) |
NT_166318.1 |
| B3 | ~1770 | No known transcript | NT_039433.7 |
| B45 | 610 | Kidney androgen regulated protein (Kap) | NM_010594.2 |
| B46 | >2000 | Solute carrier family 16 (monocarboxylic acid transporters), member 14 (Slc16a14) |
NM_027921.1 |
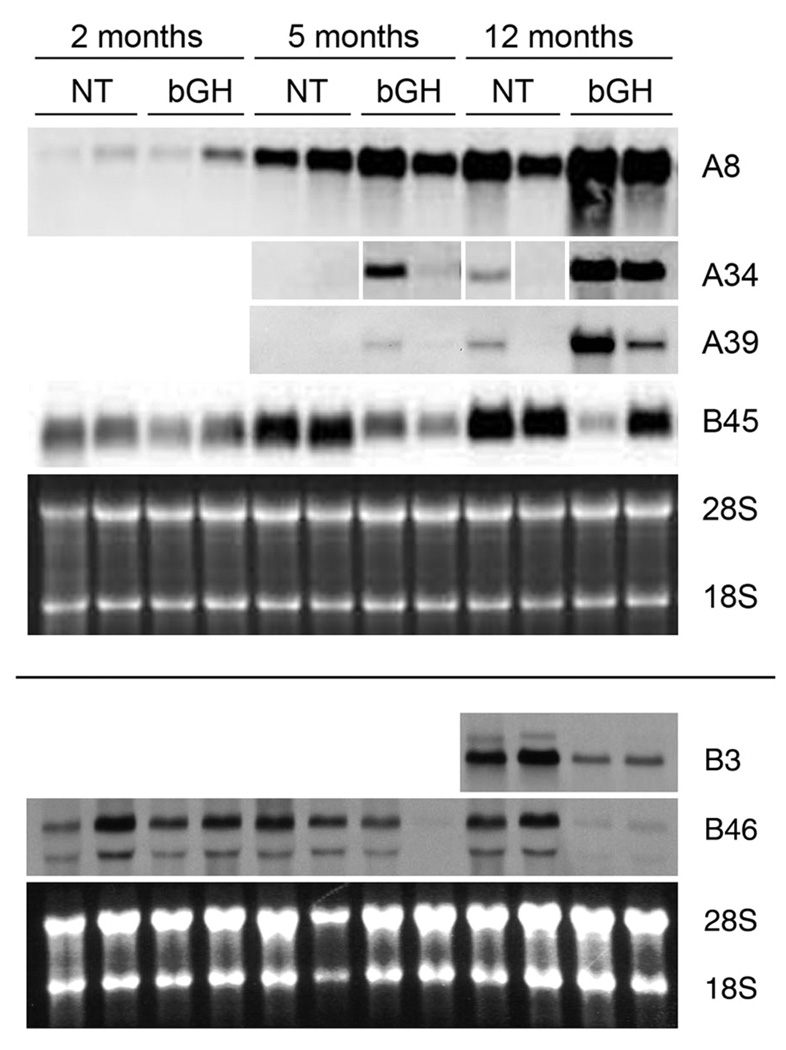
Northern blot analyses were performed to assess the expression of the isolated clones in several different circumstances. First, expression was assessed at different ages of NT and bGH mice. Expression of the four A clones (immunoglobulins) was similar in that RNA levels were greater in bGH kidneys than in age-matched NT kidneys and were greater at the older ages (Fig. 4 and data not shown). Clone B3 was only assessed at the 12 month age, and its expression was decreased in the bGH kidneys (Fig. 4). B45 expression in the NT increased with age, but expression was reduced in the age-matched bGH kidneys (Fig. 4). B46 expression was most obviously decreased in the 12 month old bGH kidneys (Fig. 4).
Fig. 4.
Northern blot analysis of expression of six clones (A8, A34, A39, B3, B45 and B46) obtained from the cDNA subtraction libraries. RNA was extracted from kidneys of NT and bGH females at 2, 5, and 12 months of age and probed with DIG-labeled clones. Representative blots of two different samples for each genotype and age are shown. The same RNA samples were used for the four blots in the top section (the samples for A34 were loaded in a different order; the panels were rearranged such that all samples align; the white gaps mark the rearrangements) and the same RNA samples were used for the two blots in the bottom section, though some of the samples differed between the top and bottom sections. Ethidium-bromide stained images of the 28S and 18S rRNA bands served as loading controls (bottom panel of each section).
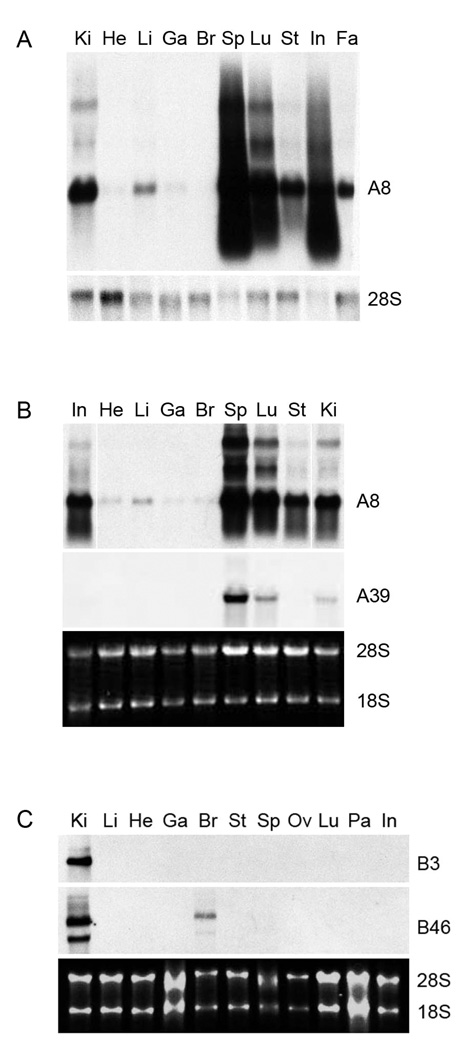
Tissue specificity was also examined for several of the clones (Fig. 5). Clone A8 was most strongly expressed in spleen, followed by lung, intestine, kidney, stomach, fat and liver of both NT and bGH mice at 12 months of age. Clone A39 was highest in spleen, followed by lung and kidney. The expression patterns of Clones A34 and A48 were very similar to those for A39 (data not shown). The high expression of the A clones in spleen agrees with their identification as immunoglobulin genes. Clone B3, the unidentified clone, was only detected in kidney. Clone B46 levels were highest in kidney but also were detectable in brain. Clone B45 was not tested, but the expression of the kidney androgen-regulated protein gene predominantly in epithelial cells of the renal proximal tubule has already been reported 29.
Fig. 5.
Northern blot analysis of expression of four clones (A8, A39, B3 and B46) obtained from the cDNA subtraction libraries. RNA was extracted from indicated tissues of a single NT (A and C) or single bGH (B) female at 12 months of age and probed with DIG-labeled clones. DIG-labeled 28S rRNA probe (bottom panel of A) or ethidium-bromide stained images of the 28S and 18S rRNA bands (bottom panel of B and C) served as loading controls. Ki, kidney; He, heart; Li, liver; Ga, gastrocnemius muscle; Br, brain; Sp, spleen; Lu, lung; St, stomach; In, intestine; Fa, fat (white adipose tissue); Ov, ovary; Pa, pancreas.
To assess expression of the clones in a diabetic mouse model, RNA levels were examined in streptozotocin-induced type 1 diabetic mice (Fig. 6). Clone B3 showed little to no change in expression in kidneys after 10 weeks of diabetes (DB) in comparison to kidneys from non-diabetic mice (ND). Clone A8 and A48 showed a decrease in expression in the diabetic samples (Fig. 6 and data not shown), just the opposite of the pattern seen in the bGH kidney damage model. RNA for clones B34 and B39 was undetectable in all four samples (data not shown). Clone B46 showed a moderate decrease in expression in the diabetic kidneys while Clone B45 showed a severe decrease in expression in the diabetic kidneys. Both Clone B45 and B46 exhibited decreases in expression in damaged kidneys, but the decrease for Clone B45 was more dramatic in the STZ-diabetes kidneys while the decrease for Clone B46 was more dramatic in the bGH kidneys (compare Fig. 4 and Fig. 6).
3.3. Identification of differentially expressed genes by microarray analysis
Microarray analysis was also used to identify genes that were differentially expressed between the kidneys of NT and bGH mice. Of 18 255 genes, 127 were increased greater than 2 fold for all three array comparisons while 26 were decreased greater than 2 fold for all three array comparisons of the bGH kidneys to the NT kidneys (Supplemental Table). Many of the genes up-regulated in the bGH kidneys, especially those showing the largest increases, were immunoglobulins, as was found with the cDNA subtraction libraries. The kidney androgen-regulated protein gene was the second most down-regulated gene in the bGH kidneys. This gene was also identified in the cDNA subtraction libraries.
3.4. Verification of differential gene expression by real-time RT-PCR analysis
Real-time RT/PCR analysis was used to quantify the differential gene expression for several of the cDNA subtraction clones and to verify the differential gene expression of some of the genes identified in the microarray analysis using RNA isolated from whole kidney of 11 month old female mice (Table 2). Clone A8 showed a significant increase of nearly 5-fold in the 11 month old bGH compared to NT kidneys. A slight but non-significant decrease was seen for Clone B3. Clone B45, corresponding to the kidney androgen regulated-protein gene, was decreased 5-fold, a little less than the 13.18-fold decrease seen in the microarray analysis. Clone B46 was decreased greater than 2-fold in the bGH kidneys, though the difference did not reach statistical significance. From the microarray analysis, five genes not related to immunoglobulins were confirmed as being significantly up-regulated in the bGH kidney and three additional genes were confirmed as being significantly down-regulated in bGH kidney. Six others showed no significant difference in expression, including the gene identified by microarray analysis as exhibiting the greatest decrease in expression in the bGH kidney (Supplemental Table). Expression of each of the genes in Table 2 was also compared between two genetic backgrounds, the mixed C57Bl6J/SJL background and the pure C57Bl6J background. The results were similar (data not shown).
3.5. Additional differential gene expression verified by real-time RT-PCR analysis
An interest in inflammation led to analysis of gene expression of nine additional genes with various roles in inflammation pathways or outcomes (Table 3). All were up-regulated in bGH kidneys, but only three showed a significant increase: chemokine (C-C motif) ligand 2 (CCL2; also known as monocyte chemotactic protein-1 or MCP-1), matrix metallopeptidase 12 (MMP-12; also known as macrophage elastase), and cluster of differentiation 68 (CD68; also known as macrosialin).
4. DISCUSSION
Evaluation of the bGH transgenic mice used in this study suggested that they developed a similar, though possibly milder, form of kidney damage as compared to reports of other GH transgenic mice generated in other labs. For example, for the mice in this study, kidney weight was increased at all of the ages examined (2, 5 and 12 months), but the increase was proportional to body weight gain. Glomerular volume was greatly increased as early as two months of age, but when normalized to body weight or kidney weight, the increase was only apparent at 2 months of age. While a greater than proportional increase in kidney weight and glomerular volume was commonly reported for some of the other transgenic lines 30, proportional changes were also observed, possibly reflecting species differences of the foreign GH or differing levels of GH overproduction in the mice 31. Increased mesangial region was obvious by 12 months of age for the mice in this study, at which time urinary albumin excretion was also elevated. Both of these are indications of kidney damage seen previously in bGH mice 32, although they seem to occur at later ages in our mice. This could be indicative of a milder form of damage or could be due to differences in the way these parameters were analyzed. Although serum bGH levels were not measured in the current study, a previous study reported serum bGH levels of 0.5–6 µg/ml for this mouse line, which was consistent with reports for transgenic lines from other labs 33. An informal perusal of mouse records for natural deaths of the bGH line used in the current study revealed a mean age of death of 213 days for bGH males (n=46) and only 61% alive at six months of age and a mean of 258 days for bGH females (n=25) and only 76% alive at six months of age, more than a two-fold reduction of the mean for NT mice (K. Coschigano, unpublished observations), and a lower survival rate than that reported for bGH mice from other labs 30. Although the small sample number was also a potential limitation of the current study, the concurrence of our results and those of an earlier study of these mice suggests that was not an issue 4.
As seen previously for other GH transgenic mice, serum IGF-I expression was elevated from 2 to 12 months of age in our bGH mice. In parallel, serum IGFBP-4 expression was also elevated at all ages. IGFBP-1 was elevated only at 2 and 5 months of age. Although appearing slightly elevated, the levels of IGFBP-2 and -3 were not significantly altered. The levels of IGFBPs differed from reports of elevated IGFBP-3 expression and decreased IGFBP-2 expression in other GH transgenic mice 31, but this might reflect differences in the GH transgenes or promoters (i.e. species, integration site, etc.). The roles of the IGFBPs are not fully elucidated, but circulating IGFBPs are thought to regulate the half-life and endocrine effects of IGF 34. Thus, the increased levels of circulating IGFBPs may be reducing the bioavailability of IGF-I in the bGH mice. It was not determined whether the increased levels of IGFBPs reduced the bioavailability of IGF-I to a level lower than seen in the NT mice, but this was probably not the case since the growth of the bGH transgenic mice was greater than the NT mice.
In our gene expression analyses, we found that the vast majority of genes with increased RNA expression in the kidneys of bGH females, as identified by cDNA subtraction and microarray analyses at the age of 11–12 months, were immunoglobulins. Their expression was greater in bGH as compared to age-matched NT samples and increased with age with the most obvious expression being in the 12 month samples. The identification of immunoglobulin RNA suggests the presence of B cells, perhaps accumulating as a secondary response to an as yet unidentified phenomonen, rather than via intrinsic expression by the kidney. The observation of mononuclear cell infiltration also suggests the presence of B cells. A report by Ogueta et al. demonstrated the development of an arthritic disorder and expression of self-antibodies in transgenic mice expressing bGH under the transcriptional control of the phosphoenolpyruvate carboxykinase promoter, a different promoter from the one used in this study 35. Their findings suggested that the kidney damage seen in those mice might be due to an autoimmune-like process caused by the chronic high GH/IGF levels. The down-regulation of DNAse I expression, which has been linked to the development of autoimmunity 36, and the up-regulation of immmunoglobin gene expression supports a similar conclusion in our bGH mice. Definitive evidence for this conclusion would require identification of self-antibodies and immune-mediated glomerular lesions in our bGH mice, studies that we plan to do in the future.
The presence of activated B cells in bGH kidney is also supported by the up-regulation of two additional genes. Expression of CC chemokine ligand 2, also known as monocyte chemotactic protein-1 or MCP-1, has been observed in mesangial cells, endothelial cells, tubular epithelial cells and infiltrating mononuclear cells of diseased kidneys 37. Frade et al. showed that MCP-1 could trigger specific B cell migration 38. CD80, also known as B7-1, has a well-established role in B cell co-stimulation, but also appears to mediate transmembrane outside-in signaling in B cells and to be involved in the development of podocyte foot process effacement 39. Thus, these two genes could be involved in B cell mobilization and accumulation within the bGH kidney. Since our studies were performed with RNA extracted from the entire kidney, future studies will be aimed at identifying the exact cells that express these two genes within the bGH kidney to help clarify their role.
To gain further insight into the molecular mechanism(s) that may be contributing to the bGH kidney damage, the possible roles of additional up-regulated genes was considered. Macrophage metalloelastase, also referred to as MMP-12, is mainly produced by infiltrating macrophages to break down extracellular matrix components such as fibronectin, laminin and collagen IV, but it has also been co-localized with MCP-1 in podocytes in the absence of macrophages but in association with glomerular basement membrane pathogenesis 40. CD53, a member of the tetraspanin family of membrane-spanning glycoproteins, is expressed mainly in lymphoid and myeloid cells 41, including mesangial cells, which are derived from the myeloid lineage 42. CD68, known as macrosialin in mice, is a glycosylated transmembrane protein also expressed mainly in myeloid cells 43. Lysozyme RNA expression and secretion is associated with activation of macrophages 44, but its expression has also been observed in the proximal tubules of the kidney, possibly reflecting the role of the kidney in clearing lysozyme from the body 45. Prostaglandin D synthetase expression has been associated with antigen-presenting cells 46, which could include mesangial cells. Speculating that inflammatory processes were involved in GH-induced kidney damage, Rezende et al. also found that a number of inflammatory markers were upregulated in the bGH kidney, several (toll-like receptors [TLR] -2 and -4, MCP-1 and smooth muscle actin [SMA]) as early as 5 weeks of age, the earliest age examined by the authors 47. Toll-like receptors are expressed on leukocyte subsets and non-immune cells and appear to be involved in both innate and adaptive immune responses 48. Considered together, the altered expression of all of these genes suggests infiltration by macrophages in addition to B cells within the bGH kidney. Again, further studies are required to confirm this supposition.
Interestingly, only one gene was identified in both the cDNA subtraction libraries and the microarray analysis as being down-regulated in the bGH kidney. It was also identified in a different set of cDNA subtraction libraries comparing RNA expression of kidneys from diabetic and non-diabetic mice (Coschigano, unpublished results). In each case, this gene, which encodes kidney androgen-regulated protein, or KAP, demonstrated decreased expression in kidneys exhibiting kidney damage. KAP expression is confined almost exclusively to epithelial cells of the renal proximal tubule 29. Although the function of KAP remains elusive, it has been demonstrated that the protein specifically interacts with cyclophilin B and that overexpression of KAP in stably transfected proximal tubule cells significantly decreases the toxic effects of cyclosporine A 49. Over-expression of the KAP gene in the kidneys of mice induced hypertension, suggesting a role in cardiovascular-renal homeostasis 50. While it is not clear what function it is performing in the kidney, the loss of KAP expression appears to associate with indicators of kidney damage.
In summary, comparison of gene expression between kidneys of female bGH mice and their non-transgenic littermates revealed a number of differentially expressed genes. Inclusion of two groups, immunoglobulins and inflammation-related genes, suggests a role of the immune system resulting in bGH kidney damage. Confirmation of the regulation of these genes by GH, elucidation of the exact roles of the differentially expressed genes in the bGH kidney, and applicability of these genes and mechanisms to other forms of kidney damage await further investigation.
Supplementary Material
ACKNOWLEDGEMENTS
We would like to thank Chad Keller for technical advice and student oversight and Drs. Maria Cecilia Courreges and Ramiro Malgor for helpful comments and discussion regarding the manuscript.
This work was supported by the State of Ohio’s Eminent Scholar program, which includes a grant from Milton and Lawrence Goll; WADA; Endocrine Society Summer Research Opportunities for Minority Students award; NIH grants 1 R15 DK061952-01, 1 R15 DK081192-01, R15DK075436, RO1 AG19899-05, and P01 AG031736-01A1; and funds from Ohio University and Ohio University’s College of Osteopathic Medicine.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
REFERENCES
- 1.Sen A, Buyukgebiz A. Albumin excretion rate, serum insulin-like growth factor-I and glomerular filtration rate in type I diabetes mellitus at puberty. J. Pediatr. Endocrinol. Metab. 1997;10:209–215. doi: 10.1515/jpem.1997.10.2.209. [DOI] [PubMed] [Google Scholar]
- 2.Cummings EA, Sochett EB, Dekker MG, Lawson ML, Daneman D. Contribution of growth hormone and IGF-I to early diabetic nephropathy in type 1 diabetes. Diabetes. 1998;47:1341–1346. doi: 10.2337/diab.47.8.1341. [DOI] [PubMed] [Google Scholar]
- 3.Doi T, Striker LJ, Quaife C, et al. Progressive glomerulosclerosis develops in transgenic mice chronically expressing growth hormone and growth hormone releasing factor but not in those expressing insulinlike growth factor-1. Am. J. Pathol. 1988;131:398–403. [PMC free article] [PubMed] [Google Scholar]
- 4.Yang CW, Striker LJ, Pesce C, et al. Glomerulosclerosis and body growth are mediated by different portions of bovine growth hormone: Studies in transgenic mice. Lab. Invest. 1993;68:62–70. [PubMed] [Google Scholar]
- 5.Pesce CM, Striker LJ, Peten E, Elliot SJ, Striker GE. Glomerulosclerosis at both early and late stages is associated with increased cell turnover in mice transgenic for growth hormone. Lab. Invest. 1991;65:601–605. [PubMed] [Google Scholar]
- 6.Yang CW, Striker GE, Chen WY, Kopchick JJ, Striker LJ. Differential expression of glomerular extracellular matrix and growth factor mRNA in rapid and slowly progressive glomerulosclerosis: Studies in mice transgenic for native or mutated growth hormone. Lab. Invest. 1997;76:467–476. [PubMed] [Google Scholar]
- 7.Ireland JT, Patnaik BK, Duncan LJ. Effect of pituitary ablation on the renal arteriolar and glomerular lesions in diabetes. Diabetes. 1967;16:636–642. doi: 10.2337/diab.16.9.636. [DOI] [PubMed] [Google Scholar]
- 8.Merimee TJ, Fineberg SE, Hollander W. Vascular disease in the chronic HGH-deficient state. Diabetes. 1973;22:813–819. doi: 10.2337/diab.22.11.813. [DOI] [PubMed] [Google Scholar]
- 9.Chen NY, Chen WY, Bellush L, et al. Effects of streptozotocin treatment in growth hormone (GH) and GH antagonist transgenic mice. Endocrinology. 1995;136:660–667. doi: 10.1210/endo.136.2.7835300. [DOI] [PubMed] [Google Scholar]
- 10.Chen NY, Chen WY, Kopchick JJ. A growth hormone antagonist protects mice against streptozotocin induced glomerulosclerosis even in the presence of elevated levels of glucose and glycated hemoglobin. Endocrinology. 1996;137:5163–5165. doi: 10.1210/endo.137.11.8895392. [DOI] [PubMed] [Google Scholar]
- 11.Liu ZH, Striker LJ, Phillips C, et al. Growth hormone expression is required for the development of diabetic glomerulosclerosis in mice. Kidney Int. 1995 Suppl.:S37–S38. 5196039526. [PubMed] [Google Scholar]
- 12.Esposito C, Liu ZH, Striker GE, et al. Inhibition of diabetic nephropathy by a GH antagonist: A molecular analysis. Kidney Int. 1996;50:506–514. doi: 10.1038/ki.1996.342. [DOI] [PubMed] [Google Scholar]
- 13.Flyvbjerg A, Bennet WF, Rasch R, Kopchick JJ, Scarlett JA. Inhibitory effect of a growth hormone receptor antagonist (G120K-PEG) on renal enlargement, glomerular hypertrophy and urinary albumin excretion in experimental diabetes in mice. Diabetes. 1999;48:377–382. doi: 10.2337/diabetes.48.2.377. [DOI] [PubMed] [Google Scholar]
- 14.Segev Y, Landau D, Rasch R, Flyvbjerg A, Phillip M. Growth hormone receptor antagonism prevents early renal changes in nonobese diabetic mice. J. Am. Soc. Nephrol. 1999;10:2374–2381. doi: 10.1681/ASN.V10112374. [DOI] [PubMed] [Google Scholar]
- 15.Bellush LL, Doublier S, Holland AN, Striker LJ, Striker GE, Kopchick JJ. Protection against diabetes-induced nephropathy in growth hormone receptor/binding protein gene-disrupted mice. Endocrinology. 2000;141:163–168. doi: 10.1210/endo.141.1.7284. [DOI] [PubMed] [Google Scholar]
- 16.Rotwein P, Chia DJ. Gene regulation by growth hormone. Pediatr. Nephrol. 2009 doi: 10.1007/s00467-009-1258-y. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Chen WY, Wight DC, Chen NY, Coleman TA, Wagner TE, Kopchick JJ. Mutations in the third alpha-helix of bovine growth hormone dramatically affect its intracellular distribution in vitro and growth enhancement in transgenic mice. J. Biol. Chem. 1991;266:2252–2258. [PubMed] [Google Scholar]
- 18.Chen NY, Chen WY, Striker LJ, Striker GE, Kopchick JJ. Co-expression of bovine growth hormone (GH) and human GH antagonist genes in transgenic mice. Endocrinology. 1997;138:851–854. doi: 10.1210/endo.138.2.5036. [DOI] [PubMed] [Google Scholar]
- 19.Machado MO, Hirata RD, Sellitti DF, et al. Growth hormone promotes glomerular lipid accumulation in bGH mice. Kidney Int. 2005;68:2019–2028. doi: 10.1111/j.1523-1755.2005.00656.x. [DOI] [PubMed] [Google Scholar]
- 20.Berryman DE, List EO, Coschigano KT, Behar K, Kim JK, Kopchick JJ. Comparing adiposity profiles in three mouse models with altered GH signaling. Growth Horm. IGF Res. 2004;14:309–318. doi: 10.1016/j.ghir.2004.02.005. [DOI] [PubMed] [Google Scholar]
- 21.Coschigano KT, Holland AN, Riders ME, List EO, Flyvbjerg A, Kopchick JJ. Deletion, but not antagonism, of the mouse growth hormone receptor results in severely decreased body weights, insulin, and insulin-like growth factor I levels and increased life span. Endocrinology. 2003;144:3799–3810. doi: 10.1210/en.2003-0374. [DOI] [PubMed] [Google Scholar]
- 22.Flyvbjerg A, Dagnaes-Hansen F, De Vriese AS, Schrijvers BF, Tilton RG, Rasch R. Amelioration of long-term renal changes in obese type 2 diabetic mice by a neutralizing vascular endothelial growth factor antibody. Diabetes. 2002;51:3090–3094. doi: 10.2337/diabetes.51.10.3090. [DOI] [PubMed] [Google Scholar]
- 23.Hossenlopp P, Seurin D, Segovia-Quinson B, Hardouin S, Binoux M. Analysis serum insulin-like growth factor binding proteins using western blotting: Use of the method for titration of the binding proteins and competitive binding studies. Anal. Biochem. 1986;154:138–143. doi: 10.1016/0003-2697(86)90507-5. [DOI] [PubMed] [Google Scholar]
- 24.Altschul SF, Madden TL, Schaffer AA, et al. Gapped BLAST and PSI-BLAST: A new generation of protein database search programs. Nucleic Acids Res. 1997;25:3389–3402. doi: 10.1093/nar/25.17.3389. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Thomas RP, Guigneaux M, Wood T, Evers BM. Age-associated changes in gene expression patterns in the liver. J. Gastrointest. Surg. 2002;6:445–453. doi: 10.1016/s1091-255x(01)00010-5. discussion 454. [DOI] [PubMed] [Google Scholar]
- 26.Lefever S, Vandesompele J, Speleman F, Pattyn F. RTPrimerDB: The portal for real-time PCR primers and probes. Nucleic Acids Res. 2009;37:D942–D945. doi: 10.1093/nar/gkn777. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Rozen S, Skaletsky HJ. Primer3 on the WWW for general users and for biologist programmers. In: Krawetz S, Misener S, editors. Bioinformatics methods and protocols: Methods in molecular biology. Totowa, NJ: Humana Press; 2000. pp. 365–386. [DOI] [PubMed] [Google Scholar]
- 28.Andersen CL, Jensen JL, Orntoft TF. Normalization of real-time quantitative reverse transcription-PCR data: A model-based variance estimation approach to identify genes suited for normalization, applied to bladder and colon cancer data sets. Cancer Res. 2004;64:5245–5250. doi: 10.1158/0008-5472.CAN-04-0496. [DOI] [PubMed] [Google Scholar]
- 29.Meseguer A, Catterall JF. Mouse kidney androgen-regulated protein messenger ribonucleic acid is expressed in the proximal convoluted tubules. Mol. Endocrinol. 1987;1:535–541. doi: 10.1210/mend-1-8-535. [DOI] [PubMed] [Google Scholar]
- 30.Wanke R, Wolf E, Hermanns W, Folger S, Buchmuller T, Brem G. The GH-transgenic mouse as an experimental model for growth research: Clinical and pathological studies. Horm. Res. 1992;37:74–87. doi: 10.1159/000182406. [DOI] [PubMed] [Google Scholar]
- 31.Wolf E, Wanke R. Growth hormone overproduction in transgenic mice: Phenotypic alterations and deduced animal models. In: Van Zutphen LFM, van der Meer M, editors. Welfare aspects of transgenic animals. Berlin: Springer-Verlag; 1997. pp. 26–47. [Google Scholar]
- 32.Doi T, Striker LJ, Gibson CC, Agodoa LY, Brinster RL, Striker GE. Glomerular lesions in mice transgenic for growth hormone and insulinlike growth factor-I. I. relationship between increased glomerular size and mesangial sclerosis. Am. J. Pathol. 1990;137:541–552. [PMC free article] [PubMed] [Google Scholar]
- 33.Chen WY, Wight DC, Wagner TE, Kopchick JJ. Expression of a mutated bovine growth hormone gene suppresses growth of transgenic mice. Proc. Natl. Acad. Sci. U. S. A. 1990;87:5061–5065. doi: 10.1073/pnas.87.13.5061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Flyvbjerg A, Khatir DS, Jensen LJ, Dagnaes-Hansen F, Gronbaek H, Rasch R. The involvement of growth hormone (GH), insulin-like growth factors (IGFs) and vascular endothelial growth factor (VEGF) in diabetic kidney disease. Curr. Pharm. Des. 2004;10:3385–3394. doi: 10.2174/1381612043383106. [DOI] [PubMed] [Google Scholar]
- 35.Ogueta S, Olazabal I, Santos I, Delgado-Baeza E, Garcia-Ruiz JP. Transgenic mice expressing bovine GH develop arthritic disorder and self-antibodies. J. Endocrinol. 2000;165:321–328. doi: 10.1677/joe.0.1650321. [DOI] [PubMed] [Google Scholar]
- 36.Walport MJ. Lupus, DNase and defective disposal of cellular debris. Nat. Genet. 2000;25:135–136. doi: 10.1038/75963. [DOI] [PubMed] [Google Scholar]
- 37.Viedt C, Orth SR. Monocyte chemoattractant protein-1 (MCP-1) in the kidney: Does it more than simply attract monocytes? Nephrol. Dial. Transplant. 2002;17:2043–2047. doi: 10.1093/ndt/17.12.2043. [DOI] [PubMed] [Google Scholar]
- 38.Frade JM, Mellado M, del Real G, Gutierrez-Ramos JC, Lind P, Martinez-A C. Characterization of the CCR2 chemokine receptor: Functional CCR2 receptor expression in B cells. J. Immunol. 1997;159:5576–5584. [PubMed] [Google Scholar]
- 39.Reiser J, Mundel P. Danger signaling by glomerular podocytes defines a novel function of inducible B7-1 in the pathogenesis of nephrotic syndrome. J. Am. Soc. Nephrol. 2004;15:2246–2248. doi: 10.1097/01.ASN.0000136312.46464.33. [DOI] [PubMed] [Google Scholar]
- 40.Uchio K, Sawada K, Manabe N. Expression of macrophage metalloelastase (MMP-12) in podocytes of hereditary nephrotic mice (ICGN strain) J. Vet. Med. Sci. 2009;71:305–312. doi: 10.1292/jvms.71.305. [DOI] [PubMed] [Google Scholar]
- 41.Maecker HT, Todd SC, Levy S. The tetraspanin superfamily: Molecular facilitators. FASEB J. 1997;11:428–442. [PubMed] [Google Scholar]
- 42.Yunta M, Rodriguez-Barbero A, Arevalo MA, Lopez-Novoa JM, Lazo PA. Induction of DNA synthesis by ligation of the CD53 tetraspanin antigen in primary cultures of mesangial cells. Kidney Int. 2003;63:534–542. doi: 10.1046/j.1523-1755.2003.00758.x. [DOI] [PubMed] [Google Scholar]
- 43.Gottfried E, Kunz-Schughart LA, Weber A, et al. Expression of CD68 in non-myeloid cell types. Scand. J. Immunol. 2008;67:453–463. doi: 10.1111/j.1365-3083.2008.02091.x. [DOI] [PubMed] [Google Scholar]
- 44.Keshav S, Chung P, Milon G, Gordon S. Lysozyme is an inducible marker of macrophage activation in murine tissues as demonstrated by in situ hybridization. J. Exp. Med. 1991;174:1049–1058. doi: 10.1084/jem.174.5.1049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Klockars M, Reitamo S. Tissue distribution of lysozyme in man. J. Histochem. Cytochem. 1975;23:932–940. doi: 10.1177/23.12.1104708. [DOI] [PubMed] [Google Scholar]
- 46.Urade Y, Ujihara M, Horiguchi Y, Ikai K, Hayaishi O. The major source of endogenous prostaglandin D2 production is likely antigen-presenting cells. localization of glutathione-requiring prostaglandin D synthetase in histiocytes, dendritic, and kupffer cells in various rat tissues. J. Immunol. 1989;143:2982–2989. [PubMed] [Google Scholar]
- 47.Rezende AA, Semino-Mora C, Hirata MH, et al. American Society of Nephrology Renal Week. San Diego, CA: 2006. Up-regulation of toll-like receptors precedes matrix expansion in a model of growth hormone-induced glomerulosclerosis. (Abstract), p. TH-PO470. [Google Scholar]
- 48.Anders HJ, Banas B, Schlondorff D. Signaling danger: Toll-like receptors and their potential roles in kidney disease. J. Am. Soc. Nephrol. 2004;15:854–867. doi: 10.1097/01.asn.0000121781.89599.16. [DOI] [PubMed] [Google Scholar]
- 49.Cebrian C, Areste C, Nicolas A, et al. Kidney androgen-regulated protein interacts with cyclophilin B and reduces cyclosporine A-mediated toxicity in proximal tubule cells. J. Biol. Chem. 2001;276:29410–2949. doi: 10.1074/jbc.M102916200. [DOI] [PubMed] [Google Scholar]
- 50.Tornavaca O, Pascual G, Barreiro ML, et al. Kidney androgen-regulated protein transgenic mice show hypertension and renal alterations mediated by oxidative stress. Circulation. 2009;119:1908–1917. doi: 10.1161/CIRCULATIONAHA.108.808543. [DOI] [PubMed] [Google Scholar]
- 51.Fujimoto M, Maezawa Y, Yokote K, et al. Mice lacking Smad3 are protected against streptozotocin-induced diabetic glomerulopathy, Biochem. Biophys. Res. Commun. 2003;305:1002–1007. doi: 10.1016/s0006-291x(03)00885-4. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.