Abstract
Background
Gastric distension has important implications for motility and satiety. The hypothesis of this study was that distension affects the amplitude and duration of gastric contraction and that these parameters are largely mediated by efferent vagus stimulation.
Methods
A novel isovolumic myograph was introduced to test these hypotheses. The isovolumic myograph isolates the stomach and records the pressure generated by the gastric contraction under isovolumic conditions. Accordingly, the phasic changes of gastric contractility can be documented. A group of 12 rats were used under in vivo conditions and isolated ex vivo conditions and with two different gastric restraints (small and large) to determine the effect of degree of restraint.
Results
The comparison of the in vivo and ex vivo contractility provided information on the efferent vagus mediation of gastric contraction, i.e., the in vivo amplitude and duration reached maximum of 12.6 ± 2.7 mmHg and 19.8 ± 5.6 s in contrast to maximum of 5.7 ± 0.9 mmHg and 7.3 ± 1.3 s in ex vivo amplitude and duration, respectively. The comparison of gastric restraint and control groups highlights the role of distension on in vivo gastric contractility. The limitation of gastric distension by restraint drastically reduced the maximal amplitude to below 2.9 ± 0.2 mmHg.
Conclusions
The results show that distension-induced gastric contractility is regulated by both central nervous system and local mechanisms with the former being more substantial. Furthermore, the gastric restraint significantly attenuates gastric contractility (decreased amplitude and shortened duration of contraction) which is mediated by the efferent vagus activation. These findings have important implications for gastric motility and physiology and may improve our understanding of satiety.
Keywords: Myograph, Motility, Gastric restraint, Contraction
Introduction
The stomach is largely dependent on extrinsic nervous inputs arising from the central nervous system. These inputs regulate the activation of gastric smooth muscles and coordinate the digestive function of the stomach by parasympathetic and sympathetic pathways [1, 2]. The excitatory neurotransmitters by efferent vagus fibers (mainly acetylcholine and tachykinins) cause rhythmic contractions of gastric smooth muscles [3–5]. The gastric smooth muscles exhibit the tone on which there is superimposition of rhythmic contractions driven by cycles of membrane depolarization and repolarization [4].
It has been known for nearly three decades that the gastric mechanoreceptors which respond to gastric muscular distension and contraction are implicated in postprandial satiety, in sensing the effectiveness of a contraction to expel contents, and in a variety of reflexes [6–8]. Electrophysiological studies in different species have shown that mechanosensitive afferent fibers located in the antrum muscle wall respond to changes in smooth muscle transmural and local tension with an increased firing rate [9–13]. Gastric distension is correlated with a firing of vagal mechanosensitive afferent fibers which play an important role in satiety [9, 11].
Bariatric surgery significantly reduces body weight in obese patients and animal models and is the most effective treatment of severe obesity [14–16]. The weight loss after bariatric surgery is attributed to the restriction of gastric capacity, marked reduction of hunger, malabsorption, or regulation of appetite and satiety [13, 17–20]. We recently developed a reverse gastric sleeve implant to mimic the benefits of vertical sleeve gastroplasty, and a significant weight loss and decrease in food intake were identified [21]. Although the benefits of restrictive procedures are acknowledged experimentally and clinically, the effect of the restriction of gastric capacity on gastric contractility has not been well studied.
In this study, we hypothesize that gastric restraint mediated by mechanotransduction can acutely attenuate gastric contractility. We introduce an isovolumic myograph to measure gastric motility in response to intraluminal distension. We studied the distension-induced gastric contractility in response to intragastric inflation pressure. We placed external restraint of two different sizes (small and large) on the gastric wall to prevent distension of different surface areas of the gastric wall to evaluate the effect of degree of restraint on gastric contractility. The comparison of contractility at in vivo with ex vivo conditions was to assess the role of efferent vagus activation.
Materials and Methods
The animal experiments were performed in accordance with the guidelines of the Institute of Laboratory Animal Research Guide, Public Health Service Policy, Animal Welfare Act and an approved IACUC protocol by Indiana University School of Medicine.
Animals
Twelve C571/B mice at 12 weeks of age were obtained from Jackson Laboratory. The animals were acclimated to the facility for approximately 1 week prior to the start of the study. The animals were housed at 22°C under a 12-h light and dark cycle and were given free access to tap water and standard rodent chow. The animals were anesthetized with xylazine (1 mg/kg, i.p.) and ketamine (9 mg/kg, i.p.) and maintained with xylazine (0.5 mg/kg) and ketamine (4.5 mg/kg) every half hour.
In Vivo Gastric Contractility
Under anesthesia, the abdominal skin and muscle layers of the animal were opened to expose the stomach. The stomach was moisturized with warm (37°C) N-2-hydroxyethylpiperazine-N-2-ethanesulfonic acid–physiological saline solution (HEPES-PSS in mmol/l: 119 NaCl, 4.7 KCl, 3 HEPES, 2.7 HEPES Na, 1.17 MgSO4, 1.6 CaCl, 5.5 dextrose). The stomach was canulated with a HEPES-PSS prefilled catheter (ID 1 mm, OD 2 mm) which was connected to the isovolumic system (see Fig. 1). A 2-mm incision was cut at fundus apex of the stomach through which the catheter (OD 2 mm) was inserted into the stomach lumen. The fundus adjacent to the incision was tied on the tube with 6-0 silk suture twice to ensure no leakage. A 2-ml HEPES-PSS was gently injected into the stomach through the tube to wash out the content. After careful dissection of visible vagal nervous fibers near the stomach, the lower esophageal sphincter and pyloric sphincter were ligated with 6-0 silk suture. The gastric mesentery was untouched to allow the stomach to work in a physiological enviroment maintaining normal circulation and vagal responses. The gastric contractility was compared before imposing the restraint and after its removal. The data were discarded if the comparison was not within 10%.
Fig. 1.
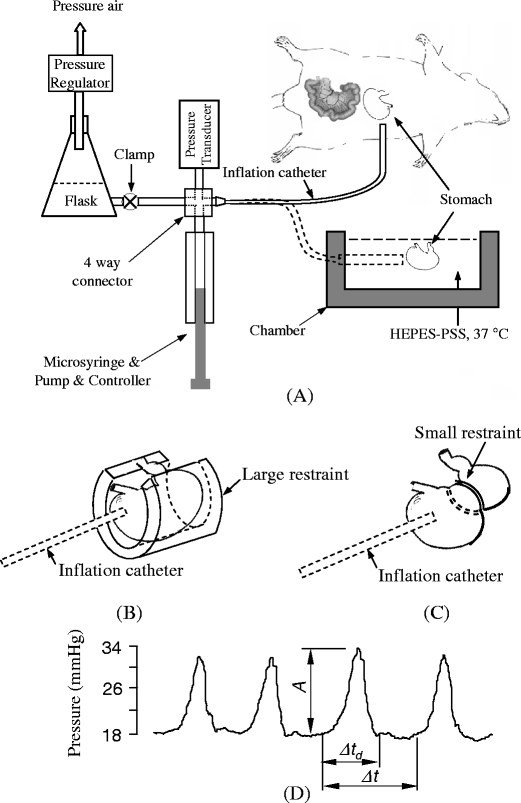
a A schematics of isovolumic myograph and in vivo and ex vivo experimental setup. The four-way connector merges the inflation flask, pressure transducer, microsyringe, and extended tubing. The chamber contains the HEPES-PSS at 37°C. The stomach and myograph are at isovolumic state when clamp is closed. b A large restraint limited gastric distension. c A small restraint applied on the stomach. d Typical pressure waves of gastric contraction. A is amplitude, Δt d is the duration of contraction, Δt is period
Ex Vivo Gastric Contractility
The animal was euthanized by overanesthesia. The stomach was excised quickly and placed in cold HEPES-PSS (4°C). The adjacent tissue was dissected with the aid of a stereomicroscope. The stomach was allowed to warm up to room temperature (22°C) slowly 10–15 min and transferred to a chamber with HEPES-PSS (22°C) of isovolumic myograph. A 2-mm incision was cut at the fundus apex of the stomach (fibrotic portion) and a catheter (ID 1 mm, OD 2 mm) was inserted into the stomach lumen through the incision. The fundus adjacent to the incision was tied on the catheter with 6-0 suture twice to avoid leakage. A 2-ml HEPES-PSS was gently injected into the stomach through the catheter to wash out the gastric content. Following drainage, the lower esophageal sphincter and pyloric sphinter were ligated with 6-0 silk suture. The stomach in the chamber was warmed to 37°C slowly (15–20 min) and equilibrated for 30 min at a basal intragastric pressure of about 2 mmHg before distension.
Isovolumic Myograph
An isovolumic myograph (Fig. 1a) consists of a chamber with a catheter on one side wall of the chamber which bridges the gastric lumen to inflation flask and pressure transducer [22]. A 50-ml inflation flask with PSS (connected to a pressure regulator) inflates the stomach to the desired pressure. The catheter, a solid state pressure transducer (SPR-524, Microtip catherter transducer, Millar Inc, TX, USA), a tube to the inflation flask, and a compensatory microsyringe were assembled in a four-way connector. A compensatory microsyringe (50 μl gastight microsyringe, UltraMicroPump III, and Micro 4TM microsyringe control, World Precision Instruments, USA) was used to stabilize the baseline of the pressure since water transport across the gastric wall reduces the intragastric pressure. The clamping of the tube between the inflation flask and the four-way connector achieves isovolumic conditions, i.e., intragastric volume is constant. As the stomach was inflated to a desired pressure (e.g., 5, 10 mmHg, etc.), the clamp was closed and the gastric contraction or relaxation was reflected by the variation of intragastric pressure recorded with a solid state pressure transducer. The isovolumic system records the periodic contractions of the stomach by the periodic variations of pressure.
Gastric Contractility in Response to Mechanical Stimulation
The mechanical stimulation of the stomach in this study was induced by an intragastric inflation pressure. The gastric contraction was quantified by the intragastric presssure under isovolumic condition, and the contractility was characterized with the amplitude and period of the pressure waveforms. The stomach was inflated to a desired pressure by a pressure regulator connected to a flask. The clamping of the tube between the inflation flask and the four-way connector maintained a constant volume of solution in the gastric lumen (isovolumic condition). The compensatory microsyringe maintains isovolumic conditions at an infusion rate of 1–3 μl/min. The data were discarded if the rate was larger than 10 μl/min since this implied damage (leakage) of the gastric wall. At isovolumic conditions, the variations of intragastric pressure were recorded with a data acquisition system (Biopac, MP100, Houston, TX, USA). The amplitude, frequency, and contractile duration of pressure waveforms were analyzed to characterize the gastric contractility.
Gastric Restraint
There were two types of loosely fitting restraints used in this study. The small restraint was a sector made of stainless steel wire (ϕ0.71 mm) and able to be closed to a circle with an inner diameter of 5.8 mm. The wire cycle was placed between the pyloric antrum and gastric body (Fig. 1c). A large restraint was made of plastic tube to study the effect of covered area of restrtaint on the gastric contraction. A cap of the tube was removed (semi-cylinder) to allow gastric mesentery pass free (Fig. 1b). The dimensions of the tube were 12.5 mm in length, 6 mm in internal diameter, and 2 mm in wall thickness. The width of the semi-cylinder is about 1 mm. The semi-cylinder was longitudinally opened up to a sector with the aid of a forceps. The semi-cylinder was passed through the gastric body. The forceps was released to allow the semi-cylinder to fully wrap the stomach. The semi-cylinder was circumferentially tied with a 6-0 silk suture to restraint the stomach into the internal space of the semi-cylinder when the stomach was inflated (Fig. 1b). The large restraint covered the side area of the stomach and limited the stretch on the gastric side wall despite an increase in intragastric pressure. The non-covered gastric wall on the two ends was distended during the increase in intragastric pressure.
Protocol of Mechanical Stimulation
The intragastric pressure was increased stepwise to 2, 5, 10, 15, 20, 30, 40, and 50 mmHg at a rate of 0.06 ml/min, respectively. The gastric contraction at isovolumic condition was recorded as the variation of the pressure at each individual inflation pressure. This protocol was applied to both in vivo and ex vivo stomach with or without gastric restraint. In the experiment of ex vivo stomach, acetylcholine (10–6 mol/l) was used to elicit non-neuroactive contraction of gastric smooth muscle at intragastric pressure of 50 mmHg to evalute contractility of gastric smooth muscle.
Data Analysis and Statistics
Figure 1d illustrates the definition of the parameters used in the analysis. The gastric contractile amplitude (A) was indicated by the amplitude of the variation of pressure. The gastric contractile period (Δt) was defined as the interval from one pressure waveform to the next pressure waveform. The gastric contractile duration (Δt d) was defined as the interval from pressure rise to fall.
The data were presented as mean ± SD and significant differences between groups were determined by Student’s t test. Significant differences between the in vivo, ex vivo, and restraint groups were determined by the use of analysis of variance (ANOVA) between groups. A probability of P < 0.05 was considered indicative of a statistically significant difference.
Results
The intragastric pressure and gastric volume in both in vivo and ex vivo are shown in Fig. 2, which reflects the global distension (compliance) of the stomach. The in vivo gastric volume was significantly different from the ex vivo volume. The ex vivo gastric volume increased in low pressure range (5–20 mmHg) while in vivo gastric volume increased almost linearly. The small restraint approximately reduced half of the gastric volume at high pressure range. The large restraint limited the gastric volume at about 30% of maximum.
Fig. 2.
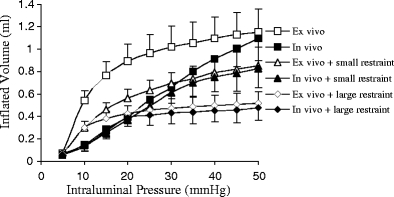
The relationship between gastric capacity (volume) and inflation pressure for various conditions. The statistical analysis (ANOVA): P < 0.05 between ex vivo and in vivo. P < 0.05 among in vivo, in vivo + small restraint, and in vivo + large restraint. P < 0.05 among ex vivo, ex vivo + small restraint, and ex vivo + large restraint. P > 0.05 between in vivo + small restraint and ex vivo + small restraint. P > 0.05 between in vivo + large restraint and ex vivo + large restraint
The intragastric pressure waveforms during gastric contraction are shown in Fig. 3. The variation of the pressure reflects the gastric contractility which is characterized by three parameters: contractile amplitude, duration, and period. The in vivo amplitude of gastric contraction increased from 1.6 to 12.5 mmHg when the inflation pressure changed from 2 to 30 mmHg (Fig. 3a), respectively. Clearly, both the small (Fig. 3b) and large (Fig. 3c) restraints significantly attenuated the amplitude of gastric pressure generation (contractility).
Fig. 3.
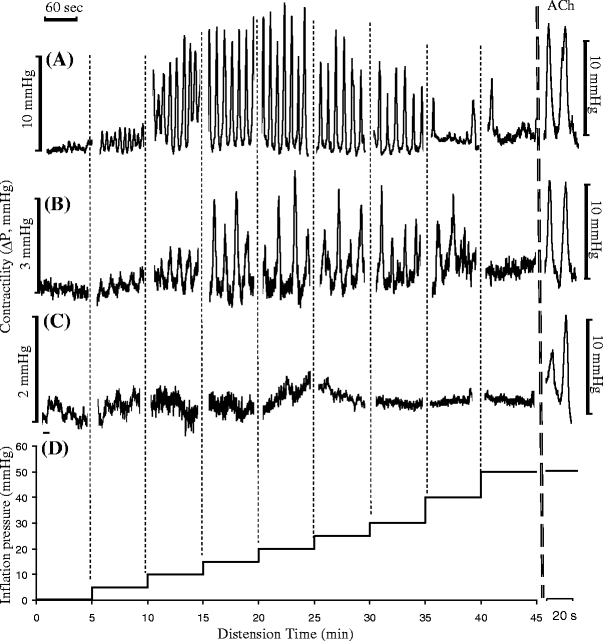
Typical in vivo contractile waves of gastric contractility at various inflation pressures. a No restraint; b when small restraint was applied, the contractile waves were significantly attenuated; c when large restraint was applied, the contractile waves were further attenuated. ACh acetylcholine administered at a concentration 10−6 mol/l. d The protocol of mechanical stimulation with stepwise inflation pressure
The contractility relation is shown in Fig. 4 both as a function of inflation pressure (Fig. 4a) and volume (Fig. 4b). The contractility increased with an increase in inflation pressure or volume and reached a maximum for the in vivo condition. The in vivo amplitude then decreased from 12.5 to 3.3 mmHg (Fig. 4a) when the inflation pressure was further increased from 30 to 50 mmHg. In ex vivo, the gastric contractile amplitude was significantly lower than that in in vivo at every inflation pressure. At 50 mmHg inflation pressure, the ex vivo contraction was completely abolished. We found that the contraction stimulated by the external administration of ACh (the right columns in Figs. 3 and 4), however, was still high. This suggests that the inhibition of gastric contraction under high distension is regulated by efferent nerves since gastric smooth muscle maintains contractility in response to ACh.
Fig. 4.
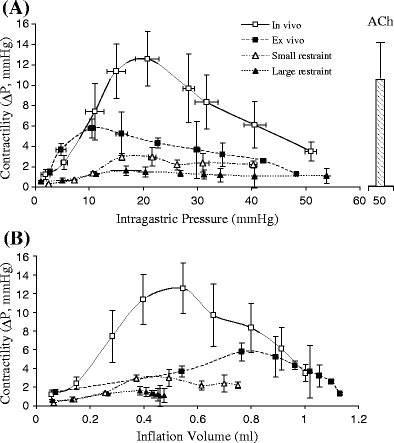
The amplitude of the contractile waves for various conditions. a The amplitude was represented as function of intragastric pressure. b The amplitude was represented as the function of inflation volume. The statistical analysis (ANOVA) is as follows: P < 0.05 between ex vivo and in vivo; P < 0.05 among in vivo, small restraint, and large restraint. The data for ex vivo small and large restraints were not shown since the amplitude of pressure wave was zero
The gastric restraint was applied externally to restrict gastric expansion during inflation. The large restraint covered over 80% of the external surface of the stomach and completely blocked the ex vivo gastric contraction and suppressed in vivo gastric contractions. To verify that the gastric nerves were not damaged during placement of large restraint, the contractility was measured again after removal of restraint. We found that the contractility remained at 95% to 100% of that before the application of the large restraint. The small restraint covered <10% of the external surface of the stomach. It is interesting that the inhibitory effects of the small restraint on gastric contraction were similar to those of the large restraint. In ex vivo experiments, the gastric contraction was completely inhibited by the large and small restraints (data not shown). But the ACh-induced contraction was similar to that of control (the right columns in Figs. 3, 4, and 5), which indicates that the agonist-induced contractility of gastric smooth muscle was not affected and the gastric restraint may block efferent activation of gastric response.
Fig. 5.
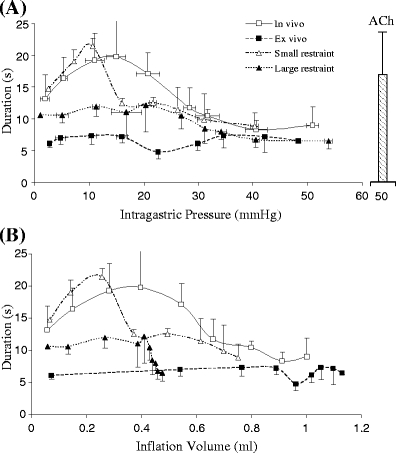
The duration of gastric contraction. a The duration was represented as function of intragastric pressure. b The duration was represented as the function of inflation volume. The legends were defined the same as Fig. 4. The statistical analysis (ANOVA) is as follows: P < 0.05 between ex vivo and in vivo; P < 0.05 among in vivo, small restraint, and large restraint. The data for ex vivo small and large restraints were not shown since the contraction was inhibited
The durations of the gastric contractility in vivo and ex vivo and under two gastric restraint conditions are presented in Fig. 5 as a function of pressure (a) and volume (b). The duration indicates the sustained interval of a single contraction wave. The in vivo duration was significantly larger than the ex vivo duration when the inflation pressure was below 35 mmHg or inflation volume was below 0.7 ml. The in vivo duration reached a maximum when inflation pressure increased from 10 to 20 mmHg (inflation volume from 0.3 to 0.5 ml). The ex vivo duration did not significantly change with the inflation. The large restraint blocked the change of in vivo duration with the inflation pressure or volume. The small restraint did not significantly affect the in vivo duration. The maximum duration with small restraint occurred at 10 mmHg of inflation pressure or 0.2 ml of inflation volume, which was lower than that without gastric restraint (~15 mmHg).
The period of the gastric contractility in vivo and ex vivo is shown in Fig. 6 as a function of pressure (a) and volume (b). The results showed that the period was similar in both in vivo and ex vivo preparation and increased with inflation. The gastric restraint eliminated the increase of period following inflation.
Fig. 6.
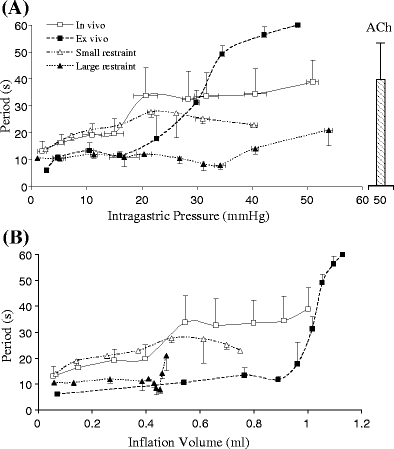
The period of gastric contractility. a The period was represented as a function of intragastric pressure. b The period was represented as the function of inflation volume. The legends were defined the same as Fig. 4. The statistical analysis (ANOVA) is as follows: P < 0.05 between ex vivo and in vivo; P < 0.05 between in vivo and large restraint; P > 0.05 between in vivo and small restraint. The data for ex vivo small and large restraints were not shown since the contraction was inhibited
Discussion
An isovolumic myograph was used to assess gastric contractility in terms of amplitude, duration, and period. The gastric restraint was shown to attenuate the mechanical stimulation-induced gastric contractility. The in vivo preparation was designed to detect the efferent neurogenic contraction and ex vivo preparation was designed to measure the efferent-independent contraction. The in vivo contractile amplitude and duration were significantly larger than those in ex vivo, indicating that contractile amplitude and duration may be efferent neurogenic. The similar period in in vivo and ex vivo preparation indicates that contractile period may be efferent-independent. The gastric restraint drastically attenuates the in vivo amplitude and duration of efferent neurogenic contraction, whereas the gastric restraint did not alter the period of the contraction.
Gastric contractility is closely coupled to the mechanosensitivity located in the gastric wall [12, 23, 24]. The myogenic response of the gastric smooth muscle and efferent neurogenic contraction are regulated by mechanoreceptors: afferent and efferent vagus nerves [1, 6, 9, 11, 12, 23, 24]. The relation between afferent vagus signals and gastric distension was identified decades ago [6, 9, 11, 12]. The mechanoreceptors in the gastric wall are primary sensors of mechanical stimulation [9, 23]. The efferent (motor) vagus signals are responses of the central nervous system to the afferent (sensory) vagus stimulation [2]. One of the physiological functions of efferent vagus signals is to regulate gastric contractility. Hence, the gastric contractility reflects the activation of the efferent vagus nerve.
In the present study, we evaluated gastric contractility in ex vivo preparation which excludes efferent vagus regulation. In the ex vivo stomach, the nerve fibers are excised and damaged, and hence, there is a loss of efferent vagus signals which appears to be significant for mechanical distension-induced contractility. The ex vivo contractility of the gastric smooth muscle was significantly attenuated due to the absence of efferent vagus signals. The strong in vivo contractility reflects the efferent vagus activation in response to mechanical stimulation sensed by afferent vagus nerves. The role of duration is interesting since it may reflect the efferent vagus activation. The duration of in vivo contraction varies with mechanical stimulation, whereas the duration of ex vivo contraction is largely unchanged in response to mechanical stimulation. The latter implies that the duration is regulated by the central nervous system. In contrast, the period seems to be independent of efferent vagus activation since both in vivo and ex vivo periods increase during the increase in distension. It appears that the contractility (Fig. 4) and duration (Fig. 5) which are dependent on efferent vagus activation are affected by gastric restraint, whereas the period (Fig. 6) is independent of efferent vagus activation.
Bariatric surgery, by restriction of gastric capacity or malabsorption, has been used to limit the amount of food intake in obesity [8, 25–28]. Weight loss and subsequent ameliorations in cardiovascular system are the major benefits of bariatric surgery [29–32]. Effects on the gastrointestinal hormonal levels and neurologic reflex response have been proposed to contribute to the mechanism of weight loss after bariatric surgery [6, 8, 12, 17–20, 33–35]. The present study shows that gastric restraint significantly attenuates gastric contractility and the decrease in the contractility is due to the downregulation of efferent vagus activation (Figs. 3 and 4). Our present findings suggest that gastric restraint may not only reduce gastric capacity but also compromises the gastric motility and delays gastric emptying. Since the peak of duration of the small restraint shifts to smaller inflation volume, this implies that efferent vagus inactivation is engaged at smaller filling volumes.
Some limitations of the present model deserve mention. Although the isovolumic myograph was used to evaluate the gastric global contractility, the regional contraction was not measured. The gastric tone (basal pressure) in the fundus and upper body was not characterized in the present study. Furthermore, the gastric contractile wave is generated in the lower stomach. Finally, the ligation on esophageal and pyloric sphincters may reduce the gastric contractility because of tiny nerve fibers in the sphincters. The variations of the contractility from animal to animal may be attributed to of the injury of nerves from ligation. Although the efferent vagus activation was evaluated mechanically, the nerve activation should be recorded electrically in future studies to further validate the present observation.
In conclusion, we introduced a novel isovolumic myograph to understand the contractility of the stomach. The in vivo and ex vivo gastric contractility in response to distension (inflation) provides evidence that gastric motility can be regulated by the central nervous system. The gastric restraint can attenuate gastric contractility (amplitude and duration of contraction), which is mediated by efferent vagus activation. The large restraint blocking gastric contractility more than the small restraint implies that the covered area of gastric restraint affects gastric motility. This study provides a better understanding of the role of gastric distension and its relation to the central nervous system.
Acknowledgment
This research was funded in part by GRest, Inc.
Conflict of Interest
The authors declare that they have no conflict of interest.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- 1.Bornstein JC, Costa M, Grider JR. Enteric motor and interneuronal circuits controlling motility. Neurogastroenterol Motil. 2004;16(Suppl 1):34–8. doi: 10.1111/j.1743-3150.2004.00472.x. [DOI] [PubMed] [Google Scholar]
- 2.Travagli RA, Hermann GE, Browning KN, et al. Brainstem circuits regulating gastric function. Annu Rev Physiol. 2006;68:279–305. doi: 10.1146/annurev.physiol.68.040504.094635. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Furness JB, Young HM, Pompolo S, et al. Plurichemical transmission and chemical coding of neurons in the digestive tract. Gastroenterology. 1995;108(2):554–63. doi: 10.1016/0016-5085(95)90086-1. [DOI] [PubMed] [Google Scholar]
- 4.Murthy KS. Signaling for contraction and relaxation in smooth muscle of the gut. Annu Rev Physiol. 2006;68:345–74. doi: 10.1146/annurev.physiol.68.040504.094707. [DOI] [PubMed] [Google Scholar]
- 5.Sarna SK. Tachykinins and in vivo gut motility. Dig Dis Sci. 1999;44(8 Suppl):114S–8. [PubMed] [Google Scholar]
- 6.Andrews PL, Grundy D, Scratcherd T. Reflex excitation of antral motility induced by gastric distension in the ferret. J Physiol. 1980;298:79–84. doi: 10.1113/jphysiol.1980.sp013068. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Leek BF. Abdominal and pelvic visceral receptors. Br Med Bull. 1977;33(2):163–8. doi: 10.1093/oxfordjournals.bmb.a071417. [DOI] [PubMed] [Google Scholar]
- 8.Woods SC. Gastrointestinal satiety signals I. An overview of gastrointestinal signals that influence food intake. Am J Physiol Gasterointest Liver Physiol. 2004;286(1):G7–13. doi: 10.1152/ajpgi.00448.2003. [DOI] [PubMed] [Google Scholar]
- 9.Andrews PL, Grundy D, Scratcherd T. Vagal afferent discharge from mechanoreceptors in different regions of the ferret stomach. J Physiol. 1980;298:513–24. doi: 10.1113/jphysiol.1980.sp013098. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Davis JD, Smith GP, Sayler JL. Reduction of intake in the rat due to gastric filling. Am J Physiol Regul Integr Comp Physiol. 1997;272:R1599–605. doi: 10.1152/ajpregu.1997.272.5.R1599. [DOI] [PubMed] [Google Scholar]
- 11.Peles S, Petersen J, Aviv R, et al. Enhancement of antral contractions and vagal afferent signaling with synchronized electrical stimulation. Am J Physiol Gastrointest Liver Physiol. 2003;285(3):G577–85. doi: 10.1152/ajpgi.00109.2003. [DOI] [PubMed] [Google Scholar]
- 12.Schwartz GJ, McHugh PR, Moran TH. Integration of vagal afferent responses to gastric loads and cholecystokinin in rats. Am J Physiol Regul Integr Comp Physiol. 1991;261:R64–9. doi: 10.1152/ajpregu.1991.261.1.R64. [DOI] [PubMed] [Google Scholar]
- 13.Taché Y, Mönnikes H, Bonaz B, et al. Role of CRF in stress-related alterations of gastric and colonic motor function. Ann NY Acad Sci. 1993;697:233–43. doi: 10.1111/j.1749-6632.1993.tb49936.x. [DOI] [PubMed] [Google Scholar]
- 14.Dixon JB, O'Brien PE. Changes in comorbidities and improvements in quality of life after LAP-BAND placement. Am J Surg. 2002;184(Suppl):S51–4. doi: 10.1016/S0002-9610(02)01181-9. [DOI] [PubMed] [Google Scholar]
- 15.Ponce J, Haynes B, Paynter S, et al. Effect of Lap-Band-induced weight loss on type II diabetes mellitus and hypertension. Obes Surg. 2004;14:1335–42. doi: 10.1381/0960892042583932. [DOI] [PubMed] [Google Scholar]
- 16.Inge TH, Krebs NF, Garcia VF, et al. Bariatric surgery for severely overweight adolescents: concerns and recommendations. Pediatrics. 2004;114:217–23. doi: 10.1542/peds.114.1.217. [DOI] [PubMed] [Google Scholar]
- 17.Cummings DE, Weigle DS, Frayo RS, et al. Plasma ghrelin levels after diet-induced weight loss or gastric bypass surgery. N Engl J Med. 2002;346:1623–30. doi: 10.1056/NEJMoa012908. [DOI] [PubMed] [Google Scholar]
- 18.Langer FB, Reza Hoda MA, Bohdjalian A, et al. Sleeve gastrectomy and gastric banding: effects on plasma ghrelin levels. Obes Surg. 2005;15:1024–9. doi: 10.1381/0960892054621125. [DOI] [PubMed] [Google Scholar]
- 19.Schindler K, Prager G, Ballaban T, et al. Impact of laparoscopic adjustable gastric banding on plasma ghrelin, eating behaviour and body weight. Eur J Clin Investig. 2004;34:549–54. doi: 10.1111/j.1365-2362.2004.01382.x. [DOI] [PubMed] [Google Scholar]
- 20.Korner J, Inabnet W, Conwell IM, et al. Differential effects of gastric bypass and banding on circulating gut hormone and leptin levels. Obesity (Silver Spring) 2006;14:1553–61. doi: 10.1038/oby.2006.179. [DOI] [PubMed] [Google Scholar]
- 21.Guo X, Mattar S, Morales C, et al. Novel gastric sleeve magnetic implant: safety and efficacy in rats. Surg Obes Relat Dis. 2009;5(6):684–91. doi: 10.1016/j.soard.2009.07.005. [DOI] [PubMed] [Google Scholar]
- 22.Lu X, Kassab GS. Vasoactivity of blood vessels using a novel isovolumic myograph. Ann Biomed Eng. 2007;35(3):356–66. doi: 10.1007/s10439-006-9243-0. [DOI] [PubMed] [Google Scholar]
- 23.Phillips RJ, Powley TL. Tension and stretch receptors in gastrointestinal smooth muscle: re-evaluating vagal mechanoreceptor electrophysiology. Brain Res Brain Res Rev. 2000;34(1–2):1–26. doi: 10.1016/S0165-0173(00)00036-9. [DOI] [PubMed] [Google Scholar]
- 24.Zhu H, Chen JD. Gastric distension alters frequency and regularity but not amplitude of the gastric slow wave. Neurogastroenterol Motil. 2004;16(6):745–52. doi: 10.1111/j.1365-2982.2004.00571.x. [DOI] [PubMed] [Google Scholar]
- 25.Chevallier JM, Zinzindohoué F, Elian N, et al. Adjustable gastric banding in a public university hospital: prospective analysis of 400 patients. Obes Surg. 2002;12(1):93–9. doi: 10.1381/096089202321144658. [DOI] [PubMed] [Google Scholar]
- 26.DeMaria EJ. Laparoscopic adjustable silicone gastric banding. Surg Clin North Am. 2001;81(5):1129–44. doi: 10.1016/S0039-6109(05)70188-2. [DOI] [PubMed] [Google Scholar]
- 27.Kanno H, Kiyama T, Fujita I, et al. Rat gastric banding model for bariatric surgery. J Nippon Med Sch. 2008;75(4):202–6. doi: 10.1272/jnms.75.202. [DOI] [PubMed] [Google Scholar]
- 28.Wolf AM, Kortner B, Kuhlmann HW. Results of bariatric surgery. Int J Obes Relat Metab Disord. 2001;25(Suppl 1):S113–4. doi: 10.1038/sj.ijo.0801713. [DOI] [PubMed] [Google Scholar]
- 29.DePaula AL, Macedo AL, Rassi N, et al. Laparoscopic treatment of type 2 diabetes mellitus for patients with a body mass index less than 35. Surg Endosc. 2008;22(3):706–16. doi: 10.1007/s00464-007-9472-9. [DOI] [PubMed] [Google Scholar]
- 30.DePaula AL, Macedo AL, Mota BR, et al. Laparoscopic ileal interposition associated to a diverted sleeve gastrectomy is an effective operation for the treatment of type 2 diabetes mellitus patients with BMI 21-29. Surg Endosc. 2009;23(6):1313–20. doi: 10.1007/s00464-008-0156-x. [DOI] [PubMed] [Google Scholar]
- 31.Fielding GA, Rhodes M, Nathanson LK. Laparoscopic gastric banding for morbid obesity. Surgical outcome in 335 cases. Surg Endosc. 1999;13(6):550–4. doi: 10.1007/s004649901038. [DOI] [PubMed] [Google Scholar]
- 32.Keating CL, Dixon JB, Moodie ML, et al. Cost-efficacy of surgically induced weight loss for the management of type 2 diabetes: a randomized controlled trial. Diab Care. 2009;32(4):580–4. doi: 10.2337/dc08-1748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Ariyasu H, Takaya K, Tagami T, et al. Stomach is a major source of circulating ghrelin, and feeding state determines plasma ghrelin-like immunoreactivity levels in humans. J Clin Endocrinol Metab. 2001;86:4753–8. doi: 10.1210/jc.86.10.4753. [DOI] [PubMed] [Google Scholar]
- 34.Wang Y, Liu J. Plasma ghrelin modulation in gastric band operation and sleeve gastrectomy. Obes Surg. 2009;19:357–62. doi: 10.1007/s11695-008-9688-3. [DOI] [PubMed] [Google Scholar]
- 35.Herron DM. The surgical management of severe obesity. Mt Sinai J Med. 2004;71(1):63–71. [PubMed] [Google Scholar]


