Abstract
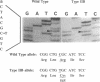
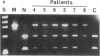
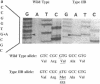
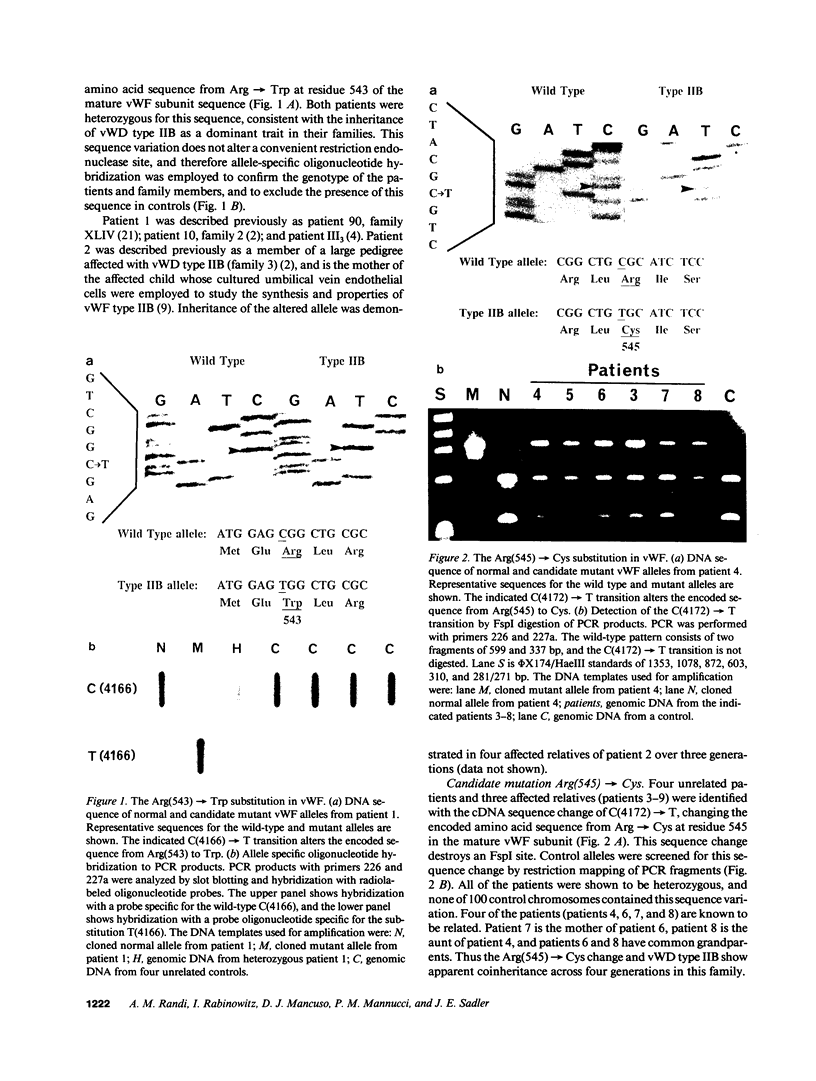
Many variants of von Willebrand disease (vWD) with qualitatively abnormal von Willebrand factor (vWF) are recognized. In vWD type IIB, the abnormal protein displays enhanced affinity for a platelet vWF receptor, the glycoprotein Ib-IX complex. 14 patients from 7 unrelated families with vWD type IIB were studied to determine the molecular basis for this phenotype. Specific oligonucleotide primers were used to amplify portions of vWF exon 28 encoding a domain that interacts with the platelet glycoprotein Ib-IX complex. Candidate missense mutations were identified for all 14 patients by DNA sequencing, allele specific oligonucleotide hybridization, and restriction endonuclease digestion. These sequence changes occur in an 11 amino acid segment within a single disulfide loop bounded by Cys(509) and Cys(695). All of these sequence changes are C----T transitions within CG dinucleotides. Six patients from two unrelated families were heterozygous for the encoded sequence Arg(543)----Trp. Seven patients from four unrelated families were heterozygous for the encoded sequence Arg(545)----Cys; this sequence change appears to have occurred independently three times, once as a new spontaneous mutation. One patient with apparently sporadic vWD type IIB was heterozygous for the encoded sequence Val(553)----Met, and this appears to be a new mutation. None of these sequence changes was found in 100 normal alleles. These findings suggest that vWD type IIB may be caused by relatively few distinct mutations, that these mutations may cluster within a specific region of one disulfide loop in vWF domain A1, and that this region can modulate the affinity of vWF for the platelet glycoprotein Ib-IX complex.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Andrews R. K., Gorman J. J., Booth W. J., Corino G. L., Castaldi P. A., Berndt M. C. Cross-linking of a monomeric 39/34-kDa dispase fragment of von Willebrand factor (Leu-480/Val-481-Gly-718) to the N-terminal region of the alpha-chain of membrane glycoprotein Ib on intact platelets with bis(sulfosuccinimidyl) suberate. Biochemistry. 1989 Oct 17;28(21):8326–8336. doi: 10.1021/bi00447a010. [DOI] [PubMed] [Google Scholar]
- Bird A. P. DNA methylation and the frequency of CpG in animal DNA. Nucleic Acids Res. 1980 Apr 11;8(7):1499–1504. doi: 10.1093/nar/8.7.1499. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Coulondre C., Miller J. H., Farabaugh P. J., Gilbert W. Molecular basis of base substitution hotspots in Escherichia coli. Nature. 1978 Aug 24;274(5673):775–780. doi: 10.1038/274775a0. [DOI] [PubMed] [Google Scholar]
- Dayhoff M. O., Barker W. C., Hunt L. T. Establishing homologies in protein sequences. Methods Enzymol. 1983;91:524–545. doi: 10.1016/s0076-6879(83)91049-2. [DOI] [PubMed] [Google Scholar]
- De Marco L., Girolami A., Zimmerman T. S., Ruggeri Z. M. Interaction of purified type IIB von Willebrand factor with the platelet membrane glycoprotein Ib induces fibrinogen binding to the glycoprotein IIb/IIIa complex and initiates aggregation. Proc Natl Acad Sci U S A. 1985 Nov;82(21):7424–7428. doi: 10.1073/pnas.82.21.7424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Donnér M., Holmberg L., Nilsson I. M. Type IIB von Willebrand's disease with probable autosomal recessive inheritance and presenting as thrombocytopenia in infancy. Br J Haematol. 1987 Jul;66(3):349–354. doi: 10.1111/j.1365-2141.1987.tb06922.x. [DOI] [PubMed] [Google Scholar]
- Federici A. B., Mannucci P. M., Bader R., Lombardi R., Lattuada A. Heterogeneity in type IIB von Willebrand disease: two unrelated cases with no family history and mild abnormalities of ristocetin-induced interaction between von Willebrand factor and platelets. Am J Hematol. 1986 Dec;23(4):381–390. doi: 10.1002/ajh.2830230410. [DOI] [PubMed] [Google Scholar]
- Fujimura Y., Titani K., Holland L. Z., Russell S. R., Roberts J. R., Elder J. H., Ruggeri Z. M., Zimmerman T. S. von Willebrand factor. A reduced and alkylated 52/48-kDa fragment beginning at amino acid residue 449 contains the domain interacting with platelet glycoprotein Ib. J Biol Chem. 1986 Jan 5;261(1):381–385. [PubMed] [Google Scholar]
- Giannelli F., Green P. M., High K. A., Lozier J. N., Lillicrap D. P., Ludwig M., Olek K., Reitsma P. H., Goossens M., Yoshioka A. Haemophilia B: database of point mutations and short additions and deletions. Nucleic Acids Res. 1990 Jul 25;18(14):4053–4059. doi: 10.1093/nar/18.14.4053. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Giles A. R., Hoogendoorn H., Benford K. Type IIB von Willebrand's disease presenting as thrombocytopenia during pregnancy. Br J Haematol. 1987 Nov;67(3):349–353. doi: 10.1111/j.1365-2141.1987.tb02356.x. [DOI] [PubMed] [Google Scholar]
- Ginsburg D., Konkle B. A., Gill J. C., Montgomery R. R., Bockenstedt P. L., Johnson T. A., Yang A. Y. Molecular basis of human von Willebrand disease: analysis of platelet von Willebrand factor mRNA. Proc Natl Acad Sci U S A. 1989 May;86(10):3723–3727. doi: 10.1073/pnas.86.10.3723. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grunebaum L., Cazenave J. P., Camerino G., Kloepfer C., Mandel J. L., Tolstoshev P., Jaye M., De la Salle H., Lecocq J. P. Carrier detection of Hemophilia B by using a restriction site polymorphism associated with the coagulation Factor IX gene. J Clin Invest. 1984 May;73(5):1491–1495. doi: 10.1172/JCI111354. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmberg L., Nilsson I. M., Borge L., Gunnarsson M., Sjörin E. Platelet aggregation induced by 1-desamino-8-D-arginine vasopressin (DDAVP) in Type IIB von Willebrand's disease. N Engl J Med. 1983 Oct 6;309(14):816–821. doi: 10.1056/NEJM198310063091402. [DOI] [PubMed] [Google Scholar]
- Hultin M. B., Sussman I. I. Postoperative thrombocytopenia in type IIB von Willebrand disease. Am J Hematol. 1990 Jan;33(1):64–68. doi: 10.1002/ajh.2830330113. [DOI] [PubMed] [Google Scholar]
- Mancuso D. J., Tuley E. A., Westfield L. A., Lester-Mancuso T. L., Le Beau M. M., Sorace J. M., Sadler J. E. Human von Willebrand factor gene and pseudogene: structural analysis and differentiation by polymerase chain reaction. Biochemistry. 1991 Jan 8;30(1):253–269. doi: 10.1021/bi00215a036. [DOI] [PubMed] [Google Scholar]
- Mancuso D. J., Tuley E. A., Westfield L. A., Worrall N. K., Shelton-Inloes B. B., Sorace J. M., Alevy Y. G., Sadler J. E. Structure of the gene for human von Willebrand factor. J Biol Chem. 1989 Nov 25;264(33):19514–19527. [PubMed] [Google Scholar]
- Marti T., Rösselet S. J., Titani K., Walsh K. A. Identification of disulfide-bridged substructures within human von Willebrand factor. Biochemistry. 1987 Dec 15;26(25):8099–8109. doi: 10.1021/bi00399a013. [DOI] [PubMed] [Google Scholar]
- Miller J. L., Castella A. Platelet-type von Willebrand's disease: characterization of a new bleeding disorder. Blood. 1982 Sep;60(3):790–794. [PubMed] [Google Scholar]
- Mohri H., Fujimura Y., Shima M., Yoshioka A., Houghten R. A., Ruggeri Z. M., Zimmerman T. S. Structure of the von Willebrand factor domain interacting with glycoprotein Ib. J Biol Chem. 1988 Dec 5;263(34):17901–17904. [PubMed] [Google Scholar]
- Mohri H., Yoshioka A., Zimmerman T. S., Ruggeri Z. M. Isolation of the von Willebrand factor domain interacting with platelet glycoprotein Ib, heparin, and collagen and characterization of its three distinct functional sites. J Biol Chem. 1989 Oct 15;264(29):17361–17367. [PubMed] [Google Scholar]
- Rick M. E., Williams S. B., Sacher R. A., McKeown L. P. Thrombocytopenia associated with pregnancy in a patient with type IIB von Willebrand's disease. Blood. 1987 Mar;69(3):786–789. [PubMed] [Google Scholar]
- Ruggeri Z. M., Lombardi R., Gatti L., Bader R., Valsecchi C., Zimmerman T. S. Type IIB von Willebrand's disease: differential clearance of endogenous versus transfused large multimer von willebrand factor. Blood. 1982 Dec;60(6):1453–1456. [PubMed] [Google Scholar]
- Ruggeri Z. M., Mannucci P. M., Lombardi R., Federici A. B., Zimmerman T. S. Multimeric composition of factor VIII/von Willebrand factor following administration of DDAVP: implications for pathophysiology and therapy of von Willebrand's disease subtypes. Blood. 1982 Jun;59(6):1272–1278. [PubMed] [Google Scholar]
- Ruggeri Z. M., Pareti F. I., Mannucci P. M., Ciavarella N., Zimmerman T. S. Heightened interaction between platelets and factor VIII/von Willebrand factor in a new subtype of von Willebrand's disease. N Engl J Med. 1980 May 8;302(19):1047–1051. doi: 10.1056/NEJM198005083021902. [DOI] [PubMed] [Google Scholar]
- Ruggeri Z. M., Zimmerman T. S. Variant von Willebrand's disease: characterization of two subtypes by analysis of multimeric composition of factor VIII/von Willebrand factor in plasma and platelets. J Clin Invest. 1980 Jun;65(6):1318–1325. doi: 10.1172/JCI109795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saba H. I., Fujimura Y., Saba S. R., Bruce A. C., Morelli G. A., Ruggeri Z. M., Zimmerman T. S. Spontaneous platelet aggregation in type IIB Tampa von Willebrand disease is inhibited by the 52/48-kDa fragment of normal von Willebrand factor, which contains the GPIb binding domain. Am J Hematol. 1989 Mar;30(3):150–153. doi: 10.1002/ajh.2830300306. [DOI] [PubMed] [Google Scholar]
- Sadler J. E., Shelton-Inloes B. B., Sorace J. M., Harlan J. M., Titani K., Davie E. W. Cloning and characterization of two cDNAs coding for human von Willebrand factor. Proc Natl Acad Sci U S A. 1985 Oct;82(19):6394–6398. doi: 10.1073/pnas.82.19.6394. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saiki R. K., Gelfand D. H., Stoffel S., Scharf S. J., Higuchi R., Horn G. T., Mullis K. B., Erlich H. A. Primer-directed enzymatic amplification of DNA with a thermostable DNA polymerase. Science. 1988 Jan 29;239(4839):487–491. doi: 10.1126/science.2448875. [DOI] [PubMed] [Google Scholar]
- Shelton-Inloes B. B., Chehab F. F., Mannucci P. M., Federici A. B., Sadler J. E. Gene deletions correlate with the development of alloantibodies in von Willebrand disease. J Clin Invest. 1987 May;79(5):1459–1465. doi: 10.1172/JCI112974. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Weiss H. J., Meyer D., Rabinowitz R., Pietu G., Girma J. P., Vicic W. J., Rogers J. Pseudo-von Willebrand's disease. An intrinsic platelet defect with aggregation by unmodified human factor VIII/von Willebrand factor and enhanced adsorption of its high-molecular-weight multimers. N Engl J Med. 1982 Feb 11;306(6):326–333. doi: 10.1056/NEJM198202113060603. [DOI] [PubMed] [Google Scholar]
- Wood W. I., Gitschier J., Lasky L. A., Lawn R. M. Base composition-independent hybridization in tetramethylammonium chloride: a method for oligonucleotide screening of highly complex gene libraries. Proc Natl Acad Sci U S A. 1985 Mar;82(6):1585–1588. doi: 10.1073/pnas.82.6.1585. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssoufian H., Kazazian H. H., Jr, Phillips D. G., Aronis S., Tsiftis G., Brown V. A., Antonarakis S. E. Recurrent mutations in haemophilia A give evidence for CpG mutation hotspots. 1986 Nov 27-Dec 3Nature. 324(6095):380–382. doi: 10.1038/324380a0. [DOI] [PubMed] [Google Scholar]
- de Groot P. G., Federici A. B., de Boer H. C., d'Alessio P., Mannucci P. M., Sixma J. J. von Willebrand factor synthesized by endothelial cells from a patient with type IIB von Willebrand disease supports platelet adhesion normally but has an increased affinity for platelets. Proc Natl Acad Sci U S A. 1989 May;86(10):3793–3797. doi: 10.1073/pnas.86.10.3793. [DOI] [PMC free article] [PubMed] [Google Scholar]