Abstract
The effects of abundant Mancozeb (Mn, Zn—bisdithiocarbamate) applications (2.5 kg ha−1week−1 for 10 years) on soil and surface-, subsurface- and groundwater pollution were monitored in a banana production region of tropical Mexico. In soils, severe manganese accumulation was observed, wheras the main metabolite ethylenethiourea was near the detection limit. Surface and subsurface water was highly polluted with ethylenethiourea, the main metabolite of Mancozeb (22.5 and 4.3 μg L−1, respectively), but not with manganese. In deep ground water, no ethylenethiourea was detected. The level of pollution in the region presents a worrisome risk for aquatic life and for human health.
Keywords: Fungicide, Soil and water pollution, Tropics
The frequent use of pesticides in tropical agro industrial regions carries the risk of accumulation of pesticides, metabolites and accompanying heavy metals in soil, water and organisms (WHO 2005). Leaching and aerial transport of pesticides applied to leaves increase the risk of dispersion of these products in the environment and affect ecosystems and human health (Mortensen et al. 1998, Konstantinou et al. 2006). Conventional production of bananas requires great quantities of agrochemicals, particularly fungicides, to control the disease Black Sigatoka. Spraying of bisdithiocarbamates to control Black Sigatoka takes place in nearly all conventional managed banana farms at high frequencies and doses. Bisdithiocarbamates such as Mancozeb have a medium life span in soil of 1–7 days, and a low toxicity (Hayes and Laws 1991). Its decomposition in the environment may take place by photolysis, hydrolysis or biological decomposition (Xu 2000a). Its main metabolite is ethylenethiourea (ETU) which is mobile in the environment because of its high solubility in water and weak adsorption in soil (Rajagopal et al. 1984, Xu 2000a). According to Shukla and Arora (2001) ETU has a high potential to cause cancer, and genetic defects in laboratory animals. The rapid decomposition of Mancozeb allows all its sub compounds, including manganese and zinc, to be released into the soil (Xu 2000a). Melgar et al. (2008) found severe accumulation of Mn in sediments of runnels, and of ETU in surface water in a tropical banana production region.
Laboratory studies suggest that pesticide degradation rates and pathways are comparable in tropical and temperate soils. However, field investigations of the fate of pesticides in tropical soils indicate that dissipation occurs (much) more rapidly under tropical conditions than under temperate conditions (Racke et al. 1997). The authors suggest that investigations on the fate and effects of pesticides in tropical soils, especially under tropical environmental conditions, should continue to be encouraged. The aim of this study was to measure the accumulation of ETU and the heavy metals Mn and Zn—from decomposing Mancozeb—in soil and water bodies in an agro industrial banana production region in tropical Mexico. The results of this study can serve as a reference for similar tropical conditions.
Materials and Methods
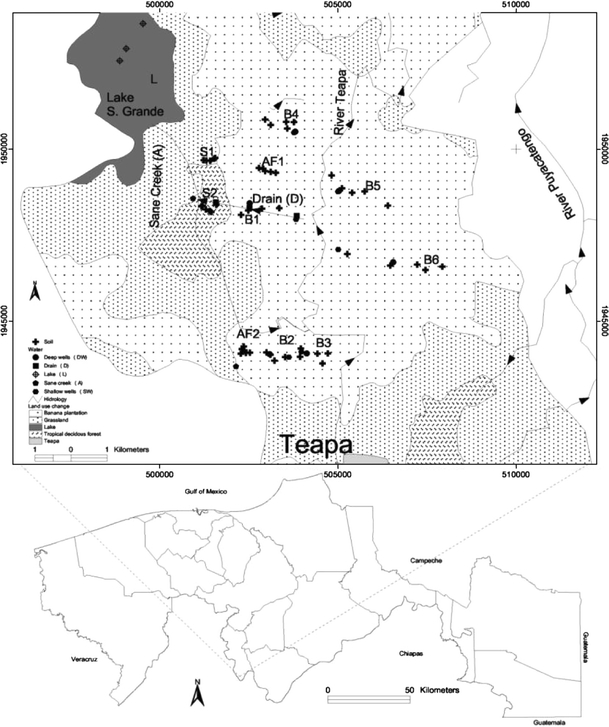
The study area is located on a plain in the state of Tabasco, SE Mexico (Fig. 1). The entire study area has a surface of 10453 ha. The climate in the region is humid warm (25.4°C mean temp., 3862 mm mean annual rainfall). There are three climatic seasons during the year: dry (April–May), tropical rains (June–October) and moderate rains (November–March). Soils are mainly Gleysols and Fluvisols. Agricultural activities are primarily banana production and agro forestry plantations (Spanish cedar and bananas). Since 1995, a weekly application of 2.5 kg ha−1 Mancozeb is sprayed over the banana plantations and agro forestry systems. The banana farms and agro forestry systems have an open ditch drainage system to decrease the frequency of yearly flooding. A system of secondary and tertiary drains lead to one main drain per farm (D). The main drains discharge into a creek (C) that ends in the Sitio Grande lake (L) (Fig. 1). Tropical riparian succession forests in the study area have no drainage system. Sub superficial water (SS) is located at a depth of 3–5 m and serves as potable water. The ground water table (GW) is 80 m deep and is separated from the subsurface water body through an impermeable clay layer.
Fig. 1.
Sane Creek (A) through Sane Creek (C)
Soil samples were taken from six banana sites (B1–B6), two agro forestry sites (AF1, AF2) and two succession forest sites (S1, S2) (Fig. 1). The soil samples from the two last sites served as references for natural Mn and Zn levels. Due to financial restrictions, analysis of ETU content was confined to banana soils. In May 2006, soil samples were taken from five randomly distributed points per site (1 ha) at two different depths (0–10 and 10–30 cm). Soil samples were collected at the end of the dry season, based on the assumption that maximum ETU content would be detected in the dry season as the result of reduced leaching.
Samples of surface and subsurface water were collected toward the end of the rainy season in February 2007 because collection in the dry season was impossible. We collected surface water samples from the principle drain (D) at site B1, from the creek (C) and from the lake (L), with three replicas per site (Fig. 1). Samples from surface water were taken at two depths (10 cm and 10–50 cm). Subsurface (SS) water samples were collected from five shallow wells (3–5 m) and ground water (GW) samples from five deep wells (80 m) randomly distributed in the watershed of the lake Sitio Grande (3 replicates per well).
Analysis of the soil samples included: pH (KCl), texture (Bouyocus), organic matter (OM) (Walkley and Black), apparent density (AD) and cation exchange capacity (CEC) using ammonium acetate extraction at pH 7 according to the Mexican Official Norms (SEMARNAT 2000). The content of Mntot and Zntot in soil was determined through digestion with HNO3/HClO4 and atomic absorption spectrophotometry. Plant available (avail) content of Mn and Zn was analyzed using DTPA extraction. Water pH was measured in the field. Total content of Mn and Zn in water was determined using atomic absorption spectrophotometer following the method established by Franson (1992).
For identification and quantification of ETU in soil and water the method proposed by Dubey et al. (1997) was followed, modified for soil and water samples. This covered the requisites of selectivity, sensibility and precision to insure the validity of the results. For the analysis of ETU in soil we used 10 g of soil, and for analysis in water 50 mL of water. After extraction with methanol (soil samples) followed a derivatisation with a liquid–liquid partition and purification in columns, and a derivation for the determining stage with a Varian CP-3800 gas chromatograph and an electron capture detector (ECD). We determined the detection limit (0.01 mg kg−1) by fortifying soil (0.124 mg kg−1) and water (0.00082 mg kg−1) samples with Ethylenethiourea and extracting it by the same technique. The limit of detection (LOD) was determined considering answers in the sample of three times the noise level (LOD = ng ETU causing an answer of three times the level of noise in the detector/10 mg equivalents of sample used in the determinative step). The limit of quantification (LOQ) (0.03 mg kg−1) was established as the ETU concentration able to produce an answer in the detector of 10% of deflection of the signal level (FSD) (LOQ = ng of ETU causing a signal of the detector of 10% FSD/10 mg equivalents of sample used in the determinative step). The recovery percentage for both matrix types was >90% using three replicates (Melgar Valdes et al. 2008).
We applied the Kolmogorov–Smirnov test to determine whether the data followed a normal distribution or not. For data with a normal distribution, analysis of variance (ANOVA) and the T3-Dunnett Post Hoc Test were applied for determination of significant differences between the sites. For data without a normal distribution, the Mann–Whitney U test was applied. The level of significance for both distributions was p < 0.05. We used the principal component analysis (PCA) to extract the principal components from soil data (pH, OM, clay content, CEC, total and available Mn and Zn, 8 variables, 50 sampling points).
Results and Discussion
Soil texture ranged from clayey silt to clayey loam. This texture is prone to low infiltration and high flow of surface water as well as preferential flow in cracks (Persson et al. 2000). The apparent density varied between 0.9 and 1.2 g cm−3, with the lowest values in the tropical forest S1 and in AF2 and the highest in the banana soil B6. The soils at B1, B3, B4, B5, B6, S1 and AF2 had a neutral pH, the soils at B2 and AF1 were strongly acidic and the soil at S2 acidic. The organic matter (OM) content in the banana and agro forestry soils was significantly lower than that in the tropical forest soils (S1 and S2) at both depths (Table 1a and b). The high content of organic matter in tropical forest soils is generated by the high abundance of leaf litter that falls from the trees and is incorporated into the system throughout the year (Geissen and Morales 2006).
Table 1.
a, b Physical–chemical soil properties of banana plantations (B1–B6), agro forestry systems (AF), succesion forest (S) at depths 0–10 cm and 10–30 cm
| Site | pH(KCl) | OM g 100 g−1 | CEC cmol kg−1 | AD (g cm−3) | Texture |
|---|---|---|---|---|---|
| (a) 0–10 cm | |||||
| S1 | 6.8c ± 0.2 | 6.1d ± 1.7 | 47.2c ± 3.76 | 0.9a ± 0.02 | Clayey silt |
| S2 | 5.5b ± 0.1 | 4.9c ± 0.3 | 48.4c ± 1.31 | 1.0b ± 0.02 | Clayey silt |
| AF1 | 4.4a ± 0.2 | 2.2a ± 0.2 | 33.2b ± 2.89 | 1.1b ± 0.05 | Clay |
| AF2 | 5.9bc ± 0.9 | 3.8b ± 0.7 | 43.6c ± 4.65 | 0.9a ± 0.06 | Clay |
| B1 | 6.7cd ± 0.5 | 3.7b ± 0.7 | 36.2b ± 9.75 | 1.0ab ± 0.07 | Silty clay |
| B2 | 5.0ab ± 0.9 | 2.2a ± 0. 3 | 32.8ab ± 3.31 | 1.0b ± 0.04 | Silty clay loam |
| B3 | 6.0bc ± 1.0 | 2.2a ± 0.3 | 29.7a ± 4.11 | 1.0ab ± 0.06 | Silty clay loam |
| B4 | 6.9c ± 0.1 | 3.0ab ± 0.9 | 34.0ab ± 4.97 | 1.1c ± 0.1 | Silty clay loam |
| B5 | 6.9cd ± 0.1 | 2.9a ± 0.6 | 36.8ab ± 8.10 | 1.1c ± 0.05 | Silty clay loam |
| B6 | 6.2bc ± 0.9 | 1.7a ± 0.5 | 34.7ab ± 3.55 | 1.2d ± 0.03 | Clay loam |
| (b) 10–30 cm | |||||
| S1 | 6.9c ± 0.2 | 5.0c ± 1.2 | 48.5c ± 4.07 | 0.9a ± 0.06 | Clayey silt |
| S2 | 5.9b ± 0.4 | 3.1b ± 0.3 | 46.3bc ± 3.04 | 1.1b ± 0.02 | Clayey silt |
| AF1 | 3.9a ± 0.1 | 2.0a ± 0.3 | 33.2ab ± 3.23 | 1.0ab ± 0.05 | Clay |
| AF2 | 6.5bc ± 0.4 | 2.5ab ± 0.2 | 45.0bc ± 4.18 | 0.9a ± 0.03 | Clayey silt |
| B1 | 6.7c ± 0.2 | 2.6ab ± 0.2 | 39.0b ± 7.56 | 0.9a ± 0.07 | Silty clay |
| B2 | 4.3a ± 0.7 | 2.0a ± 0.4 | 32.2ab ± 2.00 | 1.0ab ± 0.08 | Clay loam |
| B3 | 6.0bc ± 1.0 | 2.0a ± 0.7 | 28.6a ± 4.46 | 1.1b ± 0.07 | Silt loam |
| B4 | 6.8c ± 0.2 | 2.0a ± 0.5 | 34.1ab ± 7.73 | 1.0ab ± 0.1 | Clay loam |
| B5 | 6.9c ± 0.2 | 1.9a ± 0.6 | 32.6ab ± 11.2 | 1.2c ± 0.9 | Silt loam |
| B6 | 6.2bc ± 0.9 | 1.9a ± 0.6 | 33.5ab ± 1.84 | 1.2c ± 0.5 | Silty clay loam |
Mean and standard deviation, significant difference (p < 0.05): a < b < c < d, n = 5, OM organic matter, AD appearing density
Three principal components represent 69.5% of the total variation. The organic matter component explains 34.3% of the variation, the pH component represents 20.9% of the variation, and the third component, the total Zn and Mn content, accounts for 14.3% of the total variation. The available Zn and Mn content has a strong positive correlation to the pH component (Fig. 2).
Fig. 2.
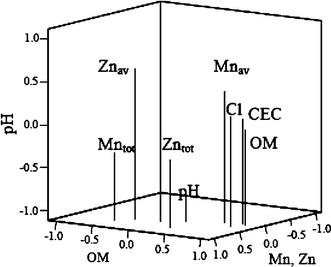
Components extracted from physical–chemical soil properties (OM organic matter, av available, tot total, Cl clay)
The ETU content in all the banana soils (both depths) was close to the detection limit of 0.01 mg kg−1 (Table 2). This low ETU content results from weak adsorption in soil and high solubility in water (2 g 100 mL−1) (Lyman and Lacoste 1974). Because of its low Koc (70 mL g−1), ETU does not adsorb to organic matter and tends to move in surface water, aquifers and the air (Rajagopal et al. 1984). The low content of ETU in the soils during the dry season is probably due to the high temperatures and high sunlight intensity that leads to decomposition of this compound while it is still on the banana leaves, as well in the soils (Calumpang et al. 1993; FAO 2000).
Table 2.
a, b ETU, total, available Mn and Zn content and ratio Mn (Zn) avail/tot in soils of banana plantations (B1–B6), agro forestry systems (AF), succesion forest (S) at depths 0–10 cm and 10–30 cm
| Site | ETU (mg kg−1) | Mn tot (mg kg−1) | Mn avail (mg kg−1) | Mn avail/Mn tot % | Zn tot (mg kg−1) | Zn avail (mg kg−1) | Zn avail/Zn tot % |
|---|---|---|---|---|---|---|---|
| (a) 0–10 cm | |||||||
| S1 | ND | 507a + 38.5 | 45.1d + 7.1 | 8.89c ± 1.1 | 101b + 15.9 | 0.4a + 0.2 | 0.41a ± 0.1 |
| S2 | ND | 474a + 72.3 | 3.1ab + 0.7 | 0.66a ± 0.1 | 81.6a + 7.2 | 0.2a + 0.02 | 0.34a ± 0.1 |
| AF1 | ND | 626b + 90.7 | 19.6c + 4.8 | 3.13b ± 0.9 | 77.6a + 7.5 | 1.6b + 0.4 | 2.07c ± 0.6 |
| AF2 | ND | 704b + 98.9 | 8.4ab + 6.5 | 1.20ab ± 1.0 | 98.2b + 6.4 | 1.4b + 0.3 | 1.44b ± 0.3 |
| B1 | <0.01 | 784b + 111 | 6.7ab + 9.4 | 0.86ab ± 0.9 | 91.9b + 9.1 | 1.2ab + 0.7 | 1.33b ± 0.6 |
| B2 | <0.01 | 651b + 66.9 | 14.3bc + 7.6 | 2.2b ± 1.3 | 77.3a + 5.8 | 1.3ab + 0.9 | 1.69b ± 1.0 |
| B3 | <0.01 | 718b + 57.5 | 7.9ab + 3.9 | 1.11ab ± 0.6 | 80.8a + 9.5 | 1.2ab + 0.2 | 1.47b ± 0.3 |
| B4 | <0.01 | 749b + 61.9 | 1.8a + 0.7 | 0.25a ± 0.1 | 83.6a + 7.8 | 1.0ab + 0.3 | 1.24b ± 0.2 |
| B5 | <0.01 | 774b + 108 | 1.6a + 0.3 | 0.21a ± 0.1 | 96.2b + 35.6 | 0.6a + 0.1 | 0.61a ± 0.2 |
| B6 | <0.01 | 707b + 106 | 2.6ab + 1.9 | 0.38a ± 0.3 | 91.9b + 20.2 | 0.4a + 0.1 | 0.47a ± 0.1 |
| (b) 10–30 cm | |||||||
| S1 | ND | 489a + 30.0 | 41.9d + 8.5 | 8.57c ± 1.3 | 90.9b + 3.5 | 0.2a + 0.1 | 0.26a ± 0.1 |
| S2 | ND | 547ab + 109 | 5.6ab + 4.3 | 1.02ab ± 0.8 | 81.5b + 9.6 | 0.1a + 0.3 | 0.15a ± 0.1 |
| AF1 | ND | 650b + 214 | 17.8c + 4.2 | 2.72b ± 1.3 | 82.9b + 9.7 | 1.0c + 0.2 | 1.28b ± 0.2 |
| AF2 | ND | 684b + 84.0 | 3.1a + 1.1 | 0.46a ± 0.2 | 95.3b + 8.1 | 0.6b + 0.1 | 0.66a ± 0.1 |
| B1 | <0.01 | 755b + 73.8 | 3.4a + 2.5 | 0.45a ± 0.3 | 91.9b + 22.2 | 0.3a + 0.2 | 0.35a ± 0.2 |
| B2 | <0.01 | 628b + 95.0 | 12.3b + 1.6 | 1.96b ± 0.6 | 71.7a + 5.0 | 0.7b + 0.2 | 1.0b ± 0.3 |
| B3 | <0.01 | 713b + 124 | 6.4ab + 4.2 | 0.9ab ± 0.5 | 81.6b + 11.0 | 0.5ab + 0.2 | 0.59a ± 0.2 |
| B4 | <0.01 | 714b + 35.2 | 1.5a + 0.7 | 0.22a ± 0.1 | 80.4b + 4.5 | 0.3a + 0.1 | 0.43a ± 0.1 |
| B5 | <0.01 | 834b + 96.6 | 1.8a + 0.3 | 0.21a ± 0.1 | 85.5b + 3.3 | 0.3a + 0.05 | 0.34a ± 0.1 |
| B6 | <0.01 | 622b + 107 | 2.5a + 1.6 | 0.4a ± 0.3 | 83.3b + 6.7 | 0.3a + 0.09 | 0.38a ± 0.1 |
Means and standard deviation, significant differences (p < 0.05): a < b < c < d), n = 5: avail available, tot total, ND no data
The Mntot content in the banana and agro forestry soils was significantly greater than in the forest soils S1 and S2 at both depths. However, the content of available Mn and the Mnavail/Mntot ratio were significantly greater in S1 soil than in all other soils at both depths. The Mnavail content and Mnavail/Mntot ratio in the soils at AF1 and B2 were significantly greater than in the other banana soils and at AF2 (Table 2a and b).
The Zntot content was significantly greater in S1 than in all other sites and significantly lower in B2 than in the other sites. The available Zn content ranged between 1.6 mg kg−1 in AF1 to 0.1 in B2, with the most significant content in AF1 (10–30 cm). In the 0–10 cm depth, contents were significantly greater in the soils in AF1 and AF2 than in the succession forests and B5 and B6 (Table 2a and b). The Znavail/Zntot ratio in AF1 was significantly greater (2.07%) than in all other sites in the 0-10 cm depth. In the lower soil depth the ratio was significantly higher in AF1 (1.28%) and B2 (1.0%) than in the other sites.
The high content of Mntot in banana and agro forestry soils is the result of the constant use of the fungicide Mancozeb over 10 years. The accumulation of Mn in the soils may be in response to the dithiocarbamates redistributing manganese, as a product of decomposition, and liberating the ions present in their structures (WHO 1988). The rapid decomposition of Mancozeb incorporates all the decomposition sub-products into the soil, including manganese and zinc ions (Xu 2000a). Considering a yearly Mancozeb application of 130 kg ha−1 (=1,300 kg in 10 years) containing 20% Mn and 2.6% Zn, we calculated an increase in the soil (0–10 cm) for Mn of 260 mg kg−1 and for Zn of 34 mg kg−1. Our results confirmed this theoretical calculation.
We measured a severe accumulation of Mntot comparing the contents of Mntot in banana and agro forestry soils with the forest reference soil. Threshold values for Mn in soils do not exist. However, Melgar et al. (2008) found Mn accumulation up to 20,000 mg kg−1 in creek sediments in the same study region. This suggests that the Mn adsorbed onto the soil is transported by preferential erosion into the creeks resulting in levels that far exceed the threshold values for aquatic life. The high content of available Mn in the soil at S1 is probably caused by the extended flooding periods which result in an increased solution and availability of Mn. The acidity of the soils at AF1 and B2 cause a high availability of Mn (Howe et al. 2004). We conclude that the frequent application of Mancozeb leads to severe accumulation of Mn in the environment causing risks for aquatic live.
No significant accumulation of Zntot was recorded in the banana and agro forestry soils because of its low concentration in Mancozeb. All Zn contents were near the threshold values for soils (98 mg kg−1) as established by the German Federal Soil Protection and Contaminated Sites Ordinance (BbodSchV 1999). The greatest available content was measured in the acid soils at AF1 and B2. This result confirmed the observations of Ahn (1993) and Howe et al. (2004) who describe a high mobility of Zn in acid soils.
The greatest concentrations of ETU in water were recorded for the drain water D (22.5 μg L−1, 15.5 μg L−1) and in the creek C (21.5 μg L−1), with values significantly greater than those in other water bodies (Table 3). The mean concentration of ETU in sub superficial water was 4.3 μg L−1. We did not detect ETU in the water of the lake (L) or in the ground water (GW). Mn concentration varied in all water bodies from 2.3 to 2.4 mg L−1, with no significant differences for all the sampled water bodies. We did not detect Zn in the water samples.
Table 3.
pH, ETU, Mn and Zn concentration in surface water of the drain (D), creek (C), lake (L) subsurface (SS) and ground water (GW)
| Site | Depth cm/m | pH | ETU (mg L−1) | Mn (mg L−1) | Zn (mg L−1) |
|---|---|---|---|---|---|
| C | 0–10 cm | 7.4 ± 3.7 | 21.5 ± 15.2c | 2.4 ± 0.0 | <0.01 |
| C | 10–50 cm | 7.3 ± 3.7 | <0.01a | ND | ND |
| D | 0–10 cm | 7.4 ± 3.7 | 15.5 ± 16.7c | 2.3 ± 0.1 | <0.01 |
| D | 10–50 cm | 7.4 ± 3.7 | 22.5 ± 25.0c | ND | ND |
| L | 0–10 cm | 7.7 ± 3.8 | <0.01a | 2.3 ± 0.1 | <0.01 |
| L | 10–50 cm | 7.1 ± 0.1 | <0.01a | ND | ND |
| SS | 3–5 m | 7.6 ± 3.9 | 4.3 ± 5.3b | 2.3 ± 0.1 | <0.01 |
| GW | 70–80 m | 7.3 ± 0.2 | <0.01a | 2.4 ± 0.1 | <0.01 |
ND No data
Detection limit: ETU 0.01 ug L−1
Pesticide limit according to the European Union (European Commission 1998): 0.1 ug L−1
Means and standard deviation, significant differences between the water bodies (p < 0.05): a < b < c n = 3
The high concentrations of ETU in the water of drain D (22.5 μg L−1, 15.5 μg L−1) and creek C (21.5 μg L−1) are evidence of the high solubility of ETU in water and its transportation in surface water. These results are different from those reported by Xu (2000b) who described a rapid photolysis and hydrolysis of ETU. These concentrations are above the threshold values for ETU for aquatic organisms such as invertebrates (1 μg L−1) and algae (1–4 μg L−1) suggested by the WHO (1988). In the lake ETU was not detected probably because of dilution. The concentration of ETU in sub-superficial water is elevated, and is a response to the high leaching rate and probable underground transport (Rajagopal et al. 1984). No ETU was detected in groundwater, as the layer of clay serves as a waterproof barrier.
As previously mentioned the water in the area is for human consumption. The concentrations recorded are markedly above the permitted limits of 0.1 μg L−1 proposed by the European Commission (1998) for potable water. The lixiviation of ETU probably results from flow through the macro pores in the soil. No data have been reported on the health of the population because of restrictions imposed by health institutions.
The frequent use of Mancozeb results in high levels of environmental pollution which present significant risks for aquatic life and human health. We strongly recommend the application of integrated management strategies to drastically reduce the use of bisdithiocarbamates.
Acknowledgments
We thank SEDAFOP Tabasco and the Fundación Produce Tabasco for financial aid.
Open Access
This article is distributed under the terms of the Creative Commons Attribution Noncommercial License which permits any noncommercial use, distribution, and reproduction in any medium, provided the original author(s) and source are credited.
References
- Ahn PM. Troical soils and fertilizer use. 1. Malaysia: Longman Group UK Limited, TCP; 1993. [Google Scholar]
- BBodSchV (1999) German federal soil protection and contaminated sites ordinance. Available via www.bmu.bund.de/files/pdfs/allgemein/…/pdf/bbodschv_uk.pdf
- Calumpang SMF, Medina MJB, Roxas NP, Magallona ED. Movement and degradation of mancozeb fungicide and its metabolites, ethylenethiourea and ethyleneurea in silty clay loam soil. Internat J Pest Man. 1993;39:161–166. doi: 10.1080/09670879309371783. [DOI] [Google Scholar]
- Dubey JK, Heberer T, Stan HJ. Determination of ethylenethiourea in food commodities by a two-step derivatization method and gas chromatography with electron-capture and nitrogen-phosphorus detection. J of Chrom. 1997;765:31–38. doi: 10.1016/S0021-9673(96)01008-4. [DOI] [PubMed] [Google Scholar]
- European Commission (1998) Council directive 98/93 EC. Drinking water directive
- FAO (2000) Evaluación de la contaminación del suelo. Manual de referencia. Documento de campo GCP/INT/650/NET producido por el Departamento de Agricultura, Roma, Italia
- Franson F (1992) Standard methods for the examination of water and wastewater. ISBBN 84.7978-031-2
- Geissen V, Morales G. Fertility of tropical soils under different land use systems-a case study of soils in Tabasco, Mexico. J Appl Soil Ecol. 2006;31:169–178. doi: 10.1016/j.apsoil.2005.02.012. [DOI] [Google Scholar]
- Hayes WJ, Laws ER. Hand book of pesticide toxicology. Classes of pesticides. New York, NY: Academic Press; 1991. [Google Scholar]
- Howe PD, Malcolm HM, Dobson S. Manganese and its compounds: environmental aspects. United Nations environment programme. Geneva: International Labour Organization and World Health Organization; 2004. [Google Scholar]
- Konstantinou IK, Hela DG, Albanis TA. The status of pesticide pollution in surface waters (rivers and lakes) of Greece. Part I. Review on occurrence and levels. Environ Poll. 2006;141:555–570. doi: 10.1016/j.envpol.2005.07.024. [DOI] [PubMed] [Google Scholar]
- Lyman WR, Lacoste RL. New developments in the chemistry and fate of ethylenebis (dithiocarbamate) fungicides. Environ Quality Safety. 1974;3:67–74. [PubMed] [Google Scholar]
- Melgar Valdes CE, Geissen V, Cram S, Sokolov M, Bastidas P, Ruiz-Suárez LE, Que Ramos FY, Jarquín-Sanchez A. Occurrence of pollutants in drainage channels after long-term application of mancozeb to banana plantations in SE Mexico. JPNSS. 2008;17(4):597–604. [Google Scholar]
- Mortensen SR, Johnson KA, Weisskopf CP, Hooper MJ, Lacher TE, Kendall PJ. Avian exposure to pesticides in Costa Rican banana plantations. Bull Environ Contam Toxicol. 1998;60:562–568. doi: 10.1007/s001289900662. [DOI] [PubMed] [Google Scholar]
- Persson M, Berndtsson R, Nasri S, Albergel J, Za P, Yumegaki Y. Solute transport and water content measurements in clay soils using time domain reflectometry. Hydrol Sci J. 2000;45(6):833–847. doi: 10.1080/02626660009492387. [DOI] [Google Scholar]
- Racke KD, Skidmore M, Hamilton DJ, Unsworth JB, Miyamoto J, Cohen SZ. Pesticide fate in tropical soils. Appl Chem. 1997;69:1349–1371. doi: 10.1351/pac199769061349. [DOI] [Google Scholar]
- Rajagopal BS, Brahmaprakash GP, Reddy BR, Singh UD, Sethunathan N. Effect and persistence of selected carbamate pesticides in soil. Residue Rev. 1984;93:1–199. [Google Scholar]
- SEMARNAT (2000) Secretaría de Medio Ambiente y Recursos Naturales. Norma Oficial Mexicana NOM-021-RECNAT-2000. Segunda sección
- Shukla Y, Arora A. Transplacental carcinogenic potential of the carbamate fungicide mancozeb. J Environ Pathol Toxicol Oncol. 2001;20(2):127–131. [PubMed] [Google Scholar]
- WHO, World Health Organization (1988) Dithiocarbamate pesticides, ethylenethiourea, and propylenethiourea: a general introduction. Environ Health Criteria, 78
- WHO, World Health Organization (2005) Ecosystems and human well being. A report of the millennium ecosystem assessment
- Xu S. Environmental fate of mancozeb. Environmental monitoring and pest management. United States: Sacramento; 2000. [Google Scholar]
- Xu S. Environmental fate of ethylenethiourea. California: California Department of Pesticide Regulation; 2000. [Google Scholar]


