Abstract
Since carbapenemase-producing Klebsiella pneumoniae strains were first reported in North Carolina, these highly resistant organisms have been isolated with increasing frequency, especially in the New York City area. Polymyxin B is one of the few antimicrobials that retain reliable activity against these organisms. However, polymyxin B MICs are elevated against K. pneumoniae isolates with increasing frequency, leaving clinicians with few therapeutic options. We investigated several antimicrobial agents for potential synergy with polymyxin B against 12 clinical strains of carbapenemase-producing K. pneumoniae. A broth microdilution assay using a 96-well plate was developed in which graded dilutions of polymyxin B and the study drug were incubated with resistant isolates in a checkerboard pattern. Polymyxin B was studied in combination with cefazolin, ceftriaxone, cefepime, imipenem, gentamicin, tigecycline, doxycycline, and rifampin. All K. pneumoniae strains tested positive for K. pneumoniae carbapenemase (KPC) genes by real-time PCR and had elevated polymyxin B MIC values ranging from 16 to 128 μg/ml. Synergy was observed with the combination of polymyxin B and rifampin as well as with polymyxin B and doxycycline, resulting in at least a 4-fold decrease in the polymyxin B MIC. For both combinations, this effect occurred at physiologically achievable concentrations. Less pronounced synergy was noted with tigecycline and polymyxin B. No synergy was observed at physiologic concentrations with the other antimicrobials studied. These results suggest that rifampin, doxycycline, and tigecycline may be useful additions to polymyxin B in the treatment of infections caused by highly resistant carbapenemase-producing K. pneumoniae. Further studies are warranted to determine if these in vitro findings translate into clinical efficacy.
The treatment of highly resistant strains of Gram-negative organisms is one of the most challenging problems in infectious diseases (5, 11). Highly resistant strains of Pseudomonas aeruginosa and Acinetobacter species have long been reported as causes of nosocomial infections. Recent years have also seen the emergence of carbapenemase-producing strains of Klebsiella pneumoniae (5, 7, 18). In the United States, carbapenemase resistance is mediated primarily by a beta-lactamase known as Klebsiella pneumoniae carbapenemase (KPC). Organisms harboring KPC genes are usually also resistant to most other drug classes, and such isolates have been reported for other members of the Enterobacteriaceae as well (1, 13, 16). The polymyxins (i.e., polymyxin B and colistin), long abandoned in favor of more effective and less toxic alternatives, have now become a mainstay of therapy in the treatment of KPC-producing K. pneumoniae infections and infections with other highly drug-resistant Gram-negative organisms (6, 8, 13, 20, 21). However, elevated polymyxin MICs are being reported with increasing frequency for strains of K. pneumoniae (2, 4, 9). Infections with these pan-resistant organisms leave clinicians with few or no therapeutic options.
In cases where there is a lack of effective agents, combinations of two or more antibiotics are often used in the hopes of achieving a synergistic effect. There is some literature exploring combination antimicrobial therapy in the treatment of multidrug-resistant strains of Acinetobacter baumannii and Pseudomonas aeruginosa (8, 14, 15, 17, 19, 21). However, there are limited data on combination therapy for KPC-producing K. pneumoniae. Clinicians must often make decisions based on the theoretical advantages of certain drug combinations, with few data to guide their choices.
Researchers studying the polymyxins face several challenges, as the pharmacokinetic and pharmacodynamic properties of the polymyxins are not well elucidated, despite their availability for clinical use since the 1960s (6, 8, 20). Furthermore, no universally accepted breakpoints for polymyxins have been put forward for susceptibility interpretations against Enterobacteriaceae. Thus, when using polymyxins to treat KPC-producing K. pneumoniae, microbiologists and infectious disease practitioners must make a determination about resistance without reference to universally accepted breakpoints.
We explored the efficacy of several two-drug combinations of antimicrobials against highly resistant K. pneumoniae isolates. The basis for choosing polymyxin B for this study was the fact that polymyxin B is the drug of choice for the polymyxin class at our institution. Despite the lack of universally accepted breakpoints for Enterobacteriaceae, one can use breakpoints for Pseudomonas aeruginosa and Acinetobacter spp. as comparators (≤2 μg/ml for susceptibility) (3). Accordingly, the isolates studied were considered to be resistant, since extremely high polymyxin B MICs (≥16 μg/ml) were observed with these organisms. We included antibiotics used by clinicians to treat infections with resistant Gram-negative organisms, including cephalosporins, carbapenems, aminoglycosides, tetracyclines, and rifampin.
MATERIALS AND METHODS
Twelve consecutive K. pneumoniae clinical isolates were examined in this study. Isolates were included if the polymyxin B MIC was ≥16 μg/ml. The isolates were selected from specimens submitted to the microbiology laboratory of Saint Vincent Medical Center, New York, NY, between January and December 2008. The isolates were stored at −80°C in brain heart infusion broth and passaged twice on 5% sheep blood agar before studies. The initial identification and susceptibility testing were performed using a Vitek 2 instrument with Gram-negative identification and AST-GN17 susceptibility cards (bioMérieux, Inc., Durham, NC) according to the manufacturer's instructions and the interpretative guidelines established by CLSI (3). Polymyxin B susceptibility testing of the isolates was carried out using Etest gradient strips (indicated for research use only; not an FDA-approved method). The presence of blaKPC genes in isolates was investigated by a real-time PCR assay performed on a SmartCycler instrument (Cepheid, Sunnyvale, CA). The real-time PCR assay was developed and validated in-house, using a blaKPC forward primer (5′-GGCACGGCAAATGACTATG-3′) and a blaKPC reverse primer (5′-GCCAATAGATGATTTTCAGAGC-3′). The molecular beacon 5′-TET-AAGGATGACAAGTACAGCGATCCTT-BHQ1-3′ was used for detection of the blaKPC-3 gene, and the beacon 5′-6-carboxyfluorescein (FAM)-AAGGATGACAAGCACAGCGATCCTT-BHQ1-3′ was used for detection of the blaKPC-2 gene. Amplification was carried out by use of Platinum Taq polymerase and a real-time PCR kit (Invitrogen, Carlsbad, CA) under the following cycling conditions: 95°C for 2 min for initial denaturation, followed by 35 cycles of 95°C for 15 s, 64°C for 30 s, and 72°C for 10 s.
Initial MIC data for antibiotics generated by Vitek 2 testing were used to screen the isolates to determine their eligibility for the study. MICs of each antibiotic used in synergy studies were also determined by broth microdilution tests, and the data obtained from broth microdilution tests were used in calculations of synergy (12). Dilutions were tested in duplicate. The method was modified to increase the testing volume to 150 μl. Briefly, using 96-well U-bottom microplates (Dynex Technologies Inc., Chantilly, VA), graded concentrations of antibiotics were mixed, each well was inoculated with 104 CFU of microorganisms, and the medium was cation-adjusted Mueller-Hinton broth. The following antibiotics were used in the assays: rifampin, doxycycline (Bedford Laboratories, Bedford, OH), polymyxin B (XGEN Pharmaceuticals Inc., Northport, NY), ceftriaxone (Sandoz Inc., Princeton, NJ), cefazolin (Baxter Healthcare Corporation, Deerfield, IL), imipenem (Merck and Company Incorporated, Whitehouse Station, NJ), cefepime (Sagent Pharmaceuticals, Inc., Schaumburg, IL), gentamicin (APP Pharmaceuticals, LLC, Schaumburg, IL), and tigecycline (Wyeth, Madison, NJ).
The plates were incubated for 24 h at 35°C in ambient air. The presence or inhibition of microbial growth was determined visually. Total fractional inhibitory concentrations (FIC) for each well were calculated according to the formula ΣFIC = FIC of agent A + FIC of agent B, where FIC of agent A or B = MIC of agent A or B in combination/MIC of agent A or B alone (12).
In interpreting the results of calculations, the following principles were used: ΣFIC values of ≤0.5 indicate synergism, ΣFIC values of 0.5 to 4 indicate indifference, and ΣFIC values of >4 indicate antagonism (12).
In all MIC determinations, Escherichia coli ATCC 252922 was included as a quality control strain, and all antibiotics were also tested against this strain simultaneously.
RESULTS
The sources of the isolates and their susceptibility profiles are shown in Table 1. The majority of the isolates were from the respiratory tract. All isolates used were resistant to all of the tested antimicrobials, with the exception of doxycycline, tigecycline, and gentamicin. Relatedness analysis by molecular DNA typing of the isolates via pulsed-field gel electrophoresis (PFGE) indicated that the isolates fell into 4 different clusters (data not shown). No difference was discernible between the isolates from different clusters with regard to the synergistic effects observed with different antibiotic combinations.
TABLE 1.
Sources of isolates and MICs of various antibioticsa
| Isolate | Source | MIC (μg/ml) |
|||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| PB | RIF | Doxy | FEP | IMI | CZ | CRO | GM | TG | CL | ||
| 1 | Sputum | 32 | >32 | 8 | 32 | 16 | ≥64 | ≥64 | ≥16 | 2 | 8 |
| 2 | Tracheal aspirate | 32 | 32 | 8 | 64 | 16 | ≥64 | ≥64 | ≥16 | 2 | 8 |
| 3 | Tracheal aspirate | 128 | 32 | 4 | 64 | 16 | ≥64 | ≥64 | ≥16 | 2 | 8 |
| 4 | Sputum | 32 | 32 | 4 | 128 | 16 | ≥64 | ≥64 | ≥16 | 2 | 6 |
| 5 | Axilla | 64 | 32 | 4 | 64 | 32 | ≥64 | ≥64 | ≥16 | 2 | 8 |
| 6 | Tracheal aspirate | 32 | 32 | 4 | 32 | 16 | ≥64 | 32 | ≥16 | 2 | 8 |
| 7 | Urine | 64 | >32 | 8 | >128 | 64 | ≥64 | ≥64 | ≥16 | 8 | 4 |
| 16 | Tracheal aspirate | 16 | 32 | 4 | 32 | 32 | ≥64 | ≥64 | ≥16 | 2 | 12 |
| 17 | Urine | 32 | >32 | 8 | 64 | 32 | ≥64 | ≥64 | 8 | 2 | 8 |
| 29 | Urine | 32 | 32 | 8 | 32 | 32 | ≥64 | ≥64 | ≥16 | 2 | 12 |
| 32 | Sputum | 64 | >32 | 8 | 32 | 32 | ≥64 | ≥64 | 8 | 8 | 12 |
| 35 | Catheter tip | 128 | >32 | 16 | 32 | 32 | ≥64 | ≥64 | 8 | 4 | 12 |
The isolates were tested against polymyxin B (PB), rifampin (RIF), doxycycline (Doxy; susceptible MIC [S] ≤ 4 μg/ml, intermediate MIC [I] = 8 μg/ml, resistant MIC [R] ≥ 16 μg/ml), cefepime (FEP; S ≤ 8 μg/ml, I = 16 μg/ml, R ≥ 32 μg/ml), imipenem (IMI; S ≤ 4 μg/ml, I = 8 μg/ml, R ≥ 16 μg/ml), cefazolin (CZ; S ≤ 8 μg/ml, I = 16 μg/ml, R ≥ 32 μg/ml), ceftriaxone (CRO; S ≤ 8 μg/ml, I = 16 to 32 μg/ml, R ≥ 64 μg/ml), gentamicin (GM; S ≤ 4 μg/ml, I = 8 μg/ml, R ≥ 16 μg/ml), tigecycline (TG; S ≤ 4 μg/ml, I = 8 μg/ml, R ≥ 16 μg/ml), and colistin (CL).
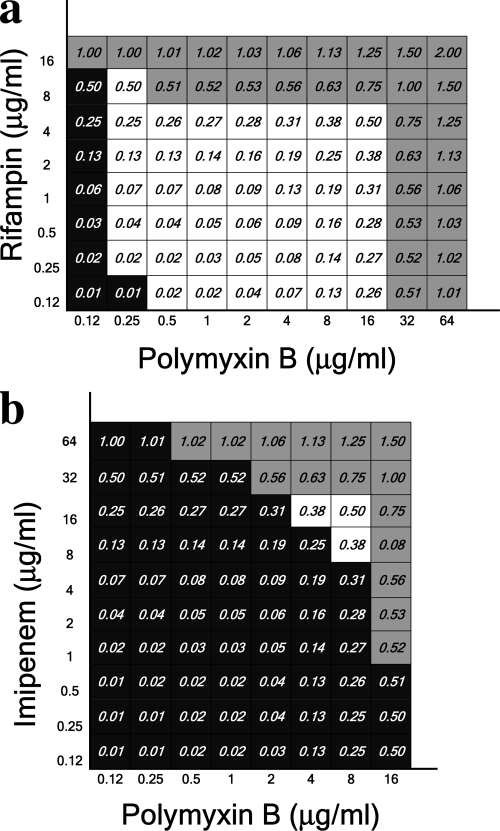
For all 12 isolates tested, there was synergism between polymyxin B and rifampin (Fig. 1a) at physiologically attainable concentrations of both drugs. This was not evident for imipenem (Fig. 1b), cefepime (not shown), cefazolin (not shown), or ceftriaxone (not shown). The data demonstrated in Fig. 1 were generated from one representative isolate (isolate 7). Similar results were achieved with the other resistant strains.
FIG. 1.
Total fractional inhibitory concentrations of polymyxin B combined with rifampin (a) and imipenem (b). Black squares indicate bacterial growth in the wells. Gray squares indicate the wells for which total FIC values exceeded 0.5, which was considered the cutoff value for a synergistic effect. White squares demonstrate the antibiotic combinations for which bacterial growth was inhibited and total FIC values were <0.5 and which, consequently, were considered the combinations producing synergy. Data obtained for isolate 7 are presented.
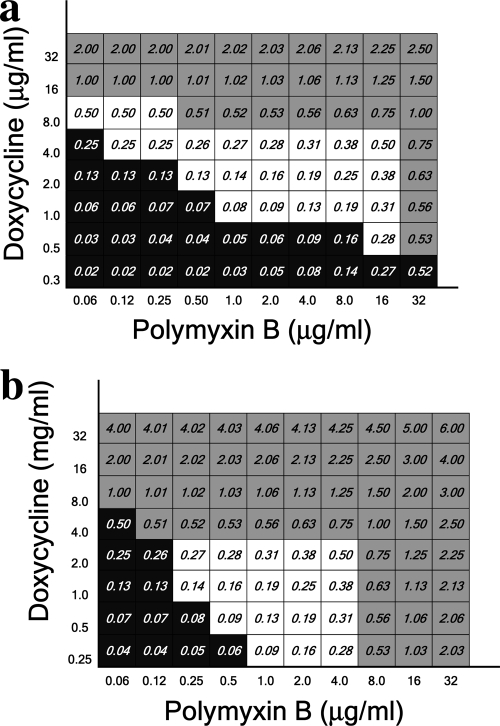
Doxycycline-polymyxin B (Fig. 2) and tigecycline-polymyxin B (Fig. 3) were also tested against the isolates. Similar to the case in the rifampin experiments, for both doxycycline-resistant and -susceptible isolates (Fig. 2a and b, respectively), the combination with polymyxin B exhibited synergy, as evidenced by a total FIC of ≤0.5 at achievable serum antibiotic concentrations for both drugs.
FIG. 2.
Total fractional inhibitory concentrations of polymyxin B combined with doxycycline-resistant (a) and doxycycline-susceptible (b) isolates. Data obtained for isolates 35 (a) and 3 (b) are presented.
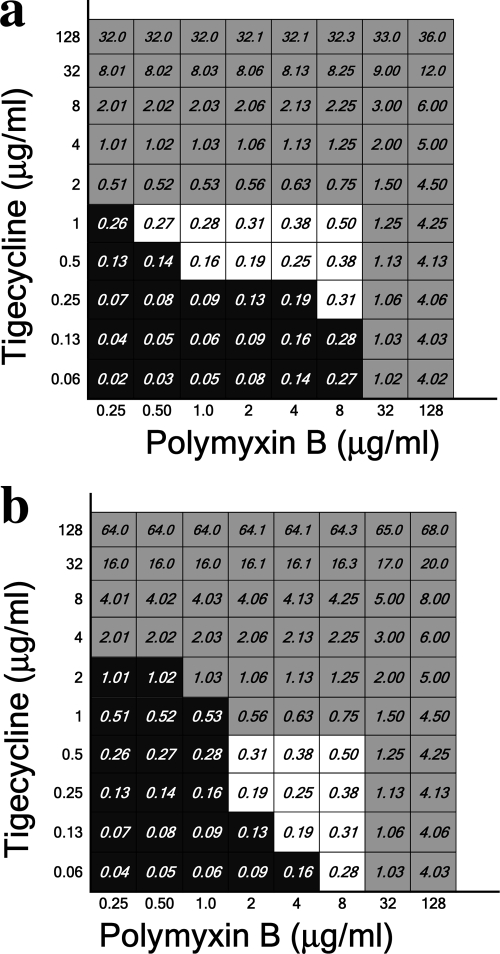
FIG. 3.
Total fractional inhibitory concentrations of polymyxin B combined with tigecycline-resistant (a) and tigecycline-susceptible (b) isolates. Data obtained for isolates 7 (a) and 3 (b) are presented.
To investigate the interaction between tigecycline and polymyxin B, graded concentrations of both drugs were tested against the isolates. The results for tigecycline-resistant and tigecycline-susceptible isolates are presented in Fig. 3a and b, respectively. For all isolates, a detectable synergism was evident at antibiotic concentrations lower than the MIC of each antibiotic by itself. However, the window of dilutions for which synergism was detected was narrower for tigecycline-polymyxin B combinations than for rifampin-polymyxin and doxycycline-polymyxin B combinations.
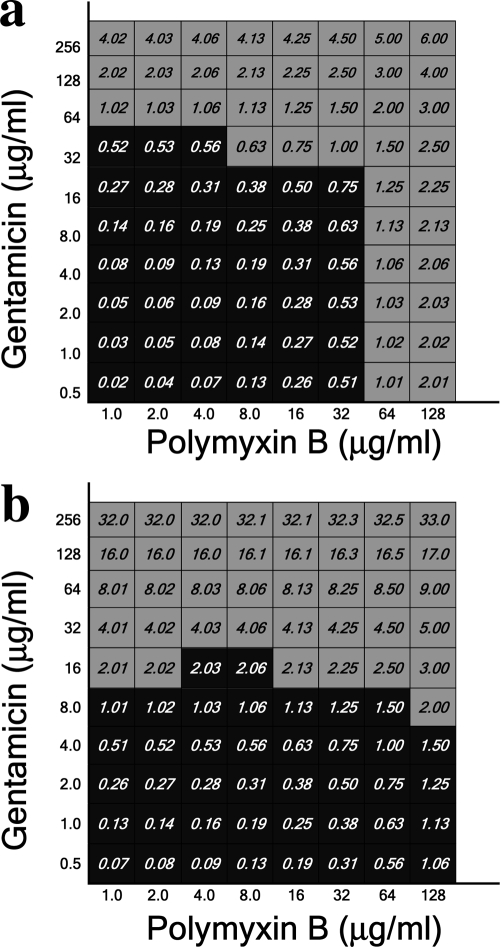
Gentamicin-resistant and intermediate isolates were also tested against polymyxin B and gentamicin combinations (Fig. 4a and b, respectively). No interactions were noticed between the two drugs.
FIG. 4.
Total fractional inhibitory concentrations of polymyxin B combined with gentamicin-resistant (a) and gentamicin-susceptible (b) isolates. Data obtained for isolates 7 (a) and 35 (b) are presented.
DISCUSSION
This study demonstrates that the combination of polymyxin B and either rifampin or doxycycline has in vitro synergistic activity against KPC-producing Klebsiella pneumoniae isolates against which polymyxin B alone has an increased MIC. This synergy was apparent at physiologically attainable concentrations of both drugs, suggesting that the results may be relevant to clinical management. Tigecycline also demonstrated synergistic activity when combined with polymyxin B, but the synergy was confined to a small number of antibiotic dilutions, whereas for rifampin-polymyxin B and doxycycline-polymyxin B combinations, synergistic activity was noticeable in a wider window of antibiotic dilutions.
To our knowledge, this is the first report indicating synergy between polymyxin B and another antibiotic against multiple KPC-producing isolates for which polymyxin B MICs are elevated. There are several reports in the literature of synergy studies performed with multidrug-resistant Acinetobacter baumannii (14, 15, 19). However, the synergy findings were not universal and may have been strain dependent (17), making it difficult to generalize the results. In addition, the use of different synergy testing methods further complicated the interpretation of the results. There is also one previously published report (2) showing synergy between rifampin and polymyxin B against K. pneumoniae strains. However, the strains tested in that study were largely susceptible to polymyxin B, with only two isolates being considered resistant. In their study, Bratu et al. (2) noticed synergy for 15 of 16 isolates with rifampin and polymyxin B combinations. Imipenem-polymyxin B combinations were also synergistic with 10 of 16 isolates and antagonistic with 3 isolates. In contrast, in the present investigation, all of the isolates were resistant to polymyxin B. While it is difficult to conclude if a combination of antibiotics might be synergistic with any given isolate, we find it promising that we achieved very similar results for all 12 tested isolates.
Polymyxin B and E (colistin) are considered to have similar modes of action, spectra of activity, mechanisms of resistance, and clinical uses (6), though there are limited comparative data. In the present report, the synergy experiments were performed only with polymyxin B. MICs of colistin against all isolates were determined, and it was noticed that all isolates were also resistant to colistin (Table 1). Though the observed synergy may also occur with colistin, this remains to be demonstrated.
Prior experience with rifampin in combination therapy has been studied primarily with Staphylococcus aureus. Rifampin is not traditionally considered a drug for use against Gram-negative organisms. However, our findings as well as the above-mentioned studies highlight a growing utility of rifampin in combination with other drugs in treating highly resistant Gram-negative organisms. While in vitro studies are limited in clinical applicability, our findings are promising in that we have shown synergy against isolates that would otherwise be very difficult to treat. Further in vitro studies with a broader range of strains, as well as clinical outcome studies, are needed before combination therapy can be recommended as a standard of care for serious KPC-positive Klebsiella pneumoniae infections.
Our results also demonstrated synergy when doxycycline was combined with polymyxin B. Furthermore, doxycycline, in contrast to rifampin, had MIC values within the susceptible range against 42% of the polymyxin B-resistant isolates. These results imply that in addition to being used in combination with polymyxin B, doxycycline alone may be of use against KPC-positive K. pneumoniae, in cases where susceptibility of the organism has been established in the laboratory. Unlike rifampin, doxycycline is well established for use as single-agent therapy against other Gram-negative organisms, including Klebsiella species. However, the clinical efficacy of doxycycline in such resistant cases has not yet been determined. We are unaware of any studies involving doxycycline in combination therapy. This may be due to a traditional hesitation on the part of clinicians to combine bacteriostatic and bactericidal drugs out of concern for the possibility of antagonism. While this may be a valid theoretical concern, in situations where therapeutic options are limited, it may be worthwhile to pursue unusual treatment options. Also, this concern may be more applicable in the context of combining a bacteriostatic drug with an agent that is dependent on active cell wall synthesis to achieve its effect. The polymyxins' mechanism of action is not dependent on a dividing cell wall and therefore may not be affected by being combined with a bacteriostatic drug. Further studies in this area are needed to determine if doxycycline should be considered for broader use in combination therapy.
Tigecycline, a glycylcycline with activity against many multidrug-resistant Gram-negative organisms, including KPC-positive Klebsiella pneumoniae isolates, also demonstrated synergistic activity in combination with polymyxin B. Tigecycline attains low serum concentrations, and sporadic reports of treatment failures have generated concern with regard to its use as a single agent in the treatment of serious multidrug-resistant infections (13). There are also increasing numbers of K. pneumoniae isolates that are resistant to tigecycline. Anecdotal data suggest that many clinicians are now using tigecycline in combination with other agents, such as the polymyxins, though there are no published synergy studies of this combination against multidrug-resistant K. pneumoniae. In the current study, most isolates were susceptible to tigecycline. The results of experiments with two isolates are presented. Polymyxin B combination reduced both polymyxin B and tigecycline MICs, suggesting that tigecycline along with polymyxin B may be an effective option for treatment.
The exact mechanism of synergy observed in the present study is not known. A possible role for polymyxin B in the generation of synergistic activity is thought to be the rapid permeabilization of the bacterial outer membrane, allowing intracellularly active drugs such as rifampin, doxycycline, and tigecycline to penetrate the cells (19). However, this has not yet been proven.
The use of combination antibiotic therapy in the hopes of achieving synergy is problematic because in vitro and in vivo results do not always correlate. Also, the testing methods described in this study are too labor-intensive to be performed in daily clinical laboratory practice, making it difficult to test specimens in a timely enough manner to be relevant for routine patient care. Further studies using time-kill methodology would be useful in confirming the above findings before recommending drug combinations. Clinical trials testing such drug combinations for treatment of infections caused by KPC-producing K. pneumoniae strains are also needed.
This study presents susceptibility and synergy data for the largest collection of polymyxin B-resistant isolates published to date. We demonstrate that rifampin, doxycycline, and tigecycline have in vitro synergistic activity with polymyxin B against KPC-producing K. pneumoniae isolates. The emergence of pan-resistant isolates has left some authors dreading a return to the preantibiotic era (8, 10, 13). It is therefore in our interest to explore any potential treatment options, especially in a time when increasingly resistant organisms are being detected in the setting of a limited pipeline of new antimicrobial agents.
Footnotes
Published ahead of print on 4 August 2010.
REFERENCES
- 1.Bratu, S., S. Brooks, S. Burney, S. Kochar, J. Gupta, D. Landman, and J. Quale. 2007. Detection and spread of Escherichia coli possessing the plasmid-borne carbapenemase KPC-2 in Brooklyn, New York. Clin. Infect. Dis. 44:972-975. [DOI] [PubMed] [Google Scholar]
- 2.Bratu, S., P. Tolaney, U. Karumudi, J. Quale, M. Mooty, S. Nichani, and D. Landman. 2005. Carbapenemase-producing Klebsiella pneumoniae in Brooklyn, NY: molecular epidemiology and in vitro activity of polymyxin B and other agents. J. Antimicrob. Chemother. 56:128-132. [DOI] [PubMed] [Google Scholar]
- 3.CLSI. 2009. Performance standards for antimicrobial susceptibility testing. Nineteenth informational supplement. M100-S18. Clinical and Laboratory Standards Institute, Wayne, PA.
- 4.Elemam, A., J. Rahimian, and W. Mandell. 2007. Infection with panresistant Klebsiella pneumoniae: a report of 2 cases and a brief review of the literature. Clin. Infect. Dis. 49:271-274. [DOI] [PubMed] [Google Scholar]
- 5.Giske, C. G., D. L. Monnet, O. Cars, and Y. Carmeli. 2008. Clinical and economic impact of common multidrug-resistant Gram-negative bacilli. Antimicrob. Agents Chemother. 52:813-821. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Kwa, A., S. K. Kasiakou, V. H. Tam, and M. E. Falagas. 2007. Polymyxin B; similarities to and differences from colistin (polymyxin E). Expert Rev. Anti Infect. Ther. 5:811-821. [DOI] [PubMed] [Google Scholar]
- 7.LaBombardi, V. J. 2007. The emergence of the KPC carbapenemases: clinical and laboratory issues. Rev. Med. Microbiol. 18:29-34. [Google Scholar]
- 8.Landman, D., C. Georgescu, D. A. Martin, and J. Quayle. 2008. Polymyxins revisited. Clin. Microbiol. Rev. 21:449-465. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Lee, J., G. Patel, S. Huprikar, D. P. Calfee, and S. G. Jenkins. 2009. Decreased susceptibility to polymyxin B during treatment for carbapenem-resistant Klebsiella pneumoniae infection. J. Clin. Microbiol. 47:1611-1612. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Livermore, D. 2009. Has the era of untreatable infections arrived? J. Antimicrob. Chemother. 64(Suppl. 1):29-36. [DOI] [PubMed] [Google Scholar]
- 11.McDonald, L. C. 2006. Trends in antimicrobial resistance in health care-associated pathogens and effect on treatment. Clin. Infect. Dis. 42(Suppl. 2):S65-S71. [DOI] [PubMed] [Google Scholar]
- 12.Moody, J. 2007. Synergism testing: broth microdilution checkerboard and broth macrodilution methods. Chapter 5.12, p. 5.12.1-5.12.23. In L. S. Garcia and H. D. Isenberg (ed.), Clinical microbiology procedures handbook, 2nd edition update. ASM Press, Washington, DC.
- 13.Nordman, P., G. Cuzon, and T. Naas. 2009. The real threat of Klebsiella pneumoniae carbapenemase producing bacteria. Lancet Infect. Dis. 9:228-236. [DOI] [PubMed] [Google Scholar]
- 14.Sands, M., Y. McCarter, and W. Sanchez. 2007. Synergy testing of multidrug resistant Acinetobacter baumanii against tigecycline and polymyxin using an E test methodology. Eur. J. Clin. Microbiol. Infect. Dis. 26:521-522. [DOI] [PubMed] [Google Scholar]
- 15.Tan, T. Y., L. S. Y. Ng, E. Tan, and G. Huang. 2007. In vitro effect of minocycline and colistin combinations on imipenem-resistant Acinetobacter baumanii clinical isolates. J. Antimicrob. Chemother. 60:421-423. [DOI] [PubMed] [Google Scholar]
- 16.Walther-Ramussen, J., and N. Hoiby. 2007. Class A carbapenemases. J. Antimicrob. Chemother. 60:470-482. [DOI] [PubMed] [Google Scholar]
- 17.Wareham, D. W., and D. C. Bean. 2006. In vitro activity of polymyxin B in combination with imipenem, rifampicin and azithromycin versus multidrug resistant strains of Acinetobacter baumannii producing OXA-23 carbapenemases. Ann. Clin. Microbiol. Antimicrob. 5:10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Woodford, N., P. M. Tierno, Jr., K. Young, L. Tysall, M. F. Palepou, E. Ward, R. E. Painter, D. F. Suber, D. Shungu, L. L. Silver, K. Inglima, J. Kornblum, and D. M. Livermore. 2004. Outbreak of Klebsiella pneumoniae producing a new carbapenem-hydrolysing class A beta lactamase, KPC-3, in a New York medical center. Antimicrob. Agents Chemother. 48:4793-4799. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Yoon, J., C. Urban, C. Terzian, N. Mariano, and J. Rahal. 2004. In vitro double and triple synergistic activities of polymyxin B, imipenem, and rifampin against multidrug-resistant Acinetobacter baumannii. Antimicrob. Agents Chemother. 48:753-757. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Yuan, Z., and V. H. Tam. 2008. Polymyxin B: a new strategy for multi-drug resistant Gram-negative organisms. Expert Opin. Invest. Drugs 17:661-668. [DOI] [PubMed] [Google Scholar]
- 21.Zavascki, A. P., L. Z. Goldani, J. Li, and R. L. Nation. 2007. Polymyxin B for the treatment of multidrug-resistant pathogens: a critical review. J. Antimicrob. Chemother. 60:1206-1215. [DOI] [PubMed] [Google Scholar]