Abstract
Venous and arterial thromboembolic diseases are still the most frequent causes of death and disability in high-income countries. Clinical anticoagulants are inhibitors of enzymes involved in the coagulation pathway, such as thrombin and factor Xa. Thrombin is a key enzyme of blood coagulation system, activating the platelets, converting the fibrinogen to the fibrin net, and amplifying its self-generation by the activation of factors V, VIII, and XI. Thrombin has long been a target for the development of oral anticoagulants. Furthermore, selective inhibitors of thrombin represent a new class of antithrombotic agents. For these reasons, a number of specific thrombin inhibitors are under evaluation for possible use as antithrombotic drugs. This paper summarizes old and new interests of specific thrombin inhibitors described in different animals.
1. Introduction
Thrombin is a multifunctional serine proteinase that cleaves substrates after arginine (Arg) or lysine (Lys) residues [1]. This enzyme plays an important role in different biological phenomena, such as hemostasis, thrombosis, inflammation, and proliferative response [2, 3].
Thrombin is the key enzyme of the blood coagulation system, presenting many important biological functions, such as the activation of platelets, conversion of fibrinogen to fibrin, and feedback amplification of coagulation. The precise generation of thrombin at vascular injury sites is the result of an ordered series of reactions collectively referred to as blood coagulation cascade [4, 5].
2. Blood Coagulation
The hemostatic process is a host defense mechanism to preserve the integrity of the closed high-pressure circulatory system. This process must remain inactive but poised to immediately minimize extravasations of blood from the vasculature following tissue injury [6].
The blood coagulation cascade is initiated when subendothelial tissue factor is exposed to the blood flow following either the damage or activation of the endothelium [7, 8]. After cellular activation by vascular trauma or an inflammatory stimulus, tissue factor becomes exposed and binds to a serine protease, factor VIIa, already present in blood [9, 10], and forms the factor VIIa-tissue factor complex, in the presence of phospholipid and calcium (extrinsic factor tenase), which activates the zymogens factor IX and factor X [11]. The limited amounts of the serine protease factor Xa produced generate picomolar concentrations of thrombin, which initiates several positive feedback reactions that sustain thrombin's own formation and facilitates the rapid growth of the blood clot or thrombus around the area of vascular damage [12]. Thrombin partially activates platelets and cleaves the procofactors factor V and factor VIII generating the active cofactors factor Va and factor VIIIa, respectively [13]. Factor VIIIa forms the intrinsic factor tenase complex with the serine protease, factor IXa, phospholipid, and calcium, on a membrane surface provided by platelets and endothelial and other cells [14], and activates factor X at a 50–100-fold higher rate than the factor VIIa-tissue factor complex [15]. Factor Xa forms the prothrombinase complex with the cofactor, factor Va, phospholipid, and calcium on the membrane surface, which is the primary activator of prothrombin [16]. The thrombin produced further amplifies its own generation by activating factor XI [17] and completing the activation of platelets and factors V and VIII [13]. Thrombin also cleaves fibrinogen [18] and factor XIII [19] to form the insoluble cross-linked fibrin clot [20] that forms the backbone of a thrombus or blood clot [21, 22].
3. Thrombin
Thrombin plays a vital role in blood coagulation by promoting platelet aggregation and by converting fibrinogen to form the fibrin clot in the final step of the coagulation cascade. In addition, thrombin influences a number of other cellular effects. Besides promoting platelet aggregation, thrombin also stimulates platelets to release mediators including thromboxane A2, platelet factor 4, PDGF (platelet-derived growth factor), and TGF-β (transforming growth factor-β). Thrombin also plays a major role in the tracking of inflammatory cells into sites of injury and is chemotactic for a number of different cell types including monocytes, macrophages, and neutrophils. It also influences several key processes of tissue repair [23].
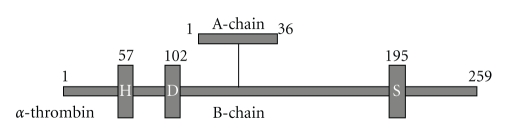
Prothrombin is synthesized in the liver as a preproprotein and undergoes a number of posttranslational processes prior to secretion. It is converted to thrombin following tissue injury by cleavage at two sites by factor Xa. The resultant 39 kDa thrombin comprises two chains A-chain, of 36 amino-acid residues in human, and B-chain, of 259 residues [2], cross-linked by four disulphide bonds (Figure 1) that house a narrow groove containing the catalytic triad consisting of His-57, Asp-102, and Ser-195. This groove is hydrophobic and exhibits a preference for apolar amino acids preceding Arg at a thrombin-susceptible bond such as Leu Asp Pro Arg/Ser (where/represents the cleavage site). The high specificity of thrombin towards its substrates and receptors is conferred by its unique anion-binding exosite. In addition, thrombin possesses three specificity pockets which interact with inhibitors [23].
Figure 1.
Thrombin linear structure scheme.
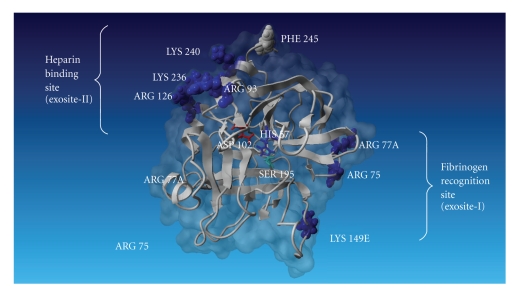
Thrombin shows the characteristic fold of the trypsin-like serine proteases. However, it has some peculiarities such as an unusual charge distribution. Although it has an almost neutral isoelectric point, charged residues are clustered, resulting in a pronounced electrostatic field that extends for a considerable distance beyond the molecular surface. The thrombin tertiary structure can be represented as shown in Figure 2, where the catalytic residues are at the edge of a negatively charged surface patch; this acidic region is sandwiched between two positive regions to the east and northwest of the active site. These regions are called exosites. The eastern positive patch at the base of the active site cleft, called exosite-1, plays a role in the interaction with fibrinogen [24–28], fibrin, hirudin [29–31], thrombomodulin [32–35], and thrombin receptor [34, 36–39].
Figure 2.
A model of human thrombin (1 ppb) is represented by ribbon in gray, and molecular surface is represented in dark gray. The catalytic triad composed by His-57, Asp-102, and Ser-195 is shown in the middle of the figure. Heparin-binding site and fibrinogen recognition site are shown at the left and right sides of thrombin, respectively (this image was done by YASARA, reference proteins 47,393-402).
The northwest patch (the heparin-binding site), called exosite-2, is strongly positive. The heparin binding accelerates complex formation of thrombin with the serine protease inhibitors, antithrombin, and heparin cofactor II [40]. Prothrombin fragment F2 also binds to this site [41], which prevents the thrombin inhibition during early coagulation stages.
Thrombin acts in the blood coagulation and fibrinolysis systems, so it has pro- and anticoagulant functions. These activities are regulated by the protease inhibitors: α2-macroglobulin and serpins. Serpins are serine protease inhibitors like antithrombin, heparin cofactor II, and proteinase nexin-I. Thrombin inhibition by serpins is enhanced by the presence of heparin [2].
4. Thrombin Inhibitors
Maintaining a balance between bleeding and clotting has always been a challenge in treating coagulation disorders [42]. The control of blood coagulation occurs not only by the presence of enzymes in zymogen forms, but also by the presence of specific plasmatic inhibitors [43]. Simultaneously, blood coagulation, fibrinolytic system, and anticoagulant proteins are activated to dissolve the clot and to limit blood coagulation activation, respectively.
Arterial and venous thrombotic occlusive diseases are the main causes for the evolution of myocardial infarction and brain vascular accidents; inclusively, venous thrombosis affects one in 100 individuals each year [44]. Anticoagulation therapy is used in many medical specialties as well as the treatment of both venous and arterial thromboembolic events. Heparins and vitamin K antagonists have long been the anticoagulants of choice [45]. Although these agents are efficacious, their use is associated with a number of drawbacks and here is a need for new anticoagulants that can overcome their limitations [45].
Thrombin inhibitors are considered excellent candidates for therapeutic applications [46–48]. Direct inhibitors of coagulation proteins targeting a single enzyme in the coagulation cascade have been developed in recent years [45]. Direct thrombin inhibitors are a new class of anticoagulants that bind directly to thrombin and block its interaction with its substrates. Direct thrombin inhibitors do not bind to plasma protein resulting in a more predictable anticoagulant response than is seen with heparin [42]. Besides endogenous thrombin inhibitors, such as antithrombin and α2Macroglobulin, many studies have been done to find an efficient and specific thrombin inhibitor.
The inhibition of thrombin activity has been useful in a number of clinical conditions. Thrombin inhibitors are predominantly used as antithrombotic therapy (anticlotting) and have been particularly important in acute myocardial infarction. Inhibitors are also used for the prophylaxis and treatment of deep venous thrombosis and pulmonary embolism and the prevention of arterial reocclusion following endarterectomies and endovascular stent deployment [23].
4.1. Anticoagulant Molecules Secreted by Blood-Sucking Animals
In nature, there are many animals adapted to a diet of fresh blood, and they had to evolve mechanisms to control their host coagulation processes. In general, a stress due to the bite will lead to a host inflammatory response with a large number of leucocytes in the injured site. Leeches challenge overtake to block peripheral noniception effect during the bite to reduce local inflammation, but they also have to synthesize anticoagulants to maintain the blood in a fluid state during intake and subsequent digestion. Thus, leeches found a way to avoid the leucocytes migration because, during the long period of time necessary for blood meal digestion, leucocytes would release protein blood-degrading enzymes [49, 50]. Concerning this issue, a variety of coagulation inhibitors have been isolated from blood-sucking animals such as bats [51], ticks [52, 53], leeches [54, 55], and hookworms [56]. Among these inhibitors, hirudin, from a leech species, was the first thrombin inhibitor isolated and studied.
Hirudin was the first thrombin inhibitor used in clinical agent development. Hirudin was originally isolated from the salivary glands of the medical leech Hirudo medicinalis [57]. It is a polypeptide composed by 65 amino acids, which tightly and specifically binds to ∝-thrombin, in a 1 : 1 stoichiometry with Ki about 20 fM. It interacts with thrombin catalytic site and exosite-1, preventing fibrinogen cleavage and consequently clot formation. Hirudin also inhibits thrombin agonist action upon the platelet aggregation and the activation of factors V, VIII, and XIII [58–60]. Other hirudin variants have been isolated from different species of leeches. These variants differ from hirudin in both length and amino-acid composition, even though they show the same high inhibitory potency [61–63]. Hirudin itself is not commercially available; however, its discovery led to the development by recombinant technology of derivatives, namely, lepirudin and desirudin. Hirudin and its synthetics peptides have been tested in clinical trials [64–67]. Lepirudin is available in the United States, whereas desirudin is available in Europe [68, 69].
The leech Hirudinaria manillensis produces two thrombin inhibitors: hirullin P6 [70] and hirullin P18 [70–73]. They showed 62% and 52% sequence homologies to hirudin, respectively. In spite of the differences in C-terminal segment, the number and distribution of acidic-amino acid residues are similar. The presence of an Arg residue in position 2 of hirullin P6 should be pointed out for its possible interaction with the active site of thrombin, which may account for the higher inhibitory potency of hirullin P6 when comparing to hirudin from Hirudo medicinalis P18 [48].
Another tight-binding thrombin inhibitor is haemadin, isolated from the leech Haemadipsia sylvestris, which also contains an Arg residue at position 2 of the sequence. It does not inhibit other proteases and does not reveal any homology to known serine protease inhibitors, including hirudin [62].
Theromin is the most potent thrombin inhibitor, which was isolated from the gut of the leech Theromyson tessulatum [63]. It is a homodimer of 67 amino-acid residues, with 16 Cys residues engaged in eight disulfide bridges. Compared with hirudin, both peptides are anionic and rich in Cys residues. Theromin does not have sequence homology with any other protease inhibitor yet sequenced throughout the animal kingdom [63].
There are also thrombin inhibitors present in blood-sucking arthropods, like triabin, a new potent thrombin inhibitor isolated from the saliva of the blood-sucking insect Triatoma pallidipenis [74]. It is a 142-residue protein, which specifically binds to thrombin forming a 1 : 1 noncovalent complex. Triabin has been shown to be a highly potent exosite thrombin inhibitor; it inhibits thrombin-induced platelet aggregation and prolongs both thrombin clotting time (TT) and activated partial thromboplastin time (APTT); meanwhile it only minimally reduces the amidolytic activity of this enzyme toward small chromogenic substrates. In addition, triabin is able to completely block trypsin-catalyzed cleavage of thrombin and to inhibit the thrombomodulin-mediated activation of protein C in a dose-dependent fashion. Triabin contacts the thrombin molecule exclusively at exosite-1. A total of 19 and 13 residues from triabin and thrombin, respectively, are in direct contact [74].
Another thrombin inhibitor isolated from a blood sucking insect is rhodniin, a highly and potent thrombin inhibitor isolated from the assassin bug Rhodinius prolixus [75]. It binds to thrombin with a peculiar interaction mechanism, presenting multiple interactions between them, forming a 1 : 1 complex. Rhodniin is a small protein of 103 amino acids, structurally organized into two Kazal-type domains, linked via an acidic extended peptide fragment [75]. In 1999, another inhibitor similar to rhodniin was described, dipetalogastin from the insect Dipetalogaster maximus [76]. Dipetalogastin acts as a slow, tight-binding inhibitor of thrombin, and its activity to thrombin is in the pM range. The cDNA of dipetalogastin codes for a large protein which consists of six Kazal-type domains. There are three tandem, homologous regions each including two domains [77]. Recently, another inhibitor similar to rhodniin was described: infestin from the kissing bug Triatoma infestans midgut, one of the most important Chagas disease vectors in Brazil [78]. Infestin 1-2 is a double Kazal-type domain; it strongly inhibits thrombin with Ki in pM range. Infestin full-length DNA revealed a protein containing seven Kazal-type domains which is pos-translational processed creating double or single domains with thrombin, factor XIIa or neutrophil elastase inhibitory activities [78–80]. Recently, it was confirmed that those intestinal Kazal-type domains, such as rhodniin, dipetalogastin, and infestin 1-2 may be important to maintain the blood fluid during acquisition and storage by gene silence using RNA interference of brasiliensin, a thrombin inhibitor from Triatoma brasiliensis [81].
A potent and specific inhibitor of the human coagulation thrombin activity was purified from salivary gland extracts of the tsetse fly, Glossina morsitans morsitans, and an important vector of African trypanosomiasis. It is a low molecular weight peptide (MW = 3,530 Da). Amino terminal sequencing of the peptide reveals no homology to any previously identified serine protease inhibitor or natural occurring anticoagulant. In addition, it is also a potent inhibitor of thrombin-induced platelet aggregation [82, 83].
Ticks are as important as insects as vectors of disease-causing agents to humans and livestock [84].
The coagulation inhibitors and platelet aggregation inhibitors (PAIs) present in the saliva of hard and soft ticks have been extensively reviewed [85–88]. Thrombin inhibitors isolated from salivary glands of Ornithodoros moubata and Ornithodoros savignyi, respectively, named ornithodorin and savignin, have been described [53, 89–91]. Both proteins are similar. They contain two Kunitz-type domains and they need both domains to inhibit thrombin in a similar manner as rhodniin. These inhibitors bind thrombin to both the active site and to exosite-1 [92]. Amblin is the first thrombin inhibitor isolated from ixodid tick haemolymph of Amblyomma hebraeum. It is composed of 151 amino acids and contains 14 cysteines and two Kunitz-like domains [93]. Another interesting thrombin inhibitor described is boophilin, from the ixodid tick, Rhipicephalus (Boophilus) microplus, which has 12 cysteines distributed in two Kunitz-type domains that interact with thrombin by different manner when compared to that of hirudin or rhodniin [94].
The crystal structure analysis highlights that many natural inhibitors from blood-sucking animals known to date bind thrombin to both the active site and to exosite-1. The latter interaction is as important as the binding to the active site, since it enhances not only the affinity of the inhibitor for thrombin, but also the selectivity, thus preventing the proteolytic cleavage of the inhibitors by other proteases [48].
4.2. Plasma Thrombin Inhibitors
Antithrombin, a serine protease inhibitor (serpin), is an important anticoagulant molecule in mammalian circulatory system [98]. The human antithrombin is a glycoprotein composed by one chain of 58 kDa, with plasmatic concentration at 29 mg/dL. Preferably, it inhibits free enzymes; meanwhile the complex tenase and prothrombinase enzymes are less accessible to it. It is the most effective, well-regulated inhibitor of coagulation proteases, such as thrombin; factors IXa, Xa, and XIa; plasmin; and plasmatic kallikrein; moreover, antithrombin is believed to play a major role in controlling blood coagulation, thereby preventing widespread thrombosis [99]. The glycosaminoglycan heparin is a positive effector, which can enhance antithrombin activity 1000-fold [100–102]. Antithrombin and heparin thus form an interdependent regulatory mechanism of the coagulation process [103]. Inhibition by antithrombin involves the formation of a stable 1 : 1 complex between the active domain of the serine protease and the reactive site of antithrombin; the protease initially recognizes the inhibitor as a substrate. During the cleavage of the antithrombin reactive site bond, a conformational change occurs in the inhibitor that traps the protease, making it an irreversible inhibitor [100]. The antithrombin physiology role is to limit the blood coagulation process to the vascular local injury and to protect the circulation from released enzymes.
Our group has isolated antithrombin from Bothrops jararaca snake plasma by affinity chromatography using HiTrap Heparin HP column and its molecular weight, amino-terminal sequence, carbohydrate content, and isoelectric point; inhibition of bovine thrombin and immunological properties were studied and compared with previously described antithrombins [95]. Bothrops jararaca antithrombin is a single-chain glycoprotein with 18% total carbohydrate content. The molecular weight of Bothrops jararaca antithrombin was 61 kDa by SDS-PAGE, and the inhibitor exhibited an acidic isoelectric point (4.5). The amino-terminal sequence determined was His-Glu-Ser-Ser-Val-Gln-Asp-Ile-Ile-Thr, which is highly homologous to the N-terminal sequences of other animal antithrombins, indicating high amino-acid homology among several animals. Immunological cross-reactivity was observed among fish, frog, chicken, human, nonvenomous snake, and Bothrops jararaca antithrombins. Bothrops jararaca antithrombin showed inhibitory activity upon human and Bothrops jararaca coagulation and human thrombin amidolytic activity using synthetic substrate S-2238 [95].
Nahas et al. (1973) have described an inhibitor in snake plasma “Bothrops jararaca”, different from antithrombin, which also interferes in thrombin coagulant activity, measured by thrombin time; however, it does not block the activation of blood coagulation extrinsic pathway, permitting the occlusion of injured vessel. This inhibitor shows similarities with heparin, like protamin susceptibility, but differs from this in being thermolabile and not being adsorbed with BaSO4 and Al(OH)3. Our group isolated this protein, the Bothrops jararaca inhibitor (BjI), a glycoprotein showing two polypeptide chains with molecular masses of 109/138 and 150/219 kDa on SDS-PAGE in reducing and nonreducing conditions, respectively. However, the BjI presents a molecular mass about 1000 kDa in native conditions, suggesting an oligomeric form containing around five 200 kDa polypeptide chains in the native BjI. Thus, the intermonomer interactions of the BjI native form should be very weak, which could explain the lower protein molecular mass in SDS-PAGE [96].
BjI prolongs the blood coagulation intrinsic pathway; by contrast, it does not affect the extrinsic pathway [104].
BjI is an anticoagulant inhibitor that affects specifically thrombin. As it does not inhibit the thrombin amidolytic activity, it does not interact with the catalytic site of thrombin [105]. This feature differentiates BjI from antithrombin and other thrombin inhibitors previously described such as ornithodorin [82] and hirudin [57]. The BjI inhibitory activity is not increased by heparin; on the other hand, it increases the thrombin amidolytic activity, which suggests a structural conformation change when BjI binds to thrombin, facilitating the access of small chromogenic substrates to the enzyme active site [96]. Regarding thrombin exosite-1 large groove geometry, it is conceivable that two different ligands may be docked simultaneously [31]. The fact that BjI inhibits coagulant thrombin activity but not platelet aggregation activity indicates that this inhibitor does not bind to exosite-1 or it binds in a subsite other than glycoprotein Ib.
A second thrombin high positive charge density surface region designated by “anion exosite-2” is involved in heparin binding [2]. Many of the basic residues involved in thrombin interaction with heparin also act in prothrombin activation fragment 2 binding [41]. Occupancy of exosite-2 by fragment 2 or chondroitin sulfate has been suggested to alter thrombin conformation resulting in the altered calcium dependence of protein C activation [106]. This is consistent with the observation that interaction with exosite-2 may induce conformational changes in the catalytic center of thrombin [107]. Therefore, exosite-2 might also act in cooperation with exosite-1 for thrombin binding to fibrinogen [25] and platelets [25]. BjI seems to interact with exosite-2 close to the heparin-binding site, which results in increased thrombin amidolytic activity to small synthetic substrates, and can interfere in the thrombin heparin binding [96].
Our group has identified proteins similar to BjI in the plasma of three different species of viperid snakes Bothrops alternatus, Bothrops jararacussu, and Crotalus durissus terrificus. These proteins exhibited molecular masses of 109 and/or 138 kDa and were immunologically related to BjI [108]. They also inhibited the coagulant activity of thrombin, evaluated by the thrombin time test. These findings demonstrated the presence of proteins similar to BjI in these three species, although such inhibitor could not be observed in all the samples of the specimens tested. Moreover, the presence of these proteins in the plasma is related to the prolongation of thrombin time, implying a relationship between these proteins and their inhibitory coagulant activity upon thrombin. Our results suggest that BjI-like proteins are widely distributed among Crotalinae snakes found in Brazil [108].
These characteristics of BjI make it an interesting protein not only for studies on the blood coagulation evolutionary view but also for its possible use as a tool in drug design to generate specific antithrombotic.
The inhibitors cited in the text were presented in Table 1.
Table 1.
Thrombin inhibitors from different animals.
| Name | Origin | Target | Size | Types of structural domains | Binding data (Ki values) | References |
|---|---|---|---|---|---|---|
| Hirudin | Hirudo medicinalis | Thrombin catalytic site and exosite-1 | 7,000 Da | — | 20 fM | [56–58, 60] |
| Hirullin P6 | Hirudinaria manillensis | — | 7,000 Da | — | — | [70] |
| Hirullin P18 | Hirudinaria manillensis | Thrombin exosite-1 | 7,000 Da | — | 7.8 pM | [70, 71] |
| Haemadin | Haemadipsia sylvestris | Thrombin catalytic site and exosite-2 | 5,000 Da | — | 100 fM | [61] |
| Theromin | Theromyson tessulatum | — | 14,941 Da | — | 12 fM | [62] |
| Triabin | Triatoma pallidipenis | Thrombin exosite-1 | 18,000 Da | — | 3 pM | [63] |
| Rhodniin | Rhodinius prolixus | Thrombin catalytic site | 11,000 Da | Kazal-type | 20 pM | [74] |
| Dipetalogastin | Dipetalogaster maximus | — | 11,800 Da | Kazal-type | 125 fM | [75, 76]] |
| Infestin | Triatoma infestans | — | 13,000 Da | Kazal-type | 43.5 pM | [77–79] |
| Brasiliensin | Triatoma brasiliensis | — | — | Kazal-type | — | [80] |
| — | Glossina morsitans morsitans | — | 3,530 Da | — | 584 fM | [81, 82] |
| Ornithodorin | Ornithodoros moubata | Thrombin catalytic site | — | Kunitz-like | 1 pM | [88] |
| Savignin | Ornithodoros savignyi | Thrombin catalytic site and exosite-1 | 12,430 Da | — | 4.89 pM | [89, 90] |
| Amblin | Amblyomma hebraeum | — | 18,000 Da | Kunitz-like | 0.02 μM | [92] |
| Boophilin | Rhipicephalus (Boophilus) microplus | Thrombin catalytic site and exosite-1 | 13,900 Da | Kunitz-like | 1.8 nM | [93] |
| Antithrombin | Bothrops jararaca | Thrombin catalytic site | 61,000 Da | — | — | [95] |
| Bothrops jararaca inhibitor (BjI) | Bothrops jararaca | Thrombin exosite-2 | 1,000,000 Da | — | — | [96, 97] |
5. Conclusions
This paper is the first in this area, collecting thrombin inhibitors from different animal sources, vertebrates and invertebrates, either with or without clear physiological function such as in hematophagous invertebrates and snakes. Human health can be helped by using animals as a source of several biodrugs. Pharmaceutical industry has been looking for active molecules by using the biodiversity of the world, which includes plants and animals. A huge number of proteins and peptides have been identified and characterized enabling the rational design of small molecular weight compounds or peptomimetics [109].
Acknowledgment
This work was supported by FAPESP (10/51659-4).
References
- 1.Fenton JW, II, Fasco MJ, Stackrow AB. Human thrombins. Production, evaluation, and properties of α thrombin. Journal of Biological Chemistry. 1977;252(11):3587–3598. [PubMed] [Google Scholar]
- 2.Stubbs MT, Bode W. The clot thickens: clues provided by thrombin structure. Trends in Biochemical Sciences. 1995;20(1):23–28. doi: 10.1016/s0968-0004(00)88945-8. [DOI] [PubMed] [Google Scholar]
- 3.Guillin M-C, Bezeaud A, Bouton M-C, Jandrot-Perrus M. Thrombin specificity. Thrombosis and Haemostasis. 1995;74(1):129–133. [PubMed] [Google Scholar]
- 4.Mann KG. Biochemistry and physiology of blood coagulation. Thrombosis and Haemostasis. 1999;82(2):165–174. [PubMed] [Google Scholar]
- 5.Furie B, Furie BC. Molecular and cellular biology of blood coagulation. New England Journal of Medicine. 1992;326(12):800–806. doi: 10.1056/NEJM199203193261205. [DOI] [PubMed] [Google Scholar]
- 6.Furie B. Pathogenesis of thrombosis. Hematology American Society of Hematology Education Program. 2009:255–258. doi: 10.1182/asheducation-2009.1.255. [DOI] [PubMed] [Google Scholar]
- 7.Yang H-L, Lu F-J, Wung S-L, Chiu H-C. Humic acid induces expression of tissue factor by cultured endothelial cells: regulation by cytosolic calcium and protein kinase C. Thrombosis and Haemostasis. 1994;71(3):325–330. [PubMed] [Google Scholar]
- 8.Camera M, Giesen PLA, Fallon J, et al. Cooperation between VEGF and TNF-α is necessary for exposure of active tissue factor on the surface of human endothelial cells. Arteriosclerosis, Thrombosis, and Vascular Biology. 1999;19(3):531–537. doi: 10.1161/01.atv.19.3.531. [DOI] [PubMed] [Google Scholar]
- 9.Morrissey JH, Macik BG, Neuenschwander PF, Comp PC. Quantitation of activated factor VII levels in plasma using a tissue factor mutant selectively deficient in promoting factor VII activation. Blood. 1993;81(3):734–744. [PubMed] [Google Scholar]
- 10.Eichinger S, Mannucci PM, Tradati F, Arbini AA, Rosenberg RD, Bauer KA. Determinants of plasma factor VIIa levels in humans. Blood. 1995;86(8):3021–3025. [PubMed] [Google Scholar]
- 11.Lawson JH, Mann KG. Cooperative activation of human factor IX by the human extrinsic pathway of blood coagulation. Journal of Biological Chemistry. 1991;266(17):11317–11327. [PubMed] [Google Scholar]
- 12.Minnema MC, Ten Cate H, Hack CE. The role of factor XI in coagulation: a matter of revision. Seminars in Thrombosis and Hemostasis. 1999;25(4):419–428. doi: 10.1055/s-2007-994944. [DOI] [PubMed] [Google Scholar]
- 13.Butenas S, Van ’t Veer C, Mann KG. Evaluation of the initiation phase of blood coagulation using ultrasensitive assays for serine proteases. Journal of Biological Chemistry. 1997;272(34):21527–21533. doi: 10.1074/jbc.272.34.21527. [DOI] [PubMed] [Google Scholar]
- 14.Hugel B, Socié G, Vu T, et al. Elevated levels of circulating procoagulant microparticles in patients with paroxysmal nocturnal hemoglobinuria and aplastic anemia. Blood. 1999;93(10):3451–3456. [PubMed] [Google Scholar]
- 15.Mann KG, Krishnaswamy S, Lawson JH. Surface-dependent hemostasis. Seminars in Hematology. 1992;29(3):213–226. [PubMed] [Google Scholar]
- 16.Kalafatis M, Swords NA, Mann KG. Membrane-dependent reactions in blood coagulation: role of the vitamin K-dependent enzyme complexes. Biochimica et Biophysica Acta. 1994;1227(3):113–129. doi: 10.1016/0925-4439(94)90086-8. [DOI] [PubMed] [Google Scholar]
- 17.Gailani D, Broze GJ., Jr. Factor XI activation in a revised model of blood coagulation. Science. 1991;253(5022):909–912. doi: 10.1126/science.1652157. [DOI] [PubMed] [Google Scholar]
- 18.Mosesson MW. The roles of fibrinogen and fibrin in hemostasis and thrombosis. Seminars in Hematology. 1992;29(3):177–188. [PubMed] [Google Scholar]
- 19.Naski MC, Lorand L, Shafer JA. Characterization of the kinetic pathway for fibrin promotion of α-thrombin-catalyzed activation of plasma factor XIII. Biochemistry. 1991;30(4):934–941. doi: 10.1021/bi00218a008. [DOI] [PubMed] [Google Scholar]
- 20.Brummel KE, Butenas S, Mann KG. An integrated study of fibrinogen during blood coagulation. Journal of Biological Chemistry. 1999;274(32):22862–22870. doi: 10.1074/jbc.274.32.22862. [DOI] [PubMed] [Google Scholar]
- 21.Gentry PA. Comparative aspects of blood coagulation. Veterinary Journal. 2004;168(3):238–251. doi: 10.1016/j.tvjl.2003.09.013. [DOI] [PubMed] [Google Scholar]
- 22.Bick RL, Murano G. Physiology of hemostasis. Clinics in Laboratory Medicine. 1994;14(4):677–707. [PubMed] [Google Scholar]
- 23.Goldsack NR, Chambers RC, Dabbagh K, Laurent GJ. Molecules in focus thrombin. International Journal of Biochemistry and Cell Biology. 1998;30(6):641–646. doi: 10.1016/s1357-2725(98)00011-9. [DOI] [PubMed] [Google Scholar]
- 24.Fenton JW., II Thrombin specificity. Annals of the New York Academy of Sciences. 1981;370:468–495. doi: 10.1111/j.1749-6632.1981.tb29757.x. [DOI] [PubMed] [Google Scholar]
- 25.Fenton JW, II, Olson TA, Zabinski MP, Wilner GD. Anion-binding exosite of human α-thrombin and fibrin(ogen) recognition. Biochemistry. 1988;27(18):7106–7112. doi: 10.1021/bi00418a066. [DOI] [PubMed] [Google Scholar]
- 26.Naski MC, Fenton JW, II, Maraganore JM, Olson ST, Shafer JA. The COOH-terminal domain of hirudin. An exosite-directed competitive inhibitor of the action of α-thrombin on fibrinogen. Journal of Biological Chemistry. 1990;265(23):13484–13489. [PubMed] [Google Scholar]
- 27.Hopfner K-P, Di Cera E. Energetics of thrombin-fibrinogen interaction. Biochemistry. 1992;31(46):11567–11571. doi: 10.1021/bi00161a040. [DOI] [PubMed] [Google Scholar]
- 28.Bouton M-C, Jandrot-Perrus M, Bezeaud A, Guillin M-C. Late-fibrin(ogen) fragment E modulates human α-thrombin specificity. European Journal of Biochemistry. 1993;215(1):143–149. doi: 10.1111/j.1432-1033.1993.tb18016.x. [DOI] [PubMed] [Google Scholar]
- 29.Rydel TJ, Ravichandran KG, Tulinsky A, et al. The structure of a complex of recombinant hirudin and human α-thrombin. Science. 1990;249(4966):277–280. doi: 10.1126/science.2374926. [DOI] [PubMed] [Google Scholar]
- 30.Rydel TJ, Tulinsky A, Bode W, Huber R. Refined structure of the Hirudin-Thrombin complex. Journal of Molecular Biology. 1991;221(2):583–601. doi: 10.1016/0022-2836(91)80074-5. [DOI] [PubMed] [Google Scholar]
- 31.Bode W, Turk D, Karshikov A. The refined 1.9-Å X-ray crystal structure of D-Phe-Pro-Arg chloromethylketone-inhibited human α-thrombin: structure analysis, overall structure, electrostatic properties, detailed active-site geometry, and structure-function relationships. Protein Science. 1992;1(4):426–471. doi: 10.1002/pro.5560010402. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 32.Hofsteenge J, Stone SR. The effect of thrombomodulin on the cleavage of fibrinogen and fibrinogen fragments by thrombin. European Journal of Biochemistry. 1987;168(1):49–56. doi: 10.1111/j.1432-1033.1987.tb13385.x. [DOI] [PubMed] [Google Scholar]
- 33.Suzuki K, Nishioka J, Hayashi T. Localization of thrombomodulin-binding site within human thrombin. Journal of Biological Chemistry. 1990;265(22):13263–13267. [PubMed] [Google Scholar]
- 34.Mathews II, Padmanabhan KP, Tulinsky A, Sadler JE. Structure of a nonadecapeptide of the fifth EGF domain of thrombomodulin complexed with thrombin. Biochemistry. 1994;33(46):13547–13552. doi: 10.1021/bi00250a006. [DOI] [PubMed] [Google Scholar]
- 35.Srinivasan J, Hu S, Hrabal R, Zhu Y, Komives EA, Ni F. Thrombin-bound structure of an EGF subdomain from human thrombomodulin determined by transferred nuclear overhauser effects. Biochemistry. 1994;33(46):13553–13560. doi: 10.1021/bi00250a007. [DOI] [PubMed] [Google Scholar]
- 36.Vu T-KH, Wheaton VI, Hung DT, Charo I, Coughlin SR. Domains specifying thrombin-receptor interaction. Nature. 1991;353(6345):674–677. doi: 10.1038/353674a0. [DOI] [PubMed] [Google Scholar]
- 37.Vu T-KH, Hung DT, Wheaton VI, Coughlin SR. Molecular cloning of a functional thrombin receptor reveals a novel proteolytic mechanism of receptor activation. Cell. 1991;64(6):1057–1068. doi: 10.1016/0092-8674(91)90261-v. [DOI] [PubMed] [Google Scholar]
- 38.Ni F, Ripoll DR, Martin PD, Edwards BFP. Solution structure of a platelet receptor peptide bound to bovine α-thrombin. Biochemistry. 1992;31(46):11551–11557. doi: 10.1021/bi00161a037. [DOI] [PubMed] [Google Scholar]
- 39.Ishihara H, Connolly AJ, Zeng D, et al. Protease-activated receptor 3 is a second thrombin receptor in humans. Nature. 1997;386(6624):502–506. doi: 10.1038/386502a0. [DOI] [PubMed] [Google Scholar]
- 40.Bourin M, Lindahl U. Glycosaminoglycans and the regulation of blood coagulation. Biochemical Journal. 1993;289(2):313–330. doi: 10.1042/bj2890313. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Arni RK, Padmanabhan K, Padmanabhan KP, Wu T-P, Tulinsky A. Structures of the noncovalent complexes of human and bovine prothrombin fragment 2 with human PPACK-thrombin. Biochemistry. 1993;32(18):4727–4737. doi: 10.1021/bi00069a006. [DOI] [PubMed] [Google Scholar]
- 42.Castellone DD, Van Cott EM. Laboratory monitoring of new anticoagulants. American Journal of Hematology. 2010;85(3):185–187. doi: 10.1002/ajh.21607. [DOI] [PubMed] [Google Scholar]
- 43.Travis J, Salvesen GS. Human plasma proteinase inhibitors. Annual Review of Biochemistry. 1983;52:655–709. doi: 10.1146/annurev.bi.52.070183.003255. [DOI] [PubMed] [Google Scholar]
- 44.Dahlbäck B. Blood coagulation. Lancet. 2000;355(9215):1627–1632. doi: 10.1016/S0140-6736(00)02225-X. [DOI] [PubMed] [Google Scholar]
- 45.Laux V, Perzborn E, Heitmeier S, et al. Direct inhibitors of coagulation proteins—the end of the heparin and low-molecular-weight heparin era for anticoagulant therapy? Thrombosis and Haemostasis. 2009;102(5):892–899. doi: 10.1160/TH09-02-0134. [DOI] [PubMed] [Google Scholar]
- 46.Fenton JW, II, Ofosu FA, Moon DG, Maraganore JM. Thrombin structure and function: why thrombin is the primary target for antithrombotics. Blood Coagulation & Fibrinolysis. 1991;2(1):69–75. [PubMed] [Google Scholar]
- 47.Bode C, Nordt TK, Runge MS. Thrombolytic therapy in acute myocardial infarction—selected recent developments. Annals of Hematology. 1994;69(4):S35–S40. doi: 10.1007/BF02215957. [DOI] [PubMed] [Google Scholar]
- 48.Lombardi A, De Simone G, Galdiero S, Staiano N, Nastri F, Pavone V. From natural to synthetic multisite thrombin inhibitors. Biopolymers. 1999;51(1):19–39. doi: 10.1002/(SICI)1097-0282(1999)51:1<19::AID-BIP4>3.0.CO;2-G. [DOI] [PubMed] [Google Scholar]
- 49.Arocha-Piñango CL, Marchi R, Carvajal Z, Guerrero B. Invertebrate compounds acting on the hemostatic mechanism. Blood Coagulation and Fibrinolysis. 1999;10(2):43–68. doi: 10.1097/00001721-199903000-00001. [DOI] [PubMed] [Google Scholar]
- 50.Iwanaga S, Okada M, Isawa H, Morita A, Yuda M, Chinzei Y. Identification and characterization of novel salivary thrombin inhibitors from the ixodidae tick, Haemaphysalis longicornis. European Journal of Biochemistry. 2003;270(9):1926–1934. doi: 10.1046/j.1432-1033.2003.03560.x. [DOI] [PubMed] [Google Scholar]
- 51.Gardell SJ, Ramjit DR, Stabilito II, et al. Effective thrombolysis without marked plasminemia after bolus intravenous administration of vampire bat salivary plasminogen activator in rabbits. Circulation. 1991;84(1):244–253. doi: 10.1161/01.cir.84.1.244. [DOI] [PubMed] [Google Scholar]
- 52.Jacobs JW, Cupp EW, Sardana M, Friedman PA. Isolation and characterization of a coagulation factor Xa inhibitor from black fly salivary glands. Thrombosis and Haemostasis. 1990;64(2):235–238. [PubMed] [Google Scholar]
- 53.Waxman L, Smith DE, Arcuri KE, Vlasuk GP. Tick anticoagulant peptide (TAP) is a novel inhibitor of blood coagulation factor Xa. Science. 1990;248(4955):593–596. doi: 10.1126/science.2333510. [DOI] [PubMed] [Google Scholar]
- 54.Sawyer RT. Leech Biology and Behavior. Vol. 1. Oxford, UK: Oxford Science Publications; 1986. [Google Scholar]
- 55.Sawyer RT. Thrombolytics and anticoagulants from leeches. Nature Biotechnology. 1991;9(6):513–518. doi: 10.1038/nbt0691-513. [DOI] [PubMed] [Google Scholar]
- 56.Cappello M, Vlasuk GP, Bergum PW, Huang S, Hotez PJ. Ancylostoma caninum anticoagulant peptide: a hookworm-derived inhibitor of human coagulation factor Xa. Proceedings of the National Academy of Sciences of the United States of America. 1995;92(13):6152–6156. doi: 10.1073/pnas.92.13.6152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 57.Markwardt F. Hirudin as an inhibitor of thrombin. Methods in Enzymology. 1970;19:p. 1970. [Google Scholar]
- 58.Markwardt F, Nowak G, Sturzebecher U, Walsmann P. Studies on the pharmacokinetics of hirudin. Biomedica Biochimica Acta. 1987;46(4):237–244. [PubMed] [Google Scholar]
- 59.Markwardt F. Development of hirudin as an antithrombotic agent. Seminars in Thrombosis and Hemostasis. 1989;15(3):269–282. doi: 10.1055/s-2007-1002719. [DOI] [PubMed] [Google Scholar]
- 60.Markwardt F, Kaiser B, Nowak G. Studies on antithrombotic effects of recombinant hirudin. Thrombosis Research. 1989;54(5):377–388. doi: 10.1016/0049-3848(89)90208-9. [DOI] [PubMed] [Google Scholar]
- 61.Scharf M, Engels J, Tripier D. Primary structures of new ’iso-hirudins’. FEBS Letters. 1989;255(1):105–110. doi: 10.1016/0014-5793(89)81070-1. [DOI] [PubMed] [Google Scholar]
- 62.Strube K-H, Kroger B, Bialojan S, Otte M, Dodt J. Isolation, sequence analysis, and cloning of haemadin. An anticoagulant peptide from the Indian leech. Journal of Biological Chemistry. 1993;268(12):8590–8595. [PubMed] [Google Scholar]
- 63.Salzet M, Chopin V, Baert J-L, Matias I, Malecha J. Theromin, a novel leech thrombin inhibitor. Journal of Biological Chemistry. 2000;275(40):30774–30780. doi: 10.1074/jbc.M000787200. [DOI] [PubMed] [Google Scholar]
- 64.Fischer K-G. The role of recombinant hirudins in the management of thrombotic disorders. BioDrugs. 2004;18(4):235–268. doi: 10.2165/00063030-200418040-00003. [DOI] [PubMed] [Google Scholar]
- 65.Deitcher SR. Clinical utility of subcutaneous hirudins. American Journal of Health-System Pharmacy. 2003;60:S27–S31. doi: 10.1093/ajhp/60.suppl_5.S27. [DOI] [PubMed] [Google Scholar]
- 66.Greinacher A, Eichler P, Albrecht D, Strobel U, Pötzsch B, Eriksson BI. Antihirudin antibodies following low-dose subcutaneous treatment with desirudin for thrombosis prophylaxis after hip-replacement surgery: incidence and clinical relevance. Blood. 2003;101(7):2617–2619. doi: 10.1182/blood-2002-04-1055. [DOI] [PubMed] [Google Scholar]
- 67.Lubenow N, Greinacher A. Management of patients with heparin-induced thrombocytopenia: focus on recombinant hirudin. Journal of Thrombosis and Thrombolysis. 2000;10(supplement 1):S47–S57. doi: 10.1023/a:1027333320023. [DOI] [PubMed] [Google Scholar]
- 68.Nutescu EA, Shapiro NL, Chevalier A, Amin AN. A pharmacologic overview of current and emerging anticoagulants. Cleveland Clinic Journal of Medicine. 2005;72(supplement 1):S2–S6. doi: 10.3949/ccjm.72.suppl_1.s2. [DOI] [PubMed] [Google Scholar]
- 69.Greinacher A, Janssens U, Berg G, et al. Lepirudin (recombinant hirudin) for parenteral anticoagulation in patients with heparin-induced thrombocytopenia. Circulation. 1999;100(6):587–593. doi: 10.1161/01.cir.100.6.587. [DOI] [PubMed] [Google Scholar]
- 70.Steiner V, Knecht R, Olaf Börnsen K, et al. Primary structure and function of novel O-glycosylated hirudins from the leech Hirudinaria manillensis. Biochemistry. 1992;31(8):2294–2298. doi: 10.1021/bi00123a012. [DOI] [PubMed] [Google Scholar]
- 71.Krstenansky JL, Owen TJ, Yates MT, Mao SJT. The C-terminal binding domain of hirullin P18. Antithrombin activity and comparison to hirudin peptides. FEBS Letters. 1990;269(2):425–429. doi: 10.1016/0014-5793(90)81208-6. [DOI] [PubMed] [Google Scholar]
- 72.Qiu X, Yin M, Padmanabhan KP, Krstenansky JL, Tulinsky A. Structures of thrombin complexes with a designed and a natural exosite peptide inhibitor. Journal of Biological Chemistry. 1993;268(27):20318–20326. [PubMed] [Google Scholar]
- 73.De Filippis V, Vindigni A, Altichieri L, Fontana A. Core domain of hirudin from the leech Hirudinaria manillensis: chemical synthesis, purification, and characterization of a Trp3 analog of fragment 1–47. Biochemistry. 1995;34(29):9552–9564. doi: 10.1021/bi00029a032. [DOI] [PubMed] [Google Scholar]
- 74.Noeske-Jungblut C, Haendler B, Donner P, Alagon A, Possani L, Schleuning W-D. Triabin, a highly potent exosite inhibitor of thrombin. Journal of Biological Chemistry. 1995;270(48):28629–28634. doi: 10.1074/jbc.270.48.28629. [DOI] [PubMed] [Google Scholar]
- 75.Friedrich T, Kroger B, Bialojan S, et al. A Kazal-type inhibitor with thrombin specificity from Rhodnius prolixus. Journal of Biological Chemistry. 1993;268(22):16216–16222. [PubMed] [Google Scholar]
- 76.Mende K, Petoukhova O, Koulitchkova V, et al. Dipetalogastin, a potent thrombin inhibitor from the blood-sucking insect Dipetalogaster maximus. cDNA cloning, expression and characterization. European Journal of Biochemistry. 1999;266(2):583–590. doi: 10.1046/j.1432-1327.1999.00895.x. [DOI] [PubMed] [Google Scholar]
- 77.Mende K, Lange U, Nowak G. Three recombinant serine proteinase inhibitors expressed from the coding region of the thrombin inhibitor dipetalogastin. Insect Biochemistry and Molecular Biology. 2004;34(9):971–979. doi: 10.1016/j.ibmb.2004.06.006. [DOI] [PubMed] [Google Scholar]
- 78.Campos ITN, Amino R, Sampaio CAM, et al. Infestin, a thrombin inhibitor presents in Triatoma infestans midgut, a Chagas’ disease vector: gene cloning, expression and characterization of the inhibitor. Insect Biochemistry and Molecular Biology. 2002;32(9):991–997. doi: 10.1016/s0965-1748(02)00035-8. [DOI] [PubMed] [Google Scholar]
- 79.Campos ITN, Guimarães BG, Medrano FJ, Tanaka AS, Barbosa JARG. Crystallization, data collection and phasing of infestin 4, a factor XIIa inhibitor. Acta Crystallographica Section D. 2004;60(11):2051–2053. doi: 10.1107/S0907444904021596. [DOI] [PubMed] [Google Scholar]
- 80.Lovato DV, Nicolau de Campos IT, Amino R, Tanaka AS. The full-length cDNA ofanticoagulant protein infestin revealed anovel releasable Kazal domain, aneutrophil elastase inhibitor lacking anticoagulant activity. Biochimie. 2006;88(6):673–681. doi: 10.1016/j.biochi.2005.11.011. [DOI] [PubMed] [Google Scholar]
- 81.Araujo RN, Campos ITN, Tanaka AS, et al. Brasiliensin: a novel intestinal thrombin inhibitor from Triatoma brasiliensis (Hemiptera: Reduviidae) with an important role in blood intake. International Journal for Parasitology. 2007;37(12):1351–1358. doi: 10.1016/j.ijpara.2007.04.017. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 82.Cappello M, Bergum PW, Vlasuk GP, Furmidge BA, Pritchard DI, Aksoy S. Isolation and characterization of the tsetse thrombin inhibitor: a potent antithrombotic peptide from the saliva of Glossina morsitans morsitans. American Journal of Tropical Medicine and Hygiene. 1996;54(5):475–480. doi: 10.4269/ajtmh.1996.54.475. [DOI] [PubMed] [Google Scholar]
- 83.Cappello M, Li S, Chen X, et al. Tsetse thrombin inhibitor: bloodmeal-induced expression of an anticoagulant in salivary glands and gut tissue of Glossina morsitans morsitans. Proceedings of the National Academy of Sciences of the United States of America. 1998;95(24):14290–14295. doi: 10.1073/pnas.95.24.14290. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 84.Bior AD, Essenberg RC, Sauer JR. Comparison of differentially expressed genes in the salivary glands of male ticks, Amblyomma americanum and Dermacentor andersoni. Insect Biochemistry and Molecular Biology. 2002;32(6):645–655. doi: 10.1016/s0965-1748(01)00143-6. [DOI] [PubMed] [Google Scholar]
- 85.Law JH, Ribeiro JMC, Wells MA. Biochemical insights derived from insect diversity. Annual Review of Biochemistry. 1992;61:87–111. doi: 10.1146/annurev.bi.61.070192.000511. [DOI] [PubMed] [Google Scholar]
- 86.Sauer JR, McSwain JL, Bowman AS, Essenberg RC. Tick salivary gland physiology. Annual Review of Entomology. 1995;40:245–267. doi: 10.1146/annurev.en.40.010195.001333. [DOI] [PubMed] [Google Scholar]
- 87.Bowman AS, Coons LB, Needham GR, Sauer JR. Tick saliva: recent advances and implications for vector competence. Medical and Veterinary Entomology. 1997;11(3):277–285. doi: 10.1111/j.1365-2915.1997.tb00407.x. [DOI] [PubMed] [Google Scholar]
- 88.Nuttall PA, Paesen GC, Lawrie CH, Wang H. Vector-host interactions in disease transmission. Journal of Molecular Microbiology and Biotechnology. 2000;2(4):381–386. [PubMed] [Google Scholar]
- 89.Van De Locht A, Stubbs MT, Bode W, et al. The ornithodorin-thrombin crystal structure, a key to the TAP enigma? EMBO Journal. 1996;15(22):6011–6017. [PMC free article] [PubMed] [Google Scholar]
- 90.Joubert AM, Louw AI, Joubert F, Neitz AWH. Cloning, nucleotide sequence and expression of the gene encoding factor Xa inhibitor from the salivary glands of the tick, Ornithodoros savignyi. Experimental and Applied Acarology. 1998;22(10):603–619. doi: 10.1023/a:1006198713791. [DOI] [PubMed] [Google Scholar]
- 91.Nienaber J, Gaspar ARM, Neitz AWH. Savignin, a potent thrombin inhibitor isolated from the salivary glands of the tick Ornithodoros savignyi (Acari: Argasidae) Experimental Parasitology. 1999;93(2):82–91. doi: 10.1006/expr.1999.4448. [DOI] [PubMed] [Google Scholar]
- 92.Van de Locht A, Lamba D, Bauer M, et al. Two heads are better than one: crystal structure of the insect derived double domain Kazal inhibitor rhodniin in complex with thrombin. EMBO Journal. 1995;14(21):5149–5157. doi: 10.1002/j.1460-2075.1995.tb00199.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 93.Lai R, Takeuchi H, Jonczy J, Rees HH, Turner PC. A thrombin inhibitor from the ixodid tick, Amblyomma hebraeum. Gene. 2004;342(2):243–249. doi: 10.1016/j.gene.2004.07.012. [DOI] [PubMed] [Google Scholar]
- 94.Horn F, Coutinho dos Santos P, Termignoni C. Boophilus microplus anticoagulant protein: an antithrombin inhibitor isolated from the cattle tick saliva. Archives of Biochemistry and Biophysics. 2000;384(1):68–73. doi: 10.1006/abbi.2000.2076. [DOI] [PubMed] [Google Scholar]
- 95.de Morais KB, Vieira CO, Hirata IY, Tanaka-Azevedo AM. Bothrops jararaca antithrombin: isolation, characterization and comparison with other animal antithrombins. Comparative Biochemistry and Physiology. 2009;152(2):171–176. doi: 10.1016/j.cbpb.2008.11.002. [DOI] [PubMed] [Google Scholar]
- 96.Tanaka-Azevedo AM, Tanaka AS, Sano-Martins IS. A new blood coagulation inhibitor from the snake Bothrops jararaca plasma: isolation and characterization. Biochemical and Biophysical Research Communications. 2003;308(4):706–712. doi: 10.1016/s0006-291x(03)01464-5. [DOI] [PubMed] [Google Scholar]
- 97.Tanaka-Azevedo AM. Characterization of Bothrops jararaca coagulation inhibitor (BjI) and presence of similar protein in plasma of other animals. Toxicon. 2004;44(3):289–294. doi: 10.1016/j.toxicon.2004.06.001. [DOI] [PubMed] [Google Scholar]
- 98.Doolittle RF. The evolution of vertebrate blood coagulation: a case of yin and yang. Thrombosis and Haemostasis. 1993;70(1):24–28. [PubMed] [Google Scholar]
- 99.Roemisch J, Gray E, Hoffmann JN, Wiedermann CJ, Kalina U. Antithrombin: a new look at the actions of a serine protease inhibitor. Blood Coagulation and Fibrinolysis. 2002;13(8):657–670. doi: 10.1097/00001721-200212000-00001. [DOI] [PubMed] [Google Scholar]
- 100.Rosenberg RD, Damus PS. The purification and mechanism of action of human antithrombin heparin cofactor. Journal of Biological Chemistry. 1973;248(18):6490–6505. [PubMed] [Google Scholar]
- 101.Barrett EF, Barrett JN. Separation of 2 voltage sensitive potassium currents, and demonstration of a tetrodotoxin resistant calcium current in frog motoneurones. Journal of Physiology. 1976;255(3):737–774. doi: 10.1113/jphysiol.1976.sp011306. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 102.Quinsey NS, Greedy AL, Bottomley SP, Whisstock JC, Pike RN. Antithrombin: in control of coagulation. International Journal of Biochemistry and Cell Biology. 2004;36(3):386–389. doi: 10.1016/s1357-2725(03)00244-9. [DOI] [PubMed] [Google Scholar]
- 103.Jordan RE. Antithrombin in vertebrate species: conservation of the heparin-dependent anticoagulant mechanism. Archives of Biochemistry and Biophysics. 1983;227(2):587–595. doi: 10.1016/0003-9861(83)90488-5. [DOI] [PubMed] [Google Scholar]
- 104.Nahas L, et al. Blood coagulation inhibitor in a snake plasma (Bothrops jararaca) Thrombosis et Diathesis Haemorrhagica. 1973;30(1):106–113. [PubMed] [Google Scholar]
- 105.Zingali RB, Jandrot-Perrus M, Guillin M-C, Bon C. Bothrojaracin, a new thrombin inhibitor isolated from Bothrops jararaca venom: characterization and mechanism of thrombin inhibition. Biochemistry. 1993;32(40):10794–10802. doi: 10.1021/bi00091a034. [DOI] [PubMed] [Google Scholar]
- 106.Liu L-W, Rezaie AR, Carson CW, Esmon NL, Esmon CT. Occupancy of anion binding exosite 2 on thrombin determines Ca2+ dependence of protein C activation. Journal of Biological Chemistry. 1994;269(16):11807–11812. [PubMed] [Google Scholar]
- 107.Bock PE. Active-site-selective labeling of blood coagulation proteinases with fluorescence probes by the use of thioester peptide chloromethyl ketones. II. Properties of thrombin derivatives as reporters of prothrombin fragment 2 binding and specificity of the labeling approach for other proteinases. The Journal of Biological Chemistry. 1992;267(21):14974–14981. [PubMed] [Google Scholar]
- 108.Batista de Morais K, Fernandes Grego K, Mitico Tanaka-Azevedo A. Identification of proteins similar to Bothrops jararaca coagulation inhibitor (BjI) in the plasmas of Bothrops alternatus, Bothrops jararacussu and Crotalus durissus terrificus snakes. Comparative Biochemistry and Physiology. B. 2008;149(2):236–240. doi: 10.1016/j.cbpb.2007.09.011. [DOI] [PubMed] [Google Scholar]
- 109.Redwan E-RM. Animal-derived pharmaceutical proteins. Journal of Immunoassay and Immunochemistry. 2009;30(3):262–290. doi: 10.1080/15321810903084400. [DOI] [PubMed] [Google Scholar]