Abstract
Cannabinoid agonists have shown some promise clinically as analgesics, in particular for cancer pain, in which they have the additional benefit of decreasing nausea. However, as for most other drugs, the long-term use of cannabinoids is limited by the development of tolerance. Several molecular mechanisms have been proposed to explain drug tolerance, including receptor downregulation. The cannabinoid 1 (CB1) receptors can be downregulated in vitro through an interaction with the G-protein-coupled receptor-associated sorting protein1, GASP1, that targets CB1 receptors for degradation after their agonist-mediated endocytosis. To investigate whether GASP1-mediated postendocytic sorting of the CB1 receptor contributes to tolerance to cannabinoid drugs in vivo, we generated a mouse with a disruption of GASP1. In wild-type mice, repeated administration of the cannabinoid agonist WIN55,212-2 promoted downregulation of CB1 receptor levels and concomitant tolerance to the effects of drug on antinociception, motor incoordination, and locomotor hypoactivity. In contrast, GASP1 knockout mice did not develop tolerance to any of these effects and showed no significant receptor downregulation. Taken together, this study provides evidence that GASP1 regulates CB1 receptor downregulation in vivo, and that postendocytic receptor trafficking has a key role in the development of tolerance to WIN55,212-2.
Keywords: CB1 receptor; downregulation; GASP; tolerance; trafficking; WIN55,212-2
INTRODUCTION
The cannabinoid 1 (CB1) receptor is one of the most abundant G-protein-coupled receptors (GPCRs) in the mammalian central nervous system. Therefore, cannabinoid ligands have the potential to treat an array of disorders including acute and chronic pain, anxiety, and metabolic diseases (Pertwee, 2009). Agonists at the CB1 receptor include endogenously produced anandamide and 2-arachidonylglycerol, cannabis-derived delta-9-tetrahydrocannabinol (Δ9-THC), as well as synthetic drugs such as WIN55,212-2 and CP55,590. Despite the vast therapeutic potential, use of cannabinoid agonists in the clinic is limited because of the development of functional tolerance (De Vry et al., 2004; Fan et al., 1996; Gonzalez et al., 2005; Maldonado, 2002; Martin et al., 2004; Rubino et al., 2005; Sim-Selley and Martin, 2002; Tappe-Theodor et al., 2007), defined as a decrease in pharmacological response after prolonged drug exposure. Another drawback of medical cannabinoid use is the number of side effects resulting from the widespread distribution of cannabinoid receptors in the brain (Herkenham et al., 1991). Thus, cannabinoid agonists may elicit some undesired effects, including hypomobility, motor incoordination, and a drop in body temperature (Iversen, 2003; Marsicano and Lutz, 2006). Importantly, it is unclear which molecular mechanism(s) are responsible for the development of tolerance, and whether tolerance to both the beneficial and deleterious side effects of cannabinoids is mediated by the same mechanism.
There is a growing body of evidence that functional tolerance to cannabinoids is due to a change in receptor sensitivity (desensitization) or availability (downregulation). Both events lead to changes in signal transduction through the CB1 receptor, but by different mechanisms. By desensitization, receptors show reduced function owing to changes in drug affinity or receptor coupling to downstream effectors. In contrast, by downregulation, the number of drug-binding sites has been reduced by means of enhanced degradation of existing receptors, by reduced synthesis, or by altered targeting of newly synthesized receptors. Most cannabinoid ligands promote receptor downregulation (Fan et al., 1996; Oviedo et al., 1993; Rodriguez de Fonseca et al., 1994; Romero et al., 1999; Romero et al., 1997; Sim-Selley and Martin, 2002), whereas receptor desensitization seems to be more ligand specific (Gonzalez et al., 2005).
Receptor trafficking can modulate both receptor desensitization and receptor downregulation. After activation, most GPCR agonists promote receptor desensitization, which is then followed by rapid receptor endocytosis. Subsequently, receptors are either resensitized and recycled back to the cell surface, ready for a new ligand encounter, or they are targeted for degradation in lysosomes. Thus, the postendocytic fate of receptors has a pivotal role in determining the effect of endocytosis on signal transduction. For receptors that are recycled, endocytosis facilitates resensitization (turning receptors back ‘on'), whereas for receptors that are targeted for degradation, endocytosis assists in signal termination (turning receptors ‘off'). As the ability to switch signal transduction either ‘on' or ‘off' has far-reaching physiological consequences, receptor trafficking is tightly regulated. Several proteins have been implicated in modulating postendocytic sorting of GPCRs. GPCR-associated sorting proteins (GASPs) have been shown to bind to the C-terminal tail of a number of GPCRs (Heydorn et al., 2004; Simonin et al., 2004) However, the functional relevance of this interaction for postendocytic lysosomal sorting has only been established for a fraction of these receptors, including the δ-opioid receptor (Whistler et al., 2002), dopamine D2 receptor (Bartlett et al., 2005), mutants of the β2 adrenergic receptor (Thompson et al., 2007), bradykinin B1 receptor (Enquist et al., 2007), and the cannabinoid CB1 receptor (Martini et al., 2007). The GASP-family may have several members (Simonin et al., 2004; Winter and Ponting, 2005), but to date, only a defined role of GASP1 has been established.
We and others have previously shown that the CB1 receptor is targeted for lysosomal degradation by GASP1 after prolonged exposure to WIN55,212-2 in heterologous expression systems (Martini et al., 2007; Tappe-Theodor et al., 2007) and that virally expressed dominant-negative GASP1 can effect cannabinoid-mediated antinociception in vivo (Tappe-Theodor et al., 2007). On the basis of these observations, our prediction was that, tolerance to the cannabinoid WIN55,212-2 would be promoted, at least in part, by GASP1-mediated CB1 receptor downregulation in vivo, and that mice deficient in the sorting protein GASP1 would not develop the same degree of tolerance. To examine this hypothesis, we generated a GASP1 knockout (KO) mouse line and investigated the role of CB1 receptor trafficking in the development of tolerance to the effects of WIN55,212-2 after repeated drug administration.
MATERIALS AND METHODS
Drugs and Chemicals
WIN55,212-2 ((R)-(+)-[2,3-dihydro-5-methyl-3-(4-morpholinylmethyl)pyrrolo[1,2,3-de]-1,4-benzoxazin-6-yl]-1-naphthalenylmethanone mesylate) and AM251 (N-(piperidin-1-yl)-5-(4-iodophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide) were purchased from Tocris Cookson (Ellisville, MO). [3H]SR141716a (N-(piperidin-1-yl)-5-(4-chlorophenyl)-1-(2,4-dichlorophenyl)-4-methyl-1H-pyrazole-3-carboxamide) was obtained from Amersham Biosciences (GE Healthcare Biosciences, Piscataway, NJ). Bovine serum albumin (BSA), MgCl2, Tris-HCl, Cremophor EL, Sigmacote, and EDTA was purchased from Sigma-Aldrich (St. Louis, MO). Isotonic saline (0.9% sodium chloride) was obtained from Hospira (Lake Forest, IL), ethanol was purchased from Goldshield Chemical (Hayward, CA), and sucrose was obtained from Fisher Scientific (Pittsburgh, PA). WIN55,212-2 was dissolved in a 1 : 1 : 18 solution of ethanol/Cremophor/saline in glass vials pretreated with Sigmacote.
Animals
Mice were housed in a climate-controlled room under a 12 : 12 h light/dark cycle with ad libitum access to food and water. All procedures were conducted in accordance with the guidelines provided by the Institutional Animal Care and Use Committee (IACUC) at the Ernest Gallo Clinic and Research Center, CA. Only male mice were used for this study, as the female hormonal cycle may affect the responsiveness to cannabinoids (Kalbasi Anaraki et al., 2008). Mice were acclimated to the observation rooms at least 1 hr before behavioral evaluation. All injections were administered either subcutanously (s.c.) or intraperitoneally (i.p.) in a volume of 10 ml/kg.
Generation of GASP1 KO Mice
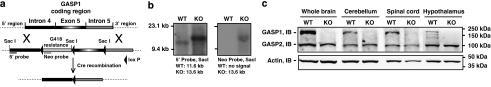
A targeting vector containing a neomycin (G418)-resistance gene flanked by loxP-Cre recombination sites was inserted into intron 4, which is upstream of the GASP1 open reading frame (ORF) on the mouse X chromosome (Figure 1a). A third loxP site was inserted in the intron downstream of the GASP1 ORF. A total of 30 μg of NotI-linearized KO vector DNA was electroporated into ∼107 C57BL/6 ES cells and selected with 200 μg/ml G418. Primary ES screening was performed by Southern blotting for the presence of hybridization band corresponding to the targeted allele, and absence of wild-type (WT) hybridization band (Figure 1b, left) and a single neo integration (Figure 1b, right). ES clones containing homologous recombination were then transfected with Cre-recombinase and Type I recombination events, in which both G418 and the GASP ORF were removed, were identified, and confirmed on expansion by PCR analysis. Blastocyst injection (using B6/Tyr blastocysts) was performed and the resulting male chimeras were bred with C57BL/6 Taconic WT females to generate heterozygous mice.
Figure 1.
Generation of GASP1 KO mice. (a) Targeting vector design for generating GASP1 KO mice. A cassette expressing the G418 resistance gene flanked by loxP sites was inserted into the intron upstream of the GASP1 open reading frame (ORF) (intron 4) and a third loxP site was inserted in the intron downstream of the GASP ORF (intron 5). ES cells from C57/Bl6 mice were transfected with this vector. Properly targeted clones (see b) were transfected with Cre-recombinase, and blastocysts from clones in which the GASP1 ORF was disrupted were implanted into C57/BL6 females. (b) Southern blotting analysis identified homologous recombination and single insertion using both (left) 5′ and (right) Neo probes. (c) Homogenates from wild-type (WT) and GASP1 knockout (KO) whole brain, cerebellum, spinal cord, and hypothalamus were analyzed by immunoblot (IB) and shows complete knockout of GASP1. Furthermore, there are no compensatory changes in the expression of the closest homolog, GASP2, in either of these regions.
Behavioral Tests
Mice were treated according to the paradigm described in Table 1. Briefly, on days 1 and 7, mice were injected with accumulative doses of WIN55,212-2 (1–6 mg/kg s.c. for antinociception and body temperature tests; 3.5–14 mg/kg i.p. for motor coordination test) and subjected to the appropriate behavioral assay. In between, on days 2–6, mice received either vehicle or WIN55,212-2 (3.5 mg/kg s.c.) twice daily (0900–1100 hours and 1600–1800 hours). Locomotor activity was assessed on day 2 and day 6, after a single injection of either vehicle or drug. Mice in the control group received vehicle only, whereas mice in the test group were injected with WIN55,212-2 only. At 24 h after the final injection, mice were killed to isolate tissue for binding analysis.
Table 1. Drug Treatment and Behavioral Testing Paradigm.
| Day | 1 | 2 | 3 | 4 | 5 | 6 | 7 |
|---|---|---|---|---|---|---|---|
| Injection | WIN dose response | WIN/Veh | WIN/Veh | WIN/Veh | WIN/Veh | WIN/Veh | WIN dose response |
| Behavioral assay | Tail flick latency | Locomotor activity | Locomotor activity | Tail flick latency | |||
| Body temperature | Body temperature | ||||||
| Motor coordination | Motor coordination |
Behavioral tests for antinociception, hypothermia, and locomotor activity were conducted essentially as described previously (Wallace et al., 2009). Briefly, antinociception was assessed by tail flick withdrawal response latencies by placing the mice on a tail flick analgesia meter (Columbus Instruments, Columbus, OH). On test days, the time between heat stimulus and rapid tail removal was recorded, and a cutoff of 10 s was employed to avoid tissue damage. First, baseline latencies were determined and subsequently at 20 min after each drug administration. Antinociception was calculated as the percent of maximal possible effect: (%MPE=((test latency−baseline latency)/(10−baseline latency)) × 100). Body temperature was measured immediately after assessment of antinociception by inserting a thermometer probe (Type J, Barnant, Barrington, IL) 2 cm into the rectum. Temperature changes were calculated by subtracting test temperature from the basal temperature. Locomotor activity was monitored using individual, open-field photocell chambers (46 cm × 46 cm, Med Associates, St Albans, VT) protected from light and sound. At 20 min postinjection, mice were placed in the chambers, and the distance traveled was recorded for 10 min counting the number of broken photobeams using Activity Monitor software (Med Associates). For motor coordination, on two consecutive days before the test day, all mice had been trained to remain for 120 s on a 3 cm diameter rotarod treadmill (Ugo Basile, Accelerating Rotarod ‘Jones & Roberts' for mice 7650) set to a steady speed of 6 rpm. On each training day, the mice were given five attempts to stay on the rotarod. On test day, the mice were placed on the rotarod set at an accelerating speed (increasing from 4 to 22.8 rpm during a 290 s period). Only the active time that the mice spent on the revolving rotarod was recorded with a cutoff at 470 s to avoid overexertion of the mice. The mice were then injected with accumulative doses of WIN55,212-2 and subjected to rotarod testing every 15 min. Cannabinoid agonists have a primarily sedative effect, but reports have described how tactile or auditory stimuli may induce hyperreflexia—also known as the ‘popcorn' effect—with increased jumping frequency (Dewey, 1986). Mice that developed such response to WIN55,212-2 were excluded from the rotarod study.
Immunoblot Analysis
Mice were killed by cervical dislocation. Whole brain, cerebellum, spinal cord, and hypothalamus were dissected, homogenized in buffer A (0.32 M sucrose, 10 m Tris-HCl; pH 7.4, with added complete protease inhibitors (Roche, Basel, Switzerland)) on ice and centrifuged at 1000 × g (whole brain, cerebellum, and spinal cord) or 10 000 × g (hypothalamus) for 10 min at 4°C. Supernatants were analyzed for protein contents by the Bradford assay (Biorad, Hercules, CA), denatured in reducing sample buffer, resolved on 4–20% Tris-glycine precast gels (Invitrogen, Carlsbad, CA), and electroblotted onto PVDF membrane (Millipore, Tamecula, CA). The blot was cut at 75 kDa, and the upper part was incubated with rabbit anti-GASP1 antibody (1 : 1000; Whistler et al., 2002), whereas the lower part was incubated with mouse anti-actin antibody (1 : 10 000; Sigma-Aldrich). After washing extensively, the blots were incubated with appropriate fluorescent secondary antibodies (1 : 5000), Alexa Fluor 680 goat anti-mouse IgG (Molecular Probes, Invitrogen) for the lower blot and IRDye800-conjugated anti-rabbit IgG (Rockland, PA) for the upper blot, washed again, dried, and visualized using the Odyssey Infrared Imaging system (LI-COR Biosciences, NE). Actin has a molecular weight of 43 kDa. GASP1 is a 1395 amino acid protein and has an apparent mass of 190 kDa (Whistler et al., 2002). GASP2 is a 838 amino acid protein and runs with an apparent molecular weight of 100 kDa when resolved by SDS-polyacrylamide gel electrophoresis.
Histology and Immunocytochemistry
Four GASP1 KO mice and four WT littermates were deeply anesthetized with an overdose of Euthasol (Virbac, Forth Worth, TX) and perfused with 0.9% NaCl, followed by 4% paraformaldehyde in phosphate buffer (PB, pH 7.4). Brains were cryopreserved in 30% sucrose and sectioned on a cryostat (CM3050, Leica Instruments, Nussloch, Germany) using coronal or sagittal orientations. Serial 50 μm-thick sections were mounted on gelatin-treated slides and counterstained with Cresyl Violet to compare the overall anatomy between the genotypes. For immunostaining, free-floating sections were first permeabilized with 50% ethanol for 20 min, rinsed in PBS, and then blocked with 10% normal donkey serum in PBS for 30 min. Sections were then incubated for 48 h at 4°C on an orbital shaker with a combination of primary antibodies. The choice of primary antibodies was determined by goals of the experiments. For the colocalization study in the preoptic anterior hypothalamus (POAH), the antibodies used were monoclonal mouse anti-NeuN antibody (neuronal nuclei marker, 1 : 1000; Millipore), polyclonal goat anti-CB1 receptor antibody (1 : 1000; a kind gift from Ken Mackie, University of Indiana), and polyclonal rabbit anti-GASP antibody (1 : 500; Bartlett et al., 2005; Martini et al., 2007). For the colocalization study of GABAergic inhibitory neurons, sections were incubated with a combination of monoclonal mouse anti-parvalbumin (1 : 500; Sigma-Aldrich), polyclonal goat anti-CB1 receptor, and rabbit anti-GASP antibodies (as described above). To check for any abnormal gliosis in the transgenic animals, we used monoclonal mouse anti-GFAP (1 : 1000; Sigma-Aldrich). After treating with primary antibodies, sections were rinsed in PBS, blocked in 2% normal donkey serum for 10 min, and incubated for 3 h with a combination of the secondary antibodies (all raised in donkey), namely, Alexa Fluor 488-labeled anti-rabbit, Alexa Fluor 594-labeled anti-goat (1 : 300; Invitrogen), and Cy5-labeled anti-mouse antibodies (1 : 250; Jackson ImmunoResearch, West Grove, PA). Sections were finally rinsed in PBS, mounted on gelatin-coated slides, air dried, and coverslipped using Vectashield mounting medium. All images were acquired using Zeiss LSM 510 META laser confocal microscope (Zeiss, Thornwood, NY) using optimal factory recommended filter configurations.
Membrane Preparation for Saturation Binding Analysis
For membrane preparation, the methods were essentially as described previously (Griffin et al., 1998). After cervical dislocation, spinal cord, and cerebella from at least three mice undergoing the same treatment paradigms were dissected and pooled into separate vials. Tissues were homogenized in buffer A using a Teflon/glass douncer and centrifuged at 1000 × g for 10 min at 4°C. The supernatants were then centrifuged at 40 000 × g for 20 min at 4°C; the resulting pellets were washed in homogenization buffer and centrifuged as described previously. The final P2 pellets were then resuspended in buffer (50 m Tris-HCl, 3 m MgCl2, 1 m EDTA; pH 7.4). Protein concentrations were determined by the Bradford method; aliquots were snap frozen and stored at −80°C until use.
Saturation Binding
Membrane suspensions were prepared by diluting 90 μg spinal cord or 25 μg cerebellum membrane protein in 450 μl assay buffer (50 m Tris-HCl, 3 m MgCl2, 1 m EDTA, 0.5% BSA; pH 7.4). Binding was initiated by adding membrane suspensions to Sigmacote-treated glass vials containing either 1 μ AM251 for non-specific binding (NSB) or DMSO for total binding (TB) and preincubated at 4°C for 10 min. Next, 50 μl of increasing concentrations of [3H]SR141716A (78 pmol to 5 n) was added and the vials were incubated at 30°C for 60 min. The reaction was terminated by the addition of ice-cold wash buffer (50 m Tris-HCl and 0.1% BSA; pH 7.4) followed by rapid filtration under vacuum through the Whatman GF/C glass fiber filters using a 12-well sampling manifold (Millipore). The tubes were rinsed three times over the filters with 5 ml of ice-cold wash buffer. Finally, the filters were placed into plastic scintillation vials, 5 ml Scintiverse II SX12-4 scintillation cocktail (Fisher Scientific) was added, and bound radioactivity was determined the next day by liquid scintillation counting. TB was performed in triplicate, whereas NSB was in duplicate determinations, and specific binding was determined by subtracting NSB from TB. Bmax values were calculated as an average of at least three binding assays performed on independent membrane preparations.
Data Analysis
All statistical analyses were performed by using Prism 4.03 (GraphPad Software, San Diego, CA). Data from tail flick withdrawal latency, motor coordination, spontaneous locomotor activity, and body temperature tests were analyzed by two-way analysis of variance (ANOVA) with Bonferroni multiple comparison posttests. Emax and EC50 values were calculated from non-linear regression analysis on sigmoidal dose–response curves (variable slope). From saturation-binding experiments, Bmax values were calculated by fitting the data to a non-linear regression model with one site binding (hyperbola). Subsequent comparisons of Bmax values between different drug treatments were based on one-way ANOVA with Bonferroni multiple comparison posttests. Significance was defined as p<0.05.
RESULTS
Acute Effects of WIN55,212-2 on Antinociception, Motor Incoordination, Hypolocomotion, and Hypothermia
To study the role of GASP1 in responsiveness to WIN55,212-2 in vivo, mice were generated with a disruption of the GASP1 gene (Figures 1a and b). GASP1 KO progeny were viable and showed no apparent difference in survival or lifespan (Boeuf et al., 2009). Microscopic examination of Nissl-counterstained sections did not reveal any gross abnormalities in the brain morphology of GASP1 KO animals, and immunostaining with neuronal (NeuN, parvalbumin) and glial (GFAP) markers also did not reveal abnormalities in the corresponding cell types of KO animals (data not shown).
Western blot analysis confirmed expression of GASP1 in whole brain, cerebellum, spinal cord, and hypothalamus homogenates in WT mice and confirmed KO of the GASP1 protein in the KO mice with no apparent change in levels of GASP2, which is the closest homolog to GASP1 (Simonin et al., 2004) (Figure 1c).
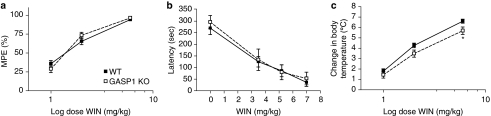
Cannabimimetic agents mediate behavioral responses in antinociception, motor coordination, spontaneous locomotor activity, and body temperature (Little et al., 1988; Patel and Hillard, 2001). WT and GASP1 KO mice, showed no genotype-specific differences in basal performance in any of these tests (Table 2). In addition, when challenged acutely with escalating doses of WIN55,212-2, there were no genotype-specific differences in drug potency or efficacy for antinociception, as assessed by latency of tail flick withdrawal (Figure 2a), or for motor incoordination, as assessed by latency to fall off an accelerating rotarod (Figure 2b). To assess the effects of WIN55,212-2 on spontaneous locomotor activity, mice were placed in an open-field chamber. As repeated exposure to the chamber on the same day reduces the novelty of the environment and, consequently, spontaneous exploration/locomotion (data not shown), only a single dose of WIN55,212-2 was tested (3.5 mg/kg). There were no genotype differences to the acute hypolocomotor effects of WIN55,212-2 (50–65% reduction in both genotypes, data not shown). Finally, WIN55,212-2 was slightly less potent at decreasing body temperature in GASP1 KO mice compared with WT mice, but significance was only reached at the highest dose tested (Figure 2c). Together, these data suggest that there are no compensatory changes in the GASP1 KO mice that affect any of these behaviors.
Table 2. Behavioral Response in Drug-Naïve Mice.
| WT | n | GASP1 KO | n | |
|---|---|---|---|---|
| Tail flick withdrawal latency (sec) | 4.5±0.9 | 56 | 4.6±1.1 | 58 |
| Spontaneous locomotor activity (cm) | 1191±237 | 37 | 1204±335 | 33 |
| Rotarod latency (sec) | 258±104 | 23 | 282±105 | 19 |
| Body temperature (°C) | 37.1±0.36 | 36 | 36.9±0.66 | 25 |
Figure 2.
Acute effect of WIN55,212-2 in antinociception, motor coordination, and body temperature. Wild-type (WT, ▪) and GASP1 knockout (KO, □) mice were injected with increasing doses of WIN55,212-2 (WIN) and analyzed for drug-induced behavioral changes. Data are presented as mean±SEM. (a) Antinociception was assessed by tail flick withdrawal latency after injection with WIN55,212-2 (1–6 mg/kg s.c.; n=38–52). There were no genotype differences in response to drug-induced antinociception. MPE, maximal possible effect. (b) Motor coordination was evaluated by placing mice on an accelerating rotarod after injection with WIN55,212-2 (3.5–7 mg/kg i.p.; n=9–13) and measuring latency to fall off the rod. There were no genotype differences in response to drug-induced motor incoordination. (c) Body temperature was measured after injection with WIN55,212-2 (1–6 mg/kg s.c.; n=25–37). There were no genotype differences in response to drug-induced hypothermia except for the highest dose of WIN55,212-2 tested (6 mg/kg, *p<0.05).
Tolerance to the Antinociceptive, Motor Incoordinating, Hypolocomotive, and Hypothermic Effects of WIN55,212-2
We next compared GASP1 KO and WT mice for the development of behavioral tolerance after undergoing repetitive administration of WIN55,212-2 (see paradigm in Table 1). Mice of both genotypes received 3.5 mg/kg WIN55,212-2, as this dose induced a significant, but not maximal, response in all behavioral tests (see Figure 2 and data not shown).
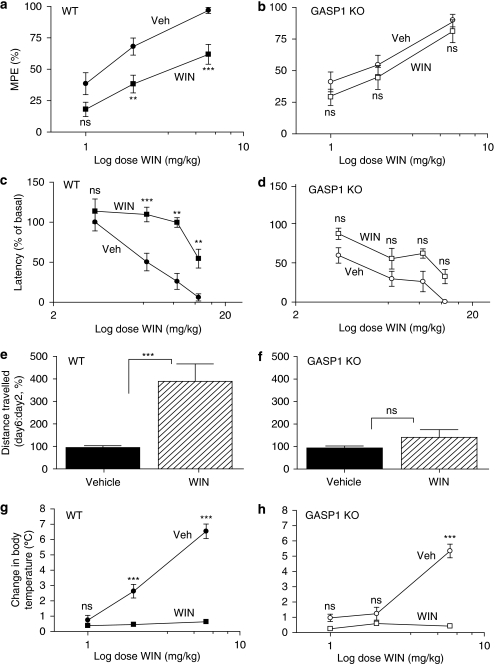
First, we assessed the antinociceptive effects of WIN55,212-2. There were no significant genotype differences in basal (drug free) tail flick withdrawal latency (Table 2) or in the acute effects of WIN55,212-2 on nociception (Figure 2a). After 5 days of treatment, mice of both genotypes showed no significant changes in the baseline (drug free) tail flick responses compared with nontreated mice (data not shown) and no significant effect of vehicle treatment (compare Figure 2a with Figure 3a-b). Chronic drug treatment promoted a high degree of antinoceptive tolerance in WT mice. This was observed as a reduction in Emax values by ∼40% (Figure 3a), but with no evident change in EC50 values (83 mg/kg in vehicle-treated and 62 mg/kg in WIN55,212-2-treated mice), consistent with a loss of receptor reserve owing to receptor downregulation (see Table 3). In contrast, GASP1 KO mice showed no significant change in Emax response to the drug (Figure 3b) and no significant loss of receptor number (see Table 3).
Figure 3.
Tolerance to the effects of WIN55,212-2 in antinociception, motor incoordination, locomotor hypoactivity, and hypothermia after repetitive administration. Wild-type (WT) and GASP1 KO mice were injected twice daily for 5 days with either vehicle (veh) or 3.5 mg/kg WIN55,212-2 (WIN) and assessed for the development of tolerance to WIN55,212-2. (•) Vehicle-treated and (▪) WIN55,212-2-treated WT mice; (○) vehicle-treated and (□) WIN55,212-2-treated GASP1 KO mice. Data are presented as mean±SEM. (a, b) On days 1 and 7, WT (a) and GASP KO (b) mice were tested for tail flick withdrawal latencies after injection with increasing doses of WIN55,212-2 (1–6 mg/kg s.c.; n=18–24 per group). MPE, maximal possible effect. GASP1 KO mice (b) treated chronically with WIN55,212-2 showed reduced antinociceptive tolerance compared with WT mice (a). ***p<0.001; **p<0.01; ns, not significant (compared with vehicle treatment). (c, d) Mice were tested for motor coordination on an accelerating rotarod after injection with increasing doses of WIN55,212-2 (3.5–14 mg/kg i.p.; n=9–12 per group). GASP1 KO mice (d) treated chronically with WIN55,212-2 showed reduced tolerance compared with WT mice (c). ***p<0.001; **p<0.01; ns: not significant (compared with vehicle treatment). (e, f) Mice chronically treated with either vehicle or drug were placed in activity chambers after injection of vehicle (solid bars) or 3.5 mg/kg WIN55,212-2 (hatched bars), respectively. The distance traveled on day 6 was compared with the distance on day 2 (n=15–16 per group). GASP1 KO mice (f) treated chronically with WIN55,212-2 showed reduced tolerance to the hypolocomotor effects of WIN55,212-2 compared with WT mice (e). ***p<0.001; ns: not significant (compared with vehicle). (g, h) On days 1 and 7, body temperature was measured after injection with increasing doses of WIN55,212-2 (1–6 mg/kg s.c.; n=8–19 per group). On day 7, both WT (g) and GASP1 KO (h) mice had developed tolerance to the hypothermic effects of WIN55,212-2 compared with vehicle-treated mice. ***p<0.001; ns: not significant (compared with vehicle treatment).
Table 3. [3H]SR141716a Saturation Binding.
|
Bmax (fmol/mg) |
Kd (nM) |
|||
|---|---|---|---|---|
| WT | GASP1 KO | WT | GASP1 KO | |
| Spinal cord | ||||
| Vehicle | 712±82 | 438±31 | 1.9±0.5 | 0.9±0.2 |
| WIN55,212-2 | 404±28 | 375±26 | 1.2±0.2 | 1.1±0.2 |
| Cerebellum | ||||
| Vehicle | 1664±144 | 1613±111 | 1.5±0.3 | 1.3±0.2 |
| WIN55,212-2 | 1379±92 | 1840±93 | 1.6±0.3 | 1.5±0.2 |
We then examined the effects of WIN55,212-2 on motor coordination. There were no significant genotype differences in the basal (drug free) performance on the rotarod (Table 2) or in the acute effects of WIN55,212-2 on motor coordination (Figure 2b). WT mice undergoing drug treatment for 5 days showed significant tolerance to the motor incoordinating effects of WIN55,212-2 compared with vehicle-treated mice (Figure 3c). In contrast, GASP1 KO mice did not show any significant development of tolerance in the same test (Figure 3d).
We next evaluated the effects of WIN55,212-2 on locomotor hypoactivity. There were no genotype-specific differences in either basal (drug free) exploration or in the acute response to WIN55,212-2 (Table 2 and data not shown). WT mice treated repeatedly with WIN55,212-2 showed significant tolerance to the locomotor inhibitory effects of the drug compared with vehicle-treated mice (Figure 3e), whereas GASP1 KO mice did not show the same degree of recovery (Figure 3f).
Finally, we studied development of tolerance to the hypothermic effects of WIN55,212-2. There were no differences in baseline body temperature between WT and GASP1 KO mice (Table 2). All drug-treated mice, regardless of genotypic background, displayed the same degree of tolerance (Figures 3g–h). This suggests that mechanisms besides—or in addition to—receptor downregulation may regulate tolerance to the hypothermic effects of cannabinoids.
In summary, these results indicate that GASP1 has a key role in the development of tolerance to most effects of WIN55,212-2, including antinociception, motor incoordination, and spontaneous locomotor inhibition, but not in hypothermia.
Colocalization of GASP and CB1 Receptors
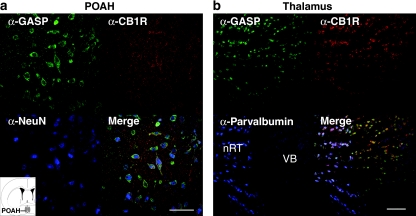
It has been reported that thermoregulation relies on neurons located at the POAH (Boulant, 1981), as destruction of this region disrupts the ability to regulate body temperature. Furthermore, infusion of WIN55,212-2 into this region leads to hypothermia, which can be blocked by SR141716a, suggesting that the effect is CB1 receptor mediated (Rawls et al., 2002). As we did not observe a difference between genotypes in the development of tolerance to the hypothermic effects of WIN55,212-2, we speculated that perhaps GASP1 was not expressed in this brain region. However, we found that GASP is expressed in POAH neurons and colocalized with the CB1 receptor (Figure 4a). Therefore, it is likely that mechanisms other than CB1 downregulation by GASP, control tolerance to the hypothermic effects of WIN55,212-2.
Figure 4.
Colocalization of GASP and CB1 receptors in the mouse central nervous system. Micrographs of coronal sections from drug-naïve WT mice (a) preoptic anterior hypothalamus (POAH, medial part) and (b) thalamus co-stained for GASP (green) and CB1 receptor (red). (a) GASP and CB1 receptor immunoreactivity is present in the majority of POAH neurons. POAH sections were also stained for NeuN (neuronal nuclei, blue). Inset: schematic showing the location of POAH (gray ovals). (b) Thalamic sections were stained for GASP (green), CB1 receptors (red), and also for parvalbumin (blue) to identify GABAergic neurons of the reticular thalamic nucleus (nRT). Both GASP and CB1 receptors were present in GABAergic neurons of nRT and also in nearby glutamatergic cells of the ventrobasal thalamus (VB). Scale bars: a, 50 μm; b,100 μm.
It has previously been reported that CB1 receptor expression in both excitatory and inhibitory neurons are important to varying degrees for the tetrad of cannabinoid-mediated behavioral effects, including locomotion, body temperature, nociception, and catalepsy (Monory et al., 2007). We therefore examined colocalization of GASP and CB1 in brain areas involved in mediating above behaviors, including the somatosensory thalamus. In the thalamus, both the excitatory and inhibitory neurons are present, and can be reliably identified by their location in the discrete areas and by co-staining with parvalbumin, a marker of inhibitory neurons (Figure 4b). We found that GASP and CB1 were coexpressed in parvalbumin-expressing GABAergic inhibitory cells of the reticular thalamic nucleus and were also colocalized in the glutamatergic cells of the ventrobasal thalamus (Figure 4b). Thus, GASP is poised to control trafficking of the CB1 receptor in different cells types.
Reduced Receptor Downregulation in GASP1 KO Mice after Repetitive WIN55,212-2 Administration
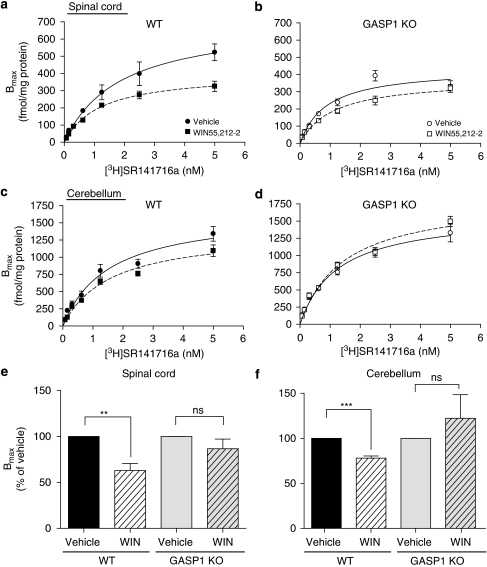
Previous research has supported a key role for GASP1 in mediating GPCR downregulation by facilitating the trafficking of receptors to lysosomal compartments after agonist-induced endocytosis. To quantify receptor levels in WT and GASP1 KO mice, we performed saturation binding experiments using the selective CB1 receptor antagonist SR141716a (Rinaldi-Carmona et al., 1996) in membrane preparations from spinal cord and cerebellum. Both regions express relatively high levels of cannabinoid receptors, and are involved in the spinal tail flick reflex (Hohmann et al., 1998) and in motor coordination (Patel and Hillard, 2001), respectively. We found that repeated administration of WIN55,212-2 promoted a significant loss of CB1 receptor-binding sites in WT mice in both spinal cord and cerebellum (Table 3 and Figure 5). In contrast, with the same treatment, GASP1 KO mice did not show any significant CB1 receptor downregulation (Table 3 and Figure 5). There were no significant changes in the binding affinity (Kd) of SR141716a in any of the treatment groups (Table 3).
Figure 5.
Downregulation of CB1 receptors in WT, but not in GASP1 KO mice, after repetitive administration of WIN55,212-2. Tissue from WT and GASP1 KO mice chronically injected with either vehicle or 3.5 mg/kg WIN55,212-2 was tested in saturation binding assays using the antagonist SR141716a to assess total number of CB1 receptors (Bmax). (a–d) WT mice treated with WIN55,212-2 showed significant downregulation of CB1 receptor number in both the spinal cord (a, e) and the cerebellum (c, f), whereas GASP1 KO mice showed reduced receptor downregulation in both tissues (b, d, e, f). (e, f) Data presented as percent Bmax±SEM in drug-treated versus vehicle-treated mice. ***p<0.001; **p<0.01; ns: not significant (compared with normalized vehicle-treated mice, n=3–4, performed on independent membrane preparations).
DISCUSSION
This study investigated the importance of the postendocytic sorting protein GASP1 in modulating tolerance to WIN55,212-2-mediated effects using mice with a disruption of GASP1. Following endocytosis, GPCRs can either be recycled to the plasma membrane or targeted to the lysosome for degradation. Numerous proteins have been identified that modulate this choice between recycling and degradation (for review see Hanyaloglu and von Zastrow, 2008). For the CB1 receptor, this choice between recycling and degradation seems to depend on the concentration and/or the duration of the action of the agonist ligand (Hsieh et al., 1999; Martini et al., 2007; Wu et al., 2008). However, studies with a virally expressed dominant-negative GASP had suggested that degradation mediated by GASP is important for regulating at least the antinociceptive responses to cannabinoids (Tappe-Theodor et al., 2007). Here, we used GASP1 KO mice to assess the role of GASP1 in the development of tolerance to several cannabinoid-mediated behaviors. We found that WT mice developed significant tolerance to WIN55,212-2-mediated antinociception, motor incoordination, locomotor inhibition, and hypothermia. Development of tolerance to these behavioral effects in GASP1 KO was reduced significantly, except for hypothermia, in which the KO mice developed tolerance similar to WT mice.
Previous studies have also shown selectivity in the development of tolerance to cannabinoids, depending on the behavioral effects studied. For example, inhibition of nitric oxide synthase (NOS) has been shown to reduce tolerance to the WIN55,212-2-mediated hypothermic effects, whereas tolerance to the antinociceptive effects was not affected (Spina et al., 1998). Thus, it is possible that different molecular mechanisms control the development of cannabinoid tolerance, possibly depending on the brain region/circuits involved in the behavioral readout. Indeed, the fact that NOS is important selectively for tolerance to the hypothermic effects of cannabinoids could explain why both WT and GASP1 KO mice develop tolerance to this effect of WIN55,212-2, but only WTs develop tolerance to the antinociceptive, incoordinative, and locomotor effects.
Importantly, GASP is regionally expressed in the rodent brain (Simonin et al., 2004) and may therefore be involved in downregulating receptors in distinct brain areas and influencing some, but not all, cannabinoid behaviors. Indeed, several studies report that the extent of receptor downregulation and cannabinoid tolerance differ spatially (Sim-Selley, 2003; Sim-Selley and Martin, 2002; Sim-Selley et al., 2006) and even temporally (Bass and Martin, 2000) between specific brain regions. Importantly, especially in light of the functional role of CB1 receptors in both excitatory and inhibitory neuronal populations, we found that GASP and CB1 receptors were colocalized in both these cell types (Figure 4). However, colocalization does not mandate that there is a functional interaction of GASP and CB1 in all places in which they are expressed together. For example, other scaffolding or accessory proteins interacting with the CB1 receptor could prevent interaction of GASP, much the way NHERF/EBP50 interferes with the ability of the β2 adrenergic receptor to interact with GASP (Thompson et al., 2007).
The precise mechanism by which GASP targets GPCRs such as CB1 receptor for degradation remains to be elucidated. The sequential activities of the members of the endosomal sorting complex required for transport (ESCRT) pathway are thought to be necessary for the recognition and sorting of ubiquitinated proteins for degradation (see Hanyaloglu and von Zastrow, 2008 for review). However, mammalian GPCRs that are degraded in the lysosome do not require ubiquitination to be degraded (see, for example, Tanowitz and von Zastrow, 2002). Hence, it is intriguing to speculate that GASP could be a part of the ESCRT machinery, whereby its function is to target non-ubiquitinated proteins for degradation. If this were the case, it is surprising that GASP has not been previously identified as a member of the various ESCRT complexes in any species. Intriguingly, although there are close GASP orthologues in numerous mammalian species, there are no apparent orthologues in any of the other genetically tractable species that were used to decipher the ESCRT sorting machinery, including Drosophila, Caenorhbditis elegans, or yeast.
Interestingly, although we found that behavioral tolerance to WIN55,212-2 in GASP1 KO mice was reduced to levels below significance, we noted that the data from drug-treated and vehicle-treated mice was not superimposable. This suggests that some degree of tolerance did occur in the GASP1 KO mice. In this regard, it is important to keep in mind that the GASP1 KO mice still express other GASP homologs, as well as other proteins involved in receptor signaling and trafficking. Indeed, regional differences in the expression of GASP (Simonin et al., 2004) or other accessory proteins could determine the extent of tolerance. Furthermore, regional differences in the density of CB1 receptors (Herkenham et al., 1991), the stoichiometry of receptors to G-proteins (Breivogel et al., 1997; Sim et al., 1996) or to other accessory proteins, could influence the rate or degree of tolerance. In addition, it has been reported that after an intraperitoneal injection of WIN55,212-2, the actual drug concentration varies throughout different regions of the CNS (Barna et al., 2009). Therefore, as drug levels seem to dictate the specific postendocytic sorting of CB1 receptors, it is possible that receptors may either recycle or degrade depending on the brain region studied and thereby, contribute to varying levels of receptor downregulation.
Surprisingly, we found that compared with naïve WT mice, naïve GASP1 KO mice showed reduced CB1 receptor levels in the spinal cord, but not in the cerebellum (Table 3). This lower receptor number did not affect the acute antinocieptive effects of WIN55,212-2, indicating that there is a substantial receptor reserve, at least in the spinal cord. Nevertheless, these findings suggest that cellular mechanisms other than GASP-mediated postendocytic receptor sorting, perhaps altered synthesis or transport of new receptors, are used to regulate CB1 receptor levels and are engaged, perhaps, to compensate for loss of GASP1 function in the GASP1 KO mice.
In summary, this study establishes a link between CB1 receptor trafficking mediated by GASP1 and the development of tolerance to many of the effects of WIN55,212-2, including antinociception, locomotor hypoactivity, and motor incoordination. However, there was no effect on hypothermia. Thus, regional differences in expression of receptors or accessory proteins may determine the specific mechanism of tolerance. By extension, selectively engaging these mechanisms could provide a means by which to either promote or decrease tolerance to specific behavioral effects of cannabinoids.
Acknowledgments
We would like to thank Ken Mackie for kindly providing the goat anti-CB1 receptor antibody. We also thank Melisa Wallace for input and suggestions, Madeline Ferwerda and Stacy Taylor for assistance with mouse breeding and genotyping and Shruti Chander for technical assistance. This study was supported by the funds provided to the state of California for medical research on alcohol and substance abuse through the University of California, San Francisco to J.L.W and to V.K. and the National Institute of Mental Health to J.L.W. L.M. is supported by the Danish Agency for Science and Technology and Innovation and The Lundbeck Foundation (Copenhagen, Denmark). D.T. is supported in part by a NARSAD young investigator award.
The authors declare no conflict of interest.
References
- Barna I, Till I, Haller J. Blood, adipose tissue and brain levels of the cannabinoid ligands WIN-55,212 and SR-141716A after their intraperitoneal injection in mice: compound-specific and area-specific distribution within the brain. Eur Neuropsychopharmacol. 2009;19:533–541. doi: 10.1016/j.euroneuro.2009.02.001. [DOI] [PubMed] [Google Scholar]
- Bartlett SE, Enquist J, Hopf FW, Lee JH, Gladher F, Kharazia V, et al. Dopamine responsiveness is regulated by targeted sorting of D2 receptors. Proc Natl Acad Sci USA. 2005;102:11521–11526. doi: 10.1073/pnas.0502418102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bass CE, Martin BR. Time course for the induction and maintenance of tolerance to Delta(9)-tetrahydrocannabinol in mice. Drug Alcohol Depend. 2000;60:113–119. doi: 10.1016/s0376-8716(99)00150-7. [DOI] [PubMed] [Google Scholar]
- Boeuf J, Trigo JM, Moreau PH, Lecourtier L, Vogel E, Cassel JC, et al. Attenuated behavioural responses to acute and chronic cocaine in GASP-1-deficient mice. Eur J Neurosci. 2009;30:860–868. doi: 10.1111/j.1460-9568.2009.06865.x. [DOI] [PubMed] [Google Scholar]
- Boulant JA. Hypothalamic mechanisms in thermoregulation. Fed Proc. 1981;40:2843–2850. [PubMed] [Google Scholar]
- Breivogel CS, Sim LJ, Childers SR. Regional differences in cannabinoid receptor/G-protein coupling in rat brain. J Pharmacol Exp Ther. 1997;282:1632–1642. [PubMed] [Google Scholar]
- De Vry J, Jentzsch KR, Kuhl E, Eckel G. Behavioral effects of cannabinoids show differential sensitivity to cannabinoid receptor blockade and tolerance development. Behav Pharmacol. 2004;15:1–12. doi: 10.1097/00008877-200402000-00001. [DOI] [PubMed] [Google Scholar]
- Dewey WL. Cannabinoid pharmacology. Pharmacol Rev. 1986;38:151–178. [PubMed] [Google Scholar]
- Enquist J, Skroder C, Whistler JL, Leeb-Lundberg LM. Kinins promote B2 receptor endocytosis and delay constitutive B1 receptor endocytosis. Mol Pharmacol. 2007;71:494–507. doi: 10.1124/mol.106.030858. [DOI] [PubMed] [Google Scholar]
- Fan F, Tao Q, Abood M, Martin BR. Cannabinoid receptor down-regulation without alteration of the inhibitory effect of CP 55 940 on adenylyl cyclase in the cerebellum of CP 55 940-tolerant mice. Brain Res. 1996;706:13–20. doi: 10.1016/0006-8993(95)01113-7. [DOI] [PubMed] [Google Scholar]
- Gonzalez S, Cebeira M, Fernandez-Ruiz J. Cannabinoid tolerance and dependence: a review of studies in laboratory animals. Pharmacol Biochem Behav. 2005;81:300–318. doi: 10.1016/j.pbb.2005.01.028. [DOI] [PubMed] [Google Scholar]
- Griffin G, Atkinson PJ, Showalter VM, Martin BR, Abood ME. Evaluation of cannabinoid receptor agonists and antagonists using the guanosine-5′-O-(3-[35S]thio)-triphosphate binding assay in rat cerebellar membranes. J Pharmacol Exp Ther. 1998;285:553–560. [PubMed] [Google Scholar]
- Hanyaloglu AC, von Zastrow M. Regulation of GPCRs by endocytic membrane trafficking and its potential implications. Annu Rev Pharmacol Toxicol. 2008;48:537–568. doi: 10.1146/annurev.pharmtox.48.113006.094830. [DOI] [PubMed] [Google Scholar]
- Herkenham M, Lynn AB, Johnson MR, Melvin LS, de Costa BR, Rice KC. Characterization and localization of cannabinoid receptors in rat brain: a quantitative in vitro autoradiographic study. J Neurosci. 1991;11:563–583. doi: 10.1523/JNEUROSCI.11-02-00563.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Heydorn A, Sondergaard BP, Ersboll B, Holst B, Nielsen FC, Haft CR, et al. A library of 7TM receptor C-terminal tails. Interactions with the proposed post-endocytic sorting proteins ERM-binding phosphoprotein 50 (EBP50), N-ethylmaleimide-sensitive factor (NSF), sorting nexin 1 (SNX1), and G protein-coupled receptor-associated sorting protein (GASP) J Biol Chem. 2004;279:54291–54303. doi: 10.1074/jbc.M406169200. [DOI] [PubMed] [Google Scholar]
- Hohmann AG, Tsou K, Walker JM. Cannabinoid modulation of wide dynamic range neurons in the lumbar dorsal horn of the rat by spinally administered WIN55,212-2. Neurosci Lett. 1998;257:119–122. doi: 10.1016/s0304-3940(98)00802-7. [DOI] [PubMed] [Google Scholar]
- Hsieh C, Brown S, Derleth C, Mackie K. Internalization and recycling of the CB1 cannabinoid receptor. J Neurochem. 1999;73:493–501. doi: 10.1046/j.1471-4159.1999.0730493.x. [DOI] [PubMed] [Google Scholar]
- Iversen L. Cannabis and the brain. Brain. 2003;126 (Pt 6:1252–1270. doi: 10.1093/brain/awg143. [DOI] [PubMed] [Google Scholar]
- Kalbasi Anaraki D, Sianati S, Sadeghi M, Ghasemi M, Javadi P, Ejtemaei Mehr S, et al. Modulation by female sex hormones of the cannabinoid-induced catalepsy and analgesia in ovariectomized mice. Eur J Pharmacol. 2008;586:189–196. doi: 10.1016/j.ejphar.2008.02.055. [DOI] [PubMed] [Google Scholar]
- Little PJ, Compton DR, Johnson MR, Melvin LS, Martin BR. Pharmacology and stereoselectivity of structurally novel cannabinoids in mice. J Pharmacol Exp Ther. 1988;247:1046–1051. [PubMed] [Google Scholar]
- Maldonado R. Study of cannabinoid dependence in animals. Pharmacol Ther. 2002;95:153–164. doi: 10.1016/s0163-7258(02)00254-1. [DOI] [PubMed] [Google Scholar]
- Marsicano G, Lutz B. Neuromodulatory functions of the endocannabinoid system. J Endocrinol Invest. 2006;29 (3 Suppl:27–46. [PubMed] [Google Scholar]
- Martin BR, Sim-Selley LJ, Selley DE. Signaling pathways involved in the development of cannabinoid tolerance. Trends Pharmacol Sci. 2004;25:325–330. doi: 10.1016/j.tips.2004.04.005. [DOI] [PubMed] [Google Scholar]
- Martini L, Waldhoer M, Pusch M, Kharazia V, Fong J, Lee JH, et al. Ligand-induced down-regulation of the cannabinoid 1 receptor is mediated by the G-protein-coupled receptor-associated sorting protein GASP1. FASEB J. 2007;21:802–811. doi: 10.1096/fj.06-7132com. [DOI] [PubMed] [Google Scholar]
- Monory K, Blaudzun H, Massa F, Kaiser N, Lemberger T, Schutz G, et al. Genetic dissection of behavioural and autonomic effects of Delta(9)-tetrahydrocannabinol in mice. PLoS Biol. 2007;5:e269. doi: 10.1371/journal.pbio.0050269. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Oviedo A, Glowa J, Herkenham M. Chronic cannabinoid administration alters cannabinoid receptor binding in rat brain: a quantitative autoradiographic study. Brain Res. 1993;616:293–302. doi: 10.1016/0006-8993(93)90220-h. [DOI] [PubMed] [Google Scholar]
- Patel S, Hillard CJ. Cannabinoid CB(1) receptor agonists produce cerebellar dysfunction in mice. J Pharmacol Exp Ther. 2001;297:629–637. [PubMed] [Google Scholar]
- Pertwee RG. Emerging strategies for exploiting cannabinoid receptor agonists as medicines. Br J Pharmacol. 2009;156:397–411. doi: 10.1111/j.1476-5381.2008.00048.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rawls SM, Cabassa J, Geller EB, Adler MW. CB1 receptors in the preoptic anterior hypothalamus regulate WIN 55212-2 [(4,5-dihydro-2-methyl-4(4-morpholinylmethyl)-1-(1-naphthalenyl-carbonyl)-6H-pyrrolo[3,2,1ij]quinolin-6-one]-induced hypothermia. J Pharmacol Exp Ther. 2002;301:963–968. doi: 10.1124/jpet.301.3.963. [DOI] [PubMed] [Google Scholar]
- Rinaldi-Carmona M, Pialot F, Congy C, Redon E, Barth F, Bachy A, et al. Characterization and distribution of binding sites for [3H]-SR 141716A, a selective brain (CB1) cannabinoid receptor antagonist, in rodent brain. Life Sci. 1996;58:1239–1247. doi: 10.1016/0024-3205(96)00085-9. [DOI] [PubMed] [Google Scholar]
- Rodriguez de Fonseca F, Gorriti MA, Fernandez-Ruiz JJ, Palomo T, Ramos JA. Downregulation of rat brain cannabinoid binding sites after chronic delta 9-tetrahydrocannabinol treatment. Pharmacol Biochem Behav. 1994;47:33–40. doi: 10.1016/0091-3057(94)90108-2. [DOI] [PubMed] [Google Scholar]
- Romero J, Berrendero F, Garcia-Gil L, Lin SY, Makriyannis A, Ramos JA, et al. Cannabinoid receptor and WIN-55,212-2-stimulated [35S]GTPgammaS binding and cannabinoid receptor mRNA levels in several brain structures of adult male rats chronically exposed to R-methanandamide. Neurochem Int. 1999;34:473–482. doi: 10.1016/s0197-0186(99)00020-0. [DOI] [PubMed] [Google Scholar]
- Romero J, Garcia-Palomero E, Castro JG, Garcia-Gil L, Ramos JA, Fernandez-Ruiz JJ. Effects of chronic exposure to delta9-tetrahydrocannabinol on cannabinoid receptor binding and mRNA levels in several rat brain regions. Brain Res Mol Brain Res. 1997;46:100–108. doi: 10.1016/s0169-328x(96)00277-x. [DOI] [PubMed] [Google Scholar]
- Rubino T, Forlani G, Vigano D, Zippel R, Parolaro D. Ras/ERK signalling in cannabinoid tolerance: from behaviour to cellular aspects. J Neurochem. 2005;93:984–991. doi: 10.1111/j.1471-4159.2005.03101.x. [DOI] [PubMed] [Google Scholar]
- Sim-Selley LJ. Regulation of cannabinoid CB1 receptors in the central nervous system by chronic cannabinoids. Crit Rev Neurobiol. 2003;15:91–119. doi: 10.1615/critrevneurobiol.v15.i2.10. [DOI] [PubMed] [Google Scholar]
- Sim-Selley LJ, Martin BR. Effect of chronic administration of R-(+)-[2,3-Dihydro-5-methyl-3-[(morpholinyl)methyl]pyrrolo[1,2,3-de]-1,4-b enzoxazinyl]-(1-naphthalenyl)methanone mesylate (WIN55,212-2) or delta(9)-tetrahydrocannabinol on cannabinoid receptor adaptation in mice. J Pharmacol Exp Ther. 2002;303:36–44. doi: 10.1124/jpet.102.035618. [DOI] [PubMed] [Google Scholar]
- Sim-Selley LJ, Schechter NS, Rorrer WK, Dalton GD, Hernandez J, Martin BR, et al. Prolonged recovery rate of CB1 receptor adaptation after cessation of long-term cannabinoid administration. Mol Pharmacol. 2006;70:986–996. doi: 10.1124/mol.105.019612. [DOI] [PubMed] [Google Scholar]
- Sim LJ, Hampson RE, Deadwyler SA, Childers SR. Effects of chronic treatment with delta9-tetrahydrocannabinol on cannabinoid-stimulated [35S]GTPgammaS autoradiography in rat brain. J Neurosci. 1996;16:8057–8066. doi: 10.1523/JNEUROSCI.16-24-08057.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Simonin F, Karcher P, Boeuf JJ, Matifas A, Kieffer BL. Identification of a novel family of G protein-coupled receptor associated sorting proteins. J Neurochem. 2004;89:766–775. doi: 10.1111/j.1471-4159.2004.02411.x. [DOI] [PubMed] [Google Scholar]
- Spina E, Trovati A, Parolaro D, Giagnoni G. A role of nitric oxide in WIN 55,212-2 tolerance in mice. Eur J Pharmacol. 1998;343:157–163. doi: 10.1016/s0014-2999(97)01543-4. [DOI] [PubMed] [Google Scholar]
- Tanowitz M, Von Zastrow M. Ubiquitination-independent trafficking of G protein-coupled receptors to lysosomes. J Biol Chem. 2002;277:50219–50222. doi: 10.1074/jbc.C200536200. [DOI] [PubMed] [Google Scholar]
- Tappe-Theodor A, Agarwal N, Katona I, Rubino T, Martini L, Swiercz J, et al. A molecular basis of analgesic tolerance to cannabinoids. J Neurosci. 2007;27:4165–4177. doi: 10.1523/JNEUROSCI.5648-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thompson D, Pusch M, Whistler JL. Changes in G protein-coupled receptor sorting protein affinity regulate postendocytic targeting of G protein-coupled receptors. J Biol Chem. 2007;282:29178–29185. doi: 10.1074/jbc.M704014200. [DOI] [PubMed] [Google Scholar]
- Wallace MJ, Newton PM, McMahon T, Connolly J, Huibers A, Whistler J, et al. PKCepsilon regulates behavioral sensitivity, binding and tolerance to the CB1 receptor agonist WIN55,212-2. Neuropsychopharmacology. 2009;34:1733–1742. doi: 10.1038/npp.2008.230. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Whistler JL, Enquist J, Marley A, Fong J, Gladher F, Tsuruda P, et al. Modulation of postendocytic sorting of G protein-coupled receptors. Science. 2002;297:615–620. doi: 10.1126/science.1073308. [DOI] [PubMed] [Google Scholar]
- Winter EE, Ponting CP. Mammalian BEX, WEX and GASP genes: coding and non-coding chimaerism sustained by gene conversion events. BMC Evol Biol. 2005;5:54. doi: 10.1186/1471-2148-5-54. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu DF, Yang LQ, Goschke A, Stumm R, Brandenburg LO, Liang YJ, et al. Role of receptor internalization in the agonist-induced desensitization of cannabinoid type 1 receptors. J Neurochem. 2008;104:1132–1143. doi: 10.1111/j.1471-4159.2007.05063.x. [DOI] [PubMed] [Google Scholar]