Abstract
Transecting the corpus callosum of postnatal day (P)1–6 rats disinhibits the production of spindle bursts (SBs) within primary somatosensory cortex (S1), most notably during periods of sleep-related myoclonic twitching. Here we investigated developmental changes in this callosally mediated disinhibition and its association with cortical plasticity. Recordings in P2–15 subjects revealed that callosotomy-induced disinhibition is a transient feature of early development that disappears abruptly after P6. This abrupt switch was accompanied by sharp decreases in myoclonic twitching and equally sharp increases in spontaneous SBs and in the number of GABAergic and glutamatergic presynaptic terminals in S1. Expression of the K+Cl− co-transporter 2 (KCC2) also increased across these ages. To determine whether these abrupt developmental changes are associated with alterations in cortical plasticity, pups were callosotomized at P1, P6, or P8, and tested over the subsequent week. Regardless of age, callosotomy immediately disrupted SBs evoked by forepaw stimulation. Over the next week, the P1 and P6 callosotomy groups exhibited full recovery of function; in contrast, the P8 group did not exhibit recovery of function, thus indicating an abrupt decrease in cortical plasticity between P6 and P8. Together, our data demonstrate that callosotomy-induced disinhibition is a transient phenomenon whose disappearance coincides with the onset of increased intrinsic connectivity, establishment of excitatory-inhibitory balance, and diminished plasticity in S1. Accordingly, our findings indicate that callosotomy-induced disinhibition of twitch-related SBs is a bioassay of somatosensory cortical plasticity and, in addition, support the hypothesis that myoclonic twitches, like retinal waves, actively contribute to cortical development and plasticity.
Keywords: plasticity, myoclonic twitching, somatosensory cortex, REM sleep, glutamate, GABA, KCC2
Activity-dependent processes guide the development of interhemispheric connectivity by the corpus callosum during the prenatal and early postnatal period in rats (Mizuno, Hirano, & Tagawa, 2007; Wang et al., 2007). In rodents, callosal fibers cross the midline soon after birth, after which they make contact with the cortical subplate and invade the contralateral cortex during the first postnatal week (Innocenti & Price, 2005; Ivy & Killackey, 1981; Wise & Jones, 1976). We recently found that transecting the corpus callosum (CCx) of postnatal day (P)1–6 rats disinhibits spontaneous spindle bursts (SBs) in the forelimb region of primary somatosensory cortex (S1) (Marcano-Reik & Blumberg, 2008); CCx also disrupted evoked S1 responses to contralateral forepaw stimulation. These findings suggested that the corpus callosum – soon after birth and at least through P6 – exerts a net inhibitory influence over homotopic cortical circuits and plays a modulatory role in sensory processing.
SBs are brief 5–25 Hz oscillatory bursts and are among the most prevalent early cortical activity patterns (Khazipov et al., 2004). SBs occur in S1 during waking but most prominently during periods of sleep-related myoclonic twitching, and they can be specifically evoked by prioprioceptive stimulation (Marcano-Reik & Blumberg, 2008). In addition to S1, SBs are produced in visual (Hanganu, Staiger, Ben-Ari, & Khazipov, 2007) and barrel (Minlebaev, Ben-Ari, & Khazipov, 2007) cortex, but they have not been detected in non-sensory cortical regions (Seelke & Blumberg, 2010).
Models of cortical injury and recovery typically involve direct damage to specific areas within cortical lobes (Kolb, Halliwell, & Gibb, 2010). Such studies have revealed that the long-term consequences of cortical damage depend in part on the stage of brain development at the time of injury. For example, in rats, functional recovery is best when cortical injury is produced during the second postnatal week in relation to the first postnatal week, that is, after neuronal migration to superficial layers is complete around P6. However, if SBs contribute to the development of somatosensory representations in S1, as has been suggested (Khazipov et al., 2004), then the dramatic CCx-induced disinhibition of SB activity that we observed before P6 might indicate a state of heightened plasticity.
Here we investigated developmental changes in CCx-induced disinhibition and its association with somatosensory cortical plasticity. We document the sudden disappearance of CCx-induced disinhibition, accompanied by behavioral, cellular, and molecular changes that are associated with a reduction of one form of cortical plasticity after P6. We propose that CCx in early development offers a new and unique model system for examining the effects of early perturbations of cortical circuitry on somatosensory processing and recovery of function. All together, these findings support the hypothesis that myoclonic twitches serve as a primary source of sensory input driving activity-dependent processes in somatosensory cortex during early postnatal development, similar to the role played by retinal waves in the development of visual cortex (Katz & Shatz, 1996; Wong, 1999).
MATERIALS AND METHODS
All experiments were performed in accordance with National Institutes of Health guidelines for the care and use of animals in research and were approved by the Institution Animal Care and Use Committee of the University of Iowa. All efforts were made to minimize the number of animals used.
For all experiments, mothers and their litters were housed in standard laboratory cages (48 × 20 × 26 cm) in the animal colony at the University of Iowa. Food and water were available ad libitum and all animals were maintained on a 12-hr light-dark cycle with lights on at 0700 h. All experiments took place during the lights-on period. Litters were culled to 8 pups within 3 days of birth (day of birth = P0).
Developmental changes in cortical activity in sham and callosotomized pups
Subjects
A total of 64 pups from 32 litters were used. Subjects were tested on P2, P4, P6, P7, P8, P9, P12, or P15 (n = 4 per age). Males and females were equally represented among the subjects.
Surgery
On the day of testing, 2 littermates underwent CCx or sham surgery (with counterbalancing of the order of surgery), as described previously (Marcano-Reik & Blumberg, 2008). Briefly, under isoflurane anesthesia, a 2–3 mm opening was created in the skull, halfway between bregma and lambda and parallel and lateral to midline. In CCx subjects, a thin surgical knife was inserted to a depth of approximately 5 mm and was swept in an anterior-to-posterior direction to transect the corpus callosum. Sham surgeries were identical except that the surgical knife was not inserted. In all subjects, 2 recording sites were prepared for placement of cortical surface electrodes over the left and right S1s. Each site consisted of 2 holes separated by approximately 2 mm and centered over the forelimb region of S1, approximately 1 mm rostral to bregma and 2–3 mm lateral to midline.
As described previously (Karlsson, Gall, Mohns, Seelke, & Blumberg, 2005; Marcano-Reik & Blumberg, 2008), a custom-built stainless steel apparatus, designed to attach to the earbar holders of a stereotaxic apparatus (David Kopf Instruments, Tujunga, CA, United States), was secured to the skull with cyanoacrylate adhesive. Bipolar stainless steel electrodes (50 μm diameter, California Fine Wire, Grover Beach, CA) were inserted into the left and right nuchal muscles. A ground wire was implanted anterior to the nuchal EMGs. Electrodes were secured with flexible collodion. At the end of surgery, the pup’s trunk was lightly wrapped in gauze while the limbs remained exposed. These surgeries lasted approximately 10 min. The pups were then placed in a humidified incubator maintained at thermoneutrality (35°C) to recover for 1 h. At the end of the recovery period, pups were transferred to an electrically shielded chamber for testing.
Procedure
As reported previously (Marcano-Reik & Blumberg, 2008), the pup’s head was fixed in the stereotaxic apparatus and its ventrum was placed on a flat support bar with its forelimbs and hindlimbs dangling freely on both sides without contacting any surface. Temperature-controlled water flowing through a concave double-walled glass chamber, situated beneath the pup, helped to control the thermal environment and, in conjunction with a heat lamp, maintain the pup’s brain temperature at 37°C throughout testing. Under these conditions, head-fixed pups sleep even more than unrestrained pups and rarely exhibit signs of distress (i.e., excessive movement or audible vocalizations). Moreover, head-fixed pups cycle regularly between sleep and wakefulness and exhibit all the basic features of sleep-wake organization, including temporally coherent twitching in multiple muscle groups against a background of muscle atonia (e.g., see Mohns & Blumberg, 2010).
Custom-made Ag/AgCl electrodes were inserted just below the cortical surface into the pre-drilled recording sites. Ground electrodes were placed in the cerebellum. Electroencephalographic (EEG) and electromyographic (EMG) electrodes were connected to differential amplifiers (A-M Systems, Carlsborg, WA; filter setting: 0.1–3000 Hz; amplification: x10,000). Neural and EMG signals were sampled at 12.5 kHz using a digital interface and Spike2 software (Cambridge Electronic Design, Cambridge, UK).
Once sleep-wake cyclicity was observed, the plantar surface of the left and right forepaws was stimulated with a fine brush, typically eliciting dorsiflexion at the wrist joint. Electrode placements within S1 were judged successful when plantar surface stimulation of the contralateral forepaw resulted in an SB. Other parts of the limb and body were also stimulated to assure the specificity of the response and recording location.
For all subjects, after at least 1 h of acclimation in the stereotaxic apparatus, the recording session began with a 15-min period of behavioral scoring (twitching and wake-related behaviors), and was followed by a 15-min period of plantar surface forepaw stimulation. EMG and EEG data were acquired continuously throughout the test. In all subjects, visual observation and EMG activity in the left and right nuchal muscles provided measures of behavioral state and myoclonic twitching (Karlsson et al., 2005; Marcano-Reik & Blumberg, 2008; Seelke & Blumberg, 2005). As described elsewhere (Gramsbergen, Schwartze, & Prechtl, 1970; Karlsson et al., 2005), myoclonic twitches of the limbs and tail, indicative of active sleep, were defined as phasic, rapid, and independent movements. High-amplitude movements, indicative of wakefulness, included locomotion, stretching, and yawning. In addition, during periods of sleep-wake observation and forepaw stimulation, the experimenter marked the events in synchrony with the electrophysiologic records by pressing a key on the keyboard during data acquisition.
Histology
Upon completion of testing, all CCx pups were overdosed with an intraperitoneal injection of sodium pentobarbital and perfused transcardially with phosphate-buffered saline, followed by a 3% formalin solution. Brains were postfixed for at least 48 h in a formalin–sucrose solution before being sliced in the coronal plane (50 μm sections). Light microscopy was then used to assess the extent of damage to the corpus callosum. Similar to what was reported previously using the same method (Marcano-Reik & Blumberg, 2008), approximately 90% of the corpus callosum was typically destroyed, beginning at the anterior portion and extending posteriorly.
Data Analysis
For each subject, 15-min periods of synchronized data comprising digital records of behavior, nuchal EMG activity, and EEG activity were created and analyzed using Spike2 software, as described previously (Marcano-Reik & Blumberg, 2008). For all analyses of sham and CCx littermate subjects, paired (within-subjects) t tests were performed using JMP 5.0 software (SAS, Cary, North Carolina, USA). For all tests, alpha was set at 0.05 and Bonferroni corrections were used to correct alpha for multiple comparisons.
Myoclonic twitches were counted by referring to both the behavioral record (i.e., limb and tail twitching) and the left and right nuchal EMG activity. Thresholds were set in Spike2 to analyze nuchal EMG activity (Mohns & Blumberg, 2008). Cursors were used to identify individual twitches in the EMG records and relate them to behaviorally scored twitches. A memory buffer was created with all the marked twitches, and an analysis was run to count all the events during the 15-min recording period.
Spontaneous and evoked SBs were identified by referring to both the raw EEG records of the left and right S1s as well as filtered records (band-pass: 5–40 Hz). Using criteria similar to those described previously (Khazipov et al., 2004; Marcano-Reik & Blumberg, 2008), SBs were defined as comprising at least 3 complete oscillations, approximately 100 ms in duration, and containing at least 1 oscillation that exceeded 50 μV in amplitude (from baseline to peak). In contrast with spontaneous SBs, evoked SBs were embedded within large, slow potentials with amplitudes ≥ 100 μV. Finally, SB oscillation frequency and duration were measured in sham and CCx subjects as described previously; consistent with our previous study (Marcano-Reik & Blumberg, 2008), SB oscillation frequency, but not duration, increased after CCx.
SB latencies were measured and compared for all subjects, as described previously (Marcano-Reik & Blumberg, 2008). For this analysis, 20 ‘anchor’ SBs in the left S1 recording were selected at random and its duration measured. Then, the latencies between the anchor SB and the prior (L−) and subsequent (L+) SBs in the right S1 recording were determined (see Figure 1, middle panel). Mean values of L− and L+ were always statistically indistinguishable.
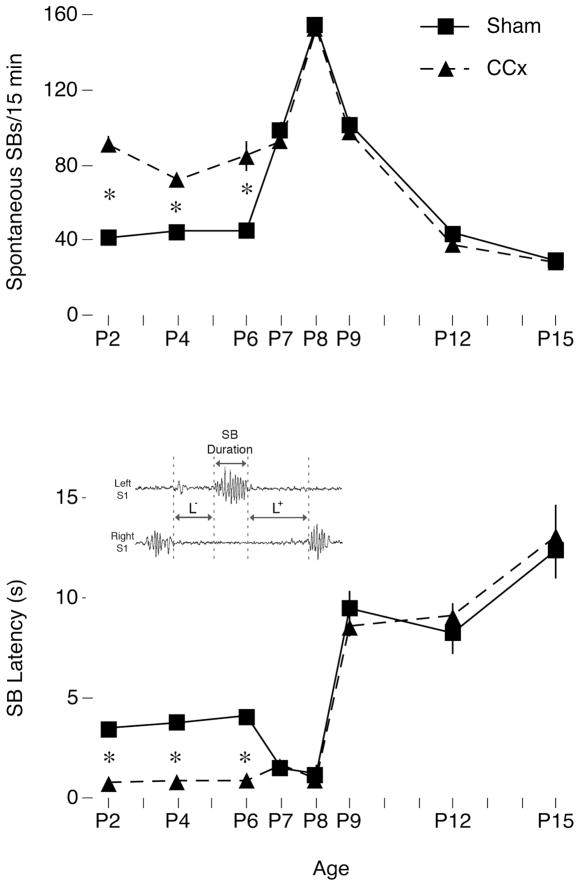
Figure 1.
Changes in SB activity in sham and callosotomized (CCx) P2–15 rats. Top Panel: The rate of spontaneous SBs doubles after CCx at or before P6 but not thereafter. Middle Panel: CCx-induced disinhibition is accompanied by decreased interhemispheric SB latencies. Inset: Illustration of the method used to score SB latencies; only mean L− values are presented in the figure. * significant difference between groups.
Effect of GABAergic agonists and antagonists on cortical activity
Subjects and surgical preparation
A total of 32 P4 rats from 8 litters were used. On the day of testing, 2 littermates experienced sham surgery and 2 additional littermates received CCx surgery under isoflurane anesthesia. All subjects were prepared for testing as described above (n = 8 per group). Surgeries were staggered to minimize the latency between surgery and testing.
Procedure
After recovery from surgery, the pup was transferred to the recording apparatus. After acclimation, the recording session began with a 1μl infusion of the vehicle, artificial cerebrospinal fluid (ACSF), into the left or right S1 and behavior was scored for 15 min immediately after the infusion. After the behavioral recording period, the subject received either a 1μl infusion of the GABAA receptor agonist, muscimol hydrobromide (0.01M mixed with ACSF; Sigma-Aldrich, St. Louis, MO, USA), or a 0.5μl infusion of the GABAA antagonist, bicuculline methiodide (10μM; Sigma-Aldrich, St. Louis, MO, USA). The volumes and concentrations for muscimol (Campolattaro & Freeman, 2008) and bicuculline (Jones & Barth, 2002) are based on previously published work. After the infusion, another 15-min period of behavioral scoring was conducted. The remaining 3 littermates were tested in succession and the order of testing (i.e., sham vs. CCx; muscimol vs. bicuculline; left vs. right S1) was randomized.
Infusions were delivered using a Hamilton microliter syringe with a 25-gauge needle (Model 7001; Hamilton, Reno, NV, USA) secured to the stereotaxic apparatus and mounted directly above the infusion site. The syringe was lowered just below the cortical surface into a pre-drilled hole located halfway between the two Ag/AgCl electrode sites. The infusion rate for all infusions was 0.1μl/s. These methods are identical to a previous report (Marcano-Reik & Blumberg, 2008) and it has been shown before that surface and intracortical infusions do not differ significantly in their effects on SB activity (Hanganu et al., 2007).
Three of the muscimol subjects received an infusion of the same concentration of fluorescently labeled muscimol (Allen et al., 2008) (BODIPY TMR-X conjugate; Invitrogen, Eugene, Oregon, USA). At the end of the recording session, the pups were overdosed with an intraperitoneal injection of sodium pentobarbital and perfused transcardially with phosphate-buffered saline, followed by a 3% formalin solution. Brains were postfixed for at least 48 h in a formalin–sucrose solution before being sliced in the coronal plane with a sliding microtome (50μm sections). A fluorescent microscope was used to visualize the distribution of muscimol throughout the brain.
Data Analysis
For all subjects, paired (within-subjects) t tests were performed using JMP 5.0 software (SAS, Cary, North Carolina, USA). For all tests, alpha was set at 0.05 and Bonferroni corrections were used to correct alpha for multiple comparisons.
Western blot analysis of K+Cl− co-transporter 2 (KCC2) expression across age
Procedure
Cortical tissue including the S1 region of P5, P6, P7, P8, and P9 rats (n=2 per age) was homogenized in RIPA buffer (150 mM NaCl, 1% Triton X-100, 0.5% deoxycholic acid, 0.1% SDS, 50 mM Tris-HCl, pH 8.0) with Complete Mini, EDTA-free protease inhibitor cocktail (Roche Applied Science, Indianapolis, IN). Concentration was determined by BCA analysis. Samples were incubated in sample buffer (80 mM Tris-HCl, 2% SDS, 10% glycerol, 5.3% β-mercaptoethanol, 0.025% bromophenol blue) at 75 C for 10 min. Samples were run, 10 μg each, on a 7.5% polyacrylamide gel and then transferred to a nitrocellulose membrane. The membrane was blocked in TBST (Tris Buffered Saline, 0.1% Tween-20) and 5% milk, then incubated overnight at 4°C with antibodies against KCC2 (Rabbit anti-KCC2, Upstate Cell Signaling Solutions, Lake Placid, NY; 1:1000 dilution in TBST) and β-tubulin (Mouse anti-β-tubulin, Sigma-Aldrich, St. Louis, MO; 1:1000 dilution in TBST). The membrane was washed in TBST, and probed with corresponding HRP-conjugated secondary antibodies (Goat anti-Rabbit HRP IgG HL, Jackson ImmunoResearch, West Grove, PA; 1:10,000 dilution in TBST with 5% milk; Goat anti-Mouse HRP IgG HL, Pierce Biotechnology, Rockford, IL; 1:5000 dilution in TBST with 5% milk) and imaged using SuperSignal West Pico Chemiluminescent Substrate (Pierce Biotechnology, Rockford, IL).
Scanned images of blots were quantified using the “Analyze Gels” functions in ImageJ (National Institutes of Health). The intensity of KCC2 bands were quantified for each lane and normalized to the b-tubulin signal. Data were then expressed as arbitrary units after normalization to the P5 signal.
Disruption and recovery of evoked responses to forepaw stimulation after callosotomy
Subjects and surgical preparation
A total of 132 P1–15 infant rats from 18 litters were used. In one set of 6 litters, a total of 60 littermates (10 per litter) received sham or CCx surgery at P1 and were tested in pairs at P1, P2, P4, P6, or P8. In a second set of 6 litters, a total of 36 littermates (6 per litter) received sham or CCx surgery at P6 and were tested in pairs at P6, P9, or P13. In a final set of 6 litters, a total of 36 littermates (6 per litter) received sham or CCx surgery at P8 and were tested in pairs at P9, P12, or P15 (this dataset was supplemented with previously collected data from 12 subjects and 6 litters in which pups received sham or CCx surgery on P8 and were tested in pairs the same day). Thus, at each age, 6 sham and 6 CCx littermates from 6 litters were tested. Males and females were equally represented among the subjects.
Surgeries were identical to those described above. However, for those subjects that received sham or CCx surgeries at P1, P6, or P8 and were to be tested at a later age, the incision was closed with Vetbond and pups, along with their other littermates, were transferred to a humidified incubator maintained at thermoneutrality (35°C) to recover for 1 h. At the end of the recovery period, all pups, with the exception of the 2 subjects to be tested that same day, were placed back in the home cage with their mother. At the appropriate time, 2 littermates were transferred to an electrically shielded chamber for acclimation and neurophysiological and behavioral testing, as described above. All procedures for recording evoked and spontaneous activity were the same as described above.
When pups were returned to their mother and homecage, daily observations were conducted to ensure that all subjects were healthy and maintained a visible milk band. Body weights were not different between sham and CCx subjects 1 week after surgery (at P8 after surgery at P1: shams: 20.2–24.1 g; CCx: 19.1–24.5 g; at P13 after surgery at P6: shams: 30.6–36.9 g; CCx: 30.5–40.2 g; at P15 after surgery at P8: shams: 37.4–42.7 g; CCx: 38.9–43.2 g).
Data Analysis
For all subjects, paired t tests were performed at each age using JMP 5.0 software (SAS, Cary, North Carolina, USA). For all tests, alpha was set at 0.05 and Bonferroni corrections were used to correct alpha for multiple comparisons.
Immunofluorescence
Procedure
To determine normal changes in presynaptic terminals and receptor populations, additional subjects were anesthetized with isofluorane and received sham surgery (i.e., the same surgical procedures as those described above) on P6, P7, P8 or P9 (n=3 at each age). Subjects were perfused transcardially with cold 0.1M phosphate buffered saline (PBS) followed by 2% paraformaldehyde in PBS (pH=7.4). Following perfusion the brains were extracted and postfixed in cold 2% paraformaldehyde in PBS overnight. The brains were then cryoprotected in 30% sucrose in PBS at 4°C and frozen in OCT compound (Tissue-Tek; Sakura-Finetek) using dry ice/ethanol-cooled isopentane.
Coronal cryostat sections (12μm) at the level of the corpus callosum were cut on a Leica CM1850 cryostat and collected with the aid of a Leica CryoJane tape transfer system. Tissue sections were air-dried and then blocked in 2.5% bovine serum albumin, 0.1% Triton X-100 in PBS for 1 h, followed by overnight incubation at 4°C with primary antibodies: guinea pig anti-vesicular glutamate transporter-1 (VGLUT-1; 1:500, Chemicon) and mouse anti-glutamic acid decarboxylase (GAD; mAb GAD-6; 1:10 Developmental Studies Hybridoma Bank) diluted in the same blocking solution. Sections were washed in PBS and incubated for 1 h at room temperature with the appropriate secondary antibodies conjugated to Alexa 488 and 568 (Invitrogen). Sections were washed in PBS containing the nuclear counterstain DAPI (4′, 6′ diamidino-2 phenylindole) and mounted in Gel/Mount (Biomeda) aqueous mounting media.
Image Acquisition and Analysis
Quantification of presynaptic (VGLUT-1-positive or GAD-positive) puncta was performed on 6 sections per animal from 3 different animals at each time point (i.e., 18 fields for each marker). Images were taken of fields within S1 cortex using 63X Plan Apo objectives on a Leica DM5000B digital epifluorescence microscope. Digital images were captured in Adobe Photoshop and similarly adjusted for brightness and contrast. Images were thresholded in NIH Image/J and synaptic puncta were counted by using the Analyze Particle module; the accuracy of these automated quantifications was confirmed in a portion of each field by manual counts.
Data Analysis
To control for field-by-field variations in cell density, we normalized the number of presynaptic terminals to the number of cell nuclei in each field. The proportion of cells that labeled with antibodies to the neuronal marker NeuN was also monitored, and did not change between P6 and P9 (data not shown), confirming that total cell density is proportional to neuronal density. ANOVAs were separately conducted for each marker (i.e., VGLUT-1 and GAD) and cortical depth (i.e., layers 2–3 and 4–6) using JMP 5.0 software (SAS, Cary, North Carolina, USA). Student’s t test was used for post hoc comparisons. Alpha was set at 0.05 and the Bonferroni correction procedure was used.
RESULTS
Abrupt disappearance of CCx-induced disinhibition of spontaneous SBs after P6
As shown previously (Marcano-Reik & Blumberg, 2008), CCx doubled the rate of spontaneous SBs in P1–6 subjects (Figure 1, Top: Ps<0.001, n=4/age). This disinhibition was reflected in shorter latencies separating SBs produced by the two hemispheres (Figure 1, bottom: P<0.0001). (Given that rates of twitching were unaffected by CCx (see below) and that SBs largely cluster during periods of myoclonic twitching, it appears that CCx increases the probability that twitches trigger SBs in the contralateral S1.) Then, abruptly at P7, CCx no longer disinhibited spontaneous SBs in relation to shams. Coincident with this abrupt disappearance of CCx-induced disinhibition, the rate of SBs in both sham and CCx subjects increased sharply and peaked at P8 before steadily decreasing over the next several days.
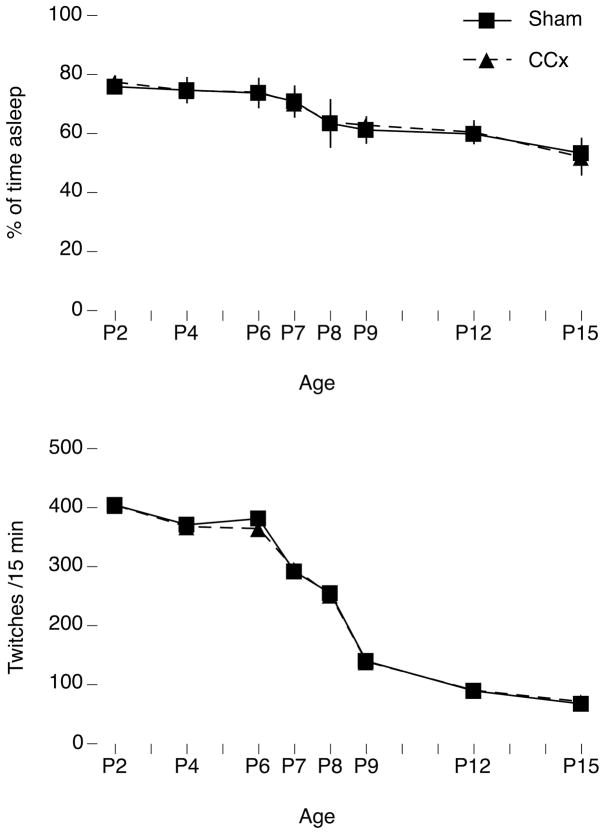
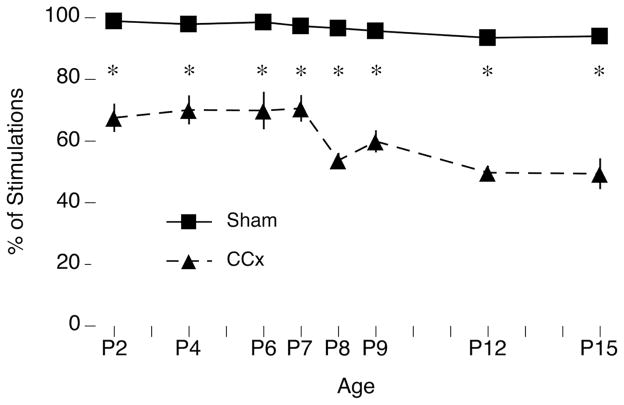
We next confirmed that the age-related effects of CCx described above were not due to alterations of sleep or rates of twitching. Given that disconnection of the cortex does not alter sleep-wake cycling in P8 rats (Karlsson, Kreider, & Blumberg, 2004), we expected CCx to have no effect on the percentage of time asleep in relation to shams; indeed, no effect was found (Figure 2, top). Also, since twitches are produced within the brainstem and are not dependent on forebrain structures (Kreider & Blumberg, 2000), we anticipated that CCx would not alter or influence their occurrence, which is what was observed (Figure 2, bottom). Interestingly, we found that over several days beginning at P6, the rate of twitching rapidly declined in both sham and CCx subjects.
Figure 2.
Developmental changes in percentage of time asleep and rates of active sleep-related twitching in sham and callosotomized (CCx) P2–15 rats. Top: In relation to shams, CCx had no effect on the percentage of time asleep across the 15-min recording period at any age. Bottom: Although the quantity of nuchal muscle twitches per 15 min decreased with age, especially after P6, CCx again had no effect on twitching at any age. Because twitches are produced within the brainstem, we anticipated that CCx would not influence their occurrence. Means ± sem.
GABAergic modulation of spontaneous spindle bursts in intact and callosotomized subjects
We hypothesized that CCx-induced disinhibition of SBs through P6 results from a reduction of excitatory drive onto GABAergic interneurons. To test this hypothesis, we unilaterally infused artificial cerebrospinal fluid (ACSF) just below the S1 cortical surface in P4 rats, followed by an infusion of the GABAA agonist muscimol or antagonist bicuculline (n=8 subjects/group). In shams, muscimol did not significantly influence spontaneous SB production whereas bicuculline significantly increased spontaneous SBs (P<0.001, n=8) across the 15-min test period in relation to ACSF (Figure 3A). On a percentage basis, muscimol decreased SBs by only 11.3% whereas bicuculline increased SBs by 60% in relation to ACSF. This finding of a floor effect with respect to GABAergic inhibition suggests that the corpus callosum exerts a powerful inhibitory influence on S1 networks at this age.
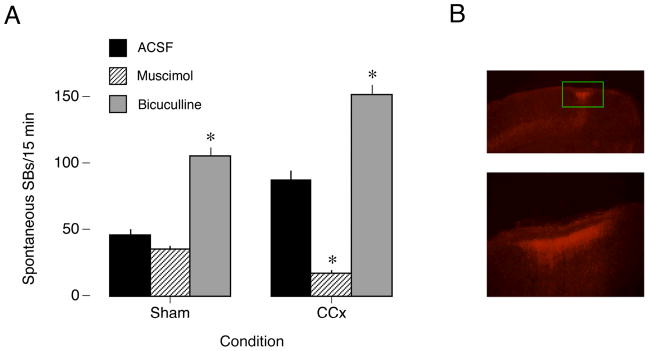
Figure 3.
(A) Effect of unilateral infusion of ACSF, muscimol, or bicuculline into S1 on spontaneous SB activity in sham and CCx P4 rats. Note that in CCx subjects, but not sham subjects, muscimol and bicuculline have equal and opposite effects on SB production. * significant difference from ACSF. Means ± sem. (B) The distribution of fluorescently labeled muscimol (0.01 M) after a 1 μl infusion into the left S1 of a P4 rat. Top: Coronal section of the left hemisphere showing the site of muscimol infusion (green box). Bottom: The boxed area above is magnified.
If the corpus callosum exerts its effects at P4 by preferentially exciting GABAergic interneurons, then it should be possible to compensate for the reduction of GABAergic modulation after CCx by infusing muscimol. This is exactly what was found. Specifically, in relation to ACSF, muscimol and bicuculline significantly reversed and exacerbated (Ps<0.001, n=8), respectively, the effect of CCx-induced disinhibition on spontaneous SBs. On a percentage basis, muscimol decreased SBs by 71% and bicuculline increased SBs by 64.5% in relation to ACSF (Figure 3A).
In a subset of the muscimol subjects (n=3), fluorescently labeled drug was infused to confirm the desired distribution of the drug (Figure 3B). Muscimol was clearly localized near the site of infusion, although some diffusion to other cortical and subcortical areas may have occurred. Diffusion to the contralateral hemisphere was not detected; indeed, consistent with this observation, neither muscimol nor bicuculline infusions affected spontaneous SB production in the non-drug hemisphere (data not shown).
To assess developmental changes in GABAergic modulation of SBs, we examined in P10 subjects the effects of muscimol and bicuculline infusion. However, in contrast with its effects at P4, infusion of bicuculline at P10 produced a hyperexcitable state that included epileptiform activity. That such activity would occur after but not before P7 in response to GABA antagonism is consistent with a previous report examining age-related differences in epileptiform activity in neocortical slices (Wong & Yamada, 2001). Unfortunately, the nearly continuous cortical activation induced by bicuculline precluded quantification of SB activity at this age.
KCC2 upregulation at the end of the first postnatal week
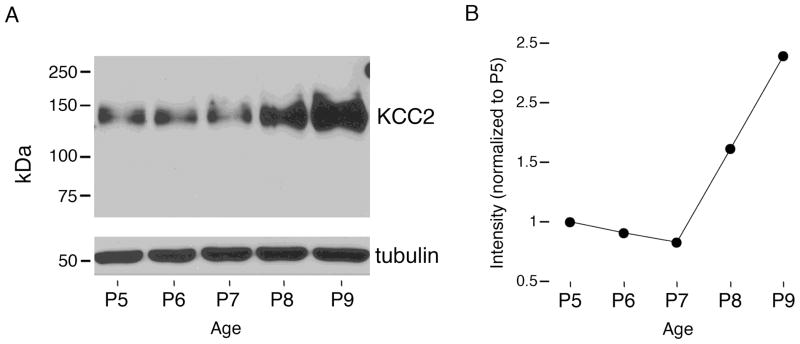
The inhibitory and excitatory effects of muscimol and bicuculline, respectively, on SB activity at P4 are consistent with GABA exerting its postsynaptic effects through either shunting inhibition or hyperpolarization. Upregulation of the K+/Cl− co-transporter KCC2, through its control of the Cl− reversal potential, is thought to be a critical factor in the postnatal emergence of GABA’s hyperpolarizing effects (Sipilä, Blaesse, & Kaila, 2010). Although KCC2 is known to be upregulated sometime during the first two postnatal weeks in the cortex (Blaesse et al., 2006; Wang et al., 2002), protein levels have not been examined on a day-by-day basis. Therefore, we assessed KCC2 levels in cortex from P5, P6, P7, P8, and P9 rats using Western blot analysis (Figure 4A). As shown in Figure 4B, levels of KCC2 increase abruptly after P7.
Figure 4.
Cortical KCC2 protein levels increase at the end of the first postnatal week. (A) Western blot analysis of KCC2 expression in rat cortex at P5, P6, P7, P8, and P9. kDa, kilodalton. (B) The intensity of KCC2 bands normalized to P5. KCC2 levels are stable between P5 and P7, but increase sharply thereafter.
Rapid developmental changes in GABAergic and glutamatergic presynaptic terminals
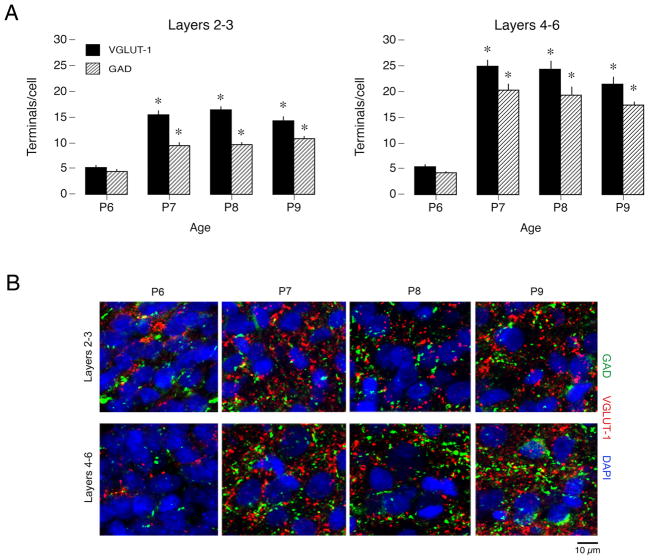
We examined whether the rapid changes in SB production between P6 and P9 are accompanied by anatomical changes in GABAergic and glutamatergic presynaptic terminals in S1. Using an additional set of sham subjects (n=3 pups/age; 6 fields/pup), we stained cryosections with antibodies directed against glutamic acid decarboxylase (GAD) and vesicular glutamate transporter-1 (VGLUT1) in layers 2–3 and 4–6 in S1. We controlled for any variation in cell density by normalizing the number of presynaptic terminals to the number of cell nuclei in each field (see Materials and Methods). We found that GABAergic and glutamatergic presynaptic terminals increased rapidly and significantly at P7 in relation to P6, especially in the deep layers (Figure 5A; Ps<0.0001).
Figure 5.
Changes in GABAergic and glutamatergic presynaptic terminals in P6–9 rats. (A) Number of S1 glutamatergic (VGLUT-1) and GABAergic (GAD) presynaptic terminals in layers 2–3 (left) and 4–6 (right), normalized to the number of cell nuclei, increase significantly at P7 in relation to P6. * significant difference from value at P6. Means ± sem. (B) Sections through S1 cortex in P6, P7, P8, or P9 sham-operated animals were stained with antibodies against inhibitory (GAD, green) and excitatory (VGlut-1, red) presynaptic terminals; nuclei were counterstained with DAPI (blue). The density of both GABAergic and glutamatergic terminals in layers 2–3 (top row) and layers 4–6 (bottom row) increases sharply between P6 and P7, with little subsequent change over the next two days.
The marked increase in GAD and VGLUT1 terminals between P6 and P7 can be seen clearly in representative immunofluorescent fields from layers 2–3 and 4–6 (Figure 5B). Along with this sharp increase in the number of presynaptic terminals, cortical cells appeared more widely spaced at P7 and later, resulting in a reduced number of cells per microscope field. This increased spacing is almost certainly due to the rapid expansion of the neuropil and continued growth of the cortex, as no apoptosis was observed between P6 and P7 (based on immunostaining for cleaved caspase-3; data not shown). As noted above, we normalized our synaptic terminal counts to the cell density in each microscope field; thus, these changes in cell density cannot account for the observed increase in the number of presynaptic terminals. Indeed, the absolute (i.e., not normalized) number of presynaptic terminals also exhibited a sharp increase after P6 despite the decrease in cell density. Thus, our histological findings indicate a rapid and steep increase in cortical innervation after P6 that coincides with the loss of the disinhibitory effects of CCx, the upregulation of KCC2, and the developmental decrease in myoclonic twitching.
Immediate disruptive effects of callosotomy on evoked spindle bursts are consistent across age
We measured the immediate effect of CCx on SB responses in the S1 forelimb region to peripheral stimulation to the plantar surface of the contralateral forepaw. In sham subjects, forepaw stimulation reliably evoked an SB response at all ages (Figure 6); in contrast, CCx significantly reduced the probability of an evoked response by 30–50% (Ps<0.0001).
Figure 6.
Callosotomy (CCx) disrupts the SB response to peripheral stimulation of the contralateral forepaw in P2–15 rats. In sham subjects the response to stimulation of the forepaw reliably evokes an SB in contralateral S1 in P2–P15 infant rats; however, CCx immediately decreases the response to forepaw stimulation at all ages. * significant difference between groups. Means ± sem.
Developmental changes in recovery of function after callosotomy
To assess recovery of function across the early postnatal period, we performed sham or CCx surgeries at P1, P6, or P8 and tested littermates on the same day or up to one week later. As shown in Figure 7A and 7B, CCx at P1 and P6 immediately disrupted evoked responding to forepaw stimulation; over the ensuing week, evoked responding in the CCx pups gradually recovered to sham levels. Also, as expected, at P1 and P6 CCx doubled the rate of spontaneous SBs in relation to shams; over the ensuing week, spontaneous SBs in CCx subjects gradually declined to levels at or below those of shams (data not shown).
Figure 7.
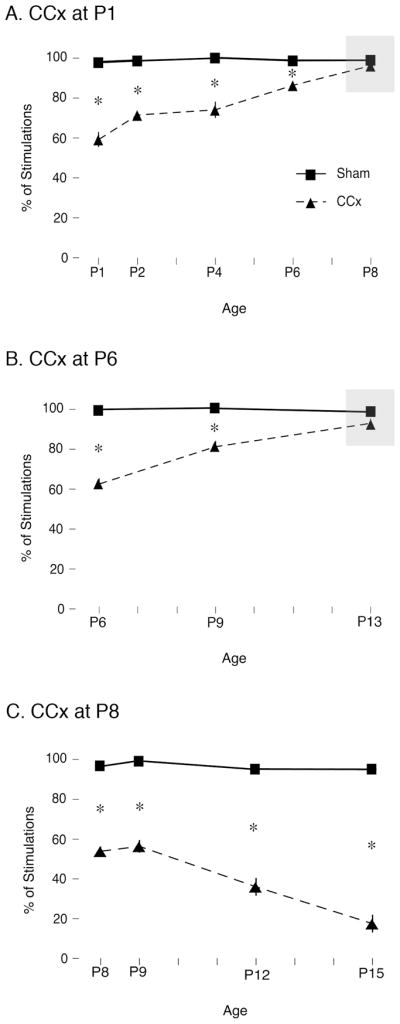
Effect of callosotomy (CCx) on evoked spindle burst (SB) responses to contralateral forepaw stimulation and recovery of function across different ages. Subjects received sham (squares) or CCx (triangles) surgery at (A) P1, (B) P6, or (C) P8 with subsequent testing over the ensuing week. In subjects that received CCx at P1, P6, and P8, evoked responding was immediately disrupted. Importantly, recovery of function was observed after 7 days in pups that received CCx at P1 or P6 (shaded boxes) but not in pups that received CCx at P8. * significant difference between groups. Means ± sem.
As shown in Figure 7C, CCx at P8 also produced an immediate disruption of evoked responding. However, in contrast to pups experiencing CCx at P1 and P6, recovery of function was not observed; instead, steady deterioration of evoked responding was observed over the ensuing week. As expected, disinhibition of spontaneous SBs by CCx was not observed at any age.
DISCUSSION
We have documented contemporaneous behavioral, neurophysiological, anatomical, and molecular changes that suggest a role for sleep-related motor activity and its associated sensory feedback in modifying the structure and function of cortical circuits during early development. Specifically, we saw a rapid decrease in myoclonic twitching after P6, at which time CCx-induced disinhibition of spontaneous SBs also disappeared abruptly. In addition, both GABAergic and glutamatergic innervation of superficial and deep cortical layers increased sharply at P7 as the rate of spontaneous SBs in both sham and CCx subjects increased dramatically, peaked at P8, and then decreased over the next week. KCC2 expression also increased across these ages, suggesting that developmental changes in GABAergic functioning are associated with changes in the intrinsic excitability of local cortical circuits involved in the production of SBs. These behavioral, neurophysiological, neuroanatomical, and molecular changes after P6 correlated with an abrupt reduction in somatosensory cortical plasticity. Importantly, the recovery of reliable evoked SBs after CCx was predicted by CCx-induced increases in the production of spontaneous SBs, thus suggesting that sensory feedback from myoclonic twitching is a participating factor in cortical plasticity and recovery of function.
Myoclonic twitch movements of the limbs, tail, and head are produced by phasic, rapid activation of skeletal muscles (Gramsbergen et al., 1970; Karlsson & Blumberg, 2002; Seelke, Karlsson, Gall, & Blumberg, 2005); for review, see Blumberg & Seelke, 2010). Neurons within the mesopontine region participate in the production of twitches during the early postnatal period (Karlsson et al., 2005; Kreider & Blumberg, 2000). Recently, it was discovered that sensory feedback from twitching produces SBs in somatosensory cortex (Khazipov et al., 2004); moreover, it appears that it is the proprioceptive feedback from limb twitching that triggers somatosensory SBs (Marcano-Reik & Blumberg, 2008). In addition, SBs are produced in visual (Hanganu et al., 2007) and barrel (Minlebaev et al., 2007) cortex, and hippocampal activity is also modulated by sensory feedback from twitching (Mohns & Blumberg, 2008, 2010). Thus, it is now clear that sensory feedback from twitching modulates neural activity throughout the neuraxis – from spinal cord (Petersson, Waldenström, Fåhraeus, & Schouenborg, 2003) to forebrain – thus supporting the hypothesis that sleep-related endogenous activity shapes and refines neural circuits in early development and across the lifespan (Blumberg & Lucas, 1996; Blumberg & Seelke, 2010; Corner et al., 1980; Roffwarg, Muzio, & Dement, 1966).
From the data in Figure 2 – and given that circadian effects on sleep-wake activity at these ages are relatively small (Gall, Todd, Ray, Coleman, & Blumberg, 2008) – we estimate that the nuchal muscle twitches over 38,000 times per 24 h at P6; this number decreases to 9,500 at P9, a four-fold decrease over 3 days. Within the forelimb region of somatosensory cortex, we estimate that nearly 4,000 SBs occur per 24 h at P6, a value that jumps to over 15,000 at P8 before declining to levels below 4,000 by P12. These values highlight the considerable quantity of endogenous activity produced and experienced by infant rats over the first two postnatal weeks. Thus, in early development, twitching may serve a similar role for the somatosensory cortex as retinal waves serve for the visual cortex (Katz & Shatz, 1996; Wong, 1999). However, further work is needed to establish a role for twitching in the development and refinement of somatosensory cortex and to determine whether its role involves permissive or instructive influences (Crair, 1999).
Our findings suggest that callosal fibers inhibit spontaneous SBs during the first postnatal week via excitatory effects on GABAergic interneurons (see Khazipov, 2009) for further evidence of a role for GABA in SB modulation. Although activation of GABAA receptors typically depolarizes postsynaptic cells in immature cortex before the developmental upregulation of KCC2, GABA-mediated inhibition can still occur as a result of shunting inhibition of glutamatergic excitatory postsynaptic currents (Akerman & Cline, 2007; Ben-Ari, 2002; Lamsa, Palva, Ruusuvuori, Kaila, & Taira, 2000; Owens & Kriegstein, 2002; Sipilä et al., 2010). For example, similar to what was found here, in the newborn hippocampus, muscimol and bicuculline decrease and increase network activity, respectively (Lamsa et al., 2000). However, the pharmacological manipulations used here are not sufficient to discern the mechanism by which GABA inhibits postsynaptic activity.
The fact that KCC2 is upregulated in the cortex at the end of the first postnatal week suggests that GABA does indeed exert depolarizing effects at earlier ages. This upregulation is also consistent with previous findings in cortical tissue across a broader range of ages. Specifically, KCC2 mRNA in cortex increases between P1 and P15 (Wang et al., 2002) and KCC2 immunoreactivity is very low at P4 and uniformly high at P12 (Blaesse et al., 2006). In rat neocortical brain slices, the GABAA receptor reversal potential approaches the resting membrane potential sometime after P4 (Owens, Boyce, Davis, & Kriegstein, 1996). Similar to what we found here in cortex, KCC2 expression in hippocampus also increases sharply between P5 and P9 (Rivera, Voipio, & Kaila, 2005).
The callosal inhibition observed here through P6 might be attributable in part to a transient GABAergic population of callosal neurons comprising approximately 21% of the callosal bundle in infant rats (Cobas, Alvarez-Bolado, & Fairen, 1988; Kimura & Baughman, 1997). Significantly, these GABAergic neurons largely disappear at or before P6 and few such fibers are detected in adults (Fabri & Manzoni, 2004). The connectivity of these transient neurons in the contralateral hemisphere is not yet known.
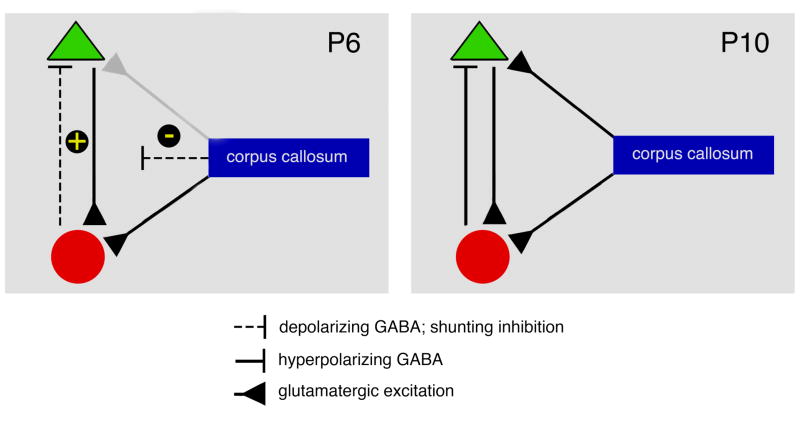
Figure 8 presents a model depicting hypothesized changes in intrinsic cortical circuitry and callosal influences on that circuitry between P6 and P10. At P6, the local circuit producing SBs comprises glutamatergic pyramidal cells and GABAergic interneurons; at this age, GABAergic inhibition occurs through shunting. The fact that CCx disinhibits SB activity at this age suggests net excitation in the intrinsic cortical circuit that is counteracted by a net inhibitory influence provided by callosal excitation of GABAergic interneurons and/or direct inhibition of pyramidal cells by the transient population of GABAergic callosal neurons. By P10, after KCC2 upregulation, the emergence of hyperpolarizing GABA, and the other changes in intrinsic connectivity described here, we hypothesize that excitatory-inhibitory balance is now expressed within the intrinsic cortical circuit. In addition, we hypothesize that callosal fibers, which are thought to exert both excitatory and inhibitory effects in adults (Bloom & Hynd, 2005; Makarov, Schmidt, Castellanos, Lopez-Aguado, & Innocenti, 2008), exert a more balanced influence on the intrinsic circuit by P10. Thus, we view excitatory-inhibitory balance as arising from complex interactions among intrinsic cortical circuits and extrinsic influences that include the corpus callosum.
Figure 8.
Model depicting hypothesized mechanisms underlying the development of excitatory-inhibitory balance in S1 between P6 and P10. The model depicts an S1 network comprising GABAergic interneurons (red circles) and glutamatergic pyramidal cells (green triangles) combined with callosal modulation of the intrinsic circuit. Left: At P6, CCx disinhibits SB activity in S1, suggesting net excitation (+) in the intrinsic cortical circuit and a net inhibitory influence (−) by the corpus callosum. Although GABA is typically depolarizing early in development, GABA-mediated inhibition can still occur as a result of shunting of glutamatergic excitatory postsynaptic currents (dashed line). According to this model, net callosal inhibition is achieved by preferential excitation of GABAergic interneurons as well as the presence of transient GABAergic callosal projections (most likely by inhibiting pyramidal cells, although such connectivity is not specified in the figure). Right: At P10 and after KCC2 upregulation, the hyperpolarizing effects of GABA predominate and the intrinsic cortical circuit has achieved excitatory-inhibitory balance. In addition, callosal inputs from the corpus callosum now exert equal effects on excitatory and inhibitory cortical neurons and GABAergic callosal projections have disappeared. As a consequence, CCx at this age has no discernible effect on spontaneous activity (although evoked responses are still affected).
In addition to their neurophysiological effects, glutamate and GABA may act as trophic signals in early development that, among other things, stimulate the release of brain-derived neurotrophic factor (BDNF) (Akerman & Cline, 2007; Ben-Ari, 2002; Marmigere, Rage, & Tapia-Arancibia, 2003; Marty, Berzaghi Mda, & Berninger, 1997; Owens & Kriegstein, 2002; Xiong et al., 2002). BDNF is produced by cortical pyramidal cells and, in an activity-dependent manner, regulates synaptic interactions among glutamatergic pyramidal cells and GABAergic interneurons (Turrigiano, 1999). As a consequence, twitch-triggered SBs in the developing neocortex may, through the release of glutamate, GABA and BDNF, contribute to the somatosensory cortical plasticity observed here during the first postnatal week. Moreover, the endogenously generated SBs may provide the activity upon which KCC2 upregulation and the emergence of hyperpolarizing GABA depend. This activity may be mediated by GABA itself (Ganguly, Schinder, Wong, & Poo, 2001), by glutamate (Kanold, 2009), or by other factors (Ludwig, Li, Saarma, Kaila, & Rivera, 2003).
After P6, as rates of twitching sharply decline and GABA no longer exerts depolarizing effects, we hypothesize that associated declines in the activity-dependent release of BDNF will lead to relative increases in excitatory drive onto pyramidal cells, as has been hypothesized for cultured cortical networks (Turrigiano, 1999). These adjustments in network organization, which may also include the observed relative increases in glutamatergic presynaptic terminals (see Figure 5), may contribute to the peak in SB activity at P8 and the emergence of excitatory-inhibitory balance (Akerman & Cline, 2007; Hensch, 2005; Owens & Kriegstein, 2002).
In light of all of these foregoing issues, we predicted that CCx-induced disinhibition during the first postnatal week would serve as a bioassay of somatosensory cortical plasticity. To test this prediction, we performed sham or CCx surgeries at P1, P6, or P8 and tested littermates immediately or over the ensuing week. Although the evoked response to contralateral forepaw stimulation is typically very reliable, CCx consistently disrupted evoked responses to forepaw stimulation. Only pups receiving CCx at P1 or P6 – ages at which CCx disinhibits spontaneous SB activity – exhibited recovery of function. The evoked SB responses of pups that received CCx at P8 deteriorated further over the week. In addition, as evoked responses recovered, inhibitory control of spontaneous responses appeared to reemerge (see Figure 7). Although we do not yet know whether the increased production of spontaneous SBs is causally related to recovery of function in S1, it does appear that this increased production can be used as a bioassay of plasticity-promoting conditions conducive to recovery of function. Moreover, the present findings suggest a new model for exploring somatosensory cortical plasticity – involving disruption of the local circuit by CCx – that complements other models of cortical plasticity (Aton et al., 2009; Feldman, 2009; Feller & Scanziani, 2005).
It is interesting that CCx before P6 doubles the expression of spontaneous SBs but decreases the likelihood of evoked SBs to contralateral forepaw stimulation (compare the developmental profiles in Figures 1A and 4). We have previously reported other pronounced differences between spontaneous and evoked SBs (Marcano-Reik & Blumberg, 2008). Such differences could reflect the contributions of non-overlapping neural circuits to the production of spontaneous and evoked SBs, including perhaps the production of an efference copy in association with spontaneous SBs. Efference copy provides a mechanism by which animals distinguish sensations arising from self-generated or passive movements (Blakemore, Wolpert, & Frith, 2000; Cullen, 2004). Thus, we hypothesize that efference copy associated with self-generated twitches, perhaps communicated through the cholinergic basal forebrain (Juliano, Ma, & Eslin, 1991; Kilgard & Merzenich, 1998; Ma, Hohmann, Coyle, & Juliano, 1989; Rasmusson & Dykes, 1988; Tremblay, Warren, & Dykes, 1990), prepares the neocortex for twitch-related sensory feedback, thereby contributing to sensorimotor development and somatotopic organization. Interestingly, cholinergic afferents from the basal forebrain facilitate spindle burst activity in primary visual cortex in P5–6 rats (Hanganu et al., 2007).
All together, the changes in twitch behavior and associated SB activity observed here suggest that the first postnatal week in rats ends with an increase in intrinsic cortical synaptic connectivity, a transformation in GABA’s functional properties, the establishment of excitatory-inhibitory balance, and a diminution of one form of somatosensory cortical plasticity. These findings also suggest that the corpus callosum participates in the activity-dependent development and refinement of cortical networks and point the way to an understanding of how malformations of the corpus callosum may contribute to a variety of neurological and psychiatric disorders (Paul et al., 2007).
Acknowledgments
We thank Matthew Campolattaro, Cynthia Shaw, and Claire Vernon for technical assistance. This work was supported by a research grant (MH50701) and an Independent Scientist Award (MH66424) from the National Institutes of Health (to M.S.B.).
Footnotes
Publisher's Disclaimer: The following manuscript is the final accepted manuscript. It has not been subjected to the final copyediting, fact-checking, and proofreading required for formal publication. It is not the definitive, publisher-authenticated version. The American Psychological Association and its Council of Editors disclaim any responsibility or liabilities for errors or omissions of this manuscript version, any version derived from this manuscript by NIH, or other third parties. The published version is available at www.apa.org/pubs/journals/bne
References
- Akerman CJ, Cline HT. Refining the roles of GABAergic signaling during neural circuit formation. Trends in Neurosciences. 2007;30:382–389. doi: 10.1016/j.tins.2007.06.002. [DOI] [PubMed] [Google Scholar]
- Allen TA, Narayanan NS, Kholodar-Smith DB, Zhao Y, Laubach M, Brown TH. Imaging the spread of reversible brain inactivations using fluorescent muscimol. Journal of Neuroscience Methods. 2008;171:30–38. doi: 10.1016/j.jneumeth.2008.01.033. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Aton SJ, Seibt J, Dumoulin M, Jha SK, Steinmetz N, Coleman T, Naidoo N, Frank MG. Mechanisms of sleep-dependent consolidation of cortical plasticity. Neuron. 2009;61:454–466. doi: 10.1016/j.neuron.2009.01.007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ben-Ari Y. Excitatory actions of GABA during development: The nature of the nurture. Nature Reviews Neuroscience. 2002;3:728–739. doi: 10.1038/nrn920. [DOI] [PubMed] [Google Scholar]
- Blaesse P, Guillemin I, Schindler J, Schweizer M, Delpire E, Khiroug L, Friauf E, Nothwang HG. Oligomerization of KCC2 correlates with development of inhibitory neurotransmission. Journal of Neuroscience. 2006;26:10407–10419. doi: 10.1523/JNEUROSCI.3257-06.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Blakemore SJ, Wolpert D, Frith C. Why can’t you tickle yourself? Neuroreport. 2000;11:R11–16. doi: 10.1097/00001756-200008030-00002. [DOI] [PubMed] [Google Scholar]
- Bloom JS, Hynd GW. The role of the corpus callosum in interhemispheric transfer of information: excitation or inhibition? Neuropsychology Review. 2005;15:59–71. doi: 10.1007/s11065-005-6252-y. [DOI] [PubMed] [Google Scholar]
- Blumberg MS, Lucas DE. A developmental and component analysis of active sleep. Developmental Psychobiology. 1996;29:1–22. doi: 10.1002/(SICI)1098-2302(199601)29:1<1::AID-DEV1>3.0.CO;2-Y. [DOI] [PubMed] [Google Scholar]
- Blumberg MS, Seelke AM. The form and function of infant sleep: From muscle to neocortex. In: Blumberg MS, Freeman JH, Robinson SR, editors. The Oxford Handbook of Developmental Behavioral Neuroscience. New York: Oxford University Press; 2010. pp. 391–423. [Google Scholar]
- Campolattaro MM, Freeman JH. Eyeblink conditioning in 12-day-old rats using pontine stimulation as the conditioned stimulus. Proceedings of the National Academy of Sciences of the United States of America. 2008;105:8120–8123. doi: 10.1073/pnas.0712006105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cobas A, Alvarez-Bolado G, Fairen A. Transient GABA-like immunoreactive axons in the corpus callosum of perinatal rats. Neuroscience Letters. 1988;93:7–12. doi: 10.1016/0304-3940(88)90003-1. [DOI] [PubMed] [Google Scholar]
- Corner MA, Mirmiran M, Bour HL, Boer GJ, van de Poll NE, van Oyen HG, Uylings HB. Does rapid-eye-movement sleep play a role in brain development? Progress in Brain Research. 1980;53:347–356. doi: 10.1016/S0079-6123(08)60074-6. [DOI] [PubMed] [Google Scholar]
- Crair MC. Neuronal activity during development: permissive or instructive? Current Opinion in Neurobiology. 1999;9:88–93. doi: 10.1016/s0959-4388(99)80011-7. [DOI] [PubMed] [Google Scholar]
- Cullen KE. Sensory signals during active versus passive movement. Current Opinion in Neurobiology. 2004;14:698–706. doi: 10.1016/j.conb.2004.10.002. [DOI] [PubMed] [Google Scholar]
- Fabri M, Manzoni T. Glutamic acid decarboxylase immunoreactivity in callosal projecting neurons of cat and rat somatic sensory areas. Neuroscience. 2004;123:557–566. doi: 10.1016/j.neuroscience.2003.09.011. [DOI] [PubMed] [Google Scholar]
- Feldman DE. Synaptic mechanisms for plasticity in neocortex. Annual Review of Neuroscience. 2009;32:33–55. doi: 10.1146/annurev.neuro.051508.135516. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Feller MB, Scanziani M. A precritical period for plasticity in visual cortex. Current Opinion in Neurobiology. 2005;15:94–100. doi: 10.1016/j.conb.2005.01.012. [DOI] [PubMed] [Google Scholar]
- Gall AJ, Todd WD, Ray B, Coleman CM, Blumberg MS. The development of day-night differences in sleep and wakefulness in Norway rats and the effect of bilateral enucleation. Journal of Biological Rhythms. 2008;23:232–241. doi: 10.1177/0748730408316954. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ganguly K, Schinder AF, Wong ST, Poo M. GABA itself promotes the developmental switch of neuronal GABAergic responses from excitation to inhibition. Cell. 2001;105:521–532. doi: 10.1016/s0092-8674(01)00341-5. [DOI] [PubMed] [Google Scholar]
- Gramsbergen A, Schwartze P, Prechtl HFR. The postnatal development of behavioral states in the rat. Developmental Psychobiology. 1970;3:267–280. doi: 10.1002/dev.420030407. [DOI] [PubMed] [Google Scholar]
- Hanganu IL, Staiger JF, Ben-Ari Y, Khazipov R. Cholinergic modulation of spindle bursts in the neonatal rat visual cortex in vivo. Journal of Neuroscience. 2007;27:5694–5705. doi: 10.1523/JNEUROSCI.5233-06.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hensch TK. Critical period plasticity in local cortical circuits. Nature Reviews Neuroscience. 2005;6:877–888. doi: 10.1038/nrn1787. [DOI] [PubMed] [Google Scholar]
- Innocenti GM, Price DJ. Exuberance in the development of cortical networks. Nature Reviews Neuroscience. 2005;6:955–965. doi: 10.1038/nrn1790. [DOI] [PubMed] [Google Scholar]
- Ivy GO, Killackey HP. The ontogeny of the distribution of callosal projection neurons in the rat parietal cortex. Journal of Comparative Neurology. 1981;195:367–389. doi: 10.1002/cne.901950302. [DOI] [PubMed] [Google Scholar]
- Jones MS, Barth DS. Effects of bicuculline methiodide on fast (>200 Hz) electrical oscillations in rat somatosensory cortex. Journal of Neurophysiology. 2002;88:1016–1025. doi: 10.1152/jn.2002.88.2.1016. [DOI] [PubMed] [Google Scholar]
- Juliano SL, Ma W, Eslin D. Cholinergic depletion prevents expansion of topographic maps in somatosensory cortex. Proceedings of the National Academy of Sciences of the United States of America. 1991;88:780–784. doi: 10.1073/pnas.88.3.780. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kanold PO. Subplate neurons: crucial regulators of cortical development and plasticity. Frontiers in Neuroanatomy. 2009;3:1–9. doi: 10.3389/neuro.05.016.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson KÆ, Blumberg MS. The union of the state: Myoclonic twitching is coupled with nuchal muscle atonia in infant rats. Behavioral Neuroscience. 2002;116:912–917. doi: 10.1037//0735-7044.116.5.912. [DOI] [PubMed] [Google Scholar]
- Karlsson KÆ, Gall AJ, Mohns EJ, Seelke AMH, Blumberg MS. The neural substrates of infant sleep in rats. PLoS Biology. 2005;3:891–901. doi: 10.1371/journal.pbio.0030143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karlsson KÆ, Kreider JC, Blumberg MS. Hypothalamic contribution to sleep- wake cycle development. Neuroscience. 2004;123:575–582. doi: 10.1016/j.neuroscience.2003.09.025. [DOI] [PubMed] [Google Scholar]
- Katz LC, Shatz CJ. Synaptic activity and the construction of cortical circuits. Science. 1996;274:1133–1138. doi: 10.1126/science.274.5290.1133. [DOI] [PubMed] [Google Scholar]
- Khazipov R. GABA and early patterns in vivo. INMED Satellite Conference of SFN; Chicago, IL. 2009. [Google Scholar]
- Khazipov R, Sirota A, Leinekugel X, Holmes GL, Ben-Ari Y, Buzsaki G. Early motor activity drives spindle-bursts in the developing somatosensory cortex. Nature. 2004;432:758–761. doi: 10.1038/nature03132. [DOI] [PubMed] [Google Scholar]
- Kilgard MP, Merzenich MM. Cortical map reorganization enabled by nucleus basalis activity. Science. 1998;279:1714–1718. doi: 10.1126/science.279.5357.1714. [DOI] [PubMed] [Google Scholar]
- Kimura F, Baughman RW. GABAergic transcallosal neurons in developing rat neocortex. European Journal of Neuroscience. 1997;9:1137–1143. doi: 10.1111/j.1460-9568.1997.tb01467.x. [DOI] [PubMed] [Google Scholar]
- Kolb B, Halliwell C, Gibb R. Factors influencing neocortical development in the normal and injured brain. In: Blumberg MS, Freeman JH, Robinson SR, editors. The Oxford Handbook of Developmental Behavioral Neuroscience. New York: Oxford University Press; 2010. pp. 375–388. [Google Scholar]
- Kreider JC, Blumberg MS. Mesopontine contribution to the expression of active ‘twitch’ sleep in decerebrate week-old rats. Brain Research. 2000;872:149–159. doi: 10.1016/s0006-8993(00)02518-x. [DOI] [PubMed] [Google Scholar]
- Lamsa K, Palva JM, Ruusuvuori E, Kaila K, Taira T. Synaptic GABA(A) activation inhibits AMPA-kainate receptor-mediated bursting in the newborn (P0–P2) rat hippocampus. Journal of Neurophysiology. 2000;83:359–366. doi: 10.1152/jn.2000.83.1.359. [DOI] [PubMed] [Google Scholar]
- Ludwig A, Li H, Saarma M, Kaila K, Rivera C. Developmental up-regulation of KCC2 in the absence of GABAergic and glutamatergic transmission. European Journal of Neuroscience. 2003;18:3199–3206. doi: 10.1111/j.1460-9568.2003.03069.x. [DOI] [PubMed] [Google Scholar]
- Ma W, Hohmann CF, Coyle JT, Juliano SL. Lesions of the basal forebrain alter stimulus-evoked metabolic activity in mouse somatosensory cortex. Journal of Comparative Neurology. 1989;288:414–427. doi: 10.1002/cne.902880305. [DOI] [PubMed] [Google Scholar]
- Makarov VA, Schmidt KE, Castellanos NP, Lopez-Aguado L, Innocenti GM. Stimulus-dependent interaction between the visual areas 17 and 18 of the 2 hemispheres of the ferret (Mustela putorius) Cerebral Cortex. 2008;18:1951–1960. doi: 10.1093/cercor/bhm222. [DOI] [PubMed] [Google Scholar]
- Marcano-Reik AJ, Blumberg MS. The corpus callosum modulates spindle-burst activity within homotopic regions of somatosensory cortex in newborn rats. European Journal of Neuroscience. 2008;28:1457–1466. doi: 10.1111/j.1460-9568.2008.06461.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marmigere F, Rage F, Tapia-Arancibia L. GABA-glutamate interaction in the control of BDNF expression in hypothalamic neurons. Neurochemistry International. 2003;42:353–358. doi: 10.1016/s0197-0186(02)00100-6. [DOI] [PubMed] [Google Scholar]
- Marty S, Berzaghi Mda P, Berninger B. Neurotrophins and activity-dependent plasticity of cortical interneurons. Trends in Neurosciences. 1997;20:198–202. doi: 10.1016/s0166-2236(96)01026-0. [DOI] [PubMed] [Google Scholar]
- Minlebaev M, Ben-Ari Y, Khazipov R. Network mechanisms of spindle-burst oscillations in the neonatal rat barrel cortex in vivo. Journal of Neurophysiology. 2007;97:692–700. doi: 10.1152/jn.00759.2006. [DOI] [PubMed] [Google Scholar]
- Mizuno H, Hirano T, Tagawa Y. Evidence for activity-dependent cortical wiring: formation of interhemispheric connections in neonatal mouse visual cortex requires projection neuron activity. Journal of Neuroscience. 2007;27:6760–6770. doi: 10.1523/JNEUROSCI.1215-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohns EJ, Blumberg MS. Synchronous bursts of neuronal activity in the developing hippocampus: Modulation by active sleep and association with emerging gamma and theta rhythms. Journal of Neuroscience. 2008;28:10134–10144. doi: 10.1523/JNEUROSCI.1967-08.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mohns EJ, Blumberg MS. Neocortical activation of the hippocampus during sleep in newborn rats. Journal of Neuroscience. 2010;30:3438–3449. doi: 10.1523/JNEUROSCI.4832-09.2010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens DF, Boyce LH, Davis MB, Kriegstein AR. Excitatory GABA responses in embryonic and neonatal cortical slices demonstrated by gramicidin perforated-patch recordings and calcium imaging. Journal of Neuroscience. 1996;16:6414–6423. doi: 10.1523/JNEUROSCI.16-20-06414.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Owens DF, Kriegstein AR. Is there more to GABA than synaptic inhibition? Nature Reviews Neuroscience. 2002;3:715–727. doi: 10.1038/nrn919. [DOI] [PubMed] [Google Scholar]
- Paul LK, Brown WS, Adolphs R, Tyszka JM, Richards LJ, Mukherjee P, Sherr EH. Agenesis of the corpus callosum: genetic, developmental and functional aspects of connectivity. Nature Reviews Neuroscience. 2007;8:287–299. doi: 10.1038/nrn2107. [DOI] [PubMed] [Google Scholar]
- Petersson P, Waldenström A, Fåhraeus C, Schouenborg J. Spontaneous muscle twitches during sleep guide spinal self-organization. Nature. 2003;424:72–75. doi: 10.1038/nature01719. [DOI] [PubMed] [Google Scholar]
- Rasmusson DD, Dykes RW. Long-term enhancement of evoked potentials in cat somatosensory cortex produced by co-activation of the basal forebrain and cutaneous receptors. Experimental Brain Research. 1988;70:276–286. doi: 10.1007/BF00248353. [DOI] [PubMed] [Google Scholar]
- Rivera C, Voipio J, Kaila K. Two developmental switches in GABAergic signalling: the K+-Cl- cotransporter KCC2 and carbonic anhydrase CAVII. Journal of Physiology. 2005;562:27–36. doi: 10.1113/jphysiol.2004.077495. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Roffwarg HP, Muzio JN, Dement WC. Ontogenetic development of the human sleep-dream cycle. Science. 1966;152:604–619. doi: 10.1126/science.152.3722.604. [DOI] [PubMed] [Google Scholar]
- Seelke AM, Blumberg MS. Developmental appearance and disappearance of cortical events and oscillations in infant rats. Brain Research. 2010;1324:34–42. doi: 10.1016/j.brainres.2010.01.088. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seelke AMH, Blumberg MS. Thermal and nutritional modulation of sleep in infant rats. Behavioral Neuroscience. 2005;19:603–611. doi: 10.1037/0735-7044.119.2.603. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seelke AMH, Karlsson KÆ, Gall AJ, Blumberg MS. Extraocular muscle activity, rapid eye movements, and the development of active and quiet sleep. European Journal of Neuroscience. 2005;22:911–920. doi: 10.1111/j.1460-9568.2005.04322.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sipilä ST, Blaesse P, Kaila K. Development of GABAergic signaling: From molecules to emerging networks. In: Blumberg MS, Freeman JH, Robinson SR, editors. The Oxford Handbook of Developmental Behavioral Neuroscience. New York: Oxford University Press; 2010. pp. 108–139. [Google Scholar]
- Tremblay N, Warren RA, Dykes RW. Electrophysiological studies of acetylcholine and the role of the basal forebrain in the somatosensory cortex of the cat. II. Cortical neurons excited by somatic stimuli. Journal of Neurophysiology. 1990;64:1212–1222. doi: 10.1152/jn.1990.64.4.1212. [DOI] [PubMed] [Google Scholar]
- Turrigiano GG. Homeostatic plasticity in neuronal networks: the more things change, the more they stay the same. Trends in Neurosciences. 1999;22:221–227. doi: 10.1016/s0166-2236(98)01341-1. [DOI] [PubMed] [Google Scholar]
- Wang C, Shimizu-Okabe C, Watanabe K, Okabe A, Matsuzaki H, Ogawa T, Mori N, Fukuda A, Sato K. Developmental changes in KCC1, KCC2, and NKCC1 mRNA expressions in the rat brain. Developmental Brain Research. 2002;139:59–66. doi: 10.1016/s0165-3806(02)00536-9. [DOI] [PubMed] [Google Scholar]
- Wang CL, Zhang L, Zhou Y, Zhou J, Yang XJ, Duan SM, Xiong ZQ, Ding YQ. Activity-dependent development of callosal projections in the somatosensory cortex. Journal of Neuroscience. 2007;27:11334–11342. doi: 10.1523/JNEUROSCI.3380-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wise SP, Jones EG. The organization and postnatal development of the commissural projection of the rat somatic sensory cortex. Journal of Comparative Neurology. 1976;168:313–343. doi: 10.1002/cne.901680302. [DOI] [PubMed] [Google Scholar]
- Wong M, Yamada KA. Developmental characteristics of epileptiform activity in immature rat neocortex: a comparison of four in vitro seizure models. Developmental Brain Research. 2001;128:113–120. doi: 10.1016/s0165-3806(01)00149-3. [DOI] [PubMed] [Google Scholar]
- Wong RO. Retinal waves and visual system development. Annual Review of Neuroscience. 1999;22:29–47. doi: 10.1146/annurev.neuro.22.1.29. [DOI] [PubMed] [Google Scholar]
- Xiong H, Futamura T, Jourdi H, Zhou H, Takei N, Diverse-Pierluissi M, Plevy S, Nawa H. Neurotrophins induce BDNF expression through the glutamate receptor pathway in neocortical neurons. Neuropharmacology. 2002;42:903–912. doi: 10.1016/s0028-3908(02)00043-6. [DOI] [PMC free article] [PubMed] [Google Scholar]