Abstract
Monocyte-derived myeloid cells play vital roles in inflammation-related autoimmune/inflammatory diseases and cancers. Here, we report that exosomes can deliver anti-inflammatory agents, such as curcumin, to activated myeloid cells in vivo. This technology provides a means for anti-inflammatory drugs, such as curcumin, to target the inflammatory cells as well as to overcome unwanted off-target effects that limit their utility. Using exosomes as a delivery vehicle, we provide evidence that curcumin delivered by exosomes is more stable and more highly concentrated in the blood. We show that the target specificity is determined by exosomes, and the improvement of curcumin activity is achieved by directing curcumin to inflammatory cells associated with therapeutic, but not toxic, effects. Furthermore, we validate the therapeutic relevance of this technique in a lipopolysaccharide (LPS)-induced septic shock mouse model. We further show that exosomes, but not lipid alone, are required for the enhanced anti-inflammatory activity of curcumin. The specificity of using exosomes as a drug carrier creates opportunities for treatments of many inflammation-related diseases without significant side effects due to innocent bystander or off-target effects.
Introduction
The treatment of inflammatory disorders must rely on targets that are present in both healthy and diseased tissues. Achieving therapeutic effects on the inflammatory cells often requires high drug doses that can induce unwanted effects on other tissues. Thus, although the current target-based drug design paradigm efficiently finds candidate drugs that are selective in a molecular sense, cell-type specificity is lacking that prevents their use in vivo at effective doses.
Monocyte-derived myeloid cells represent a large pool of scavenger and potential effector cells during inflammatory processes.1,2,3,4 Uncontrolled activation of myeloid cells leads to chronic inflammation. The process of myeloid cell–mediated inflammation has been shown to play a major role in most chronic illnesses, including neurodegenerative,5,6,7 cardiovascular,8,9,10,11,12 pulmonary,13,14,15 metabolic,15,16,17 autoimmune,18,19,20,21,22 and neoplastic diseases.23,24,25,26,27,28
Exosomes released from many different types of cells can transfer information to recipient cells. Activated monocyte-derived myeloid cells act as scavengers and have a high capacity for taking up vesicles, like exosomes circulating in the peripheral blood, through their phagocytic function.29,30,31,32,33
Curcumin, a natural polyphenol found in the rhizomes of Curcuma longa (turmeric), exhibits anti-inflammatory, antineoplastic, antioxidant, and chemopreventive activity.34,35,36,37,38 Several clinical trials dealing with cancers have addressed the pharmacokinetics, safety, and efficacy of curcumin in humans.36 Despite extensive research and development, poor solubility of curcumin, due to its hydrophobic property and preferential interaction with lipid membranes, remains a major barrier in its bioavailability and clinical efficacy.39 To increase its solubility and bioavailability, attempts have been made through encapsulation in liposomes, polymeric nanoparticles, biodegradable microspheres, cyclodextrin, and hydrogels.40,41,42,43,44,45,46,47,48,49
In this study, we show that exosomes released from a number of different cell types form a complex with curcumin. Mice treated with curcumin complexed with exosomes are protected against lipopolysaccharide (LPS)-induced septic shock. These data show that curcumin complexed with exosomes are taken up by activated monocyte-derived myeloid cells circulating in the peripheral blood and that curcumin subsequently induces apoptosis in these monocytes. In addition, the formation of exosome–curcumin complexes also leads to an increase in the stability of curcumin in vitro and bioavailability in vivo. We consider these findings to have significant impact on target-based drug development for successful in vivo drug delivery to treat inflammatory-related diseases.
Results
Incorporation of curcumin into exosomes
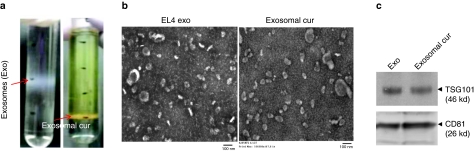
Exosomes are 30–100 nm nanoparticles secreted by cells into the extracellular environment. To exploit the possibility of nanoparticle exosomes as a carrier to entrap curcumin, curcumin was mixed with EL-4-derived exosomes at 22 °C, and then subjected to sucrose gradient centrifugation. A yellowish band (Figure 1a, right) appeared between the 45 and 60% sucrose gradients, with a weak band appeared between the 30 and 45% gradients (original exosomes, Figure 1a, left). The yellowish band was collected, washed and dissolved in phosphate-buffered saline (PBS). This fraction was designated exosomal curcumin. The morphology and the size of exosomal curcumin were similar to the original exosomes (Figure 1b). Exosomal protein markers, such as TSG101 and CD81 (Figure 1c), were identified in the exosomal curcumin. The binding capacity was estimated to be 2.9 g curcumin to 1 g exosomes. Exosomes isolated from other types of cell lines, including MDA-MB231 (human adenocarcinoma), 4T-1 (murine breast tumor cell line), and primary mouse embryonic fibroblasts, also have similar efficiency in terms of binding curcumin (data not shown).
Figure 1.
Encapsulation of curcumin into exosomes. (a) Exosomal curcumin purification. Five minutes after co-incubation of curcumin with EL-4 exosomes, the mixture was loaded on a sucrose gradient (8, 30, 45, and 60%, respectively) for centrifugation. A representative photo of exosomal curcumin is shown between the sucrose gradient layers of 45 and 60% (right, exosomal cur), instead of the 30 and 45% layers (left, original exosomes, Exo). (b) Electron microscopic images of EL-4 exosomes and exosomal curcumin. Purified exosomes and exosomal curcumin were imaged by a Hitachi H7000 electron microscope. Bar = 100 nm. (c) Western blot analysis of exosomes and exosomal curcumin protein expression. Equal amounts of exosomal protein and exosomal curcumin were loaded onto a 10% SDS-PAGE gel. TSG101 and CD81 expression were determined by blotting with monoclonal antibodies against TSG101 and CD81 (Santa Cruz, CA).
Incorporation of curcumin into exosomes can increase the solubility, stability, and bioavailability of curcumin
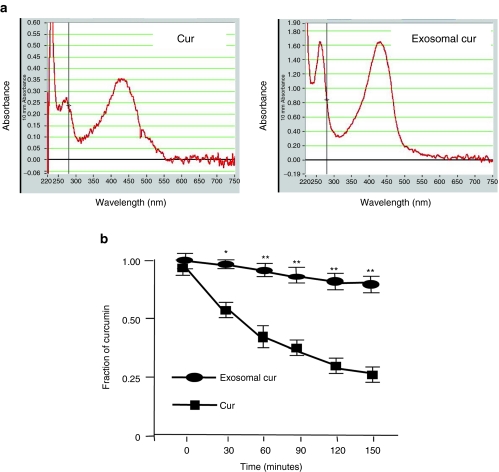
Curcumin is a hydrophobic polyphenol compound that is insoluble in aqueous solution. To determine whether the binding of curcumin to exosomes can increase curcumin's solubility, an identical amount of curcumin was mixed in an equal volume of PBS or exosomes in PBS, and the mixtures were placed on ice for 30 minutes. To estimate curcumin solubility, the concentration of curcumin in the supernatant was determined using a Nanodrop 1000 spectrophotometer. The curcumin concentration in the mixture of curcumin and exosomes was appropriately fivefold higher than curcumin alone (Figure 2a). Thus, the solubility of exosomal curcumin is higher than free curcumin and this is due to the binding of curcumin to exosomes.
Figure 2.
Encapsulation of curcumin into exosomes can increase curcumin's solubility and stability in vitro. (a) Exosomal curcumin is more soluble in phosphate-buffered saline (PBS) than free curcumin. An identical amount of curcumin was mixed with an equal volume of PBS or exosomes. Representative spectrographs of curcumin (Cur) and exosomal curcumin (exosomal cur) in the supernatant are shown, and the spectrographs were obtained using a Nanodrop 1000 spectrophotometer at OD420. (b) Exosomal curcumin is more stable than free curcumin. Curcumin and exosomal curcumin were added to 2 ml PBS to achieve a final concentration of 30 µmol/l and incubated in the dark in a 37 °C water bath. At 30, 60, 90, 120, and 150 minutes, 100 µl of each sample was taken to determine the concentration of curcumin. The concentrations of curcumin or exosomal curcumin at the beginning were set as 1.00. The fold reduction of the concentration at each time point compared to the beginning is shown. The experiments were repeated three times for each time point (*P < 0.05, **P < 0.01).
Curcumin is relatively unstable, and this is one of the major barriers for clinical use of curcumin to treat cancer and other inflammation-related diseases.15 To determine whether exosomal curcumin is more stable, free curcumin and exosomal curcumin were incubated at 37 °C over a period of 150 minutes and sampled periodically to determine the concentration of curcumin. After incubation for 150 minutes at 37 °C, we found that free curcumin in PBS degraded quickly and only 25% remained after the 150-minute incubation when compared to the 0-minute sample (set as 1.0). Curcumin in exosomal curcumin was protected from degradation with >80% remaining after the 150-minute incubation in PBS (pH 7.4, Figure 2b).
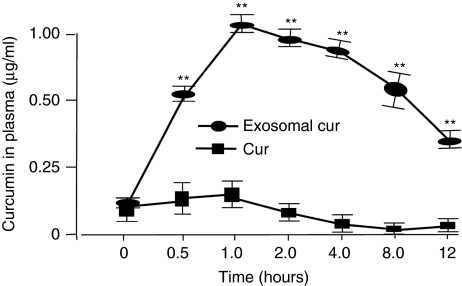
Another major barrier for clinical use of curcumin is its low systemic bioavailability.15 After administering orally, curcumin is digested in the stomach. Along with other food, curcumin then passes through the intestinal wall into the enterohepatic circulation, arriving in the liver for detoxification (the first-pass effect) and eventually gets into the blood stream. Curcumin's low bioavailability may be due to the rapid first-pass effect and the fast intestinal glucuronidation metabolism. To assess whether exosomal curcumin can increase bioavailability of curcumin, free curcumin, and exosomal curcumin (before purification, see Materials and Methods) was administered intraperitoneally (i.p.) or orally at a dose of 100 mg/kg of body weight. Due to the low bioavailability of curcumin, we used an extremely high dose of curcumin to achieve detectable curcumin in curcumin-treated mice. Curcumin in plasma was quantified at 0.5, 1, 2, 4, 8, and 12 hours after administration using an established high performance liquid chromatography (HPLC) method. Figure 3 and Supplementary Figure S1 show that at 30 minutes, i.p. administration of exosomal curcumin led to a five- to tenfold higher curcumin accumulated in peripheral blood than that of curcumin alone. At 12 hours after i.p. injection, curcumin in the plasma still remained at a much higher level in the group of mice injected with exosomal curcumin. In contrast, there was no detectable curcumin circulating in the blood of mice treated with curcumin alone. Similar results were obtained when mice were administrated with curcumin or exosomal curcumin orally (data not shown). Collectively, our data suggest that exosomal curcumin is stable in the circulation.
Figure 3.
Encapsulation of curcumin into exosomes can increase bioavailability of curcumin in vivo. Two groups (five per group) of C57BL/6j mice were injected i.p. with 100 mg curcumin or exosomal curcumin/kg of body weight. Thirty minutes, 1, 2, 4, 8, and 12 hours after injection, blood samples were taken, and the concentration of curcumin in the plasma was determined by high performance liquid chromatography as described in the Materials and Methods. Naive mice without any treatment were used as controls. A Tukey's pairwise comparison with analysis of variance was performed to determine the difference between the groups. Standard deviation (±SD) is presented as error bars. **P < 0.01.
Anti-inflammation activity of exosomal curcumin
Our data show that, as a nanoparticle drug carrier, exosomes can increase curcumin's solubility and stability in vitro, and bioavailability in vivo. We speculate that exosomal curcumin can enhance the anti-inflammatory activity of curcumin by accumulating curcumin to high levels in cellular targets. To evaluate the anti-inflammatory activity of exosomal curcumin in vitro, RAW 264.7 cells were treated with curcumin or exosomal curcumin at a concentration of 20 µmol/l for 1 hour. Subsequently, treated cells were stimulated with LPS (50 ng/ml) for an additional 6 hours. Cytokine production in the supernatant was measured 6 hours after treatment. As shown in Figure 4, exosomal curcumin–treated macrophages produced significantly less IL-6 and TNF-α in comparison with curcumin treatment alone.
Figure 4.
Encapsulated curcumin had a higher inhibitory effect on IL-6 and TNF-α secretion than free curcumin in vitro. RAW 264.7 cells were plated into 24-well plates and incubated overnight. The cells were then treated with curcumin or EL-4 exosomal curcumin at a concentration of 20 µmol/l for 1 hour and then stimulated with LPS (50 ng/ml) for an additional 6 hours. RAW 264.7 cells treated with PBS or exosomes served as controls. TNF-α and IL-6 in the culture supernatant were determined using a standard enzyme-linked immunosorbent assay. Data are represented as mean ± SEM (n = 6). *P < 0.05, **P < 0.01. LPS, lipopolysaccharide; PBS, phosphate-buffered saline.
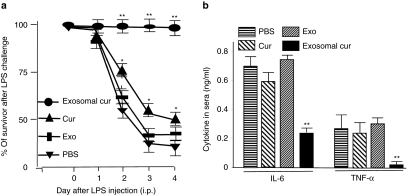
To assess the anti-inflammatory activity of exosomal curcumin in vivo, we adapted a LPS-induced septic shock model. To monitor mice mortality, a LD50 (median lethal dose) of LPS was determined first. LPS (5 mg/ml, Sigma-Aldrich) was prepared with sterile H2O, and varied amounts of LPS were i.p. injected into the same batch of commercially supplied C57BL/6j mice. Each group contained 10 mice. The LD50 was 18.75 mg of LPS/kg of body weight. C57BL/6j mice were i.p. injected with LPS (18.75 mg/kg) together with curcumin, or exosomal curcumin (4 mg/kg body weight) treatments. There was a significant survival advantage for mice treated with exosomal curcumin as compared to mice treated with an equivalent concentration of free curcumin over a 4-day period (Figure 5a). Exosomes and PBS injections serving as controls had no effects on LPS-induced mouse mortality. Sixteen hours after i.p. injection of LPS, the sera levels of IL-6 and TNF-α were similar in mice treated with free curcumin, exosomes, and PBS. However, both cytokines were significantly lower in the exosomal curcumin–treated group of mice (Figure 5b) and this finding correlated with mortality data.
Figure 5.
Exosomal curcumin can protect mice from LPS-induced septic shock. (a) Curcumin or exosomal curcumin (4 mg/kg) was injected i.p. into C57BL/6j mice together with LPS (18.5 mg/kg, Sigma-Aldrich). An equal amount of EL-4 exosomes used in the exosomal curcumin was used in the control. Mouse mortality was monitored over a period of 4 days. Data represented 10 mice/group and pooled from two experiments (**P < 0.01, *P < 0.05). (b) Exosomal curcumin can decrease IL-6 and TNF-α secretion in LPS-stimulated mice. The sera were collected 16 hours after LPS injection, and IL-6 and TNF-α concentrations in the sera were determined using an enzyme-linked immunosorbent assay (**P < 0.01).
Exosomal curcumin decreased CD11b+Gr-1+ cells in the lungs of mice
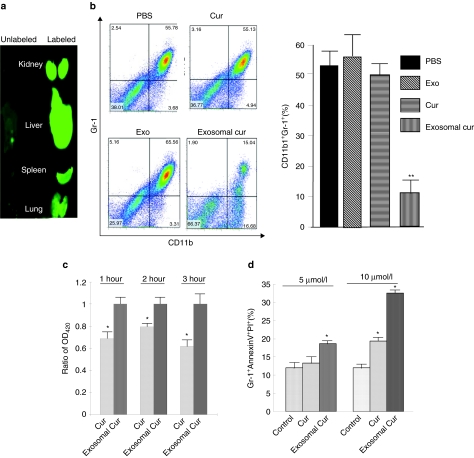
To further identify specific targeting of organs by exosomes, in vivo biodistribution of fluorescent dye IRDye800-labeled EL-4 exosomes was conducted in mice using Odyssey imaging. For these studies, we evaluated distribution i.p. injection of IRDye800-labeled EL-4 exosomes. Within an hour after injection, fluorescent signals were predominantly detected in liver, lung, kidney, and splenic tissues (Figure 6a, right panel). As we demonstrated before,32 flow cytometric analysis revealed that PKH67 fluorescent dye–labeled exosomes were taken up by CD11b+Gr-1+ cells (data not shown).
Figure 6.
Exosomal curcumin can downregulate the CD11b+Gr-1+ cell population during lipopolysaccharide (LPS)-induced inflammation. (a) Biodistribution of i.p.-injected EL-4 exosomes. A total of 100 µg of IRDye 800-labeled EL-4 exosomes or unlabeled EL-4 exosomes were injected i.p. into C57BL/6j mice. Each organ imaging was done at 1 hour after injection. (b) Exosomal curcumin treatment dramatically decreased the CD11b+Gr-1+ cell population in the lungs. At day 1 after LPS challenge, mice were killed, and the leukocytes in the lungs were isolated. The percentage of cells in the lung was determined by fluorescence-activated cell sorting (FACS) analysis, and representative analyses are shown (left). Statistical significance was analyzed as before (**P < 0.01, right). (c) Gr-1+ cells incorporated more exosomal curcumin than free curcumin. Gr-1+ cells were selected from bone marrow cells and then treated with the same concentration of free curcumin and exosomal curcumin. At 1, 2, and 3 hours, cells were collected and washed with PBS. Curcumin in the cells was extracted by methanol, and the concentration was determined using a Nanodrop 1000 spectrophotometer. At each time point, curcumin concentration in exosomal curcumin–treated cells was set as 1.0. The ratio of curcumin concentration in free curcumin–treated cells compared to exosomal curcumin–treated cells was reported. Triplicates of each sample were performed. *P < 0.05. (d) Exosomal curcumin induced more Gr-1+ cell death in culture. Gr-1+ cells were selected from bone marrow cells and then treated with free curcumin and exosomal curcumin. After 8 hours, cells were collected and immunostained with AnnexinV-FITC and PI. The percentage of AnnexinV+PI+ Gr-1+ cells was determined by FACS analysis. Triplicates of each sample were performed, and the significance compared to controls and analyzed as before (*P < 0.05).
One of the features of LPS-induced septic shock is a robust increase of the number of CD11b+Gr-1+ cells that are sequestered in the lungs leading to acute lung inflammation.26,27 In LPS-treated mice, there were significantly fewer CD11b+Gr-1+ cells in the lungs of the mice treated with exosomal curcumin when compared to mice treated with curcumin, exosomes, or PBS (Figure 6b). We and others have reported previously that tumor exosomes are taken up by CD11b+Gr-1+ cells circulating in the peripheral blood.25,28 To find whether exosomal curcumin can take advantage of this property, Gr-1+ cells were isolated from bone marrow cells and cocultured with curcumin, or exosomal curcumin at the same concentration of curcumin. At 1, 2, and 3 hours, cellular curcumin concentration was measured (Figure 6c); an increase in curcumin did not occur in cells treated with an equivalent amount of exosomes and curcumin together (data not shown). Interestingly, curcumin induces Gr-1+ cell apoptosis when Gr-1+ cells are treated with 5 and 10 µmol/l curcumin, and exosomal curcumin can significantly enhance this induction as determined by fluorescence-activated cell sorting (FACS) analysis (Figure 6d). Gr-1+ cells treated with exosomes alone had a similar apoptosis percentage as the controls (data not shown).
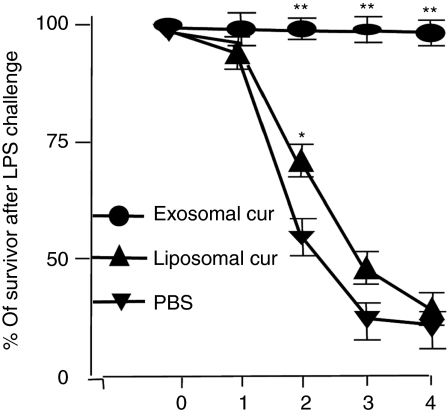
To determine whether the exosomes are functioning as a delivery vehicle or are contributing to the therapeutic effects, an in vivo liposome curcumin treatment of LPS-challenged mice was conducted. The results indicate that mice treated with exosomal curcumin have much lower mortality than mice treated with an equivalent concentration of liposome curcumin (Figure 7). The inefficiency of liposome curcumin to prevent LPS-induced septic shock is most likely not due to an insufficient inhibition of anti-inflammatory pathways because liposome curcumin treatment of bone marrow–derived macrophages has a similar effect as exosomal curcumin on the prevention of IκBα degradation as well as prevention of Cox2 induction after LPS stimulation (Supplementary Figure S2). These data suggest that exosomes are most likely not simply functioning as a curcumin carrier-like liposomes, The increase in curcumin uptake by Gr-1+ cells with an increase in cell apoptosis caused by exosomal curcumin treatment may be one of the mechanisms underlying protection of mice from LPS-induced septic death.
Figure 7.
Exosomes function as more than a curcumin carrier. Liposome curcumin or exosomal curcumin (4 mg/kg) was injected i.p. into C57BL/6j mice together with lipopolysaccharide (18.5 mg/kg, Sigma-Aldrich). An equal amount of PBS was used as diluents in the control. Mouse mortality was monitored over a period of 4 days. Data represented six mice/group and were pooled from two experiments (**P < 0.01, *P < 0.05).
Discussion
In this study, we provide evidence that naturally existing nanoparticle exosomes can carry and deliver curcumin that in turn enhances its anti-inflammatory activity through (i) increasing the solubility, stability, and bioavailability of curcumin and (ii) enhancing/increasing delivery of curcumin to activated monocytes. Our approach leads to the protection of mice from LPS-induced septic shock. Furthermore, we have extended previous knowledge by demonstrating that exosomes target not only CD11b+Gr-1+ cells in the peripheral circulation but also enhance/increase delivery of exosomal curcumin to CD11b+Gr-1+ cells thus inducing more cell death. Finally, unlike other nonhost delivery vehicles, host-derived exosomes have an advantage as a potential delivery vehicle because they would not induce an immune response with subsequent side effects.
CD11b+Gr-1+ cells are one of the major cellular populations associated with disease pathogenesis. Accumulation CD11b+Gr-1+ cells can suppress host immune responses and interrupt immunosurveillance, which could provide an explanation as to why long-term inflammation promotes tumor progression. Exosome-directed curcumin targeting to CD11b+Gr-1+ cells may provide a means to treat inflammation-related diseases, and perhaps even cancers.
Our data indicate that encapsulation of curcumin into exosomes can increase solubility, stability, and bioavailability of curcumin. There are two ways to load drugs into nanoparticles: (i) physical entrapment and (ii) chemical conjugation. Exosomes, containing a lipid bilayer, which is similar to liposomal nanoparticles, may load curcumin through physical entrapment. Through the hydrophobic interaction between the hydrophobic tails and hydrophobic drug, curcumin can be self-assembled into the lipid bilayer of exosomes and this may protect curcumin from degradation. Nanoparticles are distributed to organs in a size-dependent selective manner.46 Sizes <5 nm nanoparticles are preferentially distributed to kidney and liver. Larger size exosomes likely stay in the vasculature for an extended time. Distribution of this nature may favor exosome-drug combinations, such as curcumin, being delivered as “brick dust–candidate” drugs. “Brick-dust candidates” are small molecule drugs with poor water solubility. When combined with exosomes, an increase in curcumin water solubility and stability is achieved, resulting in better delivery to the blood stream and increased bioavailability. This observation is also supported by data published by other groups, demonstrating that increased solubility and stability can be achieved by packing curcumin into liposomes or phospholipids.40,41
The present study is fundamental research on an exosomal curcumin drug delivery system that could be a milestone for a new powerful drug delivery system. The use of exosomal curcumin may take advantage of functional properties of curcumin, exosomes, and nanoparticles in an additive way to fight various inflammation related diseases. Curcumin has been well documented as a potent anti-inflammatory, antioxidant, and anticancer reagent through regulation of multiple pathways.35,37,50 Although our data show that exosomal curcumin enhances apoptosis of CD11b+Gr-1+ cells, additional mechanisms underlying the protection of mice against LPS-induced septic shock could also play a role because curcumin modulates a number of pathways. For an example, curcumin regulates the activity of a number of crucial transcription factors, including NF-κB, STAT3, and Nrf2, and therefore their biological effects on cell proliferation, apoptosis, cytokine induction, and antioxidation may also contribute to the prevention of LPS-induced septic shock. Second, unlike liposome as a carrier, exosomes, such as dendritic and tumor cell–derived exosomes, exhibit strong tendencies to regulate immune responses and tumor progression. Therefore, selecting different types of exosomes in combination with therapeutic drugs achieves targetable and additive/syngeneic therapeutic effects. Third, because most diseases including cancer involve dysregulation of multiple pathways, exosomes can carry multiple therapeutic drugs to modulate the activity of multiple pathways in the same targeted cells. It is conceivable that using exosomes as a delivery vehicle has advantages over other carrier systems in terms of modulation of multiple pathways simultaneously in the targeted cells.
Materials and Methods
Mice. In this study, 7- to 10-week female C57BL/6j mice (Jackson Laboratory, Bar Harbor, ME) were used. All animal studies were conducted within the guidelines established by the Institutional Animal Care and Use Committee of the University of Alabama at Birmingham.
Chemicals and reagents. Curcumin and LPS were purchased from Sigma-Aldrich (St Louis, MO). Liposome curcumin was purchased from Encapsula NanoSciences (Nashville, TN).
Cell culture. The EL-4 (mouse lymphoma cell line) and RAW 264.7 (murine macrophage cell line) were maintained in vitro at 37 °C in a humidified 5% CO2 atmosphere in air with complete RPMI 1640 and DMEM medium (supplemented with 10% fetal bovine serum). Fetal bovine serum used in cell cultures to isolate exosomes was exosome-depleted prior to use by differential centrifugation using a method described previously.29
Preparation of exosomes. The cell culture supernatants were collected and used for exosome purification by differential centrifugation using a previously described method.22 Purity and integrity of sucrose gradient–isolated exosomes was analyzed using a Hitachi H7000 electron microscope (Electronic Instruments, Akishima, Japan) as previously described.29 The concentration of exosomes was determined by analyzing protein concentration using the Bio-Rad protein quantitation assay kit (Bio-Rad, Hercules, CA) with bovine serum albumin as a standard. The protein expression of exosomes was determined by western blotting analysis as described previously.29
Quantification of circulating exosomal curcumin. Plasma (200 µl) collected from mice injected i.p. with curcumin, exosomal curcumin, exosomes, or PBS were diluted to 5 ml with PBS and centrifuged at 4,000 g for 30 minutes at 4 °C. The supernatants were then transferred to 15 ml tubes and further diluted to 10 ml with cold PBS, and centrifuged at 10,000 g for 1 hour at 4 °C to remove large debris. The samples were then transferred to ultracentrifuge tubes, and exosomes were washed twice at 100,000 g for 2 hours (SW41Ti rotor; Beckman Coulter, Brea, CA). Following two washes, exosomes were resuspended in 40 µl PBS for quantification of curcumin by HPLC analysis.
Preparation of exosomal curcumin. Exosomal curcumin was prepared by mixing curcumin with EL-4 exosomes in PBS. After incubation at 22 °C for 5 minutes, the mixture was subjected to sucrose gradient (8, 30, 45, and 60%, respectively) centrifugation for 1.5 hours at 36,000 rpm. The exosomal curcumin, distinguished as a yellowish band in the sucrose gradient between 45 and 60%, was subsequently collected, washed, and dissolved with PBS. The concentration of exosomes and curcumin in the complex was determined as described. Based on the morphology and protein expression, the vesicles were determined to be exosomes.29
Analysis of curcumin concentration in vitro and in vivo. The in vitro concentration of curcumin in samples was determined using a Nanodrop 1000 spectrophotometer (Thermo Scientific, Wilmington, DE) at 420 nm. Briefly, to evaluate the concentration of curcumin, a standard curve of curcumin was plotted first. A stock solution of curcumin was diluted to a range of 5–50 µmol/l. A standard calibration curve was obtained by plotting the concentration of standard curcumin versus fluorescent absorbance at 420 nm (OD420). The curcumin quantity in cell culture supernatant or PBS was calculated based on the OD420 with respect to the concentration of curcumin in the diluted standards.
To determine the concentration of curcumin in plasma, a standard curve of curcumin was plotted first. Briefly, a stock solution of curcumin (0.5 mg/ml) in acetonitrile was diluted to a range of 0.1–5 µg/ml with acetonitrile, and then 10 µl of diluted curcumin were added to 90 µl of plasma isolated from naive C57BL/6j mice (a range of 1–500 ng/ml). The mixture was added to an equal volume of emodin (0.15 µg/ml, Sigma-Aldrich) and vortexed for 5 minutes at 22 °C. After centrifugation at 2500 g for 15 minutes to remove precipitated plasma proteins, 50 µl of each of the working solutions containing 1–500 ng/ml of curcumin was analyzed by HPLC. The chromatographic separation was performed on a C18 column (5 µm, 250 × 4.6 mm; AAPPTec, Louisville, KY) with the mobile phase composed of acetonitrile-5% acetic acid (75:25, vol/vol) at a flow rate of 1.0 ml/minute. The wavelength of detection was at 420 nm. A standard calibration curve was obtained by plotting the concentration of standard curcumin versus absorbance units.
To determine the concentration of curcumin in the samples, plasma samples collected from mice treated with exosomal curcumin or free curcumin were precipitated with emodin to remove proteins and analyzed using an identical method as described above. The concentration was calculated using the absorbance units with respect to the concentration of curcumin in the standard curve.
FACS analysis. For cell surface marker staining, isolated cells were blocked at 4 °C for 5 minutes with 10 µg/ml mouse Fc block (BD Biosciences, San Jose, CA) and then reacted with various fluochrome-labeled antibodies including appropriate isotype controls for 30 minutes at 4 °C. After washing twice, cells were fixed and analyzed using a FACSCalibur flow cytometer (BD Biosciences). Data were analyzed using FlowJo software (TreeStar, Ashland, OR). The following antibodies were used for immunostaining: FITC-AnnexinV (Invitrogen, Carlsbad, CA), APC anti-mouse CD11b and PE anti-mouse Gr-1 (eBiosciences, San Diego, CA), and propidium iodide (Sigma-Aldrich) was used for staining apoptotic cells.
In vitro stability assays. To determine the stability of free curcumin and exosomal curcumin in PBS (pH 7.4), curcumin and exosomal curcumin were added to 2 ml PBS to achieve a final concentration of 30 µmol/l and incubated in the dark in a 37 °C water bath. At different time points, 100 µl of each sample were taken to determine the concentration of curcumin. The concentrations of curcumin or exosomal curcumin at the beginning were considered as 1.00. The fold reduction of the concentration at each time was determined by comparison to the beginning value. The experiments were repeated three times for each time point (n = 3).
In vivo bioavailability assays. To determine the bioavailability of free curcumin and exosomal curcumin in vivo, two groups (five per group) of C57BL/6j mice were i.p. injected or administrated orally with 100 mg curcumin or exosomal curcumin/kg body weight. (To achieve a higher dose of curcumin treatment, curcumin prebinding with an appropriate amount of EL-4 exosomes was considered as exosomal curcumin). At 0.5, 1, 2, 4, 8, and 12 hours, blood samples were taken through eye sinus bleeding, and the concentration of curcumin in the plasma was determined by HPLC as described above. Naive mice without treatment were used as blank controls.
In vitro proinflammatory cytokine induction assays. RAW 264.7 cells were plated in 24-well plates and incubated overnight. The cells were treated with curcumin or exosomal curcumin at a concentration of 20 µmol/l for 1 hour and then stimulated with LPS (50 ng/ml) for an additional 6 hours. RAW 264.7 cells treated with PBS or exosomes served as controls. TNF-α and IL-6 levels in the cell culture supernatant were measured using a standard enzyme-linked immunosorbent assay (eBiosciences).
LPS mouse septic shock model. Curcumin or exosomal curcumin (4 mg/kg of body weight) was injected i.p. into C57BL/6j mice together with LPS (18.5 mg/kg, Sigma-Aldrich). EL-4 exosomes equal to the amount in exosomal curcumin and PBS were used as controls. Mouse mortality was monitored over a period of 4 days. The sera were collected 16 hours after LPS injection and used to determine IL-6 and TNF-α levels using an enzyme-linked immunosorbent assay as before. At day 1 after LPS challenge, three mice from each treated group were killed, and the leukocytes in the lungs were isolated using a method described previously.32 The percentage of CD11b+Gr-1+ cells in the lung was determined by FACS analysis.
Isolation of Gr-1+ cells from mouse bone marrow cells. Mouse bone marrow cells were isolated as described previously.39 The isolated bone marrow cells were resuspended to a concentration of 1 × 108 cells/ml using RPMI 1640 medium supplemented with 10% fetal bovine serum. Following the addition of a mouse FcR blocking specific antibody (5 µl/ml), a Gr-1-PE conjugated antibody (3 µg/ml) was added, mixed thoroughly and incubated at 4 °C for 15 minutes. After centrifugation at 1,500 rpm for 5 minutes, cells were resuspended in fresh medium to a concentration of 1 × 108 cells/ml. EasySep PE selection cocktail (25 µl/ml; StemCell Technologies, Seattle, WA), was added to the cells and incubated at 4 °C for 15 minutes. Afterward, EasySep magnetic nanoparticles (25 µl/ml) were added and incubated at 4 °C for another 15 minutes. Culture medium was added to a final volume of 2.5 ml and the cells mixed by gentle pipetting of the mixture 2–3 times. The uncapped polystyrene tube was placed into the EasySep magnet and set aside for 5 minutes. The supernatant containing unbound cells was removed leaving the magnetically bound Gr-1+ cells. A second round of magnetic separation was done on the supernatant. Positively selected (magnetically bound) cells were collected from the tubes, counted and cultured in RPMI 1640 supplemented with MCSF (20 ng/ml) for curcumin uptake and apoptosis assays.
Labeling of exosomes. Exosomes were labeled using an odyssey fluorescent dye IRDye800 kit (LI-COR Biosciences, Lincoln, NE). To localize EL-4 exosomes injected i.p. into mice, the IRDye 800CW-labeled EL-4 exosomes (100 µg) were injected i.p. into C57BL/6j mice, and mice were imaged over a 48-hour period using a prototype LI-COR imager (LI-COR Biosciences). Mice (five mice/group) receiving nonlabeled EL-4 exosomes served as controls.
Western blot analysis. C57BL/6 mouse bone marrow–derived macrophages were stimulated with LPS (50 ng/ml) or PBS for 15 minutes. Equal amounts of protein extract (50 µg of total protein) were electrophoresed on a 10% polyacrylamide gel and transferred to a nitrocellulose membrane. The western blot was carried out with the anti-IκBα polyclonal antibody or anti-Cox2 or β-actin antibody (Santa Cruz Biotechnology, Santa Cruz, CA).
Statistical analysis. Statistical differences between groups were determined by analysis of variance with multiple comparisons using Fisher's post hoc analysis. The Student's t-test was used for comparisons when only two parameters were evaluated. P < 0.05 was considered significant.
SUPPLEMENTARY MATERIAL Figure S1. Exosomal curcumin is more stable than free curcumin in vivo. Figure S2. Liposomal curcumin has equal effect as curcumin or exosomal curcumin on the in vitro inhibition of IκB-α degradation.
Acknowledgments
This work was supported by grants from the National Institutes of Health (NIH) (RO1CA116092, RO1CA107181, RO1CA137037, and R01AT004294); Birmingham Veterans Administration Medical Center (VAMC) Merit Review grants (H.-G.Z.); and a grant from the Susan G. Komen Breast Cancer Foundation. We thank Jerald Ainsworth for editorial assistance.
Supplementary Material
Exosomal curcumin is more stable than free curcumin in vivo.
Liposomal curcumin has equal effect as curcumin or exosomal curcumin on the in vitro inhibition of IκB-α degradation.
REFERENCES
- Iwamoto S, Iwai S, Tsujiyama K, Kurahashi C, Takeshita K, Naoe M, et al. TNF-alpha drives human CD14+ monocytes to differentiate into CD70+ dendritic cells evoking Th1 and Th17 responses. J Immunol. 2007;179:1449–1457. doi: 10.4049/jimmunol.179.3.1449. [DOI] [PubMed] [Google Scholar]
- Jongstra-Bilen J, Haidari M, Zhu SN, Chen M, Guha D., and, Cybulsky MI. Low-grade chronic inflammation in regions of the normal mouse arterial intima predisposed to atherosclerosis. J Exp Med. 2006;203:2073–2083. doi: 10.1084/jem.20060245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Xu H, Manivannan A, Dawson R, Crane IJ, Mack M, Sharp P, et al. Differentiation to the CCR2+ inflammatory phenotype in vivo is a constitutive, time-limited property of blood monocytes and is independent of local inflammatory mediators. J Immunol. 2005;175:6915–6923. doi: 10.4049/jimmunol.175.10.6915. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Taylor EL, Megson IL, Haslett C., and, Rossi AG. Nitric oxide: a key regulator of myeloid inflammatory cell apoptosis. Cell Death Differ. 2003;10:418–430. doi: 10.1038/sj.cdd.4401152. [DOI] [PubMed] [Google Scholar]
- Chen K, Huang J, Liu Y, Gong W, Cui Y., and, Wang JM. Synergy of TRIF-dependent TLR3 and MyD88-dependent TLR7 in up-regulating expression of mouse FPR2, a promiscuous G-protein-coupled receptor, in microglial cells. J Neuroimmunol. 2009;213:69–77. doi: 10.1016/j.jneuroim.2009.05.018. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pais TF, Figueiredo C, Peixoto R, Braz MH., and, Chatterjee S. Necrotic neurons enhance microglial neurotoxicity through induction of glutaminase by a MyD88-dependent pathway. J Neuroinflammation. 2008;5:43. doi: 10.1186/1742-2094-5-43. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sasahara M, Otani A, Oishi A, Kojima H, Yodoi Y, Kameda T, et al. Activation of bone marrow-derived microglia promotes photoreceptor survival in inherited retinal degeneration. Am J Pathol. 2008;172:1693–1703. doi: 10.2353/ajpath.2008.080024. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Croce K, Gao H, Wang Y, Mooroka T, Sakuma M, Shi C, et al. Myeloid-related protein-8/14 is critical for the biological response to vascular injury. Circulation. 2009;120:427–436. doi: 10.1161/CIRCULATIONAHA.108.814582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Soehnlein O., and, Weber C. Myeloid cells in atherosclerosis: initiators and decision shapers. Semin Immunopathol. 2009;31:35–47. doi: 10.1007/s00281-009-0141-z. [DOI] [PubMed] [Google Scholar]
- Nygren JM, Liuba K, Breitbach M, Stott S, Thorén L, Roell W, et al. Myeloid and lymphoid contribution to non-haematopoietic lineages through irradiation-induced heterotypic cell fusion. Nat Cell Biol. 2008;10:584–592. doi: 10.1038/ncb1721. [DOI] [PubMed] [Google Scholar]
- Boyd JH, Kan B, Roberts H, Wang Y., and, Walley KR. S100A8 and S100A9 mediate endotoxin-induced cardiomyocyte dysfunction via the receptor for advanced glycation end products. Circ Res. 2008;102:1239–1246. doi: 10.1161/CIRCRESAHA.107.167544. [DOI] [PubMed] [Google Scholar]
- Beekhuizen H, Blokland I, Corsèl-van Tilburg AJ, Koning F., and, van Furth R. CD14 contributes to the adherence of human monocytes to cytokine-stimulated endothelial cells. J Immunol. 1991;147:3761–3767. [PubMed] [Google Scholar]
- Qu P, Du H, Li Y., and, Yan C. Myeloid-specific expression of Api6/AIM/Sp alpha induces systemic inflammation and adenocarcinoma in the lung. J Immunol. 2009;182:1648–1659. doi: 10.4049/jimmunol.182.3.1648. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Medoff BD, Seung E, Hong S, Thomas SY, Sandall BP, Duffield JS, et al. CD11b+ myeloid cells are the key mediators of Th2 cell homing into the airway in allergic inflammation. J Immunol. 2009;182:623–635. doi: 10.4049/jimmunol.182.1.623. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Deng ZB, Liu Y, Liu C, Xiang X, Wang J, Cheng Z, et al. Immature myeloid cells induced by a high-fat diet contribute to liver inflammation. Hepatology. 2009;50:1412–1420. doi: 10.1002/hep.23148. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Strom SS, Yamamura Y, Kantarijian HM., and, Cortes-Franco JE. Obesity, weight gain, and risk of chronic myeloid leukemia. Cancer Epidemiol Biomarkers Prev. 2009;18:1501–1506. doi: 10.1158/1055-9965.EPI-09-0028. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Nguyen MT, Favelyukis S, Nguyen AK, Reichart D, Scott PA, Jenn A, et al. A subpopulation of macrophages infiltrates hypertrophic adipose tissue and is activated by free fatty acids via toll-like receptors 2 and 4 and JNK-dependent pathways. J Biol Chem. 2007;282:35279–35292. doi: 10.1074/jbc.M706762200. [DOI] [PubMed] [Google Scholar]
- Tzima S, Victoratos P, Kranidioti K, Alexiou M., and, Kollias G. Myeloid heme oxygenase-1 regulates innate immunity and autoimmunity by modulating IFN-beta production. J Exp Med. 2009;206:1167–1179. doi: 10.1084/jem.20081582. [DOI] [PMC free article] [PubMed] [Google Scholar]
- King IL, Dickendesher TL., and, Segal BM. Circulating Ly-6C+ myeloid precursors migrate to the CNS and play a pathogenic role during autoimmune demyelinating disease. Blood. 2009;113:3190–3197. doi: 10.1182/blood-2008-07-168575. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dominguez-Soto A., and, Corbi AL. Myeloid dendritic cell lectins and their role in immune responses. Curr Opin Investig Drugs. 2007;8:910–920. [PubMed] [Google Scholar]
- Zhu B, Bando Y, Xiao S, Yang K, Anderson AC, Kuchroo VK, et al. CD11b+Ly-6C(hi) suppressive monocytes in experimental autoimmune encephalomyelitis. J Immunol. 2007;179:5228–5237. doi: 10.4049/jimmunol.179.8.5228. [DOI] [PubMed] [Google Scholar]
- McKinlay A, Radford K, Kato M, Field K, Gardiner D, Khalil D, et al. Blood monocytes, myeloid dendritic cells and the cytokines interleukin (IL)-7 and IL-15 maintain human CD4+ T memory cells with mixed helper/regulatory function. Immunology. 2007;120:392–403. doi: 10.1111/j.1365-2567.2006.02515.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ostrand-Rosenberg S., and, Sinha P. Myeloid-derived suppressor cells: linking inflammation and cancer. J Immunol. 2009;182:4499–4506. doi: 10.4049/jimmunol.0802740. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gonzalez-Mejia ME., and, Doseff AI. Regulation of monocytes and macrophages cell fate. Front Biosci. 2009;14:2413–2431. doi: 10.2741/3387. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bunt SK, Clements VK, Hanson EM, Sinha P., and, Ostrand-Rosenberg S. Inflammation enhances myeloid-derived suppressor cell cross-talk by signaling through toll-like receptor 4. J Leukoc Biol. 2009;85:996–1004. doi: 10.1189/jlb.0708446. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gabrilovich DI., and, Nagaraj S. Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol. 2009;9:162–174. doi: 10.1038/nri2506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sinha P, Clements VK, Fulton AM., and, Ostrand-Rosenberg S. Prostaglandin E2 promotes tumor progression by inducing myeloid-derived suppressor cells. Cancer Res. 2007;67:4507–4513. doi: 10.1158/0008-5472.CAN-06-4174. [DOI] [PubMed] [Google Scholar]
- Bunt SK, Sinha P, Clements VK, Leips J., and, Ostrand-Rosenberg S. Inflammation induces myeloid-derived suppressor cells that facilitate tumor progression. J Immunol. 2006;176:284–290. doi: 10.4049/jimmunol.176.1.284. [DOI] [PubMed] [Google Scholar]
- Xiang X, Poliakov A, Liu C, Liu Y, Deng ZB, Wang J, et al. Induction of myeloid-derived suppressor cells by tumor exosomes. Int J Cancer. 2009;124:2621–2633. doi: 10.1002/ijc.24249. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Iero M, Valenti R, Huber V, Filipazzi P, Parmiani G, Fais S, et al. Tumour-released exosomes and their implications in cancer immunity. Cell Death Differ. 2008;15:80–88. doi: 10.1038/sj.cdd.4402237. [DOI] [PubMed] [Google Scholar]
- Bhatnagar S, Shinagawa K, Castellino FJ., and, Schorey JS. Exosomes released from macrophages infected with intracellular pathogens stimulate a proinflammatory response in vitro and in vivo. Blood. 2007;110:3234–3244. doi: 10.1182/blood-2007-03-079152. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yu S, Liu C, Su K, Wang J, Liu Y, Zhang L, et al. Tumor exosomes inhibit differentiation of bone marrow dendritic cells. J Immunol. 2007;178:6867–6875. doi: 10.4049/jimmunol.178.11.6867. [DOI] [PubMed] [Google Scholar]
- Valenti R, Huber V, Iero M, Filipazzi P, Parmiani G., and, Rivoltini L. Tumor-released microvesicles as vehicles of immunosuppression. Cancer Res. 2007;67:2912–2915. doi: 10.1158/0008-5472.CAN-07-0520. [DOI] [PubMed] [Google Scholar]
- Ravindran J, Prasad S., and, Aggarwal BB. Curcumin and cancer cells: how many ways can curry kill tumor cells selectively. AAPS J. 2009;11:495–510. doi: 10.1208/s12248-009-9128-x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anand P, Thomas SG, Kunnumakkara AB, Sundaram C, Harikumar KB, Sung B, et al. Biological activities of curcumin and its analogues (Congeners) made by man and Mother Nature. Biochem Pharmacol. 2008;76:1590–1611. doi: 10.1016/j.bcp.2008.08.008. [DOI] [PubMed] [Google Scholar]
- Dhillon N, Aggarwal BB, Newman RA, Wolff RA, Kunnumakkara AB, Abbruzzese JL, et al. Phase II trial of curcumin in patients with advanced pancreatic cancer. Clin Cancer Res. 2008;14:4491–4499. doi: 10.1158/1078-0432.CCR-08-0024. [DOI] [PubMed] [Google Scholar]
- Anand P, Sundaram C, Jhurani S, Kunnumakkara AB., and, Aggarwal BB. Curcumin and cancer: an “old-age” disease with an “age-old” solution. Cancer Lett. 2008;267:133–164. doi: 10.1016/j.canlet.2008.03.025. [DOI] [PubMed] [Google Scholar]
- Aggarwal BB., and, Harikumar KB. Potential therapeutic effects of curcumin, the anti-inflammatory agent, against neurodegenerative, cardiovascular, pulmonary, metabolic, autoimmune and neoplastic diseases. Int J Biochem Cell Biol. 2009;41:40–59. doi: 10.1016/j.biocel.2008.06.010. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Anand P, Kunnumakkara AB, Newman RA., and, Aggarwal BB. Bioavailability of curcumin: problems and promises. Mol Pharm. 2007;4:807–818. doi: 10.1021/mp700113r. [DOI] [PubMed] [Google Scholar]
- Mulik R, Mahadik K., and, Paradkar A. Development of curcuminoids loaded poly(butyl) cyanoacrylate nanoparticles: physicochemical characterization and stability study. Eur J Pharm Sci. 2009;37:395–404. doi: 10.1016/j.ejps.2009.03.009. [DOI] [PubMed] [Google Scholar]
- Shaikh J, Ankola DD, Beniwal V, Singh D., and, Kumar MN. Nanoparticle encapsulation improves oral bioavailability of curcumin by at least 9-fold when compared to curcumin administered with piperine as absorption enhancer. Eur J Pharm Sci. 2009;37:223–230. doi: 10.1016/j.ejps.2009.02.019. [DOI] [PubMed] [Google Scholar]
- Narayanan NK, Nargi D, Randolph C., and, Narayanan BA. Liposome encapsulation of curcumin and resveratrol in combination reduces prostate cancer incidence in PTEN knockout mice. Int J Cancer. 2009;125:1–8. doi: 10.1002/ijc.24336. [DOI] [PubMed] [Google Scholar]
- Yadav VR, Suresh S, Devi K., and, Yadav S. Novel formulation of solid lipid microparticles of curcumin for anti-angiogenic and anti-inflammatory activity for optimization of therapy of inflammatory bowel disease. J Pharm Pharmacol. 2009;61:311–321. doi: 10.1211/jpp/61.03.0005. [DOI] [PubMed] [Google Scholar]
- Cui J, Yu B, Zhao Y, Zhu W, Li H, Lou H, et al. Enhancement of oral absorption of curcumin by self-microemulsifying drug delivery systems. Int J Pharm. 2009;371:148–155. doi: 10.1016/j.ijpharm.2008.12.009. [DOI] [PubMed] [Google Scholar]
- Chen C, Johnston TD, Jeon H, Gedaly R, McHugh PP, Burke TG, et al. An in vitro study of liposomal curcumin: stability, toxicity and biological activity in human lymphocytes and Epstein-Barr virus-transformed human B-cells. Int J Pharm. 2009;366:133–139. doi: 10.1016/j.ijpharm.2008.09.009. [DOI] [PubMed] [Google Scholar]
- Choi HS, Ipe BI, Misra P, Lee JH, Bawendi MG., and, Frangioni JV. Tissue- and organ-selective biodistribution of NIR fluorescent quantum dots. Nano Lett. 2009;9:2354–2359. doi: 10.1021/nl900872r. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bisht S, Feldmann G, Soni S, Ravi R, Karikar C, Maitra A, et al. Polymeric nanoparticle-encapsulated curcumin (“nanocurcumin”): a novel strategy for human cancer therapy. J Nanobiotechnology. 2007;5:3. doi: 10.1186/1477-3155-5-3. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li L, Braiteh FS., and, Kurzrock R. Liposome-encapsulated curcumin: in vitro and in vivo effects on proliferation, apoptosis, signaling, and angiogenesis. Cancer. 2005;104:1322–1331. doi: 10.1002/cncr.21300. [DOI] [PubMed] [Google Scholar]
- Li J, Jiang Y, Wen J, Fan G, Wu Y., and, Zhang C. A rapid and simple HPLC method for the determination of curcumin in rat plasma: assay development, validation and application to a pharmacokinetic study of curcumin liposome. Biomed Chromatogr. 2009;23:1201–1207. doi: 10.1002/bmc.1244. [DOI] [PubMed] [Google Scholar]
- Epstein J, Sanderson IR., and, Macdonald TT.2010Curcumin as a therapeutic agent: the evidence from in vitro, animal and human studies Br J Nutrepub ahead of print). [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Exosomal curcumin is more stable than free curcumin in vivo.
Liposomal curcumin has equal effect as curcumin or exosomal curcumin on the in vitro inhibition of IκB-α degradation.