Abstract
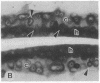
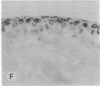
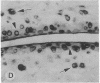
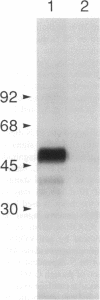
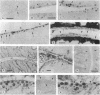
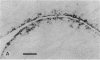
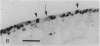
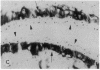
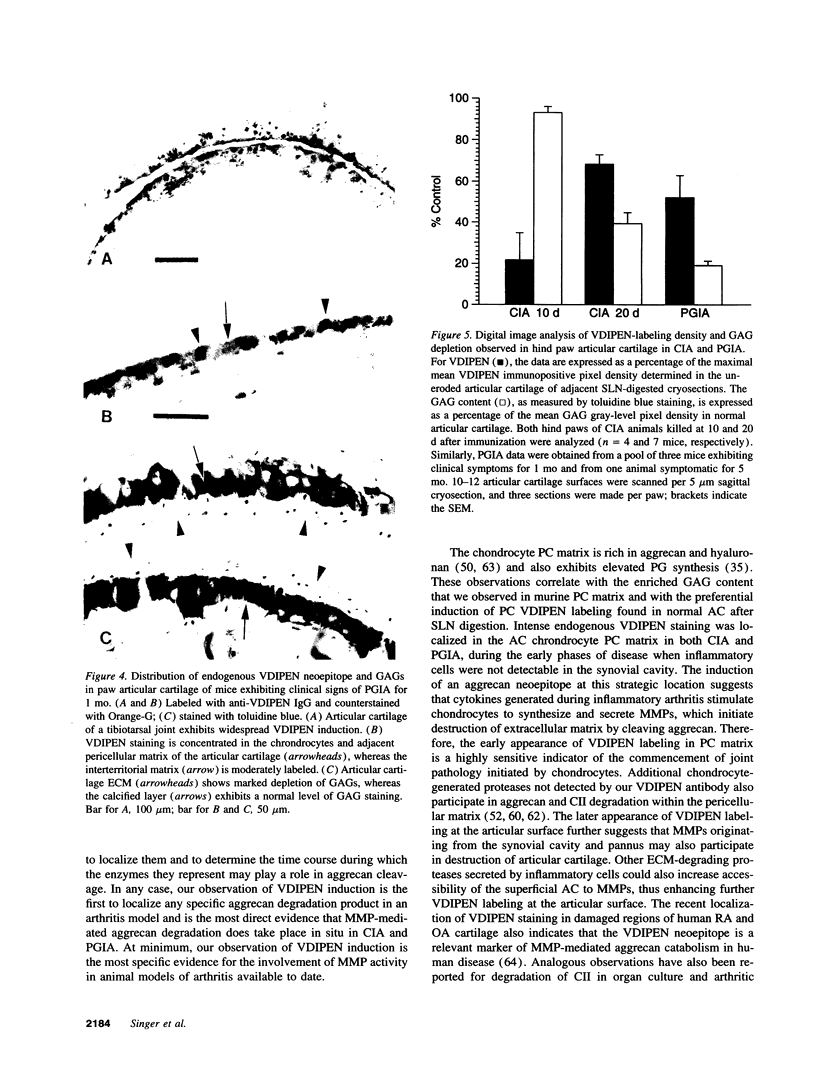
The destruction of articular cartilage in immune inflammatory arthritic disease involves the proteolytic degradation of its extracellular matrix. The role of activated matrix metalloproteinases (MMPs) in the chondrodestructive process was studied by identifying a selective cleavage product of aggrecan in murine arthritis models initiated by immunization with either type II collagen or proteoglycan. We conducted semiquantitative immunocytochemical studies of VDIPEN341 using a monospecific polyclonal antibody requiring the free COOH group of the COOH-terminal Asn for epitope detection. This antibody recognizes the aggrecan G1 domain fragment generated by MMP [i.e., stromelysin (SLN) or gelatinase A] cleavage of aggrecan between Asn341-Phe342 but does not recognize intact aggrecan. VDIPEN was undetectable in normal mouse cartilage but was observed in the articular cartilage (AC) of mice with collagen-induced arthritis 10 d after immunization, without histological damage and clinical symptoms. This aggrecan neoepitope was colocalized with high levels of glycosaminoglycans (GAGs) in pericellular matrices of AC chondrocytes but was not seen at the articular surface at this early time. Digestion of normal (VDIPEN negative) mouse paw cryosections with SLN also produced heavy pericellular VDIPEN labeling. Computer-based image analysis showed that the amount of VDIPEN expression increased dramatically by 20 d (70% of the SLN maximum) and was correlated with GAG depletion. Both infiltration of inflammatory cells into the synovial cavity and early AC erosion were also very prominent at this time. Analysis of adjacent sections showed that both induction of VDIPEN and GAG depletion were strikingly codistributed within sites of articular cartilage damage. Similar results occurred in proteoglycan-induced arthritis, a more progressive and chronic model of inflammatory arthritis. These studies demonstrate for the first time the MMP-dependent catabolism of aggrecan at sites of chondrodestruction during inflammatory arthritis.
Full text
PDF







Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Banerjee S., Wei B. Y., Hillman K., Luthra H. S., David C. S. Immunosuppression of collagen-induced arthritis in mice with an anti-IL-2 receptor antibody. J Immunol. 1988 Aug 15;141(4):1150–1154. [PubMed] [Google Scholar]
- Birkedal-Hansen H., Moore W. G., Bodden M. K., Windsor L. J., Birkedal-Hansen B., DeCarlo A., Engler J. A. Matrix metalloproteinases: a review. Crit Rev Oral Biol Med. 1993;4(2):197–250. doi: 10.1177/10454411930040020401. [DOI] [PubMed] [Google Scholar]
- Dodge G. R., Poole A. R. Immunohistochemical detection and immunochemical analysis of type II collagen degradation in human normal, rheumatoid, and osteoarthritic articular cartilages and in explants of bovine articular cartilage cultured with interleukin 1. J Clin Invest. 1989 Feb;83(2):647–661. doi: 10.1172/JCI113929. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Doege K. J., Sasaki M., Kimura T., Yamada Y. Complete coding sequence and deduced primary structure of the human cartilage large aggregating proteoglycan, aggrecan. Human-specific repeats, and additional alternatively spliced forms. J Biol Chem. 1991 Jan 15;266(2):894–902. [PubMed] [Google Scholar]
- Flannery C. R., Lark M. W., Sandy J. D. Identification of a stromelysin cleavage site within the interglobular domain of human aggrecan. Evidence for proteolysis at this site in vivo in human articular cartilage. J Biol Chem. 1992 Jan 15;267(2):1008–1014. [PubMed] [Google Scholar]
- Fosang A. J., Last K., Knäuper V., Neame P. J., Murphy G., Hardingham T. E., Tschesche H., Hamilton J. A. Fibroblast and neutrophil collagenases cleave at two sites in the cartilage aggrecan interglobular domain. Biochem J. 1993 Oct 1;295(Pt 1):273–276. doi: 10.1042/bj2950273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fosang A. J., Neame P. J., Hardingham T. E., Murphy G., Hamilton J. A. Cleavage of cartilage proteoglycan between G1 and G2 domains by stromelysins. J Biol Chem. 1991 Aug 25;266(24):15579–15582. [PubMed] [Google Scholar]
- Geiger T., Towbin H., Cosenti-Vargas A., Zingel O., Arnold J., Rordorf C., Glatt M., Vosbeck K. Neutralization of interleukin-1 beta activity in vivo with a monoclonal antibody alleviates collagen-induced arthritis in DBA/1 mice and prevents the associated acute-phase response. Clin Exp Rheumatol. 1993 Sep-Oct;11(5):515–522. [PubMed] [Google Scholar]
- Glant T. T., Mikecz K., Arzoumanian A., Poole A. R. Proteoglycan-induced arthritis in BALB/c mice. Clinical features and histopathology. Arthritis Rheum. 1987 Feb;30(2):201–212. doi: 10.1002/art.1780300211. [DOI] [PubMed] [Google Scholar]
- Goldberg G. I., Marmer B. L., Grant G. A., Eisen A. Z., Wilhelm S., He C. S. Human 72-kilodalton type IV collagenase forms a complex with a tissue inhibitor of metalloproteases designated TIMP-2. Proc Natl Acad Sci U S A. 1989 Nov;86(21):8207–8211. doi: 10.1073/pnas.86.21.8207. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Griffiths M. M., Eichwald E. J., Martin J. H., Smith C. B., DeWitt C. W. Immunogenetic control of experimental type II collagen-induced arthritis. I. Susceptibility and resistance among inbred strains of rats. Arthritis Rheum. 1981 Jun;24(6):781–789. doi: 10.1002/art.1780240605. [DOI] [PubMed] [Google Scholar]
- Hardingham T. E., Fosang A. J. Proteoglycans: many forms and many functions. FASEB J. 1992 Feb 1;6(3):861–870. [PubMed] [Google Scholar]
- Hasty K. A., Reife R. A., Kang A. H., Stuart J. M. The role of stromelysin in the cartilage destruction that accompanies inflammatory arthritis. Arthritis Rheum. 1990 Mar;33(3):388–397. doi: 10.1002/art.1780330312. [DOI] [PubMed] [Google Scholar]
- Hom J. T., Bendele A. M., Carlson D. G. In vivo administration with IL-1 accelerates the development of collagen-induced arthritis in mice. J Immunol. 1988 Aug 1;141(3):834–841. [PubMed] [Google Scholar]
- Hom J. T., Butler L. D., Riedl P. E., Bendele A. M. The progression of the inflammation in established collagen-induced arthritis can be altered by treatments with immunological or pharmacological agents which inhibit T cell activities. Eur J Immunol. 1988 Jun;18(6):881–888. doi: 10.1002/eji.1830180608. [DOI] [PubMed] [Google Scholar]
- Hunziker E. B., Herrmann W. In situ localization of cartilage extracellular matrix components by immunoelectron microscopy after cryotechnical tissue processing. J Histochem Cytochem. 1987 Jun;35(6):647–655. doi: 10.1177/35.6.3553318. [DOI] [PubMed] [Google Scholar]
- Hutchinson N. I., Lark M. W., MacNaul K. L., Harper C., Hoerrner L. A., McDonnell J., Donatelli S., Moore V., Bayne E. K. In vivo expression of stromelysin in synovium and cartilage of rabbits injected intraarticularly with interleukin-1 beta. Arthritis Rheum. 1992 Oct;35(10):1227–1233. doi: 10.1002/art.1780351016. [DOI] [PubMed] [Google Scholar]
- Knudson C. B. Hyaluronan receptor-directed assembly of chondrocyte pericellular matrix. J Cell Biol. 1993 Feb;120(3):825–834. doi: 10.1083/jcb.120.3.825. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lark M. W., Gordy J. T., Weidner J. R., Ayala J., Kimura J. H., Williams H. R., Mumford R. A., Flannery C. R., Carlson S. S., Iwata M. Cell-mediated catabolism of aggrecan. Evidence that cleavage at the "aggrecanase" site (Glu373-Ala374) is a primary event in proteolysis of the interglobular domain. J Biol Chem. 1995 Feb 10;270(6):2550–2556. doi: 10.1074/jbc.270.6.2550. [DOI] [PubMed] [Google Scholar]
- Lark M. W., Williams H., Hoernner L. A., Weidner J., Ayala J. M., Harper C. F., Christen A., Olszewski J., Konteatis Z., Webber R. Quantification of a matrix metalloproteinase-generated aggrecan G1 fragment using monospecific anti-peptide serum. Biochem J. 1995 Apr 1;307(Pt 1):245–252. doi: 10.1042/bj3070245. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lohmander L. S., Hoerrner L. A., Lark M. W. Metalloproteinases, tissue inhibitor, and proteoglycan fragments in knee synovial fluid in human osteoarthritis. Arthritis Rheum. 1993 Feb;36(2):181–189. doi: 10.1002/art.1780360207. [DOI] [PubMed] [Google Scholar]
- Lohmander L. S., Neame P. J., Sandy J. D. The structure of aggrecan fragments in human synovial fluid. Evidence that aggrecanase mediates cartilage degradation in inflammatory joint disease, joint injury, and osteoarthritis. Arthritis Rheum. 1993 Sep;36(9):1214–1222. doi: 10.1002/art.1780360906. [DOI] [PubMed] [Google Scholar]
- MacNaul K. L., Chartrain N., Lark M., Tocci M. J., Hutchinson N. I. Discoordinate expression of stromelysin, collagenase, and tissue inhibitor of metalloproteinases-1 in rheumatoid human synovial fibroblasts. Synergistic effects of interleukin-1 and tumor necrosis factor-alpha on stromelysin expression. J Biol Chem. 1990 Oct 5;265(28):17238–17245. [PubMed] [Google Scholar]
- Matrisian L. M. Metalloproteinases and their inhibitors in matrix remodeling. Trends Genet. 1990 Apr;6(4):121–125. doi: 10.1016/0168-9525(90)90126-q. [DOI] [PubMed] [Google Scholar]
- Mauritz N. J., Holmdahl R., Jonsson R., Van der Meide P. H., Scheynius A., Klareskog L. Treatment with gamma-interferon triggers the onset of collagen arthritis in mice. Arthritis Rheum. 1988 Oct;31(10):1297–1304. doi: 10.1002/art.1780311012. [DOI] [PubMed] [Google Scholar]
- McDonnell J., Hoerrner L. A., Lark M. W., Harper C., Dey T., Lobner J., Eiermann G., Kazazis D., Singer I. I., Moore V. L. Recombinant human interleukin-1 beta-induced increase in levels of proteoglycans, stromelysin, and leukocytes in rabbit synovial fluid. Arthritis Rheum. 1992 Jul;35(7):799–805. doi: 10.1002/art.1780350714. [DOI] [PubMed] [Google Scholar]
- McLean I. W., Nakane P. K. Periodate-lysine-paraformaldehyde fixative. A new fixation for immunoelectron microscopy. J Histochem Cytochem. 1974 Dec;22(12):1077–1083. doi: 10.1177/22.12.1077. [DOI] [PubMed] [Google Scholar]
- Mikecz K., Glant T. T., Poole A. R. Immunity to cartilage proteoglycans in BALB/c mice with progressive polyarthritis and ankylosing spondylitis induced by injection of human cartilage proteoglycan. Arthritis Rheum. 1987 Mar;30(3):306–318. doi: 10.1002/art.1780300310. [DOI] [PubMed] [Google Scholar]
- Moder K. G., Luthra H. S., Kubo R., Griffiths M., David C. S. Prevention of collagen induced arthritis in mice by treatment with an antibody directed against the T cell receptor alpha beta framework. Autoimmunity. 1992;11(4):219–224. doi: 10.3109/08916939209035158. [DOI] [PubMed] [Google Scholar]
- Nguyen Q., Murphy G., Roughley P. J., Mort J. S. Degradation of proteoglycan aggregate by a cartilage metalloproteinase. Evidence for the involvement of stromelysin in the generation of link protein heterogeneity in situ. Biochem J. 1989 Apr 1;259(1):61–67. doi: 10.1042/bj2590061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okada Y., Shinmei M., Tanaka O., Naka K., Kimura A., Nakanishi I., Bayliss M. T., Iwata K., Nagase H. Localization of matrix metalloproteinase 3 (stromelysin) in osteoarthritic cartilage and synovium. Lab Invest. 1992 Jun;66(6):680–690. [PubMed] [Google Scholar]
- Okada Y., Takeuchi N., Tomita K., Nakanishi I., Nagase H. Immunolocalization of matrix metalloproteinase 3 (stromelysin) in rheumatoid synovioblasts (B cells): correlation with rheumatoid arthritis. Ann Rheum Dis. 1989 Aug;48(8):645–653. doi: 10.1136/ard.48.8.645. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Reife R. A., Loutis N., Watson W. C., Hasty K. A., Stuart J. M. SWR mice are resistant to collagen-induced arthritis but produce potentially arthritogenic antibodies. Arthritis Rheum. 1991 Jun;34(6):776–781. doi: 10.1002/art.1780340621. [DOI] [PubMed] [Google Scholar]
- Sandy J. D., Flannery C. R., Neame P. J., Lohmander L. S. The structure of aggrecan fragments in human synovial fluid. Evidence for the involvement in osteoarthritis of a novel proteinase which cleaves the Glu 373-Ala 374 bond of the interglobular domain. J Clin Invest. 1992 May;89(5):1512–1516. doi: 10.1172/JCI115742. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shu S. Y., Ju G., Fan L. Z. The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the nervous system. Neurosci Lett. 1988 Feb 29;85(2):169–171. doi: 10.1016/0304-3940(88)90346-1. [DOI] [PubMed] [Google Scholar]
- Spinella D. G., Jeffers J. R., Reife R. A., Stuart J. M. The role of C5 and T-cell receptor Vb genes in susceptibility to collagen-induced arthritis. Immunogenetics. 1991;34(1):23–27. doi: 10.1007/BF00212308. [DOI] [PubMed] [Google Scholar]
- Thorbecke G. J., Shah R., Leu C. H., Kuruvilla A. P., Hardison A. M., Palladino M. A. Involvement of endogenous tumor necrosis factor alpha and transforming growth factor beta during induction of collagen type II arthritis in mice. Proc Natl Acad Sci U S A. 1992 Aug 15;89(16):7375–7379. doi: 10.1073/pnas.89.16.7375. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Umenishi F., Umeda M., Miyazaki K. Efficient purification of TIMP-2 from culture medium conditioned by human hepatoma cell line, and its inhibitory effects on metalloproteinases and in vitro tumor invasion. J Biochem. 1991 Aug;110(2):189–195. doi: 10.1093/oxfordjournals.jbchem.a123555. [DOI] [PubMed] [Google Scholar]
- Van Wart H. E., Birkedal-Hansen H. The cysteine switch: a principle of regulation of metalloproteinase activity with potential applicability to the entire matrix metalloproteinase gene family. Proc Natl Acad Sci U S A. 1990 Jul;87(14):5578–5582. doi: 10.1073/pnas.87.14.5578. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Walakovits L. A., Moore V. L., Bhardwaj N., Gallick G. S., Lark M. W. Detection of stromelysin and collagenase in synovial fluid from patients with rheumatoid arthritis and posttraumatic knee injury. Arthritis Rheum. 1992 Jan;35(1):35–42. doi: 10.1002/art.1780350106. [DOI] [PubMed] [Google Scholar]
- Walcz E., Deák F., Erhardt P., Coulter S. N., Fülöp C., Horvath P., Doege K. J., Glant T. T. Complete coding sequence, deduced primary structure, chromosomal localization, and structural analysis of murine aggrecan. Genomics. 1994 Jul 15;22(2):364–371. doi: 10.1006/geno.1994.1396. [DOI] [PubMed] [Google Scholar]
- Watson W. C., Townes A. S. Genetic susceptibility to murine collagen II autoimmune arthritis. Proposed relationship to the IgG2 autoantibody subclass response, complement C5, major histocompatibility complex (MHC) and non-MHC loci. J Exp Med. 1985 Dec 1;162(6):1878–1891. doi: 10.1084/jem.162.6.1878. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Williams R. O., Feldmann M., Maini R. N. Anti-tumor necrosis factor ameliorates joint disease in murine collagen-induced arthritis. Proc Natl Acad Sci U S A. 1992 Oct 15;89(20):9784–9788. doi: 10.1073/pnas.89.20.9784. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woessner J. F., Jr Matrix metalloproteinases and their inhibitors in connective tissue remodeling. FASEB J. 1991 May;5(8):2145–2154. [PubMed] [Google Scholar]
- Wolfe G. C., MacNaul K. L., Buechel F. F., McDonnell J., Hoerrner L. A., Lark M. W., Moore V. L., Hutchinson N. I. Differential in vivo expression of collagenase messenger RNA in synovium and cartilage. Quantitative comparison with stromelysin messenger RNA levels in human rheumatoid arthritis and osteoarthritis patients and in two animal models of acute inflammatory arthritis. Arthritis Rheum. 1993 Nov;36(11):1540–1547. doi: 10.1002/art.1780361108. [DOI] [PubMed] [Google Scholar]
- Wooley P. H. Collagen-induced arthritis in the mouse. Methods Enzymol. 1988;162:361–373. doi: 10.1016/0076-6879(88)62091-x. [DOI] [PubMed] [Google Scholar]
- Wooley P. H., Luthra H. S., Stuart J. M., David C. S. Type II collagen-induced arthritis in mice. I. Major histocompatibility complex (I region) linkage and antibody correlates. J Exp Med. 1981 Sep 1;154(3):688–700. doi: 10.1084/jem.154.3.688. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wu J. J., Lark M. W., Chun L. E., Eyre D. R. Sites of stromelysin cleavage in collagen types II, IX, X, and XI of cartilage. J Biol Chem. 1991 Mar 25;266(9):5625–5628. [PubMed] [Google Scholar]
- Yasumitsu H., Miyazaki K., Umenishi F., Koshikawa N., Umeda M. Comparison of extracellular matrix-degrading activities between 64-kDa and 90-kDa gelatinases purified in inhibitor-free forms from human schwannoma cells. J Biochem. 1992 Jan;111(1):74–80. doi: 10.1093/oxfordjournals.jbchem.a123721. [DOI] [PubMed] [Google Scholar]
- van de Loo F. A., Arntz O. J., Otterness I. G., van den Berg W. B. Protection against cartilage proteoglycan synthesis inhibition by antiinterleukin 1 antibodies in experimental arthritis. J Rheumatol. 1992 Mar;19(3):348–356. [PubMed] [Google Scholar]
- van den Berg W. B., Joosten L. A., Helsen M., van de Loo F. A. Amelioration of established murine collagen-induced arthritis with anti-IL-1 treatment. Clin Exp Immunol. 1994 Feb;95(2):237–243. doi: 10.1111/j.1365-2249.1994.tb06517.x. [DOI] [PMC free article] [PubMed] [Google Scholar]