Abstract
Objectives
Morphological studies on presbycusis, or age-related hearing loss, have been performed in several different strains of mice that demonstrate hearing loss with auditory pathology. The C57BL/6 (C57) mouse is a known model of early onset presbycusis, while the CBA mouse is characterized by relatively late onset hearing loss. We performed this study to further understand how early onset hearing loss is related with the aging process of the cochlea.
Methods
We compared C57 cochlear pathology and its accompanying apoptotic processes to those in CBA mice. Hearing thresholds and outer hair cell functions have been evaluated by auditory brainstem response (ABR) recordings and distortion product otoacoustic emission (DPOAE).
Results
ABR recordings and DPOAE studies demonstrated high frequency hearing loss in C57 mice at P3mo of age. Cochlear morphologic studies of P1mo C57 and CBA mice did not show differences in the organ of Corti, spiral ganglion, or stria vascularis. However, from P3mo and onwards, a predominant early outer hair cell degeneration at the basal turn of the cochlea in C57 mice without definitive degeneration of spiral ganglion cells and stria vascularis/spiral ligament, compared with CBA mice, was observed. Additionally, apoptotic processes in the C57 mice also demonstrated an earlier progression.
Conclusion
These data suggest that the C57 mouse could be an excellent animal model for early onset 'sensory' presbycusis in their young age until P6mo. Further studies to investigate the intrinsic or extrinsic etiologic factors that lead to the early degeneration of organ of Corti, especially in the high frequency region, in C57 mice may provide a possible pathological mechanism of early onset hearing loss.
Keywords: Cochlea, Morphology, Hearing, Aging, Apoptosis
INTRODUCTION
Mice models for the study of hearing loss were recognized approximately 50 years ago and mice are now the predominant model for hearing research (1). As mice permit increased housing densities compared with other models and have a short lifespan, they provide an excellent model for studying the effects of aging, as results are evident in a shorter timeframe and at lower cost. These features make mice ideal for studies of into age-related hearing loss (ARHL).
The short length of the mouse cochlea also means that morphologic studies from basal apical turns require fewer sampling points. Understanding the subtle morphological differences at the microstructures of the cochleae in different strains of mice models may enhance our knowledge regarding ARHL and provide some clues for the mechanism of differing vulnerabilities for cochlea pathology during the aging process. In this regard, C57BL/6 (C57) mice have been used as an animal model for human ARHL. They are homozygous for the defective Cdh23ahl allele of the gene encoding cadherin 23, which is a constituent of hair cell stereocilia (2, 3). Progressive loss of hair cells (4), degeneration of afferent neurons, the stria vascularis, and the spiral ligament (5, 6) have all been observed in the C57 mouse strain, which overlaps with each of the major types of ARHL suggested by Schuknecht and Gacek (7). Investigation of these multiform pathologies and their relationship with each other during ARHL in C57 mice may thus lead to new perspectives in the pathology of ARHL.
The pattern of progressive hearing loss is generally reported as early onset in C57 mice, unlike CBA mice that exhibit minimal hearing loss at 18 months (4, 6, 8). CBA mice have a mean life span of 22-25 months and stay relatively free of disease until at least 20-22 months. CBA mice carry an ahl-resistant allele and, like humans, gradually lose their ability to hear high-frequency sounds as they age and are not prone to premature hearing loss (9). Recent morphologic studies on the CBA mice cochlea have revealed a lack of stria vascularis degeneration and unchanged endocochlear potential until 25 months. Slow high-frequency hearing loss with a modest reduction of ganglion cell density was evident, suggesting the CBA mouse as a potential animal model of sensorineural presbycusis (10).
Several reports have suggested a relationship between apoptotic processes and age related decline in physiological function in multiple organs. Apoptosis is triggered by two major pathways: the intrinsic pathway (initiated by the loss of outer mitochondrial membrane integrity) and the extrinsic pathway (initiated through the recruitment of caspases to the membrane death inducing signaling complexes) (11). In the cochlea, ARHL development was both increased and more severe in mice possessing deletions in the superoxide dismutase genes, which are important in antioxidant defense, indicating a need to detoxify ROS to prevent earlier presbycusis (12, 13). Multiple cell death pathways induced by oxidative stress are activated in the hair cells of the cochlea in aging CBA/J mice (14). In C57 mice, induction of Bak-dependent mitochondrial fission and apoptosis, which systematically occurs in response to oxidative stress, has been suggested to be a key mechanism of ARHL (15).
Despite the use of C57 and CBA strains for the study of ARHL, no comparison of the apoptotic processes and morphologic changes of the cochleae exist in these models. Because these strains vary in terms of their progressive hearing loss, such studies are required to further the mechanism-related investigation of ARHL. In this study, we performed precise and quantitative morphological studies and analyzed the basic apoptotic processes existing in the C57 and CBA mice cochleae. Audiologic studies were also performed for the measurement of hearing thresholds and outer hair cell function.
MATERIALS AND METHODS
Animals
Male CBA/N and C57 mice were purchased from the Choongang Experimental Animal Centre (Orient Bio, Seongnam, Korea). They were kept at an animal colony of the Catholic University of Korea prior to the initiation of experimental procedures. All procedures and animal handling were performed according to the national ethic guidelines and were approved by, and complied with all protocol requirements at the Catholic University of Korea (IACUC - CUMC-20).
Hearing tests
To investigate the hearing levels of C57 and CBA mice, auditory brainstem response (ABR) and distortion product otoacoustic emissions (DPOAEs) were performed at P1mo, P3mo and P6mo (n=10-12 for each mouse group). Auditory testing was performed in a sound-proof chamber. Prior to acoustic testing, mice were anesthetized by intraperitoneal injection of a mixture of Rumpun (0.4 mL/kg) and Zoletil (0.6 mL/kg) and boosted with one-fifth of the original dose as required. Mouse body temperature was maintained with a heating pad.
Auditory brainstem response test
The evoked acoustic brainstem response thresholds were differentially recorded from the scalp of the mice. Responses were recorded using subdermal needle electrodes at the vertex, below the pinna of the left ear (reference), and below the contralateral ear (ground). The sound stimuli included click (100 µs duration; 31 Hz) and tone pips at 8, 16, and 32 kHz (1,562 µms duration; cos2 shaping; 21 Hz). ABR measurements were taken with an Intelligent Hearing System (IHS) Smart EP System, running IHS high frequency software (ver. 2.33) and using IHS high-frequency transducers (HFT9911-20-0035, IHS, Miami, FL, USA). Acoustic stimuli were presented directly to the entrance of the ear canal. Stainless steel needle electrodes were placed subcutaneously at the vertex and over the bullae, with a reference electrode at the occiput. ABR thresholds were obtained for 5 ms duration clicks and 5 ms duration tone bursts were presented at a rate of 50/seconds. Tone bursts were gated using an exact Blackman envelope (2.5 ms rise/decay, 0 ms plateau) and presented in a decreasing intensity series, beginning with levels that elicited distinct evoked potentials. Evoked potentials were amplified (×200,000); bandpass-filtered (100-3,000 Hz), and averaged over 1,024 sweeps. Recording epochs comprised the 12 ms following stimulus onset. Thresholds were determined for a broad-band click and for 8, 16, and 32 kHz pure-tone stimuli by decreasing the sound pressure level (SPL) in 10 dB decrements until the lowest level at which a distinct ABR wave pattern could be recognized by two of the investigators.
DPOAE test
DPOAE was recorded using the IHS of Smart OAE 4.26 (IHS). DPOAE measurements were conducted for pure tones from 6-32 kHz. An Etymotic 10B+ probe was inserted into the external ear canal and used in conjunction with two different types of transducer, depending on the range of the stimulation frequency. An Etymotic ER2 stimulator was used for frequencies ranging from 6-16 kHz. For frequencies ranging from 16-32 kHz, an IHS high frequency transducer was used. Stimulus response signals were sampled at a rate of 128 kHz using a 16-bit D/A converter. L1 amplitude was set to 65 dB SPL and L2 amplitude was set to 55 dB SPL. Frequencies were acquired with an F2 F1 ratio of 1.22. Five stimulation levels, ranging from 65 to 25 dB SPL in 10 dB steps, were used. In total, four blocks were acquired, each block consisting of 32 sweeps. A comparison of mean ABR thresholds and DPOAE values between CBA and C57 mice were performed using student t-test. A P-value of <0.05 was deemed to indicate statistical significance.
Light microscopy
CBA and C57 mice of P1mo (n=5), P3mo (n=5), and P6mo (n=5) were anesthetized and their cochleae isolated and dissected. The cochleae were perfused through the round and oval window with both 2% paraformaldehyde and 2% glutaraldehyde in 0.1 M phosphate buffer (pH 7.4) and incubated in the same fixative overnight at 4℃. The cochleae were then rinsed with 0.1 M PBS and incubated in 1% osmium tetroxide overnight, followed by immersion in 5% EDTA for 2-4 days. The calcified cochlea were then dehydrated in ethanol and propylene oxide and embedded in Araldite 502 resin (Electron Microscopy Sciences, Fort Washington, PA, USA). The cochlea were sectioned at 5 µm, stained with toluidine blue and mounted in Permount on microscope slides. The Image Processing and Analysis in Java (ImageJ; National Institute of Health, Bethesda, MD, USA) was used for morphologic observations.
Morphometry of the organ of Corti
Three different regions of the cochlea from mid-modiolar sections with clearly visible whole organs of Corti (OC) (base to apex) were chosen for image magnification (×40 objective). Six sections from each cochlea were analyzed and the mean values were used. A modified rank-order grading method (16) was used to rate the condition of both the supporting cells and the general shape of the OC from P3mo and P6mo mice. Briefly, numbers were assigned to indicate the following conditions: 5) normal cytoarchitecture of the OC with intact hair cells; 4) maintenance of normal cytoarchitecture of the OC with all supporting cells intact, but loss of hair cells; 3) partial collapse of the OC, but with subtypes of supporting cells still recognizable; 2) a low cuboidal cell layer without recognizable supporting cells; and 1) complete degeneration of the OC into a single flattened and undifferentiated cell layer. The averaged regional values for OC in the base, middle and apical turns were compared between the C57 and CBA mice.
Spiral ganglion cell counts
Spiral ganglion cells (SGC) were counted from the same plastic sections as used for the OC morphometric study. Rosenthal's canals from basal, mid, and apical turns were circled to determine the total area (Image J) and all spiral ganglion cells were counted. The number of spiral ganglion cells was then divided by the area measured to obtain the spiral ganglion cell density. Sections from C57 and CBA mice of P3mo and P6mo of ages were matched for their location in the cochlea and a total of six sections for each mouse assessed.
Morphometry of the stria vascularis and spiral ligament
Plastic sections prepared for OC morphometry and spiral ganglion cell counts were used in the morphometric analysis of the lateral wall of the cochlea. Six representative sections from each cochlea were analyzed. The combined area of the spiral ligament (SL) and stria vascularis (SV) was determined from images at a 10× magnification and the strial area alone from images at a 25× magnification using ImageJ.
Loss of hair cells
The cochleae of CBA and C57 mice of P1mo, P3mo, and P6mo (n=5) were perfused with 4% PFA and maintained in the fixative overnight at 4℃. After rinsing with PBS, the cochleae were decalcified with 5% EDTA for 2-4 days. The otic capsule was then removed, followed by the removal of the lateral wall, Reissner's membrane and the tectorial membrane. The remaining OC was stained with rhodamine-phalloidin (2 U/mL) for 1 hour, and after rinsing with PBS, each turn of the cochleae was whole-mounted. Hair cells in the OC were visualized under a microscope equipped with epifluorescence. Missing hair cells were observed through Z-stacked images viewed under a 10× objective at each turn using a Zeiss confocal microscope with LSM image software. Identical settings were maintained throughout image capture.
Apoptosis assays
Western blot for caspase 3
Total protein extracts were prepared from five mice cochlea of each mouse group at P6mo lysed with 20 mM Tris HCl, pH 7.4, 5 mM EDTA, 10 mM Na2P2O7, 100 mM NaF, 2 mM Na3VO4, 1% NP-40, 1 mM PMSF, aprotinin, and leupeptin. The cochlea homogenate was centrifuged (6,500 rpm, 4℃, 20 seconds) and protein concentration in the supernatant determined by a BCA protein assay. Equal amounts of protein were resolved by SDS-PAGE, and transferred to a nitrocellulose membrane. Membranes were probed with an anti-caspase 3 (1:1,000) primary antibody and appropriate HRP-conjugated secondary antibodies as per the manufacturer's recommendations. All Western blots were visualized using an enhanced chemiluminescence system (Las 3000, Fujifilm, Tokyo, Japan).
TUNEL assay
To evaluate nuclear DNA fragmentation (a known marker of apoptotis) we examined the cochleae of CBA and C57 mice using a terminal deoxynucleotidyl transferase-mediated deoxyuridine triphosphate nick end labeling (TUNEL) assay. TUNEL staining for apoptotic nuclei was performed using the DeadEnd Colorimetric TUNEL System (Promega, Madison, WI, USA) according to the manufacturer's instructions. Briefly, stained sections were viewed by light microscopy and the OC in the basal turns was evaluated for TUNEL positivity.
Statistical analyses
SPSS ver. 15.0 (SPSS Inc., Chicago, IL, USA) was used for all statistical analyses. Group comparisons between CBA and C57 mice were made using Student's t-test or Mann-Whitney test. A P-value of <0.05 was deemed to indicate statistical significance.
RESULTS
C57 mice display early onset high frequency hearing loss
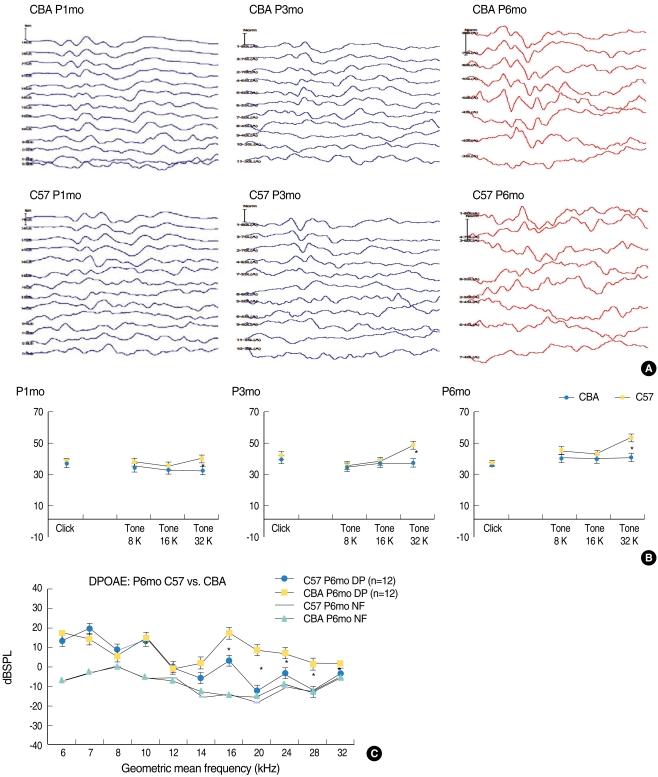
At the age of P1mo, C57 mice did not show any significant difference in hearing compared with CBA mice. However, ABR threshold testing through P6mo for CBA and C57 mice showed an elevated threshold. Raw data from the 32 kHz tone burst ABR displayed both an elevated threshold and abnormal wave forms in C57 mice from P3mo onwards. C57 mice were shown to demonstrate a significantly elevated hearing threshold at the 32 kHz tone burst ABR from P1mo onwards, but not for 8, 16 kHz or click.
In serial ABR threshold testing through P6mo, C57 mice demonstrated a significantly different high frequency (32 kHz) sensorineural hearing loss from P3mo onwards, but not for 16 kHz, 8 kHz, or clicks (Fig. 1A). Representative serial raw ABR data at 32 kHz tonal stimuli (Fig. 1B) are shown for better visualization of the differences observed in ABR. Additionally, P6mo C57 mice showed a defect in the function of outer hair cells (OHCs), shown as DPOAE levels decreased compared with CBA mice (Fig. 1C). Significant level differences in the DPOAE test between C57 and CBA mice were observed at 24-32 kHz at P6mo. These data indicate that C57 mice display relatively rapid progression of OHC dysfunction at high frequency areas when compared with CBA mice.
Fig. 1.
Auditory brainstem response (ABR) threshold testing through P6mo for CBA and C57 mice. Raw data for the 32 kHz tone burst ABR show an elevated threshold and abnormal wave form in C57 mice from P3mo onwards (A). C57 mice demonstrate a significantly elevated hearing threshold at a 32 kHz tone burst ABR from P1mo onwards, but not for 8, 16 kHz, or clicks (B). C57 mice at P6mo show a clear defect in outer hair cell function from the mid-to-high frequency region, which has been observed as low distortion product otoacoustic emission (DPOAE) levels compared with the CBA mice (C). *P<0.05. NF: noise floor.
C57 mice display earlier degeneration in the organ of Corti and spiral ganglion, but not in the stria vascularis or spiral ligament
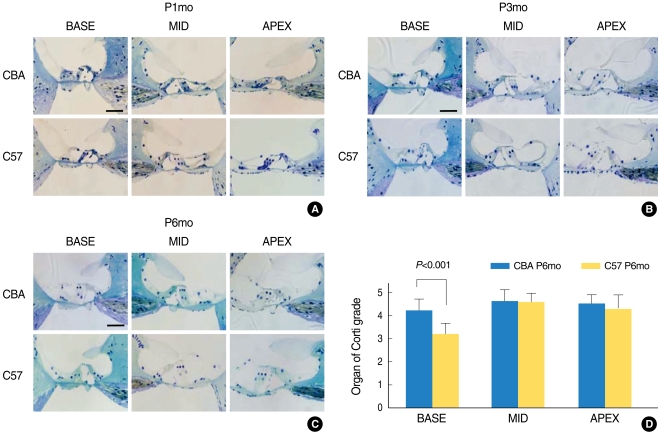
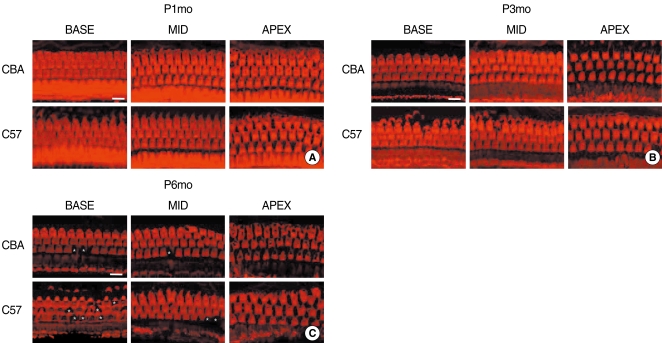
Serial light microscopic changes of the OC from CBA (Fig. 2, upper panels) versus C57 (lower panels) mice from base, mid and apical regions of the cochlea showed no difference at P1mo. However, from P3mo (B), C57 mice showed degeneration of the outer hair cells at the base of the cochlea. At P6mo (C), C57 mice consistently displayed more severe degeneration in the region of the outer hair cells, compared with those of CBA mice (Fig. 2A-C). The averaged value for regional supporting cell condition in the base of C57 mice was significantly lower than that of CBA mice at P6mo (Mann Whitney test; P<0.001) (Fig. 2D).
Fig. 2.
Serial light microscopy changes of the organ of Corti from CBA (upper panels) versus C57 (lower panels) mice from base, mid, and apical regions of the cochlea. At P1mo (A), the morphology of the organ of Corti did not show differences between CBA and C57 strains. However, from P3mo (B), C57 mice showed degeneration of outer hair cells at the base of the cochlea. At P6mo (C), C57 mice consistently showed more severe damage in the region of the outer hair cells, compared with CBA. A rank-order grading method was used to rate the condition of supporting cells and the general shape of the organ of Corti. The average value for regional supporting cell condition in the base of C57 mice was significantly lower than that of CBA mice at P6mo. (D) Six mid-modiolar sections were counted and averaged to obtain a value from one animal. Scale bar 50 µm.
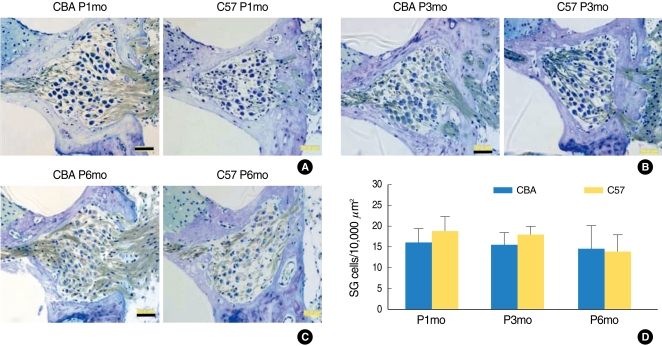
Serial light microscopic changes of the SGC from base, mid, and apical regions of the cochlea showed that C57 mice displayed both earlier and more severe degeneration (Fig. 3A-C). SGC densities (cells/10.000 µm2) decreased in both mice strains; however, no significant differences in this decrease were apparent (Fig. 3D). Interestingly, SGC density in C57 mice was higher than that in CBA mice until P3mo. At P6mo, the density of SGC from both mice strains was reversed, which may indicate an earlier and more abrupt degeneration of SGC in the C57 mice.
Fig. 3.
Serial light microscopy changes of the spiral ganglion cells from CBA versus C57 mice from base, mid, and apical regions of the cochlea. C57 mice appear to have earlier and more severe degeneration of spiral ganglion cells (SGC) in the light micrographs (A-C). SGC densities showed a decreasing tendency in both mice strains, but no significant difference between them was evident (D). SGC density in C57 mice was higher than that of CBA mice until P3mo. At P6mo, the density of SGC was reversed, indicating earlier and more abrupt degeneration of SGC in C57 mice. Scale bar 50 µm.
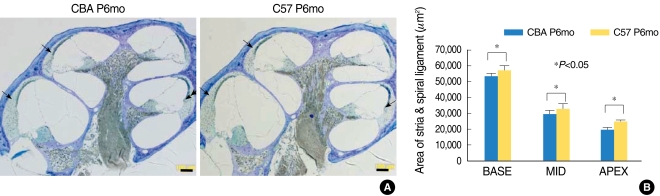
Morphological studies of the SV and SL showed significantly larger mean values in the C57 mice (Fig. 4). No evidence of SV or SL degeneration was evident until P6mo in both strains (data not shown). To further assess the status of hair cell degeneration, we performed surface preparations of the cochlea at P1mo, P3mo, and P6mo. No hair cell loss was evident in mice strains at P1mo. However, the P3mo C57 mice showed hair cell degeneration at the basal turn of the cochlea that was more pronounced at P6mo of age, when hair cell loss at the mid turn of the cochleae was observed (Fig. 5). CBA mice showed only occasional hair cell loss, which was restricted until P6mo.
Fig. 4.
Morphology of the strain vascularis and spiral ligament of P6mo in CBA and C57 mice. C57 mice displayed a larger stria vascularis and spiral ligament until P6mo, compared with CBA mice (A). Quantification of the area showed significantly larger values in C57 mice (B). Scale bar 100 µm.
Fig. 5.
Hair cells of P1mo CBA and C57 mice were intact from base to apex when stained with rhodamine-phalloidin (A). At P3mo, C57 mice showed occasional hair cell loss at the basal turn of the cochlea (B). At P6mo, multiple hair cell loss at the basal and mid turn of the cochlea was observed in C57 mice (C). CBA mice showed occasional hair cell loss at P6mo of age. *Representative sites of hair cell loss. Scale bar 10 µm.
C57 mice display earlier apoptotic hair cell death
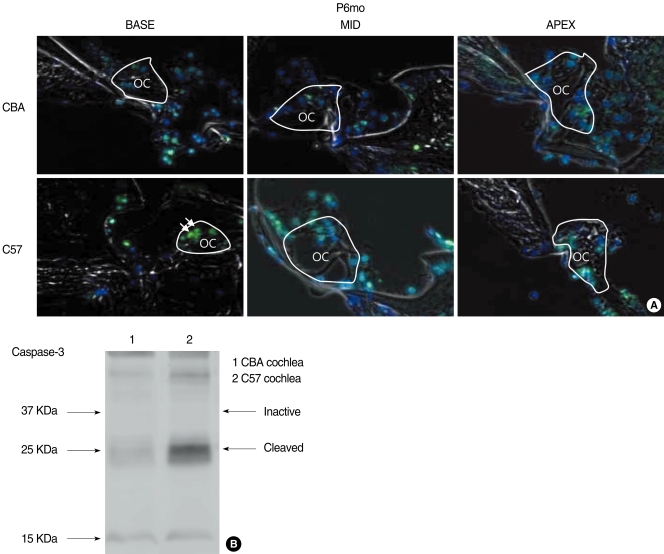
Same anatomical site of basal, mid and apical turns of cochleae at P1mo, P3mo, and P6mo have been compared for the study of TUNEL assay. When the number of apoptotic hair cells from P1mo to P6mo in the cochlea of mouse strains was measured, the TUNEL assay demonstrated that the nuclei of the outer hair cells of the OC were apoptotic from P1mo in C57 mice. More severe apoptosis was evident in C57 when compared with CBA mice (Fig. 6A-C), suggesting enhanced cell death in the C57 mice. To further clarify the enhancement of apoptosis in the C57 mice we analyzed the levels of activated (cleaved) caspase-3 in the cochlea of both mouse strains during the aging process. While we detected cleaved caspase-3 in the cochlear homogenates of both mice strains, the levels of activated caspase-3 in the cochlear homogenate of C57 mice was consistently higher than in CBA mice (Fig. 6D). These data are consistent with the occurrence of increased numbers of apoptotic hair cells in the C57 mice.
Fig. 6.
Apoptotic hair cell death at P6mo in the cochlea of CBA and C57 mice. Apoptotic cells were observed by either the TUNEL assay (A) or Western blot for cleaved caspase-3 (B). Strongly stained nuclei of the outer hair cells (arrows) which represent the more severe apoptosis were observed at the basal turn of the cochlea in C57 mice compared with CBA mice (A). Activated caspase-3 was more evident in C57 mice than in CBA mice at P6mo (B). OC: organ of Corti.
DISCUSSION
C57 mice have been used as the animal model for human ARHL. The pattern of progressive hearing loss is generally reported to be early onset hearing in C57 mice, unlike CBA mice that exhibit minimal hearing loss (4, 6, 8). The current study was conducted to compare morphological differences of the cochlea between these two commercial strains as a means for further studies regarding the mechanisms of ARHL. Our functional studies of ABR and DPOAE were well-matched to data obtained from the morphological study results. To our knowledge, this is the first reported investigation into the morphology of the entire cochlea with a quantitative comparison in young C57 and CBA mouse strains.
As C57 mice show high frequency hearing loss from P1mo onwards, they provide a good model for early onset hearing loss. Additionally, the pronounced high frequency hearing loss of C57 mice suggests they may provide a model of sensory presbycusis among different types of ARHL, as suggested by Schukenecht (7). Compared with CBA mice, C57 mice show an earlier onset of high frequency hearing loss, observed at ABR with a tone burst stimulus of 32 kHz. Outer hair cell functions observed from DPOAE studies also demonstrate that the earlier functional loss of OHCs in C57 mice occurs, particularly at high frequency areas. Through this functional study, we can suggest the use of C57 mice as a model of early onset hearing loss.
ABR threshold shifts in several strains of mice including CBA, C57, and BALB at different ages have been observed in other studies (16, 17). The hearing loss over time in CBA and C57 strains is similar to the elevated hearing thresholds observed with age in humans. The C57 mice demonstrate a rapid auditory decline, while the CBA mice display a wide range of high-frequency responses from near-normal to severely impaired, even at 23 months. The effects of age on DPOAE amplitudes in the CBA and C57 strains were also evaluated in this study. We confirmed an earlier onset of hearing loss and decreased outer hair cell function in C57 mice. The DPOAE levels for CBA mice displayed higher DPOAE magnitudes for the high frequency region, relative to the C57 mice. It has been shown that a reduction in OAEs is associated with a cochlea-based impairment that specifically involves the OHCs (18, 19). Due to their specificity for measuring OHC junction, OAEs represent an ideal response measurement for studying the progressive OHC damage associated with ARHL. In this study, C57 mice displayed progressive hearing impairment that began in high-frequency regions as early as P3mo. This impaired OHC function in DPOAE is consistent with an ABR threshold shift. The present study demonstrated that DPOAE levels of C57 mice were similar to CBA mice at P1mo, but then declined by P3mos. Thus, at P3mo, C57 and CBA strains display different courses of ARHL, suggesting that C57 mice are a more suitable model of early onset hearing loss or sensory ARHL.
We observed the morphological status of the cochleae in C57 and CBA stains from P1mo to P6mo. Quantitative analysis of morphologic differences in OC, spiral ganglion (SG), and SV showed that predominant and early OHC degeneration observed at the basal turn of the cochlea in C57 mice was present, without definitive degeneration of spiral ganglion cells or stria vascularis/spiral ligament, compared with the CBA mice. Thus, the C57 mouse appears to be a good animal model of sensory hearing loss. The two strains of mice showed a different morphological status of the cochlea from P3mo to P6mo and a good correlation was evident between audiologic study results and HC degeneration. Previous morphologic studies regarding CBA mice display a decrease in OHC cell density at 18 months, a time when OHC loss is evident. No degeneration of SV was observed until 25 months of age and a reduction of ganglion cell density following OHC degeneration was observed, suggesting that CBA mice could provide a model of late onset sensorineural ARHL (10, 20). In this study, no morphological degeneration of CBA mice cochlea was observed until P6mo, and thus CBA were used as morphological and functional control animals for the studies of early onset hearing loss in C57 mice.
Hair cell degeneration was measured from both mouse strains. Neither strain showed hair cell loss at P1mo; C57 mice at P3mo showed hair cell degeneration at the basal turn of the cochlea. At P6mo, significant hair cell loss was evident at the basal turn, while some loss of hair cells was evident at the mid turn. Compared with C57 mice, CBA mice did not show hair cell loss at any turn of the cochlea until P6mo, consistent with previous quantitative measuresments (4). These studies, however, did not evaluate the precise morphological status of OC, SG, or SV and, as such, the present study provides a quantitative description of cochlear morphology in C57 and CBA mice. A statistical difference in the grade of OC in the basal turns of the cochlea and their characteristic early degeneration of OHCs and hair cell loss with concomitant high frequency hearing loss provides evidence that C57 mice are a good animal model of early onset sensory hearing loss.
Our results show that the cochleae of C57 mice demonstrated an earlier apoptotic process of aging, when compared with CBA mice, as determined by the TUNEL assay and the abundance of cleaved caspase-3. As C57 mice displayed apoptotic cell death in their cochlea at young ages of P3mo and P6mo it is interesting to suggest that such a phenomenon may be characteristic of early onset sensory hearing loss. Several reports exist regarding cell death pathways in the cochlea. It has been suggested that the intrinsic and extrinsic cell death pathways at the age of 12 months and 18-26 months are increased in CBA mice. Additionally, an increase of cytosolic cytochrome c and activated caspase 9 with an increase of calpain I and calpain II has been demonstrated in middle aged or old CBA mice, suggesting that the activation of multiple cell death pathways linked to oxidative stress, are involved in the aging process in mice cochlea (14). Apoptosis related genes have been previously observed in aging CBA mice (4). Indeed, 35 genes displayed significantly different expression in ARHL from GeneChip assays, particularly caspase-4 apoptosis-related cysteine proteases, calpain II, and the tumor necrosis factor receptor superfamilies. Additionally, the expression of apoptosis-related proteins, the suppression of bcl-2, and the activation of caspase-3p20 have been described in the aged cochlea of Mongolian gerbils (21). However, the current study is the first to describe an early onset apoptotic pathways in the cochlea of C57 mice and as such suggests that the intrinsic apoptosis pathway may be related to inner ear degeneration in this mouse strain. In this regard it should be noted that the early onset of apoptosis was elevated in C57 mice when compared with young CBA mice.
In conclusion, we demonstrated that the C57 mouse model of early onset sensory hearing loss shows different functional and morphologic phenotypes from the CBA mouse. Understanding the differences between different mice strains may provide important clues to understand the individual variance in and susceptibility to ARHL.
ACKNOWLEDGEMENTS
This research was funded by a grant from the alumni of the Department of Otolaryngology, The Catholic University of Korea School of Medicine, Seoul, Korea, and by the Catholic Medical Center Research Foundation made in the program year 2009.
Footnotes
Results in this paper were presented at the 2010 ARO midwinter meeting held in Anaheim, USA, Feb. 2010.
No potential conflict of interest relevant to this article was reported.
References
- 1.Kocher W. Research on the genetics and pathology of the development of delayed hereditary deafness in the mouse (Mus musculus) Arch Ohren Nasen Kehlkopfheilkd. 1960;177:108–145. [PubMed] [Google Scholar]
- 2.Johnson KR, Erway LC, Cook SA, Willott JF, Zheng QY. A major gene affecting age-related hearing loss in C57BL/6J mice. Hear Res. 997 Dec;114(1-2):83–92. doi: 10.1016/s0378-5955(97)00155-x. [DOI] [PubMed] [Google Scholar]
- 3.Siemens J, Lillo C, Dumont RA, Reynolds A, Williams DS, Gillespie PG, et al. Cadherin 23 is a component of the tip link in hair-cell stereocilia. Nature. 2004 Apr 29;428(6986):950–955. doi: 10.1038/nature02483. [DOI] [PubMed] [Google Scholar]
- 4.Spongr VP, Flood DG, Frisina RD, Salvi RJ. Quantitative measures of hair cell loss in CBA and C57BL/6 mice throughout their life spans. J Acoust Soc Am. 997 Jun;101(6):3546–3553. doi: 10.1121/1.418315. [DOI] [PubMed] [Google Scholar]
- 5.Ichimiya I, Suzuki M, Mogi G. Age-related changes in the murine cochlear lateral wall. Hear Res. 000 Jan;139(1-2):116–122. doi: 10.1016/s0378-5955(99)00170-7. [DOI] [PubMed] [Google Scholar]
- 6.Li HS, Hultcrantz M. Age-related degeneration of the organ of Corti in two genotypes of mice. ORL J Otorhinolaryngol Relat Spec. 994 Mar–Apr;56(2):61–67. doi: 10.1159/000276611. [DOI] [PubMed] [Google Scholar]
- 7.Schuknecht HF, Gacek MR. Cochlear pathology in presbycusis. Ann Otol Rhinol Laryngol. 993 Jan;102(1 Pt 2):1–16. doi: 10.1177/00034894931020S101. [DOI] [PubMed] [Google Scholar]
- 8.Li HS, Borg E. Age-related loss of auditory sensitivity in two mouse genotypes. Acta Otolaryngol. 1991;111(5):827–834. doi: 10.3109/00016489109138418. [DOI] [PubMed] [Google Scholar]
- 9.Bronson RT. Rate of occurrence of lesions in 20 inbred and hybrid genotypes of rats and mice sacrificed at 6 month intervals during the first years of life. In: Harrison DE, editor. Genetic effects on aging II. Caldwell (NJ): Telford Press; 1990. pp. 279–357. [Google Scholar]
- 10.Sha SH, Kanicki A, Dootz G, Talaska AE, Halsey K, Dolan D, et al. Age-related auditory pathology in the CBA/J mouse. Hear Res. 008 Sep;243(1-2):87–94. doi: 10.1016/j.heares.2008.06.001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 11.Youle RJ, Strasser A. The BCL-2 protein family: opposing activities that mediate cell death. Nat Rev Mol Cell Biol. 008 Jan;9(1):47–59. doi: 10.1038/nrm2308. [DOI] [PubMed] [Google Scholar]
- 12.McFadden SL, Ding D, Burkard RF, Jiang H, Reaume AG, Flood DG, et al. Cu/Zn SOD deficiency potentiates hearing loss and cochlear pathology in aged 129,CD-1 mice. J Comp Neurol. 1999 Oct 11;413(1):101–112. [PubMed] [Google Scholar]
- 13.McFadden SL, Ding D, Reaume AG, Flood DG, Salvi RJ. Age-related cochlear hair cell loss is enhanced in mice lacking copper/zinc superoxide dismutase. Neurobiol Aging. 999 Jan–Feb;20(1):1–8. doi: 10.1016/s0197-4580(99)00018-4. [DOI] [PubMed] [Google Scholar]
- 14.Sha SH, Chen FQ, Schacht J. Activation of cell death pathways in the inner ear of the aging CBA/J mouse. Hear Res. 2009 Aug;254(1-2):92–99. doi: 10.1016/j.heares.2009.04.019. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Someya S, Xu J, Kondo K, Ding D, Salvi RJ, Yamasoba T, et al. Age-related hearing loss in C57BL/6J mice is mediated by Bak-dependent mitochondrial apoptosis. Proc Natl Acad Sci U S A. 2009 Nov 17;106(46):19432–19437. doi: 10.1073/pnas.0908786106. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Ohlemiller KK, Gagnon PM. Divergence of hearing sensitivity and endocochlear potential (EP) in CBA/J and CBA/CaJ mice after 12 months of age. Assoc Res Otolaryngol. 2007 Abstract 454. [Google Scholar]
- 17.Hultcrantz M, Li HS. Inner ear morphology in CBA/Ca and C57BL/6J mice in relationship to noise, age and phenotype. Eur Arch Otorhinolaryngol. 1993;250(5):257–264. doi: 10.1007/BF00186222. [DOI] [PubMed] [Google Scholar]
- 18.Anderson SD, Kemp DT. The evoked cochlear mechanical response in laboratory primates: a preliminary report. Arch Otorhinolaryngol. 1979;224(1-2):47–54. doi: 10.1007/BF00455223. [DOI] [PubMed] [Google Scholar]
- 19.Brownell WE. Outer hair cell electromotility and otoacoustic emissions. Ear Hear. 990 Apr;11(2):82–92. doi: 10.1097/00003446-199004000-00003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Willott JF, Erway LC. Genetics of age-related hearing loss in mice: IV. cochlear pathology and hearing loss in 25 BXD recombinant inbred mouse strains. Hear Res. 998 May;119(1-2):27–36. doi: 10.1016/s0378-5955(98)00029-x. [DOI] [PubMed] [Google Scholar]
- 21.Tadros SF, D'Souza M, Zhu X, Frisina RD. Apoptosis-related genes change their expression with age and hearing loss in the mouse cochlea. Apoptosis. 008 Nov;13(11):1303–1321. doi: 10.1007/s10495-008-0266-x. [DOI] [PMC free article] [PubMed] [Google Scholar]