Abstract
There have been numerous types of animal models of choroidal neovascularization (CNV) and retinal neovascularization (RNV). Understanding the pathobiology of CNV and RNV is important when evaluating and utilizing these models. Both CNV and RNV are dynamic processes. A break or defect in Bruchs’ membrane is necessary for CNV to develop. This may be induced with a laser, mechanically via surgery, or in the setting of transgenic mice. Some of the transgenic mouse models spontaneously develop RNV and/or retinal angiomatous proliferation (RAP)-like lesions. The pathogenesis of RNV is well-known and is generally related to ischemic retinopathy. Models of oxygen induced retinopathy (OIR) closely resemble retinopathy of prematurity (ROP). The streptozotocin (STZ) rat model develops features similar to diabetic retinopathy. This review summarizes general categories and specific examples of animal models of CNV and RNV. There are no perfect models of CNV or RNV and individual investigators are encouraged to choose the model that best suits their needs.
Keywords: choroidal neovascularization, retinal neovascularization, animal models
1. Introduction
The development of animal models of choroidal neovascularization (CNV) and retinal neovascularization (RNV) has paralleled and contributed to the understanding of the biology of these conditions. These models have also been developed in order to test new treatments. Although the models recapitulate many of the clinical manifestations of CNV and RNV in humans, the time to develop, course of progression, size and appearance of the lesion vary among the models. In this review, various categories of animal models of CNV and RNV are presented along with specific examples in each category, including their strengths and weaknesses. This review is meant to be comprehensive, but not encyclopedic. It should be used as a guide for investigators to further elucidate the biology of CNV and RNV with emphasis on evaluation of novel treatments for these conditions.
2. Animal Models of Choroidal Neovascularization
2.1 Overview of Choroidal Neovascularization
Choroidal neovascularization (CNV) represents a non-specific response to a specific stimulus (Grossniklaus and Green 2004). CNV is encountered in numerous chorioretinal diseases (Green and Wilson 1986; Green 2006) and is a dynamic process with initiation, maintenance and involution stages (Grossniklaus et al 2002). The involutional stage is characterized by a decrease in cytokine production with associated scarring and fibrosis. The CNV may arise de novo or reactivate in areas of inactive CNV. Our laboratory has estimated that CNV in patients with age-related macular degeneration grows at a rate of approximately 0.2 disc areas (DAs) per month (unpublished data). There are varying degrees of inflammation in the CNV depending on the underlying disease and dynamic stage of CNV evolution (Grossniklaus and Green 2004).
Understanding the pathobiology of CNV is important with regard to developing suitable animal models of the process. The evolution of CNV begins with a break or defect in Bruch’s membrane. This may be secondary to a traumatic break, a degenerative process, tissue traction and/or inflammation (Penfold, Killingsworth and Sarks 1984, Spraul and Grossniklaus 1997, Grossniklaus et al 2000, Kilarski et al 2009). When this occurs, choriocapillary endothelial cells, pericytes, fibrocytes and inflammatory cells are introduced into the sub-retinal pigment epithelium (sub-RPE) and/or sub-retinal spaces. There are inflammatory, angiogenic and extracellular matrix components of the CNV. These components exist together in a microcosm where there are competing factors that induce or suppress the processes. The combination of endothelial cell growth and coverage by pericytes surrounded by the extracellular matrix (ECM) as mediated by vascular endothelial growth factor (VEGF) and platelet derived growth factor (PDGF) seems essential for CNV formation (Greenberg et al 2008).
The systems involved with the formation of CNV may be divided into three general categories: inflammation, angiogenesis and proteolysis. These systems are interrelated. Several inflammatory subsystems have been implicated in CNV, including the complement system, cytokines, and chemokines (Hageman et al 2001, Mullins et al 2000). Pro-angiogenic factors that stimulate the proliferation of endothelial cells in CNV include VEGF, basic fibroblast growth factor, (bFGF) and platelet derived growth factor (PDGF) (Amin et al 1994, Kvanta et al 1996, Lopez et al 1996). The proteolysis component includes a provisional fibrin matrix laid down in the area that will become the physical location of the CNV (Schlingemann 2004). The fibrin acts as a scaffold upon which the CNV will grow. The fibrin arises from conversion of circulating fibrinogen by tissue factor (TF). The TF cell receptor is a co-activator of serine proteases promoting both thrombin and fibrin. TF is also involved in inflammation, cell adhesion and VEGF secretion (Carmeliet and Collen 1998). Although thrombospondin has anti-angiogenic properties, it also enhances release of the pro-angiogenic molecule, matrix metalloproteinase 2 (MMP-2), from the extracellular matrix (Bein and Simons 2000). Cells critical for the pathogenesis of CNV include macrophages and RPE Macrophages appear to play a key role in this process, as they express the angiogenic cytokines VEGF and TNFα (Penfold et al 2001, Grossniklaus et al 2002). The RPE is a critical regulator of the process, as it expresses VEGF, MCP1 and IL8, all of which regulate monocyte and endothelial cell recruitment (Elner et al 1996, Kvanta et al 1996, Lopez et al 1996, Grossniklaus et al 2002). The dynamic stages of CNV (initiation, maintenance and involution) depend on cell signals being turned on or off during the process (Grossniklaus et al 2002). These opposing signals include pro- and anti- inflammatory, angiogenic and proteolytic. Oxidative damage-induced inflammation may initiate age-related macular degeneration (Hollyfield et al 2008) and there appear to be immune-related mechanisms involved with angiogenesis in CNV (Ferguson and Apte 2008). The most widely studied inflammatory cascades in age-related macular degeneration are the complement pathways (Schol et al 2008, Baird et al 2008, Park et al 2009) and there are competing proteins that potentiate or inhibit these pathways. VEGF is the primary initiator of angiogenesis in CNV (Kvanta et al 1996, Lopez et al 1996, Grossniklaus et al 2002). Angiogenic remodeling factors include angiopoeitin 1 and 2, ephrin 1, the tyrosine kinases Tie 1, basic fibroblastic growth factor (bFGF), platelet derived growth factor (PDGF), KDR/Flt-1, and others (Folkman et al 1996, Holash et al 1999, Gal and Yancopoulos 1999, Wada et al 1999). Endogenous angiogenic inhibitors include PEDF, angiostatin and endostatin (Cao 2001). Pigment epithelium-derived factor (PEDF) is a naturally occurring anti-angiogenic protein (Mori et al 2001). Interestingly, PEDF also exhibits anti-inflammatory properties by modulating macrophage activity (Zamiri et al 2006). The MMPs and tissue inhibitors of matrix metalloproteases (TIMPs) are involved with pro- and anti-angiogenesis as well as pro- and anti- tissue degradation (Sven et al 1998, Bergers et al 2000). These dynamic processes of inflammation, angiogenesis, and proteolysis relate to the initiation, maintenance, and involution stages of CNV formation (Grossniklaus et al 2002) and should be kept in mind when assessing animal models of CNV. There are many conditions that lead to CNV in humans, including age-related macular degeneration, ocular histoplasmosis syndrome, myopia and idiopatic CNV. The clinical features of these conditions should also considered when creating animal models of CNV.
In addition to recapitulating the pathobiology of human CNV, it is desirable that animal models of CNV be efficient and reproducible, stable and sustainable over time, exhibit similar pathologic findings including growth patterns to human CNV (Grossniklaus and Green 2004), be inexpensive to produce and be able to be followed in vivo with imaging techniques including fluorescein angiography (FA) and ocular coherence tomography (OCT). There are generally three categories of animal models of CNV: laser induced models, surgically induced models and genetically engineered animals. These models may be modified by introduction of cytokines, age, diet and other factors. The following will summarize pertinent features of each of these types of models for CNV with specific examples including their strengths and weaknesses.
2.2 Laser and Light Induced Models of CNV
2.2.1 Rat and Mouse
After the first models of CNV were developed in primates (Ryan 1979), Dobi and associated created a rat model of CNV in 1989 (Dobi et al 1989). Those authors created argon laser photocoagulation spots (647nm, 100μm 50 to 100 mW, 0.1s) through a dilated pupil with a coverslip over the cornea. The spots created breaks in Bruchs membrane, with a central bubble formation with or without intra-retinal or choroidal hemorrhage. There was fluorescein angiographic evidence of CNV in 24% of the created lesions. Examination of enucleated eyes by light and electron microscopy showed pathologic evidence of CNV in 60% of the lesions. Frank and co-workers also developed a rat model of CNV in 1989 (Frank, Das and Weber, 1989). That model used a krypton laser (blue green or green wavelength, 500μm, 50mW, 0.02s) to create breaks in Bruchs membrane and CNV. Campochiaro’s group and other groups essentially used the same technique to create experimental CNV in mice, with minor modifications to the laser spot parameters. For instance, using a krypton laser in C57BL6 mice with modified settings including a smaller spot size, higher intensity, and shorter duration (blue green or green wavelength, 50μm 50 to 350 to 400mW 0.05s) resulted in a higher frequency (100%) of histologic evidence of CNV (Tobe et al 1998, Seo et al 1999). The rat laser induced CNV model has been used to study the role of tissue inhibitor metalloproteinase 3 (Takahashi et al 2000) and tissue factor (Bora et al 2003) in CNV formation. It has also been used in pharmacologic intervention experiments (Yanagi et al 2002). VEGF has been shown to be the major stimulator of CNV in the mouse model (Figure 1) (Kwak et al 2000). Immunohistochemical staining in this model, especially in the context of transgenic mice, has been used to study various components of CNV including basic fibroblast growth factor 2 (FGF2) (Tobe et al 1998) and matrix metalloproteinases (Berglin et al 2003). Image analysis software may be used to analyze the histologic area of CNV in these models, especially in the context of testing potential pharmacologic therapies of CNV (Seo et al 1999). The model has evolved to where the current standard of measurement is determining the area of CNV by confocal scanning electron microscopy image analysis of flat-mounts of the CNV/RPE and choroid after intravenous infusion with fluorescently labeled dextran. A diode laser may be used to create the CNV (532 nm, 100μm 50 to 100 mW, 0.1s) and this model has been used to assess aging as it relates to CNV formation (Espinosa-Heidmann et al 2002), macrophage contribution to CNV formation (Sakurai et al 2003, Espinosa-Heidmann et al 2003), and gene therapy (Bainbridge et al 2003), siRNA targeting (Reich et al 2003), novel delivery platforms, such as AAV mediated transfections (Mori et al 2002) and nanoparticles (Singh et al 2009) in the treatment of CNV. Advantages include that the model is relatively simple to create, inexpensive, reproducible, efficient, may be used in conjunction with immune manipulation (Bora et al 2005) or transgenic mice to elucidate the pathobiology of CNV (Sengupta et al 2003, Rakic et al 2003), and experiments may be done in short time frames, such as several weeks. Disadvantages include the artificial nature of the model, the small size of the rat/mouse eye compared to the human eye, mice and rats do not have a macula, and laser photocoagulation has been used to treat CNV as well as create it. Nevertheless, the rat/mouse laser model of CNV is the currently the standard animal model of CNV for most treatment evaluation experiments. Recently, Albert and co-workers described CNV formation in a cyclic light induced rat model (Albert et al 2010). In that model, Albino rats were exposed to 12 hours of 3000-lux cyclic light for 1, 3 or 6 months. Results showed microscopic sub-RPE neovascularization at 1 month, extension of the neovascularization into the outer retina at 3 months, and multiple areas of neovascularization with associated connects with retina vessels and tissue bands at 6 months. The retinas of the light-exposed animals immunostained for 4-hydroxy-2-nonenal (HNE), a measure of ω-6 polyunsaturated fatty acid peroxidation, indicating oxidative stress. The cyclic light also induced other retinal atrophy and drusen-like deposits. An advantage to this model is the potential to study the biologic progression of CNV in non-genetically altered animal model without laser or mechanical perforation of Bruch’s membrane.
Figure 1. Mouse laser model of CNV.
A. Control mouse with laser induced CNV (between arrows). B. A mouse is treated with a drug that inhibits VEGF and PDGF develops smaller CNV (between arrows) after laser induction. (From Kwak et al, 2000) C. A flat mount preparation of laser induced CNV in the mouse after perfusion with fluorescent dextran shows the horizontal extent of the CNV. (From Singh et al, 2009)
2.2.2 Primate, rabbit and pig
The primate model of experimental CNV, created by Ryan and colleagues, was termed “subretinal neovascularization” at the time and was the first animal model of CNV (Ryan 1979, Ryan 1982, Miller, Miller and Ryan 1986). Original work in this area included transscleral and transvitreal injection of collagenase and hyalurondiase into the subretinal space (Ryan 1979). This resulted in mechanical or enzymatic damage to Bruchs membrane. These invasive techniques resulted in low incidences of CNV and led to the development of laser disruption of Bruchs membrane as a model of CNV. Ryan was able to induce CNV in Macaca speciosa monkeys using argon laser (488nm and 514nm, 50 to 200μm, 200-950mW, 0.1 to 0.5s) disruption of Bruchs membrane. Approximately 40% of the monkeys developed angiographic evidence of CNV and up to 90% developed histologic/ultrastructural evidence of CNV. These studies provided early experimental evidence of the role of ischemia (later studies showing VEGF in response to ischemia), inflammation including macrophages, and the RPE (Miller, Miller and Ryan 1986) in the role of CNV formation. The primate laser model of CNV played an essential role in the development of photodynamic therapy as a treatment for CNV (Miller et al 1995). It has also been used, in conjunction with a surgically induced rabbit model of CNV to evaluate an integrin inhibitor to treat CNV (Zhan et al 2009). Criswell and co-workers created CNV in new-world squirrel monkeys and compared them with CNV created in old-world cynomolgus Macaca speciosa monkeys (Criswell et al 2004). With their optimized laser parameters for the squirrel monkeys (532nm, 0.05s, 75μm, 650mW), they elicited fibrovascular tissue (CNV) in 65% of squirrel monkey laser spot sites compared with 37% of cynomolgus laser spot sites (Figure 2). Many of these CNV lesions expanded beyond the size of the original photocoagulation sites including diffuse subretinal fibrovascular tissue. Advantages of the primate model include the close approximation of the monkey retina and macula to the human, size of the primate eye for drug delivery studies, and utility of the model for development of human clinical trials (such as for PDT and anti-angiogenics). Disadvantages include the expense of the animals/animal care/husbandry, length of experiments and ethical issues regarding use of primates when rodents (mice and rats) are reliable models. An intermediate laser-induced CNV model between rodents and primates has been developed. This is the rabbit model of laser-induced CNV (El Dirini et al 1991). This model avoids the expense and ethical issues of the primate model, although the rabbit lacks a macula and the rabbit retina is supplied by a medullary ray, unlike the vascular supply of the retina to primates and rodents. The model utilized subretinal endophotocoagulation to create histologically identified CNV. Another intermediate laser-induced CNV model is the pig (Saishin et al 2003, Kiilgaard et al 2005). Saishin and coworkers used a diode laser (75μm, 400mW, 0.1s) to create defects in Bruchs membrane in the pig eye and establish histologic evidence of CNV in 100% of lesions. Image analysis of histologic sections was used to evaluate drug delivery for the treatment of the CNV (Saishin et al 2003). Kiilgaard and coworkers studied xenon laser versus diode laser versus mechanical disruption of Bruchs membrane in a pig model of CNV (Kiilgaard et al 2005). They found histopathogic evidence of CNV in 54%, 83% and 100% of the animals, respectively (Kiilgaard 2005). The pig eye is approximately the same size as the human eye and the retina vascularization is similar to the human. Like the rabbit, the pig circumvents the cost and ethical issues of the primate. The utility of the pig is for drug delivery experiments. However, the pig lacks the advantages of high throughput, short duration experiments as available in rat and mouse models of laser induced CNV.
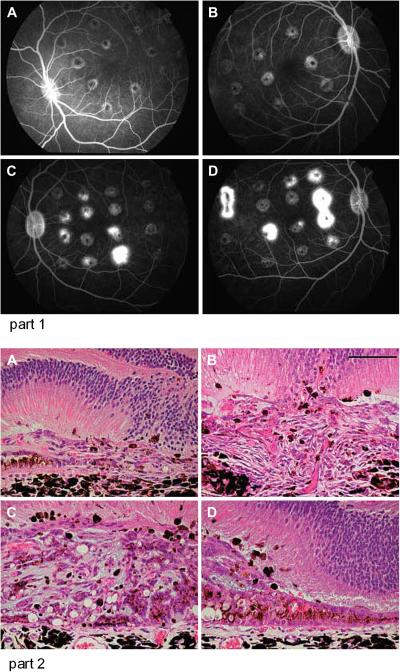
Figure 2. Cynomolgus monkey laser model of CNV.
Part 1. Fluorescein angiography 21 days after laser induction of CNV shows partial localized (A and B) or mixed localized and diffuse CNV (C and D) Part 2. Histologic examination of enucleate eyes shows CNV between the retina and a reflected layer of RPE (A), extension of the CNV into the retina (B), and numerous vascular channels within the CNV which also contains pigmented macrophages (C and D). (From Criswell et al, 2004)
2.3 Surgically Induced Models of CNV
2.3.1 Rat and Mouse
Subretinal and/or choroidal neovascularization has been immunologically and mechanically induced in rat and mouse models, primarily by injection of synthetic peptides, viral vectors containing VEGF, cells and inert synthetic materials. Chan and colleagues reported and illustrated subretinal neovascularization in 10% of mice immunized with interphotoreceptor retinoid-binding protein (IRB) and developed experimental autoimmune uveoretinitis (EAU) (Chan et al, 1990). Sakamoto and co-workers immunized Lewis rats with a hind foot pad injection of an IRBP-derived synthetic peptide and intravenous Bordetella persusis (Sakamoto et al 1994). They found histologic evidence of inflammatory destruction of Bruchs membrane and subretinal neovascularization in 38% of the rats. This model may be similar to ocular inflammatory diseases that result in subretinal neovascularization, such as punctate inner choroidopathy, Harada disease, and others, and supports the concept that inflammatory disruption of Bruchs membrane may be associated with CNV. Spilbury and co-workers performed injections of a recombinant adenovirus vector expressing rat VEGF164 driven by a CMV promotor into the subretinal space of rats (Spilsbury et al 2000). They found fluorescein angiographic and histologic evidence of CNV in 80% of the eyes, although they could not exclude the possibility of needle puncture to Bruchs membrane as helping to cause the CNV. Baffi and co-workers injected 5 to 10 μl of an adenoviral vector expressing VEGF165 into the subretinal space of the rat eye (Baffi et al 2000). Examination by fluorescein angiography, histology and electron microscopy showed CNV in most of the eyes. Those authors used the FITC-labeled dextran retinal flatmount technique to examine some specimens, which is now the standard technique. Wang and colleagues performed similar studies, using a 2μl injection of an adenoviral vector expressing human VEGF165 (or GFP) with a CMV promoter into the subretinal space in rats (Wang et al 2003). Their extensive study demonstrated VEGF expression in the RPE and angiographic/histologic evidence of CNV in approximately 90% of the injected rats. Additionally, electroretinogram (ERG) a and b waves were reduced by approximately 50% in the rats with CNV compared with controls. It is likely that the subretinal injection itself causes trauma to Bruchs membrane sufficient for CNV. Our laboratory has induced choroidal neovascularization in C57BL6 mice by subretinal injection of retinal pigment epithelium and polystyrene microbeads (Figure 3) (Schmack et al 2009). In our study, we found that the combination of RPE, which expressed VEGF, and polystyrene beads, resulted in larger CNV lesions than RPE or the polystyrene beads alone. Additionally, subretinal injection of PBS resulted in small CNV lesions, thus supporting the concept that the trauma of subretinal injection alone damages Bruchs membrane and may result in CNV. Subretinal/RPE injection of matrigel, a basement membrane extract, that solidifies after implantation in tissue and stimulates local angiogenesis, is reported to induce CNV in B6 and Ccl2 deficient mice with 31% and 53% incidences, respectively. That report supports the involvement of macrophages and chemotaxis in CNV (Shen et al, 2006).
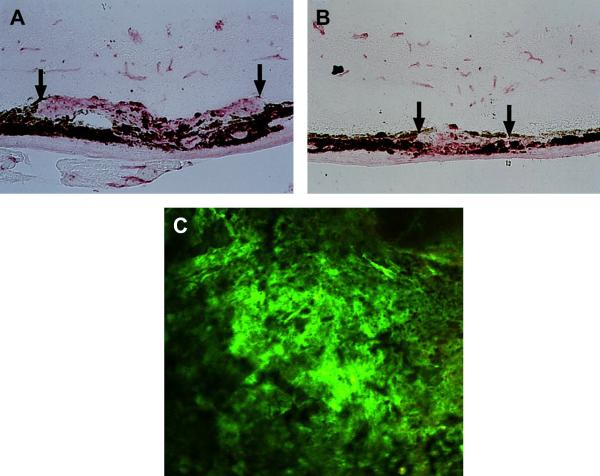
Figure 3. Mouse surgical model of CNV.
A. After subretinal injection of RPE and polystyrene beads, CNV is located between the retina and a reflected layer of RPE. Vascular channels are indicated by arrows. B. There are vascular channels (arrows), mononuclear inflammatory cells, and beads (asterisks) located within the CNV. C. A fluorescent labeled dextran flatmount preparation shows the extent of the CNV. (From Schmack et al, 2009)
2.3.2 Rabbit
Some of the most extensive work regarding the creation of CNV after subretinal injections has been performed in rabbits. Early studies showed that subretinal injection of vitreous would induce retinal pigment epithelial proliferations and microscopically identified CNV in rabbit eyes (Zhu et al, 1989). In 1995, Kimura and co-workers injected 50μl suspensions of 2.5 μl bFGF impregnated gelatin microspheres into the subretinal space in rabbits via a transscleral route (Kimuira et al 1995). They found angiographic and histologic evidence of CNV in 83% to 100% of the eyes (Kimura et al 1995, Kimura et al 1999). The injection technique created a break in Bruchs membrane in their model. The cellular response in that model was studied by immunohistochemistry, which showed CD31 positive endothelial cells first present at 14 days after inoculation and forming neovascular channels that persisted for up to 8 weeks (Kimura et al 1999). Other studies have shown that a break in Bruchs membrane is needed for traumatically induced CNV in the rabbit (Hsu et al 1989). Tamai and co-workers found that subretinal injections of 12.5 to 25μg of lineloic acid hydroperoxide (LHP) into the subretinal space in rabbit eyes induced CNV (Tamia et al 2002). They demonstrated angiographic evidence of CNV in 46% of the eyes, and TNFα, PDGF, VEGF, TGFβ, bFGF and IL1α were upregulated by the LHP. The areas of the CNV in Kimura’s and Tamai’s models were relatively small. There have been several models created that produce large areas of CNV in the rabbit eye. Ni and coworkers injected a 50μl cocktail of endotoxin, growth factor, heparin beads, FGF-2, 100ng LPS with 50μg of heparin sepharose beads transvitreally into the subretinal space of the rabbit eye (Figure 4) (Ni et al 2005). Fluorescein angiography, ocular coherence tomography (OCT) and histology demonstrated two types of extensive CNV growth in 100% of the eyes. Primary CNV extending into the subretinal space and associated with injury to Bruchs membrane at the injection site was visible by 2-weeks and stable at 3-months post-injection. Secondary CNV extending into the subRPE space away from the injection site developed in all eyes by 8-months post-injection. The CNV persisted for up to 3 years. The CNV was characterized by initiation, growth and maintenance stages, similar to human CNV (Grossniklaus et al 2002), and was inhibited by subcutaneous dexamethasone. Qui and coworkers performed transvitreal injections of 10μl of Matrigel and 750μg of VEGF into the subretinal space of the rabbit eye (Qui et al 2006). In addition to fluorescein angiography and histology, they used OCT to demostrate CNV in 100% of eyes at 1 to 9 weeks post-inoculation. Unlike Ni’s model, the CNV in Qui’s model was relatively small. Qui and coworkers noted that the Matrigel acted as a scaffold upon which the CNV grew. Julien and coworkers performed transvitreal injections of 5×106 IU of a high capacity adenoviral vector construct expressing VEGF-A165 driven by an EF1αHTLV promoter into the subretinal space of the rabbit eye (Julien et al 2008). Those investigators found angiographic evidence of CNV in 85% of the injected eyes, which was confirmed by histology, immunohistochemistry and electron microscopy. ICG angiography and electron microscopy showed intra-choroidal changes in the model. The CNV in that model, like Qui’s, was relatively small compared with Ni’s model. An advantage to the rabbit model is that potential large size and long duration of the experimentally induced CNV. Disadvantages include a lack of ability for high throughput experiments, lack of a macula and lack of retinal vasculature similar to the human eye.
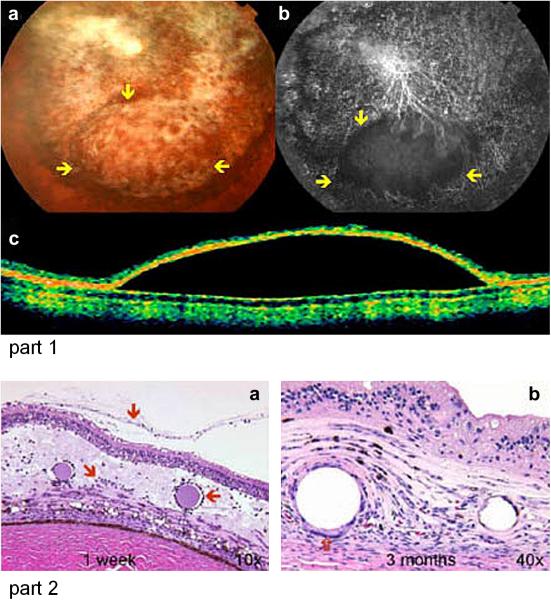
Figure 4. Rabbit surgical model of CNV.
Part 1. A color fundus photograph (a), fluorescein-dextran angiogram (b) and OCT show that there is a separation of the neurosensory from the underlying Bruchs membrane and choroid. Part 2. CNV is present in the subretinal space 1 week (a) and 3 months (b) after subretinal injection of 100 ng FGF-2/100 ng LPS. There are inflammatory cells present (arrows) in the retina, CNV, and surrounding Heparin-sepharose beads at the injection area in the 1 week old CNV. There are giant cells (open arrow) in the 3 month old CNV. (From Ni et al, 2005)
2.3.3 Pig
The mechanically-induced pig model of CNV has mainly been developed in Denmark (Kiilgaard et al 2005, Lassota et al 2006, Lassota et al 2007, Lassota 2008, Lassota et al 2008). Earlier work by Sourbane and coworkers showed that suprachoroidal injection of basic fibroblast growth factor caused intrachoroidal angiogenesis, but not CNV, since there was no break in Bruchs membrane (Sourbane et al 1994). As previously mentioned, Kiilgaard and coworkers showed that in pigs, CNV arose after xenon laser photocoagulation, diode laser photocoagulation and surgical debridement of the RPE followed by mechanical rupture of Bruchs membrane in 54%, 83% and 100% of eyes, respectively, thus confirming that a break in Bruchs membrane is needed for CNV development (Kiilgaard et al 2005). The mechanical model was induced by transvitreal subretinal debridement of the RPE with a retinal scraper after retinal detachment with subretinal injection of isotonic saline. Further investigation of that mechanically induced model by fundus examination, fluorescein angiography, and histology showed correlation of the location of the fibrovascular tissue (CNV) with fundus and angiographic findings (Figure 5) (Lassota et al 2006). Studies showed histologic evidence of CNV in 100% of eyes using a technique in which Bruchs membrane was perforated and the RPE was left intact compared with RPE removal alone and RPE removal/Bruchs perforation (Lassota et al 2007). Although all three techniques resulted in CNV, perforation of Bruchs without prior RPE removal was the easiest to reproduce and involved the fewest variables. Mechanical disruption of Bruchs membrane with a retinal perforator alone was found to reliably produce relatively large areas of CNV in the pig model. The fundus, fluorescein angiographic, and histologic aspects of that model were studied over an extended period of time (Lassota 2008, Lassota et al 2008). The lesion was angiographically visualized in 92% of eyes and the vasculature of the pig retina more closely approximates the human retina than does the rabbit retina (Lassota 2008, Lassota et al 2008). Immunohistochemical studies showed cytokine production and configuration of the CNV to be analogous to human CNV. Additionally, the pig eye, like the rabbit eye, is large enough to perform for drug delivery experiments. Disadvantages are similar to the rabbit model of mechanically induced CNV.
Figure 5. Pig surgical model of CNV.
Part 1. A fundus photograph (upper left) and fluorsecein angiogram early (upper right), middle (bottom left) and late (bottom right) phases show a yellow, early hyperfluorescent with late leakage CNV lesion in the pig model with perforation of Bruchs membrane only. Part 2. Histology and immunohistochemical staining for CD34 that shows vascular channels shows tha the CNV lesion is smaller and has fewer vascular channels after RPE removal/Bruchs perforation (upper and lower left) compared with Bruchs membrane perforation alone (upper and lower right). (From Lassota et al 2007)
2.3.4 Primate
Ryan originally attempted to create CNV in a primate model by subretinal injection of collagenase and hyaluronidase into the subretinal space (Ryan 1979). Cui and colleagues further explored the feasibility of a primate CNV model using subretinal injections (Cui et al 2000). That group injected an 80μl suspension of VEGF-impregnated gelatin mcirospheres transvitreally through a retinotomy into the subretinal space of Macacca mulatta monkeys. They were able to find fundus and angiographic evidence of CNV in 92% of the injected eyes. The CNV was relatively small, and there has been limited use of this model due to expense and ethical issues. This model is more difficult to produce and does not offer any advantages over the laser CNV model in the primate.
2.4 Transgenic and Knockout Mouse Models of CNV
2.4.1 VEGF164RPE65
Although there are several transgenic mouse models of age-related macular degeneration (AMD) (Rakoczy et al 2006), only a relatively few of the models spontaneously develop CNV. It has become apparent that overexpression of VEGF by the retina or RPE is not enough to elicit CNV in these models and there is a central role of compromised Bruchs membrane in the development of CNV (Rakoczy et al 2006). The advantages of these models are the ability to study various biologic components of CNV by comparison with controls and cross breeding experiments. Disadvantages relate to the length of time for the CNV to develop, the relatively small percentages of eyes that develop CNV and the small size of the CNV. These models have supported the concept that the best models of CNV are those that incorporate physical disruption of Bruchs membrane into the model (Rakoczy et al 2006). Schwesinger and colleagues described a transgenic mouse expressing VEGF164 driven by an RPE65 promoter (Schwesinger et al 1994). All of the mice developed histologically identified intrachoroidal vascular anomalies, which were described as intrachoroidal neovascularization, although they did not develop CNV. Some consider the intrachoroidal vascular anomalies in this model to represent developmental (vasculogenic) changes, and not neovascularization.
2.4.2 Ccr2/Ccl2 deficient
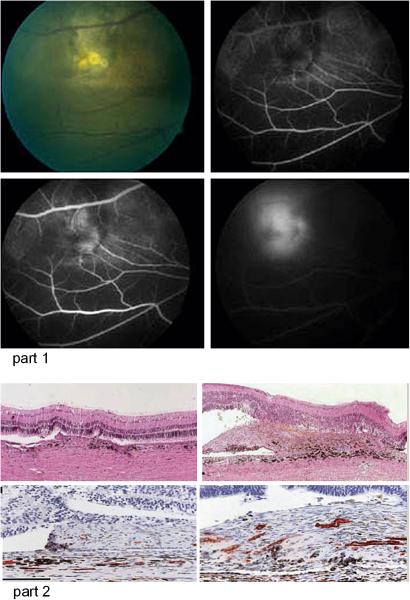
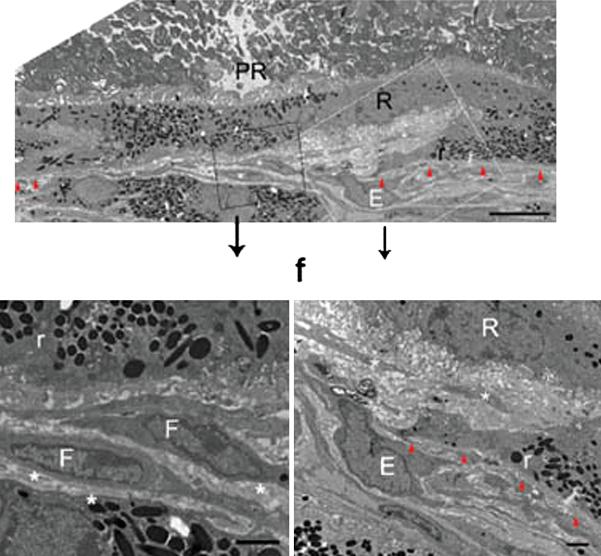
The Ccr2/Ccl2 deficient mouse model of AMD, developed by Ambati and co-workers, has received considerable attention (Ambati et al 2003,Takeda et al 2009). In that model, transgenic mice deficient in either Ccl2 or Ccr2 fail to recruit macrophages to the area of the RPE and Bruchs membrane. This allows for accumulation of C5a and IgG, both of which induce VEGF production. Histologic, immunofluorescent and ultrastructural examination of either of these transgenic mice showed that approximately 25% of the mice developed microscopic areas of CNV (Ambati et al 2003). These findings have helped with the understanding of the pathobiology of CNV, expecially with regard to macrophage recruitment. Although the areas of CNV are very small, recent work has shown that CCR3 is specifically expressed in choroidal neovascular endothelial cells in either of these deficient mice (Takeda et al 2009). In vivo imaging of CCR3-targeted quantum dots locate the CNV that is not able to be visualized by standard fluorescein angiography (Figure 6) (Takeda et al 2009). This is a good example of how an animal model of CNV may translate to the possibility of imaging and possibly treating human CNV.
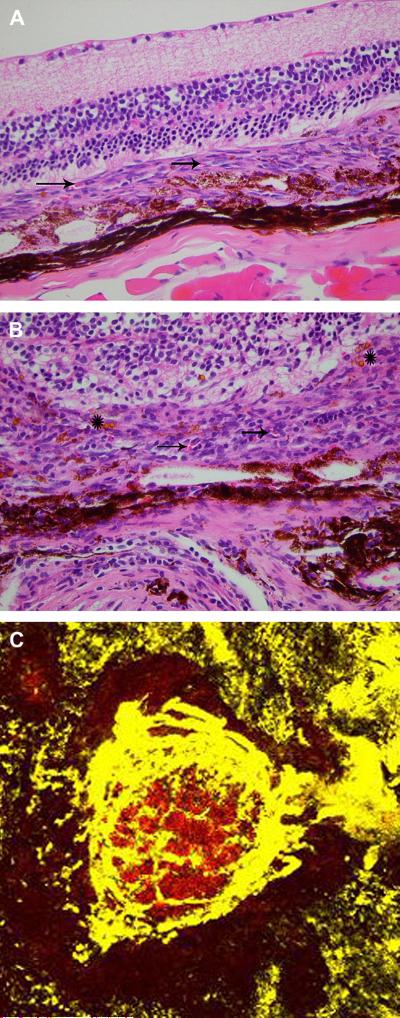
Figure 6.
Mouse CNV in Ccr2/CCl2 transgenic model Electron micrographs of CNV shows endothelial cells (E) and fibrocytes (F) from the choroid breaking through Bruchs membrane and extending into the sub-RPE (R) space. Photoreceptors (PR) are present internal to the RPE. (From Ambati et al, 2003)
2.4.3 ApoE overexpression
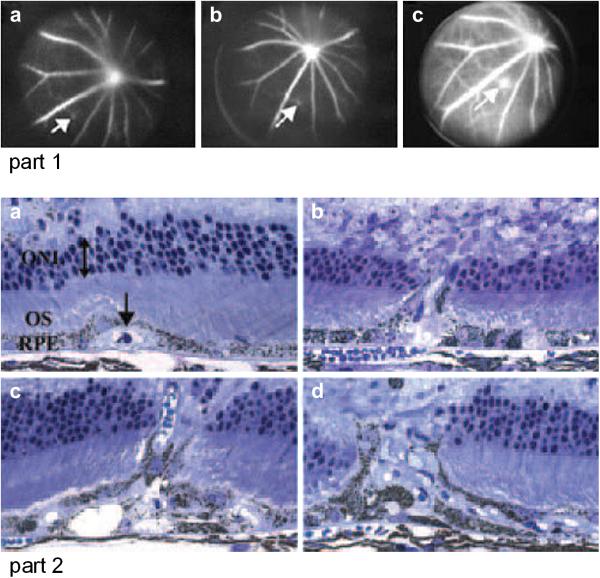
Our laboratory and others have demonstrated AMD-like changes in Bruchs membrane and the area of the RPE in hypercholesterolemic mice (Dithmar et al 2000). Interestingly, Malek and co-workers have shown that in ApoE4 over-expressing transgenic mice fed a high-fat cholesterol rich diet and allowed to age 65-127 weeks developed CNV in addition to drusen-like and basal laminar-like depostis (Figure 7) (Malek et al 2005). The CNV developed in 19% of male and 18% of female mice and was demonstrated by fluorescein angiography, histology, immunohistochemistry, and electron microscopic examination. This model is an important model to study mechanisms of spontaneous CNV formation in the setting of AMD-like lesions.
Figure 7. Mouse ApoE overexpression transgenic model of CNV.
Part 1. Early (a), mid (b) and late-phase (c) fluorescein angiogram demonstrates area of hyperfluorescence and leakage indicative of CNV. Part 2. Histology showing CNV under RPE (a), extending from the sub-RPE space into the retina (b and c), and extensive CNV (d). (From Malek et al, 2005)
2.4.4 Cp−/− Heph-/Y knockout
Hahn and co-workers have shown that disruption of ceruloplasmin and hephaestin causing iron overaload causes AMD-like changes in transgenic mice. These mice developed RPE changes and photoreceptor degeneration. The mice developed funduscopically evident lesions at the level of the RPE and 100% of the mice developed histologically identified subretinal neovascularization, although it is not apparent if the neovascularization arose from the retina or choroid (Hahn et al 2004). Additionally, the mice do not appear to develop the drusen-like or basal laminar deposit-like lesions as seen in AMD.
2.4.5 Spontaneous Bst chromosome 16 mutant
The ocular histopathologic findings in the spontaneous Bst chromosome 16 mutant mouse have been extensively studied by Smith and coworkers (Smith et al 2000). This mouse was found to spontaneously develop retinal lesions identified by fundus examination in the Jackson Laboratories in Bar Harbor, Maine. The spontaneous belly spot and tail (Bst) mutation arose in C57BLKS inbred strains of mice. The eyes of the mice were studied by fundus examination, fluorescein angiography and histology. Approximately 88% of the mice developed histologically identified subretinal neovascularization. The neovascularization was associated with RPE abnormalities, retinal hamartoma-like lesions, and connections of the neovascularization with the retina and choroid through defects in Bruchs membrane. Although the neovasuclarization was age-related, there were no drusen-like or basal laminar deposit-like lesions in this mouse, thus the neovascularization appears to be more similar to that seen in retinal angiomatous proliferation (RAP) than CNV in AMD.
2.4.6 Vldlr −/− targeted mutant
This mouse was developed by Dr. Joachim Herz at the University of Texas, Southwestern. These are homozygous strains of mice with targeted mutations for very low density lipoprotein receptor gene (VldlrTm1Her) The mice develop new blood vessels in the area of the outer plexiform layer of the retina and choroidal anastomoses by 3 months (Heckenlively et al 2003). Like the spontaneous Bst mouse, these appear similar to RAP lesions.
2.4.7 Ccl2/Cx3cr1-deficient mice
Chan and coworkers have generated a Ccl2/Cx3cr1 deficient double knockout (DKO) transgenic mouse that developed increased N-retinylidene-N-retinylethanomalmine (A2E) and decreased ERp29 levels in the RPE (Chan et al 2008). These mice, when fed a diet low in omega-3 polyunsaturated fatty acids (PUFAs), developed funduscopically and histologically-ultrastructurally visible RPE changes and drusen-like lesions. Approximately 15% of the mice developed histologic evidence of neovascularization (Figure 8). The lesions in this mouse develop in 6 weeks, a short time span in the transgenic mouse world, thus making it a potentially attractive model for spontaneous development of neovascularization in a transgenic mouse, although further work is warranted to study the nature of this neovascularization.
Figure 8. Mouse Ccl2Cx3cr1 transgenic mouse model of subretinal neovascualrization.
There is neovascularization in the deep retina with unvolvement of the underlying RPE (A). These vascular channels (arrow) are present under the photoreceptors and appear to arise from the area of the RPE (arrow) (B). Courtesy of Chi Chao Chan MD
2.4.8 Tet/VMD2/VEGF & Tet/VMD2/VEGF/Ang2
In a sophisticated set of experiments, Oshima and coworkers studied CNV in double (Tet/VMD2/VEGF) and triple (Tet/VMD2/VEGF/Ang2) transgenic mice (Oshima et al 2004). Adult mice with increased expression of VEGF in the RPE had normal retinas and choroids and did not develop CNV. However, subretinal injection of an adenoviral vector containing an expression construct for Ang 2 resulted in histologically identified CNV in 100% of the Tet/VMD2/VEGF mice whereas Tet/VMD2/VEGF/Ang2 triple transgenic mice did not spontaneously develop CNV. The investigators used immunohistochemistry and flourescein dextran perfused flatmounts to demonstrate the presence and size of CNV. This model provides the important information that overexpression of VEGF is not sufficient for the development of CNV; there must be mechanical and/or other factors at the level of Bruchs membrane to induce CNV. Exogenously administered tetracycline or doxacycline turns of the genetic construct in the transgenic animal. One caveat is that doxacycline downregulates CNV and animal models using Tet-on/Tet-off for CNV should be carefully evaluated.
2.4.9 Rhodopsin promoter VEGF overexpression
Okamoto, Tobe and colleagues generated rhodopsin promoter/VEGF fusion gene transgenic mice (Okamoto et al, 1997, Tobe, Okamoto et al 1998). Progeny of these mice crossbred with C57BL/6 mice developed angiographically visible intraretinal neovascularization that extended into the subretinal space. This lesion, similar to RAP, demonstrated that overexpression of VEGF in the retina is sufficient to cause intraretinal and subretinal neovascularization. It is possible that neovuascularization my originate in the choroid of the animal as well as the retina thus resulting in chorio-retinal anastamosis. Some investigators choose to use both a laser-induced CNV model, such as in a rat or mouse, and a transgenic model, such as this rhodopsin promoter/VEGF mouse, when evaluating new treatments for CNV.
2.4.10 senecient SOD1−/ − mice
Imamura and co-workers studied the fundus, fluorescein angiographic, histologic, ultrastructural and immunohistochemical features of the retina, RPE, Burch’s membrane and choroidal areas of Cu, Zn-superoxide dismutase (SOD1) deficient mice (Imamura et al, 2006). These investigators found that there was fundus and histologic evidence of CNV in 8.3% and 10% of these senecient Sod−/− mice, respectively. A marker of oxidative damage to DNA, 8-hydroxy-2′-2-deoxyguanosine (I-OHdG) was detected in the RPE of senescent Sod−/− but not in controls. The CNV appeared to connect with retinal vessels in 16 month old compared with 12 month old mice.
2.5 Summary of Animal Models of CNV
Representative animal models of CNV are shown in Table 1. There is no ideal animal model of CNV. The models studied to date indicate that VEGF overexpression is insufficient for the development of CNV; there needs to be a compromise to Bruchs membrane, either mechanically or biologically induced, to induce CNV. Furthermore, there appears to be a central role in Bruchs membrane in the formation of human CNV. All models require compromise to Bruchs membrane, VEGF expression and inflammatory cytokine mediation. The emphasis of which plays a predominant role in the CNV formation depends on the model. For instance, some models are based on laser and/or mechanically induction of a break in Bruchs membrane, which is the dominant feature in those models. Some models are based on over-expression of VEGF, either by subretinal introduction of VEGF or materials impregnated with VEGF, or transgenic animals with over-expression of VEGF driven by retina or RPE specific promoters. Other models are based on inflammatory cytokine production as the dominant feature causing CNV, such as those that utilize subretinal injection of biomaterials and possibly those based on oxidative stress. Investigators should be aware of the relative importance of the various components of CNV formation (i.e. mechanical injury to Bruchs membrane, VEGF production, inflammatory cytokine mediation) when utilizing an animal model of CNV.
Table 1.
Animal Models of Choroidal Neovascularization
| reference | animal | method | success rate |
quantification | mechanism |
|---|---|---|---|---|---|
| Dobi et al 1989 | rat | krypton laser | 24%-60% | FA-path | Bruchs |
| Frank et al 1989 | rat | krypton laser | 43% | path | Bruchs |
| Tobe et al 1998 | mouse | krypton laser | 57% | path | Bruchs |
| Seo et al 1999 | mouse | krypton laser | 100% | path | Bruchs |
| Albert et al 2010 | rat | cyclic light | n/a | path | cytokines |
| Ryan 1979 | monkey | argon laser | 40-90% | FA-path | Bruchs |
| Criswell et al 2004 | monkey | diode laser | 37-65% | path | Bruchs |
| El Dirini et al 199 | rabbit | subretinal argon laser | 100% | path | Bruchs |
| Saishin et 2003 | pig | diode laser | 100% | path | Bruchs |
| Kiilgaard et al 2005 | pig | xenon or diode laser | 54% to 83% | path | Bruchs |
| Chan et al 1990 | mouse | immunization | 10% | path | cytokines |
| Sakamoto et al 1994 | rat | immunization | 38% | path | cytokines |
| Spilsbury et al 2000 | rat | subretinal injection | 86% | path | VEGF |
| Baffi et al 2000 | rat | subretinal injection | most eyes | path | VEGF |
| Wang et al 2003 | rat | subretinal injection | 90% | path | VEGF |
| Schmack et al 2009 | mouse | subretinal beads | 100% | path | cytokines |
| Shen et al 2006 | mice | subretinal matrigel | 31% to 53% | path | cytokines |
| Zhu et al 1989 | rabbit | subretinal vitreous | 31% | path | cytokines |
| Kimura et al 1995 | rabbit | subretinal microspheres | 83%-100% | FA-path | cytokines |
| Hsu et al 1989 | rabbit | penetrating retinal injury | n/a | path | Bruchs |
| Tamai et al 2002 | rabbit | subretinal linoleic acid | 46% | FA | cytokines |
| Ni et al 2005 | rabbit | transvitreal subretinal beads | 100% | FA, OCT,path | cytokines |
| Qui et al 2006 | rabbit | subretinal matrigel/VEGF | 100% | FA | cytokines |
| Julien et al 2008 | rabbit | transvitreal subretinal VEGF | 85% | FA | VEGF |
| Lassota et al 2006 | pig | subretinal Bruchs perforation | 100% | path | Bruchs |
| Cui et al 2000 | monkey | subretinal microspheres | 92% | FA | VEGF |
| Ambati et al 2003 | mouse | transgenic | 25% | path | cytokines |
| Malek et al 2000 | mouse | transgenic | 18-19% | FA/path | cytokines |
| Hahn et al 2004 | mouse | knockout | 100% | path | cytokines |
| Smith et al 2000 | mouse | spontaneous mutant | 88% | path | Bruchs |
| Heckenlively et al 2005 |
mouse | targeted mutant | n/a | n/a | Bruchs |
| Chan et al 2008 | mouse | transgenic | 15% | path | cytokines |
| Oshima et al 2004 | mouse | transgenic | 100% | path | Bruchs |
| Okamoto et al 1997 | mouse | transgenic | n/a | FA | VEGF |
| Imamura et al 2006 | mouse | transgenic | 8.3%-10% | path | cytokines |
n/a=not available; FA=fluorescein angiography; path=pathology; OCT=ocular coherence tomography
Each currently available model has advantages and disadvantages as mentioned above. Considerations when choosing an animal model are related to what the model will be used for. Small, reproducible models, such as the mouse laser-induced CNV model, may be chosen for high throughput drug screening studies. These models are reasonably consistent and result in CNV in approximately 80% of lesions. Laser induction of CNV may be performed in transgenic mice as well, thus providing the ability to study various contributions of inflammatory, angiogenic and proteolytic pathways to CNV formation. The rabbit model that includes delivery of angiogenic factors and insult to Bruchs membrane results in CNV that may persist for months. That type of model may be useful for longitudinal studies, where doses and durations of pharmacologic agents need to be studied in vivo using angiography and/or OCT. These larger models, such as the rabbit and pig, are also useful for drug delivery studies. If a model is needed with a macula, the primate may be used, although there are cost and ethical issues pertinent to primate use. The three features needed for successful animal models of CNV are 1) sustained angiogenesis by VEGF production; 2) an inflammatory cytokine component; 3) compromise of Bruch’s membrane. A given laboratory’s capabilities including budget, animal housing facilities, and experimental goals are all important when deciding which animal model of CNV to utilize.
3. Animal Models of Retinal Neovascularization
3.1 Overview of Retinal Neovascularization
The unique feature of the retina with dual blood supply makes it possible to identify two types of neovascularization: retinal neovascularization, which originates from the retinal vessels, usually extends through the internal limiting membrane (ILM) and grows into the vitreous (under some conditions, the vessels grow in the opposite direction into the subretinal space); and choroidal neovascularization, as described in previous section.
The pathogenesis of retinal neovascularization is better understood than that of CNV (Campochiaro, 2000) (Figure 9). Numerous clinical and experimental observations have indicated that ischemia is the main cause for retinal neovascularization (Michaelson, 1948; Ashton, 1957; Shimizu et al, 1981). Attenuation of retinal vessels resulting in retinal ischemia is a common feature shared by each of the diseases where retinal neovascularization develops, including diabetic retinopathy, retinopathy of prematurity (ROP), central retinal vein occlusion, and branch retinal vein occlusion, diseases that are referred to as ischemic retinopathy.
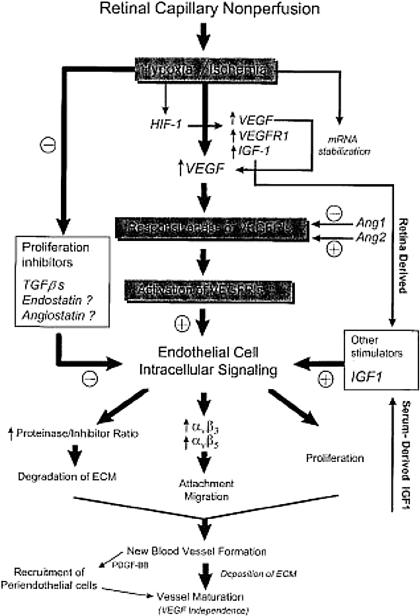
Figure 9. Schematic diagram of the cascade of events that have been implicated in the development of retinal neovascularization.
Abbreviations: HIF-1, hypoxia-inducible factor 1; IGF-1, insulin-like growth factor 1; PDGF-BB, platelet-derived factor B chain homodimer; TGF-βs, members of the transforming growth factor beta super family; VEGF, vascular endothelial growth factor; VEGFR1, VEGF receptor 1. (From Campochiaro, 2000)
One retinal neovascular disease that is developmental in origin is retinopathy of prematurity (Smith, 2002). Retinal vascularization completes by 36 weeks of gestation in the nasal quadrant and slightly later in the temporal quadrant. If an infant is born prematurely before the retinal vascularization is completed, normal vascular maturation process can be disrupted. This neovascularization process occurs in two phases: a vaso-obliterative phase and a vaso-proliferative phase (Ashton et al, 1954). After birth, the premature infant is exposed to the relative hyperoxia of extrauterine environment (55-80 mmHg of PO2) compared to the in utero condition (35 mmHg of PO2), which is exacerbated by supplemental oxygen therapy. During this exposure to hyperoxia, the expression of hypoxia-driven angiogenic factors is downregulated, leading to an arrest or retardation of existing retinal blood growth (Madan and Penn, 2003). Increased metabolic requirement of the developing retina stimulate new vessel growth in response to hypoxia caused by undervascularized retina. This is followed by the vaso-proliferative phase of compensatory, often uncontrolled neovascularization. These new vessels, due to inappropriate development and patterning, cannot sufficiently oxygenate the retina and can bleed easily, causing the degeneration of retinal ganglion cells and photoreceptors (Ashton, 1966; Hellstrom et al, 2001). The vaso-attenuative effect of hyperoxia in neonatal retina with subsequent neovascularization has been used to generate animal models of ischemic retinopathy that not only recapitulates critical events of ROP, but also those of proliferative diabetic retinopathy.
Diabetic retinopathy is the leading cause of acquired blindness in young adults and one of the most severe ocular complications of diabetes (The Early Treatment Diabetic Retinopathy Study Research Group, 1987). In early stages, retinal capillaries are damaged as a result of the microvasculopathy characteristic of diabetes, leading to retinal hypoxia. This is followed by the vaso-proliferative stage of diabetic retinopathy where abnormal, immature new blood vessels grow along the retina and into the vitreous. These vessels can easily bleed, sometimes complicated by fibrovascular membrane and tractional retinal detachment. The microvascular damage also increases vascular permeability. Retinal edema and thickening in the macular area leads to significant visual loss, which is the most common cause of vision loss in non-proliferative diabetic retinopathy (Sarraf, 2001). There are currently no perfect models for diabetic retinopathy; the most commonly used model for diabetic retinopathy is the model induced by intraperitoneal injection of streptozotocin in the rats. The diabetogenic action of the nitrourea derivative streptozotocin (STZ) is used to induce diabetes in the rats, and the animals later develop features similar to diabetic retinopathy (Sosula et al, 1972). However, OIR models are used to study specific pathological events that commonly occur during diabetic retinopathy (Feit-Leichman et al, 2005).
Occlusion of retinal veins by either laser photocoagulation or photodynamic therapy can be performed to induce retinal neovascularization associated with retinal vein occlusion. Other animal models includes transgenic mouse models with overexpression of vascular endothelial growth factor (VEGF), intraocular injection of pro-angiogenic molecules, and inflammation-associated models.
3.2 Oxygen-induced Models of Retinal Neovascularization
Animal models of oxygen-induced retinopathy (OIR) have become major models for pathological angiogenesis resulting from ischemia. This model has been developed first in the kitten (Ashton et al, 1954), then extended to other animal species, including rat (Penn et al, 1993), mouse (Smith et al, 1994), beagle puppy (McLeod et al, 1996), and zebrafish (Cao et al, 2008). Each of the models in different animal species shares the same basic mechanism by which exposure to hyperoxia during early retinal development leads to the arrest or retardation of normal retinal vascular development. When the animals are returned to the normoxic environment, they are under a relative hypoxic situation where the retina now lacks its normal vasculature that is required to adequately support the neural tissue in normoxic conditions. This ischemic situation results in unregulated, abnormal neovascularization. The neovascular response is very consistent, reproducible and quantifiable, and it has become integral for studying disease mechanisms and potential treatments for ischemic retinopathy. Neovascularization occurs at the junction between the vascularized and nonvascularized retina, followed by normalization of the vascular pattern with regression of the abnormal vessels in mice (Smith et al, 1994). Retinal detachment which can be seen in the late stage of ROP occurs in a beagle puppy OIR model, but not in the mouse or kitten OIR model (McLeod et al, 1998; Madan and Penn, 2003).
This ischemia-mediated pathological neovascularization makes the OIR models useful for studying other ischemic retinopathies such as diabetic retinopathy, the leading cause of blindness in younger adults. The mouse OIR model described by Smith et al has become the most common model for studying pathologic angiogenesis associated with ROP (Smith et al, 1994). The use of this model has been extended to the general study of ischemic retinopathies and related anti-angiogenic treatments.
3.2.1 mouse
The most widely used models of OIR are the mouse model originally described by Smith in 1994 (Smith et al, 1994) and the rat model developed by Penn (Penn et al, 1993). The main advantage of the mouse OIR model is the ease of genetic manipulation; transgenic mice can be used to directly study the roles of different genes on pathological neovascularization. Other advantages are the reproducibility of the phenotype, relative costs, the size of the animals, and a reliable method of quantifying retinal neovascularization (Smith et al, 1994; Aguilar et al, 2008). A major disadvantage of this model is the fact that in mice, the central retinal vessels, rather than the peripheral vessels, are obliterated during exposure to hyperoxia. This is different from what happens in ROP where the peripheral retinal vessels fail to develop. Despite this key difference, the mouse OIR model very closely recapitulates the pathologic events that occur during ischemia-induced neovascularization (Aguilar et al, 2008).
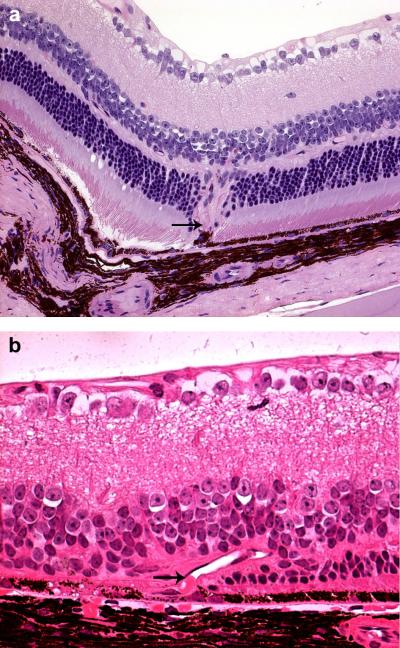
C57BL/6J mouse pups and their mothers are placed in 75% oxygen beginning at postnatal day 7 (P7), and remain in the oxygen chamber for 5 days with constant 75% oxygen levels. Neovascularization is noted five days after return to room air. Some strains of mice, such as BALB/cByJ, react differently to this protocol and do not develop pathological neovascularization at all despite hyperoxia-induced vascular obliteration (Ritter et al, 2006). The original model utilized alternating high/low oxygen exposure; however, similar exposure to constant levels of 75% oxygen for 5 days followed by 5 days in room air demonstrated comparable results (Smith et al, 1994) (Figure 10).
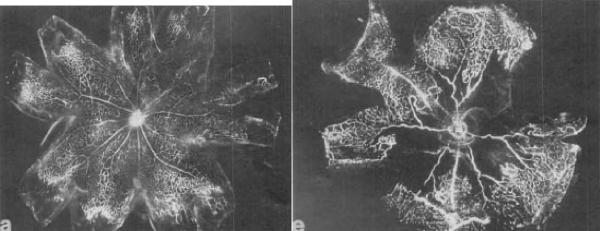
Figure 10. Time course of retinal vascular response to hyperoxia in fluorescein-dextran perfused flat mounts.
Left: normal P7 retina before hyperoxia exposure. Note area of nonperfusion in periphery where blood vessels have not yet developed. Right: P14 retina after 5 days of hyperoxia exposure, followed by 2 days in room air. Note increased perfusion in the periphery and dilation and tortuosity of radial vessels and persistent absence of central perfusion. (Adapted from Smith et al,1994)
3.2.2 rat
While the mouse model utilizes exposure to the constant hyperoxia for 5 days before returning to the room air, the rat OIR model uses alternating hyperoxia-hypoxia cycles. The main advantage of the rat OIR model is that similar to the ROP, the peripheral vasculature becomes obliterated upon exposure to hyperoxia, whereas the central retinal vessels are obliterated in the mouse model (Madan and Penn, 2003) (Figure 11). Removal from oxygen exposure to room air results in neovascular growth at the boundary between vascular and avascular areas in the mid-peripheral retina.
Figure 11. Retinal flat mount of rat OIR model.

Peripheral neovascularization is seen in the rat retina. (From Madan and Penn, 2003)
The protocol of the oxygen exposure, both duration and age at which the rat pups are initially exposed to hyperoxia, is different from the mouse OIR protocol. Penn and coworkers generated the rat OIR model by exposing the animals to the alternating cycle of 80% and 40% oxygen for 12-h for the first 14 days followed by exposure to the room air (Penn et al, 1993). The revised protocol also uses alternating oxygen cycles, but these cycles now utilize 12-hour cycles of 50% and 10% oxygen which show a higher incidence and severity of neovascularization than 80%/40% cycle (Madan and Penn, 2003). The severity of neovascularization can be augmented with increased CO2 concentrations or with growth retardation (Holmes and Duffner, 1996; Holmes et al, 1997). The rat OIR model has been used extensively to identify factors involved in hypoxia-induced neovascularization as well as to test various ocular angiostatics. (Penn et al, 2001; Penn and Rajaratnam, 2003).
3.2.3 feline model
The kitten model was first introduced by Ashton and coworkers, and was one of the first to be used for the study of oxygen-induced retinal neovascularization (Ashton et al, 1954). The kittens are exposed to 70-80% of oxygen within hours after birth for 96 hours, and then returned to room air. The vascular changes occur in successive stages: closure and obliteration during hyperoxia, vasoproliferation induced by hypoxia, and normalization after the relief of hypoxia with distinct cellular mechanisms and stimuli (Chan-Ling et al, 1992).
3.2.4 canine model
McLeod and colleagues studied the pattern of vaso-obliteration in the canine model of OIR. All vascular components showed constriction after 1 hour of hyperoxia, and capillary closure peaked by 24 hours. The pattern and severity of the reaction of the developing retinal vessels to hyperoxia is similar to that observed in the kitten and the premature babies (McLeod et al, 1996). When the newborn pups were exposed to 95-100% oxygen for 4 days and returned to room air until 22-45 days of age, tractional retinal folds and intravitreal vascularized membranes were present in the eyes (McLeod et al, 1998).
3.2.5 zebrafish
Cao and colleagues generated a model of hypoxia-induced retinal angiogenesis in the adult zebrafish. Nitrogen gas was perfused directly into the water in a sealed aquarium. Adult Fli-EGFP-Tg zebrafish were first placed in normoxic water and the O2 tension was gradually reduced to the final 10% air saturation. The fish were exposed in the hypoxic condition from 3-15 days. At day 12, retinal neovascularization reached a plateau of maximal angiogenic responses (Cao et al, 2008).
3.3 Vascular occlusion models of retinal neovascularization
3.3.1 rat
Retinal vein occlusion by laser photocoagulation or photodynamic therapy has been served as a useful model of preretinal neovascularization (Shen et al, 1996; Ham et al, 1997; Saito et al, 1997). Occlusion of retinal veins in rats causes microvascular changes similar to the findings seen in branch retinal vein occlusion clinically, which includes venous dilation, vascular tortuosity, and extravasation of fluid from retinal vessels. These result in reduced blood flow, retinal ischemia, and subsequent neovascularization within the retina or iris. Depending on the number of retinal veins treated, the model can be a model of either branch retinal vein occlusion or central vein occlusion.
After anesthesia, rose bengal dye is injected through tail vein or sodium fluorescein is injected intraperitoneally and laser spots are applied to two or three major vein as they emerge from the optic nerve up to one disc diameter. The laser parameters should be adjusted to achieve venous occlusion; if the laser photocoagulation is too weak, occlusion will not be achieved, and too strong settings can extensively damage other areas of the retina or cause bleeding from the vein. Retinal edema became evident 10-30 minutes after treatment in the sectors associated with the occluded veins, and later resulting in retinal neovascularization and tractional retinal detachment.
3.3.2 pig
Laser induced retinal vein occlusion in pigs has also been shown to cause retinal neovascularization. Retinal neovascularization occurred after occlusion of a single retinal vein or multiple retinal veins with argon laser coagulation (Pournaras et al, 1990) or photodynamic therapy (Danis et al, 1993). Retinal hemorrhage and edema were observed hours after the occlusion, and later preretinal and intravitreal new vessels developed on the ischemic areas.
3.4 Transgenic Mouse Models of Retinal Neovascularization
3.4.1 rho/VEGF Transgenic Mice
As mentioned previously (see 2.4.9), Okamoto and coworkers observed the development of intraretinal and subretinal neovascularization in the transgenic mice with VEGF driven by the rhodopsin promoter (rho/VEGF mice).
Overexpression of VEGF in the retinal photoreceptors demonstrated new vessels originating from the deep capillary bed of the retina extending into the subretinal space. This model may be related more closely to the disease entity seen in patients with retinal angiomatous proliferation (RAP) (Miller, 1997; Okamoto et al, 1997). Tobe et al showed that VEGF transgene mRNA was first detected in the retina on postnatal day 6 (P6). On P14, sprouts from retinal vessels in the deep capillary bed were found, reaching the subretinal space by P18. Invasion of blood vessels from the choroid was not seen (Tobe et al, 1998).
3.4.2 rho/rtTA-TRE/VEGF or IRBP/rtTA-TRE/VEGF Transgenic Mice
Ohno-Matsui et al used the reverse tetracycline transactivator (rtTA) inducible promoter system couple to either the rhodopsin or interphotoreceptor retinoid-binding protein (IRBP) promoter to control the time of onset of VEGF transgene expression in the retina. Without doxycycline, the adult transgenic mice showed little VEGF expression. The addition of doxycycline to the mice resulted in prominent VEGF transgene expression with evidence of more extensive neovascularization. It was also associated with total retinal detachment and higher ocular levels of VEGF mRNA and protein compared to those of rho/VEGF mice (Ohno-Matsui et al, 2002).
3.4.3 VEGF165 (hVEGF) overexpressing mice
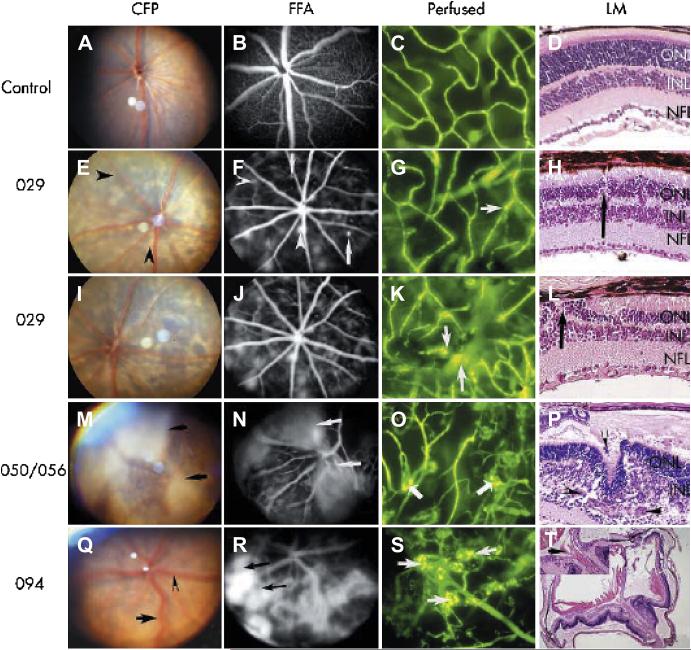
Lai et al developed four human VEGF165 (hVEGF) over-expressing transgenic mouse lines with phenotypes ranging from mild to severe retinal neovascularization. These transgenic mice were generated via microinjection of a DNA construct containing the hVEGF gene driven by a truncated mouse rhodopsin promoter. One transgenic line with low hVEGF showed mild changes such as focal fluorescein leakage, microaneurysms, venous tortuosity; three other lines with high hVEGF levels presented with severe phenotypes, as well as extensive neovascularization, hemorrhage, and retinal detachment (Lai et al, 2005) (Figure 12).
Figure 12.
Characterization of different transgenic mouse lines by color fundus photography, fluorescein fundus angiography, fluorescein micrographs of flat mounted, fluorescein labeled dextran perfused eyes, and light micrographs of hematoxylin and eosin stained paraffin sections. (From Lai et al, 2005)
Bai et al investigated the role of the Müller cell-derived VEGF in ischemia-induced retinal neovascularization in conditional VEGF knockout mice. The transgenic mice were generated by mating Müller cell-expressing Cre mice with floxed VEGF mice, and the VEGF production in the Müller cell was disrupted by feeding 2 mg/ml doxycycline from embryonic day 15 to postnatal day 1. Disruption of VEGF in Müller cell did not cause any detectable retinal degeneration with significant inhibition of the ischemia-induced retinal neovascularization.
3.4.4 Nrl−/−Grk1−/− double knockout mice
A double knockout Nrl−/−Grk1−/− mice were generated to study the cone G-protein coupled receptor signaling pathway. G-protein coupled receptor kinase (Grk1) is involved in the inactivation and normal recovery of cone pigments, and the neural retina leucine zipper knockout mouse (Nrl−/−) retina is biochemically “all-cones”, because rod phototransduction components are absent in this retina. The absence of Grk1 expression on the Nrl−/− showed age-related and light independent cone dystrophy. Retinal neovascularization was observed in all retinal layers by 1 month of age, which was mediated by inflammatory pathway (Yetemian et al, 2010).
3.5 Intraocular injection of pro-angiogenic molecules
3.5.1 Rabbit
Retinal neovascularization was induced after intravitreal injection of fibroblast. Tissue-cultured skin fibroblasts were injected into the vitreous cavity in rabbits, and strand formation in the mid-vitreous cavity was observed, extending toward the retina. Once it attached to the retina, neovascularization began with growth of vessels along the fibrous strand. New vessels regressed after three months (Tano et al, 1981). Antoszyk and coworkers modified the technique by producing partial posterior vitreous detachment with repeated injection and aspiration of hyaluronidase. Rabbit eyes were injected with repeated injection and aspiration of hyaluronidase before injection of homologous tissue-cultured dermal fibroblasts. Ninety-five percent of the eyes developed definite neovascularization by day 14 (Antoszyk et al, 1991). A drawback of this technique is that the neovascular response is more traumatic and inflammatory, rather than ischemic, and retinal detachment is needed for the development of neovascularization (Miller, 1997).
3.5.2 Mouse
In order to recapitulate the vascular changes in the diabetic retinopathy, a binary recombinant adeno-associated virus construct producing green fluorescent protein (GFP) and VEGF under the control of the human cytomegalovirus promoter was injected into the subretinal space of C57BL6 mice. The neovascular changes were more gradual and less pronounced in these mice than other transgenic mouse models with high VEGF levels. This model demonstrated that the development of different stages of diabetic retinopathy is closely correlated with an increased VEGF level in the retina (Rakoczy et al, 2003).
3.6 Other Models of Retinal Neovascularization
3.6.1 Inflammatory Models of Retinal Neovascularization
As mentioned previously (see 2.3.1), the inflammatory model consists in immunization of rats with interphotoreceptor retinoid binding protein peptide to induce mild inflammation. The inflammation was most prominent on day 14, with inflammatory cells in the anterior and posterior segment of the eye. The inflammation decreased in intensity on day 18, but branches of retinal vessels were seen extending into the choroid. On day 45, there was no appreciable inflammation, but new vessels were present in 38% of the eyes (Sakamoto et al, 1994).
3.6.2 Radiation retinopathy model
Irvine and Wood studied the clinical and histological changes of the experimental radiation retinopathy in monkeys. The first change observed after radiation was the focal loss of capillary endothelial cells and pericytes, and as the areas of capillary loss became confluent, large areas of retinal capillary nonperfusion appeared. Later, intraretinal neovascularization and rubeosis iris with neovascular glaucoma developed (Irvine and Wood, 1987).
3.6.3 Diabetic retinopathy model
In early stages of STZ-diabetic rats, there is capillary basement membrane thickening as well as capillary dilatation (Sosula et al, 1972). Vascular occlusion of retinal capillaries and increased vascular leakage from retinal blood vessels are also present (Bek, 1997; Zhang et al, 2005; Masuzawa et al, 2006).
A spontaneously diabetic strain of Sprague-Dawley (SD) rat was recognized in 1988 and named as the Spontaneously Diabetic Torii (SDT) rat. Male SDT rats develop hyperglycemia and glycosuria at approximately 20 weeks of age. The cumulative incidence of diabetes is 100% by 40 weeks of age in the male rats, whereas 33.3% by 65 weeks of age in the female rats (Shinohara et al, 2000). Histologic examination showed cataract and large retinal folds mimicking tractional retinal detachment with extensive leakage of fluorescein around the optic disk (Kakehashi et al, 2006). The SDT rat develops retinal neovascularization without retinal ischemia, which can be attributed to the change in the vitreo-retinal interface, where the persistent vitreous adhesion can be served as the platform for vascular proliferation (Yamada et al, 2005).
3.7 Summary of Animal Models of Retinal Neoascularization
Representative animal models of retinal neovascularization are shown in Table 2. Animal models of retinal neovascularization are well established compared to those for choroidal neovascularization. Ischemia plays a central role in the development of retinal neovascularization. Disease processes in which retinal neovascularization occurs, including diabetic retinopathy, ROP, central retinal vein occlusion, branch retinal vein occlusion, are collectively referred to as ischemic retinopathies. The methods inducing ischemia in animal models include hyperoxia, laser, inflammation, and radiation. There has been a prevalence of rodent studies versus other species in recent years. These models are well characterized, readily quantifiable, and easily reproducible.
Table 2.
Animal Models of Retinal Neovascularization
| reference | animal | method | success rate | quantification | mechanism |
|---|---|---|---|---|---|
| Smith et al 1994 | mouse | hyperoxia (75% oxygen) | 100% | path | ischemia |
| Penn et al, 1993 | rat | cyclic oxygen (80%/40%) | 66% | path | ischemia-reperfusion |
| Holmes et al, 1997 | rat | cyclic oxygen (80%/10%) + high CO2 |
85% | path | ischemia |
| Ashton et al, 1954 | kitten | hyperoxia (70-80%) | 100% | path | ischemia |
| McLeod et al, 1996 | puppy | hyperoxia (95-100%) | N/A | path | ischemia |
| Cao et al, 2008 | zebrafish | hypoxia (10%) | N/A | path | ischemia |
| Shen et al, 1996 | rat | argon laser + rose bengal | 39% | FA | ischemia |
| Ham et al, 1997 | rat | argon laser + rose bengal | 17% | FA | ischemia |
| Saito et al, 1997 | rat | argon laser | 70% | FA + path | ischemia |
| Pournaras et al, 1990 | pig | argon laser | N/A | PO2 measurement |
ischemia |
| Danis et al, 1993 | pig | argon laser + rose bengal | 100% | FA + path | ischemia |
| Okamoto et al 1997 | mouse | transgenic | N/A | FA | VEGF |
| Ohno-Matsui et al, 2002 | mouse | transgenic | 47-100% | path | VEGF |
| Lai et al, 2005 | mouse | transgenic | N/A | FA + path | VEGF |
| Tano et al, 1981 | rabbit | intravitreal injection of fibroblast | 72% | fundus + path | inflammation |
| Antoszyk et al, 1990 | rabbit | intravitreal injection of fibroblast + hyaluronidase |
95% | FA | inflammation |
| Rakoczy et al, 2003 | subretinal injection of recombinant adeno-associated virus-mediated VEGF |
90% | FA + path | VEGF | |
| Sakamoto et al, 1994 | rat | immunization | 38% | path | cytokines |
| Irvine & Wood, 1987 | monkey | radiation | N/A | fundus + path | ischemia |
| Shinohara et al, 2000 | rat | spontaneous | 100% | path | hyperglycemia |
N/A=not available; FA=fluorescein angiography; path=pathology
2. Conclusions and Future Directions
The two main types of periretinal neovascularization are choroidal neovasuclarization (CVN) and retinal neovascularization (RNV). CNV originates in the choroid from the choriocapillaris circulation, penetrates through Bruch’s membrane, and grows into the subRPE and/or subretinal space. RNV originates in the retina from the retinal circulation and extends onto the posterior hyaloid face of the vitreous or into the vitreous. CNV is generally well-confined, like due to a limited extent of cytokine diffusion and RPE control of the size and extent of the lesion. RNV grows in a more haphazard, wildfire-like manner, likely due to diffusion of angiogenic cytokines into the vitreous. A third type of periretinal neovascularization may occur in the setting of CNV and/or RNV and when it forms anastamoses between the retinal and choroidal circulation, it may be classified as retinal angiomatous proliferation, or RAP. When evaluating animal models of CNV and RNV, one should consider these features. Many laser, surgical, dietary and transgenic models of CNV and RNV have been developed and are well described. There is no perfect animal model of CNV or RNV. Investigators are encouraged to carefully consider the hypotheses to be tested, strengths and weaknesses of the various models, available funding, facilities and time available prior to choosing an animal model of CNV or RNV. This will facilitate experimentation and provide the best opportunity for meaningful results. Animal models of CNV and RNV provide an intermediate step between in vivo experimentation and human clinical trials. These current animal models and those that will be developed will continue to provide usefully scientific information for the foreseeable future. The key will be to match scientific interest and funding availability with the model. For those interested in biologic mechanisms, transgenic and knockdown models will continue to be useful. For those interested in drug development, animal models that quickly develop CNV or RNV, such as laser-induced models will continue to be important, especially since these animal models usually develop neovascularization in days to weeks as opposed to the usual months to years it takes for transgenic and knockdown models. As new drugs are developed and become successful, it will become increasingly more difficult to prove superiority of one drug versus another. However, in order to do this, animal models will continue to be needed to test safety and efficacy. Targeted drug delivery to the back of the eye is a rapidly growing area that will require larger animal models of CNV and RNV, such as in the rabbit or pig, in order to simulate the size of the human eye for introduction of investigational drug delivery devices. New, reproducible animal models will continue to be developed, especially larger animals for drug delivery studies.
Acknowledgements
Supported in part by NIH NEIP30EY06360, NEIR24EY017045 and an unrestricted departmental grant from Research to Prevent Blindness, Inc
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Aguilar E, Dorrell MI, Friedlander D, Jacobson RA, Johnson A, Marchetti V, Moreno SK, Ritter MR, Friedlander M. Ocular models of angiogenesis. Methods Enzymol. 2008;444:115–158. doi: 10.1016/S0076-6879(08)02806-1. [DOI] [PubMed] [Google Scholar]
- Albert DM, Neekhar A, Wang S, Darjatmoko SR, Sorenson CM, Subielzig RR, Sheibani N. Development of choroidal neovascularizaiton in rats with advanced intense cyclic light-induced retinal degeneration. Arch Ophthalmol. 2010;128:212–222. doi: 10.1001/archophthalmol.2009.395. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ambati J, Anand A, Fernandez S, et al. An animal model of age-related macular degeneration in senescent Ccl-2- or Ccr-2- deficient mice. Nat Med. 2003;11:1290–1397. doi: 10.1038/nm950. [DOI] [PubMed] [Google Scholar]
- Amin R, Pucklin JE, et al. Growth factor localization in choroidal noevascular membranes of age-related macular degeneration. Inv Ophthalmol Vis Sci. 1994;35:3178–3188. [PubMed] [Google Scholar]
- Antoszyk AN, Gottlieb JL, Machemer R, Hatchell DL. The effects of intravitreal triamcinolone acetonide on experimental pre-retinal neovascularization. Graefes Arch Clin Exp Ophthalmol. 1993;231:34–40. doi: 10.1007/BF01681698. [DOI] [PubMed] [Google Scholar]
- Ashton N. Retinal vascularization in health and disease. Am J Ophthalmol. 1957;44:7–17. doi: 10.1016/0002-9394(57)90426-9. [DOI] [PubMed] [Google Scholar]
- Ashton N. Oxygen and the growth and development of retinal vessels. In vivo and in vitro studies. The XX Francis I. Proctor Lecture. Am J Ophthalmol. 1966;62:412–435. doi: 10.1016/0002-9394(66)91322-5. [DOI] [PubMed] [Google Scholar]
- Ashton N, Ward B, Serpell G. Effect of oxygen on developing retinal vessels with particular reference to the problem of retrolental fibroplasia. Br J Ophthalmol. 1954;38:397–432. doi: 10.1136/bjo.38.7.397. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baffi J, Byrnes G, Chan CC, Csaky KG. Choroidal neovascularization in the rat induced by adenovirus mediate expression of vascular endothelial growth factor. Inv Ophthalmol Vis Sci. 2000;41:3582–3589. [PubMed] [Google Scholar]
- Bai Y, Ma JX, Guo J, Wang J, Zhu M, Chen Y, Le YZ. Müller cell-derived VEGF is a significant contributor to retinal neovascularization. J Pathol. 2009;219:446–54. doi: 10.1002/path.2611. [DOI] [PubMed] [Google Scholar]
- Bainbridge JW, Mistry AR, Thrasher AJ, Ali RR. Gene therapy for ocular angiogenesis. Clin Sci (Lond) 2003;104:561–575. doi: 10.1042/CS20020314. [DOI] [PubMed] [Google Scholar]
- Baird PN, Robman LD, Richardson AJ, et al. Gene environment interaction in progression of AMD: the CFH gene, smoking and exposure to chronic infection. Hum Mol Genet. 2008;17:1299–1305. doi: 10.1093/hmg/ddn018. [DOI] [PubMed] [Google Scholar]
- Bein K, Simons M. Thrombospondin type 1 repeats interact with matrix metalloproteinase 2. Regulation of metalloproteinase activity. J Biol Chem. 2000;275:32167–32173. doi: 10.1074/jbc.M003834200. [DOI] [PubMed] [Google Scholar]
- Bek T. Glial cell involvement in vascular occlusion of diabetic retinopathy. Acta Ophthalmol Scand. 1997;75:239–243. doi: 10.1111/j.1600-0420.1997.tb00764.x. [DOI] [PubMed] [Google Scholar]
- Bergers G, Brekken R, et al. Matrix metalloproteinase-9 triggers the angiogenic switch during carcinogenesis. Nat Cell Biol. 2000;2:737–744. doi: 10.1038/35036374. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Berglin L, Sarman S, gan der Ploeg I, et al. Reduced choroidal neovascular membrane formation in matrix metalloproteinase-2 deficient mice. Inv Ophthalmol Vis Sci. 2003;44:403–408. doi: 10.1167/iovs.02-0180. [DOI] [PubMed] [Google Scholar]
- Bora PS, Sohn JG, Curz JM, et al. role of complement and complement membrane attack complex in laser-induced choroidal neovascularization. J Immunol. 2005;174:491–497. doi: 10.4049/jimmunol.174.1.491. [DOI] [PubMed] [Google Scholar]
- Bora PS, Hu Z, Tezel TH, Sohn JH, Kang SG, Cruz JM, boar NS, Garen A, Kaplan HJ. Immunotherapy for choroidal neovascularization in a laser-induced mouse model simulating exudative (wet) macular degeneration. PNAS. 2003;100:2679–2684. doi: 10.1073/pnas.0438014100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campochiaro PA. Retinal and choroidal neovascularization. J Cell Physiol. 2000;184:301–310. doi: 10.1002/1097-4652(200009)184:3<301::AID-JCP3>3.0.CO;2-H. J Cell Physiol 2000;184:301-310. [DOI] [PubMed] [Google Scholar]
- Cao R, Jensen L, Söll I, Hauptmann G, Cao Y. Hypoxia-induced retinal angiogenesis in zebrafish as a model to study retinopathy. PLoS ONE. 2008;3:e2748. doi: 10.1371/journal.pone.0002748. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cao Y. Endogenous angiogenesis inhibitors and their therapeutic implications. Int J Biochem Cell Biol. 2001;33:357–369. doi: 10.1016/s1357-2725(01)00023-1. [DOI] [PubMed] [Google Scholar]
- Carmeliet P, Collen D. Tissue Factor. Int J Biochem Cell Biol. 1998;30:661–667. doi: 10.1016/s1357-2725(97)00121-0. [DOI] [PubMed] [Google Scholar]
- Chan CC, Caspi RR, Ni M, Leake WC, Wiggert B, Chader GJ, Nussenblatt RB. Pathology of experimental autoimmunity in mice. J Autoimmune. 1990;3:247–255. doi: 10.1016/0896-8411(90)90144-h. [DOI] [PubMed] [Google Scholar]
- Chan CC, Ross RJ, Shen D, et al. Cl2/Cx3cr1-deficient mice: an animal model for age-related macular degeneration. Ophthamic Res. 2008;40:124–128. doi: 10.1159/000119862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chan-Ling T, Tout S, Holländer H, Stone J. Vascular changes and their mechanisms in the feline model of retinopathy of prematurity. Invest Ophthalmol Vis Sci. 1992;33:2128–2147. [PubMed] [Google Scholar]
- Criswell MH, Ciulla TA, Hill TE, et al. The squirrel monkey: characterization of a new-world primate model of experimental choroidal neovascularization and comparison with the macaque. Inv Ophthalmol Vis Sci. 2004;45:625–634. doi: 10.1167/iovs.03-0718. [DOI] [PubMed] [Google Scholar]
- Cui JZ, Kimura H, Spee C, et al. Natural history of choroidal neovascularization induced by vascular endothelial growth factor in the primate. Graefe’s Arch Clin Exp Ophthlamol. 2000;238:326–333. doi: 10.1007/s004170050360. [DOI] [PubMed] [Google Scholar]
- Danis R, Yang Y, Massicotte S, Boldt C. Preretinal and optic nerve head neovascularization induced by photodynamic venous thrombosis in domestic pigs. Arch Ophthalmol. 1993;111:539–543. doi: 10.1001/archopht.1993.01090040131047. [DOI] [PubMed] [Google Scholar]
- Dithmar S, Curcio CA, Le NA, et al. Ultrastructural changes in Bruchs membrane of apoplipoprotein E-deficient mice. Inv Ophthalol Vis Sci. 2000;41:2035–2042. [PubMed] [Google Scholar]
- Dobi ET, Puliafito CA, Destro M. A new model of experimental choroidal neovascularization in the rat. Arch Ophthalmol. 1989;107:264–269. doi: 10.1001/archopht.1989.01070010270035. [DOI] [PubMed] [Google Scholar]
- El Diniri AA, Ogden TE, Ryan SJ. Subretinal endophotocoagulation. A new model of subretinal neovascularization in the rabbit. Retina. 1991;11:244–249. [PubMed] [Google Scholar]
- Elner VM, Streiten RM, Elner SG, et al. Neutrophil chemotactic factor (IL-8) gene expression by cytokine-treated retinal pigment epithelial cells. Am J Pathol. 1996;136:745–750. [PMC free article] [PubMed] [Google Scholar]
- Espinosa-Heidmann DG, Suner I, Hernandez EP, et al. Age as an independent risk factor for severity of experimental choroidal neovascularization. Inv Ophthalmol Vis Sci. 2002;43:1567–1573. [PubMed] [Google Scholar]
- Espinosa-Heidmann DG, Suner I, Hernandez EP, et al. macrophage depletion dimishes lesion size and severity in experimental choroidal neovascularization. Inv Ophthalmol Vis Sci. 2003;44:3586–3592. doi: 10.1167/iovs.03-0038. [DOI] [PubMed] [Google Scholar]
- Feit-Leichman RA, Kinouchi R, Takeda M, Fan Z, Mohr S, Kern TS, Chen DF. Vascular damage in a mouse model of diabetic retinopathy: Relation to neuronal and glial changes. Invest Ophthalmol Vis Sci. 2005;46:4281–4287. doi: 10.1167/iovs.04-1361. [DOI] [PubMed] [Google Scholar]
- Ferguson T, Apte RS. Angiogenesis in eye disease: immunity gained or immunity lost? Semin Immunopathol. 2008;30:111–119. doi: 10.1007/s00281-008-0113-8. [DOI] [PubMed] [Google Scholar]
- Folkman J, D’Amore PA, et al. Blood vessel formation: what is its molecular basis? Cell. 1996;87:1153–1155. doi: 10.1016/s0092-8674(00)81810-3. [DOI] [PubMed] [Google Scholar]
- Frank RN, Das A, Weber ML. A model of subretinal neovascularization in the pigmented rat. Curr Eye Res. 1989;8:239–247. doi: 10.3109/02713688908997565. [DOI] [PubMed] [Google Scholar]
- Gale NW, Yancopoulos GD. Growth factors activing via endothelial cell-specific receptor tyrosine kinases: VEGFs, angiopoietins, and ephrins in vascular development. Genes Dev. 1999;13:1055–1066. doi: 10.1101/gad.13.9.1055. [DOI] [PubMed] [Google Scholar]
- Green WR. The uveal tract. In: Spencer WR, editor. Ophthalmic Pathology: An Atlas and Textbook. WB Saunders; Philadelphia: 2006. pp. 1597–1607. [Google Scholar]
- Green WR, Wilson DJ. Choroidal neovascularization. Ophthalmology. 1986;93:169–1176. doi: 10.1016/s0161-6420(86)33609-1. [DOI] [PubMed] [Google Scholar]
- Greenberg JI, Shields DJ, Barillas SG, et al. A role for VEGF as a negative regulator of pericyte function and vessel maturation. Nature. 2008;456:809–813. doi: 10.1038/nature07424. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Grossniklaus HE, Cingle KA, Yoon YD, Ketkar N, L’Hernault N, Brown S. Correlation of histologic 2-dimensional reconstruction and confocal scanning laser microscopic imaging of choroidal neovascularization in eyes with age-related maculopathy. Arch Ophthalmol. 2000;118:625–629. doi: 10.1001/archopht.118.5.625. [DOI] [PubMed] [Google Scholar]
- Grossniklaus HE, Green WR. Choroidal neovascularization. Am J Ophthalmol. 2004;137:496–503. doi: 10.1016/j.ajo.2003.09.042. [DOI] [PubMed] [Google Scholar]
- Grossniklaus HE, Ling JX, Wallace TM, et al. Macrophage and retinal pigment epithelium expression of angiogenic cytokines in choroidal neovascularization. Mol Vis. 2002;8:119–126. [PubMed] [Google Scholar]
- Hageman GS, Luthert PJ, et al. An integrated hypothesis that considers drusen as biomarkers of immune-meidated processes at the RPE-Bruch’s membrane interface in aging and age-related macular degeneration. Prog Retina Eye Res. 2001;20:705–732. doi: 10.1016/s1350-9462(01)00010-6. [DOI] [PubMed] [Google Scholar]
- Hahn P, Qian Y, Dentchev T, et al. Disruption of ceruloplasmin and hehaestin in mice causes retinal iron overload and retinal degeneration with features of age-realted macular degeneration. PNAS. 2004;101:13850–13855. doi: 10.1073/pnas.0405146101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ham DI, Chang K, Chung H. Preretinal neovascularization induced by experimental retinal vein occlusion in albino rats. Korean J Ophthalmol. 1997;11:60–64. doi: 10.3341/kjo.1997.11.1.60. [DOI] [PubMed] [Google Scholar]
- Heckenlively JR, Hawes NL, Friedlander M, Nusinowitz S, Hurd R, Davisson M, Chang B. Mouse model of subretinal neovascularization with choroidal anastomosis. Retina. 2003;23:518–522. doi: 10.1097/00006982-200308000-00012. [DOI] [PubMed] [Google Scholar]
- Hellstrom A, Perruzzi C, Ju M, Engstrom E, Hard AL, Liu JL, Albersson-Wikland K, Carlsson B, Niklasson A, Sjodell L, LeRoith D, Senger DR, et al. Low IGF-I suppresses VEGF-survival signaling in retinal endothelial cells: Direct correlation with clinical retinopathy of prematurity. Proc Natl Acad Sci USA. 2001;98:5804–5808. doi: 10.1073/pnas.101113998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holash J, Maisonpierre PC, et al. Vessel cooption, regression, and growth in tumors mediated by angiopoietins and VEGF. Science. 1999;284:1994–1998. doi: 10.1126/science.284.5422.1994. [DOI] [PubMed] [Google Scholar]
- Hollyfield JG, Bonilha VL, Rayborn ME, et al. Oxidative damage-induced inflammation initiates age-related macular degeneration. Nat Med. 2008;14:194–198. doi: 10.1038/nm1709. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holmes JM, Duffner LA. The effect of postnatal growth retardation on abnormal neovascularization in the oxygen exposed neonatal rat. Curr Eye Res. 1996;15:403–409. doi: 10.3109/02713689608995831. [DOI] [PubMed] [Google Scholar]
- Holmes JM, Zhang S, Leske DA, Lanier WL. The effect of carbon dioxide on oxygen-induced retinopathy in the neonatal rat. Curr Eye Res. 1997;16:725–732. doi: 10.1076/ceyr.16.7.725.5054. [DOI] [PubMed] [Google Scholar]
- Hsu HT, Goodnight R, Ryan SJ. Subretinal choroidal neovascularization as a response to penetrating retinal injury in the pigmented rabbit. Jpn J Ophthalmol. 1989;33:358–366. [PubMed] [Google Scholar]
- Imamura Y, Noda S, Hashizume K, Shinoda K, Yamaguchi M, Uchiyama S, Shimizu T, Mizushima Y, Shirasawa T, Tsuboto K. Drusen, choroidal neovascularization, and retinal pigment epithelium dysfunction in SOD1-deficient mice: a model of ageprelated macular degeneration. PNAS. 2006;103:11282–11287. doi: 10.1073/pnas.0602131103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Irvine AR, Wood IS. Radiation retinopathy as an experimental model for ischemic proliferative retinopathy and rubeosis iridis. Am J Ophthalmol. 1987;103:790–797. doi: 10.1016/s0002-9394(14)74395-8. [DOI] [PubMed] [Google Scholar]
- Julien S, Kreppel F, Beck S, et al. A reproducible and quantifiable model of choroidal neovascularization induced by VEGF A165 after subretinal adenoviral gene transfer in the rabbit. Mol Vis. 2008;14:1358–1372. [PMC free article] [PubMed] [Google Scholar]
- Kakehashi A, Saito Y, Mori K, Sugi N, Ono R, Yamagami H, Shinohara M, Tamemoto H, Ishikawa S, Kawakami M, Kanazawa Y. Characteristics of diabetic retinopathy in SDT rats. Diabetes Metab Res Rev. 2006;22:455–461. doi: 10.1002/dmrr.638. [DOI] [PubMed] [Google Scholar]
- Kiilgaard JF, Nis Andersen MV, Wiencke AK, et al. A new animal model of choroidal neovascularization. Acta Ophthalmol Scand. 2005;83:297–704. doi: 10.1111/j.1600-0420.2005.00566.x. [DOI] [PubMed] [Google Scholar]
- Kilarski WWW, Samolov B, et al. Biochemical regulation of blood vessel growth during tissue vascularization. Nat Med. 2009;15:657–664. doi: 10.1038/nm.1985. [DOI] [PubMed] [Google Scholar]
- Kimura H, Sakamoto T, Hinton DR, et al. A new model of subretinal neovascularzation in the rabbit. Inv Ophthalmol Vis Sci. 1995;36:2110–2119. [PubMed] [Google Scholar]
- Kimura H, Spee C, Sakamoto T, et al. Cellular response in subretinal neovascularization induced by bFGF-impregnated microspheres. Inv Ophthalmol Vis Sci. 1999;40:524–528. [PubMed] [Google Scholar]
- Kvanta A, Algvere PV, Berglin L, Seregard S. Subfoveal fibrovascualr membranes in age-related macular degeneration express vascular endothelial growth factor. Inv Ophthalmol Vis Sci. 1996;37:1929–1934. [PubMed] [Google Scholar]
- Kwak N, Okamoto N, Wood JM, Campochiaro PA. VEGF is major stimulator in model of choroidal neovascularization. Inv Ophthalmol Vis Sci. 2000;41:3158–3164. [PubMed] [Google Scholar]
- Lai CM, Dunlop SA, May LA, et al. Generation of transgenic mice with mild and severe retinal neovascularisation. Br J Ophthalmol. 2005;89:911–916. doi: 10.1136/bjo.2004.059089. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lassota N. Clinical and histological aspects of CNV formation: studies in an animal model. Acta Ophthalmologica. 2008:1–24. doi: 10.1111/j.1755-3768.2008.01412.x. [DOI] [PubMed] [Google Scholar]
- Lassota N, Kiilgaard J, la Cour M, Scherfig E, Prause JU. Natural history of chorodal neovascularization after surgical inducation in an animal model. Acta Ophthalmologica. 2008;86:295–503. doi: 10.1111/j.1600-0420.2007.01127.x. [DOI] [PubMed] [Google Scholar]
- Lassota N, Kiilgaard J, Prause JU, la Cour M. Correlation between clinical and histological features in a pig model of choroidal neovascularization. Graefe’s Arch Clin Exp Ophthalmol. 2006;244:394–398. doi: 10.1007/s00417-005-0061-0. [DOI] [PubMed] [Google Scholar]
- Lassota N, Kiilgaard J, Prause JU, et al. Surgical induction of choroidal neovascularization in a procine model. Graefe’s Arch Clin Exp Ophthalmol. 2007;245:1189–1198. doi: 10.1007/s00417-006-0518-9. [DOI] [PubMed] [Google Scholar]
- Lopez PF, Sippy BD, Lambert HM, et al. Transdifferentiated retinal pigment epithelial cells are immunoreactive for vascular endothelial growth factor in surgically-excised age-related macular degeneration-related choroidal neovascular membranes. Inv Ophthalmol Vis Sci. 1996;37:855–868. [PubMed] [Google Scholar]
- Madan A, Penn JS. Animal models of oxygen-induced retinopathy. Front Biosci. 2003;8:d1030–d1043. doi: 10.2741/1056. [DOI] [PubMed] [Google Scholar]
- Malek G, Johnson LV, Mace BE, et al. Apolipoprotein E allele-dependent pathogenesis: a model for age-related retinal degeneration. PNAS. 2005;102:11900–11905. doi: 10.1073/pnas.0503015102. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Masuzawa K, Jesmin S, Maeda S, Zaedi S, Shimojo N, Miyauchi T, et al. Effect of endothelin dual receptor antagonist on VEGF levels in streptozotocin-induced diabetic rat retina. Exp Biol Med. 2006;231:1090–1094. [PubMed] [Google Scholar]
- McLeod DS, Brownstein R, Lutty GA. Vaso-obliteration in the canine model of oxygen-induced retinopathy. Invest Ophthalmol Vis Sci. 1996;37:300–311. [PubMed] [Google Scholar]
- McLeod DS, D’Anna SA, Lutty GA. Clinical and histopathologic features of canine oxygen-induced proliferative retinopathy. Invest Ophthalmol Vis Sci. 1998;39:1918–1932. [PubMed] [Google Scholar]
- Michaelson IC. The mode of development of the vascular system of the retina with some observations on its significance for certain retinal diseases. Trans Ophthalmol Soc UK. 1948;68:137–180. [Google Scholar]
- Miller H, Miller B, Ryan SJ. The role of retinal pigment epithelium in the involution of subretinal neovascularization. Inv Ophthalmol Vis Sci. 1986;27:1644–1652. [PubMed] [Google Scholar]
- Miller JW. Vascular endothelial growth factor and ocular neovascularization. Am J Pathol. 1997;151:13–23. [PMC free article] [PubMed] [Google Scholar]
- Mori K, Duh E, et al. Pigment epithelium-derived factor inhibits retinal and choroidal neovascularization. J Cell Physiol. 2001;188:253–263. doi: 10.1002/jcp.1114. [DOI] [PubMed] [Google Scholar]
- Mori K, Gelbach P, Yamamoto S, et al. AAV-mediated gene transfer of pigment epithelium-derived factor inhibits choroidal neovascularization. Inv Ophthalmol Vis Sci. 2002;43:1994–2000. [PubMed] [Google Scholar]
- Mullins RF, Russell SR, et al. Drusen associated with aging and age-related macular degeneration contain proteins common to extracellular deposits associated with atherosclerosis, elastosis, amyloidosis and dense deposit disease. FASEB. 2000;14:835–846. [PubMed] [Google Scholar]
- Ni M, Holland M, Jarstadmarken H, De Vries G. Time-course of experimental choroidal neovascularzation in Dutch-Belted rabbit: clinical and histological evaluation. Exp Eye Res. 2005;81:286–297. doi: 10.1016/j.exer.2005.01.027. [DOI] [PubMed] [Google Scholar]
- Ohno-Matsui K, Hirose A, Yamamoto S, et al. Inducible expression of vascular endothelial growth factor in adult mice causes severe proliferative retinopathy and retinal detachment. Am J Pathol. 2002;160:711–719. doi: 10.1016/S0002-9440(10)64891-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Okamoto N, Tobe T, Hackett SF, et al. Transgenic mice with increased expression of vascular endothelial growth factor in the retina. A new model of intraretinal and subretinal neovascularization. Am J Pathol. 1997;151:281–291. [PMC free article] [PubMed] [Google Scholar]
- Oshima Y, Oshima S, Nambu H, et al. Increased expression of VEGF in retinal pigemented epithelial cells is not sufficient to cause choroidal neovascularization. J Cell Physiol. 2004;201:393–400. doi: 10.1002/jcp.20110. [DOI] [PubMed] [Google Scholar]
- Park KH, Fridley BL, Ryu E, et al. Complement component 3 (C3) haplotypes and risk of advanced age-related macular degeneration. Inv Ophthalmol Vis Sci. 2009;50:3386–3393. doi: 10.1167/iovs.08-3231. [DOI] [PubMed] [Google Scholar]
- Penfold P, Killingsworth M, Sarks S. An ulstrastructural study of the role of leucocytes and fibroblasts in the breakdown of Bruch’s membrane. Aust J Ophthalmol. 1984;12:23–31. [PubMed] [Google Scholar]
- Penfold PL, Madigan MC, et al. Immunological and aetiological aspects of macular degeneration. Prog Retin Eye Res. 2001;20:385–414. doi: 10.1016/s1350-9462(00)00025-2. [DOI] [PubMed] [Google Scholar]
- Penn JS, Rajaratnam VS. Inhibition of retinal neovascularization by intravitreal injection of human rPAI-1 in a rat model of retinopathy of prematurity. Invest Ophthalmol Vis Sci. 2003;44:5423–5429. doi: 10.1167/iovs.02-0804. [DOI] [PubMed] [Google Scholar]
- Penn JS, Rajaratnam VS, Collier RJ, Clark AF. The effect of an angiostatic steroid on neovascularization in a rat model of retinopathy of prematurity. Invest Ophthalmol Vis Sci. 2001;42:283–290. [PubMed] [Google Scholar]
- Penn JS, Tolman BL, Lowery LA. Variable oxygen exposure causes preretinal neovascularization in the newborn rat. Invest Ophthalmol Vis Sci. 1993;34:576–585. [PubMed] [Google Scholar]
- Pournaras CJ, Tsacopoulos M, Strommer K, Gilodi N, Leuenberger PM. Experimental retinal branch vein occlusion in miniature pigs induces local tissue hypoxia and vasoproliferative microangiopathy. Ophthalmology. 1990;97:1321–1328. doi: 10.1016/s0161-6420(90)32415-6. [DOI] [PubMed] [Google Scholar]
- Qui G, Stewart JM, Vas Sadda S, et al. A new model of experimental subretinal neovascularization in the rabbit. Exp Eye Res. 2006;83:141–142. doi: 10.1016/j.exer.2005.11.014. [DOI] [PubMed] [Google Scholar]
- Rakic JM, Lambert V, Munaut C, et al. Mice without upa, tpa or plasminigen genes are resistant to experimental choroidal neovascularization. Inv Ophthlamol Vis Sci. 2003;44:1732–1739. doi: 10.1167/iovs.02-0809. [DOI] [PubMed] [Google Scholar]
- Rakoczy PE, Brankov M, Fonceca A, Zaknich T, Rae BC, Lai CM. Enhanced recombinant adeno-associated virus-mediated vascular endothelial growth factor expression in the adult mouse retina: a potential model for diabetic retinopathy. Diabetes. 2003;52:857–863. doi: 10.2337/diabetes.52.3.857. [DOI] [PubMed] [Google Scholar]
- Rakoczy PE, Yu MJT, Nusinowitz S, Chang B, Heckenlively JR. Mouse models of age-related macular degeneration. Exp Eye Res. 2006;82:741–752. doi: 10.1016/j.exer.2005.10.012. [DOI] [PubMed] [Google Scholar]
- Reich SJ, Fosnot J, Kuroki A, tang W, yang X, Maguire AM, Bennett J, Tolentino MJ. Small interfering RNA (SiRNA) targeting VEGF effectively inhibits ocular neovascularization in a mouse model. Mol Vis. 2003;9:210–216. [PubMed] [Google Scholar]
- Ritter MR, Banin E, Moreno SK, Aguilar E, Dorrell MI, Friedlander M. Myeloid progenitors differentiate into microglia and promote vascular repair in a model of ischemic retinopathy. J Clin Invest. 2006;116:3266–3276. doi: 10.1172/JCI29683. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ryan SR. Subretinal neovascularization. Natural history of an experimental model. Arch Ophthalmol. 1982:1804–1809. doi: 10.1001/archopht.1982.01030040784015. [DOI] [PubMed] [Google Scholar]
- Ryan SJ. The development of an experimental model of subretinal neovascualrization in disciform macular degeneration. Trans Am Ophthalmol Soc. 1979;127:707–745. [PMC free article] [PubMed] [Google Scholar]
- Saishin Y, Silva RL, Saishin Y, et al. Periocular injection of micropsheres containing PKC412 inhibits choroidal neovascularization in a porcine model. Inv Ophthlmol Vis Sci. 2003;44:4989–4993. doi: 10.1167/iovs.03-0600. [DOI] [PubMed] [Google Scholar]
- Saito Y, Park L, Skolik SA, et al. Experimental preretinal neovascularization by laser-induced venous thrombosis in rats. Curr Eye Res. 1997;16:26–33. doi: 10.1076/ceyr.16.1.26.5116. [DOI] [PubMed] [Google Scholar]
- Sakamoto T, Sanui H, Ishibashi T, et al. Subretinal neovascularization in the rat induced by IRBP synthetic peptides. Exp Eye Res. 1994;58:155–160. doi: 10.1006/exer.1994.1003. [DOI] [PubMed] [Google Scholar]
- Sakurai E, Anand A, Ambati BK, van Rooigen N, Ambati J. Macrophage depeletion inhibits experimental choroidal neovascularization. Inv Ophthalmol Vis Sci. 2003;44:3578–3585. doi: 10.1167/iovs.03-0097. [DOI] [PubMed] [Google Scholar]
- Sarraf D. Diabetes and Eye Disease. American Academy of Ophthalmology; San Francisco, CA: 2001. pp. 3–5.pp. 8–20. [Google Scholar]
- Schlingemann RO. Role of growth factors and the wound healing response in age-related macular degeneration. Graefes Arch Clin Exp Ophthalmol. 2004;242:91–101. doi: 10.1007/s00417-003-0828-0. [DOI] [PubMed] [Google Scholar]
- Scholl HPN, Charbel IP, Walier M, et al. Systemic complement activation in age-related macular degeneration. PLOS. 2008;3:e2593. doi: 10.1371/journal.pone.0002593. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schmack I, Berglin L, Nie X, et al. Modulation of choroidal neovascularization by subretinal injection of retinal pigment epithelium and polystyrene microbeads. Mol Vis. 2009;15:146–161. [PMC free article] [PubMed] [Google Scholar]
- Schwesinger C, Yee C, Rohan RM, et al. Intrachoroidal neovascualrization in transgenic mice overexpressing vascular endothelial growth factor in the retinal pigment epithelium. Am J Pathol. 1994;158:1161–1172. doi: 10.1016/S0002-9440(10)64063-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seo MS, Kwak N, Ozaki H, et al. Dramatic inhibition of retinal and choroidal neovascularization by oral administration of a kinase inhibitor. Am J Pathol. 1999;154:1743–1753. doi: 10.1016/S0002-9440(10)65430-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sengupta N, Caballero S, Mames RN, et al. The role of adult bone marrow-derived stem cells in choroidal neovascularuzation. Inv Ophthalmol Vis Sci. 2003;44:4908–4913. doi: 10.1167/iovs.03-0342. [DOI] [PubMed] [Google Scholar]
- Shen W, He S, Han S, Ma Z. Preretinal neovascularisation induced by photodynamic venous thrombosis in pigmented rat. Aust N Z J Ophthalmol. 1996;24:50–52. doi: 10.1111/j.1442-9071.1996.tb00994.x. [DOI] [PubMed] [Google Scholar]
- Shen D, Wen R, Tuo J, Bojanowski CM, Chan CC. Exacerbation of retinal degeneration and choroidal neovascularization by subretinal injection of Matrigel in CCL2/MCP-1-deficient mice. Ophthal Res. 2006;38:71–73. doi: 10.1159/000090266. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shimizu K, Kobayashi Y, Muraoka K. Midperipheral fundus involvement in diabetic retinopathy. Ophthalmology. 1981;88:601–612. doi: 10.1016/s0161-6420(81)34983-5. [DOI] [PubMed] [Google Scholar]
- Shinohara M, Masuyama T, Shoda T, Takahashi T, Katsuda Y, Komeda K, Kuroki M, Kakehashi A, Kanazawa Y. A new spontaneously diabetic non-obese Torii rat strain with severe ocular complications. Int J Exp Diabetes Res. 2000;1:89–100. doi: 10.1155/EDR.2000.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Singh SR, Grossniklaus HE, Kang SJ, et al. Intravenous transferrin, RGD peptide and dual-targeted nanoparticles enhance anti-VEGF intraceptor gene delivery to laser-induced CNV. Gene Therapy. 2009;16:645–659. doi: 10.1038/gt.2008.185. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Smith LE. Pathogenesis of retinopathy of prematurity. Acta Paediatr Suppl. 2002;91:26–28. doi: 10.1111/j.1651-2227.2002.tb00157.x. [DOI] [PubMed] [Google Scholar]
- Smith LE, Wesolowski E, McLellan A, Kostyk SK, D’Amato R, Sullivan R, D’Amato PA. Oxygen-induced retinopathy in the mouse. Invest Ophthalmol Vis Sci. 1994;35:101–111. [PubMed] [Google Scholar]
- Smith RS, John SWM, Zabeleta A, et al. The Bst locus on mouse chromosome 16 is associated with age-related subretinal neovascularization. PNAS. 2000;97:2191–2195. doi: 10.1073/pnas.040531597. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sosula L, Beaumont P, Hollows FC, Jonson KM. Dilatation and endothelial proliferation of retinal capillaries in streptozotocin-diabetic rats: quantitative electron microscopy. Invest Ophthalmol Vis Sci. 1972;11:926–35. [PubMed] [Google Scholar]
- Sourbane G, Cohen SY, Delayer T, et al. Basic fibroblast growth factor experimentally induced choroidal angiogenesis. Curr Eye Res. 1994;13:183–195. doi: 10.3109/02713689408995776. [DOI] [PubMed] [Google Scholar]
- Spilsbury K, Garrett KL, Shen W, et al. Overexpression of vascular endothelial growth factor (VEGF) in the retinal pigment epithelium leads to the development of choroidal neovascularzation. Am J Pathol. 2000;157:135–144. doi: 10.1016/S0002-9440(10)64525-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Spraul CW, Grossniklaus HE. Characteristics of Drusen and Bruch’s membrane in post-mortem eyes with age-related macular degeneration. Arch Ophthalmol. 1997;115:267–273. doi: 10.1001/archopht.1997.01100150269022. [DOI] [PubMed] [Google Scholar]
- Sven B, Sejersen S, Berglin L, Seregard S, Kvanta A. Matrix metalloproteinases and metalloproteinase inhibitors in choroidal neovascular membranes. Inv Ophthalmol Vis Sci. 1998;29:2194–2200. [PubMed] [Google Scholar]
- Takahashi T, nakamura T, Hayashi A, Kamei M, Nakabayashi M, Okada AA, Tomita T, Karenda Y, Tano Y. Inhibition of experimental choroidal neovasculaurization by overexpression of tissue inhibitor of metalloproteinases-3 in retinal pigment epithelium cells. Am J Ophthalmol. 2000;130:774–781. doi: 10.1016/s0002-9394(00)00772-8. [DOI] [PubMed] [Google Scholar]
- Takeda A, Baffi JZ, Kleinman ME, et al. CCR3 is a target for age-related macular degeneration diagnosis and therapy. Nature. 2009;460:225–231. doi: 10.1038/nature08151. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tamai K, Spaide R, Ellis EA, et al. Lipid hydroperoxide stimulates subretinal chorodal neovascularization in the rabbit. Exp Eye Res. 2002;74:301–308. doi: 10.1006/exer.2001.1121. [DOI] [PubMed] [Google Scholar]
- Tano Y, Chandler DB, Machemer R. Retinal neovascularization after intravitreal fibroblast injection. Am J Ophthalmol. 1981;92:103–109. doi: 10.1016/s0002-9394(14)75913-6. [DOI] [PubMed] [Google Scholar]
- The Early Treatment Diabetic Retinopathy Study Research Group Photocoagulation for diabetic macular edema: Early Treatment Diabetic Retinopathy Study report no. 4. Int Ophthalmol Clin. 1987;27:265–272. doi: 10.1097/00004397-198702740-00006. [DOI] [PubMed] [Google Scholar]
- Tobe T, Okamoto N, Vinores MA, et al. Evolution of neovascularization in mice with overexpression of vascular endothelial growth factor in photoreceptors. Inv Ophthalmol Vis Sci. 39:180–188. 2998. [PubMed] [Google Scholar]
- Tobe T, Ortega S, Luna J, et al. Targeted disruption of the FGF2 gene does not prevent choroidal neovascularization in a murine model. Am J Pathol. 1998;153:141–1646. doi: 10.1016/S0002-9440(10)65753-7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wada M, Ogata N, Otsuji T, Uyama M. Expression of vascular endothelial growth factor and its receptor (KDR/flk-1) mRNA in experimental choroidal neovascularization. Curr Eye Res. 1999;18:203–213. doi: 10.1076/ceyr.18.3.203.5368. [DOI] [PubMed] [Google Scholar]
- Wang F, Rendahl KG, Manning WC, et al. AAV-mediated expression of vascular endothelial growth factor induces choroidal neovascularization in rat. Invest Ophthalmol Vis Sci. 2003;44:781–790. doi: 10.1167/iovs.02-0281. [DOI] [PubMed] [Google Scholar]
- Yamada H, Yamada E, Higuchi A, Matsumura M. Retinal neovascularisation without ischaemia in the spontaneously diabetic Torii rat. Diabetologia. 2005;48:1663–1668. doi: 10.1007/s00125-005-1809-0. [DOI] [PubMed] [Google Scholar]
- Yetemian RM, Brown BM, Craft CM. Loss of G-protein coupled receptor kinase in Nrl−/− mice leads to neovascularization, enhanced inflammatory response, and age-related cone dystrophy. Invest Ophthalmol Vis Sci. 2010 doi: 10.1167/iovs.10-5452. (in press) [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yanagi Y, tamaki Y, Obata R, Muranaka K, Humma N, Matsuoka H, Mano H. Subconjunctival administration of bucillamine suppresses choroidal neovascularization. Inv Ophthalmol Vis Sci. 2002;43:3495–3499. [PubMed] [Google Scholar]
- Zamiri P, Masli S, Streilein JW, Taylor AW. Pigment epithelial growth factor suppresses inflammation by modulating macrophage activity. Inv Ophthalmol Vis Sci. 2006;47:3912–3918. doi: 10.1167/iovs.05-1267. [DOI] [PubMed] [Google Scholar]
- Zhan G, Vossmeyer D, Stragies R, et al. Preclinical evaluation of the new small-moleculae integrin alpha 5 beta 1 inhibitor JSM6427 in monkey and rabbit models of choroidal neovascularization. Arch Ophthalmol. 2009;127:1329–1235. doi: 10.1001/archophthalmol.2009.265. [DOI] [PubMed] [Google Scholar]
- Zhang SX, Sima J, Wang JJ, Shao C, Fant J, Ma JX. Systemic and periocular deliveries of plasminogen kringle 5 reduce vascular leakage in rat models of oxygen-induced retinopathy and diabetes. Curr Eye Res. 2005;30:681–689. doi: 10.1080/02713680590934102. [DOI] [PubMed] [Google Scholar]
- Zhu ZR, Goodnight R, Sorgente N, et al. Morphotoic observations of retinal pigment epithelial proliferation and neovascularization in the rabbit. Retina. 1989;9:319–327. doi: 10.1097/00006982-198909040-00013. [DOI] [PubMed] [Google Scholar]