Abstract
This guideline focuses on the primary prevention of venous thromboembolism (VTE) in Korea. The guidelines should be individualized and aim at patients scheduled for major surgery, as well as patients with a history of trauma, high-risk pregnancy, cancer, or other severe medical illnesses. Currently, no nation-wide data on the incidence of VTE exist, and randomized controlled trials aiming at the prevention of VTE in Korea have yielded few results. Therefore, these guidelines were based on the second edition of the Japanese Guidelines for the Prevention of VTE and the eighth edition of the American College of Chest Physicians (ACCP) Evidenced-Based Clinical Practice Guidelines. These guidelines establish low-, moderate-, and high-risk groups, and recommend appropriate thromboprophylaxis for each group.
Keywords: Guideline, Prevention, Venous Thromboembolism
INTRODUCTION
Venous thromboembolism (VTE) is a thrombotic disorder of the venous system, which includes deep vein thrombosis (DVT) and pulmonary embolism (PE). Approximately half of all untreated DVT cases are complicated by PE, and conversely, 50% to 80% of all untreated PE cases are associated with DVT (1, 2).
VTE is a well-recognized, public health issue in developed countries. In the United States of America, 200,000 new cases of PE occur each year, and 50,000 of these result in death. PE is the third most common fatal vascular disorder following coronary artery disease (CAD) and cerebrovascular accident (CVA) (3). Thromboprophylaxis has been recommended for the following reasons: the high incidence of VTE in hospitalized patients; the difficulty of early diagnosis due to vague symptomatology; the cost-effectiveness of medical prophylaxis; and, the high mortality of PE without early diagnosis and adequate treatment. Health authorities in developed countries have established guidelines based on the available evidence and recommended thromboprophylaxis to medical societies in order to improve health standards and reduce health costs (4, 5).
The incidence of VTE in the Korean population has known to be lower than in the Caucasian population; however, it appears to be rapidly increasing in large part from the widespread adoption of the Western lifestyle. Additionally, the elderly comprise the largest proportion of the Korean population, and advanced age has been recognized as a risk factor for VTE. Despite the rise in the incidence of VTE, many physicians in Korea still are not aware of the significance of VTE and the risk of sudden death associated with inappropriate management of this condition. Recently, we published a guidline which was a revised version of the Japanese Guidelines for the Prevention of VTE; however, it was cumbersome for physicians to read (6). The following guidelines represent a simplified, practical version of the aforementioned guidelines, complete with risk stratification (low, moderate, and high) and thromboprophylaxis recommendations for each group.
GENERAL GUIDELINES
Risk stratification
Accurate and prompt stratification of thrombotic risk should be undertaken for every patient based on available evidence. The method for risk evaluation should be simple, efficient, and cost-effective. Most hospitalized patients have at least one risk factor for VTE, and decisions regarding the risk of VTE should include considerations of current and future risks (7). Accepted risk factors for the development of VTE include previous VTE; major surgery; pelvis or femur fracture; major trauma; cancer; pregnancy and the postpartum period; long-term immobilization; comorbidities such as congestive heart failure (CHF), chronic obstructive pulmonary disease (COPD) requiring mechanical ventilation, or CVA; spinal cord injury (SCI), old age, obesity, estrogen replacement therapy, varicose veins, and other acquired or hereditary thrombophilic conditions.
Selection of the appropriate thromboprophylaxis depends upon the level of thrombotic risk taken on by each patient (8-10). The general consensus establishes obesity, exogenous estrogen, and varicose veins as low-risk conditions; advanced age, prolonged immobilization, CHF, COPD, central venous catheter placement, chemotherapy, and sepsis as moderate-risk conditions; and, previous VTE, hereditary thrombophilia, and lower extremity paralysis as high-risk conditions.
The American College of Chest Physicians (ACCP) Evidenced-Based Clinical Practice Guidelines (8th Edition) for the Prevention of VTE uses three thrombotic risk groups (low, moderate, and high) based on clinical evidence in the Caucasian population. Thromboprophylaxis is recommended for all patients with a moderate or high risk for VTE (11). However, the Japanese Guideline recommends that active prophylaxis should be initiated at one level higher than the ACCP guidelines because the Japanese population is less susceptible to VTE than the Caucasian population. The Korean Society on Thrombosis and Hemostasis (KSTH) adopted the Japanese risk stratification guidelines due to the ethnic similarity between the Korean and Japanese populations.
Thromboprophylaxis
Screening compression ultrasonography (CUS) or pulmonary computed tomography (CT) angiography can be used to detect VTE in some patients, like only in high-risk patients who can not receive thrombophylaxis; however, these modalities are expensive and unreliable at times. These are not routinely recommended for detection of VTE instead of pharmacoprophylaxsis. Non-pharmacologic methods are applicable in patients with a high bleeding risk. However, supportive evidence is limited, and they are not cost-effective. Pharmacologic methods are reasonable and cost-effective, and are therefore recommended as the initial form of prophylaxis in most patients without a high bleeding risk.
Non-pharmacologic methods
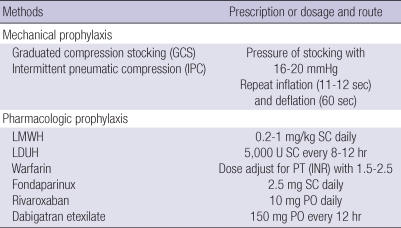
Exercise increases venous blood flow and reduces venous stasis, and thus, helps prevent VTE. However, no standardized exercise has been implemented for the prevention of VTE in hospitalized patients. The application of graduated compression stockings (GCS) at the time of admission can prevent VTE in at-risk patients. Use of intermittent pneumatic compression (IPC) can be helpful in at-risk surgical patients (Table 1) (12). Placement of an inferior vena cava (IVC) filter is only recommended for patients at high-risk of VTE when pharmacologic thromboprophylaxis is contraindicated (13-16).
Table 1.
Methods of thromboprophylaxis
LMWH, low-molecular-weight heparin; LDUH, low-dose unfractionated heparin; SC, subcutaneously; PT, prothrombin time; INR, international normalized ratio; PO, per os.
Pharmacologic methods
Pharmacologic methods for thromboprophylaxis include low-molecular weight heparin (LMWH), low-dose unfractionated heparin (LDUH), fondaparinux, warfarin, dabigatran, and rivaroxaban. The recommended daily doses are LMWH 20-100 U/kg (0.2-1 mg/kg) subcutaneously (SC) daily; LDUH 5,000 U every 8 to 12 hr SC, fondaparinux 2.5 mg SC daily; dabigatran etexilate 150 mg every 12 hr orally; and, rivaroxaban 10 mg orally each day. Warfarin should be dosed daily to maintain an international normalized ratio (INR) in the range 1.5 to 2.5 (Table 1). The duration of thromboprophylaxis depends upon the perceived benefits of anticoagulation versus the risks of bleeding and overall cost.
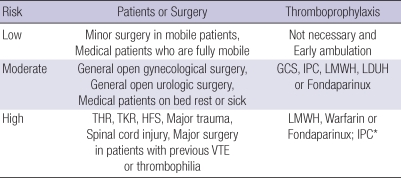
Stratification of VTE risk in hospitalized patients
Based on clinical evidence, the risk of VTE in surgical patients can be stratified from low to high (Table 2). High-risk situations include major surgery in patients with previous VTE or a hypercoagulable state, major orthopedic surgery, CVA, major trauma, or SCI.
Table 2.
VTE risk-stratification and recommended prophylactic methods for each risk group
*Recommended for patients with a risk of bleeding; consider switching to anticoagulants when the bleeding risk abates.
VTE, venous thromboembolism; GCS, graduated compression stockings; IPC, intermittent pneumatic compression; LMWH, low-molecular-weight heparin; LDUH, low-dose unfractionated heparin; THR, total hip replacement; TKR, total knee replacement; HFS, hip fracture surgery.
The Korean guidelines are not based upon clinical evidence, but rather upon a consensus in experts panel of Korean Society of Thrombosis and Hemostasis (KSTH). Therefore, these guidelines represent the most current opinions for good clinical practices for the prevention of VTE. The ultimate decision regarding thromboprophylaxis should be individualized by case and determined by the attending physician.
GENERAL SURGERY
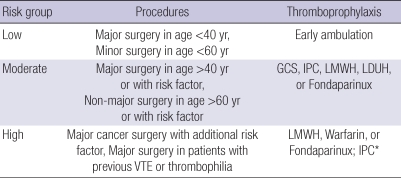
The principles of risk assessment for general surgery are based on the type of surgery (minor or major), age (<40 yr, 40-60 yr, and ≥60 yr), and the presence of additional risk factors, such as cancer or previous VTE (11). According to these principles, cases were classified into three risk groups (Table 3). Early and frequent ambulation was recommended for low-risk patients. Mechanical methods (GCS and/or IPC) or pharmacologic methods (LMWH, LDUH, or fondaparinux) are recommended for moderate-risk patients. LMWH, fondaparinux, or warfarin are recommended for high-risk patients scheduled for major cancer surgery with additional risk factors for VTE and for high-risk patients with a previous VTE or thrombophilia. When anticoagulation is contraindicated in a high-risk patient, IPC is recommended. For patients undergoing entirely laparoscopic procedures who do not have additional thromboembolic risk factors, routine use of thromboprophylaxis (other than early and frequent ambulation) is discouraged (17).
Table 3.
Levels of VTE risk and recommendations for general surgery
*Recommended in patients with a risk of bleeding; consider switching to anticoagulants when the bleeding risk abates.
VTE, venous thromboembolism; GCS, graduated compression stockings; IPC, intermittent pneumatic compression; LMWH, low-molecular-weight heparin; LDUH, low-dose unfractionated heparin; VTE, venous thromboembolism.
ORTHOPEDIC SURGERY
Patients scheduled for major orthopedic surgery, including total hip replacement (THR), total knee replacement (TKR), and hip fracture surgery (HFS), represent a group at particularly high risk of VTE. In the West, the incidence of VTE among patients that undergo major orthopedic surgery ranges from 41% to 60%, but the epidemiological data on the incidence of VTE in the Asian population varies (11). A recent Korean study found the incidence of postoperative VTE in TKR, THR, and HFS, to be 40.4%, 8.7%, and 16.4%, respectively, using CT pulmonary angiography and indirect CT venography (18).
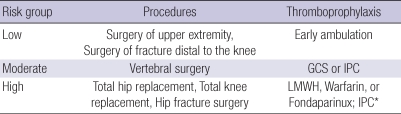
For patients scheduled to undergo TKR, THR, or HFS, LMWH, warfarin or fondaparinux is recommended for thromboprophylaxis (Table 4) (19, 20). GCS and/or IPC are recommended in patients with a risk of bleeding. The current recommended duration for anticoagulation is 7 to 10 days, but extended use through 35 days has been proposed in the 8th ACCP guideline for the prevention of VTE after discharge from the hospital (4).
Table 4.
Levels of VTE risk and recommendations for orthopedic surgery
*Recommended in patients with a risk of bleeding; consider switching to anticoagulants when the bleeding risk abates.
VTE, venous thromboembolism; GCS, graduated compression stockings; IPC, intermittent pneumatic compression; LMWH, low-molecular-weight heparin; LDUH, low-dose unfractionated heparin.
Patients scheduled for vertebral surgery have a moderate risk for VTE and should wear GCS and/or utilize IPC. Patients scheduled for surgery of the upper extremity or lower leg (distal to the knee) has a low risk for VTE and do not require prophylaxis. Routine CUS screening is not helpful in these patients.
NEUROSURGERY
The incidence of DVT and subsequent PE in neurosurgery patients has been reported as high as 25%, and the PE mortality rate has been reported from 9% to 50% (21, 22). The risk factors that contribute to the high frequency of VTE in neurosurgical patients include a prior VTE; type of surgery (cranial, spinal, or vascular); duration of surgery; malignancy; infection; immobilization; venous stasis; chronic lower extremity swelling; lower extremity trauma; advanced age; CHF; obesity; and, sleep apnea.
Early and frequent ambulation for low-risk neurosurgery patients who have undergone surgery (not craniotomy) is recommended. For moderate-risk patients scheduled for craniotomy (for reasons other than brain tumor), GCS and/or IPC is recommended. IPC, LDUH or LMWH are suggested for patients scheduled for craniotomy for a brain tumor. For high-risk patients scheduled for craniotomy with a concomitant history of VTE or thrombophilia, a combined thromboprophylactic approach with a mechanical method (GCS and/or IPC) and a pharmacologic method (LMWH or LDUH) is recommended.
UROLOGIC SURGERY
Risk factors for VTE in urologic surgery patients include an age ≥40 yr, obesity, malignancy, recent surgery, previous VTE, an open (vs transurethral) procedure, general anesthesia, and a long operation time. Major urologic surgery may present higher risks of PE and VTE than general surgery.
The risk of VTE has been associated with operation time (<45 min and ≥45 min). For patients scheduled for a low-risk procedure, early ambulation and exercise without any other form of thromboprophylaxis is recommended. On the other hand, for patients scheduled for major surgery, thromboprophylaxis with GCS or IPC; LMWH, LDUH, or fondaparinux; or a combination of mechanical and pharmacologic thromboprophylaxis is indicated. For abdominal (open) surgery on the kidney or retroperitoneum, the aforementioned principles outlined for general surgery should be followed. Nephrectomy presents the same level of risk as radical prostatectomy.
OBSTETRIC AND GYNECOLOGIC SURGERY
The incidence of VTE is high in pregnancy and may occur at any stage during pregnancy or in the weeks following delivery. The risk factors for VTE in pregnant women include previous VTE, a family history of VTE, presence of anti-phospholipid antibody, age ≥40 yr, prolonged bed rest, placenta previa, Caesarian section, and lower extremities varicosities. For women with a history of abortion, intrauterine death, toxemia, placenta previa, and intrauterine growth retardation, monitoring for thrombophilia and evaluating for thrombosis must be continued throughout the pregnancy.
Giant uterine myoma, previous surgery for an ovarian tumor, ovarian cancer, uterine or cervical cancer, severe intrapelvic adhesions, ovarian hyperstimulation syndrome, hormonal therapy, and particularly, protracted lymph node dissection requiring transfusion are considered important risk factors for VTE (23, 24). Estrogen or progesterone should be administered with caution in postmenopausal women with a high risk of VTE.
For patients with risk factors, general thromboprophylaxis, such as, lower extremity exercise on a bed, GCS, IPC, and adequate hydration postpartum are recommended. Early ambulation should be encouraged even after a normal delivery in low-risk patients. In addition, pharmacologic thromboprophylaxis with LMWH or warfarin should be considered in patients with risk factors other than Caesarian section. For high-risk pregnancies with documented thrombophilia, such as, positive anti-phospholipid antibody or previous VTE, pharmacologic thromboprophylaxis with LMWH is recommended (25). Warfarin is contraindicated during pregnancy (category X) (26). However, warfarin can replace LMWH after delivery and be used for 6 weeks to 3 months for continued postpartum thromboprophylaxis (25). For patients with gynecologic disease, management strategies should follow those outlined for general surgery patients.
MEDICAL CONDITIONS
Acutely ill medical patients represent a clinically heterogeneous group and demonstrate differing risks of VTE. Despite extensive studies in medical patients, the morbidity and mortality of VTE remains significant. Without prophylaxis, the incidence of DVT and PE in general medical patients has been reported to range from 10% to 30% (5, 27, 28).
The risk of VTE was determined by assessing the probability of VTE in acutely ill medical patients according to predisposing risk factors (age >70 yr, obesity, long-term immobility, tobacco use, varicosities, dehydration, estrogens, cancer, previous DVT, paraplegia, congenital or acquired thrombophilia, and inflammatory bowel disease [IBD]) and acute medical illnesses currently under treatment (COPD exacerbation, mechanical ventilator therapy, infection, CHF, and CVA) (Table 5). Acute ischemic stroke patients with restricted mobility should have LDUH or LMWH administrated 24 hr after thrombolytic therapy. If a patient has an acute intracranial hemorrhage or a contraindication to anticoagulation, GCS and/or IPC can be used. However, GCS and IPC can increase venous return and should be used carefully in cases of fluid overload, such as severe CHF. For ambulatory cancer patients with cyclic chemotherapy, routine thromboprophylaxis is not recommended; however, cancer patients with restricted mobility due to other acute medical illnesses are considered high-risk and should have thromboprophylaxis administered (4).
Table 5.
Risk factors of VTE in general medical patients
*congenital thrombophilia such as antithrombin III deficiency or protein C or S deficiency, or acquired thrombophilia like antiphospholipid antibody syndrome.
VTE, venous thromboembolism; HRT, hormone replacement therapy; OCs, oral contraceptives; COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; CHF, congestive heart failure; NYHA, New York Heart Association, VTE, venous thromboembolism.
Patients with CHF, COPD, sepsis, or IBD also have a higher prevalence of VTE, and thromboprophylaxis using GCS, IPC, LMWH, LDUH, or fondaparinux is recommended (29, 30). The majority of patients admitted to the intensive care unit (ICU) have multiple risk factors for VTE. These patients should be routinely assessed and offered thromboprophylaxis with LDUH or LMWH. GCS and/or IPC can be used when there is a contraindication to anticoagulation.
MAJOR TRAUMA AND SPINAL CORD INJURY
Patients with major trauma are classified as high-risk. Major trauma includes multiple serious injuries, head trauma with mental status changes, severe pelvic fracture, and multiple fractures of a lower extremity. VTE can cause significant morbidity in patients experiencing major trauma and occurs in up to 50% of patients without thromboprophylaxis (31). Furthermore, PE represents the third-leading cause of death among those who survive beyond 24 hr.
Routine thromboprophylaxis is recommended for all major trauma patients. Those patients with a high bleeding risk should receive LMWH or LDUH after primary hemostasis. On the other hand, if pharmacologic thromboprophylaxis is contraindicated due to active bleeding or a sustained high risk of bleeding, mechanical (GCS or IPC) thromboprophylaxis should be implemented.
All patients with acute SCI should receive routine thromboprophylaxis. Initially, GCS and/or IPC and careful observation for bleeding is recommended, followed by LMWH thromboprophylaxis when primary hemostasis is complete. For patients with a high bleeding risk, such as those with intra-abdominal bleeding, traumatic subarachnoid hemorrhage, or spinal hematoma, pharmacologic thromboprophylaxis should be delayed until the risk of further bleeding has diminished.
NEURAXIAL ANESTHESIA
Neuraxial anesthesia is a comprehensive term used for spinal, epidural, and caudal blocks. The risk for the development of spinal or epidural hematoma may be elevated by the concomitant use of anticoagulants and antiplatelet agents (32, 33). The established risk factors for spinal or epidural hematoma after neuraxial blockade include an underlying hemostatic disorder, an anatomically-deformed vertebral column, traumatic insertion of a needle or catheter, repeated insertion attempts, concomitant anticoagulation, continuous use of epidural catheters, and old age (11, 34).
To improve the safety of neuraxial blockade in patients receiving or scheduled to receive anticoagulant prophylaxis, several guidelines have been established. Neuraxial anesthesia/analgesia should generally be avoided in patients with a bleeding disorder and in situations when preoperative hemostasis is impaired by antithrombotic drugs. An epidural catheter should be removed when the anticoagulant effect is minimal (usually just before the next scheduled SC injection). Anticoagulation prophylaxis should be delayed for at least 2 hr after the removal of a spinal needle or epidural catheter. If warfarin is required, continuous epidural analgesia should not be used for longer than 1 or 2 days. Finally, monitoring for cord compression syndrome is required when patients are receiving anticoagulation medication (32, 35).
SUMMARY
These guidelines emphasize the primary prevention of VTE with thromboprophylaxis for Korean patients experiencing surgery, obstetric delivery, trauma, cancer, and severe medical illness. Based on VTE risk factors (age, immobility, history of VTE, comorbid illness, and type of surgery or trauma), patients can be stratified into low-, moderate-, and high-risk groups. For high-risk patients (major orthopedic surgery, major trauma, SCI, and major surgery with a history of VTE or thrombophilia), thromboprophylaxis with LMWH, warfarin, or fondaparinux is recommended. Mechanical methods of thromboprophylaxis should be used primarily in patients with a high bleeding risk. For moderate-risk patients (general open gynecological surgery, general open urologic surgery, and medical patients on bed rest), thromboprophylaxis with either a mechanical method (GCS and/or IPC) or a pharmacologic method (LMWH, LDUH, or fondaparinux) can be utilized. For low-risk patients (minor surgery in mobile patients and medical patients who are fully mobile), early and frequent ambulation is the only recommended thromboprophylaxis. In conclusion, this article outlines the first Korean guidelines issued for primary VTE prevention and provides a useful reference for clinicians. These guidelines need to be updated based on results from well controlled studies conducted in Korea.
References
- 1.Huisman MV, Buller HR, ten Cate JW, van Royen EA, Vreeken J, Kersten MJ, Bakx B. Unexpected high prevalence of silent pulmonary embolism in patients with deep venous thrombosis. Chest. 1989;95:498–502. doi: 10.1378/chest.95.3.498. [DOI] [PubMed] [Google Scholar]
- 2.Moser KM, Fedullo PF, LitteJohn JK, Crawford R. Frequent asymptomatic pulmonary embolism in patients with deep venous thrombosis. JAMA. 1994;271:223–225. [PubMed] [Google Scholar]
- 3.Anderson FA, Jr, Wheeler HB, Goldberg RJ, Hosmer DW, Patwardhan NA, Jovanovic B, Forcier A, Dalen JE. A population-based perspective of the hospital incidence and case-fatality rates of deep vein thrombosis and pulmonary embolism. The Worcester DVT Study. Arch Intern Med. 1991;151:933–938. [PubMed] [Google Scholar]
- 4.Geerts WH, Bergqvist D, Pineo GF, Heit JA, Samama CM, Lassen MR, Colwell CW American College of Chest Physicians. Prevention of venous thromboembolism: American College of Chest Physicians Evidence-Based Clinical Practice Guidelines (8th Edition) Chest. 2008;133(6 Suppl):381S–453S. doi: 10.1378/chest.08-0656. [DOI] [PubMed] [Google Scholar]
- 5.Nicolaides AN, Breddin HK, Fareed J, Goldhaber S, Haas S, Hull R, Kalodiki E, Myers K, Samama M, Sasahara A Cardiovascular Disease Educational and Research Trust and the International Union of Angiology. Prevention of venous thromboembolism. International Consensus Statement. Guidelines compiled in accordance with the scientific evidence. Int Angiol. 2001;20:1–37. [PubMed] [Google Scholar]
- 6.The Korea Society on Thrombosis and Hemostasis. Japanese guideline for prevention of venous thromboembolism. Korean J Thromb Hemost. 2009;16:Supplement 1. [Google Scholar]
- 7.Anderson FA, Jr, Wheeler HB, Goldberg RJ, Hosmer DW, Forcier A. The prevalence of risk factors for venous thromboembolism among hospital patients. Arch Intern Med. 1992;152:1660–1664. [PubMed] [Google Scholar]
- 8.Zhan C, Miller MR. Excess length of stay, charges, and mortality attributable to medical injuries during hospitalization. JAMA. 2003;290:1868–1874. doi: 10.1001/jama.290.14.1868. [DOI] [PubMed] [Google Scholar]
- 9.Samama MM, Dahl OE, Quinlan DJ, Mismetti P, Rosencher N. Quantification of risk factors for venous thromboembolism: a preliminary study for the development of a risk assessment tool. Haematologica. 2003;88:1410–1421. [PubMed] [Google Scholar]
- 10.Heit JA, O'Fallon WM, Petterson TM, Lohse CM, Silverstein MD, Mohr DN, Melton LJ., 3rd Relative impact of risk factors for deep vein thrombosis and pulmonary embolism: a population-based study. Arch Intern Med. 2002;162:1245–1248. doi: 10.1001/archinte.162.11.1245. [DOI] [PubMed] [Google Scholar]
- 11.Geerts WH, Pineo GF, Heit JA, Bergqvist D, Lassen MR, Colwell CW, Ray JG. Prevention of venous thromboembolism: the Seventh ACCP Conference on Antithrombotic and Thrombolytic Therapy. Chest. 2004;126(3 Suppl):338S–400S. doi: 10.1378/chest.126.3_suppl.338S. [DOI] [PubMed] [Google Scholar]
- 12.Scurr JH, Coleridge-Smith PD, Hasty JH. Regimen for improved effectiveness of intermittent pneumatic compression in deep venous thrombosis prophylaxis. Surgery. 1987;102:816–820. [PubMed] [Google Scholar]
- 13.Rodriguez JL, Lopez JM, Proctor MC, Conley JL, Gerndt SJ, Marx MV, Taheri PA, Greenfield LJ. Early placement of prophylactic vena caval filters in injured patients at high risk for pulmonary embolism. J Trauma. 1996;40:797–802. doi: 10.1097/00005373-199605000-00020. [DOI] [PubMed] [Google Scholar]
- 14.Carlin AM, Tyburski JG, Wilson RF, Steffes C. Prophylactic and therapeutic inferior vena cava filters to prevent pulmonary emboli in trauma patients. Arch Surg. 2002;137:521–525. doi: 10.1001/archsurg.137.5.521. [DOI] [PubMed] [Google Scholar]
- 15.Rogers FB, Shackford SR, Ricci MA, Wilson JT, Parsons S. Routine prophylactic vena cava filter insertion in severely injured trauma patients decreases the incidence of pulmonary embolism. J Am Coll Surg. 1995;180:641–647. [PubMed] [Google Scholar]
- 16.Giannoudis PV, Pountos I, Pape HC, Patel JV. Safety and efficacy of vena cava filters in trauma patients. Injury. 2007;38:7–18. doi: 10.1016/j.injury.2006.08.054. [DOI] [PubMed] [Google Scholar]
- 17.Lee KW, Bang SM, Kim S, Lee HJ, Shin DY, Koh Y, Lee YG, Cha Y, Kim YJ, Kim JH, Park DJ, Kim HH, Oh D, Lee JS. The incidence, risk factors and prognostic implications of venous thromboembolism in patients with gastric cancer. J Thromb Haemost. 2010;8:540–547. doi: 10.1111/j.1538-7836.2009.03731.x. [DOI] [PubMed] [Google Scholar]
- 18.Cha SI, Lee SY, Kim CH, Park JY, Jung TH, Yi JH, Lee J, Huh S, Lee HJ, Kim SY. Venous thromboembolism in Korean patients undergoing major orthopedic surgery: a prospective observational study using computed tomographic (CT) pulmonary angiography and indirect CT venography. J Korean Med Sci. 2010;25:28–34. doi: 10.3346/jkms.2010.25.1.28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Warwick D. New concepts in orthopaedic thromboprophylaxis. J Bone Joint Surg Br. 2004;86:788–792. doi: 10.1302/0301-620x.86b6.15085. [DOI] [PubMed] [Google Scholar]
- 20.Lieberman JR, Hsu WK. Prevention of venous thromboembolic disease after total hip and knee arthroplasty. J Bone Joint Surg Am. 2005;87:2097–2112. doi: 10.2106/JBJS.D.03033. [DOI] [PubMed] [Google Scholar]
- 21.Agnelli G, Piovella F, Buoncristiani P, Severi P, Pini M, D'Angelo A, Beltrametti C, Damiani M, Andrioli GC, Pugliese R, Iorio A, Brambilla G. Enoxaparin plus compression stockings compared with compression stockings alone in the prevention of venous thromboembolism after elective neurosurgery. N Engl J Med. 1998;339:80–85. doi: 10.1056/NEJM199807093390204. [DOI] [PubMed] [Google Scholar]
- 22.Black PM, Baker MF, Snook CP. Experience with external pneumatic calf compression in neurology and neurosurgery. Neurosurgery. 1986;18:440–444. doi: 10.1227/00006123-198604000-00008. [DOI] [PubMed] [Google Scholar]
- 23.Schorge JO, Goldhaber SZ, Duska LR, Goodman A, Feldman S. Clinically significant venous thromboembolism after gynecologic surgery. J Reprod Med. 1999;44:669–673. [PubMed] [Google Scholar]
- 24.Ageno W, Manfredi E, Dentali F, Silingardi M, Ghezzi F, Camporese G, Bolis P, Venco A. The incidence of venous thromboembolism following gynecologic laparoscopy: a multicenter, prospective cohort study. J Thromb Haemost. 2007;5:503–506. doi: 10.1111/j.1538-7836.2007.02312.x. [DOI] [PubMed] [Google Scholar]
- 25.Leonhardt G, Gaul C, Nietsch HH, Buerke M, Schleussner E. Thrombolytic therapy in pregnancy. J Thromb Thrombolysis. 2006;21:271–276. doi: 10.1007/s11239-006-5709-z. [DOI] [PubMed] [Google Scholar]
- 26.Ageno W, Crotti S, Turpie AG. The safety of antithrombotic therapy during pregnancy. Expert Opin Drug Saf. 2004;3:113–118. doi: 10.1517/eods.3.2.113.27343. [DOI] [PubMed] [Google Scholar]
- 27.Caprini JA, Arcelus JI, Reyna JJ. Effective risk stratification of surgical and nonsurgical patients for venous thromboembolic disease. Semin Hematol. 2001;38(2 Suppl 5):12–19. doi: 10.1016/s0037-1963(01)90094-0. [DOI] [PubMed] [Google Scholar]
- 28.Samama MM, Cohen AT, Darmon JY, Desjardins L, Eldor A, Janbon C, Leizorovicz A, Nguyen H, Olsson CG, Turpie AG, Weisslinger N. A comparison of enoxaparin with placebo for the prevention of venous thromboembolism in acutely ill medical patients. Prophylaxis in Medical Patients with Enoxaparin Study Group. N Engl J Med. 1999;341:793–800. doi: 10.1056/NEJM199909093411103. [DOI] [PubMed] [Google Scholar]
- 29.Kniffin WD, Jr, Baron JA, Barrett J, Birkmeyer JD, Anderson FA., Jr The epidemiology of diagnosed pulmonary embolism and deep venous thrombosis in the elderly. Arch Intern Med. 1994;154:861–866. [PubMed] [Google Scholar]
- 30.Morpurgo M, Schmid C, Mandelli V. Factors influencing the clinical diagnosis of pulmonary embolism: analysis of 229 postmortem cases. Int J Cardiol. 1998;65(Suppl 1):S79–S82. doi: 10.1016/s0167-5273(98)00069-2. [DOI] [PubMed] [Google Scholar]
- 31.Geerts WH, Code KI, Jay RM, Chen E, Szalai JP. A prospective study of venous thromboembolism after major trauma. N Engl J Med. 1994;331:1601–1606. doi: 10.1056/NEJM199412153312401. [DOI] [PubMed] [Google Scholar]
- 32.Horlocker TT, Wedel DJ, Benzon H, Brown DL, Enneking FK, Heit JA, Mulroy MF, Rosenquist RW, Rowlingson J, Tryba M, Yuan CS. Regional anesthesia in the anticoagulated patient: defining the risks (the second ASRA Consensus Conference on Neuraxial Anesthesia and Anticoagulation) Reg Anesth Pain Med. 2003;28:172–197. doi: 10.1053/rapm.2003.50046. [DOI] [PubMed] [Google Scholar]
- 33.Moen V, Dahlgren N, Irestedt L. Severe neurological complications after central neuraxial blockades in Sweden 1990-1999. Anesthesiology. 2004;101:950–959. doi: 10.1097/00000542-200410000-00021. [DOI] [PubMed] [Google Scholar]
- 34.Vandermeulen EP, Van Aken H, Vermylen J. Anticoagulants and spinal-epidural anesthesia. Anesth Analg. 1994;79:1165–1177. doi: 10.1213/00000539-199412000-00024. [DOI] [PubMed] [Google Scholar]
- 35.Rowlingson JC, Hanson PB. Neuraxial anesthesia and low-molecular-weight heparin prophylaxis in major orthopedic surgery in the wake of the latest American Society of Regional Anesthesia guidelines. Anesth Analg. 2005;100:1482–1488. doi: 10.1213/01.ANE.0000148683.54686.0F. [DOI] [PubMed] [Google Scholar]