Abstract
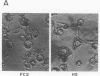
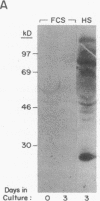
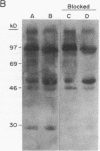
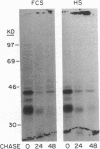
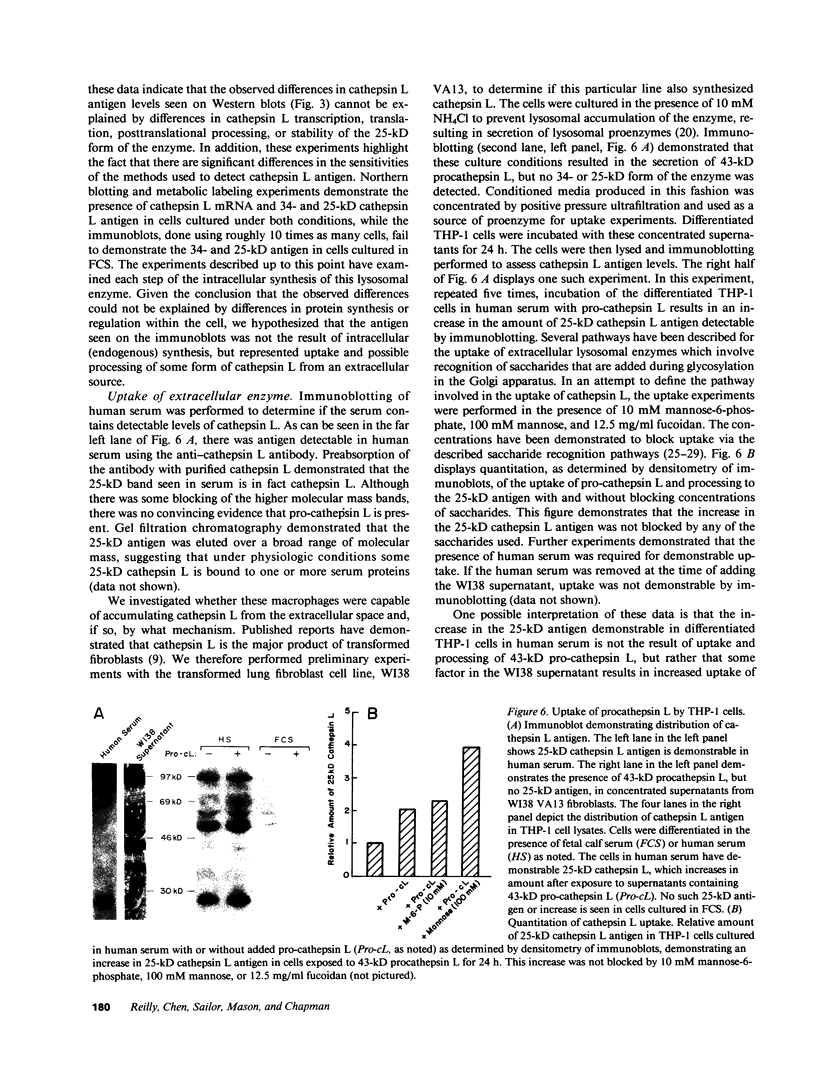
The phorbol myristate acetate (PMA)-differentiated myelomonocytic cell line, THP-1, and human alveolar macrophages contain the cysteine proteinase cathepsin L. This enzyme is synthesized as a 43-kD proenzyme and processed to the active 25-kD form. Differentiation of THP-1 cells in the presence of human serum resulted in an increase in the size of the vacuolar compartment and the accumulation of more 25-kD cathepsin L antigen, as compared with THP-1 cells differentiated in the presence of fetal calf serum. Cells cultured in both types of sera have equivalent levels of cathepsin L mRNA. Metabolic labeling experiments demonstrated equivalent rates of synthesis, processing to the active form, and persistence in both culture conditions. An extracellular source of enzyme was documented by immunoblotting human serum which demonstrated 25-kD cathepsin L antigen; furthermore, we demonstrated that both THP-1 cells, differentiated in human serum, and human alveolar macrophages take up the 43-kD proenzyme and process it to the 25-kD form. Thus, human serum contains a factor(s) that induces both a marked increase in the size of the vacuolar compartment in differentiated THP-1 cells and a novel pathway that is responsible for the uptake and processing of extracellular cathepsin L. The activity of this inducible pathway is a major determinant of levels of intracellular cathepsin L. Cathepsin L is a potent elastase and the regulation of its uptake and processing may play a role in the pathogenesis of disease processes characterized by the destruction of elastin, such as pulmonary emphysema.
Full text
PDF





Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Ahearn M. J., Hamilton T. H., Biesele J. J. Serum-induced formation of lysosomes in HeLa cells: a process sensitive to actinomycin D. Proc Natl Acad Sci U S A. 1966 Apr;55(4):852–857. doi: 10.1073/pnas.55.4.852. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Baron R., Neff L., Brown W., Courtoy P. J., Louvard D., Farquhar M. G. Polarized secretion of lysosomal enzymes: co-distribution of cation-independent mannose-6-phosphate receptors and lysosomal enzymes along the osteoclast exocytic pathway. J Cell Biol. 1988 Jun;106(6):1863–1872. doi: 10.1083/jcb.106.6.1863. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Burnette W. N. "Western blotting": electrophoretic transfer of proteins from sodium dodecyl sulfate--polyacrylamide gels to unmodified nitrocellulose and radiographic detection with antibody and radioiodinated protein A. Anal Biochem. 1981 Apr;112(2):195–203. doi: 10.1016/0003-2697(81)90281-5. [DOI] [PubMed] [Google Scholar]
- Campbell E. J. Human leukocyte elastase, cathepsin G, and lactoferrin: family of neutrophil granule glycoproteins that bind to an alveolar macrophage receptor. Proc Natl Acad Sci U S A. 1982 Nov;79(22):6941–6945. doi: 10.1073/pnas.79.22.6941. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Campbell E. J., Wald M. S. Hypoxic injury to human alveolar macrophages accelerates release of previously bound neutrophil elastase. Implications for lung connective tissue injury including pulmonary emphysema. Am Rev Respir Dis. 1983 May;127(5):631–635. doi: 10.1164/arrd.1983.127.5.631. [DOI] [PubMed] [Google Scholar]
- Campbell E. J., White R. R., Senior R. M., Rodriguez R. J., Kuhn C. Receptor-mediated binding and internalization of leukocyte elastase by alveolar macrophages in vitro. J Clin Invest. 1979 Sep;64(3):824–833. doi: 10.1172/JCI109530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Reilly J. J., Jr, Kobzik L. Role of plasminogen activator in degradation of extracellular matrix protein by live human alveolar macrophages. Am Rev Respir Dis. 1988 Feb;137(2):412–419. doi: 10.1164/ajrccm/137.2.412. [DOI] [PubMed] [Google Scholar]
- Chapman H. A., Jr, Stone O. L., Vavrin Z. Degradation of fibrin and elastin by intact human alveolar macrophages in vitro. Characterization of a plasminogen activator and its role in matrix degradation. J Clin Invest. 1984 Mar;73(3):806–815. doi: 10.1172/JCI111275. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chirgwin J. M., Przybyla A. E., MacDonald R. J., Rutter W. J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry. 1979 Nov 27;18(24):5294–5299. doi: 10.1021/bi00591a005. [DOI] [PubMed] [Google Scholar]
- Fertsch D., Schoenberg D. R., Germain R. N., Tou J. Y., Vogel S. N. Induction of macrophage Ia antigen expression by rIFN-gamma and down-regulation by IFN-alpha/beta and dexamethasone are mediated by changes in steady-state levels of Ia mRNA. J Immunol. 1987 Jul 1;139(1):244–249. [PubMed] [Google Scholar]
- Gabel C. A., Goldberg D. E., Kornfeld S. Identification and characterization of cells deficient in the mannose 6-phosphate receptor: evidence for an alternate pathway for lysosomal enzyme targeting. Proc Natl Acad Sci U S A. 1983 Feb;80(3):775–779. doi: 10.1073/pnas.80.3.775. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Geisow M. J. Fluorescein conjugates as indicators of subcellular pH. A critical evaluation. Exp Cell Res. 1984 Jan;150(1):29–35. doi: 10.1016/0014-4827(84)90698-0. [DOI] [PubMed] [Google Scholar]
- Gonzalez-Noriega A., Grubb J. H., Talkad V., Sly W. S. Chloroquine inhibits lysosomal enzyme pinocytosis and enhances lysosomal enzyme secretion by impairing receptor recycling. J Cell Biol. 1980 Jun;85(3):839–852. doi: 10.1083/jcb.85.3.839. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Janoff A. Elastases and emphysema. Current assessment of the protease-antiprotease hypothesis. Am Rev Respir Dis. 1985 Aug;132(2):417–433. doi: 10.1164/arrd.1985.132.2.417. [DOI] [PubMed] [Google Scholar]
- Jones P. A., DeClerck Y. A. Destruction of extracellular matrices containing glycoproteins, elastin, and collagen by metastatic human tumor cells. Cancer Res. 1980 Sep;40(9):3222–3227. [PubMed] [Google Scholar]
- Joseph L. J., Chang L. C., Stamenkovich D., Sukhatme V. P. Complete nucleotide and deduced amino acid sequences of human and murine preprocathepsin L. An abundant transcript induced by transformation of fibroblasts. J Clin Invest. 1988 May;81(5):1621–1629. doi: 10.1172/JCI113497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaplan A., Achord D. T., Sly W. S. Phosphohexosyl components of a lysosomal enzyme are recognized by pinocytosis receptors on human fibroblasts. Proc Natl Acad Sci U S A. 1977 May;74(5):2026–2030. doi: 10.1073/pnas.74.5.2026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kornfeld S. Trafficking of lysosomal enzymes. FASEB J. 1987 Dec;1(6):462–468. doi: 10.1096/fasebj.1.6.3315809. [DOI] [PubMed] [Google Scholar]
- Kuhlman M., Joiner K., Ezekowitz R. A. The human mannose-binding protein functions as an opsonin. J Exp Med. 1989 May 1;169(5):1733–1745. doi: 10.1084/jem.169.5.1733. [DOI] [PMC free article] [PubMed] [Google Scholar]
- LOWRY O. H., ROSEBROUGH N. J., FARR A. L., RANDALL R. J. Protein measurement with the Folin phenol reagent. J Biol Chem. 1951 Nov;193(1):265–275. [PubMed] [Google Scholar]
- Livi G. P., Cardelli J. A., Mierendorf R. C., Jr, Dimond R. L. Regulation of lysosomal alpha-mannosidase-1 synthesis during development in Dictyostelium discoideum. Dev Biol. 1985 Aug;110(2):514–520. doi: 10.1016/0012-1606(85)90110-1. [DOI] [PubMed] [Google Scholar]
- MacDonald R. G., Pfeffer S. R., Coussens L., Tepper M. A., Brocklebank C. M., Mole J. E., Anderson J. K., Chen E., Czech M. P., Ullrich A. A single receptor binds both insulin-like growth factor II and mannose-6-phosphate. Science. 1988 Mar 4;239(4844):1134–1137. doi: 10.1126/science.2964083. [DOI] [PubMed] [Google Scholar]
- Mason R. W., Gal S., Gottesman M. M. The identification of the major excreted protein (MEP) from a transformed mouse fibroblast cell line as a catalytically active precursor form of cathepsin L. Biochem J. 1987 Dec 1;248(2):449–454. doi: 10.1042/bj2480449. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mason R. W., Johnson D. A., Barrett A. J., Chapman H. A. Elastinolytic activity of human cathepsin L. Biochem J. 1986 Feb 1;233(3):925–927. doi: 10.1042/bj2330925. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McCoy K. L., Schwartz R. H. The role of intracellular acidification in antigen processing. Immunol Rev. 1988 Dec;106:129–147. doi: 10.1111/j.1600-065x.1988.tb00777.x. [DOI] [PubMed] [Google Scholar]
- Musson R. A. Human serum induces maturation of human monocytes in vitro. Changes in cytolytic activity, intracellular lysosomal enzymes, and nonspecific esterase activity. Am J Pathol. 1983 Jun;111(3):331–340. [PMC free article] [PubMed] [Google Scholar]
- Mørland B., Mørland J. Selective induction of lysosomal enzyme activities in mouse peritoneal macrophages. J Reticuloendothel Soc. 1978 Jun;23(6):469–477. [PubMed] [Google Scholar]
- Müller W. E., Renneisen K., Kreuter M. H., Schröder H. C., Winkler I. The D-mannose-specific lectin from Gerardia savaglia blocks binding of human immunodeficiency virus type I to H9 cells and human lymphocytes in vitro. J Acquir Immune Defic Syndr. 1988;1(5):453–458. [PubMed] [Google Scholar]
- Ohkuma S., Poole B. Fluorescence probe measurement of the intralysosomal pH in living cells and the perturbation of pH by various agents. Proc Natl Acad Sci U S A. 1978 Jul;75(7):3327–3331. doi: 10.1073/pnas.75.7.3327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ohsumi Y., Lee Y. C. Mannose-receptor ligands stimulate secretion of lysosomal enzymes from rabbit alveolar macrophages. J Biol Chem. 1987 Jun 15;262(17):7955–7962. [PubMed] [Google Scholar]
- Owada M., Neufeld E. F. Is there a mechanism for introducing acid hydrolases into liver lysosomes that is independent of mannose 6-phosphate recognition? Evidence from I-cell disease. Biochem Biophys Res Commun. 1982 Apr 14;105(3):814–820. doi: 10.1016/0006-291x(82)91042-7. [DOI] [PubMed] [Google Scholar]
- Reilly J. J., Chapman H. A., Jr Association between alveolar macrophage plasminogen activator activity and indices of lung function in young cigarette smokers. Am Rev Respir Dis. 1988 Dec;138(6):1422–1428. doi: 10.1164/ajrccm/138.6.1422. [DOI] [PubMed] [Google Scholar]
- Reilly J. J., Jr, Mason R. W., Chen P., Joseph L. J., Sukhatme V. P., Yee R., Chapman H. A., Jr Synthesis and processing of cathepsin L, an elastase, by human alveolar macrophages. Biochem J. 1989 Jan 15;257(2):493–498. doi: 10.1042/bj2570493. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saraiva E. M., Andrade A. F., de Souza W. Involvement of the macrophage mannose-6-phosphate receptor in the recognition of Leishmania mexicana amazonensis. Parasitol Res. 1987;73(5):411–416. doi: 10.1007/BF00538197. [DOI] [PubMed] [Google Scholar]
- Senior R. M., Connolly N. L., Cury J. D., Welgus H. G., Campbell E. J. Elastin degradation by human alveolar macrophages. A prominent role of metalloproteinase activity. Am Rev Respir Dis. 1989 May;139(5):1251–1256. doi: 10.1164/ajrccm/139.5.1251. [DOI] [PubMed] [Google Scholar]
- Siripont J., Tebo J. M., Mortensen R. F. Receptor-mediated binding of the acute-phase reactant mouse serum amyloid P-component (SAP) to macrophages. Cell Immunol. 1988 Dec;117(2):239–252. doi: 10.1016/0008-8749(88)90115-3. [DOI] [PubMed] [Google Scholar]
- Stahl P. D., Rodman J. S., Miller M. J., Schlesinger P. H. Evidence for receptor-mediated binding of glycoproteins, glycoconjugates, and lysosomal glycosidases by alveolar macrophages. Proc Natl Acad Sci U S A. 1978 Mar;75(3):1399–1403. doi: 10.1073/pnas.75.3.1399. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson J. A., Yirinec B. D., Silverstein S. C. Phorbol esters and horseradish peroxidase stimulate pinocytosis and redirect the flow of pinocytosed fluid in macrophages. J Cell Biol. 1985 Mar;100(3):851–859. doi: 10.1083/jcb.100.3.851. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Swanson J., Burke E., Silverstein S. C. Tubular lysosomes accompany stimulated pinocytosis in macrophages. J Cell Biol. 1987 May;104(5):1217–1222. doi: 10.1083/jcb.104.5.1217. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tsuchiya S., Yamabe M., Yamaguchi Y., Kobayashi Y., Konno T., Tada K. Establishment and characterization of a human acute monocytic leukemia cell line (THP-1). Int J Cancer. 1980 Aug;26(2):171–176. doi: 10.1002/ijc.2910260208. [DOI] [PubMed] [Google Scholar]
- Ullrich K., Mersmann G., Weber E., Von Figura K. Evidence for lysosomal enzyme recognition by human fibroblasts via a phosphorylated carbohydrate moiety. Biochem J. 1978 Mar 15;170(3):643–650. doi: 10.1042/bj1700643. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wessel D., Flügge U. I. A method for the quantitative recovery of protein in dilute solution in the presence of detergents and lipids. Anal Biochem. 1984 Apr;138(1):141–143. doi: 10.1016/0003-2697(84)90782-6. [DOI] [PubMed] [Google Scholar]
- Yednock T. A., Stoolman L. M., Rosen S. D. Phosphomannosyl-derivatized beads detect a receptor involved in lymphocyte homing. J Cell Biol. 1987 Mar;104(3):713–723. doi: 10.1083/jcb.104.3.713. [DOI] [PMC free article] [PubMed] [Google Scholar]
- pdessey R. Regulation of lysosomal proteolysis in burn injury. Metabolism. 1987 Jul;36(7):670–676. doi: 10.1016/0026-0495(87)90152-1. [DOI] [PubMed] [Google Scholar]
- von Figura K., Weber E. An alternative hypothesis of cellular transport of lysosomal enzymes in fibroblasts. Effect of inhibitors of lysosomal enzyme endocytosis on intra- and extra-cellular lysosomal enzyme activities. Biochem J. 1978 Dec 15;176(3):943–950. doi: 10.1042/bj1760943. [DOI] [PMC free article] [PubMed] [Google Scholar]