Abstract
The cytochrome P4501A (CYP1A) enzymes play important roles in the metabolic activation and detoxification of numerous environmental carcinogens, including polycyclic aromatic hydrocarbons (PAHs). In this study, we tested the hypothesis that hepatic CYP1A2 differentially regulates mouse hepatic and pulmonary CYP1A1 expression and suppresses transcriptional activation of human CYP1A1 (hCYP1A1) promoter in response to 3-methylcholanthrene (MC) in vivo. Administration of wild-type (WT) (C57BL/6J) or Cyp1a2-null mice with a single dose of MC (100 μmol/kg i.p.) caused significant increases in hepatic CYP1A1/1A2 activities, apoprotein content, and mRNA levels 1 day after carcinogen withdrawal compared with vehicle-treated controls. The induction persisted in the WT, but not Cyp1a2-null, animals, for up to 15 days. In the lung, MC caused persistent CYP1A1 induction for up to 8 days in both genotypes, with Cyp1a2-null mice displaying a greater extent of CYP1A1 expression. It is noteworthy that MC caused significant augmentation of human CYP1A1 promoter activation in transgenic mice expressing the hCYP1A1 and the reporter luciferase gene on a Cyp1a2-null background, compared with transgenic mice on the WT background. In contrast, the mouse endogenous hepatic, but not pulmonary, persistent CYP1A1 expression was repressed by MC in the hCYP1A1-Cyp1a2-null mice. Liquid chromatography–mass spectrometry experiments showed that CYP1A2 catalyzed the formation of 1-hydroxy-3-MC and/or 2-hydroxy-3-MC, a metabolite that may contribute to the regulation of CYP1A1 expression. In conclusion, the results suggest that CYP1A2 plays a pivotal role in the regulation of hepatic and pulmonary CYP1A1 by PAHs, a phenomenon that potentially has important implications for PAH-mediated carcinogenesis.
Introduction
Polycyclic aromatic hydrocarbons (PAHs) are commonly found in cigarette smoke, coke ovens, diesel exhausts, and charcoal-broiled meats, and numerous PAHs are human carcinogens (Hemminki, 1993). 3-Methylcholanthrene (MC) is one of the most potent PAH carcinogens known to date (Harvey, 1982; Liu et al., 2010). Metabolism of MC by cytochrome P450 (CYP)1 enzymes leads to the formation of highly reactive metabolites that form DNA adducts, a critical event in the initiation of carcinogenesis (Guengerich, 1988). CYP1A enzymes also play an important role in the detoxification of PAHs (Nebert et al., 2004; Shi et al., 2010).
The CYP1A subfamily consists of two proteins, CYP1A1 and CYP1A2, which play important roles in carcinogen bioactivation and detoxification (Guengerich, 1988). The hepatic P4501A1/1A2 enzymes are inducible by a number of chemicals, including the hepatocarcinogen 2,3,7,8-tetrachlorodibenzo-p-dioxin (TCDD) (Poland and Glover, 1974; Postlind et al., 1993), MC (Poland and Glover, 1974; Moorthy, 2000, 2008; Kondraganti et al., 2002), benzo[a]pyrene (Conney 1986, Shi et al., 2010), and cigarette smoke (Kawajiri et al., 1991).
The mechanistic aspects occurring during the CYP1A1 induction process by MC or TCDD have been investigated extensively (Nebert et al., 2004; Fujii-Kuriyama and Mimura 2005; Nukaya et al., 2009), but relatively little is known regarding the decline of drug-metabolizing enzymes to preinduction levels after termination of xenobiotic exposure. We earlier reported that MC elicits a long-term induction of hepatic CYP1A enzymes in rats for up to 45 days (Moorthy et al., 1993). Furthermore, we demonstrated that the sustained induction of CYP1A enzymes is mediated by mechanisms other than the persistence of the parent compound (Moorthy, 2000). We also showed that multiple treatments with MC of mice result in a prolonged induction of CYP1A enzymes in mice (Kondraganti et al., 2002). In Cyp1a2-null mice, the persistent induction of hepatic CYP1A1 was abolished, whereas induction of pulmonary CYP1A1 expression was maintained (Kondraganti et al., 2002). Because the lung is a target organ for pulmonary carcinogenesis mediated by PAHs (Huggins et al., 1961; Rasmussen et al., 1984), it is highly likely that persistent induction of CYP1A1 in the lung contributes to carcinogenesis.
We showed recently that in rats MC causes sustained induction of phase II enzymes, which are also encoded by the Ah gene battery, presumably by persistent activation of the AHR (Kondraganti et al., 2008). We also reported that MC elicits sustained CYP1A1 and CYP1A2 induction in vivo via transcriptional activation of the corresponding promoters (Jiang et al., 2009). Furthermore, we showed that MC elicits persistent induction of CYP1A1 in HepG2 cells in vitro by mechanisms entailing sustained transcriptional activation of the CYP1A1 promoter (Fazili et al., 2010).
Earlier, we postulated that a CYP1A2-dependent metabolite of MC specifically targets the AHREs of the CYP1A1 promoter and regulates CYP1A1 expression by interacting covalently with AHREs (Moorthy, 2002, 2008; Moorthy et al., 2007). Other studies have demonstrated a role for CYP1A2 as a hepatic binding protein for TCDD and polychlorinated dibenzofurans (DeVito et al., 1998; Diliberto et al., 1999). However, CYP1A2 has no effect on the pharmacokinetic behavior of nondioxin-like compounds such as 2,2′,4,4′,5,5′-hexachlorobiphenyl (PCB-153) (Diliberto et al., 1999).
In view of the multiple functions of CYP1A2, we investigated whether CYP1A2 would play a role in the in vivo regulation of CYP1A1 by MC. To this end, we determined whether CYP1A2 plays a mechanistic role in the persistent induction of endogenous hepatic and/or pulmonary CYP1A1 by single or multiple doses of MC in mice in vivo and whether CYP1A2 plays a role in the regulation of human CYP1A1 promoter in vivo.
Materials and Methods
Chemicals.
MC, Calcium chloride, Tris, sucrose, NADP+, ethoxyresorufin, glucose 6-phosphate, and glucose 6-phosphate dehydrogenase were purchased from Sigma-Aldrich (St. Louis, MO). Methoxyresorufin was purchased from Molecular Probes (Carlsbad, CA). Polyvinylidene difluoride membranes and buffer components for electrophoresis and Western blotting were obtained from Bio-Rad Laboratories (Hercules, CA). The primary monoclonal antibody to CYP1A1 was a gift from Dr. P. E. Thomas (Rutgers University, New Brunswick, NJ). Goat anti-mouse IgG conjugated with horseradish peroxidase was from Bio-Rad Laboratories. Anti-AHR antibodies were from Santa Cruz Biotechnology (Santa Cruz, CA). [γ-32P] ATP was obtained from PerkinElmer Life and Analytical Sciences (Waltham, MA). MC metabolite standards, i.e., 1-hydroxy-3-MC (1-OH-MC), 2-hydroxy-3-MC (2-OH-MC), 11-hydroxy-3-MC (11-OH-MC), 11,12-dihydro-11, 12-dihydroxy-3-MC (MC-11.12-diol), 1-one-3-MC (MC-1-one), 11,12-epoxy-11,12-dihydro-3-MC (MC-11,12-didydroepoxide), 11,12-dione-3-MC (MC-11,12-dione), and 11,12-dialdehyde-3-MC (MC-11,12-dialdehyde), were purchased from Chemsyn (Lawrence, KS).
Animals.
Two-month-old wild-type (WT) (C57BL/6J) female inbred mice were obtained from The Jackson Laboratory (Bar Harbor, ME). Breeding pairs of Cyp1a2-null mice of mixed background (C57BL/6J/Sv129) were obtained from Dr. Frank J. Gonzalez (National Cancer Institute, Bethesda, MD). The mice were back-crossed with C57BL/6J mice for 12 generations to generate theoretically a >99.2% C57BL/6J background. Transgenic mice expressing the human 13.2-kb CYP1A1 promoter on a CD-1 background were generated via pronuclear microinjection by using Crl:CD-1 strain oocytes and were obtained from Xenogen Corporation (Alameda, CA) (Zhang et al., 2004; Jiang et al., 2009). The hCYP1A1-luc mice were backcrossed with Cyp1a2-null on the C57BL/6J background for several generations, such that two types of transgenic mice were generated: hCYP1A1-luc-WT (B6:CD-1) and hCYP1A1-luc-Cyp1a2-null (B6:CD-1).
Animal Treatment.
WT and Cyp1a2-null mice were treated with a single dose of MC (100 μmol/kg i.p.) in corn oil (CO) (2 ml/kg) or given an equal volume of CO. At 1, 8, or 15 days after treatment, the animals were sacrificed, and microsomes from lung and liver were isolated. A portion of liver and lung tissues was stored at −80°C for later isolation of nuclei and RNA for electrophoretic mobility-shift assays (EMSAs) and real-time RT-PCR, respectively. For experiments involving transgenic mice, 12-week-old male WT [hCYP1A1-luc-WT (B6:CD-1)] or [hCYP1A1-luc-Cyp1a2-null (B6: CD-1)] mice were treated with MC (100 μmol/kg i.p.) dissolved in CO (2 ml/kg) or CO alone as control once daily for 4 days, and mice were sacrificed at 1, 8, or 15 days after CO or MC withdrawal.
Bioluminescent Imaging.
The substrate luciferin was administered into the peritoneal cavity at a dose of 150 mg/kg (20 mg/ml) approximately 5 min before imaging. Mice were anesthetized with isoflurane/oxygen and placed on the imaging station. Ventral and dorsal images were collected for 1 s by using the IVIS imaging system 100 (Xenogen Corporation) (Zhang et al., 2004; Jiang et al., 2009), and luciferase expression was determined in vivo by bioluminescent imaging (Xenogen Corporation) (Zhang et al., 2004). The animals were acclimatized for 7 days before the start of the experiment and randomized into three groups of six animals each. Tap water and food (Purina Rodent Lab Chow 5001; Purina, St. Louis, MO) were available ad libitum. A 12-h light-to-dark cycle was maintained.
Preparation of Microsomes.
The livers were excised, weighed, and homogenized in 10 mM Tris-HCl, pH 7.4, containing 0.25 M sucrose, and microsomes were prepared by the calcium chloride precipitation method (Cinti et al., 1972; Moorthy et al., 1993; Moorthy, 2000). Microsomes from lungs were prepared by differential centrifugation (Matsubara et al., 1974). Lungs were perfused with cold saline before excision and subsequent preparation of microsomes. Protein concentrations were estimated by the Bradford dye-binding method (Bradford, 1976).
Enzyme Assays.
Ethoxyresorufin O-deethylase (EROD) and methoxyresorufin O-demethylase (MROD) activities in microsomes were assayed essentially as described previously (Moorthy, 2000; Kondraganti et al., 2002; Jiang et al., 2009). Each assay was conducted in duplicate using individual liver or lung microsomes from at least three individual mice for each group.
Electrophoresis and Western Blotting.
Liver (10 μg) or lung microsomes (20 μg of protein) were subjected to SDS-polyacrylamide gel electrophoresis (PAGE) in 7.5% acrylamide gels, followed by Western blotting, as we reported previously (Moorthy, 2000; Kondraganti et al., 2002; Bhakta et al., 2008). Detection of CYP1A1 proteins on the Western blots was accomplished by the use of a monoclonal antibody to CYP1A1 that cross-reacts with CYP1A2 as the primary antibody and goat anti-mouse IgG conjugated with horseradish peroxidase as the secondary antibody. The blots were developed with the color reagent 4-chloro-1-naphthlol or by chemiluminescence (Moorthy, 2000; Kondraganti et al., 2002; Bhakta et al., 2008). The Western blots were conducted in liver or lung microsomes from at least three individual animals or each group.
Real-Time Reverse Transcriptase-Polymerase Chain Reaction.
Total RNA was isolated by using TRIzol reagent (Invitrogen, Carlsbad, CA) according to the manufacturer's instructions. Total RNA (500 ng) from the livers or lungs of MC- or CO-treated controls was subjected to one-step, real-time quantitative TaqMan RT-PCR. The ABI PRISM 7700 Sequence Detection System (Applied Biosystems, Foster City, CA) was used for the RT-PCRs. Gene-specific primers in the presence of TaqMan reverse transcription reagents and RT reaction mix (Applied Biosystems) were used to reverse-transcribe RNA, and TaqMan Universal PCR Master Mix and Assays-on-Demand Gene Expression probes (Applied Biosystems) were used for PCR amplification. The assays included a no RT control (Bookout et al., 2006). After a RT hold for 30 min at 48°C, the samples were denatured at 95°C for 10 min. The thermal cycling step was for 40 cycles at 95°C for 15 s and 40 cycles at 60°C for 1 min. Serial dilutions were used to optimize and validate RT-PCR conditions for CYP1A1 and 18S genes. Each data point was repeated three times. Quantitative values were obtained from the threshold PCR cycle number (Ct) at which the increase in signal was associated with an exponential growth of PCR product after it started to be detected (Bookout et al., 2006). The relative mRNA levels for CYP1A1 were normalized to their 18S content. The relative expression levels of the target gene were calculated as reported previously (Jiang et al., 2004, 2009; Bhakta et al., 2008).
Isolation of Nuclei.
Nuclei and nuclear protein extracts from whole cells were prepared by using commercially available kits (Pierce, Rockford, IL) following the manufacturer's instructions. The nuclear protein extracts were stored at −80°C until use.
Electrophoretic Mobility-Shift Assays.
EMSAs were performed with nuclear extracts as described previously (Moorthy, 2000; Bhakta et al., 2008). In brief, the nuclear proteins (10 μg) were preincubated with poly(dI-dC) (2 μg) on ice for 10 min, followed by incubation at room temperature for 30 min with γ-32P-labeled double-stranded oligonucleotides that contained the AHREs (Pendurthi et al., 1993) in the presence of EMSA buffer (25 mM HEPES, 0.5 mM EDTA, 0.5 mM dithiothreitol, 10% glycerol, 50 mM KCl, 5 mM MgCl2, pH 7.5). In some experiments, the nuclear extracts were incubated with a 50-fold excess of cold AHRE-specific oligonucleotide before the addition of γ-32P-labeled probe. The labeled samples were separated by nondenaturing PAGE (6%) at 200 V for 3 h. The gel was dried, and radioactivity was located by autoradiography using Kodak X-ray film (Eastman Kodak, Rochester, NY). The EMSAs were performed by using nuclear extracts from cells treated in triplicate with dimethyl sulfoxide or MC in three independent experiments for each time point.
Liquid Chromatography-Mass Spectrometry Analyses of Parent MC and Its Metabolites.
MC and its metabolites were detected and quantified from liver and lung tissues by LC-MS/MS using a QuattroPremierXE triple quadrupole mass spectrometer equipped with an Acuity UPLC (Waters, Milford, MA). The compounds were detected in positive electrospray ionization by using the selected reaction monitoring mode. Tissue samples were mechanically homogenized in phosphate-buffered saline buffer to a 100 mg/ml concentration (wt/vol). One hundred microliters of homogenate was extracted twice by using 5 ml of methyl tertiary butyl ether. The extracts were combined and evaporated to dryness under nitrogen gas stream. The dried extracts were reconstituted with 100 μl of methanol and analyzed by LC-MS/MS.
MC and its metabolites were chromatographically resolved by using a Luna C5 3μ 2 × 50-mm analytical column (Phenomenex, Torrance, CA) using a 0.2% formic acid in water (A) and methanol (B) linear gradient. Column temperature was maintained at 50°C, and sample temperature was maintained at 5°C. Initial gradient conditions consisted of 70% B for 0.5 min then increased linearly to 98% B in 3.5 min and were held at this level for an additional 2 min before returning to initial conditions. The identity of the MC UV peak was confirmed on the basis of retention time, and precursor-product ion transition was measured by mass spectrometry (m/z 269.2 > 253.9, corresponding to M + 1 and M − 15); retention time at UV = 2.37 min (Fazili et al., 2010). MC metabolites were also identified on the basis of specific mass transitions and retention times as described under Results.
Statistical Analysis.
Data obtained from individual animals are expressed as means ± S.E. Statistical significance between control and treated groups for each time point was assessed by two-way analyses of variance (ANOVA) or Students' t tests, with P < 0.05 considered significant.
Results
Hepatic CYP1A1 and 1A2 Enzyme Activities.
Hepatic EROD and MROD activities were assayed as indicators of CYP1A1 and 1A2 activities, respectively (Sinclair et al., 1998; Moorthy, 2000). On the first day after a single dose of MC, EROD activities of WT mice were 3.4 times greater than those in animals treated with vehicle CO (Fig. 1). The EROD activities in the MC samples were persistently higher, being 1- and 0.8-fold greater than those of controls at 8 and 15 days, respectively (Fig. 1A). In Cyp1a2-null animals, MC treatment led to a 6-fold increase in EROD activities over those of vehicle-treated animals at the 1-day time point. By day 8, the induction in the MC-treated samples declined, being 60% higher than controls, and by day 15, the EROD activities were not significantly different from controls (Fig. 1A).
Fig. 1.
Effect of MC on hepatic EROD/MROD activities and CYP1A apoprotein contents. Eight-week-old female WT or Cyp1a2-null mice were treated with a single dose of MC (100 μmol/kg) or vehicle (CO) and sacrificed 1, 8, or 15 days after dosing. A and B, EROD (A) and MROD (B) activities were estimated in the liver microsomes of these animals. Values represent mean ± S.E. (n = 3). ∗, Statistically significant differences between MC- and vehicle-treated samples at P < 0.05, as determined by two-way ANOVA and modified t tests. C, representative Western blot showing the effect of MC on CYP1A1/1A2 protein expression in liver microsomes. WT and Cyp1a2-null animals were treated with MC as described above, and CYP1A1/1A2 apoprotein expression was analyzed in microsomes (10 μg) at the indicated time points by Western blotting.
MC administration also resulted in persistent elevation of hepatic MROD activities in the WT animals (Fig. 1B). The hepatic CYP1A2 activities in the MC-treated animals were 3.3-, 0.6-, and 0.2-fold higher than the corresponding controls at the 1-, 8-, and 15-day time points, respectively (Fig. 1B). In the Cyp1a2-null animals, MROD activities were barely detectable in the vehicle-treated mice. In MC-treated animals, MROD activities were significantly higher at the 1-day time point, but similar to control at the 8- and 15-day time points (Fig. 1B).
Hepatic CYP1A1/1A2 Apoproteins.
As shown in Fig. 1C, liver microsomes from WT animals treated with vehicle displayed a band corresponding to CYP1A1/CYP1A2 proteins (52 kDa). Treatment of animals with MC led to a significant increase in the hepatic expression of CYP1A1 and CYP1A2 at the 1-day time point in WT mice (Fig. 1C). The induction of CYP1A1 declined by day 8 in both WT and Cyp1a2-null mice (Fig. 1C). Similar trends were noticed for CYP1A2 in the WT mice (Fig. 1C). In the Cyp1a2-null animals, CYP1A2 protein was not detectable in vehicle- or MC-treated mice (Fig. 1C).
Hepatic CYP1A1/1A2 mRNA Levels.
To determine whether the sustained induction of CYP1A1/1A2 enzyme activities and apoprotein contents was accompanied by a corresponding modulation in mRNA levels, we performed real-time RT-PCR analyses on total RNA from livers isolated from control and MC-exposed WT and Cyp1a2-null animals using probes specific for CYP1A1 or CYP1A2 mRNA. A single dose of MC treatment led to strong induction of CYP1A1 mRNA species (Table 1). The induction was strongest at 1 day (460-fold) and continued through 8 days (15-fold increase), and even after 15 days, the expression of CYP1A1 was elevated in MC-treated samples (Table 1). In the Cyp1a2-null animals, MC exposure led to a much greater extent of induction, being 1800-, 32-, and 5-fold higher than the corresponding controls at the 1-, 8-, and 15-day time points, respectively (Table 1). MC also caused significant induction of CYP1A2, albeit to a much lesser extent than CYP1A1 (Table 1). CYP1A2 mRNA was not detectable in the Cyp1a2-null mice (Table 1).
TABLE 1.
Effect of MC on hepatic CYP1A mRNA expression
WT or Cyp1a2-null mice were treated with vehicle CO or a single dose of MC (100 μmol/kg), and hepatic CYP1A1 or CYP1A2 mRNA expression was determined by real-time RT-PCR, as described under Materials and Methods. Values mean represent mean ± S.E. of fold increases in gene expression in the MC-treated animals compared with CO controls.
| Genotype | Fold Induction |
|||||
|---|---|---|---|---|---|---|
| CYP1A1 |
CYP1A2 |
|||||
| 1 Day | 8 Days | 15 Days | 1 Day | 8 Days | 15 Days | |
| WT | 460 ± 52* | 15 ± 2.2* | 3 ± 0.4* | 28 ± 3.2* | 3.1 ± 0.4* | 1.8 ± 0.2* |
| Cyp1a2-null | 1800 ± 200* | 32 ± 4.0* | 5 ± 0.7* | N.D. | N.D. | N.D. |
N.D., not determined.
Statistically significant differences between MC- and CO-treated samples at P < 0.05, as determined by one-way ANOVA.
Pulmonary CYP1A1 Expression.
The effects of MC on EROD activities in lungs of WT and Cyp1a2-null animals at selected time points are shown in Fig. 2. Comparison of EROD activities between MC-exposed and control WT animals revealed a 5-fold induction of EROD by MC at 1 day (Fig. 2A). On day 8, the EROD activities were persistently induced, being 14 times higher than those of vehicle-treated animals. In the Cyp1a2-null animals, MC caused a 2-fold increase in EROD activities at the 1-day time point (Fig. 2A). At the 8-day time point, EROD activities were approximately 6-fold higher than control (Fig. 2A). The EROD activities declined to control in MC-treated WT and Cyp1a2-null mice (Fig. 2A).
Fig. 2.
Effect of MC on pulmonary EROD activities (A) and CYP1A1 apoprotein content (B). A, eight-week-old female WT or Cyp1a2-null mice were treated with a single dose of MC as described in the legend to Fig. 1, and EROD activities were estimated in the lung microsomes of the animals. Values represent mean ± S.E. (n = 3). ∗, Statistically significant differences between MC- and vehicle-treated samples at P < 0.05, as determined by two-way ANOVA and modified t tests. B, representative Western blot showing the effect of MC on CYP1A1 protein expression in lung microsomes. WT and Cyp1a2-null animals were treated with MC as described above, and CYP1A1 apoprotein expression was analyzed in the microsomes (10 μg) at the indicated time points by Western blotting.
CYP1A1 apoprotein expression was analyzed in the lung microsomes by Western blotting. As shown in Fig. 2B, MC caused a marked induction of CYP1A1 apoprotein in the WT animals at the 1-day time point. The augmented expression was maintained at 8 days. Even at the 15-day time point, although the magnitude of induction declined, the expression was still higher than control (Fig. 2B). In the Cyp1a2-null mice, the induction of CYP1A1 expression was much higher at 1 and 8 days, compared with the WT counterparts (Fig. 2B). By 15 days, the expression of CYP1A1 in the MC-treated mice was similar to controls (Fig. 2B). It is noteworthy that even in vehicle controls the CYP1A1 expression in the Cyp1a2-null mice was much higher than that observed in WT mice (Fig. 2B).
The effects of MC on pulmonary CYP1A1 mRNA expression was studied by real-time RT-PCR. As shown in Table 2, MC elicited a 600-fold induction of CYP1A1 mRNA at the 1-day time point in the WT mice. The extent of induction at the 8- and 15-day time points was 26- and 2-fold, respectively over controls. In Cyp1a2-null mice, MC caused a 360- and 13-fold induction, respectively, at the 1- and 8-day time points (Table 2). By 15 days, the CYP1A1 mRNA levels were similar to controls.
TABLE 2.
Effect of MC on pulmonary CYP1A1 mRNA expression
WT or Cyp1a2-null mice were treated with vehicle CO or a single dose of MC (100 μmol/kg), and pulmonary CYP1A1 mRNA expression was determined by real-time RT-PCR, as described under Materials and Methods. Values mean represent mean ± S.E. of fold increases in gene expression in the MC-treated animals compared with CO controls.
| Genotype | Fold Induction |
||
|---|---|---|---|
| 1 Day | 8 Days | 15 Days | |
| WT | 600 ± 70* | 26 ± 3 | 2 ± 0.4* |
| Cyp1a2-null | 360 ± 40* | 13 ± 1.4* | 1.1 ± 0.2 |
Statistically significant differences between MC- and CO-treated samples at P < 0.05, as determined by one-way ANOVA.
MC-AHR Levels In Vivo by EMSA.
To determine whether the sustained elevation of hepatic CYP1A1 expression by MC in the WT mice was caused by persistent binding of the MC–AHR–ARNT complex to the AHRE elements on the CYP1A1 promoter, we conducted EMSAs using nuclear extracts from the livers of CO- and MC-treated WT and Cyp1a2-null mice at different time points. As shown in Fig. 3, MC-treated samples in WT and Cyp1a2-null mice showed a band shift corresponding to the binding of the MC–AHR–ARNT complex to AHREs present in the labeled oligonucleotides that we used. It is noteworthy that the intensity of the band shift was stronger in the Cyp1a2-null mice. The band shift was competed off in the presence of excess cold probe. At later time points, the intensities of the band got weaker and were similar to control by 15 days in both WT and Cyp1a2-null mice treated with MC (Fig. 3).
Fig. 3.
Representative EMSA of hepatic nuclear extracts from CO- and MC-treated mice. WT and Cyp1a2-null mice were treated with CO or MC as described in the legend to Fig. 1. Nuclear protein extracts from treated livers were subjected to EMSA, as described under Materials and Methods. The labeled nuclear proteins were separated by PAGE, and the gels were dried. The gels were exposed to autoradiography at −80°C for 24 h. The arrow indicates the interaction with the AHREs of an MC-specific nuclear protein that seems to be the MC–AHR–ARNT complex, which is competed off in the presence of 50-fold excess of cold AHRE-specific oligonucleotide (see lanes labeled Cold Probe).
In Vivo Levels of MC in Hepatic and Pulmonary Tissues of Mice.
Levels of parent MC were measured by LC-MS/MS to determine whether the persistent induction of CYP1A1 by MC in the WT or Cyp1a2-null mice was caused by persistence of the parent MC in the livers and lungs of MC-exposed mice. Figure 4A shows a representative LC of MC, which had a retention time of 2.3 min. MC was identified by MS/MS (Fig. 4B), which had a characteristic M + 1 peak at 269, and M + 1-15 peak at 253.9. As shown in Fig. 4C, MC levels in the livers of WT and Cyp1a2-null mice were 2 and 2.8 ng/mg tissue, respectively. In the lung, MC levels were 4 and 2 ng/mg tissue, respectively, in the WT and Cyp1a2-null mice (Fig. 4C). MC was not detectable in WT or Cyp1a2-null mice at 8 (not shown) or 15 days (Fig. 4C).
Fig. 4.
LC-MS/MS analyses of MC. Parent MC in livers and lungs was analyzed by LC-MS/MS. A, UV chromatogram of MC standard (I). B, multiple reaction monitoring transition showing the primary product ion MC at m/z 253.9, which represents M+ + 1-15 (loss of methyl group) fragment. C, parent MC was estimated in the livers and lungs of WT and Cyp1a2-null mice exposed to MC at 1 and 15 days after treatment. Values represent mean ± S.E. (n = 3).
In Vivo Levels of MC Metabolites in Hepatic and Pulmonary Tissues of Mice.
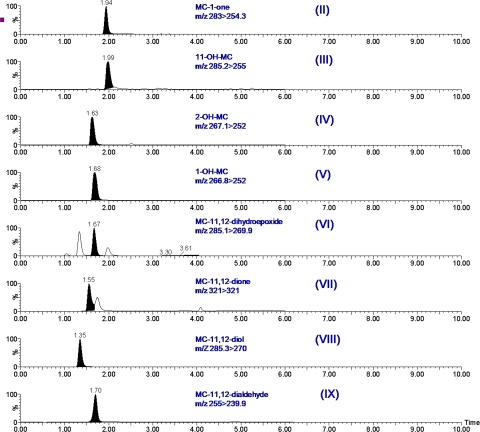
We tested the hypothesis that a CYP1A2-dependent metabolite of MC contributes to the suppression of CYP1A1 induction by MC. For this, liver and lung tissues of vehicle- and MC-treated WT or Cyp1a2-null mice were analyzed for the presence of MC metabolites by LC-MS/MS, as described under Materials and Methods. MC metabolites in tissues were identified and quantified by using eight MC metabolite standards (Fig. 5s) that were subjected to LC-MS/MS (Fig. 6). The chromatographic profiles, retention times, and mass transitions of the metabolites are depicted in Fig. 6. With the exception of 1-OH-MC and 2-OH-MC, which could not be separated chromatographically under the experimental conditions we used, each of the other MC metabolites could be resolved (Fig. 6).
Fig. 5.
Chemical structures of MC (I) and the various metabolite standards (II–IX) used in the study. I, MC; II, MC-1-one; III, 11-hydroxy-3-MC; IV, 2-OH-MC; V, 1-OH-MC; 1-hydroxy-3-MC; VI, MC-11,12-dihydroepoxide; VII, 11,12-dione-3-MC; VIII, 11,12-dihydro-11,12-dihydroxy-3-MC; and IX, MC-11,12-dialdehyde.
Fig. 6.
LC-MS/MS analyses of MC metabolites. The different MC metabolite standards (II–IX) depicted in Fig. 5 were analyzed by LC-MS/MS, and the mass transitions and retention times of each metabolite are shown.
Among the metabolites that we estimated in the tissues, 1-OH-MC and 2-OH-MC were detectable and quantifiable. MC-1-one, MC-11,12-diol, and MC-11,12-dialdehyde were detectable in liver and lung tissues, but the levels were too low to allow quantification (not shown). As shown in Fig. 7, 1-OH-MC and 2-OH-MC were detectable in livers of WT mice at 1 and 15 days, but the combined levels were markedly lower in the Cyp1a2-null mice. The levels of these metabolites in the lung were much lower than those of liver (Fig. 7). In both tissues, the levels of the metabolites were diminished by day 15.
Fig. 7.
Levels of 1-OH-MC and 2-OH-MC mouse tissues exposed to MC. WT or Cyp1a2-null mice were treated with MC as described in the legend to Fig. 1, and combined levels of hepatic and pulmonary 1-OH-MC and 2-OH-MC were estimated at the 1- and 15-day time points by LC-MS/MS. Values represent mean ± S.E. (n = 3). ∗, Statistically significant differences between WT and Cyp1a2-null groups at P < 0.05, determined by Student's t test. ND, not detected.
Effect of MC on hCYP1A1-luc Expression in WT and Cyp1a2-null Mice.
Because our results with Cyp1a2-null mice showed a mechanistic role for CYP1A2 in the regulation of endogenous and chemically inducible CYP1A1, we tested the hypothesis that mouse CYP1A2 will repress the expression of human CYP1A1 in vivo. For this we used transgenic mice expressing the human CYP1A1 promoter and the reporter luciferase gene (hCYP1A1-Luc) that were on WT (CD-1/C57BL/6J) (hCYP1A1-luc-WT) or Cyp1a2-null (hCYP1A1-luc-Cyp1a2-null) backgrounds. As shown in Fig. 8A, MC persistently induced luciferase expression in WT mice for up to 15 days. In mice that were on the Cyp1a2-null background, MC elicited luciferase expression at a much higher extent than that observed in mice on the WT background (Fig. 8B). Quantitative analyses indicated that MC caused a 5.6-, 2.5-, and 0.6-fold induction of luciferase at the 1-, 8-, and 15-day time points in the WT mice (Fig. 8C). In mice on the Cyp1a2-null background, MC elicited 36.5- and 5-fold induction in luciferase expression at the 1- and 8-day time points. At 15 days, luciferase expression had declined to less than control levels (Fig. 8C). Although the bioluminescent imaging was done to study the expression of luciferase in different internal organs, the observations in Fig. 8 suggest, based on the location of light emission from the mouse, that a substantial extent of luciferase expression was contributed by the liver. The specific expression of luciferase in each of the other organs, including lung, was not known because we did not conduct ex vivo luciferase assays in the excised tissues.
Fig. 8.
Bioluminescent imaging of hCYP1A1-luc-WT and hCYP1A1-luc-Cyp1a2-null mice after MC treatment. A and B, adult male hCYP1A1-luc-WT (A) or hCYP1A1-luc-Cyp1a2-null (B) mice were treated with CO or MC (100 μmol/kg), once daily for 4 days, and luciferase expression was analyzed by bioluminescent imaging in real time at 1, 8, or 15 days after MC or CO withdrawal. C, quantitation of bioluminescent imaging data was conducted with IVIS imaging software. Values represent mean ± S.E. (n = 5). ∗, Statistically significant differences between MC- and CO-treated mice at P < 0.05, as determined by two-way ANOVA and modified t tests.
Effect of MC on Endogenous Hepatic CYP1A Expression in Transgenic Mice.
We tested the hypothesis that MC will differentially regulate hepatic mouse endogenous CYP1A1 and CYP1A2 in transgenic mice expressing the hCYP1A1 promoter that are on the WT or Cyp1a2-null background. As shown in Fig. 9A, MC caused a 100-, 44-, and 3-fold induction of EROD activities at the 1-, 8-, and 15-day time points in hCYP1A1-luc-WT mice. In hCYP1A1-luc-Cyp1a2-null mice, MC caused a 100-, 24-, and 1-fold induction of hepatic EROD activities (Fig. 9C). MC also caused persistent induction of MROD activities in the hCYP1A1-luc-WT mice (Fig. 9B). The induction of MROD in the Cyp1a2-null mice also reflected CYP1A1 activities, because these mice were devoid of CYP1A2. Western blotting analyses showed that EROD and MROD activities in the MC-treated WT mice correlated with CYP1A1 and CYP1A2 apoprotein contents. In Cyp1a2-null mice, MC caused induction of CYP1A1 to a lesser extent, and the induction was less persistent (Fig. 9C).
Fig. 9.
Effect of MC on endogenous hepatic EROD (CYP1A1), MROD, and CYP1A apoprotein contents. A and B, adult male hCYP1A1-luc-WT (A) or hCYP1A1-luc-Cyp1a2-null (B) mice were treated with CO or MC (100 μmol/kg), once daily for 4 days, and EROD (A) and MROD (B) activities were analyzed as described under Materials and Methods. Data represent mean ± S.E. of EROD or MROD activities from at least four individual animals. C, representative Western blots showing the effects of MC on endogenous hepatic CYP1A1 and CYP1A2 (left) and pulmonary CYP1A1 protein (right) expression in mice. hCYP1A1-luc-WT (left) or hCYP1A1-luc-Cyp1a2-null (right) mice were treated with CO or MC as described above, and CYP1A1/1A2 apoprotein expression was analyzed in the microsomes (20 μg) at the indicated time points by Western blotting.
Effect of MC on Endogenous Pulmonary CYP1A1 Expression in Transgenic Mice.
We determined the effects of MC on endogenous pulmonary CYP1A1 expression in hCYP1A1-Luc-WT and hCYP1A1-Cyp1a2-null mice. As shown in Fig. 10A, MC caused a 1.4-, 1.9-, and 1.7-fold induction of endogenous pulmonary EROD (CYP1A1) expression in the WT mice at the 1-, 8-, and 15-day time points, respectively. In the Cyp1a2-null mice, MC caused 5.4-, 6.1-, and 7.8-fold induction of CYP1A1 at the 1-, 8-, and 15-day time points, respectively (Fig. 10A). Western blotting analyses (Fig. 10B) showed that CYP1A1 apoprotein levels correlated with EROD activities.
Fig. 10.
Effect of MC on endogenous pulmonary EROD (CYP1A1) and CYP1A1 apoprotein content. A, adult male hCYP1A1-luc-WT or hCYP1A1-luc-Cyp1a2-null mice were treated with CO or MC (100 μmol/kg), once daily for 4 days, and EROD activities were analyzed as described under Materials and Methods. Data represent mean ± S.E. of EROD activities from at least four individual animals. B, representative Western blot showing the effect of MC on endogenous pulmonary CYP1A1 protein expression in mice. hCYP1A1-luc-WT (left) or hCYP1A1-luc-Cyp1a2-null (right) mice were treated with CO or MC as described above, and CYP1A1 apoprotein expression was analyzed in the microsomes (20 μg) at the indicated time points by Western blotting.
Discussion
In the present study, using the Cyp1a2-null mouse model, we investigated the mechanisms of sustained CYP1A1 induction by MC and tested the hypothesis that CYP1A2, which is expressed primarily in the liver, would play a regulatory role in the induction of hepatic and pulmonary CYP1A1 induction by MC. The marked increases in hepatic EROD activities in WT animals exposed to MC for up to 15 days (Fig. 1A) suggest that MC elicited a persistent induction of CYP1A1 enzyme in mice. This finding was in agreement with our previous findings showing sustained induction of CYP1A enzymes by MC in the rodent models (Moorthy et al., 1993; Moorthy, 2000; Kondraganti et al., 2002). The lack of a persistent effect of MC on hepatic EROD activities in Cyp1a2-null mice provides strong evidence for an important role for CYP1A2 in the maintenance of CYP1A1 induction by MC (Kondraganti et al., 2002). The fact that MC elicited induction of hepatic MROD activities in WT mice for up to 15 days (Fig. 1B) supported the hypothesis that this PAH persistently induces CYP1A2 in mice.
Our finding that sustained induction of the enzyme activities in the WT animals was paralleled by persistent induction of CYP1A1/1A2 apoprotein supports the idea that the phenomenon of persistent induction of CYP1A1/1A2 activities is a result of long-term induction of their corresponding apoproteins. The real-time RT-PCR experiments showing sustained induction of hepatic CYP1A1 and CYP1A2 mRNAs by MC in the WT animals (Table 1) indicate that the persistent induction of CYP1A1 and CYP1A2 enzymes was preceded by prolonged up-regulation of the corresponding genes. Our finding that MC elicited induction of CYP1A1 mRNA to a much greater extent in Cyp1a2-null mice than WT mice at the 1-day time point supported the hypothesis that in the WT animal CYP1A2 suppresses hepatic CYP1A1 gene expression. Liang et al. (1997) did not see any differences between WT and Cyp1a2-null mice in the sensitivity of mRNA inducibility by TCDD. The differences in our studies and those of Liang et al. (1997) could be attributable to the different compounds that were used to induce CYP1A1. Although the level of CYP1A1 mRNA in the Cyp1a2-null mice exposed to MC was much higher than WT at the 1-day time point (Table 1), the mRNA levels fell drastically by days 8 and 15, suggesting that relative to the time point, the CYP1A1 mRNA levels in the Cyp1a2-null mice were not persistent.
The observation that MC elicited sustained induction of EROD (CYP1A1) (Fig. 2A), CYP1A1 apoprotein content (Fig. 2B), and mRNA (Table 2) in the lungs of WT and Cyp1a2-null animals suggested that this phenomenon is of relevance to carcinogenesis because lung is a target organ for MC-mediated and other PAH-mediated cancers (Rasmussen et al., 1984; Shi et al., 2010). In fact, we observed a greater expression of pulmonary CYP1A1 in untreated Cyp1a2-null animals than in WT mice, supporting the hypothesis that hepatic CYP1A2 suppresses the expression of endogenous CYP1A1 expression in the lung, because the lung tissue is devoid of CYP1A2.
To determine whether the sustained induction of CYP1A1 in the livers of MC-treated mice was preceded by sustained binding of the MC–AHR–ARNT complex to the CYP1A1 promoter in the nucleus, we conducted EMSAs with labeled oligonucleotides containing multiple AHREs. Our observation (Fig. 3) showing a band shift corresponding to the MC–AHR–ARNT complex at 1 day, but not at later time points, in WT and Cyp1a2-null mice suggested that the persistent induction of CYP1A1 by MC was mediated by AHR-independent mechanisms. The greater intensity of the band shift in the Cyp1a2-null mice at the 1-day time point compared with that observed in WT mice was consistent with the greater magnitude of induction of CYP1A1 mRNA and protein expression in the Cyp1a2-null animals. Our LC-MS/MS experiments (Fig. 4) showing the absence of parent compound at the 15-day time point supported the hypothesis that the sustained CYP1A1/1A2 induction was not caused by persistence of MC in the hepatic and pulmonary tissues. These data were also in agreement with our findings pertaining to the uptake, distribution, and elimination of [3H]MC in vivo in the rat (Moorthy, 2000) and HepG2 cells (Fazili et al., 2010).
The mechanisms underlying the role of CYP1A2 in the regulation of hepatic or pulmonary CYP1A1 expression have not yet been determined. Our LC-MS/MS experiments on the metabolism of MC in vivo (Fig. 7) clearly showed an important role for CYP1A2 in the formation of 1-OH-MC or 2-OH-MC (Fig. 6), because the levels of these metabolites were substantially lower in the livers or lungs of Cyp1a2-null mice. It is likely that a 2-OH-MC was the major metabolite, because previous studies (Gangarosa and Stoming, 1983; Shou and Yang, 1990) have shown 2-OH-MC to be formed to a much higher extent than 1-OH-MC. In the lung, which is devoid of CYP1A2, it is possible that the 2-OH-MC generated in liver is transported to the lung where the effects are exerted. The possibility that this metabolite was generated by another closely related CYP enzyme (e.g., CYP1A1, CYP1B1) in the lung has not been excluded. The fact that we could not detect appreciable amounts of other MC metabolites (Figs. 5 and 6) could have been caused in part by the excretion of these metabolites in the urine or feces (Kinoshita et al., 1980) or covalent binding to protein or DNA (Moorthy et al., 1993).
To determine the role of hepatic CYP1A2 in the regulation of human CYP1A1, we treated mice expressing the human CYP1A1-luc on a WT (CD-1/B6) or Cyp1a2-null background. Our finding that MC elicited sustained induction of luciferase expression (Fig. 8) for up to 15 days indicated that MC caused persistent transcriptional activation of human CYP1A1 promoter in vivo, a phenomenon that was in agreement with our observations in vitro in HepG2 cells (Fazili et al., 2010). Our observation that MC elicited luciferase expression to a much greater extent in transgenic mice on the Cyp1a2-null background than those on a WT background suggested that CYP1A2 repressed the transcription of human CYP1A1 in the WT mice, a phenomenon that was also seen in the inducibility of mouse hepatic CYP1A1 by MC in the mouse (Fig. 1; Table 1). The fact that mouse MC-inducible luciferase expression was not sustained in the hCYP1A1-luc-Cyp1a2-null mice was consistent with similar observations in the hepatic expression of CYP1A1 in MC-treated Cyp1a2-null mice (Fig. 1). The augmented induction of pulmonary CYP1A1 in the Cyp1a2-null mice in the MC-treated animals supported the hypothesis that CYP1A2 suppresses CYP1A1 expression.
We postulate the following mechanism for suppression of hepatic and pulmonary CYP1A1 expression by CYP1A2. In the liver, CYP1A2 catalyzes the formation of 2-OH-MC, which could undergo further metabolism to 2-OH-MC-X, which then complexes with the AHR and gets targeted to the nucleus, wherein it could covalently bind to the AHREs on the CYP1A1 promoter and suppress CYP1A1 gene expression (Fig. 11). Because the lung is devoid of CYP1A2, it is possible that 2-OH-MC, generated by hepatic CYP1A2, is transported to the lung, wherein it could metabolized to 2-OH-MC-X, eventually leading to the suppression of CYP1A1 expression via sequence-specific DNA adduct formation on the CYP1A1 promoter. In fact, our LC-MS/MS data (Fig. 7) did show the presence of 2-OH-MC in the lungs of WT mice, but not in Cyp1a2-null mice. Support for the sequence-specific nature of DNA adducts on the CYP1A1 promoter stems from our finding that MC, upon CYP1A2-dependent metabolic activation in vitro, yields metabolites that covalently bind to plasmids (DNA adducts) containing CYP1A1 promoter (Moorthy, 2002, 2008).
Fig. 11.
Possible mechanisms of the role of hepatic CYP1A2 in the regulation of hepatic and pulmonary CYP1A1 by MC. We hypothesize that MC upon entry into liver is metabolized by the liver-specific CYP1A2 to 2-OH-MC, which upon further metabolism results in the formation of a reactive metabolite such as 2-OH-MC-X, which complexes with the AHR and is transported to the nucleus. In the nucleus, the 2-OH-MC-X binds covalently to the AHREs, resulting in suppression of hepatic CYP1A1 induction. Because the lung is devoid of CYP1A2, it is possible that the 2-OH-MC generated in the liver is transported to the lung, wherein it gets activated to 2-OH-MC-X, which in turn suppresses CYP1A1 expression, eventually leading to decreased persistence of DNA adducts and attenuation of pulmonary tumorigenesis.
The suppression of pulmonary CYP1A1 could result in attenuation of persistent DNA adducts and pulmonary tumorigenesis. In fact, our studies showing increased susceptibility of Cyp1a2-null mice to PAH-induced pulmonary tumorigenesis (Moorthy et al., 2010; unpublished results) supports our hypothesis regarding the protective role of CYP1A2 against pulmonary carcinogenesis.
Although our data support the hypothesis that CYP1A2 suppresses CYP1A1 induction in the liver and lung by MC, we did observe differences in the persistence of CYP1A1 induction in the two organs, with CYP1A2 contributing to sustained induction of CYP1A1 in liver, but not lung (Figs. 9 and 10). We postulate that in the liver CYP1A2 catalyzes the formation of a different MC metabolite that could form adducts on the CYP1A1 promoter region that is normally occupied by chromatin remodeling factors such as histone deacetylase I and/or DNA methyltransferase I, which silence gene expression in the noninduced state (Schnekenburger et al., 2007). These adducts may have prevented the binding of histone deacetylase I and/or DNA methyltransferase to the CYP1A1 promoter, resulting in continued activation of the CYP1A1 gene.
In conclusion, the results of the present study strongly suggest that hepatic CYP1A2 plays a critical role in the differential regulation of hepatic and pulmonary CYP1A1 expression by MC. The potential significance of these observations in pulmonary carcinogenesis warrants further in-depth investigations to elucidate the molecular regulation of CYP1A1/1A2 by PAHs.
Acknowledgments
We thank Dr. P. E. Thomas (Rutgers University, New Brunswick, NJ) for monoclonal antibodies against CYP1A1/1A2; Dr. F. J. Gonzalez (National Cancer Institute, Bethesda, MD) for breeding pairs of Cyp1a2-null mice; Dr. Weisheng Zhang (Xenogen Corporation, Alameda, CA) for breeding pairs of hCYP1A1-Luc mice; and Ms. Danielle Gregory and Nsikak E. Inman for help in the preparation of this manuscript.
This work was supported in part by the National Institutes of Health National Institute of Environmental Health Sciences [Grant RO1-ES009132] and the National Institutes of Health National Heart Lung and Blood Institute [Grants RO1-HL087174, RO1-HL070921] (to B.M.).
Parts of this work were presented previously: Moorthy B, Wang L, and Jiang W (2010) Reciprocal roles of cytochromes P4501A1 and 1A2 in polycyclic aromatic hydrocarbon (PAH)-mediated tumorigenesis in mice: Implications for lung cancer in humans, at the 101st Annual American Association for Cancer Research Meeting; 2010 April 17–21; Washington, DC. American Association for Cancer Research, Philadelphia, PA.
Article, publication date, and citation information can be found at http://jpet.aspetjournals.org.
doi:10.1124/jpet.110.171173
- CYP
- cytochrome P450
- hCYP1A1
- human CYP1A1
- PAH
- polycyclic aromatic hydrocarbon
- WT
- wild type
- TCDD
- 2,3,7,8-tetrachlorodibenzo-p-dioxin
- AHR
- Ah receptor
- EROD
- ethoxyresorufin O-demethylase
- MROD
- methoxyresorufin O-demethylase
- ANOVA
- analyses of variance
- AHRE
- Ah response element
- ARNT
- Ah receptor nuclear translocator
- LC-MS/MS
- liquid chromatography-mass spectrometry/mass spectrometry
- MC
- 3-methylcholanthrene
- 1-OH-MC
- 1-hydroxy-3-MC
- 2-OH-MC
- 2-hydroxy-3-MC
- MC-1-one
- 1-one-3-MC
- MC-11
- 12-epoxide, 11,12-epoxy-11,12-dihydro-3-MC
- MC-11
- 12-dialdehyde, 11,12-dialdehyde-3-MC
- 11-OH-MC
- 11-hydroxy-3-MC
- MC-11.12-diol
- 11,12-dihydro-11,12-dihydroxy-3-MC
- MC-11
- 12-didydroepoxide, 11,12-epoxy-11,12-dihydro-3-MC
- MC-11
- 12-dione, 11,12-dione-3-MC
- RT-PCR
- reverse transcriptase-polymerase chain reaction
- EMSA
- electrophoretic mobility-shift assay
- PCB-153
- 2,2′,4,4′,5,5′-hexachlorobiphenyl
- CO
- corn oil
- PAGE
- polyacrylamide gel electrophoresis.
References
- Bhakta KY, Jiang W, Couroucli XI, Fazili IS, Muthiah K, Moorthy B. ( 2008) Regulation of cytochrome P4501A1 expression by hyperoxia in human lung cell lines: implications for hyperoxic lung injury. Toxicol Appl Pharmacol 233: 169– 178 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bookout AL, Cummins CL, Mangelsdorf DJ, Pesola JM, Kramer MF. ( 2006) High-throughput real-time quantitative reverse transcription PCR. Curr Protoc Mol Biol Chapter 15, Unit 15.18 [DOI] [PubMed] [Google Scholar]
- Bradford MM. ( 1976) A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem 72: 248– 254 [DOI] [PubMed] [Google Scholar]
- Cinti DL, Moldeus P, Schenkman JB. ( 1972) Kinetic parameters of drug-metabolizing enzymes in Ca2+-sedimented microsomes from rat liver. Biochem Pharmacol 21: 3249– 3256 [DOI] [PubMed] [Google Scholar]
- Conney AH. ( 1986) Induction of microsomal cytochrome P-450 enzymes: the first Bernard B. Brodie lecture at Pennsylvania State University. Life Sci 39: 2493– 2518 [DOI] [PubMed] [Google Scholar]
- DeVito MJ, Ross DG, Dupuy AE, Jr, Ferrario J, McDaniel D, Birnbaum LS. ( 1998) Dose-response relationships for disposition and hepatic sequestration of polyhalogenated dibenzo-p-dioxins, dibenzofurans, and biphenyls following subchronic treatment in mice. Toxicol Sci 46: 223– 234 [DOI] [PubMed] [Google Scholar]
- Diliberto JJ, Burgin DE, Birnbaum LS. ( 1999) Effects of CYP1A2 on disposition of 2,3,7,8-tetrachlorodibenzo-p-dioxin, 2,3,4,7,8-pentachlorodibenzofuran, and 2,2′,4,4′,5,5′-hexachlorobiphenyl in CYP1A2 knockout and parental (C57BL/6N and 129/Sv) strains of mice. Toxicol Appl Pharmacol 159: 52– 64 [DOI] [PubMed] [Google Scholar]
- Fazili IS, Jiang W, Wang L, Felix EA, Khatlani T, Coumoul X, Barouki R, Moorthy B. ( 2010) Persistent induction of cytochrome P4501A1 in human hepatoma cells by 3-methylcholanthrene: evidence for sustained transcriptional activation of the CYP1A1 promoter. J Pharmacol Exp Ther 333: 99– 109 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fujii-Kuriyama Y, Mimura J. ( 2005) Molecular mechanisms of AhR functions in the regulation of cytochrome P450 genes. Biochem Biophys Res Commun 338: 311– 317 [DOI] [PubMed] [Google Scholar]
- Gangarosa MA, Stoming TA. ( 1983) The metabolism of 3-methylcholanthrene by liver and lung microsomes: effect of enzyme inducing agents. Cancer Lett 20: 323– 331 [DOI] [PubMed] [Google Scholar]
- Guengerich FP. ( 1988) Roles of cytochrome P-450 enzymes in chemical carcinogenesis and cancer chemotherapy. Cancer Res 48: 2946– 2954 [PubMed] [Google Scholar]
- Harvey RG. ( 1982) Polycyclic hydrocarbons and cancer. Am Sci 70: 386– 393 [PubMed] [Google Scholar]
- Hemminki K. ( 1993) DNA adducts, mutations and cancer. Carcinogenesis 14: 2007– 2012 [DOI] [PubMed] [Google Scholar]
- Huggins C, Grand LC, Brillantes FP. ( 1961) Mammary cancer induced by a single feeding of polymucular hydrocarbons, and its suppression. Nature 189: 204– 207 [DOI] [PubMed] [Google Scholar]
- Jiang W, Wang L, Zhang W, Coffee R, Fazili IS, Moorthy B. ( 2009) Persistent induction of cytochrome P450 (CYP)1A enzymes by 3-methylcholanthrene in vivo in mice is mediated by sustained transcriptional activation of the corresponding promoters. Biochem Biophys Res Commun 390: 1419– 1424 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jiang W, Welty SE, Couroucli XI, Barrios R, Kondraganti SR, Muthiah K, Yu L, Avery SE, Moorthy B. ( 2004) Disruption of the Ah receptor gene alters the susceptibility of mice to oxygen-mediated regulation of pulmonary and hepatic cytochromes P4501A expression and exacerbates hyperoxic lung injury. J Pharmacol Exp Ther 310: 512– 519 [DOI] [PubMed] [Google Scholar]
- Kawajiri K, Natachi K, Imai K, Hayashi S, Watanabe J. ( 1991) The individual difference of susceptibility to lung cancer in relation to polymorphisms of P4501A1 gene and cigarette dose, in Xenobiotics and Cancer ( Ernester L, Gelboin HV, Sugimura T. eds) pp 55– 61, Japan Society Press, Tokyo, Japan [Google Scholar]
- Kinoshita K, Hashimoto K, Takahashi G, Yasuhira K. ( 1980) Gas chromatography-mass spectrometric analysis of 3-methyl-cholanthrene metabolism in vivo. Gann 71: 181– 189 [PubMed] [Google Scholar]
- Kondraganti SR, Jiang W, Jaiswal AK, Moorthy B. ( 2008) Persistent induction of hepatic and pulmonary phase II enzymes by 3-methylcholanthrene in rats. Toxicol Sci 102: 337– 344 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kondraganti SR, Jiang W, Moorthy B. ( 2002) Differential regulation of expression of hepatic and pulmonary cytochrome P4501A enzymes by 3-methylcholanthrene in mice lacking the CYP1A2 gene. J Pharmacol Exp Ther 303: 945– 951 [DOI] [PubMed] [Google Scholar]
- Liang HC, McKinnon RA, Nebert DW. ( 1997) Sensitivity of CYP1A1 mRNA inducibility by dioxin is the same in Cyp1a2(+/+) wild-type and Cyp1a2(−/−) null mutant mice. Biochem Pharmacol 54: 1127– 1131 [DOI] [PubMed] [Google Scholar]
- Liu WB, Liu JY, Ao L, Zhou ZY, Zhou YH, Cui ZH, Cao J. ( 2010) Epigenetic silencing of cell cycle regulatory genes during 3-methylcholanthrene and diethylnitrosamine-induced multistep rat lung cancer. Mol Carcinog 49: 556– 565 [DOI] [PubMed] [Google Scholar]
- Matsubara T, Prough RA, Burke MD, Estabrook RW. ( 1974) The preparation of microsomal fractions of rodent respiratory tract and their characterization. Cancer Res 34: 2196– 2203 [PubMed] [Google Scholar]
- Moorthy B. ( 2000) Persistent expression of 3-methylcholanthrene-inducible cytochromes P4501A in rat hepatic and extrahepatic tissues. J Pharmacol Exp Ther 294: 313– 322 [PubMed] [Google Scholar]
- Moorthy B. ( 2002) 3-Methylcholanthrene-inducible hepatic DNA adducts: a mechanistic hypothesis linking sequence-specific DNA adducts to sustained cytochrome P4501A1 induction by 3-methylcholanthrene. Redox Rep 7: 9– 13 [DOI] [PubMed] [Google Scholar]
- Moorthy B. ( 2008) The CYP1A subfamily, in Cytochromes P450. Role in Drug Metabolism and Toxicity of Drugs and Other Xenobiotics ( Ioannides C. ed) pp 97– 135, Royal Society of Chemistry, Cambridge, UK [Google Scholar]
- Moorthy B, Chen S, Li D, Randerath K. ( 1993) 3-Methylcholanthrene-inducible liver cytochrome(s) P450 in female Sprague-Dawley rats: possible link between P450 turnover and formation of DNA adducts and I-compounds. Carcinogenesis 14: 879– 886 [DOI] [PubMed] [Google Scholar]
- Moorthy B, Muthiah K, Fazili IS, Kondraganti SR, Wang L, Couroucli XI, Jiang W. ( 2007) 3-Methylcholanthrene elicits DNA adduct formation in the CYP1A1 promoter region and attenuates reporter gene expression in rat H4IIE cells. Biochem Biophys Res Commun 354: 1071– 1077 [DOI] [PubMed] [Google Scholar]
- Nebert DW, Dalton TP, Okey AB, Gonzalez FJ. ( 2004) Role of aryl hydrocarbon receptor-mediated induction of the CYP1 enzymes in environmental toxicity and cancer. J Biol Chem 279: 23847– 23850 [DOI] [PubMed] [Google Scholar]
- Nukaya M, Moran S, Bradfield CA. ( 2009) The role of the dioxin-responsive element cluster between the Cyp1a1 and Cyp1a2 loci in aryl hydrocarbon receptor biology. Proc Natl Acad Sci USA 106: 4923– 4928 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pendurthi UR, Okino ST, Tukey RH. ( 1993) Accumulation of the nuclear dioxin (Ah) receptor and transcriptional activation of the mouse Cyp1a-1 and Cyp1a-2 genes. Arch Biochem Biophys 306: 65– 69 [DOI] [PubMed] [Google Scholar]
- Poland A, Glover E. ( 1974) Comparison of 2,3,7,8-tetrachlorodibenzo-p-dioxin, a potent inducer of aryl hydrocarbon hydroxylase, with 3-methylcholanthrene. Mol Pharmacol 10: 349– 359 [PubMed] [Google Scholar]
- Postlind H, Vu TP, Tukey RH, Quattrochi LC. ( 1993) Response of human CYP1-luciferase plasmids to 2,3,7,8-tetrachlorodibenzo-p-dioxin and polycyclic aromatic hydrocarbons. Toxicol Appl Pharmacol 118: 255– 262 [DOI] [PubMed] [Google Scholar]
- Rasmussen RE, Anderson J, Kinkead ER, MacEwen JD, Bruner RH. ( 1984) Carcinogenesis, benzo[a]pyrene metabolism, and sister chromatid exchanges in lungs of rats after intratracheal 3-methylcholanthrene. J Natl Cancer Inst 73: 257– 264 [PubMed] [Google Scholar]
- Schnekenburger M, Peng L, Puga A. ( 2007) HDAC1 bound to the Cyp1a1 promoter blocks histone acetylation associated with Ah receptor-mediated trans-activation. Biochim Biophys Acta 1769: 569– 578 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shi Z, Dragin N, Miller ML, Stringer KF, Johansson E, Chen J, Uno S, Gonzalez FJ, Rubio CA, Nebert DW. ( 2010) Oral benzo[a]pyrene-induced cancer: two distinct types in different target organs depend on the mouse Cyp1 genotype. Int J Cancer doi: 10.1002/ijc.25222 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shou M, Yang SK. ( 1990) 1-Hydroxy- and 2-hydroxy-3-methylcholanthrene: regioselective and stereoselective formations in the metabolism of 3-methylcholanthrene and enantioselective disposition in rat liver microsomes. Carcinogenesis 11: 933– 940 [DOI] [PubMed] [Google Scholar]
- Sinclair PR, Gorman N, Dalton T, Walton HS, Bement WJ, Sinclair JF, Smith AG, Nebert DW. ( 1998) Uroporphyria produced in mice by iron and 5-aminolevulinic acid does not occur in Cyp1a2 (−/−) null mutant mice. Biochem J 330: 149– 153 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang W, Moorthy B, Chen M, Muthiah K, Coffee R, Purchio AF, West DB. ( 2004) A Cyp1a2-luciferase transgenic CD-1 mouse model: responses to aryl hydrocarbons similar to the humanized AhR mice. Toxicol Sci 82: 297– 307 [DOI] [PubMed] [Google Scholar]