Abstract
In contrast to the numerous reports on the pharmacological effects of Δ9-tetrahydrocannabinol (THC), the pharmacological activity of another substituent of Cannabis sativa, cannabichromene (CBC) remains comparatively unknown. In the present study, we investigated whether CBC elicits cannabinoid activity in the tetrad assay, which consists of the following four endpoints: hypomotility, antinociception, catalepsy, and hypothermia. Because cannabinoids are well documented to possess anti-inflammatory properties, we examined CBC, THC, and combination of both phytocannabinoids in the lipopolysaccharide (LPS) paw edema assay. CBC elicited activity in the tetrad that was not blocked by the CB1 receptor antagonist, rimonabant. Moreover, a behaviorally inactive dose of THC augmented the effects of CBC in the tetrad that was associated with an increase in THC brain concentrations. Both CBC and THC elicited dose-dependent anti-inflammatory effects in the LPS-induced paw edema model. The CB2 receptor, SR144528 blocked the anti-edematous actions of THC, but not those produced by CBC. Isobolographic analysis revealed that the anti-edematous effects of these cannabinoids in combination were additive. Although CBC produced pharmacological effects, unlike THC, its underlying mechanism of action did not involve CB1 or CB2 receptors. In addition, there was evidence of a possible pharmacokinetic component in which CBC dose-dependently increased THC brain levels following an i.v. injection of 0.3 mg/kg THC. In conclusion, CBC produced a subset of behavioral activity in the tetrad assay and reduced LPS-induced paw edema through a noncannabinoid receptor mechanism of action. These effects were augmented when CBC and THC were co-administered.
Keywords: Cannabis sativa, marijuana, phytocannabinoid, cannabichromene, delta-9-tetrahydrocannabinol (THC), CB1 cannabinoid receptor, CB2 cannabinoid receptor, anti-inflammatory
Introduction
While Δ9-tetrahydrocannabinol (THC) is recognized as the primary psychoactive constituent in Cannabis sativa (Gaoni et al., 1964), the issue of whether other constituents in this plant contribute to its pharmacological effects, have pharmacological effects of their own, or modulate the effects of THC remains of interest from a drug abuse perspective as well as for the development of cannabinoid-based medications. One particular class of constituents in marijuana, the phytocannabinoids (of which THC is the prototypical type), has received considerable attention for their contribution to the pharmacological effects of marijuana. Of the approximately 70 identified phytocannabinoids in marijuana, many are present in trace amounts, and some of these compounds can be quantitatively abundant in the plant (McPartland et al., 2001; ElSohly et al., 2005). For example, the respective percentages of THC and cannabidiol (CBD), a non-psychoactive phytocannabinoid, from confiscated cannabis preparations are 8.8% and 0.4% (Mehmedic, et al., 2010). CBD is documented to possess anxiolytic-like, antinociceptive, anti-psychotic-like, anti-inflammatory, and anti-oxidant effects (McPartland et al,. 2001; Mechoulam et al., 2007).
The pharmacological actions of another phytocannabinoid, cannabichromene (CBC) (Mechoulam et al., 1968), present at relatively high concentrations in certain strains of marijuana remain to be extensively investigated. Turner (1980) and Holley (1975) noted that “drug type” marijuana primarily contained higher concentrations of CBC than CBD, while “fiber type” marijuana contained higher concentrations of CBD than CBC. This classification of “drug type” or “fiber type” marijuana is related to some strains of marijuana that have more potent psychotropic effects compared to other strains that cultivated as hemp for their fiber. While CBC is the second most abundant cannabinoid in some strains of marijuana growing in the United States (Brown et al., 1990), it represents 0.3% of the constituents from confiscated cannabis preparations (Mehmedic, et al., 2010).
Despite the presence of CBC in certain marijuana strains, relatively few studies have investigated the pharmacological effects of this compound. CBC was shown to have analgesic properties and potentiated the analgesic effects of THC in the mouse tail-flick assay (Davis et al., 1983). It also was found to prolong hexabarbital-induced hypnosis (Hatoum et al., 1981). In addition, it potentiated bradycardia caused by THC, though it did not induce bradycardia on its own (O'Neil et al., 1979). CBC was reported to induce sedation and ataxia in canines (Gaoni et al., 1966); however, subsequent studies failed to replicate these effects (Gaoni et al., 1971). Of relevance to the present study, CBC and its analogs have been reported to have anti-inflammatory properties in vivo and in vitro (Turner et al., 1981; Wirth et al., 1980a; Wirth et al., 1980b).
THC, other naturally occurring psychoactive cannabinoids, and synthetic cannabinoids produce their pharmacological effects predominantly through CB1 and CB2 receptors, with the former receptor largely responsible for behavioral and metabolic actions of these drugs, and latter receptor generally associated with the modulation of immune responses. This class of drugs produces a myriad of effects, including locomotor suppression, catalepsy, antinociception, and hypothermia, which collectively are known as the tetrad assay and is used to screen cannabinoid activity (Fride, 2006; Martin et al., 1991). The pharmacological effects of cannabinoids in each of these four in vivo tests yield a high positive correlation with their affinity for the CB1 receptor (Compton et al., 1993). Importantly, CB1 receptor binding affinity also positively correlates with drug discrimination studies in rats and psychoactivity in humans. Accordingly, the tetrad assay has had great utility because of its high predictive value in identifying compounds that possess marijuana-like effects. However, there are currently no published reports that evaluated CBC in the tetrad.
Phytocannabinoids, such as THC, have been effective anti-inflammatory agents in a variety of inflammatory models and there is substantial in vivo and in vitro evidence that both cannabinoid receptors (CB1 and CB2) are involved in anti-inflammatory processes (Zurier, 2003). Although both cannabinoid receptors are found on various populations of immune cells, CB2 receptors are far more abundant than CB1 receptors (Croxford et al., 2005). Many studies have shown that stimulation of CB2 receptors inhibits pro-inflammatory cytokine release triggered by inflammatory agents, such as lipopolysaccharide (LPS), a constituent of the outer membrane of the cell wall of most gram negative bacteria (Puffenbargar et al., 2000). Intraplantar administration of LPS elicits paw edema that has been utilized to investigate the anti-inflammatory effects of various classes of anti-inflammatory compounds including steroidal and non-steroidal anti-inflammatory drugs (Kanaan et al., 1997), as well as cannabinoid receptor agonists (Naidu et al., under review). However, there are presently no published reports on the possible anti-inflammatory effects of THC or CBC in this LPS-induced inflammatory paw model.
There were five objectives of the present study. The first goal was to determine whether CBC produces cannabinoid activity in the tetrad (locomotor suppression, catalepsy, antinociception, and hypothermia). Second, we evaluated whether this phytocannabinoid would elicit anti-edematous effects in the LPS-induced paw edema model. The third aim was to determine whether CBC produces its pharmacological effects in the aforementioned assays through CB1 or CB2 receptor mechanisms of action, using the respective receptor antagonists, rimonabant and SR144528. The fourth goal was to investigate the ability of CBC to modulate the pharmacological effects of THC in the tetrad and the LPS-induced paw edema model. The final goal was to examine whether co-administration of THC and CBC alters blood and brain levels of each phytocannabinoid.
Materials and Methods
2.1. Animals
Studies utilized male ICR mice (Harlan Laboratories, Dublin, VA) weighing 20 – 30 g that were housed six to a cage in the animal care facility maintained at 22 ± 2°C on a 12 h light/dark cycle. Food and water were available ad libitum and mice were given 24 h to acclimate to the test environment (22 ± 2°C) before analysis; animals were housed in the test environment until the termination of experimental procedures for LPS-induced inflammation studies. All animal studies were approved by the Institutional Animal Care and Use Committee of Virginia Commonwealth University in accordance with the National Institute of Health Guide for the Care and Use of Laboratory Animals.
2.2. Drugs
CBC was synthesized from Organix Inc. (Woburn, MA), and THC, rimonabant, and SR144528 were supplied by the National Institute on Drug Abuse (Bethesda, MD). All compounds were dissolved in a vehicle consisting of absolute ethanol, alkamuls-620 (Rhone-Poulenc, Princeton, NJ), and saline in a ratio of 1:1:18. For tetrad experiments, all injections were administered intravenously in a volume of 0.1 ml per 10 g of body weight. For LPS-induced inflammation studies, drugs were administered intraperitoneally in a volume of 0.1 ml per 10 g of body weight. LPS from Escherichia coli 026:B6 (Sigma-Alrich, St. Louis) was suspended in 0.9% saline for paw administration.
2.3. Tetrad procedure
Pretreatment baseline tail-flick response to radiant heat (D'Amour et al., 1941) and rectal temperature were recorded. The intensity of the radiant heat stimulus was held constant to yield control latencies of 2 – 4 seconds with an automated cut-off of 10 s to prevent tissue damage to the tail. Rectal temperatures were taken with a digital thermometer (Fisher Scientific, Pittsburgh, PA) at a depth of 2 cm and recorded to the nearest 0.1°C. Locomotor suppression, antinociception, hypothermia, and catalepsy were evaluated (in that respective order) after the mice received drug treatment. Five min post-drug administration, mice were placed in individual activity chambers (plastic mouse cages in a dark cabinet) surrounded by photocell beams (Digiscan Animal Activity Monitor; Omnitech Electronics, Inc., Columbus, OH). Locomotor suppression was measured for 10 min and the data were expressed as the total number of photocell beam breaks. Tail-flick latency was measured at 20 min after drug administration. Antinociception was calculated as a percent of maximum possible effect (%MPE) by the following formula: %MPE = [(test latency-pretreatment latency)/(10-pretreatment latency)] × 100}. At 30 min, rectal temperature was measured and data were expressed as the difference between pretreatment rectal temperature and post-injection rectal temperature. At 40 min, mice were evaluated for catalepsy in a 5 min ring immobility test (Gill et al., 1970; Pertwee, 1972) in which each subject was placed on a metal ring (5.5 cm diameter) that was suspended 16 cm from the benchtop with a black background board attached (for contrast with the albino mice). The total duration of time spent motionless (no voluntary movement) was timed using a stopwatch.
2.4. Extraction Procedure, GC/MS Analysis
Mice were decapitated at two different time points (discussed in results section) and the blood was collected in heparinized tubes (Fisher Scientific, Pennsylvania). Whole brains were harvested and stored on ice until homogenized. Brain tissue was homogenized less than two h following the harvest in a 2:1 ratio (vol/wt) of saline to brain tissue. Aliquots (0.5 ml) were prepared from heparinized mouse whole blood or homogenized brain tissue. Calibrators were prepared by adding drug standards to naïve mouse blood and brain tissue. Fifty µl of internal standard (THC-d3; Cerilliant, Round Rock, Texas) was added to all calibrators and samples (minus blank samples). All tubes were equilibrated overnight at 5°C.
“Ice cold” acetonitrile (2 ml) was added dropwise while vortexing and the mixture was centrifuged at 3,000 rpm for 10 minutes. The mixture was stored overnight at −30°C to assist in further separation of the layers. The top layer (acetonitrile layer) was isolated and 2 ml of 0.2 N NaOH was added while vortexing the tubes. Four ml of organic solvent (9:1, hexane:ethyl acetate) was added and the tubes were rotated at 30 rpm for 30 min. The tubes were then centrifuged at 3,000 rpm for 10 min. The organic layer was isolated and evaporated to dryness in a Savant AES1000. Fifty µl of derivatizing agent (RC3; Regis Technologies, Inc., Morton Grove, IL) was added to each tube and samples were heated at 70°C for 20 min. Samples were then transferred to microvials for GC/MS analysis.
Microvials were loaded on a Shimadzu QP2010 GC/MS utilizing a splitless injection port and DB-5 column (30 m × 0.25 mm I.D. × 25 µm film thickness). The GC/MS was operated under the following temperatures: initial, 190°C; ramp, 30°C/min to 330°C then hold 1 min; injection port, 250°C; transfer line, 280°C; and ion source, 260°C. Ions monitored in SIM mode: 315, 343, 386 m/z for THC; 303, 246, 371 m/z for CBC; and 346, 389 m/z for THC-d3. Calibration curves were constructed with at least six calibrators to extend the linearity range for appropriate experiments.
2.5. Inflammation Induction and Measurement Procedure
Baseline body weights, as well as paw thicknesses of both hind paws were measured two h before LPS injection and again 24 h later. Digital calipers (Fisher Scientific, Inc., Pittsburgh, PA) were utilized to measure paw thickness to the nearest 0.01 mm. In accordance with a previously described method (Naidu et al., under review), mice received a subcutaneous injection (utilizing a 30 gauge needle) of LPS (25 µg in 50 µl of saline) into the plantar region of a hind paw. As a control, the contralateral hind paw received a 50 µl injection of saline in the same manner as previously stated. Paw thickness data are depicted as the change in paw thickness from baseline for the LPS-injected (ipsilateral) or saline-injected (contralateral) hind paws.
Mice received intraperitoneal injections of test drugs 1 h before, as well as 6 and 23 h after LPS injection. In experiments evaluating cannabinoid receptor mechanisms of action, rimonabant, SR144528, or vehicle was administered 30 min prior to each injection of vehicle, THC, or CBC at the three respective drug administration time points. In the drug interaction study, CBC and THC doses were combined and administered as a single injection at the three respective time points.
2.6. Statistical Analyses
Behavioral data and concentrations of cannabinoids in blood and brain matrices were statistically analyzed using ANOVA in Statview 5.0 (SAS Institute Inc., Cary, N.C.) or Prism 5 (Graphpad Software, Inc., San Diego, CA). When ANOVA yielded significant results, Dunnett’s test (comparison of all treatment groups to a vehicle group in dose-response studies) or Tukey’s test (comparison of all groups) was used for post hoc analysis. Bonferonni’s t-test was used for planned comparisons. A p value of less than 0.05 was denoted as significant for all analyses. Linear regression analysis of calibration curves and cannabinoid concentrations in blood and brain tissue were determined in Prism 5. For all experiments, unless otherwise indicated, n=6 mice per group.
Isobologram analysis (Tallarida et al., 1989; Tallarida, 2001; Tallarida, 2006) was used to determine the nature of the interaction between co-administration of THC and CBC in the above assays. Experimental ED50 values from combination doses that are along the line of additivity confer an additive relationship, below the line a synergistic relationship, and above the line an antagonistic relationship. A theoretical additive ED50 value of both drugs in combination (calculated from the ED50 value of each drug’s separate dose-response curve) was compared to the experimental ED50 value of both drugs in combination. A 1:1 ratio of three combination drug doses (calculated from each drug’s ED50 value and two descending log doses) was utilized in the experiment to determine the experimental ED50. The experimental ED50 (with confidence intervals) was then graphically compared to the theoretical additive ED50 value (with confidence intervals) to determine if these values overlapped (indicative of an additive relationship between the two drugs). Prism 5 and PharmTools Pro 1.1 (PharmSoft, Wynnewood, PA) were utilized for isobologram analysis.
3. Results
3.1. CBC elicits pharmacological effects through a non-CB1 receptor mechanism of action in the tetrad assay
The effects of intravenously administered CBC (3, 10, 30, or 100 mg/kg) in the tetrad assay are shown in Figure 1. CBC caused significant locomotor suppression (Panel A; F[4,25]=7.9, p<0.001), catalepsy (Panel B; F[4,25]=6.7, p<0.001), antinociception (Panel C; F[4,25]=3.6, p<0.05), and hypothermia (Panel D; F[4,25]=14.0, p<0.001). However, only the largest dose of CBC tested (i.e., 100 mg/kg) produced significant effects in each of these measures (Dunnett’s post hoc, p<0.05). The effects of the high dose of CBC (100 mg/kg) in the tetrad assay were then compared to those produced by THC. Significant effects of treatment for locomotor suppression (Supplementary Figure 1A1; F[5,30]=14.1, p<0.001), catalepsy (Figure 1B1; F[5,30]=36.1, p<0.001), antinociception (Figure 1C1; F[5,30]=38.0, p<0.001), and hypothermia (Figure 1D1; F[5,30]=59.2, p<0.001) were found. Dunnett’s post hoc analysis (p<0.05) revealed that THC (3 and 10 mg/kg) and CBC (100 mg/kg) produced significant locomotor suppression, catalepsy, and hypothermia compared to vehicle control mice. However, in this experiment, CBC did not elevate tail-flick latencies, though THC (1, 3, and 10 mg/kg) produced significant antinociception.
Figure 1.
Comparison between the dose-response of intravenously administered CBC alone and in combination with an inactive dose of THC (i.e., 0.3 mg/kg) in the mouse tetrad. When given alone, only 100 mg/kg CBC produced locomotor suppression (A), catalepsy (B), antinociception (C), and hypothermia (D) in mice. The inactive dose of THC (0.3 mg/kg) produced a leftward shift in the dose-response curve of CBC when compared to the dose-response of CBC administered alone for catalepsy (B), antinociception (C), and hypothermia (D), but not for hypomotility (A). The results are presented as mean ± S.E.M., n=6 mice per group. * p < 0.05 for CBC alone vs. vehicle control mice. # p < 0.05 for CBC + THC (0.3 mg/kg) vs. vehicle + vehicle control mice.
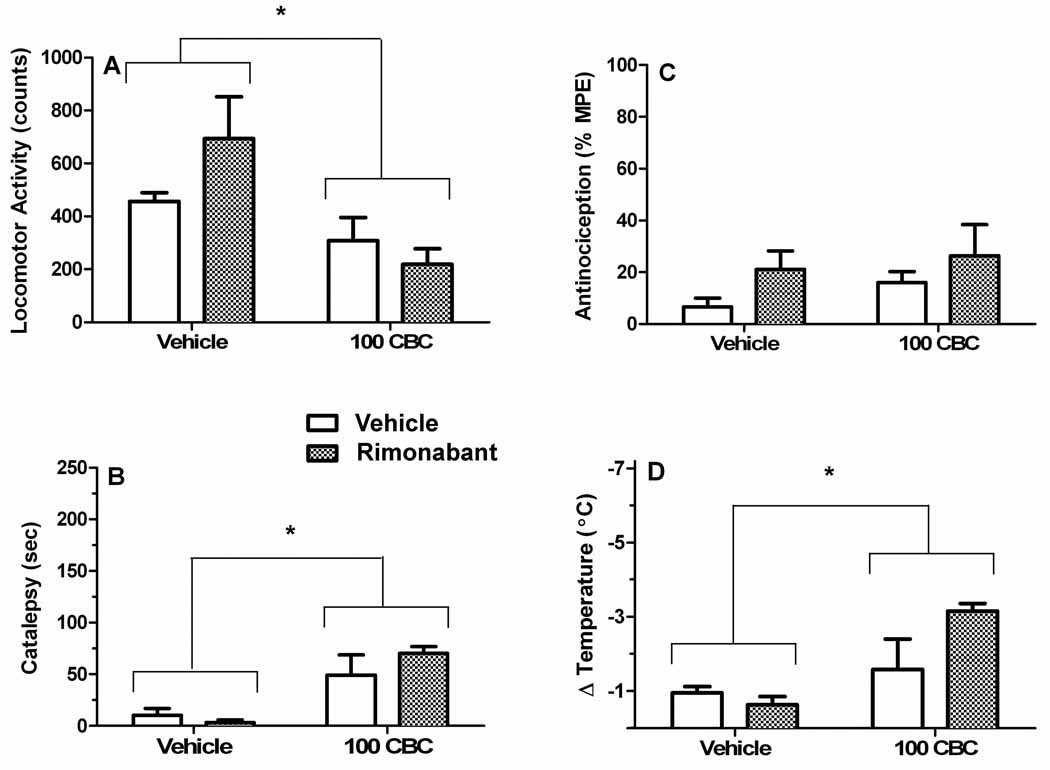
In the next experiment, we investigated whether CB1 receptors mediate the effects of CBC in the tetrad assay. Mice were given an injection of vehicle or rimonabant (3 mg/kg) followed 10 min later by a second injection of vehicle or CBC (100 mg/kg). This dose of the CB1 receptor antagonist is sufficient to block the pharmacological effects of THC and other cannabinoid receptor agonists in the tetrad assay (Compton et al., 1996). As can be seen in Figure 2, CBC produced significant hypomotility (Panel A; F[1,19]=9.7, p<0.01), catalepsy (Panel B; F[1,19]=21.5, p<0.001), and hypothermia (Panel D;1 2F[1,19]=11.5, p<0.01), but not antinociception (Panel C; p=0.09). In contrast, there were no significant main effects of rimonabant or interactions between CBC and rimonabant for any of the four dependent 3 measures, indicating that CBC produces these pharmacological effects through a non CB1 receptor mechanism of action.
Figure 2.
The pharmacological effects of CBC for locomotor suppression (A), catalepsy (B), antinociception (C), and hypothermia (D) are not mediated by the CB1 receptor. Mice were intravenously administered an i.p. injection of vehicle or rimonabant (3 mg/kg) followed 10 min later by an i.v. injection of vehicle or CBC (100 mg/kg). ANOVA revealed a significant main effect of CBC treatment (represented as asterisks) for locomotor suppression (A), catalepsy (B), and hypothermia (D), but not for antinociception (C). Rimonabant (3 mg/kg) failed to block the pharmacological effects of CBC. The results are presented as mean ± S.E.M., n=6 mice per group.
3.2. THC enhances the pharmacological effects of CBC
In the next experiment, we sought to determine whether a threshold dose of THC could augment the pharmacological effects of CBC in the tetrad assay. Based on the THC dose-response curves (Supplementary Figure 1), 0.3 mg/kg THC was selected as the threshold dose. Subjects received an i.v. injection of vehicle or CBC (3, 10, 30, or 100 mg/kg) 10 min before an i.v. injection of 0.3 mg/kg THC. As can be seen in Figure 1, 0.3 mg/kg THC produced a leftward shift in the CBC dose-response curves of CBC for catalepsy, antinociception, and hypothermia, but not hypomotility. Significant effects were found for locomotor activity (Figure 1A; F[5,30]=3.5, p<0.05), catalepsy (Figure 1B; F[5,30]=34.4, p<0.0001), antinociception (Figure 1C; F[5,30]=8.2, p<0.0001), and hypothermia (Figure 1D; F[5,30]=13.8, p<0.0001).
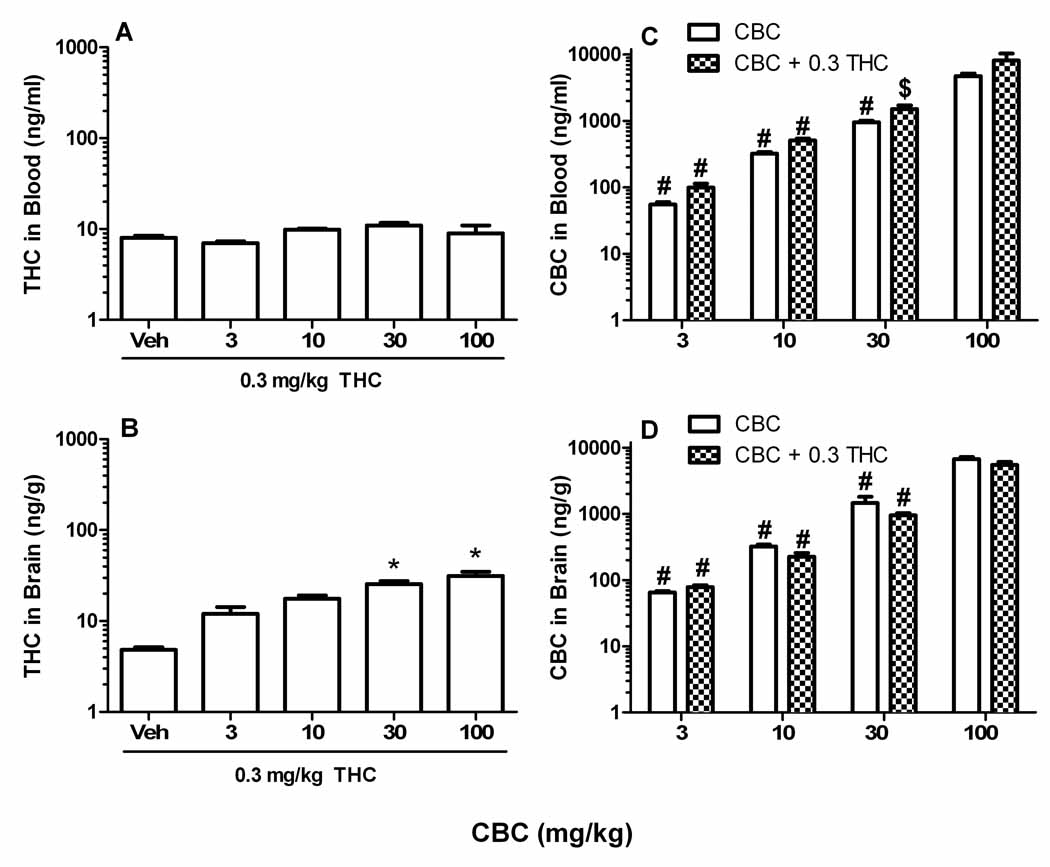
Because a threshold dose of THC significantly enhanced most of the pharmacological effects of CBC, the concentrations of both phytocannabinoids in blood and brain tissue were quantified to determine whether the drugs given in combination may have altered the pharmacokinetics of each other. Mice received an injection of CBC (vehicle, 3, 10, 30, or 100 mg/kg) followed 10 min later with a second injection of THC (0.3 mg/kg) or no injection. Tissues were harvested 30 min after CBC. Although CBC did not significantly affect THC blood levels (Figure 3A; p=0.07), it led to a dose-related increase in THC brain levels (Figure 3B; F[4,24]=9.9, p<0.001). Not surprisingly, increasing doses of CBC led to significant increases of this compound in blood (F[7,39]=12.6, p<0.001; Figure 3C) and brain (F[7,40]=67.0, p<0.0001; Figure 3D), but there were no effects of THC on CBC blood and brain levels. These results indicate that CBC dose-dependently elevated THC brain levels, but the threshold dose of THC did not affect CBC brain or blood levels.
Figure 3.
Quantification of blood and brain tissue concentrations of THC and CBC in mice administered a dose range of CBC with and without an inactive dose of THC. Mice were given an i.v. injection of CBC (vehicle, 3, 10, 30, or 100 mg/kg) followed by a second i.v. injection 10 min later of vehicle or THC (0.3 mg/kg). Blood and brain tissue were harvested at 30 min after the first injection. A. THC blood levels. B. THC Brain levels. C. CBC blood levels with and without THC (0.3 mg/kg). D. CBC brain levels with and without THC (0.3 mg/kg). The results are presented as mean ± S.E.M., n=5 or 6 mice per group. *p < 0.05 vs. vehicle + 0.3 mg/kg THC; #p < 0.0001 vs. 100 mg/kg CBC (with or without 0.3 mg/kg THC; $p < 0.0001 vs. 100 mg/kg CBC.
3.3. Evaluation of CBC and THC on LPS-induced paw edema
Both CBC (F[4,25]=8.3, p<0.001) and THC (F[4,25]=10.7, p<0.0001) produced dose-related decreases in LPS-induced paw edema (Figure 4A), but neither drug affected the paw thickness of the saline-injected paw (data not shown). Post hoc analyses indicated significant decreases in paw thickness of the LPS-injected paw following 10, 30, and 100 mg/kg CBC as well as 10 and 30 mg/kg THC compared to the respective vehicle control groups.
Figure 4.
Distinct receptor mechanisms of action mediate the anti-edematous effects of THC and CBC. A. Anti-edematous effects of i.p. administered CBC and THC. Neither drug affected paw thickness in the contralateral paw (data not shown). The results are presented as mean ± S.E.M., n=6 mice per group. Mean pre-injection paw thickness ± SEM values for LPS-injected and saline-injected paws for the mice in the CBC experiment were 1.82 mm ± 0.008 and 1.82 mm ± 0.008, respectively. Mean pre-injection paw thickness ± SEM values for LPS-injected and saline-injected paws for the mice in the THC experiment were 1.99 mm ± 0.013 and 1.99 mm ± 0.012, respectively. *p < 0.05 vs. vehicle control group (THC); #p < 0.05 vs. vehicle control group (CBC). B. The anti-inflammatory effects of THC were blocked by SR144528 (SR2), but not by rimonabant (SR), indicating a CB2 receptor mediated effect. C. The anti-inflammatory effects of CBC were not blocked by either rimonabant (SR1) or SR144528 (SR2), indicating that neither CB1 nor CB2 receptors played a necessary role in CBC’s actions. Collective baseline mean paw thickness values for mice depicted in the experiments in Panels B and C were 1.89 ± 0.008 mm and 1.85 ± 0.010 mm. The results are presented as mean ± S.E.M., n=6 mice per group. *p < 0.05 vs. vehicle/vehicle group; #p < 0.05 vs. vehicle/THC group.
In order to determine whether CB1 or CB2 receptors mediated the anti-edematous effects of CBC and THC, mice were administered vehicle, rimonabant (3 mg/kg), or SR144528 (3 mg/kg) 30 min before each of the three injections of vehicle, CBC (100 mg/kg), or THC (10 mg/kg). A significant effect of treatment was found for the THC experiment (Figure 4B; F[5,30]=18.6, p<0.0001). Neither rimonabant nor SR144528 given alone affected LPS paw edema. SR144528, but not rimonabant, significantly reduced the anti-edematous effects of THC, indicating a CB2 receptor mechanism of action.
As shown in Figure 4C, a significant effect of treatment was found in the CBC experiment (F[3,20]=7.3, p<0.01). However, neither rimonabant nor SR144528 ameliorated the anti-inflammatory effects of CBC, suggesting that the anti-edematous effects of this phytocannabinoid do not involve either CB1 or CB2 receptors.
3.4. Combination of CBC and THC on LPS-induced paw edema
In the final experiment, we examined whether equipotent doses of CBC and THC given in combination would to reduce LPS-induced paw edema in an additive or synergistic fashion. The dose-response relationship of the drugs given in 1:1 equipotent combination doses (4 mg/kg CBC:0.4 mg/kg THC, 12 mg/kg CBC:1.2 mg/kg THC, or 40 mg/kg CBC:4 mg/kg THC) are depicted in Figure 5. A significant effect of treatment (F[3,20]=5.4, p<0.0001) was found. Dunnett’s post hoc test revealed that the group treated with 40 mg/kg CBC:4 mg/kg THC had significantly less paw edema than the vehicle + vehicle control group (p<0.05). The ED50 values (expressed as ED50 mg/kg (95% confidence intervals)) of CBC and THC calculated from their individual dose-response curves were 38.7 (23.9 to 62.5) and 4.0 (2.7 to 5.9) mg/kg, respectively. These ED50 values were utilized to establish a line of additivity on the isobologram (Figure 5C). The experimental ED50 value (95% confidence intervals) of CBC and THC in a 1:1 combination was 20.8 (11.1 to 38.7) mg/kg. The CBC and THC coordinates of the experimental ED50 were calculated as 18.9 (10.1 to 35.2) and 1.9 (1.0 to 3.5) mg/kg, respectively. The theoretical ED50 (located on the line of additivity was calculated as 21.3 (15.6 to 29.1) mg/kg and its CBC and THC coordinates were 19.3 (14.2 to 26.4) and 2.0 (1.5 to 2.7) mg/kg, respectively. Because the experimental and theoretical additive ED50 values and confidences limits overlapped, the relationship between the two phytocannabinoids was additive.
Figure 5.
The anti-inflammatory dose-response of CBC and THC administered in combination yield an additive interaction. Dose response curves from CBC alone (A) and THC alone (B) from Figure 4A. In the 1:1 combination study, mice were administered vehicle, 4 mg/kg CBC:0.4 mg/kg THC, 12 mg/kg CBC:1.2 mg/kg THC or 40 mg/kg CBC:4 mg/kg THC. The results are presented as mean ± S.E.M., n=6 mice per group. The respective baseline mean ± S.E.M. paw thickness values for mice in the 1:1 combination study, CBC alone, and THC alone experiments were 1.88 ± 0.008, 1.82 ± 0.008, and 1.99 ± 0.013 mm. *p < 0.05 vs. vehicle control group in the single drug experiments; # p < 0.05 vs. vehicle control group combination drug experiment. C. Isobologram analysis of the anti-inflammatory effects of CBC and THC dosed in an equipotent combination. The ED50 values for CBC and THC were calculated from the dose-response curve of each individual drug. The experimental ED50 value overlapped with the theoretical additive ED50 value indicating an additive relationship between the two drugs.
4. Discussion
The degree to which constituents other than THC contribute to the overall pharmacological effects of marijuana, have pharmacological effects of their own, and interact with the pharmacological effects of THC remain important questions. Few of the phytocannabinoids present in marijuana have been extensively evaluated in established in vivo models indicative of cannabinoid activity, such as the mouse tetrad, which consists of four in vivo assays: locomotor activity, catalepsy, antinociception, and hypothermia (Fride, 2006). CBC is a phytocannabinoid found in most common strains of marijuana but can also occur in concentrations equal to that of THC (Brown et al., 1990; Holley et al., 1975; Turner et al., 1980). The overall goals of the present study were to investigate if CBC produces cannabinoid activity in the tetrad, whether CBC possesses anti-inflammatory properties, and whether THC augments the effects of CBC in these assays. Accordingly, we investigated CBC, as well as its effects in combination with THC, in the mouse tetrad assay and in the LPS-induced paw edema model (Naidu et al., under review). Blood and brain tissue concentrations of the two cannabinoids were quantified to associate the concentrations to tetrad activity and to investigate potential pharmacokinetic interactions.
4.1. Evaluation of pharmacological effects of CBC in the tetrad assay
CBC produced significant pharmacological effects only when administered at a high dose (i.e., 100 mg/kg), which consistently produced locomotor suppression, catalepsy, and hypothermia, but only occasionally produced a small magnitude of antinociception. The observation that CBC sporadically produced antinociception may indicate that there is a different threshold dose needed to produce this effect compared to the other three indices. Other cannabinoids have been shown to have different thresholds or potencies within each measure that make up the tetrad assay. For example, anandamide, which is an endogenous cannabinoid, had similar ED50 values for hypomotility, catalepsy, and hypothermia, but was 3 – 4 fold less potent in producing antinociception than the other measures after i.v. administration in wild type mice (Smith et al., 1994). In contrast, THC dose-dependently produced significant effects for each of the four measures, with high efficacy. These findings are consistent with previously reported tetrad effects of THC (Fan et al., 1994; Martin et al., 1991).
Considering the great disparity between mean percentages of THC (i.e., 8.8%) and CBC (i.e., 0.3%) from confiscated cannabis preparations seized in 2008 (Mehmedic, et al., 2010) as well as our observation that CBC is substantially less potent than THC in the tetrad, it is unlikely that CBC by itself relevantly contributes to the psychoactive effects of marijuana. This conclusion is in agreement with a previous study that reported the tetrad effects of marijuana were primarily accounted for by its THC content and not by the other constituents of this plant (Varvel et al., 2005). Another study that utilized human subjects varied the concentration of CBC in marijuana cigarettes and found that the CBC concentration in their cigarettes (0.01 – 2.31%) did not affect behavioral (cognitive tasks) or neurophysical measures (EEG, ERP) in the study (Ilan et al., 2005).
The CB1 receptor antagonist, rimonabant failed to block the tetrad activity of CBC, indicating a non CB1 receptor mechanism of action. These results are consistent with the observation that CBC possesses poor affinity to the CB1 receptor (i.e., Ki value > 10,000 nM; Booker et al., 2009). In comparison, rimonabant has been reported to have a Ki value of 2 nM and has been shown to block the tetrad effects of THC and other cannabinoids (Compton et al., 1996; Rinaldi-Carmona et al., 1994; Wiley et al., 1998). Various noncannabinoid classes of drugs have also been shown to have partial tetrad effects including central nervous system depressants and anti-psychotics that were not blocked by rimonabant (Wiley et al., 2003). Because the tetrad effects of CBC were not blocked by a CB1 receptor antagonist, the mechanism by which CBC elicited tetrad effects remains to be established in future studies.
Another objective of the present study was to examine whether a threshold dose of THC would modify the pharmacological actions of CBC in the tetrad. An inactive dose of THC (i.e., 0.3 mg/kg) was selected based on a dose-response study of this compound in the tetrad assay. Administration of this dose of THC produced leftward shifts in the CBC dose-response relationship for catalepsy, antinociception, and hypothermia; however it did not enhance CBC-induced locomotor suppression. In order to test whether the enhanced pharmacological results occurred because of a pharmacokinetic interaction, we quantified blood and brain levels of CBC and THC given in combination. Although the behaviorally inactive dose of THC did not significantly alter CBC blood or brain concentrations, 30 or 100 mg/kg CBC led to significantly elevated THC levels in brain, but did not quite achieve statistical significance in affecting THC blood levels (p<0.07). These results suggest that THC had distributed out of the blood and into lipophilic matrices, such as brain, which is consistent with the reported pharmacokinetics of THC and its rapid distribution phase (Grotenhermen, 2003). About 90% of THC in blood is bound to plasma (10% to red blood cells) and 95 – 99% of plasma THC is bound to lipoproteins (Hunt et al., 1980; Widman et al., 1974). A high dose of CBC (30 or 100 mg/kg) may compete with THC (0.3 mg/kg) for the lipoprotein binding sites, which ultimately leads to the distribution of THC out of the blood and into lipophilic tissue such as the brain. Subsequently, the higher concentrations of THC in brain tissue might have interacted with CB1 receptors to elicit significant tetrad activity. It is noteworthy that these same doses of CBC produced enhanced pharmacological effects when given in combination with the threshold dose of THC. Thus, it is plausible that a pharmacokinetic interaction between the two cannabinoids contributed to the leftward shift in the CBC dose response in the presence of 0.3 mg/kg THC. Taken together, these data suggest that high doses of CBC may augment a threshold dose of THC to produce significant cannabinoid activity in the tetrad.
4.2. Evaluation of CBC in the LPS-induced paw edema model
Recent research has demonstrated that WIN55,212-2, a potent mixed CB1/CB2 receptor agonist, URB597, a FAAH inhibitor, and dexamethasone, a synthetic glucocorticoid hormone ameliorated LPS induced paw edema (Naidu et al., under review). CBC as well as THC dose-dependently decreased LPS-induced inflammation in the LPS-injected paw. There are no prior reports in the literature that examined the anti-inflammatory effects of either of these cannabinoids in this inflammation model. The anti-edematous effects of THC were blocked by SR144528, but not by rimonabant, indicating that a CB2 receptor mechanism of action. Other studies have also reported that the anti-inflammatory effects of THC are mediated though a CB2 receptor mechanism (Croxford et al., 2005; Guzman et al., 2002; Klein, 2005). In marked contrast, the anti-inflammatory effects of CBC were not blocked by either SR144528 or rimonabant, indicating that its anti-inflammatory effects are mediated through a noncannabinoid receptor mechanism of action. Other phytocannabinoids have also been reported to have anti-inflammatory effects that are cannabinoid receptor independent. For example, the well documented anti-inflammatory effects of CBD are not blocked by cannabinoid receptor antagonists (Costa et al., 2004; Costa et al., 2007), though capsazepine, a TRPV1 receptor antagonist, has been found to block the anti-inflammatory actions of CBD in the carrageenan model. Furthermore, phytocannabinoids that exist as carboxylic acids have been reported to have anti-inflammatory effects that are not blocked by cannabinoid receptor antagonists. For example, a primary metabolite of THC, THC-COOH has been reported to have anti-inflammatory properties that may be mediated through suppression of cyclooxygenase and lipooxygenase in vitro (Burstein et al., 1986; Doyle et al., 1990) or through the activation of peroxisome-proliferative-activated receptor-γ (Liu et al., 2003). Because CBC is structurally related to these other phytocannabinoids, there may be a similar structure activity relationship in which the anti-inflammatory effects of CBC are mediated through receptors or processes similar to those underlying CBD or THC-COOH.
Isobolographic analysis was used to investigate anti-edematous interactions between CBC and THC. Examining the dose-response relationship of both compounds in which equipotent doses of the compounds were co-administered revealed an additive relationship. Accordingly, a benefit of this additive relationship is the possibility that these drugs administered in combination might produce anti-inflammatory effects at lower doses than each drug alone. Of course, it will be important to establish whether the psychotropic effects of THC are minimized, while still retaining anti-inflammatory efficacy.
4.3. Conclusions
In summary, CBC produced a subset of effects in the mouse tetrad assay and significantly reduced LPS-induced paw edema. Moreover, both of these effects were enhanced when CBC was given in combination with THC. The tetrad effects of CBC were not CB1 receptor mediated and its anti-inflammatory effects were not CB1 or CB2 receptor mediated. In contrast, we determined that THC produced its anti-inflammatory effects in the LPS-induced paw edema model via CB2 receptor activation, a finding that had not been previously reported in this assay. Isobolographic analysis indicated an additive relationship between the anti-inflammatory effects of CBC and THC. A threshold dose of THC augmented the tetrad effects of CBC. However, the observation that high doses of CBC led to increased brain levels of THC suggests a potential pharmacokinetic interaction for the augmented tetrad effects of the two drugs given in combination.
In conclusion, CBC produced a subset of behavioral activity in the tetrad assay and reduced LPS-induced paw edema through a noncannabinoid receptor mechanism of action. Moreover, combination of CBC and THC leads to enhanced tetrad and anti-inflammatory actions.
Supplementary Material
Acknowledgements
Special thanks to Ramona Winckler for her help with intravenous injections and tetrad studies.
Role of Funding Source
This work was supported by the National Institute on Drug Abuse R01DA002396, R01DA03672, and R01 DA015683. NIDA had no further role in study design; in the collection, analysis and interpretation of the data; in the writing of the report; or in the decision to submit the paper for publication.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
A supplementary data figure is available with the online version of this article at doi:xxx/j.drugalcdep.xxx … Please see Appendix A.
See Appendix A.
Contributors
G.T. DeLong conducted the bulk of the studies, analysis of data, and writing of the manuscript. C.E. Wolf contributed to the laboratory procedures for analytical studies. A. Poklis contributed to the design of the analytical studies. A.H. Lichtman oversaw the study, along with contributing to the writing of the manuscript.
Conflict of Interest
None of the authors report a conflict of interest that could have influenced, or be perceived to influence, this work.
References
- Bisogno T, Hanus L, De Petrocellis L, Tchilibon S, Ponde DE, Brandi I, Moriello AS, Davis JB, Mechoulam R, Di Marzo V. Molecular targets for cannabidiol and its synthetic analogues: effect on vanilloid VR1 receptors and on the cellular uptake and enzymatic hydrolysis of anandamide. Br. J. Pharmacol. 2001;134:845–852. doi: 10.1038/sj.bjp.0704327. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Booker L, Naidu PS, Razdan RK, Mahadevan A, Lichtman AH. Evaluation of prevalent phytocannabinoids in the acetic acid model of visceral nociception. Drug Alcohol Depend. 2009;105:42–47. doi: 10.1016/j.drugalcdep.2009.06.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brown NK, Harvey DJ. In vitro metabolism of cannabichromene in seven common laboratory animals. Drug Metab. Dispos. 1990;18:1065–1070. [PubMed] [Google Scholar]
- Burstein S, Hunter SA, Latham V, Renzulli L. Prostaglandins and cannabis - XVI: antagonism of Δ1-tetrahydrocannabinol action by its metabolites. Biochem. Pharmacol. 1986;35:2553–2558. doi: 10.1016/0006-2952(86)90053-5. [DOI] [PubMed] [Google Scholar]
- Burston JJ, Sim-Selley LJ, Harloe JP, Mahadevan A, Razdan RK, Selley DE, Wiley JL. N-arachidonyl maleimide potentiates the pharmacological and biochemical effects of the endocannbinoid 2-arachidonylglycerol through inhibition of monoacylglycerol lipase. J. Pharmacol. Exp. Ther. 2008;327:546–553. doi: 10.1124/jpet.108.141382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Colpaert FC, Meert T, DeWitte P, Schmitt P. Further evidence validating adjuvant arthritis as an experimental model of chronic pain in the rat. Life Sci. 1982;31:67–75. doi: 10.1016/0024-3205(82)90402-7. [DOI] [PubMed] [Google Scholar]
- Compton DR, Rice KC, De Costa BR, Razdan RK, Melvin LS, Johnson MR, Martin BR. Cannabinoid structure-activity relationships: correlation of receptor binding and in vivo activities. J. Pharmacol. Exp. Ther. 1993;265:218–226. [PubMed] [Google Scholar]
- Compton D, Aceto M, Lowe J, Martin B. In vivo characterization of a specific cannabinoid receptor antagonist (SR141716A): inhibition of Δ9-tetrahdrocannabinol-induced responses and apparent agonist activity. J. Pharmacol. Exp. Ther. 1996;277:586–594. [PubMed] [Google Scholar]
- Costa B, Giagnoni G, Franke C, Trovato AE, Colleoni M. Vanilloid TRPV1 receptor mediates the antihyperalgesic effect of the nonpsychoactive cannabinoid, cannabidiol, in a rat model of acute inflammation. Br. J. Pharmacol. 2004;143:247–250. doi: 10.1038/sj.bjp.0705920. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Costa B, Trovato AE, Comelli F, Giagnoni G, Colleoni M. The non-psychoactive cannabis constituent cannabidiol is an orally effective therapeutic agent in rat chronic inflammatory and neuropathic pain. Eur. J. Pharmacol. 2007;556:75–83. doi: 10.1016/j.ejphar.2006.11.006. [DOI] [PubMed] [Google Scholar]
- Croxford JL, Yamamura T. Cannabinoids and the immune system: potential for the treatment of inflammatory diseases? J. Neuroimmunol. 2005;166:3–18. doi: 10.1016/j.jneuroim.2005.04.023. [DOI] [PubMed] [Google Scholar]
- Davis WM, Hatoum NS. Neurobehavioral actions of cannabichromene and interactions with Δ9-tetrahydrocannabinol. Gen. Pharmacol. 1983;14:247–252. doi: 10.1016/0306-3623(83)90004-6. [DOI] [PubMed] [Google Scholar]
- Demuth DG, Molleman A. Cannabinoid signalling. Life Sci. 2006;78:549–563. doi: 10.1016/j.lfs.2005.05.055. [DOI] [PubMed] [Google Scholar]
- Di Marzo V, Breivogel CS, Tao Q, Bridgen DT, Razdan RK, Zimmer AM, Zimmer A, Martin BR. Levels, metabolism, and pharmacological activity of anandamide in CB(1) cannabinoid receptor knockout mice: evidence for non-CB(1), non-CB(2) receptor-mediated actions of anandamide in mouse brain. J. Neurochem. 2000;75:2434–2444. doi: 10.1046/j.1471-4159.2000.0752434.x. [DOI] [PubMed] [Google Scholar]
- Di Marzo V, Bisogno T, De Petrocellis L, Brandi I, Jefferson RG, Winckler RL, Davis JB, Dasse O, Mahadevan A, Razdan RK, Martin BR. Highly selective CB(1) cannabinoid receptor ligands and novel CB(1)/VR(1) vanilloid receptor "hybrid" ligands. Biochem. Biophys. Res. Commun. 2001;281:444–451. doi: 10.1006/bbrc.2001.4354. [DOI] [PubMed] [Google Scholar]
- Doyle SA, Burstein SH, Dewey WL, Welch SP. Further studies on the antinociceptive effects of Δ6-THC-7-oic acid. Agents Actions. 1990;31:157–163. doi: 10.1007/BF02003237. [DOI] [PubMed] [Google Scholar]
- ElSohly MA, Slade D. Chemical constituents of marijuana: the complex mixture of natural cannabinoids. Life Sci. 2005;78:539–548. doi: 10.1016/j.lfs.2005.09.011. [DOI] [PubMed] [Google Scholar]
- Fan F, Compton D, Ward S, Melvin L, Martin B. Development of cross-tolerance between delta 9-tetrahydrocannabinol, CP 55,940 and WIN 55,212. J. Pharmacol. Exp. Ther. 1994;271:1383–1390. [PubMed] [Google Scholar]
- Fride E. Behavioral methods in cannabinoid research. Methods Mol. Med. 2006;123:269–290. doi: 10.1385/1-59259-999-0:269. [DOI] [PubMed] [Google Scholar]
- Gaoni Y, Mechoulam R. Isolation, structure, and partial synthesis of an active constituent of hashish. J. Amer. Chem. Soc. 1964;86:1646–1647. [Google Scholar]
- Gaoni Y, Mechoulam R. Cannabichromene, a new active principle in hashish. Chem. Commun. 1966;1:20–21. [Google Scholar]
- Gaoni Y, Mechoulam R. The isolation and structure of delta-1-tetrahydrocannabinol and other neutral cannabinoids from hashish. J. Amer. Chem. Soc. 1971;93:217–224. doi: 10.1021/ja00730a036. [DOI] [PubMed] [Google Scholar]
- Gill EW, Paton WD, Pertwee RG. Preliminary experiments on the chemistry and pharmacology of cannabis. Nature. 1970;228:134–136. doi: 10.1038/228134a0. [DOI] [PubMed] [Google Scholar]
- Grotenhermen F. Pharmacokinetics and pharmacodynamics of cannabinoids. Clin. Pharmacokinet. 2003;42:327–360. doi: 10.2165/00003088-200342040-00003. [DOI] [PubMed] [Google Scholar]
- Guzman M, Sanchez C, Galve-Roperh I. Cannabinoids and cell fate. Pharmacol. Ther. 2002;95:175–184. doi: 10.1016/s0163-7258(02)00256-5. [DOI] [PubMed] [Google Scholar]
- Hargreaves K, Dubner R, Brown F, Flores C, Joris J. A new and sensitive method for measuring thermal nociception in cutaneous hyperalgesia. Pain. 1988;32:77–88. doi: 10.1016/0304-3959(88)90026-7. [DOI] [PubMed] [Google Scholar]
- Hatoum NS, Davis WM, Waters IW, Elsohly MA, Turner CE. Synergism of cannabichromene and CNS depressants in mice. Gen. Pharmacol. 1981;12:351–356. doi: 10.1016/0306-3623(81)90089-6. [DOI] [PubMed] [Google Scholar]
- Holley J, Hadley K, Turner CE. Constituents of cannabis sativa L., XI. Cannabidiol and cannabichromene in samples of known geographical origin. J. Pharm. Sci. 1975;64:892–894. doi: 10.1002/jps.2600640546. [DOI] [PubMed] [Google Scholar]
- Hunt CA, Jones RT. Tolerance and disposition of tetrahydrocannabinol in man. J. Pharmacol. Exp. Ther. 1980;215:35–44. [PubMed] [Google Scholar]
- Hylden JLK, Nahin RL, Traub RJ, Dubner R. Expansion of receptive fields of spinal lamina I projection neurons in rats with unilateral adjuvant-induced inflammation: the contribution of dorsal horn mechanisms. Pain. 1989;37:229–243. doi: 10.1016/0304-3959(89)90135-8. [DOI] [PubMed] [Google Scholar]
- Ilan AB, Gevins A, Coleman M, ElSohly MA, De Wit H. Neurophysiological and subjective profile of marijuana with varying concentrations of cannabinoids. Behav. Pharmacol. 2005;16:487–496. doi: 10.1097/00008877-200509000-00023. [DOI] [PubMed] [Google Scholar]
- Kanaan SA, Saade NE, Haddad JJ, Abdelnoor AM, Atweh SF, Jabbur SJ, Safieh-Garabedian B. Endotoxin-induced local inflammation and hyperalgesia in rats and mice: a new model for inflammatory pain. Pain. 1996;66:373–379. doi: 10.1016/0304-3959(96)03068-0. [DOI] [PubMed] [Google Scholar]
- Kanaan SA, Safieh-Garabedian B, Haddad JJ, Atweh SF, Abdelnoor AM, Jabbur SJ, Saade NE. Effects of various analgesic and anti-inflammatory drugs on endotoxin-induced hyperalgesia in rats and mice. Pharmacology. 1997;54:285–297. doi: 10.1159/000139498. [DOI] [PubMed] [Google Scholar]
- Klein TW. Cannabinoid-based drugs as anti-inflammatory therapeutics. Nat. Rev. Immunol. 2005;5:400–411. doi: 10.1038/nri1602. [DOI] [PubMed] [Google Scholar]
- Liu J, Li H, Burstein SH, Zurier RB, Chen JD. Activation and binding of peroxisome proliferator-activated receptor gamma by synthetic cannabinoid ajulemic acid. Mol. Pharmacol. 2003;63:983–992. doi: 10.1124/mol.63.5.983. [DOI] [PubMed] [Google Scholar]
- Martin BR, Compton DR, Thomas BF, Prescott WR, Little PJ, Razdan RK, Johnson MR, Melvin LS, Mechoulam R, Ward SJ. Behavioral, biochemical, and molecular modeling evaluations of cannabinoid analogs. Pharmacol. Biochem. Behav. 1991;40:471–478. doi: 10.1016/0091-3057(91)90349-7. [DOI] [PubMed] [Google Scholar]
- McPartland JM, Russo EB. Cannabis and cannabis extract: greater than the sum of the parts? J. Cannabis Therapuetics. 2001;1:103–132. [Google Scholar]
- Mechoulam R, Yagnitinsky B, Gaoni Y. Stereoelectronic factor in the chloranil dehydrogenation of cannabinoids. Total synthesis of dl-cannabichromene. J. Am. Chem. Soc. 1968;90:2418–2420. doi: 10.1021/ja01011a037. [DOI] [PubMed] [Google Scholar]
- Mechoulam R, Peters M, Murillo-Rodriguez E, O’Hanuscaron L. Cannabidiol - recent advances. Chem. Biodivers. 2007;8:1678–1692. doi: 10.1002/cbdv.200790147. [DOI] [PubMed] [Google Scholar]
- Mehmedic Z, Chandra S, Slade D, Denham H, Foster S, Patel AS, Ross SA, Khan IA, Elsohly MA. Potency Trends of Delta(9)-THC and Other Cannabinoids in Confiscated Cannabis Preparations from 1993 to 2008*. J Forensic Sci. doi: 10.1111/j.1556-4029.2010.01441.x. in press. [DOI] [PubMed] [Google Scholar]
- Naidu PS, Kinsey SG, Guo TL, Cravatt BF, Lichtman AH. Hierarchical regulation of inflammatory pain by inhibition of fatty acid amide hydrolase (FAAH) J. Pharmacol. Exp. Ther. 2009 December; doi: 10.1124/jpet.109.164806. (under review) submitted to. [DOI] [PMC free article] [PubMed] [Google Scholar]
- O'Neil JD, Dalton WS, Forney RB. The effect of cannabichromene on mean blood pressure, heart rate, and respiration rate responses to tetrahydrocannabinol in the anesthetized rat. Tox. Appl. Pharmacol. 1979;49:265–270. doi: 10.1016/0041-008x(79)90250-3. [DOI] [PubMed] [Google Scholar]
- Pertwee RG. The ring test: a quantitative method for assessing the 'cataleptic' effect of cannabis in mice. Br. J. Pharmacol. 1972;46:753–763. doi: 10.1111/j.1476-5381.1972.tb06900.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pertwee RG, Ross RA. Cannabinoid receptors and their ligands. Prostaglandins Leukot. Essent. Fatty Acids. 2002;66:101–121. doi: 10.1054/plef.2001.0341. [DOI] [PubMed] [Google Scholar]
- Piomelli D, Tarzia G, Duranti A, Tontini A, Mor M, Compton TR, Dasse O, Monaghan EP, Parrot JA, Putman D. Pharmacological profile of the selective FAAH inhibitor KDS-4103 (URB597) CNS Drug Rev. 2006;12:21–38. doi: 10.1111/j.1527-3458.2006.00021.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Puffenbarger RA, Boothe AC, Cabral GA. Cannabinoids inhibit LPS-inducible cytokine mRNA expression in rat mircroglial cells. Glia. 2000;29:58–69. [PubMed] [Google Scholar]
- Rietschel ET, Kirikae T, Schade FU, Mamat U, Schmidt G, Loppnow H, Ulmer AJ, Zahringer U, Seydel U, Di Padova F. Bacterial endotoxin: molecular relationships of structure to activity and function. FASEB J. 1994;2:217–225. doi: 10.1096/fasebj.8.2.8119492. [DOI] [PubMed] [Google Scholar]
- Rinaldi-Carmona M, Barth F, Heaulme M, Shire D, Calandra B, Congy C, Martinez S, Maruani J, Neliat G, Caput D, Ferrara P, Soubrie P, Breliere JC, Le Fur G. SR141716A, a potent and selective antagonist of the brain cannabinoid receptor. FEBS Lett. 1994;350:240–244. doi: 10.1016/0014-5793(94)00773-x. [DOI] [PubMed] [Google Scholar]
- Smith PB, Compton DR, Welch SP, Razdan RK, Mechoulam R, Martin BR. The pharmacological activity of anandamide, a putative endogenous cannabinoid, in mice. J. Pharmacol. Exp. Ther. 1994;270:219–227. [PubMed] [Google Scholar]
- Spencer SJ, Mouihate A, Galic MA, Ellis SL, Pittman QJ. Neonatal immune challenge does not effect body weight regulation in rats. Am. J. Physiol. Regu. Integr. Comp Physiol. 2007;293:R581–R589. doi: 10.1152/ajpregu.00262.2007. [DOI] [PubMed] [Google Scholar]
- Tallarida RJ, Porreca F, Cowan A. Statistical analysis of drug-drug and site-site interactions with isobolograms. Life Sci. 1989;45:947–961. doi: 10.1016/0024-3205(89)90148-3. [DOI] [PubMed] [Google Scholar]
- Tallarida RJ. Drug synergism: its detection and applications. J. Pharmacol. Exp. Ther. 2001;298:865–872. [PubMed] [Google Scholar]
- Tallarida RJ. An overview of drug combination analysis with isobolograms. J. Pharmacol. Exp. Ther. 2006;319:1–7. doi: 10.1124/jpet.106.104117. [DOI] [PubMed] [Google Scholar]
- Turner CE, ElSohly MA, Boeren EG. Constituents of Cannabis sativa L. XVII. a review of the natural constituents. J. Nat. Prod. 1980;43:169–234. doi: 10.1021/np50008a001. [DOI] [PubMed] [Google Scholar]
- Turner CE, ElSohly MA. Biological activity of cannabichromene, its homologs and isomers. J. Clin. Pharmacol. 1981;21:283S–291S. doi: 10.1002/j.1552-4604.1981.tb02606.x. [DOI] [PubMed] [Google Scholar]
- Varvel SA, Bridgen DT, Tao Q, Thomas BF, Martin BR, Lichtman AH. Δ9-Tetrahydrocannbinol accounts for the antinociceptive, hypothermic, and cataleptic effects of marijuana in mice. J. Pharmacol. Exp. Ther. 2005;314:329–337. doi: 10.1124/jpet.104.080739. [DOI] [PubMed] [Google Scholar]
- Varvel S, Wiley J, Yang R, Bridgen D, Long K, Lichtman A, Martin B. Interactions between THC and cannabidiol in mouse models of cannabinoid activity. Psychopharmacology (Berl) 2006;186:226–234. doi: 10.1007/s00213-006-0356-9. [DOI] [PubMed] [Google Scholar]
- Widman M, Agurell S, Ehrnebo M, Jones G. Binding of (+) and (−)-Δ1-tetrahydrocannabinols and (−)-7-hydroxy-Δ1-tetrahydrocannabinol to blood cells and plasma proteins in man. J. Pharm. Pharmacol. 1974;26:914–916. doi: 10.1111/j.2042-7158.1974.tb09207.x. [DOI] [PubMed] [Google Scholar]
- Wiley JL, Compton DR, Dai D, Lainton JAH, Phillips M, Huffman JW, Martin BR. Structure-activity relationships of indole- and pyrrole-derived cannabinoids. J. Pharmacol. Exp. Ther. 1998;3:995–1004. [PubMed] [Google Scholar]
- Wiley JL, Martin BR. Cannabinoid pharmacology: implications for additional cannabinoid receptor subtypes. Chem. Phys. Lip. 2002;121:57–63. doi: 10.1016/s0009-3084(02)00146-9. [DOI] [PubMed] [Google Scholar]
- Wiley JL, Martin BR. Cannabinoid pharmacological properties common to other centrally acting drugs. Eur. J. Pharmacol. 2003;471:185–193. doi: 10.1016/s0014-2999(03)01856-9. [DOI] [PubMed] [Google Scholar]
- Wirth PW, Sue Watson E, ElSohly M, Turner CE, Murphy JC. Anti-inflammatory properties of cannabichromene. Life Sci. 1980a;26:1991–1995. doi: 10.1016/0024-3205(80)90631-1. [DOI] [PubMed] [Google Scholar]
- Wirth PW, Watson ES, ElSohly MA, Seidel R, Murphy JC, Turner CE. Anti-inflammatory activity of cannabichromene homologs. J. Pharm. Sci. 1980b;69:1359–1360. doi: 10.1002/jps.2600691136. [DOI] [PubMed] [Google Scholar]
- Zurier RB. Prospects for cannabinoids as anti-inflammatory agents. J. Cell Biochem. 2003;88:462–466. doi: 10.1002/jcb.10291. [DOI] [PubMed] [Google Scholar]
- Zygmunt PM, Peterson J, Andersson DA, Chuang HH, Sorgard M, DiMarzo V, Julius D. Vanilloid receptors on sensory nerves mediate the vasodilator action of anandamide. Nature. 1999;400:452–457. doi: 10.1038/22761. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.