Abstract
Although the incidence of cancer in the United States is high, improvements in early diagnosis and treatment have significantly increased survival rates in recent years. Many survivors of cancer experience lasting, adverse effects caused by either their disease or its treatment. Physical therapy interventions, both established and new, often can reverse or ameliorate the impairments (body function and structure) found in these patients, improving their ability to carry out daily tasks and actions (activity) and to participate in life situations (participation). Measuring the efficacy of physical therapy interventions in each of these dimensions is challenging but essential for developing and delivering optimal care for these patients. This article describes the acute and long-term effects of cancer and its treatment and the use of the World Health Organization's International Classification of Functioning, Disability and Health (ICF) as a basis for selection of assessment or outcome tools and diagnostic or screening tools in this population.
Cancer has a high incidence in the United States, where 46% of all males and 41% of all females can expect to develop either an invasive or in situ cancer.1 An estimated 1.4 million new cases of cancer are diagnosed each year, with nearly 13,500 of these cases occurring in individuals younger than 20 years of age.2 In years past, survival following a diagnosis of cancer was problematic; however, dramatic progress in the ability to diagnose cancers earlier and to provide more-effective and targeted treatments has led to substantial increases in survival. The National Cancer Institutes Surveillance, Epidemiology, and End Results Program estimates that 65.3% of adults diagnosed with cancer between the years 2001 and 2005 will survive for at least 5 years.1 In addition, about 80% of people younger than 19 years of age who are diagnosed with cancer today are expected to survive for 5 years or longer.1,3 All told, an estimated 10 million people are living in the United States today who have or have had a diagnosis of cancer.1 As the population ages and treatments improve, these numbers are expected to continue to rise. Currently available medical interventions for cancer are designed to eliminate or control disease by suppressing cell growth (chemotherapy, irradiation) or directly removing the tumor (surgery).4–15 These treatments may lack specificity and can damage normal tissue.16–19 Thus, cancer is no longer an acute disease, with mortality as the primary outcome. Rather, treatment successes have made cancer a chronic disease, with many survivors developing significant sequelae to either the disease itself or to the treatment.20–23
Oncology rehabilitation has long been a part of the management of cancer, but with increased survivorship, these efforts have evolved from simple supportive and palliative care to now include complex rehabilitation interventions designed to restore the integrity of organ structure and function, to remediate functional loss, and to adapt to the environment so as to allow full participation in daily activities and life roles. In the current medical environment, demonstrating treatment efficacy by means of quantifiable outcome measures is increasingly important. As such, the expansion of interventions provided to patients with cancer and survivors of cancer must be accompanied by the appropriate application of new and existing measures. Because the information generated by these tools may be seen by many health care professionals and can extend across broad spans of time, the utility of such information is greatest when it is presented within a framework of standardized language and concepts. Such a framework can be found in the International Classification of Functioning, Disability and Health (ICF).24 This classification system is designed to describe health and health-related status from biological, personal, and societal perspectives. Disorders across the domains of body structure and function, activities, and participation are referred to as impairments, limitations, and restrictions, respectively. “Functioning” is an umbrella term that encompasses these 3 domains. Health conditions or disease states, personal factors, and the environment interact with these constructs to determine whether disordered functioning will result in disability.24
The primary purpose of this article is to use the ICF framework and its language to describe outcome measures and diagnostic screening tools that the rehabilitation therapist will find useful in assessing patients with an oncology diagnosis. Some of these outcome measures may be new to therapists; others may already be part of their routine assessment. However, factors unique to a diagnosis of cancer or to its treatment may influence how and when such routine measures are used. Thus, the second purpose of this article is to provide greater understanding of the clinical issues common to the oncology population. Collectively, we hope to improve clinical care, facilitate communication across different rehabilitation disciplines, and encourage further study in the area of oncology rehabilitation.
The ICF Function Classification Framework
The ICF was developed by the World Health Organization24 to provide a framework to describe health and health-related states and to suggest standardized language to describe these states. The ICF framework (Figure) is increasingly being used in the rehabilitation field and has recently been endorsed by the American Physical Therapy Association (APTA) House of Delegates for incorporation into all relevant Association publications, documents, and communications.25
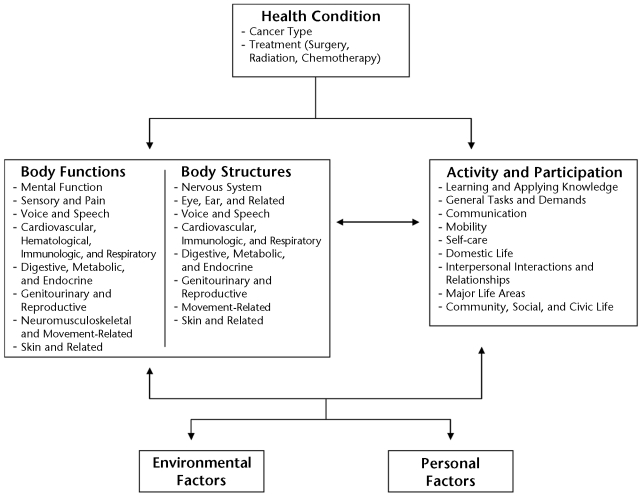
Figure.
International Classification of Functioning, Disability and Health (ICF) model24 modified for populations of people with cancer. Modified and reprinted with permission of the World Health Organization from: International Classification of Functioning, Disability and Health: ICF. Geneva, Switzerland: World Health Organization; 2001.
Based on the work of Nagi,26,27 the ICF model shifts the focus of disablement from cause to impact, from disability to health and function, and from a static process to a dynamic process.24,28 As stated previously, the ICF defines 3 domains of human function (Figure): body function and structure, activity, and participation. Body function and structure refers to the anatomical and physiological function of the body systems, and these body functions and body structures are categorized into the subdomains listed in the Figure. Deficits in this domain are called “impairments” (eg, muscle weakness, restricted joint motion, poor cardiorespiratory fitness) and often are identified, measured, and treated by physical therapists. The activity domain describes the ability of an individual to perform specific tasks such as sweeping the floor, raking the yard, or putting away groceries. Decrements in the activity domain are called “limitations” and describe the difficulty an individual has performing a particular task.24 Physical therapy goals often are aimed at reversing or normalizing such activity limitations. The participation domain describes the ability of a person to be involved in life situations. Participation restrictions describe the reduced ability of a person to maintain normal role functions and interact with society.24,29,30 Physical therapy interventions are designed, directly or indirectly, to enhance participation levels for every client at home, school, or work; on the athletic field; or in any community setting. The activity and participation subdomains are given as a single list (Figure), and their use will be discussed in the “Measurement of Activity and Participation” section of this article.
In the ICF model, health conditions, personal factors, and the environment interact dynamically across the 3 domains of body function to help determine whether disordered function results in disability. For example, if a cancer treatment (eg, chemotherapy) causes a patient to develop unresolved peripheral neuropathy and ankle weakness,31 this patient may have a limited ability to walk (limitation) and may require long-term use of an ankle brace. Limited ability to walk could result in an employment restriction for a firefighter, but not for a computer programmer. Participation restrictions occur when activity limitations cannot be sufficiently overcome to maintain role functions in the person's normal environment.29,30
Formal work is emerging that uses the ICF classification scheme to describe overall function of populations who have specific chronic health conditions, including, but not limited to, multiple sclerosis,32 stroke,33,34 osteoarthritis,35 diabetes,36 low back pain,37 obesity,38 osteoporosis,39 and rheumatoid arthritis.40,41 This growing body of literature uses the ICF framework to identify measurements relevant to a specific illness. The ICF Core Sets provide clinicians and researchers with comprehensive but concise measurement categories that describe a patient's global function from a biopsychosocial view. Some investigators42–46 have used the ICF Core Sets as the comparison standard for the assessment of function and disability when evaluating the content of a previous or newly developed measurement tool.
A limited number of ICF Core Sets have been developed for patients with head and neck cancer47 and breast cancer.48 Although the ICF Core Sets have not been widely used in the US physical therapy or oncology communities, the ICF framework is a useful model for describing global function in patients with a new or previous cancer diagnosis.49 Consideration of the interaction among cancer as a health condition, impairments in body function and structure, activity limitations, and participation restrictions in the context of the person and the environment are essential to the design of an effective oncology rehabilitation intervention.50
Selecting Appropriate Measures
In this article, we describe measures as potential descriptors of particular ICF function domains. We encourage therapists to use this schema to assist them in deciding which measures to include in their baseline, continuing, and final outcome assessments of their patients and clients. To do this, the therapist should review the primary goals of the intervention and determine how these goals fit into the ICF domains. That is, which of the ICF domains is the intervention intended to affect? If the intervention is designed to make a change at the tissue level, then the appropriate measure would assess a specific change at the body function and structure level. For example, a patient with restricted shoulder mobility (decreased range of motion [ROM]) after a mastectomy may be treated with a regimen of stretching and scar tissue mobilization where the intended outcome is lengthened tissue, making ROM an appropriate measure. By increasing ROM, this intervention also may improve the patient's ability to reach overhead, making certain daily tasks possible (an activity-level measure), which, in turn, may increase the patient's ability or willingness to engage in life activities such as work or education (a participation-level measure). In this example, outcome measures at each level would be appropriate, and such information would speak to the efficacy of the intervention across functional domains.
Selecting an outcome measure also requires consideration of the psychometric properties of the instrument or tool the therapist is planning to use. Validity, reliability, and responsiveness are 3 properties the therapist should consider.51 The measure should make sense (face validity), be accepted by experts in the field (content validity), and correlate with an expected outcome (predictive validity) and with other measures that evaluate the same construct (concurrent validity). The instrument should yield the same results (reliability) when repeated by separate examiners (interrater reliability), by the same examiner on the same patient (intrarater reliability), or on separate occasions within a time period when no changes would be expected (test-retest reliability). The therapist also will want to select an instrument that is capable of detecting change resulting from an intervention (responsiveness).51 Instruments that place individuals into a limited number of categories,51 such as the Functional Independence Measure,52 tend not to be responsive because very large changes are required to move from one category to another. Additionally, instruments should not have a ceiling effect. If many respondents initially score at the highest level, there is no room for improvement, and change will not be detected.51
It is important to understand that the psychometric properties of validity for diagnostic and screening measures are different than for outcome measures.53 Clinicians need to know how accurate the diagnostic tool is in identifying the presence or absence of the target condition. Often a new tool is compared with a gold standard, and its validity is described using sensitivity and specificity. Sensitivity, often referred to as a “true positive rate,” is defined as a test's ability to correctly identify the target condition when the target condition is present. A high sensitivity is desirable, as it will rarely miss someone who has the condition. Specificity describes a test's ability to identify those without the target condition who really do not have the target condition, a “true negative rate.” If an instrument has a high specificity, then this instrument will rarely test positive when a person does not have the disease (ie, a low chance of false positive predictions).
In this article, we provide examples of measures that are relevant to particular impairments, limitations, and restrictions experienced by patients with cancer or survivors of cancer. The list is not exhaustive and is not restricted by documented reliability, validity, or responsiveness of the particular instrument; however, it does include instruments commonly used by physical therapists, some specifically developed for oncology populations. When choosing a measurement tool, the therapist should investigate its psychometric properties in relation to the population of interest. The references given in Tables 1, 2, and 3 provide a starting point for those searches.
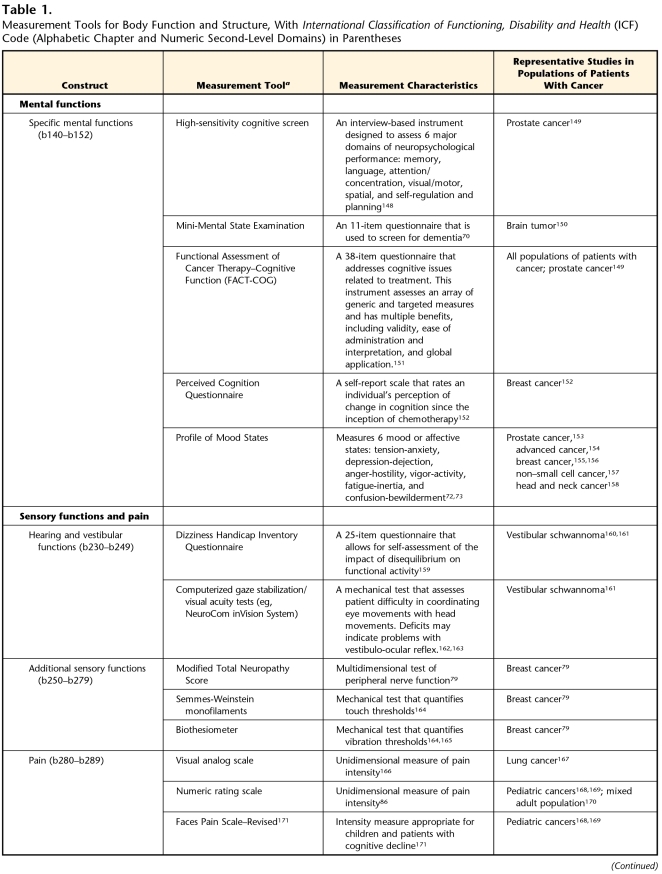
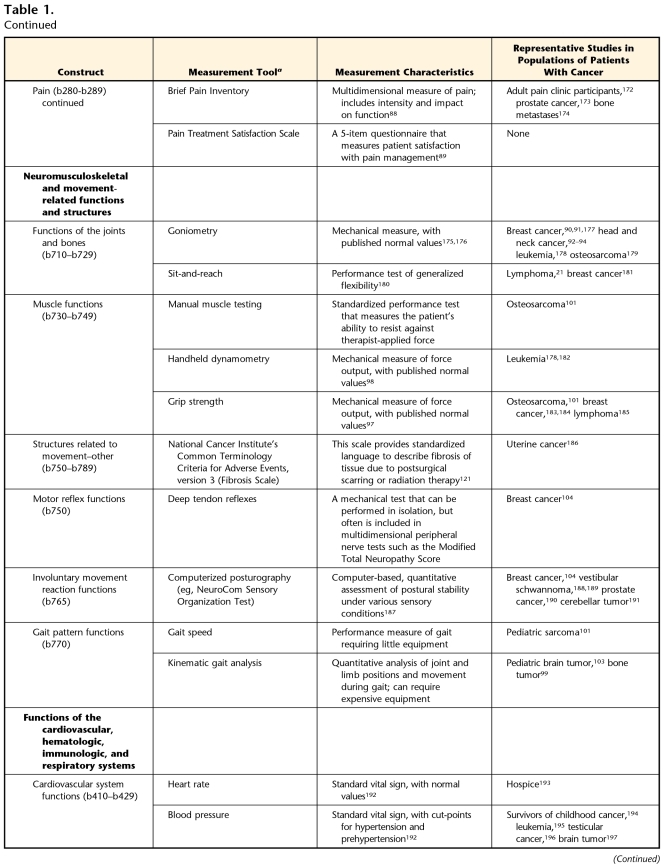
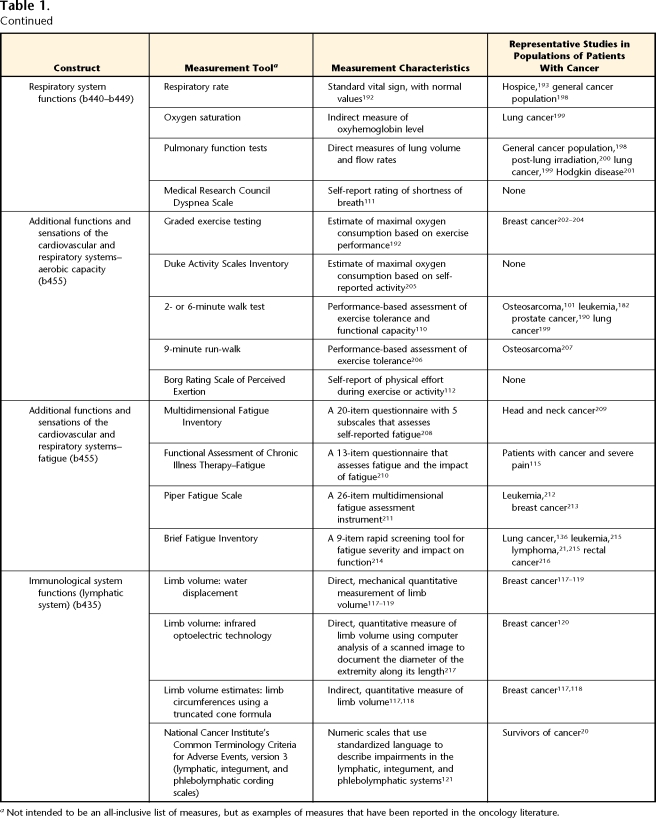
Table 1.
Measurement Tools for Body Function and Structure, With International Classification of Functioning, Disability and Health (ICF) Code (Alphabetic Chapter and Numeric Second-Level Domains) in Parentheses
aNot intended to be an all-inclusive list of measures, but as examples of measures that have been reported in the oncology literature.
Table 2.
Physician-Performed Diagnostic Measures of Body Structure and Function Indicating “Red Flags” or “Yellow Flags” for Physical Therapists, With International Classification of Functioning, Disability and Health (ICF) Code (Alphabetic Chapter and Numeric Second-Level Domains) in Parentheses
aThese tests are performed by a physician, but yield important information for the physical therapist.
Table 3.
Measurement of Activity and Participation, With International Classification of Functioning, Disability and Health (ICF) Code (Alphabetic Chapter and Numeric Second-Level Domains) in Parentheses
aNot intended to be an all inclusive list of measures, but as examples of measures that have been used in the oncology literature.
Measurement of Body Function and Structure
The specific tests and measures used by the physical therapist to measure body function and structure in patients with a cancer diagnosis often are not unique to the assessment of this population. However, these measures provide relevant information about cancer-related impairments, prognostic considerations, and safety factors. This section highlights some common cancer-related changes in body function and structure and suggests some appropriate measurement tools for assessing these impairments.
Mental Functions
Mental functions (Tab. 1, Mental Functions), although not the primary interest of most physical therapists, play an important role in determining how best to interact with and provide interventions for our patients. Both radiation and chemotherapy can alter the structure and function of the central nervous system and may result in impaired mental function in patients during or following treatment for their cancer.54–65 Specific mental function sequelae, including impaired memory and difficulty with sustained attention (concentration), may be evident years after treatment.58,66 Proposed mechanisms for these impairments include chemical toxicity, oxidative damage, inflammation, and destructive autoimmune responses.67–69 The Mini-Mental State Examination70 is a simple tool for screening mental functions and has been used by physical therapists. Although an array of more-complex and detailed neuropsychological tests are available to measure the various domains of cognitive function, information is lacking regarding the sensitivity and specificity of the tests to detect changes in cognitive function from chemotherapy. The identification of sensitive neuropsychological tests is crucial to further understanding of chemotherapy-induced cognitive impairments.71
Emotional functions also may affect the ability of a patient to participate in the physical therapy intervention. A tool that has been used to evaluate emotional functions in patients with cancer is the Profile of Mood States.72,73 This self-report instrument is easy to use and may provide insight into our patient's ability to respond to and participate in a physical therapy program.
Sensory Functions and Pain
Table 1 (Sensory Functions and Pain) describes several potential measures for vestibular, somatosensory, and pain impairments. These impairments are common in patients who are undergoing cancer treatment or have a history of cancer.
Hearing and vestibular functions can be affected by tumor growth or by chemotherapy. Although auditory impairments are infrequently targets of physical therapy assessment, vestibular impairments and their relationship to balance dysfunction should be considered. Vestibular schwannoma, a relatively rare benign tumor, can impair vestibular function, usually unilaterally. Cisplatin, a chemotherapy drug used to treat many types of tumors (eg, lung, breast, ovarian) has been associated with both vestibular toxicity and ototoxicity.74,75 Tests of vestibular function can help physical therapists document change during or after treatment (Tab. 1). It also is important for therapists to use a measure of balance for patients with these impairments (see the “Neuromusculoskeletal and Movement-Related Functions and Structures” section for more information on balance measures).
Treatment-induced peripheral nerve impairments are common. Several chemotherapy drugs (ie, taxanes, platinum agents, vinca alkaloids, and thalidomide) can damage peripheral axons and nerve cell bodies.76 Chemotherapy-induced peripheral neuropathy (CIPN) is characterized by sensory impairments, including paresthesias, dysesthesias, decreased touch thresholds, decreased vibration thresholds, and reduced deep tendon reflexes.77–79 As CIPN worsens, muscle weakness and limb movement disorders, such as foot drop, may develop and require the use of an orthosis. Multidimensional tests, such as the modified Total Neuropathy Scale, may be beneficial in fully describing the severity of CIPN (Tab. 1).77,80,81 Anesthesia or dysesthesias may occur when compression or surgical dissection of a nerve occurs.82 Radiation plexopathies also may occur but are much less common, as radiation oncologists have developed techniques to shield delicate neural structures.83
Many patients with cancer, particularly those with advanced or metastatic disease, have increased levels of pain.84 Cancer-related pain may arise from the tumor itself or as a side effect of treatment. Some forms of cancer are inherently more painful, specifically any cancer originating in or metastasizing to the bone. Pain can have a large impact on mobility, and some researchers have even established cut-points for moderate and severe pain based on its interference with daily activity.85 Evaluation of pain in this population is essential and should be multifaceted (Tab. 1, Pain). Although many scales, such as visual analog scales and numeric rating scales,86,87 specifically focus on pain intensity, other scales are multidimensional and include questions on interference with daily activity88 or acceptability of pain treatments.89
Neuromusculoskeletal and Movement-Related Functions and Structures
Table 1 (Neuromusculoskeletal and Movement-Related Functions and Structures) describes useful measures for evaluating potential changes in neuromusculoskeletal and movement-related functions and structures. Patients with cancer or a history of cancer may experience a number of impairments in this subdomain, including loss of ROM, decreased strength (force-generating capacity), gait pattern abnormalities, and balance deficits.
Deficits in ROM may be present in patients who have undergone surgery, chemotherapy, or radiation therapy. Such deficits may result from the formation of scar tissue following surgery, disuse of a joint following chemotherapy or surgery, or fibrosis caused by irradiation. Decreased ROM may occur coincident with treatment or after the completion of treatment. Seemingly less-invasive surgeries (lumpectomy versus mastectomy) can affect ROM as much as more-invasive procedures.90 Decreased ROM also should be considered if radiation treatment has involved a joint.91 This loss of ROM may occur after radiation is completed and can extend beyond the immediately irradiated joint. For example, patients who have completed surgery or radiation for a head and neck tumor may have impaired shoulder abduction and flexion in addition to the more obvious loss of cervical ROM. These problems are more severe after surgeries involving radical neck dissections and the removal of the spinal accessory nerve.92–94
Muscle strength deficits can arise from tumor-produced inflammatory intermediates that are catabolic, resulting in muscle wasting (cachexia).95 Surgical interventions also may damage muscle groups and peripheral nerves, leading to loss of strength. Radiation and chemotherapy (especially the vinca alkaloids, taxanes, and platinum agents) can reduce strength by damaging muscle or peripheral nerve tissue.91 Corticosteroids preferentially damage proximal limb muscles, limiting activities such as sit-to-stand and overhead reaching.96 Additionally, pain, fear, and fatigue lead to inactivity, which, in turn, causes further loss of muscle strength and aerobic capacity. Although techniques for manual muscle testing are widely used by therapists to measure strength, measures of dynamometry and grip force provide quantitative documentation of strength deficits.97,98
Cancer or cancer treatments can alter gait characteristics by adversely affecting the function and structure of the lower extremity or the nervous system. The few studies that have assessed these changes have shown deficits in patients with bone tumor lesions of the lower extremity and tumors of the nervous system.99–103 Traditional gait evaluation tools, such as kinematic analysis or gait speed measurements, are appropriate for patients with cancer.
Balance can be disrupted in many patients with cancer or a history of cancer and may arise from impairments in multiple body systems. Problems with sensory input, central processing of balance-related information, ROM limitations, orthostatic hypotension, and muscle force production can all contribute to this multifactorial issue. Specific to the neuromuscular system, patients with taxane-induced peripheral neuropathy have limitations in postural stability.104 It is important for physical therapists to measure postural control in a variety of challenging positions to detect and treat balance limitations in patients, especially after chemotherapy. Because the oncology population often is at risk for falls,105 screening for balance disorders is very important. We have included measures that are intended to identify balance impairments and their underlying structural problems in Table 1 (Measurement Tools for Body Function and Structure: Involuntary Movement Reaction Functions) and tests that use mobility skills to rate the level of balance dysfunction in Table 3 (Measurement of Activity and Participation: Mobility–Changing and Maintaining Body Positions). In either case, in the ICF model, a balance disorder is classified as a body function and structure impairment.
Functions of the Cardiovascular, Hematologic, Immunologic, and Respiratory Systems
Cardiotoxicity is a well-known late effect of several chemotherapeutic agents, particularly the anthracyclines (Adriamycin*) and trastuzumab (Herceptin†). These compounds may damage cardiac myocytes and ultimately can result in congestive heart failure.106,107 Similarly, radiation striking the heart can cause cardiac and coronary artery scarring, leading to restrictive cardiac disease and coronary artery disease.108 In older patients, these cardiovascular changes may be superimposed on already existing cardiovascular disease, further amplifying the impairments associated with this disease. It is important for therapists to ask the patient's physician for the results of cardiac testing, performed both before and after treatment with cardiotoxic agents (Tab. 2).
Primary tumors of the lung are frequent, with 215,020 new cases estimated for 2008 in the United States.1 These space-occupying tumors cause respiratory impairments by limiting the expansion of the thoracic cavity, compressing the airways, and reducing the surface area of the lungs available for gas exchange. As these tumors grow and impinge on other mediastinal structures, they can decrease cardiac function secondary to cardiac and vascular compression and cause upper-extremity musculoskeletal injury secondary to brachial plexus compression and infiltration.
The respiratory system also can be adversely affected by chemotherapy and radiation treatment for cancers not involving the lung. Chemotherapeutic agents such as bleomycin, methotrexate, and docetaxel can damage pneumocytes and the pulmonary parenchema.109 Such damage can lead to obliteration of alveoli and dilation of air spaces. Likewise, chest wall irradiation can damage the lining of the alveoli, leading to toxicities such as pneumonitis and fibrosis,109 as well as causing fibrosis of integumentary and musculoskeletal structures that contribute to ventilation.
Measurements of vital signs (heart rate, blood pressure, respiratory rate, and oxygen saturation) provide insight into the cardiorespiratory status of patients with cancer. The presence of hemodynamic instability at rest (altered blood pressure, tachycardia, light-headedness, cyanosis) suggests that action should be taken to protect the patient. Impairments in cardiorespiratory status may manifest themselves only with increased exertion. For this reason, assessment involving testing under conditions of increased exertional demand (Tab. 1) is preferred and may involve formal exercise testing, self-report of activity levels, or results from a 6-minute walk test or similar aerobic capacity test.110 Failure to meet normal range values for these assessment tools suggests impaired cardiovascular and respiratory function. Patient report of breathing difficulties (Dyspnea Scale111) and of exertional demand (Borg Rating of Perceived Exertion112) during a 6-minute walk test provide further insight into these impairments.
Fatigue is a well-documented, multisystem impairment commonly reported in a wide variety of cancers, both acutely and long after cancer treatments have ended.113 Exercise is an effective intervention for cancer-related fatigue, and it is recommended that a multidimentional measure be used to capture the physical, emotional, and mental aspects of fatigue.114 One such measure is the fatigue subscale of the Functional Assessment of Chronic Illness Therapy (FACIT-F), which initially was developed for the oncology population and has been used in patients with a variety of cancer types115 and in survivors of cancer.116
In the ICF, the function of the lymphatic vessels and nodes are classified under immunologic function. Defects may involve tumor obstruction of lymphatic vessels, but they more likely occur secondary to surgical resection of lymph nodes or radiation-induced fibrotic changes that affect lymphatic vessels. In any case, regional lymphatic drainage is reduced, leading to lymphatic fluid accumulation and regional swelling. Such swelling compromises the integument by increasing the likelihood of inflammation, infection, skin breakdown, limits in joint ROM, and decreased ability to move the affected limb. Lymphedema may be most associated with surgical resection of the breast and surrounding lymph nodes; however, surgical resection of a variety of tumors, including head and neck, genitourinary, and reproductive tumors, can result in lymphedema. Localized swelling is the most common impairment of lymphedema; therefore, measures of this impairment focus on quantifying limb volume (Tab. 1, Immunological Systems Functions). The water displacement method is a highly reliable method for determining the volume of an extremity with lymphedema.117–119 However, this method requires specific equipment and precise methods to obtain reliable measurements. Methods using light-emitting diodes to calculate limb volume have shown early evidence in detecting subclinical lymphedema, allowing for early intervention and prevention of symptomatic lymphedema.120 Volume estimates made by a truncated cone formula using several limb circumference measures correlate highly with those determined by water displacement.117,118 Limb circumference measurements may be more practical for some clinicians, given its simplicity and minimal equipment requirements. An important component to early detection is the timing of volume measurements. It has been shown that preoperative measurements assist with early detection and successful treatment of lymphedema.120
Volume measures are only one method used to describe the severity of lymphatic impairments. The National Cancer Institute's Common Terminology Criteria for Adverse Events, version 3,121 has expanded the number of scales to grade the severity of lymphatic and integumentary toxicity (ICF subdomain “skin and related structures”). There are separate scales for volume of lymphedema in extremities, trunk and genital region, head and neck, and viscera. In addition, there are scales to grade the severity of skin color changes, lymph leakage, lymphocele, fibrosis, and phlebolymphatic cording.121 A weakness of these scales is that the categories are broad and, therefore, not sensitive to small differences that may be clinically important. However, they do provide standardization of language to describe changes to lymphatic tissues and integumentary that may be clinically useful, particularly for long-term goals and clear communication among colleagues.
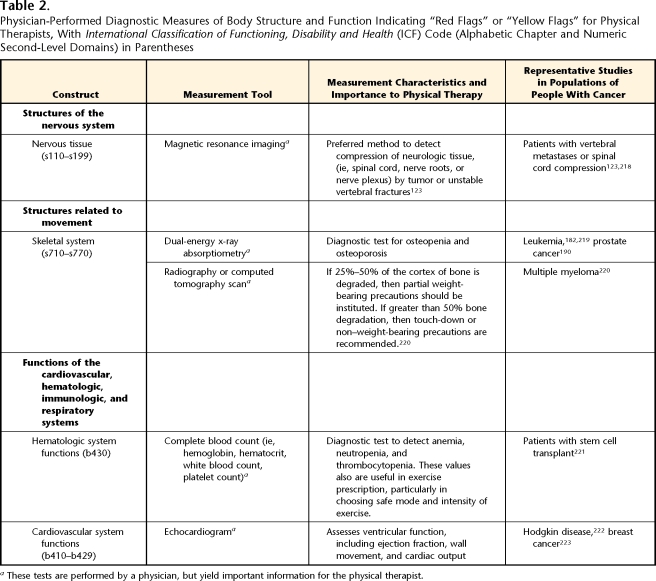
Diagnostic Measures of Body Function and Structure Indicating “Red Flags” or “Yellow Flags” for Physical Therapists
Body function and structure impairments identified through diagnostic tests performed by a physician may have significant implications for the examination by a physical therapist and the physical therapy plan of care (Tab. 2). Conversely, the therapist may identify concerning “red flags” or “yellow flags” during the examination that would warrant recommending that the patient return to his or her physician for further diagnostic testing. Both situations affect patient safety and, therefore, are described below and in Table 2.
Some tumors cause neural impairment by compressing or infiltrating a peripheral nerve, nerve plexus, or a nerve tract or nucleus within the central nervous system. The impairment may be sensory, motor, or autonomic, depending on the location, size, and structure of the tumor. Physical therapists must consider common neurological sites at increased risk for tumor compression. For example, breast and lung tumors can compress the brachial plexus, and the lumbosacral plexus is sometimes affected by colorectal tumors, gynecologic tumors, sarcomas, and lymphomas.122 Regardless of the site, the cardinal sign of neural compression is unrelenting pain, particularly at night and later focal sensory disturbances or weakness in the distribution of the plexus or spinal cord segment involved.122,123 These signs and symptoms are particularly important to consider in patients with a history of cancer who may enter the clinic with a seemingly unrelated musculoskeletal problem. If neural compression from a tumor is suspected, the therapist needs to refer the patient back to the primary physician so that further medical tests, such as magnetic resonance imaging, and appropriate treatment may be performed.
Skeletal impairments often accompany a cancer diagnosis and reflect a disease-associated loss of bony material (lytic tumor) or invasion of bone (sclerotic tumor) by a primary or secondary tumor. Communication with the medical team can help therapists navigate through the many risks associated with tumor invasion of skeletal structures. It is advantageous for therapists to be familiar with common patterns of cancer-related skeletal system involvement (eg, prostate, breast, lung, and colon cancer often metastasize to the spine; sarcomas commonly present in the femur). As the tumor invades the normal structure of bone, there is reduced bone strength and increased risk of pathological fracture.124 Although there are no definitive guidelines to predict pathologic fracture risk, it is helpful to monitor the amount of cortex that has been disrupted by tumor growth in long bones used for functional tasks (eg, the femur for gait, the humerus if an assistive device is being used). This can be calculated by a radiologist, using advanced imaging techniques. Table 2 summarizes specific weight-bearing guidelines. Tumor invasion of the vertebrae also can affect the physical therapy plan of care. If the tumor invades the vertebral arch, the segment may become unstable and possibly compress the spinal cord or adjacent nerve roots, creating a medical emergency. Unrelenting back pain often is the primary or presenting symptom of these lesions, and if a therapist suspects neurologic involvement, a segmental motor, sensory, and autonomic examination should be performed.123 If neural impingement is suspected, the medical team should be notified immediately.125
Osteonecrosis and reduced bone mineral density are common among patients with cancer. Both cancer and cancer treatments increase the risk for developing osteonecrosis in a variety of locations, including proximal or distal femur, proximal humerus, jaw, and metatarsals.126,127 New-onset pain and decreased weight-bearing ability should alert therapists to the possibility of osteonecrosis; however, this condition is not always symptomatic.128 Pharmaceutical therapies (eg, corticosteroids, hormonal therapies, androgen therapy) and radiation are associated with reduced bone mineral density.68–70 Therefore, dual-energy x-ray absorptiometry or computed tomography test results can alert therapists to this problem and allow for appropriate intervention planning.
Tests such as a complete blood count can help physical therapists determine safe exercise guidelines, particularly for patients who are undergoing or have just completed chemotherapy, radiation therapy, or bone marrow transplant.129,130 Each medical center or rehabilitation department has its own criteria for exercise prescription. These values often are the same as those used for the general acute care population, as—to our best knowledge—there are no evidence-based recommendations specific for patients with cancer.129,130 In addition to checking for anemia, patients not tolerating aerobic exercise should be screened for current or past use of cardiotoxic or pneumotoxic chemotherapy medications and referred as appropriate for further testing (see cardiovascular and respiratory discussion above). Patients should avoid exposure to infectious pathogens while neutropenic (eg, avoid public gyms, health caregivers should avoid patient contact if they are ill). If the patient is thrombocytopenic, high-impact activities or contact sports should be avoided to prevent excessive bleeding.
Measurement of Activity and Participation
The activity and participation domains encompass the ability to execute tasks, such as walking or bathing (activity), and the ability to participate in life situations, such as regularly attending work or school and conducting interpersonal relationships (participation). The subdomains for activity and participation (such as mobility and domestic life) are given in a single list in the Figure, with each component being able to denote activity, participation, or both.24 This flexibility allows for individual tailoring and operational differentiation of activity and participation.28,131,132 The ICF beginners guide suggests that clinicians, researchers, and policymakers may use this single list for their needs and purposes to “A) designate some domains as Activities and others as Participation and not allow overlap; B) make this designation but allow overlap in particular cases; C) designate detailed (third- or fourth-level) categories within a domain as Activities and broad (second-level) categories in the domain as Participation; or D) designate all domains as potentially both Activity and Participation.”24(p127) Impairments in body function and structure discussed in the previous sections can result in changes at both the activity and participation levels. Therefore, assessing change in these constructs is important.
Physical therapists typically select primary outcome measures at the activity and participation levels when their intervention plan as a whole is directed toward improving a person's physical capacity or performance. Individually tailored rehabilitation goals, commonly seen in physical therapy, take into account personal and environmental factors unique to the patient; however, the use of standardized measures allows for the comparison of individual activity and participation performance to what might be expected from control or population-specific values. The ability to make such comparisons may assist the therapist in gauging patient progress during the course of rehabilitation.
Important activity and participation domains typically addressed by physical therapy interventions include: (1) mobility, for example changing and maintaining a body position, carrying objects, or walking and moving around; (2) self-care, such as dressing, bathing, and toileting; (3) domestic life (eg, carrying a child, doing dishes); and (4) major life areas such as the ability of a child to access a classroom or the ability of an adult to perform specific tasks related to paid employment (Tab. 3). Currently available measures of activity and participation are rarely limited to a specific subdomain, and most instruments include portions of multiple constructs (eg, mobility and self-care).131
Measuring activity limitations and participation restrictions can be done by timed or clinician-observed evaluation or by patient self-report. For example, the Functional Mobility Assessment requires patients to physically perform specific tasks and to answer questions, quantifying their level of function.133 In contrast, the Toronto Extremity Salvage Score, lower-extremity version, is a self-administered questionnaire that asks patients to indicate the level of difficulty they experience in dressing, grooming, mobility, work, sports, and leisure.134,135 Both methods of measurement have different positive attributes. In using quantitative measurement of limitations or restrictions, a therapist removes some of the potential influences of symptom distress or cognitive changes.136 However, the therapist must take into account that performance-based measures are effort dependent and require that the activity be done in a standard way. Severe cognitive problems may make a performance-based measure difficult or impossible to do. Qualitative measures also are important, as patient-reported outcomes reflect the patient's own perspective on his or her limitations and restrictions. Additionally, some symptoms, such as pain, can be measured only by self-report. By adding the patient's perspective, we can better document the perceived burden of cancer and meaningful impact of interventions.137
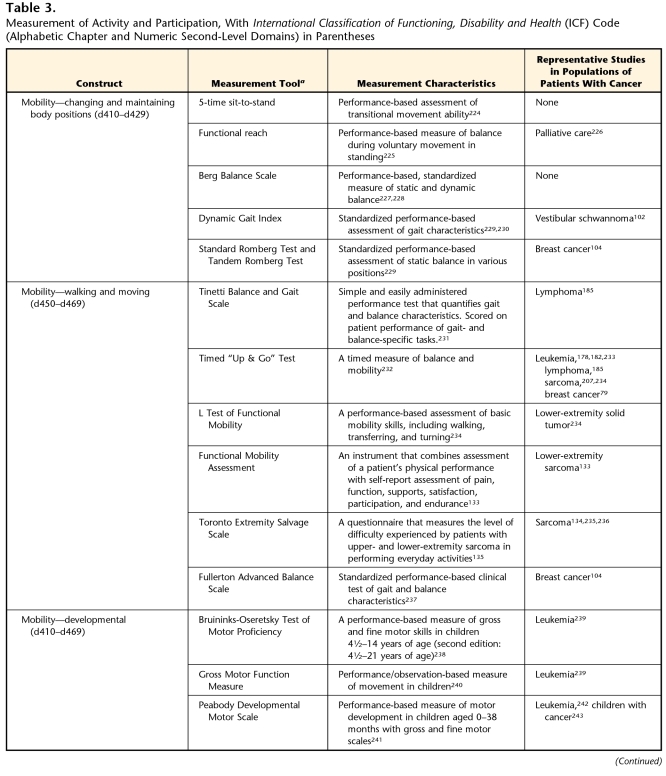
Mobility
The mobility subdomain includes the following constructs: changing and maintaining body positions; carrying, moving, and handling objects; walking and moving; and moving around using transportation. We will discuss the changing and maintaining body positions and walking and moving constructs, as they are assessed most commonly by physical therapists.
Changing and maintaining body positions incorporates both the concepts of maintaining balance and transferring between surfaces. Because the balance deficits discussed in the body function and structure section lead to impaired ability to change and maintain body positions, this is a critical area to explore in this population. Several appropriate activity-based measures of maintaining and changing body positions, including those that relate to balance impairments, are described in Table 3.
The concepts of transferring between surfaces and walking and moving often are combined in rehabilitation outcome measures, although they are separate categories in the ICF model. A few examples of combined transfer and mobility status measures include the Timed “Up & Go” Test and the L Test of Functional Mobility (Tab. 3).
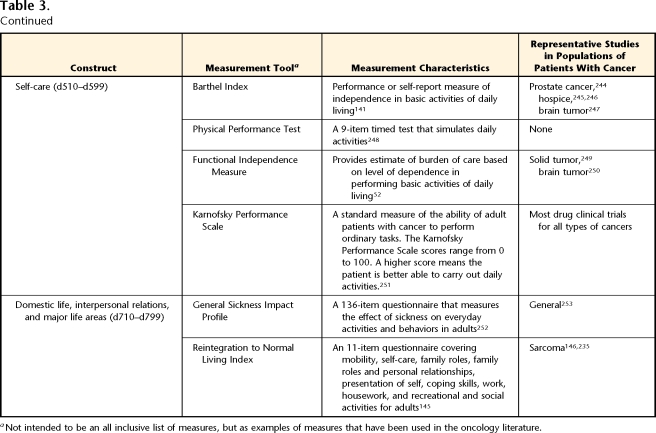
Self-care
The ability to care for one's self is a construct often measured in rehabilitation settings. A few commonly reported measures are listed in Table 3 (Self-care). The Karnofsky Performance Scale138 has been a “gold standard” measure of overall performance status in cancer treatment trials. In its mid-range values, scores indicate the ability of a person to perform self-care. Because of its limited scope, some authors139,140 have reported that it is potentially limited in its responsiveness, a factor that may make it less useful for measuring rehabilitation outcomes. Other measures, such as the Barthel Index,141 have multiple components, including large representations of self-care activities in their content, and are likely to be more responsive to changes seen with rehabilitation. Although these scales are used often in inpatient rehabilitation research, they have relevance for oncology populations that may or may not be seen in such a setting.
Domestic Life, Interpersonal Relations, and Major Life Areas
Few measures typically used by physical therapists attempt to quantify the capacity of a person to live as a family member and as a member of society (Tab. 3). Restrictions in the ability of an individual to interact with the environment or participate fully in life situations increase the disease burden on the individual, the family, and society. Indeed, people with participation restrictions are more likely to report poor health142 and bouts of depression.143 It is generally recognized that patients and survivors of cancer have restrictions in these domains,144 yet there is a paucity of outcome measures targeted here.
A measurement tool that is focused specifically on the return to lifes roles after a major health change is the Reintegration to Normal Living Index.145 This tool measures adults’ perception of their ability to resume their life roles after a serious illness or trauma. It has been used sparingly in populations of people with cancer.146,147 Because performance of activities and participation in life roles often are the main goals of rehabilitation, measurement of pertinent activity and participation subdomains provides useful information regarding the need for and effectiveness of oncology rehabilitation.
Conclusion
This article uses the ICF model to describe outcome measures that allow for broad quantification of global function and methods to document progression in patients with cancer and survivors of cancer. Understanding and documenting how these structural or anatomic deficits restrict activities (grooming, dressing, child care) and participation (attending community activities, reduced job expectations) provide a broader view of the patient's abilities. Therapists need to be adept at understanding the intended focus of their therapeutic interventions and using the most appropriate tools to assess the effectiveness of those interventions.
All authors provided concept/idea/project design and writing. Dr Gilchrist and Dr Galantino provided project management. Dr Ness provided consultation (including review of manuscript before submission).
As the Research Committee of the Oncology Section of the American Physical Therapy Association, the authors thank the Oncology Section for their assistance and support in the development of the manuscript.
Pharmacia Inc, Kalamazoo, MI 49001.
Genentech Inc, 1 DNA Way, South San Francisco, CA 94080-4990.
References
- 1.Ries L, Melbert D, Krapcho M, et al. SEER Cancer Statistics Review, 1975–2005. Available at: http://seer.cancer.gov/csr/1975_2005/. Accessed July 1, 2008, based on November 2007 SEER data submission, posted to the SEER Web site, 2008.
- 2.US Cancer Statistics Working Group. United States Cancer Statistics: 1999–2004. Incidence and mortality Web-based report. Available at: www.cdc.gov/uscs. Accessed November 26, 2008.
- 3.Robison LL. Cancer survivorship: unique opportunities for research. Cancer Epidemiol Biomarkers Prev. 2004;13:1093. [PubMed] [Google Scholar]
- 4.Merchant TE. Current management of childhood ependymoma. Oncology (Willison Park). 2002;16:629–642, 644; discussion 645–646, 648. [PubMed] [Google Scholar]
- 5.van den Berg H. Biology and therapy of malignant solid tumors in childhood. Cancer Chemother Biol Response Modif. 2003;21:683–707. [DOI] [PubMed] [Google Scholar]
- 6.Sklar CA. Childhood brain tumors. J Pediatr Endocrinol Metab. 2002;15:669–673. [DOI] [PubMed] [Google Scholar]
- 7.Freeman CR, Taylor RE, Kortmann RD, Carrie C. Radiotherapy for medulloblastoma in children: a perspective on current international clinical research efforts. Med Pediatr Oncol. 2002;39:99–108. [DOI] [PubMed] [Google Scholar]
- 8.Habrand JL, De Crevoisier R. Radiation therapy in the management of childhood brain tumors. Childs Nerv Syst. 2001;17:121–133. [DOI] [PubMed] [Google Scholar]
- 9.Kalapurakal JA, Dome JS, Perlman EJ, et al. Management of Wilms’ tumour: current practice and future goals. Lancet Oncol. 2004;5:37–46. [DOI] [PubMed] [Google Scholar]
- 10.Schwartz CL. Health status of childhood cancer survivors: cure is more than the eradication of cancer. JAMA. 2003;290:1641–1643. [DOI] [PubMed] [Google Scholar]
- 11.Schwartz CL. The management of Hodgkin disease in the young child. Curr Opin Pediatr. 2003;15:10–16. [DOI] [PubMed] [Google Scholar]
- 12.Alcoser PW, Rodgers C. Treatment strategies in childhood cancer. J Pediatr Nurs. 2003;18:103–112. [DOI] [PubMed] [Google Scholar]
- 13.Rao BN, Rodriguez-Galindo C. Local control in childhood extremity sarcomas: salvaging limbs and sparing function. Med Pediatr Oncol. 2003;41:584–587. [DOI] [PubMed] [Google Scholar]
- 14.Meyer WH, Spunt SL. Soft tissue sarcomas of childhood. Cancer Treat Rev. 2004;30:269–280. [DOI] [PubMed] [Google Scholar]
- 15.Rutqvist LE, Rose C, Cavallin-Stahl E. A systematic overview of radiation therapy effects in breast cancer. Acta Oncol. 2003;42:532–545. [DOI] [PubMed] [Google Scholar]
- 16.Yeh E. Cardiotoxicity induced by chemotherapy and antibody therapy. Ann Rev Med. 2006:485–498. [DOI] [PubMed]
- 17.Sklar CA, LaQuaglia MP. The long-term complications of chemotherapy in childhood genitourinary tumors. Urol Clin North Am. 2000;27:563–568. [DOI] [PubMed] [Google Scholar]
- 18.Marina N. Long-term survivors of childhood cancer: the medical consequences of cure. Pediatr Clin North Am. 1997;44:1021–1042. [DOI] [PubMed] [Google Scholar]
- 19.Dieckmann K, Widder J, Potter R. Long-term side effects of radiotherapy in survivors of childhood cancer. Front Radiat Ther Oncol. 2002;37:57–68. [DOI] [PubMed] [Google Scholar]
- 20.Oeffinger KC, Mertens AC, Sklar CA, et al. Chronic health conditions in adult survivors of childhood cancer. N Engl J Med. 2006;355:1572–1582. [DOI] [PubMed] [Google Scholar]
- 21.Lee JQ, Simmonds MJ, Wang XS, Novy DM. Differences in physical performance between men and women with and without lymphoma. Arch Phys Med Rehabil. 2003;84:1747–1752. [DOI] [PubMed] [Google Scholar]
- 22.Oeffinger KC, Mertens AC, Hudson MM, et al. Health care of young adult survivors of childhood cancer: a report from the Childhood Cancer Survivor Study. Ann Fam Med. 2004;2:61–70. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Oeffinger KC, Eshelman DA, Tomlinson GE, et al. Providing primary care for long-term survivors of childhood acute lymphoblastic leukemia. J Fam Pract. 2000;49:1133–1146. [PubMed] [Google Scholar]
- 24.International Classification of Functioning, Disability and Health: ICF. Geneva, Switzerland: World Health Organization; 2001.
- 25.American Physical Therapy Association. APTA Endorses ICF Model. PT Bulletin. Vol 9, issue 26, 2008. Available at: http://www.apta.org/AM/Template.cfm?Section=Archives2&Template=/Customsource/TaggedPage/PTIssue.cfm&Issue=06/17/2008#article49312.
- 26.Nagi S. A study in the evaluation of disability and rehabilitation potential: concepts, methods, and procedures. Am J Public Health Nations Health. 1964;54:1568–1579. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Nagi S. Some Conceptual Issues in Disability and Rehabilitation. Washington, DC: American Sociological Association; 1965.
- 28.Jette AM. Toward a common language for function, disability, and health. Phys Ther. 2006;86:726–734. [PubMed] [Google Scholar]
- 29.Nagi SZ. An epidemiology of disability among adults in the United States. Milbank Mem Fund Q Health Soc. 1976;54:439–467. [PubMed] [Google Scholar]
- 30.Nagi SZ. Congruency in medical and self-assessment of disability. IMS Ind Med Surg. 1969;38:27–36. [PubMed] [Google Scholar]
- 31.Quasthoff S, Hartung HP. Chemotherapy-induced peripheral neuropathy. J Neurol. 2002;249:9–17. [DOI] [PubMed] [Google Scholar]
- 32.Kesselring J, Coenen M, Cieza A, et al. Developing the ICF Core Sets for multiple sclerosis to specify functioning. Mult Scler. 2008;14:252–254. [DOI] [PubMed] [Google Scholar]
- 33.Geyh S, Cieza A, Schouten J, et al. ICF Core Sets for stroke. J Rehabil Med. 2004:135–141. [DOI] [PubMed]
- 34.Starrost K, Geyh S, Trautwein A, et al. Interrater reliability of the extended ICF Core Set for stroke applied by physical therapists. Phys Ther. 2008;88:841–851. [DOI] [PubMed] [Google Scholar]
- 35.Dreinhofer K, Stucki G, Ewert T, et al. ICF Core Sets for osteoarthritis. J Rehabil Med. 2004:75–80. [DOI] [PubMed]
- 36.Ruof J, Cieza A, Wolff B, et al. ICF Core Sets for diabetes mellitus. J Rehabil Med. 2004:100–106. [DOI] [PubMed]
- 37.Cieza A, Stucki G, Weigl M, et al. ICF Core Sets for low back pain. J Rehabil Med. 2004:69–74. [DOI] [PubMed]
- 38.Stucki A, Daansen P, Fuessl M, et al. ICF Core Sets for obesity. J Rehabil Med. 2004:107–113. [DOI] [PubMed]
- 39.Cieza A, Schwarzkopf S, Sigl T, et al. ICF Core Sets for osteoporosis. J Rehabil Med. 2004:81–86. [DOI] [PubMed]
- 40.Kirchberger I, Glaessel A, Stucki G, Cieza A. Validation of the comprehensive International Classification of Functioning, Disability and Health Core Set for rheumatoid arthritis: the perspective of physical therapists. Phys Ther. 2007;87:368–384. [DOI] [PubMed] [Google Scholar]
- 41.Uhlig T, Lillemo S, Moe RH, et al. Reliability of the ICF Core Set for rheumatoid arthritis. Ann Rheum Dis. 2007;66:1078–1084. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 42.Stamm T, Geyh S, Cieza A, et al. Measuring functioning in patients with hand osteoarthritis: content comparison of questionnaires based on the International Classification of Functioning, Disability and Health (ICF). Rheumatology (Oxford). 2006;45:1534–1541. [DOI] [PubMed] [Google Scholar]
- 43.Stucki A, Borchers M, Stucki G, et al. Content comparison of health status measures for obesity based on the International Classification of Functioning, Disability and Health. Int J Obes (Lond). 2006;30:1791–1799. [DOI] [PubMed] [Google Scholar]
- 44.Sigl T, Cieza A, Brockow T, et al. Content comparison of low back pain-specific measures based on the International Classification of Functioning, Disability and Health (ICF). Clin J Pain. 2006;22:147–153. [DOI] [PubMed] [Google Scholar]
- 45.Borchers M, Cieza A, Sigl T, et al. Content comparison of osteoporosis-targeted health status measures in relation to the International Classification of Functioning, Disability and Health (ICF). Clin Rheumatol. 2005;24:139–144. [DOI] [PubMed] [Google Scholar]
- 46.Cieza A, Geyh S, Chatterji S, et al. ICF linking rules: an update based on lessons learned. J Rehabil Med. 2005;37:212–218. [DOI] [PubMed] [Google Scholar]
- 47.Tschiesner U, Cieza A, Rogers SN, et al. Developing core sets for patients with head and neck cancer based on the International Classification of Functioning, Disability and Health (ICF). Eur Arch Otorhinolaryngol. 2007;264:1215–1222. [DOI] [PubMed] [Google Scholar]
- 48.Brach M, Cieza A, Stucki G, et al. ICF Core Sets for breast cancer. J Rehabil Med. 2004:121–127. [DOI] [PubMed]
- 49.Stucki G, Melvin J. The International Classification of Functioning, Disability and Health: a unifying model for the conceptual description of physical and rehabilitation medicine. J Rehabil Med. 2007;39:286–292. [DOI] [PubMed] [Google Scholar]
- 50.Finger ME, Cieza A, Stoll J, et al. Identification of intervention categories for physical therapy, based on the International Classification of Functioning, Disability and Health: a Delphi exercise. Phys Ther. 2006;86:1203–1220. [DOI] [PubMed] [Google Scholar]
- 51.Roach K. Measurement of health outcomes:reliability, validity and responsiveness. J Prosth Orthot. 2006;18:8–12. [Google Scholar]
- 52.Keith R, Granger C, Hamilton B, Sherwin F. The Functional Independence Measure: a new tool for rehabilitation. In: Eisenberg M, Grzesiak R, eds. Advances in Clinical Rehabilitation. New York, NY: Springer Publishing Co; 1987:6–18. [PubMed]
- 53.Portney L, Watkins M. Foundations of Clinical Research Applications to Practice. 2nd ed. Upper Saddle River, NJ: Prentice Hall Health; 2000.
- 54.Ahles T, Saykin A. Breast cancer chemotherapy-related cognitive dysfunction. Clin Breast Cancer. 2002;3:S84–S90. [DOI] [PubMed] [Google Scholar]
- 55.Barton D, Loprinizi C. Novel approaches to preventing chemotherapy-induced cognitive dysfunction in breast cancer: the art of the possible. Clin Breast Cancer. 2002;3:S121–S127. [DOI] [PubMed] [Google Scholar]
- 56.Castellon S, Ganz P, Bower J, et al. Neurocognitive performance in breast cancer survivors exposed to adjuvant chemotherapy and tamoxifen. J Clin Exp Neuropsychol. 2004;26:955–969. [DOI] [PubMed] [Google Scholar]
- 57.Dam FV, Schagen S, Muller M, et al. Impairment of cognitive function in women receiving adjuvant treatment for high-risk breast cancer: high-dose versus standard-dose chemotherapy. J Nat Cancer Inst. 1998;90:210–218. [DOI] [PubMed] [Google Scholar]
- 58.Freeman J, Broshek D. Assessing cognitive dysfunction in breast cancer: What are the tools? Clin Breast Cancer Suppl. 2002;3:91–99. [DOI] [PubMed] [Google Scholar]
- 59.Olin J. Cognitive function after systemic therapy for breast cancer. Oncology. 2001;15:613–618. [PubMed] [Google Scholar]
- 60.Rugo H, Ahles T. The impact of adjuvant therapy for breast cancer on cognitive function: current evidence and directions for research. Semin Oncol. 2003;30:749–762. [DOI] [PubMed] [Google Scholar]
- 61.Schagen S, Van Dam F, Muller M, et al. Cognitive deficits after postoperative adjuvant chemotherapy for breast carcinoma. Cancer. 1999;85:640–650. [DOI] [PubMed] [Google Scholar]
- 62.Stewart A, Bielajew C, Collins B, et al. A meta-analysis of the neuropsychological effects of adjuvant chemotherapy treatment in women treated for breast cancer. Clin Neuropsychol. 2006;20:76–89. [DOI] [PubMed] [Google Scholar]
- 63.Tchen N, Juffs H, Downie F, et al. Cognitive function, fatigue, and menopausal symptoms in women receiving adjuvant chemotherapy for breast cancer. J Clin Oncol. 2003;21:4175–4183. [DOI] [PubMed] [Google Scholar]
- 64.Wefel J, Lenxi R, Theriault R, et al. The cognitive sequelae of standard-dose adjuvant chemotherapy in women with breast carcinoma: results of a prospective, randomized, longitudinal trial. Cancer. 2004;100:2292–2299. [DOI] [PubMed] [Google Scholar]
- 65.Wieneke M, Dienst E. Neuropsychological assessment of cognitive functioning following chemotherapy for breast cancer. Psycho-oncology. 1995;4:61–66. [Google Scholar]
- 66.Paganini-Hill A, Clark L. Preliminary assessment of cognitive function in breast cancer patients treated with tamoxifen. Breast Cancer Res Treat. 2000;64:165–176. [DOI] [PubMed] [Google Scholar]
- 67.Ahles T, Saykin A, Furstenberg C, et al. Neuropsychologic impact of standard-dose systemic chemotherapy in long-term survivors of breast cancer and lymphoma. J Clin Oncol. 2002;20:485–493. [DOI] [PubMed] [Google Scholar]
- 68.O'Shaughnessy J. Chemotherapy-related cognitive dysfunction in breast cancer. Semin Oncol Nurs. 2003;19:17–24. [DOI] [PubMed] [Google Scholar]
- 69.O'Shaughnessy J. Chemotherapy-related cognitive dysfunction: a clearer picture. Clin Breast Cancer. 2003;4(suppl 2):S89–S84. [DOI] [PubMed] [Google Scholar]
- 70.Folstein M, Folstein S, McHugh P. Mini-Mental State: a practical method for grading the state of patients for the clinician J Psychiatr Res. 1975;12:189–198. [DOI] [PubMed] [Google Scholar]
- 71.Jansen C, Miaskowski C, Dodd M, Dowling G. A meta-analysis of the sensitivity of various neuropsychological tests uses to detect chemotherapay-induced cognitive impairment in patients with breast cancer. Oncol Nurs Forum. 2007;34:997–1005. [DOI] [PubMed] [Google Scholar]
- 72.Cella D, Tross S, Orov E, et al. Mood states of patients after the diagnosis of cancer. J Psychosoc Oncol. 1989;7:45–53. [Google Scholar]
- 73.McNair D, Loor M, Droppleman L. Profile of Mood States. San Diego, CA: Educational and Industrial Testing Service; 1971.
- 74.Sergi B, Ferraresi A, Troiani D, et al. Cisplatin ototoxicity in the guinea pig: vestibular and cochlear damage. Hear Res. 2003;182:56–64. [DOI] [PubMed] [Google Scholar]
- 75.FDA-Approved Label for Cisplatin. 2007. Available at: http://www.fda.gov/cder/ogd/rld/18057s68.pdf. Accessed November 26, 2008.
- 76.Verstappen CCP, Heimans JJ, Hoekman K, Postma TJ. Neurotoxic complications of chemotherapy in patients with cancer. Drugs. 2003;63:1549–1563. [DOI] [PubMed] [Google Scholar]
- 77.Chaudhry V, Rowinsky EK, Sartorius SE, et al. Peripheral neuropathy from taxol and cisplatin combination chemotherapy: clinical and electrophysiological studies. Ann Neurol. 1994;35:304–311. [DOI] [PubMed] [Google Scholar]
- 78.Cavaletti G, Jann S, Pace A, et al. Multi-center assessment of the Total Neuropathy Score for chemotherapy-induced peripheral neurotoxicity. J Peripher Nerv Syst. 2006;11:135–141. [DOI] [PubMed] [Google Scholar]
- 79.Wampler M, Miaskowski C, Hamel K, et al. The modified Total Neuropathy Score: a clinically feasible and valid measure of taxane-induced peripheral neuropathy in women with breast cancer. J Support Oncol. 2006;4:397–402. [Google Scholar]
- 80.Chaudhry V, Chaudhry M, Crawford TO, et al. Toxic neuropathy in patients with pre-existing neuropathy. Neurology. 2003;60:337–340. [DOI] [PubMed] [Google Scholar]
- 81.Cavaletti G, Bogliun G, Marzorati L, et al. Grading of chemotherapy-induced peripheral neurotoxicity using the Total Neuropathy Scale. Neurology. 2003;61:1297–1300. [DOI] [PubMed] [Google Scholar]
- 82.Stevens P, Dibble S, Miaskowski C. Prevalence, characteristics, and impact of postmastectomy pain syndrome: an investigation of women's experiences. Pain. 1995;61:61–68. [DOI] [PubMed] [Google Scholar]
- 83.Galecki J, Hicer-Grzenkowicz J, Grudzien-Kowalska M, et al. Radiation-induced brachial plexopathy and hypofractionated regimens in adjuvant irradiation of patients with breast cancer: a review. Acta Oncol. 2006;45:280–284. [DOI] [PubMed] [Google Scholar]
- 84.Cherny N. The management of cancer pain. CA Cancer J Clin. 2000;50:70–116. [DOI] [PubMed] [Google Scholar]
- 85.Serlin R, Mendoza T, Nakamura Y, et al. When is cancer pain mild, moderate or severe? Grading pain severity by its interference with function. Pain. 1995;61:277–284. [DOI] [PubMed] [Google Scholar]
- 86.Jensen M, Karoly P, Braver S. The measurement of clinical pain intensity: a comparison of six methods. Pain. 1986;27:117–126. [DOI] [PubMed] [Google Scholar]
- 87.Jensen M, Turner J, Romano J, Fisher L. Comparative reliability and validity of chronic pain intensity measures. Pain. 1999;83:157–162. [DOI] [PubMed] [Google Scholar]
- 88.Cleeland C. Measurement and prevalence of pain in cancer. Semin Oncol Nurs. 1985;1:87–92. [DOI] [PubMed] [Google Scholar]
- 89.Evans C, Trudeau E, Mertzanis P, et al. Development and validation of the Pain Treatment Satisfaction Scale (PTSS): a patient satisfaction questionnaire for use in patients with chronic or acute pain. Pain. 2004;112:254–266. [DOI] [PubMed] [Google Scholar]
- 90.Rietman JS, Dijkstra PU, Geertzen JH, et al. Short-term morbidity of the upper limb after sentinel lymph node biopsy or axillary lymph node dissection for stage I or II breast carcinoma. Cancer. 2003;98:690–696. [DOI] [PubMed] [Google Scholar]
- 91.Blomqvist L, Stark B, Engler N, Malm M. Evaluation of arm and shoulder mobility and strength after modified radical mastectomy and radiotherapy. Acta Oncol. 2004;43:280–283. [DOI] [PubMed] [Google Scholar]
- 92.Dijkstra PU, van Wilgen PC, Buijs RP, et al. Incidence of shoulder pain after neck dissection: a clinical explorative study for risk factors. Head Neck. 2001;23:947–953. [DOI] [PubMed] [Google Scholar]
- 93.Erisen L, Basel B, Irdesel J, et al. Shoulder function after accessory nerve-sparing neck dissections. Head Neck. 2004;26:967–971. [DOI] [PubMed] [Google Scholar]
- 94.Guldiken Y, Orhan KS, Demirel T, et al. Assessment of shoulder impairment after functional neck dissection: long term results. Auris Nasus Larynx. 2005;32:387–391. [DOI] [PubMed] [Google Scholar]
- 95.Inui A. Cancer anorexia-cachexia syndrome: current issues in research and management. CA Cancer J Clin. 2002;52:72–91. [DOI] [PubMed] [Google Scholar]
- 96.Owczarek J, Jasinska M, Orszulak-Michalak D. Drug-induced myopathies: an overview of possible mechanisms. Pharmacol Rep. 2005;57:23–34. [PubMed] [Google Scholar]
- 97.Mathiowetz V, Kashman N, Volland G, et al. Grip and pinch strength: normative data for adults. Arch Phys Med Rehabil. 1985;66:69–72. [PubMed] [Google Scholar]
- 98.Bohannon RW. Reference values for extremity muscle strength obtained by hand-held dynamometry from adults aged 20 to 79 years. Arch Phys Med Rehabil. 1997;78:26–32. [DOI] [PubMed] [Google Scholar]
- 99.Ochs BG, Simank HG, Kopp-Schneider A, et al. Gait analysis in limb preserving tumour surgery: kinematic gait patterns after resection of malignant bone tumours near the knee joint. Z Orthop Unfall. 2007;145:763–771. [DOI] [PubMed] [Google Scholar]
- 100.Benedetti MG, Catani F, Donati D, et al. Muscle performance about the knee joint in patients who had distal femoral replacement after resection of a bone tumor: an objective study with use of gait analysis. J Bone Joint Surg Am. 2000;82:1619–1625. [DOI] [PubMed] [Google Scholar]
- 101.Gerber L, Hoffman K, Chaudry U, et al. Functional outcomes and life satisfaction in long-term survivors of pediatric sarcoma. Arch Phys Med Rehabil. 2006;87:1611–1617. [DOI] [PubMed] [Google Scholar]
- 102.Choy N, Johnson N, Treleaven J, et al. Balance, mobility and gaze stability deficits remain following surgical removal of vestibular schwannoma (acoustic neuroma): an observational study Aust J Physiother. 2006;52:211–216. [DOI] [PubMed] [Google Scholar]
- 103.Syczewska M, Dembowska-Baginska B, Perek-Polnik M, Perek D. Functional status of children after treatment for a malignant tumor of the CNS: a preliminary report. Gait Posture. 2006;23:206–210. [DOI] [PubMed] [Google Scholar]
- 104.Wampler MA, Topp KS, Miaskowski C, et al. Quantitative and clinical description of postural instability in women with breast cancer treated with taxane chemotherapy. Arch Phys Med Rehabil. 2007;88:1002–1008. [DOI] [PubMed] [Google Scholar]
- 105.Holley S. A look at the problem of falls among people with cancer. Clin J Oncol Nurs. 2002;6:193–197. [DOI] [PubMed] [Google Scholar]
- 106.Camp-Sorrell D. Cardiorespiratory effects in cancer survivors. Am J Nurs. 2006;106:55–59. [DOI] [PubMed] [Google Scholar]
- 107.Floyd J, Nguyen D, Lobins R, et al. Cardiotoxicity of cancer therapy. J Clin Oncol. 2005;23:7685–7696. [DOI] [PubMed] [Google Scholar]
- 108.Harris E, Correa C, Hwang W, et al. Late cardiac mortality and morbidity in early-stage breast cancer patients after breast conservation treatment. J Clin Oncol. 2006;24:4100–4106. [DOI] [PubMed] [Google Scholar]
- 109.Limper A. Chemotherapy induced lung disease. Clin Chest Med. 2004;25:53–64. [DOI] [PubMed] [Google Scholar]
- 110.Butland R, Pang J, Gross E, et al. Two-, six-, and twelve-minute walking tests in respiratory disease. BMJ. 1982;284:1607–1608. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 111.Medical Research Council. Instructions for Use of the Questionaire on Respiratory Symptoms. Dawlish, England: WJ Holman; 1966.
- 112.Borg G. Psychophysical bases of perceived exertion. Med Sci Sports Exerc. 1982;4:377–381. [PubMed] [Google Scholar]
- 113.Dimeo F, Schmittel A, Fietz T, et al. Physical performance, depression, immune status and fatigue in patients with hematological malignancies after treatment. Ann Oncol. 2004;15:1237–1242. [DOI] [PubMed] [Google Scholar]
- 114.Cramp F, Daniel J. Exercise for the management of cancer-related fatigue in adults. Cochrane Database Syst Rev. 2008:CD006145. [DOI] [PubMed]
- 115.Bruera E, Strasser F, Shen L, et al. The effect of donepezil on sedation and other symptoms in patients receiving opioids for cancer pain: a pilot study. J Pain Symptom Manage. 2003;26:1049–1054. [DOI] [PubMed] [Google Scholar]
- 116.Mulrooney DA, Ness KK, Neglia JP, et al. Fatigue and sleep disturbance in adult survivors of childhood cancer: a report from the Childhood Cancer Survivor Study (CCSS). Sleep. 2008;31:271–281. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 117.Taylor R, Jayasinghe UW, Koelmeyer L, et al. Reliability and validity of arm volume measurements for assessment of lymphedema. Phys Ther. 2006;86:205–214. [PubMed] [Google Scholar]
- 118.Karges JR, Mark BE, Stikeleather SJ, Worrell TW. Concurrent validity of upper-extremity volume estimates: comparison of calculated volume derived from girth measurements and water displacement volume. Phys Ther. 2003;83:134–145. [PubMed] [Google Scholar]
- 119.Megens AM, Harris SR, Kim-Sing C, McKenzie DC. Measurement of upper extremity volume in women after axillary dissection for breast cancer. Arch Phys Med Rehabil. 2001;82:1639–1644. [DOI] [PubMed] [Google Scholar]
- 120.Stout-Gerich NL, Pfalzer LA, McGarvey C, et al. Preoperative assessment enables the early diagnosis and successful treatment of lymphedema. Cancer Invest. 2008;112:2809–2019. [DOI] [PubMed] [Google Scholar]
- 121.Common Terminology Criteria for Adverse Events, version 3.0 (CTCAE v3.0). Available at: http://ctep.cancer.gov/forms/CTCAEv3.pdf. Accessed March 14, 2007.
- 122.Jaeckle KA. Neurological manifestations of neoplastic and radiation-induced plexopathies. Semin Neurol. 2004;24:385–393. [DOI] [PubMed] [Google Scholar]
- 123.Lowey SE. Spinal cord compression: an oncologic emergency associated with metastatic cancer: evaluation and management for the home health clinician. Home Healthc Nurse. 2006;24:439–446. [DOI] [PubMed] [Google Scholar]
- 124.Hipp J, Springfield D, Hayes W. Predicting pathologic fracture risk in the management of metastatic bone defects. Clin Orthop Relat Res. 1995:120–135. [PubMed]
- 125.Rades D, Veninga T, Stalpers L, et al. Improved posttreatment functional outcomes is associated with better survival in patients irradiated for metastatic spinal cord compression. Int J Radiat Oncol Biol Phys. 2007;67:1506–1509. [DOI] [PubMed] [Google Scholar]
- 126.Majhail N, Ness K, Burns L, et al. Late effects in survivors of Hodgkin and non-Hodgkin lymphoma: a report from the Bone Marrow Transplant Survivor Study. Biol Blood Marrow Transplant. 2007;13:1153–1159. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 127.Friedrich R, Blake F. Avascular mandibular osteonecrosis in association with bisphosphonate therapy: a report on four patients. Anticancer Res. 2007;27:1841–1845. [PubMed] [Google Scholar]
- 128.Marchese VG, Connolly B, Able C, et al. Relationships among severity of osteonecrosis, pain, range of motion, and functional mobility in children, adolescents, and young adults with acute lymphoblastic leukemia. Phys Ther. 2008;88:341–350. [DOI] [PubMed] [Google Scholar]
- 129.Goodman C, Snyder T. Differential Diagnosis for Physical Therapists. 4th ed. St Louis, MO: Elsevier; 2007.
- 130.Piech D, Sanders B, Ghazinouri R, et al. Lab values interpretation resources. Available at: http://www.acutept.org/labvalues.pdf. Accessed October 2, 2008.
- 131.Perenboom RJ, Chorus AM. Measuring participation according to the International Classification of Functioning, Disability and Health (ICF). Disability Rehabil. 2003;25:577–587. [DOI] [PubMed] [Google Scholar]
- 132.Badley EM. Enhancing the conceptual clarity of the activity and participation components of the International Classification of Functioning, Disability and Health. Soc Sci Med. 2008;66:2335–2345. [DOI] [PubMed] [Google Scholar]
- 133.Marchese VG, Rai SN, Carlson CA, et al. Assessing functional mobility in survivors of lower-extremity sarcoma: reliability and validity of a new assessment tool. Pediatr Blood Cancer. 2007;49:183–189. [DOI] [PubMed] [Google Scholar]
- 134.Davis AM, Sennik S, Griffin AM, et al. Predictors of functional outcomes following limb salvage surgery for lower-extremity soft tissue sarcoma. J Surg Oncol. 2000;73:206–211. [DOI] [PubMed] [Google Scholar]
- 135.Davis AM, Wright JG, Williams JI, et al. Development of a measure of physical function for patients with bone and soft tissue sarcoma. Qual Life Res. 1996;5:508–516. [DOI] [PubMed] [Google Scholar]
- 136.Montoya M, Fossella F, Palmer J, et al. Objective evaluation of physical function in patients with advanced lung cancer: a preliminary report. J Palliat Med. 2006;9:309–316. [DOI] [PubMed] [Google Scholar]
- 137.Lipscomb J, Gotay CC, Snyder CF. Patient-reported outcomes in cancer: a review of recent research and policy initiatives. CA Cancer J Clin. 2007;57:278–300. [DOI] [PubMed] [Google Scholar]
- 138.Hwang SC, Scot CB, Chang VT, et al. Predition of survival for advanced cancer patients by recursive portioning analysis: role of Karnofsky performance status, quality of life and symptom distress. Cancer Invest 2004;22:678–687. [DOI] [PubMed] [Google Scholar]
- 139.Hassan SJ, Weymuller EA Jr. Assessments of quality of life in head and neck cancer. Head Neck. 1993;15:485–496. [DOI] [PubMed] [Google Scholar]
- 140.Kassa T, Wessel J. The Edmonton Functional Assessment Tool: further development and validation for use in palliative care. J Palliat Care. 2001;17:5–11. [PubMed] [Google Scholar]
- 141.Mahoney F, Barthel D. Functional evaluation: the Barthel Index. MD State Med J. 1965;14:61–65. [PubMed] [Google Scholar]
- 142.Larson S, Lakin K, Anderson L, et al. Prevalence of mental retardation and developmental disabilities: estimates from the 1994/1995 national health interview survey disability supplements. Am J Ment Retard. 2001;106:231–252. [DOI] [PubMed] [Google Scholar]
- 143.Lenze E, Rogers J, Martire L, et al. The association of late-life depression and anxiety with physical disability: a review of the literature and prospectus for future research. Am J Geriatr Psychiatry. 2001;9:113–135. [PubMed] [Google Scholar]
- 144.Ness K, Mertens A, Hudson M, et al. Limitations on physical performance and daily activities among long-term survivors of childhood cancer. Ann Int Med. 2006;143:639–647. [DOI] [PubMed] [Google Scholar]
- 145.Wood-Dauphinée SL, Opzoomer M, Williams J, et al. Assessment of global function: the Reintegration to Normal Living Index. Arch Phys Med Rehabil. 1988;69:583–590. [PubMed] [Google Scholar]
- 146.Schreiber D, Bell R, Wunder J, et al. Evaluating function and health-related quality of life in patients treated for extremity soft tissue sarcoma. Qual Life Res. 2006;15:1439–1446. [DOI] [PubMed] [Google Scholar]
- 147.Tunn PU, Pomraenke D, Goerling U, Hohenberger P. Functional outcome after endoprosthetic limb-salvage therapy of primary bone tumours: a comparative analysis using the MSTS score, the TESS and the RNL index. Int Orthop. 2008;32:619–625. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 148.Faust D, Fogel BS. The development and initial validation of a sensitive bedside cognitive screening test. J Nerv Ment Dis. 1989;177:25–31. [DOI] [PubMed] [Google Scholar]
- 149.Shilling V, Jenkins V. Impact of androgen deprivation therapy on physical and cognitive function, as well as quality of life of patients with nonmetastatic prostate cancer. J Urol. 2006;176:2443–2447. [DOI] [PubMed] [Google Scholar]
- 150.Meyers CA, Wefel JS. The Use of the Mini-Mental State Examination to Assess Cognitive Functioning in Cancer Trials: No Ifs, Ands, Buts, or Sensitivity. J Clin Oncol. 2003;21:3557–3558. [DOI] [PubMed] [Google Scholar]
- 151.Jacobs SR, Jacobsen PB, Booth-Jones M, et al. Evaluation of the Functional Assessment of Cancer Therapy Cognitive Scale with hematopoetic stem cell transplant patients. J Pain Symptom Manage. 2007;33:13–23. [DOI] [PubMed] [Google Scholar]
- 152.Galantino M, Brown D, Stricker C, Farrar J. Development and testing of a cancer cognition questionnaire. Rehab Oncol. 2006;24:15–22. [Google Scholar]
- 153.Ahles T, Silberfarb P, Herndon Jn, et al. Psychological adjustment of survivors of localised prostate cancer: investigating the role of dyadic adjustment, cognitive appraisal and coping style. Psychooncology. 2007;16:994–1002. [DOI] [PubMed] [Google Scholar]
- 154.Brown P, Clark M, Atherton P, et al. Will improvement in quality of life (QOL) impact fatigue in patients receiving radiation therapy for advanced cancer? Am J Clin Oncol. 2006;29:52–58. [DOI] [PubMed] [Google Scholar]
- 155.Hack T, Degner L. Coping responses following breast cancer diagnosis predict psychological adjustment three years later. Psychooncology. 2004;13:235–247. [DOI] [PubMed] [Google Scholar]
- 156.Brezden C, Phillips K, Abdolell M, et al. Cognitive function in breast cancer patients receiving adjuvant chemotherapy. J Clin Oncol. 2000;18:2695–2701. [DOI] [PubMed] [Google Scholar]
- 157.Ahles T, Silberfarb P, Herndon J, et al. Psychologic and neuropsychologic functioning of patients with limited small-cell lung cancer treated with chemotherapy and radiation therapy with or without warfarin: a study by the Cancer and Leukemia Group B. J Clin Oncol. 1998;16:1954–1960. [DOI] [PubMed] [Google Scholar]
- 158.Kurnatowski P, Putyński L, Piotrowski S. The evaluation of certain psychological indices concerning cognitive and emotional behaviors in patients after radical neck dissection. Otolaryngol Pol. 1997;51:58–63. [PubMed] [Google Scholar]
- 159.Jacobson G, Newman C, Hunter L, Balzer G. Balance function test correlates of the Dizziness Handicap Inventory. J Am Acad Audiol. 1991;2:253–260. [PubMed] [Google Scholar]
- 160.Humphriss R, Baguley D, Moffat D. Change in dizziness handicap after vestibular schwannoma excision. Otol Neurotol. 2003;24:661–665. [DOI] [PubMed] [Google Scholar]
- 161.Enticott J, O'Leary S, Briggs R. Effects of vestibulo-ocular reflex exercises on vestibular compensation after vestibular schwannoma surgery. Otol Neurotol. 2005;26:265–269. [DOI] [PubMed] [Google Scholar]
- 162.Roberts RA, Gans RE, Johnson EL, Chisolm TH. Computerized dynamic visual acuity with volitional head movement in patients with vestibular dysfunction. Ann Otol Rhinol Laryngol. 2006;115:658–666. [DOI] [PubMed] [Google Scholar]
- 163.Goebel JA, Tungsiripat N, Sinks B, Carmody J. Gaze stabilization test: a new clinical test of unilateral vestibular dysfunction. Otol Neurotol. 2007;28:68–73. [DOI] [PubMed] [Google Scholar]
- 164.Shy ME, Frohman EM, So YT, et al. Quantitative sensory testing: report of the Therapeutics and Technology Assessment Subcommittee of the American Academy of Neurology. Neurology. 2003;60:898–904. [DOI] [PubMed] [Google Scholar]
- 165.Bloom S, Till S, Sonksen P, Smith S. Use of a biothesiometer to measure individual vibration thresholds and their variation in 519 non-diabetic subjects. Br Med J (Clin Res Ed). 1984;288:1793–1795. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 166.Scott J, Huskisson E. Graphic representation of pain. Pain. 1976;2:175–184. [PubMed] [Google Scholar]
- 167.Camps C, Caballero C, Blasco A, et al. Weekly paclitaxel as second/third-line treatment in advanced non-small cell lung cancer patients: efficacy and tolerability. Anticancer Res. 2005;25:4611–4614. [PubMed] [Google Scholar]
- 168.Zernikow B, Meyerhoff U, Michel E, et al. Pain in pediatric oncology: children's and parents’ perspectives. Eur J Pain. 2005;9:395–406. [DOI] [PubMed] [Google Scholar]
- 169.Zernikow B, Smale H, Michel E, et al. Paediatric cancer pain management using the WHO analgesic ladder: results of a prospective analysis from 2,265 treatment days during a quality improvement study. Eur J Pain. 2006;10:587–595. [DOI] [PubMed] [Google Scholar]
- 170.Villars P, Dodd M, West C, et al. Differences in the prevalence and severity of side effects based on type of analgesic prescription in patients with chronic cancer pain. J Pain Symptom Manage. 2007;33:67–77. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 171.Hicks C, von Bayer C, Spafford P, et al. The faces pain scale-revised:toward a common metric in pediatric pain measurement. Pain. 2001;93:173–183. [DOI] [PubMed] [Google Scholar]
- 172.Caraceni A, Portenoy R; a working group of the IASP Task Force on Cancer Pain. An international survey of cancer pain characteristics and syndromes. Pain. 1999;82:263–274. [DOI] [PubMed] [Google Scholar]
- 173.Borden L, Clark P, Lovato J, et al. Vinorelbine, doxorubicin, and prednisone in androgen-independent prostate cancer. Cancer. 2006;107:1093–1100. [DOI] [PubMed] [Google Scholar]
- 174.Miaskowski C, Dodd M, West C, et al. The use of a responder analysis to identify differences in patient outcomes following a self-care intervention to improve cancer pain management. Pain. 2007;129:55–63. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 175.Joint Motion: Method of Measuring and Recording. Chicago, IL: American Academy of Orthopaedic Surgeons; 1965.
- 176.Reese N, Bandy W. Joint Range of Motion and Muscle Length Testing. Philadelphia, PA: WB Saunders Co; 2002.
- 177.Leidenius M, Leppanen E, Krogerus L, von Smitten K. Motion restriction and axillary web syndrome after sentinel node biopsy and axillary clearance in breast cancer. Am J Surg. 2003;185:127–130.12559441 [Google Scholar]
- 178.Marchese VG, Chiarello L. Relationships between specific measures of body function, activity, and participation in children with acute lymphoblastic leukemia. Rehabil Oncol. 2004;22:5–9. [Google Scholar]
- 179.Paulino A. Late effects of radiotherapy for pediatric extremity sarcomas. Int J Rad Oncol Biol Phys. 2004;60:265–274. [DOI] [PubMed] [Google Scholar]
- 180.Golding L, Myers C, Sinning W. Y's Way to Physical Fitness. Champaign, IL: Human Kinetics Inc; 1989.
- 181.Kolden G, Straiam T, Ward A, et al. A pilot study of group exercise training for women with primary breast cancer: feasibility and health benefits. Psychooncology. 2006;11:447–456. [DOI] [PubMed] [Google Scholar]
- 182.Ness KK, Baker KS, Dengel D, et al. Body composition, muscle strength deficits and mobility limitations in adult survivors of childhood acute lymphoblastic leukemia. Pediatr Blood Cancer. 2007;49:975–981. [DOI] [PubMed] [Google Scholar]
- 183.Hayes S, Battistutta D, Newman B. Objective and subjective upper body function six months following diagnosis of breast cancer. Breast Cancer Res Treat. 2005;94:1–10. [DOI] [PubMed] [Google Scholar]
- 184.Rietman J, Geertzen J, Hoekstra H, et al. Long-term treatment-related upper limb morbidity and quality of life after sentinal lymph node biopsy for stage I or II breast cancer. Eur J Surg Oncol. 2006;32:148–152. [DOI] [PubMed] [Google Scholar]
- 185.Siegel AB, Lachs M, Coleman M, Leonard JP. Lymphoma in elderly patients: novel functional assessment techniques provide better discrimination among patients than traditional performance status measures. Clin Lymphoma Myeloma. 2006;7:65–69. [DOI] [PubMed] [Google Scholar]
- 186.Abu-Rustum N, Alektiar K, Iasonos A, et al. The incidence of symptomatic lower-extremity lymphedema following treatment of uterine corpus malignancies: a 12-year experience at Memorial Sloan-Kettering Cancer Center. Gynecol Oncol. 2006;103:714–718. [DOI] [PubMed] [Google Scholar]
- 187.Monsell EM, Furman JM, Herdman SJ, et al. Computerized dynamic platform posturography. Otolaryngol Head Neck Surg. 1997;117:394–398. [DOI] [PubMed] [Google Scholar]
- 188.Gouveris H, Akkafa S, Lippold R, Mann W. Influence of nerve of origin and tumor size of vestibular schwannoma on dynamic posturography findings. Acta Otolaryngol. 2006;126:1281–1285. [DOI] [PubMed] [Google Scholar]
- 189.Bergson E, Sataloff R. Preoperative computerized dynamic posturography as a prognostic indicator of balance function in patients with acoustic neuroma. Ear Nose Throat J. 2005;84:154–156. [PubMed] [Google Scholar]
- 190.Galvao D, Nosaka K, Taaffe D, et al. Resistance training and reduction of treatment side effects in prostate cancer patients. Med Sci Sports Exerc. 2006;38:2045–2052. [DOI] [PubMed] [Google Scholar]
- 191.Konczak J, Schoch B, Dimitrova A, et al. Functional recovery of children and adolescents after cerebellar tumour resection. Brain. 2005;128:1428–1441. [DOI] [PubMed] [Google Scholar]
- 192.American College of Sports Medicine. ACSM's Guidelines for Exercise Testing and Prescription. Philadelphia, PA: Lippincott Williams & Wilkins; 2000.
- 193.de Miguel Sanchez C, Elustondo S, Estirado A, et al. Palliative performance status, heart rate and respiratory rate as predictive factors of survival time in terminally ill cancer patients. J Pain Symptom Manage. 2006;31:485–492. [DOI] [PubMed] [Google Scholar]
- 194.Haddy T, Mosher R, Reaman G. Hypertension and pre-hypertension in long-term survivors of childhood and adolescent cancer. Pediatr Blood Cancer. 2007;49:79–83. [DOI] [PubMed] [Google Scholar]
- 195.Kourti M, Tragiannidis A, Makedou A, et al. Metabolic syndrome in children and adolescents with acute lymphoblastic leukemia after the completion of chemotherapy. J Pediatr Hematol Oncol. 2005;27:499–501. [DOI] [PubMed] [Google Scholar]
- 196.Sagstuen H, Aass N, Fossa S, et al. Blood pressure and body mass index in long-term survivors of testicular cancer. J Clin Oncol. 2005;23:4980–4990. [DOI] [PubMed] [Google Scholar]
- 197.Pietila S, Ala-Houhala M, Lenko H, et al. Renal impairment and hypertension in brain tumor patients treated in childhood are mainly associated with cisplatin treatment. Pediatr Blood Cancer. 2005;44:363–369. [DOI] [PubMed] [Google Scholar]
- 198.Dudgeon D, Lertzman M, Askew G. Physiologic changes and clinical correlations of dyspnea in cancer outpatients. J Pain Symptom Manage. 2001;21:373–379. [DOI] [PubMed] [Google Scholar]
- 199.Nomori H, Watanabe K, Ohtsuka T, et al. Six-minute walk and pulmonary function test outcomes during the early period after lung cancer surgery with special reference to patients with chronic obstructive pulmonary disease. Jpn J Thorac Cardiovasc Surg. 2004;52:113–119. [DOI] [PubMed] [Google Scholar]
- 200.Weiner D, Maity A, Carlson C, Ginsberg J. Pulmonary function abnormalities in children treated with whole lung irradiation. Pediatr Blood Cancer. 2006;46:222–227. [DOI] [PubMed] [Google Scholar]
- 201.Bossi G, Cerveri I, Volpini E, et al. Long-term pulmonary sequelae after treatment of childhood Hodgkin's disease. Ann Oncol. 1997;8:19–24. [PubMed] [Google Scholar]
- 202.Herrero F, San Juan A, Fleck S, et al. Effects of detraining on the functional capacity of previously trained breast cancer survivors. Int J Sports Med. 2007;28:257–264. [DOI] [PubMed] [Google Scholar]
- 203.Herrero F, San Juan A, Fleck S, et al. Combined aerobic and resistance training in breast cancer survivors: a randomized, controlled pilot trial. Int J Sports Med. 2006;27:573–580. [DOI] [PubMed] [Google Scholar]
- 204.Cheema B, Gaul C. Full-body exercise training improves fitness and quality of life in survivors of breast cancer. J Strength Cond Res. 2006;20:14–21. [DOI] [PubMed] [Google Scholar]
- 205.Hlatky M, Boineau R, Higginbotham M, et al. A brief self-administered questionnaire to determine functional capacity (the Duke Activity Status Index). Am J Cardiol. 1989;64:651–654. [DOI] [PubMed] [Google Scholar]
- 206.Health Related Physical Fitness: Test Manual. Reston, VA: American Alliance for Health, Physical Education, Recreation and Dance; 1980.
- 207.Marchese V, Ogle S, Womer R, et al. An examination of outcome measures to asess functional mobility in childhood survivors of osteosarcoma. Pediatr Blood Cancer. 2004;42:41–45. [DOI] [PubMed] [Google Scholar]
- 208.Smets E, Garssen B, Bonke B, et al. The Multidimensional Fatigue Inventory (MFI) psychometric qualities of an instrument to assess fatigue. J Psychosom Res. 1995;39:315–325. [DOI] [PubMed] [Google Scholar]
- 209.Jereczek-Fossa B, Santoro L, Alterio D, et al. Fatigue during head-and-neck radiotherapy: prospective study on 117 consecutive patients. Int J Radiat Oncol Biol Phys. 2007;68:403–415. [DOI] [PubMed] [Google Scholar]
- 210.Yellen S, Cella D, Webster K, et al. Measuring fatigue and other anemia-related symptoms with the Functional Assessment of Cancer Therapy (FACT) measurement system. J Pain Symptom Manage. 1997;13:63–74. [DOI] [PubMed] [Google Scholar]
- 211.Piper B, Dibble S, Dodd M, et al. The revised Piper Fatigue Scale: psychometric evaluation in women with breast cancer. Oncol Nurs Forum. 1998;25:677–684. [PubMed] [Google Scholar]
- 212.Meeske K, Siegel S, Globe D, et al. Prevalence and correlates of fatigue in long-term survivors of childhood leukemia. J Clin Oncol. 2005;23:5501–5510. [DOI] [PubMed] [Google Scholar]
- 213.Berger A, Farr L, Kuhn B, et al. Values of sleep/wake, activity/rest, circadian rhythms, and fatigue prior to adjuvant breast cancer chemotherapy. J Pain Symptom Manage. 2007;33:398–409. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 214.Mendoza T, Wang X, Cleeland C, et al. The rapid assessment of fatigue severity in cancer patients: use of the Brief Fatigue Inventory. Cancer. 1999;85:1186–1196. [DOI] [PubMed] [Google Scholar]
- 215.Wang X, Giralt S, Mendoza T, et al. Clinical factors associated with cancer-related fatigue in patients being treated for leukemia and non-Hodgkin's lymphoma. J Clin Oncol. 2002;20:1319–1328. [DOI] [PubMed] [Google Scholar]
- 216.Wang X, Janjan N, Guo H, et al. Fatigue during preoperative chemoradiation for resectable rectal cancer. Cancer. 2001;92:1725–1732. [DOI] [PubMed] [Google Scholar]
- 217.Stanton AW, Northfield JW, Holroyd B, et al. Validation of an optoelectric limb volumeter (Perometer). Lymphology. 1997;30:77–97. [PubMed] [Google Scholar]
- 218.Ratliff J, Cooper P. Metastatic spine tumors. South Med J. 2004;97:246–253. [DOI] [PubMed] [Google Scholar]
- 219.Maniadaki I, Stiakaki E, Germankis I, Kalmanti M. Evaluation of bone mineral density at different phases of therapy for childhood ALL. Pediatr Hematol Oncol. 2006;23:11–18. [DOI] [PubMed] [Google Scholar]
- 220.Karavatas SG, Reicherter A, White N, Strong A. Physical therapy management of patients with multiple myeloma: musculoskeletal considerations. Rehab Oncol. 2006;24:11–16. [Google Scholar]
- 221.Dimeo F, Tilmann M, Bertz H, et al. Aerobic exercise in the rehabilitation of cancer patients after high dose chemotherapy and autologous peripheral stem cell transplantation. Cancer. 1997;79:1717–1722. [PubMed] [Google Scholar]
- 222.Elbl L, Vasova I, Kral Z, et al. Evaluation of acute and early cardiotoxicity in survivors of Hodgkin's disease treated with ABVD or BEACOPP regimens. J Chemother. 2006;18:199–208. [DOI] [PubMed] [Google Scholar]
- 223.Dang C, Fornier M, Sugarman S, et al. The safety of dose-dense doxorubicin and cyclophosphamide followed by paclitaxel with trastuzumab in HER-2/neu overexpressed/amplified breast cancer. J Clin Oncol. 2008;26:1216–1222. [DOI] [PubMed] [Google Scholar]
- 224.Whitney SL, Wrisley DM, Marchetti GF, et al. Clinical measurement of sit-to-stand performance in people with balance disorders: validity of data for the Five-Times-Sit-to-Stand Test. Phys Ther. 2005;85:1034–1045. [PubMed] [Google Scholar]
- 225.Duncan P, Weiner D, Chandler J, Studenski S. Functional reach: a new clinical measure of balance. J Gerontol. 1990;45:M192–M197. [DOI] [PubMed] [Google Scholar]
- 226.Oldervoll LM, Loge JH, Paltiel H, et al. The effect of a physical exercise program in palliative care: a phase II study. J Pain Symptom Manage. 2006;31:421–430. [DOI] [PubMed] [Google Scholar]
- 227.Berg K, Wood-Dauphinée SL, Williams J, Gayton D. Measuring balance in the elderly: preliminary development of an instrument. Physiother Can. 1989;41:304–311. [Google Scholar]
- 228.Berg K, Maki B, Williams J, et al. Clinical and laboratory measures of postural balance in an elderly population. Arch Phys Med Rehabil. 1992;73:1073–1080. [PubMed] [Google Scholar]
- 229.Shumway-Cook A, Woollacott MH. Motor Control: Theory and Practical Applications. 2nd ed. Baltimore, MD: Lippincott Williams & Wilkins; 2001.
- 230.Whitney SL, Wrisley DM, Furman J. Concurrent validity of the Berg Balance Scale and the Dynamic Gait Index in people with vestibular dysfunction. Physiother Res Int. 2003;8:178–186. [DOI] [PubMed] [Google Scholar]
- 231.Tinetti M. Tinetti performance-oriented mobility assessment in elderly patients. J Am Geriatr Soc. 1986;34:119–126. [DOI] [PubMed] [Google Scholar]
- 232.Podsiadlo D, Richardson S. The timed “up and go”: a test of basic functional mobility for frail elderly persons. J Am Geriatr Soc. 1991;39:142–148. [DOI] [PubMed] [Google Scholar]
- 233.Marchese VG, Chiarello L, Lange B. Effects of physical therapy intervention for children with acute lymphoblastic leukemia. Pediatr Blood Cancer. 2004;42:127–133. [DOI] [PubMed] [Google Scholar]
- 234.Deathe AB, Miller WC. The L Test of Functional Mobility measurement properties of a modified version of the Timed “Up & Go” Test designed for people with lower-extremity amputations. Phys Ther. 2005;85:626–635. [PubMed] [Google Scholar]
- 235.Davis A, Devlin M, Griffin A, et al. Functional outcome in amputation versus limb sparing of patients with lower extremity sarcoma: a matched case-control study Arch Phys Med Rehabil. 1999;80:615–618. [DOI] [PubMed] [Google Scholar]
- 236.Nagarajan R, Clohisy D, Neglia J, et al. Function and quality-of-life of survivors of pelvic and lower extremity osteosarcoma and Ewing's sarcoma: the Childhood Cancer Survivor Study. Br J Cancer. 2004;91:1858–1865. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 237.Rose DJ. Fall Proof! A Comprehensive Balance and Mobility Training Program. Champaign, IL: Human Kinetics Inc; 2003.
- 238.Dietz JC, Kartin D, Kopp K. Review of the Bruininks-Oseretsky Test of Motor Proficiency. Phys Occup Ther Pediatr. 2007;27:87–102. [PubMed] [Google Scholar]
- 239.Wright M, Halton J, Martin R, Barr R. Long-term gross motor performance following treatment for acute lymphoblastic leukemia. Med Pediatr Oncol. 1998;31:86–90. [DOI] [PubMed] [Google Scholar]
- 240.Russell D, Rosenbaum P, Cadman D, et al. The Gross Motor Function Measure: a means to evaluate the effect of therapy. Dev Med Child Neurol. 1989;31:341–352. [DOI] [PubMed] [Google Scholar]
- 241.Palisano RJ, Kolobe TH, Haley SM, et al. Validity of the Peabody Developmental Gross Motor Scale as an evaluative measure of infants receiving physical therapy. Phys Ther. 1995;75:939–951. [DOI] [PubMed] [Google Scholar]
- 242.MacLean WJ, Noll R, Stehbens J, et al; the Children's Cancer Group. Neuropsychological effects of cranial irradiation in young children with acute lymphoblastic leukemia 9 months after diagnosis. Arch Neurol. 1995;52:156–160. [DOI] [PubMed] [Google Scholar]
- 243.Crisp J, Ungerer J, Goodnow J. The impact of experience on children's understanding of illness. J Pediatr Psychol. 1996;21:57–72. [DOI] [PubMed] [Google Scholar]
- 244.Aass N, Fossa S. Pre- and post-treatment daily life function in patients with hormone-resistant prostate carcinoma treated with radiotherapy for spinal cord compression. Radiother Oncol. 2005;74:259–265. [DOI] [PubMed] [Google Scholar]
- 245.Bennett M, Ryall N. Using the modified Barthel index to estimate survival in cancer patients in hospice: observational study. BMJ. 2000;321:1381–1382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 246.Yoshioka H. Rehabilitation for the terminal cancer patient. Am J Phys Med Rehabil. 1994;73:199–206. [DOI] [PubMed] [Google Scholar]
- 247.Osoba D, Aaronson N, Muller M, et al. Effect of neurological dysfunction on health-related quality of life in patients with high-grade glioma. J Neurooncol. 1997;34:263–278. [DOI] [PubMed] [Google Scholar]
- 248.Reuben D, Siu A. An objective measure of physical function of elderly outpatients. J Am Geriatr Soc. 1990;38:1105–1112. [DOI] [PubMed] [Google Scholar]
- 249.Smith D, Ehde D, Hanley M. Efficacy of gabapentin in treating chronic phantom limb and residual limb pain. J Rehabil Res. 2005;42:645–654. [DOI] [PubMed] [Google Scholar]
- 250.Huang M, Wartella J, Kreutzer J. Functional outcomes and quality of life in patients with brain tumor: a preliminary report. Arch Phys Med Rehabil. 2001;82:1540–1546. [DOI] [PubMed] [Google Scholar]
- 251.Yates JW, Chalmer B, McKegney FP. Evaluation of patients with advanced cancer using the Karnofsky performance status. Cancer. 1980;45:2220–2224. [DOI] [PubMed] [Google Scholar]
- 252.Bergner M, Bobbitt R, Carter W, et al. The Sickness Impact Profile: development and final revision of a health status measure. Med Care. 1981;19:787–805. [DOI] [PubMed] [Google Scholar]
- 253.De Bruin A, de Witte L, Stevens F, et al. Sickness Impact Profile: the state of the art of a generic functional status measure. Soc Sci Med. 1992;35:1003–1014. [DOI] [PubMed] [Google Scholar]