Abstract
This study was conducted to develop a noninvasive marker of hepatic microvesicular lipid accumulation (MVLA), a histopathological effect currently diagnosed in humans following liver biopsy. MVLA is detected in animal studies of chemicals and drugs and occurs in some humans exposed to chemicals or pharmaceuticals. Because MVLA is a reversible histopathology, early detection of MVLA using a noninvasive method, could aid clinicians in the treatment of patients taking drugs that are known to induce this injury. Isoniazid (INH) was selected as a model compound for this investigation, because MVLA occurs in tuberculosis (TB) patients treated with a combination therapy, which includes INH. This study used male rats dosed daily with INH at 0, 10, or 300 mg/kg/day for up to 8 days. Urine, blood, and liver were obtained following 1 and 8 days. NMR metabolomics of urine revealed markers that correlated (100%) with the findings of MVLA in the right, left, and median liver lobes in 4/9 rats administered the high dose of INH for 8 days. Metabolomics of liver extracts also revealed markers that correlated with the MVLA injury. Serum enzymes that are clinically used to assess liver injury were not consistently correlated to the findings of MVLA. Metabolite changes consistent with the presence of MVLA correlated with interruptions in inositol, carbohydrate, glycerolipid, and glyoxylate metabolism. This study reveals markers that could find pre-clinical use, provides insights into mechanisms involved in MVLA, and demonstrates the need for the validation of noninvasive MVLA markers in human patients.
Keywords: Tuberculosis, Isoniazid, Liver injury, Metabolomics
1 Introduction
Microvesicular lipid accumulation (MVLA) is a reversible histopathological lesion that may precede irreversible liver damage, and hence, early detection of MVLA is important in the clinical management of liver injury. Current methods for detecting MVLA require biopsy and histopathological evaluation which can be painful, time consuming, expensive and includes some level of risk to the patient. The specific aim of our study was to use metabolomics of urine to identify noninvasive marker(s) of MVLA liver injury, and to determine how the noninvasive urinary marker(s) correlate with metabolomic perturbations in liver tissue. A noninvasive method for detection of MVLA could greatly aid clinicians in the management of patients taking drugs that are known or suspected to induce this injury, and could improve screening for this injury in animals in pre-clinical studies during drug development.
Isoniazid (INH) was selected as a model compound for this investigation because it is known to induce MVLA and because of its widespread clinical use that is sometimes associated with hepatic injury. INH is used in a combination therapy for treatment of tuberculosis (TB); an infectious disease with a significant mortality rate. There is an estimated 2 billion people with latent TB, and an estimated 8–9 million new cases of TB reported yearly with more than one third of the world’s population being at risk for developing TB at some point in life (World Health Organization 2003, 2008). Complications arise during treatment due to difficulties with compliance during the long course of treatment, and due to the potential for hepatotoxicity. In developed nations and as possible in developing nations, patients taking INH are monitored for elevation in alanine amino transferase (ALT), an enzyme that increases in the serum during and following INH treatment regimens. In some patients, acute liver injury (defined as serum ALT >5 times the upper limits of normal) has a causal association with INH therapy. If not recognized early, such liver injury can result in catastrophic liver failure, requiring liver transplantation (Russo et al. 2004). In the US, INH is one of the clinically prescribed drugs with the highest causal relation (up to 9% in studied patient populations) for patients taking medication that result in drug induced liver injury (DILI) and liver transplantation(Russo et al. 2004). In developing nations, DILI frequency from usage of INH is even greater, with up to 33% of patients taking INH experiencing hepatotoxicity (Saukkonen et al. 2006).
The mechanisms of INH liver injury may involve the metabolism of INH to acetylisoniazid, hydrolysis to acetyl hydrazine and isonicotinic acid. Acetyl hydrazine can undergo acetylation to diacetylhydrazine, or hydrolysis to hydrazine (Timbrell 1979; Preziosi 2007). Hydrazine can undergo oxidation to radicals and conversion to ammonia. The acetylation of proteins and other molecules by acetylhydrazine has also been described, and the depletion of pyridoxal phosphate (PLP) can occur with INH administration. Other metabolites that have been described include the formation of hydrazones of keto acids, primarily pyruvate and α-ketoglutarate (2-oxoglutarate) (Timbrell 1979; Preziosi 2007). Cytochrome P4502E1 mediates INH-induced hepatotoxicity in male Wistar rats following a dose of 100 mg/kg/day for up to 21 days (Yue et al. 2004). Hepatotoxicity was measured by elevated serum enzymes (ALT, and AST) and liver histopathology. Hydrazine is a metabolite INH in rabbits (Sarich et al. 1996), and hydrazine levels in liver and CYP2E1 activity correlated with the severity of hepatocellular damage. Kleno et al. (2004) demonstrated that a single dose of hydrazine in rats causes gene, protein, and metabolite changes, which can be related to glucose metabolism, lipid metabolism, and oxidative stress. Mechanisms of INH liver injury could involve interference with the target, mycolic acids. The inhibition of the synthesis of mycolic acids in the therapeutic action of INH has been investigated, and may involve the inhibition of mycobacterial enoyl acyl carrier protein reductase. The inhibition may be caused by the formation of an adduct between INH and NAD+ or NADP+ (Argyrou et al. 2006, 2007). A single administration of 110 mg/kg INH to male Wistar rats results in a decrease in succinate, α-ketoglutarate, and hippurate (Schoonen et al. 2007).
The current practice of liver biopsy and histopathological evaluation for the clinical diagnosis of MVLA is avoided in patients with infectious diseases and is impractical in large populations due to the expense and logistics. Therefore, there is a need for practical methods to monitor patients for the presence or onset of MVLA during treatment regiments. The results described here demonstrate a urinary marker profile for MVLA in rats administered INH, and the relation to markers and pathways that could be used for the early detection of this reversible liver injury.
2 Materials and methods
2.1 Study design and sample collection
Male Sprague Dawley rats were obtained from Charles River, Raleigh, NC, and arrived at 8 weeks of age. They were provided food (Purina rodent chow 5002) and water ad lib and housed in microisolator cages for 2 weeks. At least six rats per dose and time point were administered by gavage 0, 10, or 300 mg INH (dissolved in water) per kilogram body weight (mg/kg) per day for up to 8 days. Urine was collected over dry ice (0–6 and 6–24 h) from rats in all glass metabolism cages prior to the first dose (pre-dose), and again following day 1 (day 1 urine) and following day 8 (day 8 urine) of dosing, and stored at −70°C. The focus of the metabolomics analysis was on urine collected 6–24 h following administration of INH.
2.2 Serum enzymes and histopathology
Groups of rats were sacrificed following the 24 h urine collection on day 1 (termed the day 1 group) and day 8 (termed the day 8 group) for the collection of blood and liver. Blood was processed to serum for the measurement of alanine aminotransferase (ALT), aspartate aminotransferase (AST), and sorbitol dehydrogenase (SDH) using standard commercial kits and procedures. Sections of the right, median, and left lobes of the liver were collected and histopathologic assessment was made by evaluating approximately 50 histological parameters of the liver by Experimental Histopathology Laboratories (EPL), Inc. (Research Triangle Park, NC). MVLA was expected following INH treatment based on previous publications identifying this histological effect in animals and humans. The extensive list of histological diagnoses in this investigation was utilized in order to correlate the metabolomics profile with MVLA and to be certain that the metabolomic profile was not also correlated with other histopathology effects. The individual histological observations were given a “score” of 1–3 to indicate the magnitude of the effect as determined by the semi-quantitative histological observation. A score of 1 indicates a minimal effect while a score of three indicates the greatest change noted for each histological observation.
2.3 Metabolomics: sample preparation, data acquisition, and processing
Urine samples were prepared for NMR analysis by mixing an aliquot (~630 µl) of urine with 70 µl of a solvent buffer solution containing two internal standards (4,4-dimethyl-4-silapentane-1-sulfonic acid, DSS, and imidazole) for line shape analysis and spectral assignment and quantitation using the library in NMR Suite 4.6 Professional software. The left lobe, right lobe, and median lobe were prepared for NMR analysis by homogenization of each liver lobe (3:1 by weight; water:liver) and extraction with acetonitrile. Following extraction, the sample was dried under N2(g) and reconstituted in 630 µl D2O with 70 µl of the solvent buffer solution (DSS and imidazole). NMR spectra were acquired on a Varian Inova 600 MHz instrument located in the NMR facility at Duke University, Durham, NC. 1H-NMR spectra were acquired using the first increment of a NOESY sequence, with a 100 ms mixing time, 1 s relaxation delay, a spectral width of 12 ppm, and 32 transients. The water resonance was suppressed using resonance irradiation during the relaxation delay. All spectra are acquired at 25°C, and the quality of each NMR spectrum was assessed for the level of noise and alignment of identified markers. Spectra were accessed for missing data and underwent quality checks.
NMR data was processed using a traditional binning approach (The Handbook of Metabonomics and Metabolomics 2007) by automated integration (increments of 0.04 ppm) over the spectral window (producing 237 bins after excluding urea and the region of water suppression), and normalized to the total spectral intensity. NMR data were also processed using NMR Suite 4.6 Professional software, which deconvolutes the entire spectrum based on chemical shift and coupling patterns, and then matches signals to a reference library of approximately 300 low molecular weight metabolites. The internal standards (DSS and imidazole) were used as the references for performing the library matching. This software contains an internal library adjustment for increments in chemical shift based on pH variations (Weljie et al. 2006). A concentration determination for each metabolite was made by relative integration of the analyte to the internal standard, where the library of concentrations was developed to account for differences in integral values as related to the relaxation time of the signal (Weljie et al. 2006). This method has some advantageous over binning in two major ways. First, small increments in pH can result in portions of metabolite signals aligning with different bins when using the binning approach, while deconvolution circumvents this problem. A second advantage is that this analysis depends on the concentration of each metabolite, while the binning approach results in situations where each metabolite has multiple signals that fall within separate bins. For subsequent data reduction of the data derived from library matching, the concentration of each metabolite was normalized to total creatinine.
2.4 Data reduction and visualization
Data captured by NMR (metabolite id and concentration; or bin region and integral value) were transferred to software (Umetrics) for data reduction and visualization using SIMCA-P 11.5. Several approaches were taken to analyze the NMR data to provide the best set of analytes that could distinguish the study groups (predose, day 1 vehicle, day 1 low dose, day 1 high dose, day 8 vehicle, day 8 low dose, day 8 high dose) and that could provide the best correlation between the metabolomic profiles and the histopathological findings. For analysis with binned data, the integral was normalized to the total integral for each spectrum. For analysis using data from library matching, the concentration of each metabolite was normalized to creatinine. Principal component analysis (PCA) and partial least squares projection to latent structures was conducted using SIMCA-P 11.5 for the binned and metabolite data. Loadings and variable importance plots were examined to determine the bins or metabolites that best correlated with dose groups, and to find the bins and metabolites that best correlated with histopathological findings. PC1 and PC2 accounted for ~60% of the variance, and PC1–PC3 accounted for ~85% of the variance in the cases shown herein.
Subsequently, PCA using only the bins or analytes selected from the loadings and variable importance plots was conducted to demonstrate that the analysis with the subset of bins or metabolites could provide clear separation of the dose groups, and could provide correlation with the histopathological findings. Metabolites identified as important for the separation of dose groups or for correlation with histopathological findings were mapped to biochemical pathways.
3 Results and discussion
3.1 Metabolomics of urine
Metabolomics analysis was conducted on urine collected from rats administered by gavage 0, 10, or 300 mg/kg INH for 1 or 8 days.
3.2 Binning approach
Principal component analysis was conducted using binned data normalized to the total intensity, and excluding the region of water suppression, for each spectrum of urine (Fig. 1). PCA analysis using bin data (Fig. 1) clearly showed distinction of the predose, vehicle control and low dose groups (teal, purple and green, respectively) from the 300 mg/kg dose groups (blue, 1 day; red, 8 day). Five rats in the 300 mg/kg 8 day group were clustered closer to the 300 mg/kg 1 day group, while four rats in the 300 mg/kg 8 day group (Z4509, Z4507, Z3513 and Z3511) were significantly different from other rats within this dose and time group. Review of the histopathology data (Fig. 2a, b) revealed that the four rats (Z4509, Z4507, Z3513 and Z3511) in the 300 mg/kg 8 day group that had urinary profiles that differentiated them from the remainder of their respective dose/time group were rats with MVLA of the right, left, and/or median lobes of the liver. Analysis of ALT (Fig. 3a), AST (Fig. 3b), and SDH (Fig. 3c) indicated very little to equivocal elevation of serum enzymes. It is important to note that MVLA did not correlate with the serum concentration of liver enzymes. At the highest dose, ALT was reduced, but did not provide identification of the individual animals with MVLA. Reduction in ALT following INH administration is consistent with findings by O’Brien and co-workers (2002), and attributed to the involvement of the cofactor pyridoxal phosphate.
Fig. 1.
PCA analysis of bin data (excluding the region of water) normalized to total intensity for each spectrum of urine (6–24 h) from rats prior to and following administration of INH at 0, 10, or 300 mg/kg per day for 1 or 8 days. Four rats administered INH for 8 days (Z4509, Z4507, Z3513 and Z3511) were separated from the remainder of the dose and time groups
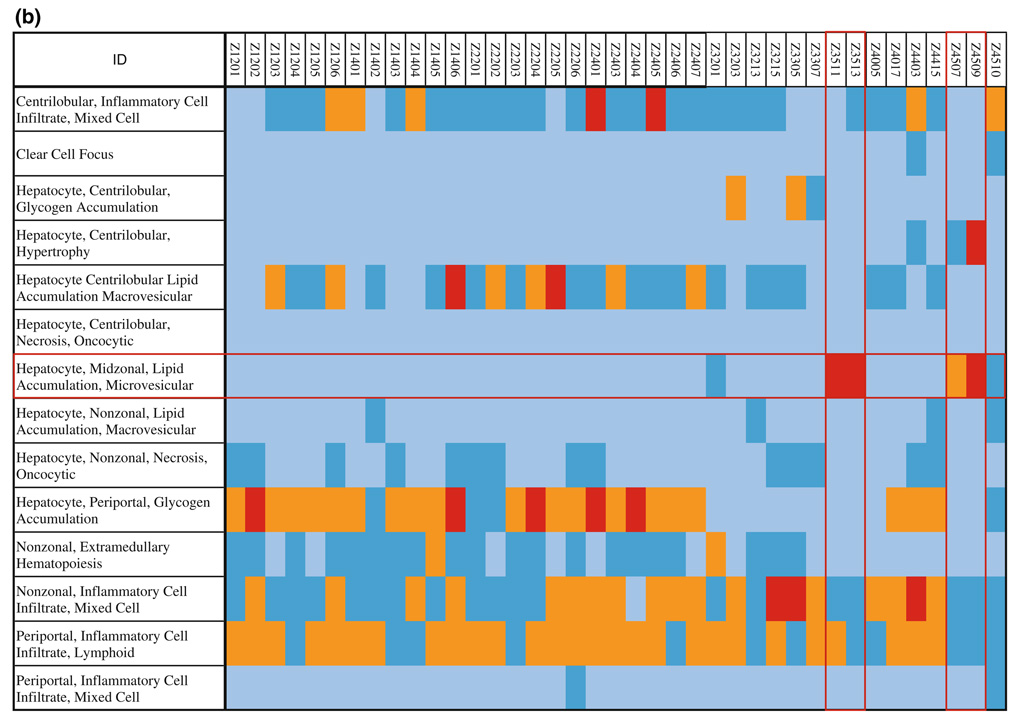
Fig. 2.
a Histopathology revealed that the four rats (Z4509, Z4507, Z3513 and Z3511) had midzonal MVLA. Periportal glycogen depletion was found for nearly all rats administered the high dose of INH: 11/15 left lobe, 13/15 median lobe; 12/15 right lobe. Histopathology findings are indicated by color as no findings (light blue); score of 1 (blue), score of 2 (orange) and score of 3 (red). b Histopathology results were added across the three lobes and normalized by the weight of the liver lobes. The summary of histopathology findings are indicated by color as no findings (light blue); score of 1 (blue), score of 2 (orange) and score of 3 (red): again showing that the most significant correlation for distinct biochemical profiles for the 4 rats (Z4509, Z4507, Z3513 and Z3511) was with MVLA. Periportal glycogen depletion was present in 11/15 rats administered the high dose. (Color figure online)
Fig. 3.
Analysis of ALT (Fig. 3a), AST (Fig. 3b), and SDH (Fig. 3c) indicated very little to equivocal elevation of serum enzymes. The highest serum enzyme elevations did not correlate with the histopathology findings of MVLA for rats Z4509, Z4507, Z3513 and Z3511. At the highest dose, ALT was reduced, but did not provide identification of the individual animals with MVLA
Loadings plots and variable importance plots were evaluated to determine the sub-set of bins that best defined MVLA. Subsequently, PCA analysis was conducted with only the sub-set of bins (Fig. 4) and demonstrated that this sub-set of data provided a greater differentiation of the four rats with MVLA from all other rats in the study.
Fig. 4.
PCA analysis of bin data selected from variable importance plots and loadings plots that demonstrated the most significant contribution to differentiation of rats with microvesicular lipid accumulation demonstrated an increase separation for rats with MVLA (Z4509, Z4507, Z3513 and Z3511) from other study groups
3.3 Library matching
Full library matching using the NMR Suite 4.6 Professional Software (Chenomx, Edmonton, Alberta, Canada) provided a list of metabolites (and their respective concentrations) that demonstrated a match within each of the urine spectra. The concentration for each metabolite was normalized to the creatinine concentration for each urine sample and PCA analysis was conducted using the concentration based data. The PCA analysis (Fig. 5) using urinary metabolite concentration data also clearly differentiated the four rats with right, left, and/or median lobe MVLA (Z4509, Z4507, Z3513 and Z3511) from all other rats dosed with INH. Specifically, the rats in the 8 day high dose group with the presence of MVLA could be clearly differentiated from the rats in the 8 day high dose group without the findings of MVLA. Inspection of the PCA plot in comparison with histopathological findings also revealed that rats with periportal glycogen depletion were well separated from rats without periportal glycogen depletion, on the t[1] axis.
Fig. 5.
PCA analysis of metabolite concentration data (normalized to creatinine) for urine (6–24 h) from rats prior to and following administration of INH at 0, 10, or 300 mg/kg per day for 1 or 8 days; indicated a clear separation for rats with MVLA (Z4509, Z4507, Z3513 and Z3511) from other study groups
3.4 Determining urinary markers that define the study groups
Loadings plots and variable importance plots were examined to determine the metabolites that best define each of the dose and time groups, and that best define the groups based on histopathology findings. These metabolites and their directionality of change are listed in Fig. 6 (under trajectories) between the control and INH low dose group, between the INH low dose group and the INH high dose group following 1 day of exposure, and between the INH low dose group and the INH high dose group following 8 days of exposure. In terms of significant histopathology findings, the suite of metabolites that could be correlated with the finding of MVLA are listed in Fig. 6 (listed under Z4509, Z4507, Z3513 and Z3511). Rats with MVLA showed an increase in urinary creatine, aminoadipate, xanthine, acetoacetate, oxaloacetate, glycolate, ornithine, and homoserine; and a decrease in cytosine, glutamine, trigonelline, fructose, pyridoxate, and hydroxybutyrate. When multivariate analysis was conducted using only this subset of metabolites, the rats with MVLA showed an even greater separation from the remainder of the study groups. The subset of metabolites that best separated the high dose and low dose groups are listed at the top of Fig. 6 (under the heading of low dose to high dose 1 day). This separation corresponds to the findings of periportal glycogen depletion in a significant portion (11 out of 15) of the rats that received a high dose of INH. The subset of metabolites that best separated the high dose 1 day group from the high dose 8 day group (without MVLA) are also shown in Fig. 6 (under the heading of high dose 1 day to high dose 8 day). These metabolites may best define the on-set of MVLA.
Fig. 6.
Inspection of variable importance plots and loadings plots enable the selection of metabolites most important to defining each of the study groups, the metabolites most important to defining the presence of MVLA for rats with MVLA (Z4509, Z4507, Z3513 and Z3511) from other study groups and the metabolites most important for defining the outcome of glycogen depletion
To determine if the two sub-sets of metabolites that defined either MVLA or periportal glycogen depletion would also be diagnostic of different types of liver injuries, these two sub-sets of metabolites were tested using data from our ongoing investigations of acetaminophen-, phenytoin-, valproic acid-, and clofibrate-induced liver injury. Dose- and time- to response studies (conducted analogous to the INH study) using these four drugs resulted in pathology findings of hypertrophy, necrosis, and macrovesicular lipid accumulation. MVLA and periportal glycogen depletion were pathology findings in the investigation with INH, but were not observed in the studies with the other four drugs. The biochemical profiles or urine obtained from the analogous studies with acetaminophen, phenytoin, valproic acid, and clofibrate were examined to determine if the sub-sets of metabolites that defined MVLA or periportal glycogen depletion would also be predictive of the different pathology findings (hypertrophy, necrosis, and macrovesicular lipid accumulation) from those investigations. The subsets of metabolites that correlate with pathology findings of MVLA and periportal glycogen depletion could not be associated with any other types of pathology findings determined for INH or the other four drugs.
3.5 Defining pathways relevant to the urinary markers
Metabolites that define MVLA were mapped to pathways that suggested interruptions in amino acid metabolism, alkanoate metabolism, purine metabolism, glycolate and dicarboxylate metabolism, urea cycle, citrate cycle, glutamate metabolism and pyrimidine metabolism (listed in Fig. 7). Metabolites that differentiate the rats in the high dose groups (with the presence of periportal glycogen depletion) from rats in the low dose groups (absence of periportal glycogen depletion) were mapped to purine metabolism, amino acid metabolism, citrate cycle, and alkanoate metabolism. Pathways perturbations that occurred between the high dose 1 day and high dose 8 day groups also included amino acid metabolism, citrate cycle, glycolate and dicarboxylate metabolism. In addition, metabolites that differentiated the high dose 1 day and high dose 8 day groups suggested interruptions in ascorbate and aldarate metabolism, inositol metabolism, and glycerolipid metabolism. The pattern (Fig. 7) revealed by the metabolomics analysis indicates that the onset of MVLA may occur following periportal glycogen depletion, associated with interruptions in amino acid, purine, and alkanoate metabolism. Continued dosing results in additional effects on ascorbate/aldarate metabolism, inositol metabolism, carbohydrate metabolism, glycerolipid metabolism, and glyoxylate/dicarboxylate metabolism; which may have the causal relation to the on-set of MVLA.
Fig. 7.
Pathways most important to defining each of the study groups, the presence of MVLA for rats with MVLA (Z4509, Z4507, Z3513 and Z3511) from other study groups, and the outcome of glycogen depletion were determined
3.6 Analysis of liver extracts
The left lobe, right lobe, and median lobes of liver were individually homogenized (3:1 by weight; water:liver), extracted with acetonitrile, dried under N2(g), and reconstituted with D2O and a solvent buffer containing DSS and imidazole. Metabolomics analysis of the liver was conducted as described for urine. Multivariant analysis (Fig. 8) was conducted independently for the (a) right lobes, (b) left lobes, or (c) median lobes. Analysis of the right lobe (Fig. 8a) demonstrated separation of the liver extracts for Z4509, Z4507, Z3513 and Z3511 from the remainder of the study groups; consistent with the findings of significant right lobe MVLA histopathology for each of these four rats (Fig. 2a; Z4509, Z4507, Z3513 or Z3511). Analysis of the left lobe (Fig. 8b) revealed most significant separation of Z4509, Z4511, Z3513 from the remainder of the rats; these rats all having a MVLA histopathology score of 2. Analysis of the median lobe (Fig. 8c) also demonstrated significant separation of Z3511, Z3513, Z4509, and Z4507 from the remainder of the study rats.
Fig. 8.
PCA analysis of metabolomics data acquired for liver extracts from the a right lobe, b left lobe, and c median lobes
Inspections of the loadings and variable importance plots revealed, overall, that the metabolites that best separate the dose groups were independent of the liver lobe analyzed. Metabolites that were increased in the liver lobe for the high dose groups (compared with time-matched controls) included arginine (6–13 fold increase between control and high dose day 8; urea and amino acid metabolism), aminoadipate (24–38 fold increase between control and high dose day 8; lysine metabolism), hydroxybutyrate (20-fold increase; butanoate metabolism). Rats with MVLA has a decrease in serine, mannitol, oxalacetate, glycerol, methylmalonate, lactate, sulphocysteine, taurine, and cystine; indicating an impact on amino acid metabolism, glycine, serine, threonine metabolism, pyrimidine metabolism, glycolysis, cysteine metabolism, tryptophan metabolism, and Krebs cycle. Of these metabolites, aminoadipate, hydroxybutyrate, oxalacetate, methylmalonate, taurine, cystine, and lactate were increased or decreased at some time-points in the urine of rats administered 10 or 300 mg/kg INH for 1 or 8 days. Differences in the specific metabolites in liver or in urine that best associate with the presence of MVLA were not surprising, in part because the urine sample was integrated over a 24 h period while the extract of liver was taken at one time only, at 24 h after the last dose.
3.7 Biochemical interpretation
Some of the changes in metabolites encountered with treatment with INH may result from direct reaction with INH, or hydrazine, whereas others may result from interference with the metabolism of enzymes that involve PLP as cofactor (6). This class of enzymes includes aminotransferases, and enzymes that catalyze beta-elimination reactions such as the reactions carried out by serine dehydratase. Yet other effects may result from depletion of essential substrates in the liver. 2-Aminoadipate transaminase, the enzyme involved in the oxidation of 2-aminoadipate, and is an intermediate in the biosynthesis and degradation of lysine, requires PLP as a coenzyme. Alterations in the metabolism of amino acids can plausibly be linked to changes in the activity of enzymes requiring PLP as a cofactor. Other changes may result from direct reaction of INH or its metabolites with keto acids, such as pyruvate, and oxoglutarate.
The multivariant analyses revealed amino acids as important metabolites defining the INH exposed rats from control. Methionine was one of the most significantly elevated amino acids for both the low and high dose groups (approximately 3-fold over control). Multivariate analysis also revealed 2-aminoadipate as an important metabolite in defining the 8 day high dose group. Specifically, 2-aminoadipate levels in urine increased 6-fold in the Day 8 high-dose group, compared with the controls, and did not increase in any of the other dose groups. Furthermore 2-aminoadipate was over 3-fold higher in rats with MVLA compared with non-MVLA rats in the same dose group. 2-Aminoadipate is an intermediate in the metabolism of lysine (Higashino et al. 1971), and undergoes metabolism by 2-aminoadipate transaminase (2.6.1.39), a pyridoxal phosphate containing enzyme (Tobes and Mason 1977), to form 2-oxoadipate, converting 2-oxoglutarate to glutamate (Nakatani et al. 1970) [l-2-aminoadipate + 2-oxoglutarate 2-oxoadipate + l-glutamate]. 2-Aminoadipate was also substantially increased in liver of the day 1 and day 8 high dose groups (3–4 fold and 24–38 fold, respectively), showing an increase that paralleled the change seen in urine.
Creatine, glycolate, glutarate, dimethylxanthine, acetoacetate adipate, and 4-aminobutyrate were increased in urine for animals with MVLA compared with non-MVLA animals in the same dose group. Creatine increased approximately 7-fold in urine from the 8-day high dose rats, compared with the controls. Rats with MVLA had an 11-fold increase (over control) in creatine levels, while non-MVLA rats in the same dose group exhibited only a 4-fold increase over control.
Urinary glycolate increased approximately 5-fold in the rats with MVLA, compared to controls; while non-MVLA rats in the same group increased only 1.4 fold. Glutarate increased 6-fold in the MVLA rats in the high dose 8-day group, versus 2.7-fold in the non-MVLA rats in the same group. Glutarate is formed via two routes: one from 2-oxoadipate in lysine catabolism, via oxidative decarboxylation; the second in the degradation of tryptophan via 2-aminomuconate (Goodman et al. 1975). Acetoacetate in urine was profoundly increased in the rats with MVLA (approximately 5-fold higher than the rats without MVLA), as was acetone (5.8-fold higher), and to a lesser extent 3-hydroxybutyrate (1.7-fold higher). The synthesis of ketone bodies is generally associated with high rates of fatty acid oxidation in the liver (Yeh and Zee 1976, 1977). Mean adipate concentrations increased approximately 3-fold in rats with MVLA while rats in the same group without MVLA increased 1.3-fold.
4 Concluding remarks
This study has demonstrated a promising non-invasive urinary marker profile that could be further evaluated for the detection of the reversible histopathology MVLA in human subjects, and provide an improved method for the detection of MVLA over assessment via traditional liver serum enzyme assays. DILI in a portion of the patients taking INH creates a challenge for the safe use of this drug where alternative therapies are not available. A biomarker that indicates a liver effect prior to nonreversible liver injury would greatly enhance the safe use of this therapy. Our studies reveal that the liver from rats with MVLA (induced using INH) has a significant perturbation in glycine, serine, threonine metabolism, pyrimidine metabolism, glycolysis, cysteine metabolism, tryptophan metabolism, and Krebs cycle metabolism. Urinary metabolites consistent with the presence of MVLA suggested interruptions in some of the same pathways (amino acid metabolism, pyrimidine metabolism); suggesting the promise of developing a non-invasive surrogate marker for MVLA. Using a metabolomics approach for the validation of the marker profile will enable the determination of any additional metabolites that should be included in a marker profile to reveal MVLA histopathologies in human investigations.
Acknowledgement
The authors are grateful for support of this research through funding via an NIH Roadmap Grant and the National Institute of General Medical Sciences (Grant 5R21GM75903).
Contributor Information
Susan J. Sumner, Email: ssumner@rti.org, Discovery Sciences, RTI International, 3040 Cornwallis Drive, Research Triangle Park, NC 27709, USA.
Jason P. Burgess, Discovery Sciences, RTI International, 3040 Cornwallis Drive, Research Triangle Park, NC 27709, USA
Rodney W. Snyder, Discovery Sciences, RTI International, 3040 Cornwallis Drive, research Triangle Park, NC 27709, USA
James A. Popp, Stratoxon LLC, Lancaster, PA 17601, USA
Timothy R. Fennell, Discovery Sciences, RTI International, 3040 Cornwallis Drive, research Triangle Park, NC 27709, USA
References
- Argyrou A, Jin L, Siconilfi-Baez L, Angeletti RH, Blanchard JS. Proteome-wide profiling of isoniazid targets in Mycobacterium tuberculosis. Biochemistry. 2006;45:13947–13953. doi: 10.1021/bi061874m. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Argyrou A, Vetting MW, Blanchard JS. New insight into the mechanism of action of and resistance to isoniazid: Interaction of Mycobacterium tuberculosis enoyl-ACP reductase with INH-NADP. Journal of the American Chemical Society. 2007;129:9582–9583. doi: 10.1021/ja073160k. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Goodman SI, Markey SP, Moe PG, Miles BS, Teng CC. Glutaric aciduria; A “new” disorder of amino acid metabolism. Biochemical Medicine. 1975;12:12–21. doi: 10.1016/0006-2944(75)90091-5. [DOI] [PubMed] [Google Scholar]
- Higashino K, Fujioka M, Yamamura Y. The conversion of l-lysine to saccharopine and alpha-aminoadipate in mouse. Archives of Biochemistry and Biophysics. 1971;142:606–614. doi: 10.1016/0003-9861(71)90525-x. [DOI] [PubMed] [Google Scholar]
- Kleno TG, Kiehr B, Baunsgaard D, Sidelmann UG. Combination of ‘omics’ data to investigate the mechanism(s) of hydrazine-induced hepatotoxicity in rats and to identify potential biomarkers. Biomarkers. 2004;9(2):116–138. doi: 10.1080/13547500410001728408. [DOI] [PubMed] [Google Scholar]
- Lindon JC, Nicholson JK, Holmes E, editors. The handbook of metabonomics and metabolomics. Published by Elsevier; 2007. p. 561. Edition: illustrated ISBN 0444528415, 978044452841. [Google Scholar]
- Nakatani Y, Fujioka M, Higashino K. Alpha-aminoadipate aminotransferase of rat liver mitochondria. Biochimica et Biophysica Acta. 1970;198:219–228. doi: 10.1016/0005-2744(70)90054-9. [DOI] [PubMed] [Google Scholar]
- O’Brien PJ, Slaughter MR, Polley SR, Kramer K. Advantages of glutamate dehydrogenase as a blood biomarker of acute hepatic injury in rats. Laboratory Animals. 2002;36:313–321. doi: 10.1258/002367702320162414. [DOI] [PubMed] [Google Scholar]
- Preziosi P. Isoniazid: Metabolic aspects and toxicological correlates. Current Drug Metabolism. 2007;8:839–851. doi: 10.2174/138920007782798216. [DOI] [PubMed] [Google Scholar]
- Russo MW, Galanko JA, Shrestha R, Fried MW, Watkins P. Liver transplantation for acute liver failure from drug induced liver injury in the United States. Liver Transplantation. 2004;10(8):1018–1023. doi: 10.1002/lt.20204. [DOI] [PubMed] [Google Scholar]
- Sarich TC, Youssefi M, Zhou T, Adams SP, Wall RA, Wright JM. Role of hydrazine in the mechanism of isoniazid hepatotoxicity in rabbits. Archives of Toxicology. 1996;70(12):835–840. doi: 10.1007/s002040050347. [DOI] [PubMed] [Google Scholar]
- Saukkonen JJ, Cohn DL, Jasmer RM, et al. on behalf of the ATS Hepatotoxicity of Antituberculosis Therapy Subcommittee. An Official ATS Statement: Hepatotoxicity of Antituberculosis Therapy. American Journal of Respiratory and Critical Care Medicine. 2006;174:935–952. doi: 10.1164/rccm.200510-1666ST. [DOI] [PubMed] [Google Scholar]
- Schoonen WG, Kloks CP, Ploemen JP, et al. Uniform procedure of (1)H NMR analysis of rat urine and toxicometabonomics. Part II: Comparison of NMR profiles for classification of hepatotoxicity. Toxicological Sciences. 2007;98:286–297. doi: 10.1093/toxsci/kfm077. [DOI] [PubMed] [Google Scholar]
- Timbrell JA. The role of metabolism in the hepatotoxicity of isoniazid and iproniazid. Drug Metabolism Reviews. 1979;10:125–147. doi: 10.3109/03602537908993904. [DOI] [PubMed] [Google Scholar]
- Tobes MC, Mason M. Alpha-aminoadipate aminotransferase and kynurenine aminotransferase. Purification, characterization, and further evidence for identity. Journal of Biological Chemistry. 1977;252:4591–4599. [PubMed] [Google Scholar]
- Weljie AM, Newton J, Mercier PM, Carlson E, Slupsky CM. Targeted profiling: Quantitative analysis of 1H-NMR metabolomics data. Analytical Chemistry. 2006;78(13):4430–4442. doi: 10.1021/ac060209g. [DOI] [PubMed] [Google Scholar]
- World Health Organization. Treatment of tuberculosis: Guidelines for national programmes. Geneva: World Health Organization; 2003 WHO/CDS/TB/2003.313.
- World Health Organization. Global tuberculosis control—surveillance, planning, financing. WHO Report 2008. 2008 WHO/HTM/TB/2008.393.
- Yeh YY, Streuli VL, Zee P. Relative utilization of fatty acids for synthesis of ketone bodies and complex lipids in the liver of developing rats. Lipids. 1977;12:367–374. doi: 10.1007/BF02533640. [DOI] [PubMed] [Google Scholar]
- Yeh YY, Zee P. Relation of ketosis to metabolic changes induced by acute medium-chain triglyceride feeding in rats. Journal of Nutrition. 1976;106:58–67. doi: 10.1093/jn/106.1.58. [DOI] [PubMed] [Google Scholar]
- Yue J, Peng RX, Yang J, Kong R, Liu J. CYP2E1 mediated isoniazid-induced hepatotoxicity in rats. Acta Pharmacologica Sinica. 2004;25(5):699–704. [PubMed] [Google Scholar]