Abstract
The analysis of the title chiral auxiliary compound, C13H15NOS2, has enabled the determination of the absolute configuration at the benzyl-bearing ring C atom as S. In the crystal structure, molecules aggregate into helical chains along the b axis via C—H⋯O contacts.
Related literature
For background to the use of N-acyl thiazolidinethiones as versatile chiral auxiliaries for asymmetric aldol reactions, see: Crimmins & Chaudhary (2000 ▶); Crimmins et al. (2005 ▶); Crimmins & Haley (2006 ▶); Crimmins & Dechert (2009 ▶). For the synthesis, see: McKennon & Meyer (1993 ▶); Delaunay et al. (1995 ▶); Lu et al. (2009 ▶).
Experimental
Crystal data
C13H15NOS2
M r = 265.39
Monoclinic,

a = 8.850 (6) Å
b = 7.189 (5) Å
c = 10.595 (7) Å
β = 95.537 (6)°
V = 670.9 (8) Å3
Z = 2
Mo Kα radiation
μ = 0.38 mm−1
T = 298 K
0.50 × 0.40 × 0.20 mm
Data collection
Rigaku Mercury diffractometer
Absorption correction: multi-scan (Jacobson, 1998 ▶) T min = 0.831, T max = 0.925
7301 measured reflections
2734 independent reflections
2361 reflections with F 2 > 2σ(F 2)
R int = 0.038
Refinement
R[F 2 > 2σ(F 2)] = 0.041
wR(F 2) = 0.048
S = 0.86
2734 reflections
170 parameters
H-atom parameters constrained
Δρmax = 0.32 e Å−3
Δρmin = −0.35 e Å−3
Absolute structure: Flack (1983 ▶), 1138 Friedel pairs
Flack parameter: −0.05 (6)
Data collection: CrystalClear (Pflugrath, 1999 ▶); cell refinement: CrystalClear; data reduction: CrystalStructure (Molecular Structure Corporation & Rigaku, 2006 ▶); program(s) used to solve structure: SIR2004 (Burla et al., 2005 ▶); program(s) used to refine structure: CRYSTALS (Betteridge et al., 2003 ▶); molecular graphics: X-SEED (Barbour et al., 2001 ▶); software used to prepare material for publication: CrystalStructure.
Supplementary Material
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536809030104/tk2514sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536809030104/tk2514Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Table 1. Hydrogen-bond geometry (Å, °).
| D—H⋯A | D—H | H⋯A | D⋯A | D—H⋯A |
|---|---|---|---|---|
| C9—H9⋯O1i | 0.95 | 2.55 | 3.408 (4) | 150 |
Symmetry code: (i)  .
.
Acknowledgments
NRG thanks the Institute of Life Sciences for allowing him to pursue this work as part of his PhD thesis.
supplementary crystallographic information
Comment
N-Acyl thiazolidinethiones, e.g. (I), are versatile chiral auxiliaries for asymmetric aldol reactions Crimmins & Chaudhary (2000). Many complex natural products have been synthesized using these auxiliaries (Crimmins et al. 2005; Crimmins & Haley, 2006; Crimmins & Dechert, 2009). The synthesis of (I) starts from amino alcohol 2 which was converted to thiazolidinethione 3 by reacting with carbon disulfide followed by treatment with propionyl chloride (Fig. 3) (Crimmins & Chaudhary, 2000).
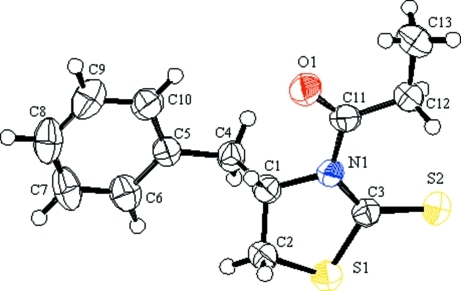
The single crystal analysis of (I), Fig. 1, allowed the determination of the absolute configuration of C1 as S. The crystal structure shows the molecules to aggregate into helical chains along the screw axis via C9—H9···O1 contacts (Fig. 2, Table 1).
Experimental
To a solution of β-amino alcohol 2 (10 mmol) (McKennon & Meyer, 1993) in aqueous 1.0 N potassium hydroxide (50 ml) was added carbon disulfide (50 mmol, 3.0 ml) slowly. The reaction mixture was refluxed at 110 °C for 12 h to give the desired thiazolidinethione 3 (Delaunay et al. 1995). To a solution of compound 3 (0.478 mmol) in dichloromethane (DCM, 3 ml) was added triethylamine (0.956 mmol) and the temperature was maintained at -40 to -78 °C. To that mixture was added propionyl chloride (0.574 mmol) drop wise. The mixture was stirred for 1–2 h, diluted with DCM (10 ml), washed with water (2 x 10 ml), dried over anhydrous Na2SO4 and concentrated low vacuum to give (I) as a light-yellow solid; mp. 374–376 K (lit. mp. 374.1 K (Lu et al. 2009)).
Compound (I) (50 mg) was dissolved in 2:1 DCM/EtOAC (1.0 ml) and left in freezer (10 °C) until fine crystals appeared. Crystals were separated from soluton and washed with hexane and dried under vacuum.
Refinement
The H atoms were positioned geometrically and refined in the riding model approximation with C—H = 0.95 Å, and with Uiso(H) set to 1.2Ueq(C).
Figures
Fig. 1.
Molecular structure of (I) showing the atom numbering scheme. The displacement ellipsoids are drawn at the 50% probability level. H-atoms are shown by small circles of arbitrary radii.
Fig. 2.
Crystal packing of (I) showing the formation of helical chains. The C-H···O contacts are shown as dashed lines.
Fig. 3.
Synthesis of (I).
Crystal data
| C13H15NOS2 | F(000) = 280 |
| Mr = 265.39 | Dx = 1.314 Mg m−3 |
| Monoclinic, P21 | Mo Kα radiation, λ = 0.71070 Å |
| Hall symbol: P 2yb | Cell parameters from 3674 reflections |
| a = 8.850 (6) Å | θ = 2.3–27.4° |
| b = 7.189 (5) Å | µ = 0.38 mm−1 |
| c = 10.595 (7) Å | T = 298 K |
| β = 95.537 (6)° | Prism, yellow |
| V = 670.9 (8) Å3 | 0.50 × 0.40 × 0.20 mm |
| Z = 2 |
Data collection
| Rigaku Mercury diffractometer | 2361 reflections with F2 > 2σ(F2) |
| Detector resolution: 7.31 pixels mm-1 | Rint = 0.038 |
| ω scans | θmax = 27.4° |
| Absorption correction: multi-scan (Jacobson, 1998) | h = −11→11 |
| Tmin = 0.831, Tmax = 0.925 | k = −6→9 |
| 7301 measured reflections | l = −13→13 |
| 2734 independent reflections |
Refinement
| Refinement on F2 | Chebychev polynomial with 3 parameters (Carruthers & Watkin, 1979) 10359.0000 14093.9000 3595.6900 |
| R[F2 > 2σ(F2)] = 0.041 | (Δ/σ)max < 0.001 |
| wR(F2) = 0.048 | Δρmax = 0.32 e Å−3 |
| S = 0.86 | Δρmin = −0.35 e Å−3 |
| 2734 reflections | Absolute structure: Flack (1983), 1138 Friedel pairs |
| 170 parameters | Flack parameter: −0.05 (6) |
| H-atom parameters constrained |
Special details
| Geometry. Bond distances, angles etc. have been calculated using the rounded fractional coordinates. All su's are estimated from the variances of the (full) variance-covariance matrix. The cell e.s.d.'s are taken into account in the estimation of distances, angles and torsion angles |
| Refinement. Refinement was performed using all reflections. The weighted R-factor (wR) and goodness of fit (S) are based on F2. R-factor (gt) are based on F. The threshold expression of F2 > 2.0 σ(F2) is used only for calculating R-factor (gt). |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| S1 | 0.55104 (6) | 1.08609 (10) | 0.94969 (5) | 0.0574 (2) | |
| S2 | 0.80540 (7) | 1.31770 (10) | 1.04864 (6) | 0.0630 (2) | |
| O1 | 0.96079 (17) | 1.0605 (2) | 0.69074 (15) | 0.0720 (6) | |
| N1 | 0.79082 (15) | 1.1039 (2) | 0.83154 (13) | 0.0410 (4) | |
| C1 | 0.6994 (2) | 0.9543 (2) | 0.76431 (19) | 0.0436 (6) | |
| C2 | 0.5366 (2) | 0.9806 (2) | 0.79429 (19) | 0.0498 (7) | |
| C3 | 0.7324 (2) | 1.1748 (2) | 0.93706 (19) | 0.0462 (6) | |
| C4 | 0.7616 (2) | 0.7610 (2) | 0.80159 (19) | 0.0469 (6) | |
| C5 | 0.7183 (2) | 0.6180 (2) | 0.70140 (17) | 0.0441 (6) | |
| C6 | 0.5872 (2) | 0.5119 (2) | 0.7014 (2) | 0.0561 (7) | |
| C7 | 0.5469 (3) | 0.3871 (3) | 0.6051 (2) | 0.0673 (9) | |
| C8 | 0.6379 (3) | 0.3610 (3) | 0.5087 (2) | 0.0704 (9) | |
| C9 | 0.7667 (3) | 0.4651 (3) | 0.5063 (2) | 0.0719 (9) | |
| C10 | 0.8074 (2) | 0.5935 (3) | 0.60097 (19) | 0.0569 (7) | |
| C11 | 0.9289 (2) | 1.1496 (2) | 0.7801 (2) | 0.0501 (7) | |
| C12 | 1.0320 (2) | 1.3001 (3) | 0.8384 (2) | 0.0545 (7) | |
| C13 | 1.1629 (2) | 1.3369 (4) | 0.7610 (2) | 0.0861 (10) | |
| H1 | 0.70330 | 0.96970 | 0.67560 | 0.0520* | |
| H6 | 0.52430 | 0.52600 | 0.76860 | 0.0670* | |
| H7 | 0.45570 | 0.31750 | 0.60530 | 0.0790* | |
| H8 | 0.61000 | 0.27260 | 0.44400 | 0.0830* | |
| H9 | 0.83030 | 0.44600 | 0.44020 | 0.0870* | |
| H10 | 0.89540 | 0.66810 | 0.59680 | 0.0690* | |
| H21 | 0.48380 | 1.06070 | 0.73390 | 0.0600* | |
| H22 | 0.48550 | 0.86440 | 0.79480 | 0.0590* | |
| H41 | 0.72130 | 0.72350 | 0.87750 | 0.0550* | |
| H42 | 0.86910 | 0.76750 | 0.81570 | 0.0560* | |
| H121 | 1.07080 | 1.26210 | 0.92110 | 0.0650* | |
| H122 | 0.97480 | 1.41110 | 0.84380 | 0.0650* | |
| H131 | 1.19300 | 1.46340 | 0.77050 | 0.1050* | |
| H132 | 1.24580 | 1.25860 | 0.78930 | 0.1050* | |
| H133 | 1.13230 | 1.31200 | 0.67430 | 0.1050* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| S1 | 0.0512 (2) | 0.0621 (3) | 0.0611 (3) | −0.0066 (3) | 0.0167 (2) | −0.0116 (3) |
| S2 | 0.0587 (3) | 0.0640 (4) | 0.0664 (3) | −0.0025 (3) | 0.0059 (2) | −0.0247 (3) |
| O1 | 0.0664 (9) | 0.0751 (11) | 0.0796 (10) | −0.0150 (9) | 0.0330 (8) | −0.0265 (10) |
| N1 | 0.0401 (7) | 0.0364 (8) | 0.0470 (8) | −0.0001 (7) | 0.0062 (6) | −0.0019 (7) |
| C1 | 0.0419 (10) | 0.0430 (11) | 0.0458 (10) | −0.0008 (8) | 0.0035 (8) | 0.0002 (8) |
| C2 | 0.0417 (10) | 0.0508 (13) | 0.0561 (12) | −0.0027 (9) | 0.0011 (8) | −0.0019 (9) |
| C3 | 0.0427 (10) | 0.0438 (11) | 0.0513 (11) | 0.0037 (8) | 0.0008 (8) | −0.0042 (9) |
| C4 | 0.0510 (11) | 0.0393 (11) | 0.0487 (11) | 0.0028 (8) | −0.0031 (8) | −0.0011 (8) |
| C5 | 0.0453 (10) | 0.0389 (12) | 0.0467 (10) | 0.0008 (8) | −0.0027 (8) | 0.0025 (8) |
| C6 | 0.0647 (13) | 0.0460 (13) | 0.0569 (12) | −0.0083 (10) | 0.0017 (10) | 0.0087 (9) |
| C7 | 0.0828 (17) | 0.0465 (14) | 0.0678 (15) | −0.0207 (11) | −0.0179 (13) | 0.0093 (11) |
| C8 | 0.104 (2) | 0.0511 (15) | 0.0517 (13) | −0.0052 (13) | −0.0146 (13) | −0.0034 (11) |
| C9 | 0.0992 (19) | 0.0603 (15) | 0.0580 (14) | 0.0079 (15) | 0.0176 (13) | −0.0055 (12) |
| C10 | 0.0579 (11) | 0.0527 (12) | 0.0617 (12) | 0.0015 (12) | 0.0134 (9) | −0.0026 (12) |
| C11 | 0.0415 (11) | 0.0480 (13) | 0.0617 (12) | 0.0026 (8) | 0.0095 (9) | −0.0011 (9) |
| C12 | 0.0430 (10) | 0.0488 (12) | 0.0711 (13) | −0.0016 (10) | 0.0032 (9) | −0.0073 (11) |
| C13 | 0.0543 (13) | 0.086 (2) | 0.121 (2) | −0.0223 (15) | 0.0242 (13) | −0.0216 (19) |
Geometric parameters (Å, °)
| S1—C2 | 1.806 (2) | C12—C13 | 1.506 (3) |
| S1—C3 | 1.744 (2) | C1—H1 | 0.9500 |
| S2—C3 | 1.650 (2) | C2—H21 | 0.9500 |
| O1—C11 | 1.199 (3) | C2—H22 | 0.9500 |
| N1—C1 | 1.486 (2) | C4—H41 | 0.9500 |
| N1—C3 | 1.374 (3) | C4—H42 | 0.9500 |
| N1—C11 | 1.424 (2) | C6—H6 | 0.9500 |
| C1—C2 | 1.517 (3) | C7—H7 | 0.9500 |
| C1—C4 | 1.532 (2) | C8—H8 | 0.9500 |
| C4—C5 | 1.501 (3) | C9—H9 | 0.9500 |
| C5—C6 | 1.389 (3) | C10—H10 | 0.9500 |
| C5—C10 | 1.395 (3) | C12—H121 | 0.9500 |
| C6—C7 | 1.380 (3) | C12—H122 | 0.9500 |
| C7—C8 | 1.373 (3) | C13—H131 | 0.9500 |
| C8—C9 | 1.366 (4) | C13—H132 | 0.9500 |
| C9—C10 | 1.385 (3) | C13—H133 | 0.9500 |
| C11—C12 | 1.509 (3) | ||
| C2—S1—C3 | 93.92 (9) | C1—C2—H21 | 110.00 |
| C1—N1—C3 | 115.36 (14) | C1—C2—H22 | 111.00 |
| C1—N1—C11 | 115.55 (14) | H21—C2—H22 | 109.00 |
| C3—N1—C11 | 129.05 (14) | C1—C4—H41 | 109.00 |
| N1—C1—C2 | 107.08 (13) | C1—C4—H42 | 109.00 |
| N1—C1—C4 | 111.56 (15) | C5—C4—H41 | 108.00 |
| C2—C1—C4 | 112.58 (14) | C5—C4—H42 | 109.00 |
| S1—C2—C1 | 104.96 (13) | H41—C4—H42 | 109.00 |
| S1—C3—S2 | 118.18 (11) | C5—C6—H6 | 119.00 |
| S1—C3—N1 | 110.37 (12) | C7—C6—H6 | 120.00 |
| S2—C3—N1 | 131.43 (14) | C6—C7—H7 | 120.00 |
| C1—C4—C5 | 112.21 (15) | C8—C7—H7 | 119.00 |
| C4—C5—C6 | 122.19 (16) | C7—C8—H8 | 120.00 |
| C4—C5—C10 | 120.09 (16) | C9—C8—H8 | 121.00 |
| C6—C5—C10 | 117.67 (16) | C8—C9—H9 | 119.00 |
| C5—C6—C7 | 120.91 (19) | C10—C9—H9 | 120.00 |
| C6—C7—C8 | 120.7 (2) | C5—C10—H10 | 119.00 |
| C7—C8—C9 | 119.3 (2) | C9—C10—H10 | 120.00 |
| C8—C9—C10 | 120.7 (2) | C11—C12—H121 | 109.00 |
| C5—C10—C9 | 120.65 (18) | C11—C12—H122 | 109.00 |
| O1—C11—N1 | 117.07 (15) | C13—C12—H121 | 109.00 |
| O1—C11—C12 | 121.82 (17) | C13—C12—H122 | 109.00 |
| N1—C11—C12 | 121.09 (16) | H121—C12—H122 | 109.00 |
| C11—C12—C13 | 111.70 (18) | C12—C13—H131 | 109.00 |
| N1—C1—H1 | 109.00 | C12—C13—H132 | 110.00 |
| C2—C1—H1 | 109.00 | C12—C13—H133 | 109.00 |
| C4—C1—H1 | 108.00 | H131—C13—H132 | 109.00 |
| S1—C2—H21 | 110.00 | H131—C13—H133 | 110.00 |
| S1—C2—H22 | 111.00 | H132—C13—H133 | 109.00 |
| C3—S1—C2—C1 | 22.91 (12) | C4—C1—C2—S1 | 93.51 (15) |
| C2—S1—C3—S2 | 171.92 (11) | N1—C1—C4—C5 | −155.93 (15) |
| C2—S1—C3—N1 | −9.64 (13) | C2—C1—C4—C5 | 83.7 (2) |
| C3—N1—C1—C2 | 25.15 (19) | C1—C4—C5—C6 | −91.3 (2) |
| C3—N1—C1—C4 | −98.44 (17) | C1—C4—C5—C10 | 86.0 (2) |
| C11—N1—C1—C2 | −156.77 (15) | C4—C5—C6—C7 | 177.20 (17) |
| C11—N1—C1—C4 | 79.65 (19) | C10—C5—C6—C7 | −0.1 (3) |
| C1—N1—C3—S1 | −7.69 (18) | C4—C5—C10—C9 | −178.46 (18) |
| C1—N1—C3—S2 | 170.47 (14) | C6—C5—C10—C9 | −1.1 (3) |
| C11—N1—C3—S1 | 174.53 (14) | C5—C6—C7—C8 | 1.9 (3) |
| C11—N1—C3—S2 | −7.3 (3) | C6—C7—C8—C9 | −2.4 (3) |
| C1—N1—C11—O1 | −2.2 (2) | C7—C8—C9—C10 | 1.2 (3) |
| C1—N1—C11—C12 | 179.47 (16) | C8—C9—C10—C5 | 0.6 (3) |
| C3—N1—C11—O1 | 175.56 (17) | O1—C11—C12—C13 | 7.3 (3) |
| C3—N1—C11—C12 | −2.8 (3) | N1—C11—C12—C13 | −174.42 (17) |
| N1—C1—C2—S1 | −29.44 (15) |
Hydrogen-bond geometry (Å, °)
| D—H···A | D—H | H···A | D···A | D—H···A |
| C9—H9···O1i | 0.95 | 2.55 | 3.408 (4) | 150 |
Symmetry codes: (i) −x+2, y−1/2, −z+1.
Footnotes
ILS Publication No. ILS-MCO-0904.
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: TK2514).
References
- Barbour, L. J. (2001). J. Supramol. Chem.1, 189-191.
- Betteridge, P. W., Carruthers, J. R., Cooper, R. I., Prout, K. & Watkin, D. J. (2003). J. Appl. Cryst.36, 1487.
- Burla, M. C., Caliandro, R., Camalli, M., Carrozzini, B., Cascarano, G. L., De Caro, L., Giacovazzo, C., Polidori, G. & Spagna, R. (2005). J. Appl. Cryst.38, 381–388.
- Crimmins, M. T. & Chaudhary, K. (2000). Org. Lett.2, 775–777. [DOI] [PubMed]
- Crimmins, M. T., Christie, H. S., Chaudhary, K. & Long, A. (2005). J. Am. Chem. Soc.127, 13810–13812. [DOI] [PubMed]
- Crimmins, M. T. & Dechert, A.-M. R. (2009). Org. Lett.11, 1635–1638. [DOI] [PMC free article] [PubMed]
- Crimmins, M. T. & Haley, M. W. (2006). Org. Lett.8, 4223–4225. [DOI] [PubMed]
- Delaunay, D., Toupet, L. & Le Corre, M. J. (1995). Org. Chem.60, 6604–6607.
- Flack, H. D. (1983). Acta Cryst. A39, 876–881.
- Jacobson, R. (1998). Private communication to the Rigaku Corporation, Tokyo, Japan.
- Lu, C., Nie, J., Yang, G. & Chen, Z. (2009). Can. J. Chem.87, 30–32.
- McKennon, M. J. & Meyer, A. I. (1993). J. Org. Chem.58, 3568–3571.
- Molecular Structure Corporation & Rigaku (2006). CrystalStructure MSC, The Woodlands, Texas, USA, and Rigaku Corporation, Tokyo, Japan.
- Pflugrath, J. W. (1999). Acta Cryst. D55, 1718–1725. [DOI] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536809030104/tk2514sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536809030104/tk2514Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report