Abstract
Daily timing of the mammalian circadian clock of the suprachiasmatic nucleus (SCN) is regulated by photic input from the retina via the retinohypothalamic tract. This signaling is mediated by glutamate which activates SCN retinorecipient units communicating to pacemaker cells in part through the release of gastrin-releasing peptide (GRP). Efferent signaling from the SCN involves another SCN-containing peptide, arginine vasopressin (AVP). It is notable that little is known concerning the mechanisms regulating these peptides, as literature on in vivo peptide release in the SCN is sparse. Here, microdialysis-radioimmunoassay procedures were used to characterize mechanisms controlling GRP and AVP release in the hamster SCN. In animals housed under a 14hr:10hr 24hr LD cycle both peptides exhibited daily fluctuations of release, with levels increasing during the morning to peak around midday. Under constant darkness, this pattern persisted for AVP, but rhythmicity was altered for GRP, characterized by a broad plateau throughout the subjective night and early subjective day. Neuronal release of the peptides was confirmed by their suppression with reverse-microdialysis perfusion of calcium blockers and stimulation with depolarizing agents. Reverse-microdialysis perfusion with the 5-HT1A,7 agonist, 8-OH-DPAT, during the day significantly suppressed GRP but had little effect on AVP. Also, perfusion with the glutamate agonist NMDA, or exposure to light at night, increased GRP but did not affect AVP. These analyses reveal distinct daily rhythms of SCN peptidergic activity, with GRP but not AVP release attenuated by serotonergic activation that inhibits photic phase-resetting, and activated by glutamatergic and photic stimulation that mediate this phase-resetting.
Keywords: suprachiasmatic, microdialysis, neuropeptide, serotonin, glutamate, photic
The master circadian clock in mammals is located within the suprachiasmatic nucleus (SCN) of the anterior hypothalamus (Klein et al., 1991; Moore, 1983; Rusak and Zucker 1979). Clock timing is synchronized to the 24 hr light-dark cycle directly by photic signaling relayed from the retina to the SCN via the retinohypothalamic tract (RHT; Johnson et al., 1988; Moore and Lenn 1972; Pickard, 1982), and indirectly from the intergeniculate leaflet via the geniculohypothalamic tract (GHT; Card and Moore, 1982; Johnson et al., 1989). The SCN also receives nonphotic entraining input from the GHT and the midbrain raphe nuclei (Albers and Ferris, 1984; Biello et al., 1994; Marchant et al., 1997; Meyer-Bernstein and Morin, 1996). The mechanism by which this diverse input is processed by the SCN to produce a stably entrained rhythm involves interplay among these systems, including three populations of peptidergic neurons indigenous to the SCN. These include cells producing gastrin-releasing peptide (GRP), vasoactive intestinal polypeptide (VIP) and arginine vasopressin (AVP). The former two peptides are implicated in photic signaling, while AVP is primarily associated with output signaling from the SCN (reviewed by Reghunandanan and Reghunandanan, 2006).
The SCN photic signaling cascade is initiated by glutamate release from RHT terminals located primarily in the ventral SCN core region. This is confirmed by observations that glutamate in the SCN induces time-dependent phase-shifts in the circadian activity rhythm and that glutamate antagonists block the behavioral and neurophysiological phase-resetting actions of light (Abe et al., 1991; Abe et al., 1992; Ding et al., 1994; Gamble et al., 2004; Rea et al., 1993). Principal among the retinorecipient targets of glutamate are GRP-containing neurons in the SCN core (Piggins et al., 1995). When photically stimulated, these neurons provide input to a dorsal cap region, where GRP is released to induce downstream signaling events, including expression of per1 and immediate-early genes as well as activation of extracellular signal-related kinases (ERK; Antle et al., 2005; Earnest et al., 1993; Hamada et al., 2004). Collectively the above observations provide a model whereby GRP neurons driven by external photic cues transmit entraining information relayed to SCN pacemaker cells, delineated by AVP, to regulate clock phase. Rhythmic signaling of these pacemaker cells to regions outside the SCN, in turn, involves the AVP-containing neurons (Buijs, 1996; Kalsbeek et al., 2006). AVP is also thought to play a regulatory role within the SCN by synchronizing oscillations generated by SCN neurons (Shirakawa et al., 2001).
Despite the extensive literature on the circadian roles of GRP and AVP, there is a gap in current understanding of how their daily activities in the SCN are regulated. Specifically (with the exception of a single study on AVP; Kalsbeek, 1995), the nature of their in vivo release in the SCN of freely behaving animals has not been characterized. To approach this question, we have adapted brain microdialysis procedures to measure the 24 hr profiles of GRP and AVP in the hamster SCN under light entrained and constant dark conditions, and to assess the effects of localized administration of glutamatergic and serotonergic agents that stimulate and inhibit photic signaling, respectively. Such information is basic to understanding how these pathways contribute to the regulation of SCN timekeeping activity.
EXPERIMENTAL PROCEDURES
Animals
Adult male Syrian hamsters (Mesocricetus auratus), raised from breeder pairs obtained from Harlan Sprague-Dawley (Madison, IL), were used in these studies. Animals were maintained in a climate-controlled (20–22°C) environmental chamber under a 14hr:10hr 24hr LD cycle (LD; 200-250 lux illuminance) and prior to experimentation were individually housed in circular polycarbonate cages (Raturn; Bioanalytical Systems Inc.; West Lafayette, IN). Rodent chow (Prolab 3000; PMI Feeds, Inc.; St. Louis, MO) and water were provided ad libitum. The experiments were approved by the Kent State Institutional Animal Care and Use Committee and were conducted using the National Institutes of Health Guidelines for the Care and Use of Laboratory Animals.
SCN Microdialysis
The microdialysis procedures used here are similar to those described in previous studies on SCN monoamine neurotransmitter release (Glass et al., 2003), except that probes having larger molecular weight cutoff were used (CMA/12; 20KDa cutoff; CMA Microdialysis, Inc. North Chelmsford, MA). The in vitro efficiencies for GRP and AVP recovery in microdialysate were determined by incubating probes in an ACSF solution of 125I-labeled peptides at 37°C and measuring radioactive label recovery in the microdialysate at a flowrate of 1 μl/min. Values averaged 8.6% and 12.9% for GRP and AVP, respectively. For surgery, animals were anesthetized with sodium pentobarbital (Nembutal; 85 mg/kg) and received a probe implant with the tip aimed at the SCN (coordinates: AP: +0.03 mm from bregma, L: +0.03 mm from midline, H: −8.0 mm from dura; head level). Surgical screws and dental acrylic were used to fasten the probe to the skull. The animals were allowed to recover for 48–72 hr prior to microdialysis sampling. Over this period the blood-brain barrier is reestablished (Benveniste, 1989), and animals exhibit normal levels of circadian behavioral rhythmicity, as confirmed by actogram analysis. For microdialysis sampling, animals housed in a rotating bowl were connected via a tether attached to a harness that protected the inflow and outflow tubings. Artificial cerebral spinal fluid (ACSF; NaCl, 126.5 mM; NaHCO3, 27.5 mM; KCl, 2.4 mM; KH2PO4, 0.5 mM; CaCl2, 1.1 mM; MgCl2, 0.85 mM; Na2SO4, 0.5 mM; D-(+)-glucose, 5.9 mM; bovine serum albumin, 0.1%; pH 7.5) was perfused through the probe at a flowrate of 1.0 μl/min using a calibrated syringe pump (CMA Microdialysis) and gas-tight syringe (Hamilton). The microdialysate flowed from the probe via PFTE tubing leading to 250 μl polyethylene non-stick tubes contained in an automated refrigerated fraction collector (BAS). Samples were frozen at −70°C until analysis.
SCN microdialysate neuropeptide measurements
The contents of GRP and AVP in SCN microdialysate were measured by radioimmunoassay (RIA; Phoenix Pharmaceuticals, Inc. Burlingame, CA). The assays are highly specific for these peptides, with low cross-reactivities of the primary antibodies with other peptides (data supplied with the assays [GRP, RK-027-07; AVP, RK-065-07]). Diluting the primary antibodies shifted the standard curves to the left, increasing the sensitivity of each assay to ~0.2 pg, with IC50’s of 10 pg for GRP, and 3 pg for AVP. The intra-assay coefficients of variability of these assays were 10.8% and 6.7%, respectively. The typical yield range for the peptides in SCN microdialysate was substantially above the limit of detection (1–60 pg/sample). The standard protocol for each assay was that standards and microdialysis samples were incubated in primary antibody for 48 hr at 4°C. Samples of ACSF and drug cocktails equal in volume to microdialysis samples (60 μl) were run as controls, in addition to the positive controls with known peptide content. Next, samples were incubated with 125I-labeled peptide for 24 hr at 4°C, and then incubated with normal rabbit serum and goat-anti-rabbit secondary antibody for 90 min at room temperature. Tubes were centrifuged, the supernatant aspirated, and radioactivity of the pellets measured using a Packard Instruments Cobra II gamma counter.
Circadian locomotor activity measurements
In experiments where 24–48 hr profiles of peptide release under LD and DD were characterized, general locomotor activity rhythms were monitored from 4–5 days and 2 wks, respectively, prior to surgery and during the period of microdialysis sampling to verify a normal pattern of circadian behavioral activity during sampling, and to establish subjective circadian time (CT) under DD. Locomotor activity was monitored using infrared motion detectors interfaced with a computerized data acquisition system (Clocklab; Coulbourn Instruments, Whitehall, PA). In animals under DD, the onset of activity (defined as the first ≥10 min period of activity that was followed ≤ 20 min later by a period of at least 1 hr of sustained activity) was designated as CT 12.
Experimental protocols
Verification of neuronal SCN peptide release
Two separate pharmacological treatments delivered via intra-SCN reverse-microdialysis were used to confirm that peptides measured in the SCN microdialysates were from neuronal release (as opposed to procedural artifact). First, a calcium blocking mixture was used (diltiazem 200μM, verapamil 200μM, cinnarizine 15μM, flunarizine 12μM) in calcium-free ACSF containing EDTA (10 mM). Second, SCN peptide release was measured concomitantly with a pulse of depolarizing medium consisting of ACSF with high [K+] (150 mM) and veratridine (100 μM; Sigma). Two days after microdialysis probe implantation, sampling for both treatments was initiated with a 2 hr equilibration period of normal ACSF. This was followed by 1 hr of baseline collection, then sample collection during a 1 hr pulse of Ca2+ blocking medium (perfusion from ZT 2–3 for both peptides; n=3 for each peptide) or depolarizing medium (perfusion from ZT 13–14 for GRP, n=2; perfusion from ZT 6–7 for AVP, n=4), followed by a 3 hr post-stimulation sample collection period with normal ACSF. Controls sampled using normal ACSF throughout were run in parallel.
Daily profiles of SCN peptide release under LD and DD
For these experiments, animals were kept in their home cage and in the same chamber to minimize any procedural behavioral stimulation. Also, samples were collected automatically and the system operation was monitored remotely in the light and dark phases by video to minimize contact with the animals. On the day of experimentation, the probe lining was connected to a syringe pump at ZT 4 under LD (GRP n=4; AVP n=6) or CT 4 under DD (n=3 for both peptides) and perfusion with ACSF was initiated. After a 2 hr equilibration period, sample collection was initiated at ZT 6 or CT 6 with an interval of 1 hr at a flow rate of 1.0 μl/min over the ensuing experiment. Each animal was run for a total of 48 hr and the first set of 24 samples were analyzed for one peptide, while the second set of 24 samples was analyzed for the other, to minimize the unnecessary use of extra animals. The automated fraction collector was set with a time delay to account for the lag-time of effluent flow from the probe to reach the collection vial. Microdialysis probe implantation under DD was undertaken by anesthetizing the animals in darkness, and masking the eyes with black tape to prevent retinal photic stimulation during the surgery. The microdialysis procedures, including probe connection were undertaken under dim red light (0.5 lux).
Photic and nonphotic treatment effects on SCN peptide release
Photic (glutamatergic or light) and nonphotic (serotonergic) neurotransmitter mechanisms involved with regulating SCN neuropeptide release were studied pharmacologically using reverse microdialysis. Hamsters outfitted with an SCN microdialysis probe underwent 3 hr of equilibration perfusion with ACSF, 2 hr baseline collection preceding 1 hr perfusion with 8-OH-DPAT (1.2 mM; ZT 6–7 for GRP; n=4 and ZT 1–2 for AVP; n=3) or NMDA (200 μM; ZT 13–14 for GRP; n=2 and ZT 18–19 for AVP; n=3) followed by 3 hr of post-treatment sampling. Additionally, animals were exposed to a 1 hr light pulse (~280 lux) during the dark phase (ZT 13–14, n=5 for GRP; the time when GRP SCN microinjection induces large delay shifts [Piggins et al., 1995]; and ZT 18–19, n=3 for AVP; the time when light could advance the morning release peak) following 2 hr baseline collection. This pulse was followed by 3 hr of post-treatment sampling.
Histological evaluations of intracranial implant sites
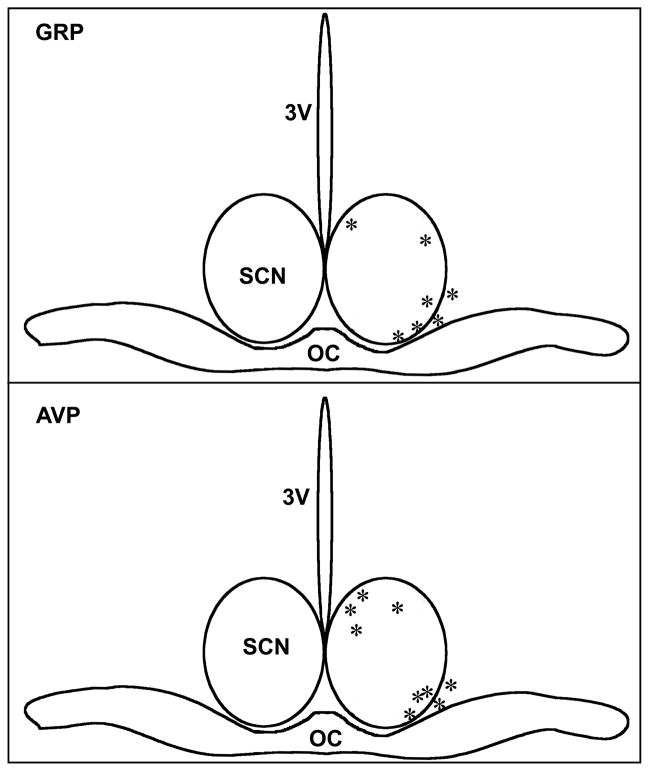
After completion of each experiment, microdialysis probe location was verified histologically. Hamsters were deeply anesthetized with Nembutal and brains were extracted, fixed in 4% paraformaldehyde overnight and then cryoprotected in 30% sucrose. Cryostat sections of the SCN (20 μm) were stained with cresyl violet for light microscopic verification of implant placement, diagrammed in Fig. 1.
Figure 1.
Diagram of the SCN demonstrating location of microdialysis probe tip sites (*) in animals sampled for GRP (top) and AVP (bottom). SCN, suprachiasmatic nucleus; OC, optic chiasma; 3V, third ventricle.
Statistics
Drug effects were normalized as a percentage of the pretreatment baseline collections (1 hr) and were analyzed using a one-way repeated measures ANOVA. Treatment effects were determined using the Student Newman-Keuls test. Individual 24 hr profiles of peptide release were normalized by expressing values as a percentage of the daily mean. Daily variations in this release were analyzed as in previous experiments (Dudley et al., 1998) using a one-way repeated measures ANOVA followed by Dunnet’s test procedure for comparing multiple group means (Zar, 1983). For all procedures the level of significance was set at p<0.05.
RESULTS
Neuronal release of peptides from the SCN
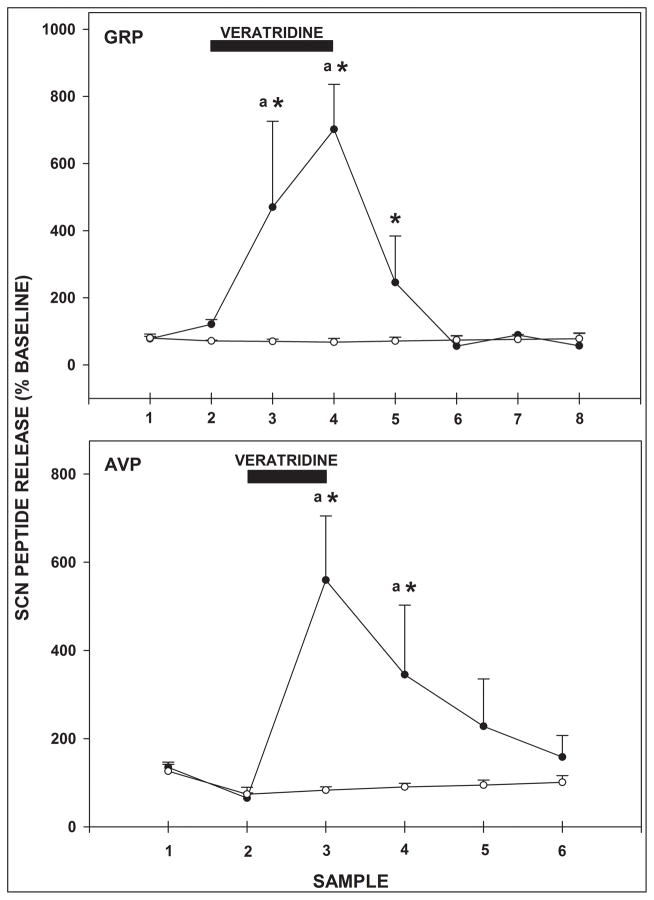
Neuronal (synaptic) release of GRP and AVP from the SCN was evaluated by reverse-dialysis with ACSF containing Ca2+ blockers. This treatment significantly reduced neuronal peptide output compared to pretreatment baseline levels (32±8% of baseline; F5,15=6.500; P=0.002 for GRP, 42±9% of baseline; F2,5=9.605; P=0.002 for AVP; Fig. 2). Conversely, depolarizing neurons with a similar perfusion of high [K+] ACSF containing veratridine markedly increased the output of each of the peptides above pretreatment baseline levels (702±134% of baseline; F6,9=5.659; P=0.011 for GRP, 559±145% of baseline; F3,5=6.041; P=0.003 for AVP; Fig. 3).
Figure 2.
Suppressive effects of reverse-microdialysis perfusion of the SCN with ACSF containing a calcium blocking cocktail (closed circles) on GRP and AVP release. Filled bars represent duration of perfusion. “a”, significantly different from pretreatment baseline (p<0.05); “*”, significantly different from normal ACSF control (open circles; p<0.05). Sampling intervals for GRP and AVP were 30 min and 60 min, respectively. Values are the mean±SEM.
Figure 3.
Stimulatory effects of reverse-microdialysis perfusion of the SCN with high [K+] ACSF containing veratridine (closed circles) on GRP and AVP release. Filled bars represent duration of perfusion. “a”, significantly different from pretreatment baseline (p<0.05); “*”, significantly different from normal ACSF control (open circles; p<0.05). Sampling intervals for GRP and AVP were 30 min and 60 min, respectively. Values are the mean±SEM.
Daily profiles of peptide release in the SCN
GRP release
Output of GRP was rhythmic under LD, with each maximal release near midday. In LD, output began increasing at lights-on and rose gradually to an acrophase at ZT 4 (176±54% of the daily mean; F3,23=2.317; P=0.004). Thereafter, output dropped sharply to a nadir at ZT 7 (68±11% of the daily mean; Fig. 4), and remained low throughout the night. Under DD, peak release shifted from the day to the subjective night, and was prolonged, lasting throughout much of the night (129±14% of the daily mean; F2,23=3.106; P<0.001). Lowest levels of release occurred from CT 7–14 (68±6% of the daily mean; Fig. 4).
Figure 4.
Averaged daily profile of GRP release from the SCN under LD (top) and DD (bottom). The 24 hr profiles are double-plotted to highlight the rhythmic daily profile of this release. Filled bars represent the dark phase under LD. Inserted below each profile is a representative actogram of an animal undergoing the microdialysis sampling. The sampling interval was 1 hr. Data points are the mean±SEM. “*”, p<0.05 vs. non-peak timepoints.
AVP release
AVP output increased during the night, reaching a broad acrophase spanning ZT 0–4 (149±22% of the daily mean; F5,23=2.534; P<0.001). Output then decreased abruptly to a nadir at ZT 12 (65±14% of the daily mean; Fig. 5). Output of AVP was rhythmic in animals maintained under DD for 14 days and displayed a pattern similar to that under LD, such that peak levels occurred from CT 1–4 (245±21% of the daily mean; F2,23=4.050; P<0.001). Lowest output extended from subjective midday to the end of the subjective night (Fig. 5).
Figure 5.
Averaged daily profile of AVP release from the SCN under LD (top) and DD (bottom). The 24 hr profiles are double-plotted to highlight the rhythmic daily profile of this release. Filled bars represent the dark phase under LD. Inserted below each profile is a representative actogram of an animal undergoing the microdialysis sampling. The sampling interval was 1 hr. Data points are the mean±SEM. “*”, p<0.05 vs. non-peak timepoints.
For both peptides, several animals received probe implants outside (>500 μm distant from) the lateral SCN margin. No statistically significant daily pattern of neuropeptide release was evident for either peptide (F2,23=0.957; P=0.532 for GRP; F2,23=0.988; P=0.497 for AVP; n=3 for both peptides; Fig. 6). Also, microdialysate concentrations of both peptides were lower than those measured using probes within the SCN domain (maximum [GRP], 32.2±9.9 pg vs. 1.8±0.1 pg [F1,5=6.78; P=0.048] and maximum [AVP], 2.2±0.3 vs. 0.9±0.1 pg [F1,7=6.82; P=0.035].
Figure 6.
Averaged daily profile of GRP release (top) and AVP release (bottom) of animals with microdialysis probe implant site >500μm from the lateral extent of the SCN. The 24 hr profiles do not exhibit significant daily rhythmicity. Filled bars represent the dark phase. Inserted below each profile is a representative actogram of an animal undergoing the microdialysis sampling. The sampling interval was 1 hr. Data points are the mean±SEM.
Serotonergic and glutamatergic/photic effects on SCN peptide release
8-OH-DPAT
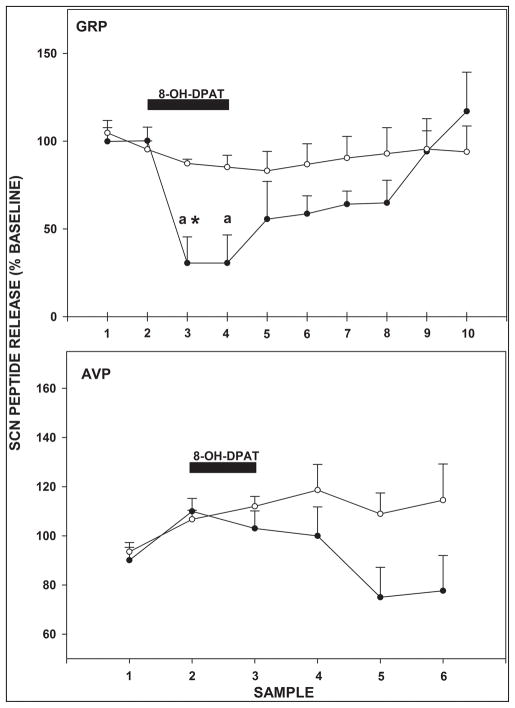
Reverse microdialysis perfusion of the SCN with the 5-HT1,7 agonist, 8-OH-DPAT, for 1 hr caused a marked suppression of GRP release (31±15% of baseline; F3,9=4.495; P=0.001; Fig. 7) that persisted for at least 2 hr after the end of drug perfusion. For AVP, 8-OH-DPAT did not significantly affect AVP release compared to baseline levels (F2,5=2.657; P=0.088; Fig. 7).
Figure 7.
Effects of reverse-microdialysis perfusion of the SCN with ACSF containing 8-OH-DPAT on GRP (perfusion from ZT 6–7) and AVP (perfusion from ZT 0–1) release. Filled bars represent duration of perfusion; “a” significantly different from pretreatment baseline (p<0.05); “*”, significantly different from normal ACSF control (open circles; p<0.05). Sampling intervals for GRP and AVP were 30 min and 60 min, respectively. Values are the mean±SEM.
NMDA
Reverse microdialysis of the SCN with the glutamate agonist, NMDA, differentially affected peptide release. For GRP, NMDA perfusion during the early dark phase caused a significant, yet transient, increase in release (186±3% of baseline; F1,7=8.288; P<0.005; Fig. 8). This treatment had no effect on AVP release (F7,4=1.294; P=0.327; Fig. 8).
Figure 8.
Effects of reverse-microdialysis perfusion of the SCN with ACSF containing NMDA on GRP (perfusion from ZT 13–14) and AVP (perfusion from ZT 18–19) release. Filled bars represent duration of perfusion; “a” significantly different from pretreatment baseline (p<0.05); “*”, significantly different from normal ACSF control (open circles; p<0.05). Sampling intervals for GRP and AVP were 30 min and 60 min, respectively. Values are the mean±SEM.
Light
A 1 hr light pulse during the dark phase (ZT 13–14) caused a significant increase in GRP release from the SCN (157±17% of baseline; F9,6=4.910; P=0.002; Fig. 9). This treatment at ZT 18–19 had no effect on AVP release (F2,5=1.965; P=0.17; Fig. 9).
Figure 9.
Effects of a 1 hr light pulse on GRP (ZT 13–14) and AVP (ZT 18–19) release from the SCN. Filled bars represent duration of light pulse; “a” significantly different from pretreatment baseline (p<0.05); “*”, significantly different from normal ACSF control (open circles; p<0.05). Sampling intervals for GRP and AVP were 30 min and 60 min, respectively. Values are the mean±SEM.
DISCUSSION
The nature of in vivo synaptic release of GRP and AVP from the SCN was assessed for the first time in freely-behaving hamsters using microdialysis procedures optimized for neuropeptide measurement. These peptides have long received extensive attention, as they are implicated as key players in SCN neuronal integration and output signaling. To date, however, there is scant in vivo information on the nature of their daily regulation in the SCN. Under LD, the peptides exhibited distinct daily rhythms of synaptic release, with peak output occurring from early morning to midday. Under DD, GRP had a broadened rhythm with a plateau extending throughout much of the subjective night and morning. Rhythmicity of AVP under DD was similar to that under LD, with the peak near subjective midday and the nadir during the subjective night. These results suggest that the timing of peak GRP neuronal activity is strongly influenced by the LD cycle, and that the oscillator driving its activity is distinct from that regulating AVP neuronal activity. This latter conclusion is consistent with the study of Shinohara et al. (1995) that provided evidence for separate oscillators controlling the output of AVP and VIP (another mediator of photic signaling) in the cultured rat SCN.
Gastrin-releasing peptide
Cells expressing GRP are located in the retinorecipient core region of the SCN where they receive direct retinal contact (Abrahamson and Moore, 2001; Johnson et al., 1988; Karatsoreos et al., 2004; Morin et al., 2006; Tanaka et al., 1997). Several lines of evidence strongly implicate these cells in the neurophysiological cascade for photic entrainment of the clock. GRP neurons are associated with calbindin-D28K that is necessary for photic signaling (Hamada et al., 2003; LeSauter et al., 2002). Expression of c-fos as well as per1 is also observed in GRP neurons following phase-resetting light pulse exposure (Aioun et al., 1998; Dardente et al., 2002). Application of GRP to the SCN in vivo and in vitro mimics the effects of light (Piggins et al., 1994), including the induction of phase-delays and phase-advances (McArthur et al., 2000), increases in per gene (Gamble et al., 2007) and Fos protein expression (Piggins et al., 2005). Correspondingly, the phase-shifting responses to exogenous GRP are blocked by GRP antagonists (McArthur et al., 2000). In addition, mice lacking the GRP receptor show reduced entrainment responses to light (Aida et al., 2002). Consistent with the present results, the production of GRP appears to be regulated by light, as there is a diurnal rhythm in its content, immunoreactivity and mRNA expression under LD (with higher levels during the day) but not under DD (Shinohara et al., 1993). Notably, mouse GRP receptor levels and binding also exhibit diurnal rhythmicity under LD, both increasing throughout the day to peak near ZT 12 (Karatsoreos et al., 2006). SCN GRP neurons project dorsally to a cap region that reacts to GRP injection in a manner involving phosphorylation of extracellular signal-related kinases (ERK) that is downstream from events in retinorecipient cells. It is proposed that GRP is an output signal of retinorecipient units that activate a group of cells in the dorsal cap region that in turn relay photic entraining information to pacemaker cells of the SCN characterized by AVP expression (Moore et al., 2002). Additionally, GRP-containing neurons originating in the SCN have been identified in the paraventricular nucleus, which is an integrator of signals in the SCN-to-pineal gland pathway (Kalsbeek et al., 1993). This may indicate a role for GRP in the transmission of photic information to the pineal gland, which in turn could affect melatonin production and secretion.
After 2 wks of DD the hamster SCN GRP release rhythm is markedly altered, with an extended 11 hr plateau of relatively elevated output extending throughout much of the subjective night and morning with trough-to-peak amplitude attenuated compared to that under LD (190% vs. 330%, respectively). This release pattern differs qualitatively with observations that the daily rhythm of SCN GRP content in rats under DD for 3 days is lost, rather than blunted (Shinohara et al., 1993). The reason for the difference in the degree of effect of DD on GRP rhythmicity between this and the present study is not clear, but may relate to species and/or methodological differences. It is also not understood if/how tissue content measurements reflect release synaptic activity. Nevertheless, both results support the view that GRP neuronal activity is strongly influenced by the LD cycle, which is consistent with demonstrations that GRP neurons receive direct excitatory retinal input (Karatsoreos et al., 2004; Mikkelson et al., 1991; Morin et al., 2006; Nishino and Koizumi, 1977; Shibata et al., 1984), that they are activated by photic stimuli (Shinohara et al., 1993), and that GRP release is stimulated by localized perfusion of the SCN with NMDA or by a light pulse at ZT 13–14 (present results). In this context, the persistence of the blunted daily fluctuation of GRP release in the absence of photic cues seen here suggests that hamster GRP neuronal activity is timed by an underlying pacemaker whose phase is normally synchronized to the LD cycle. Although the functional significance of the (rhythmic) GRP release under DD is unclear, it would be of value to undertake a future experiment to determine if circadian activity under DD could be entrained to a regimen of exogenous GRP pulses delivered daily to the SCN at CT 4, corresponding to the time of peak GRP output under LD.
The effect of serotonergic stimulation on GRP release was investigated in view of the established role of 5-HT in the regulation of the SCN photic signaling cascade. We and others have shown that the activation of presynaptic 5-HTB receptors on SCN RHT terminals, and 5-HT1A and/or 5-HT7 receptors on retinorecipient target cells significantly attenuates photic phase-resetting responses (Rea and Pickard, 2000; Rea et al., 1994; Selim et al., 1993). Correspondingly, treatment with 5-HT antagonists enhances photic phase-resetting (Gannon, 2003; Rea et al., 1995; Sterniczuk et al., 2008). Notably, it has been also been shown in hamsters that the 5-HT1A,2,7 receptor antagonist, NAN-190 potentiates NMDA-induced Fos expression in SCN retinorecipient (GRP) cells, suggesting that the activity of these cells is attenuated by one or more of these receptor subtypes (Sterniczuk et al., 2008). This fits with the present observation that GRP release is acutely suppressed by intra-SCN perfusion with the 5-HT1A,7 receptor agonist, 8-OH-DPAT.
Arginine vasopressin
Circadian patterns of AVP have been measured in cerebrospinal fluid of cats, guinea pigs, monkeys, rabbits, and rats (Cayetanot et al., 2005; Gerkema et al., 1994; Gunther et al., 1984; Perlow et al., 1982; Schwartz et al., 1983). For each, levels were highest in the morning, and lowest at the end of the light period. The SCN is responsible for timing this rhythm, as it is abolished after SCN lesioning (Jolkkonen et al., 1988; Schwartz and Reppert, 1985). Similar rhythmic output of AVP intrinsic to the SCN has also been confirmed by studies of its in vitro secretion (Earnest and Sladek, 1987; Gillette and Reppert, 1987; Watanabe et al., 1993) and by in vivo microdialysis in rats (Kalsbeek et al., 1995), where output was highest around midday, and low during the early night. The present results in hamsters under LD are consistent with these findings, in that output was highest from early morning to midday and lowest at lights-off. In addition, the present results extend those of the previous microdialysis study by verifying that the AVP (and GRP) measured in SCN microdialysate were from synaptic sources (as opposed to procedural artifact), and that essentially the same temporal expression pattern in AVP output persists after long-term (2 wk) DD exposure. This result is consistent with other in vivo and in vitro studies on the effects of constant conditions on AVP content, and its mRNA expression (Cagampang et al., 1994; Dardente et al., 2004; Tominaga et al., 1992; Yambe et al., 2002) and per1 mRNA expression in AVP cells (Hamada et al., 2004). It has been suggested that the daily rhythm of AVP release involves gap junctional coupling between AVP and other cells in the SCN, as gap junction blockers abolish this rhythm in vitro (Shinohara et al., 2000). Collectively these results, including ours, demonstrate that the rhythm in SCN AVP activity is clock driven (Jin et al., 1999) and independent of photic cues. The present results show further that AVP neuronal activity may also be independent of any direct effect of SCN regulatory photic signaling since localized reverse-microdialysis perfusion of the SCN with 8-OH-DPAT did not acutely affect AVP release. A similar result was obtained in vitro, where NMDA did not acutely alter SCN AVP secretion, although it significantly shifted the period of the AVP secretory rhythm in vitro (Shinohara et al., 1995). Also, it has been shown that AVP cells do not respond immediately (in the form of immediate early gene or clock gene expression) to photic stimulation (Antle and Silver, 2005).
In terms of its function, AVP plays critical roles in the regulation of SCN circadian timekeeping. Notably, this peptide serves as one of the major efferent signals of the SCN to other brain areas (Buijis, 1996), including the paraventricular nucleus and dorsomedial hypothalmus that are associated with hypothalamo-pituitary-adrenal axis regulation (Kalsbeek et al., 2008; Palm et al., 1999). It has been shown that in rats, increased AVP release from the SCN during the morning inhibits corticosterone secretion possibly by acting through a GABAergic intermediate (Kalsbeek et al., 2008). Correspondingly, decreasing AVP release at the end of the light period promotes the daily rise in corticosterone seen in nocturnal animals (Kalsbeek et al., 1996; 1998; 2006). This is also within the time-frame for the beginning of the daily nadir of the SCN AVP rhythm we report here for the hamster under LD and DD. Thus, the daily rhythm in SCN AVP release apparently times the occurrence of the daily corticosterone peak that begins around ZT 6, and importantly, this same circadian pattern persists under free-running conditions. Interestingly, in a diurnal rodent species (Arvicanthis ansorgei), SCN AVP has a stimulatory effect on corticosterone secretion, suggesting that in diurnals it acts through an excitatory neuronal pathway (Kalsbeek et al., 2008). The present finding that reverse-microdialysis perfusion of the SCN with 8-OH-DPAT suppresses the daytime peak in AVP release indicates that serotonergic input could modulate the amplitude of the AVP rhythm. Such a serotonergic inhibition of AVP would theoretically increase corticosterone secretion, which is consistent with the stimulatory effect of the 5-HT agonists on blood corticosterone levels (van de Kar, 1991).
In addition to its efferent signaling actions discussed above, there is morphological (Castel et al., 1990) and neurophysiological evidence (see below) that AVP acts locally in the SCN to modulate its own and other neuronal activities. The daily rhythm of spontaneous electrical activity in SCN neurons is similar to that of AVP release (both peaking near the middle of the light phase) and it has also been suggested that AVP also plays a role in intra-SCN communication by synchronizing oscillations generated by individual SCN neurons (Mihai et al., 1994). It is notable that while AVP mRNA and synaptic release both peak during the day, the V1a receptor through which it acts is expressed 180° out of phase at night (Young et al., 1993). The physiological basis for this temporal ligand-receptor expression pattern is unclear, but a slow rate of receptor turnover throughout the dark phase when AVP tonus is relatively low could theoretically result in a large pool of up-regulated receptors in the morning during peak AVP release, leading to the potentiation of neuronal electrical activity seen at this time. Additionally, circadian locomotor activity and prokineticin 2 gene expression were attenuated in mice lacking the V1a receptor, suggesting that AVP released within the SCN may regulate the expression of clock-related genes (Li et al., 2009).
In summary, the present microdialysis-RIA procedures reveal that GRP and the clock-driven peptide, AVP, exhibit daily rhythmic changes in release under LD in the hamster SCN, each peaking near midday. It is notable that neurons producing these peptides express the clock gene, per1 (Hamada et al., 2004), and it is therefore important to consider their rhythmic activity in the context of recent findings that the firing of mouse SCN neurons containing per1 is moderate in the morning, silenced in the afternoon and moderate again at dusk (Belle et al., 2009). The release profile of AVP corresponds to this daytime firing pattern of per1-producing neurons, as peptide release increases steadily during early morning and drops abruptly near the time when per1 cells have stopped firing_(assuming this also happens in hamsters). Thus, the underlying rhythm in per1 expression could be important to the timing of peptide release in the SCN.
Acknowledgments
Supported by National Institutes of Health grant NS-35229 to J.D. Glass
Abbreviations
- ACSF
artificial cerebrospinal fluid
- AVP
arginine vasopressin
- CT
circadian time
- DD
constant darkness
- ERK
extracellular signal-related kinases
- GHT
geniculohypothalamic tract
- GRP
gastrin-releasing peptide
- LD, light
dark cycle
- NMDA
N-methyl-D-aspartic acid
- RHT
retinohypothalamic tract
- SCN
suprachiasmatic nucleus
- VIP
vasoactive intestinal polypeptide
- ZT
zeitgeber time
- 5-HT
serotonin
- 8-OH-DPAT
(+)-8-hydroxydipropylaminotetralin hydrobromide
References
- Abe H, Rusak B, Robertson HA. Photic induction of fos protein in the suprachiasmatic nucleus is inhibited by the NMDA receptor antagonist MK-801. Neurosci Lett. 1991;127:9–12. doi: 10.1016/0304-3940(91)90881-s. [DOI] [PubMed] [Google Scholar]
- Abe H, Robertson HA, Rusak B. NMDA and non-NMDA receptor antagonists inhibit photic induction of Fos protein in the hamster suprachiasmatic nucleus. Brain Res Bull. 1992;28:831–835. doi: 10.1016/0361-9230(92)90269-4. [DOI] [PubMed] [Google Scholar]
- Abrahamson EE, Moore RY. Suprachiasmatic nucleus in the mouse: retinal innervation, intrinsic organization and efferent projections. Brain Res. 2001;916:172–191. doi: 10.1016/s0006-8993(01)02890-6. [DOI] [PubMed] [Google Scholar]
- Aida R, Moriya T, Araki M, Akiyama M, Wada K, Wada E, Shibata S. Gastrin-releasing peptide mediates photic entrainable signals to dorsal subsets of suprachiasmatic nucleus via induction of Period gene in mice. Mol Pharmacol. 2002;61:26–34. doi: 10.1124/mol.61.1.26. [DOI] [PubMed] [Google Scholar]
- Aioun J, Chambille I, Peytevin J, Martinet L. Neurons containing gastrin-releasing peptide and vasoactive intestinal polypeptide are involved in the reception of the photic signal in the suprachiasmatic nucleus of the Syrian hamster: an immunocytochemical ultrastructural study. Cell Tissue Res. 1998;291:239–253. doi: 10.1007/s004410050994. [DOI] [PubMed] [Google Scholar]
- Albers HE, Ferris CF. Neuropeptide Y: role in light-dark cycle entrainment of hamster circadian rhythms. Neurosci Lett. 1984;50:163–168. doi: 10.1016/0304-3940(84)90480-4. [DOI] [PubMed] [Google Scholar]
- Antle MC, Kriegsfeld LJ, Silver R. Signaling within the master clock of the brain: localized activation of mitogen-activated protein kinase by gastrin-releasing peptide. J Neurosci. 2005;25:2447–2454. doi: 10.1523/JNEUROSCI.4696-04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Antle MC, Silver R. Orchestrating time: arrangements of the brain circadian clock. Trends Neurosci. 2005;28:145–151. doi: 10.1016/j.tins.2005.01.003. [DOI] [PubMed] [Google Scholar]
- Belle MDC, Diekman CO, Forger DB, Piggins HD. Daily electrical silencing in the mammalian circadian clock. Science. 2009;326:281–284. doi: 10.1126/science.1169657. [DOI] [PubMed] [Google Scholar]
- Benveniste H. Brain microdialysis. J Neurochem. 1989;52:1667–1669. doi: 10.1111/j.1471-4159.1989.tb07243.x. [DOI] [PubMed] [Google Scholar]
- Biello SM, Janik D, Mrosovsky N. Neuropeptide Y and behaviorally induced phase shifts. Neuroscience. 1994;62:273–279. doi: 10.1016/0306-4522(94)90331-x. [DOI] [PubMed] [Google Scholar]
- Buijs RM. The anatomical basis for the expression of circadian rhythms: the efferent projections of the suprachiasmatic nucleus. Prog Brain Res. 1996;111:229–240. doi: 10.1016/s0079-6123(08)60411-2. [DOI] [PubMed] [Google Scholar]
- Cagampang FRA, Yang J, Nakayama Y, Fukuhara C, Inouye S. Circadian variation of arginine-vasopressin messenger RNA in the rat suprachiasmatic nucleus. Mol Brain Res. 1994;24:179–184. doi: 10.1016/0169-328x(94)90130-9. [DOI] [PubMed] [Google Scholar]
- Card JP, Moore RY. Ventral lateral geniculate nucleus efferents to the rat suprachiasmatic nucleus exhibit avian pancreatic polypeptide-like immunoreactivity. J Comp Neurol. 1982;206:390–396. doi: 10.1002/cne.902060407. [DOI] [PubMed] [Google Scholar]
- Castel M, Feinstein N, Cohen S, Harari N. Vasopressinergic innervation of the mouse suprachiasmatic nucleus: an immuno-electron microscopic analysis. J Comp Neur. 1990;298:172–187. doi: 10.1002/cne.902980204. [DOI] [PubMed] [Google Scholar]
- Cayetanot F, Bentivoglio M, Aujard F. Arginine-vasopressin and vasointestinal polypeptide rhythms in the suprachiasmatic nucleus of the mouse lemur reveal aging-related alterations of circadian pacemaker neurons in a non-human primate. Eur J Neurosci. 2005;22:902–910. doi: 10.1111/j.1460-9568.2005.04268.x. [DOI] [PubMed] [Google Scholar]
- Dardente H, Poirel V, Klosen P, Pévet P, Masson-Pévet M. Per and neuropeptide expression in the rat suprachiasmatic nuclei: compartmentalization and differential cellular induction by light. Brain Res. 2002;958:261–271. doi: 10.1016/s0006-8993(02)03563-1. [DOI] [PubMed] [Google Scholar]
- Dardente H, Menet JS, Challet E, Tournier BB, Pévet P, Masson-Pévet M. Daily and circadian expression of neuropeptides in the suprachiasmatic nuclei of nocturnal and diurnal rodents. Mol Brain Res. 2004;124:143–151. doi: 10.1016/j.molbrainres.2004.01.010. [DOI] [PubMed] [Google Scholar]
- Ding JM, Chen D, Weber ET, Faiman LE, Rea MA, Gillette MU. Resetting the biological clock: mediation of nocturnal circadian shifts by glutamate and NO. Science. 1994;266:1713–1717. doi: 10.1126/science.7527589. [DOI] [PubMed] [Google Scholar]
- Dudley TE, DiNardo LA, Glass JD. Endogenous regulation of serotonin release in the hamster suprachiasmatic nucleus. J Neurosci. 1998;18:5045–5052. doi: 10.1523/JNEUROSCI.18-13-05045.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Earnest DJ, Sladek CD. Circadian rhythms of vasopressin release from individual rat suprachiasmatic explants in vitro. Brain Res. 1987;382:129–133. doi: 10.1016/0006-8993(86)90119-8. [DOI] [PubMed] [Google Scholar]
- Earnest DJ, DiGiorgio S, Olschowka JA. Light induces expression of fos-related proteins within gastrin-releasing peptide neurons in the rat suprachiasmatic nucleus. Brain Res. 1993;627:205–209. doi: 10.1016/0006-8993(93)90322-e. [DOI] [PubMed] [Google Scholar]
- Gamble KL, Novak CM, Albers HA. Neuropeptide Y and N-methyl-D-aspartic acid interact within the suprachiasmatic nuclei to alter circadian phase. Neuroscience. 2004;126:559–565. doi: 10.1016/j.neuroscience.2004.04.018. [DOI] [PubMed] [Google Scholar]
- Gamble KL, Allen GC, Zhou T, McMahon DG. Gastrin-releasing peptide mediates light-like phase resetting of the suprachiasmatic nucleus circadian pacemaker through cAMP response element-binding protein and Per1 activation. J Neurosci. 2007;27:12078–12087. doi: 10.1523/JNEUROSCI.1109-07.2007. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gannon RL. Serotonergic serotonin1A mixed agonists/antagonists elicit large-magnitude phase shifts in hamster circadian wheel-running rhythms. Neuroscience. 2003;119:567–576. doi: 10.1016/s0306-4522(03)00161-1. [DOI] [PubMed] [Google Scholar]
- Gerkema MP, Van der Zee EA, Feitsma LE. Expression of circadian rhythmicity correlates with the number of arginine-vasopressin-immunoreactive cells in the suprachiasmatic nucleus of common voles, Microtus arvalis. Brain Res. 1994;639:93–101. doi: 10.1016/0006-8993(94)91768-x. [DOI] [PubMed] [Google Scholar]
- Gillette MU, Reppert SM. The hypothalamic suprachiasmatic nuclei: circadian patterns of vasopressin secretion and neuronal activity in vitro. Brain Res Bull. 1987;19:135–139. doi: 10.1016/0361-9230(87)90176-6. [DOI] [PubMed] [Google Scholar]
- Glass JD, Grossman G, Farnbauch L, DiNardo L. Midbrain raphe modulation of nonphotic circadian clock resetting and 5-HT release in the mammalian suprachiasmatic nucleus. J Neurosci. 2003;23:7451–7460. doi: 10.1523/JNEUROSCI.23-20-07451.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunther O, Landgraf R, Schuart J, Unger H. Vasopressin in cerebrospinal fluid (CSF) and plasma of conscious rabbits--circadian variations. Exp Clin Endocrinol. 1984;83:367–369. doi: 10.1055/s-0029-1210357. [DOI] [PubMed] [Google Scholar]
- Hamada T, LeSauter J, Lokshin M, Romero MT, Yan L, Venuti JM, Silver R. Calbindin influences response to photic input in suprachiasmatic nucleus. J Neurosci. 2003;23:8820–8826. doi: 10.1523/JNEUROSCI.23-26-08820.2003. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hamada T, Antle M, Silver R. The role of Period1 in nonphotic resetting of the hamster circadian pacemaker in the suprachiasmatic nucleus. Neursci Lett. 2004;362:87–90. doi: 10.1016/j.neulet.2004.02.061. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jin X, Shearman LP, Weaver DR, Zylka MJ, De Vries GJ. A molecular mechanism regulating output from the suprachiasmatic circadian clock. Cell. 1999;96:57–68. doi: 10.1016/s0092-8674(00)80959-9. [DOI] [PubMed] [Google Scholar]
- Johnson RF, Morin LP, Moore RY. Retinohypothalamic projections in the hamster and rat demonstrated using cholera toxin. Brain Res. 1988;462:301–312. doi: 10.1016/0006-8993(88)90558-6. [DOI] [PubMed] [Google Scholar]
- Johnson RF, Moore RY, Morin LP. Lateral geniculate lesions alter circadian activity rhythms in the hamster. Brain Res Bull. 1989;22:411–422. doi: 10.1016/0361-9230(89)90068-3. [DOI] [PubMed] [Google Scholar]
- Jolkkonen J, Tuomisto L, van Wimersma Greidanus TN, Riekkinen PJ. Vasopressin levels in the cerebrospinal fluids of rats with lesions of the paraventricular and suprachiasmatic nuclei. Neurosci Lett. 1988;86:184–188. doi: 10.1016/0304-3940(88)90568-x. [DOI] [PubMed] [Google Scholar]
- Kalsbeek A, Teclemariam-Mesbah R, Pévet P. Efferent projections of the suprachiasmatic nucleus in the golden hamster (Mesocricetus auratus) J Comp Neurol. 1993;332:293–314. doi: 10.1002/cne.903320304. [DOI] [PubMed] [Google Scholar]
- Kalsbeek A, Buijs RM, Engelmann M, Wotjak CT, Landgraf R. In vivo measurement of a diurnal variation in vasopressin release in the rat suprachiasmatic nucleus. Brain Res. 1995;682:75–82. doi: 10.1016/0006-8993(95)00324-j. [DOI] [PubMed] [Google Scholar]
- Kalsbeek A, van Heerikhuize J, Wortel J, Buijs R. A diurnal rhythm of stimulatory input to the hypothalamo-pituitary-adrenal system as revealed by time intrahypothalamic administration of the vasopressin V1 antagonist. J Neurosci. 1996;16:5555–5565. doi: 10.1523/JNEUROSCI.16-17-05555.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kalsbeek A, van Heerikhuize JJ, Wortel J, Buijs RM. Restricted daytime feeding modifies suprachiasmatic nucleus vasopressin release in rats. J Biol Rhythms. 1998;13:18–29. doi: 10.1177/074873098128999880. [DOI] [PubMed] [Google Scholar]
- Kalsbeek A, Palm IF, La Fleur SE, Scheer FAJL, Perreau-Lenz S, Ruiter M, Kreier F, Cailotto C, Buijs RM. SCN outputs and the hypothalamic balance of life. J Biol Rhythms. 2006;21:458–469. doi: 10.1177/0748730406293854. [DOI] [PubMed] [Google Scholar]
- Kalsbeek A, Verhagen L, Schalij I, Foppen E, Saboureau M, Bothorel B, Buijs R, Pévet P. Opposite actions of hypothalamic vasopressin on circadian corticosterone rhythm in nocturnal versus diurnal species. Eur J Neursci. 2008;27:818–827. doi: 10.1111/j.1460-9568.2008.06057.x. [DOI] [PubMed] [Google Scholar]
- Karatsoreos IN, Yan L, LeSauter J, Silver R. Phenotype matters: identification of light-responsive cells in the mouse suprachiasmatic nucleus. J Neurosci. 2004;24:68–75. doi: 10.1523/JNEUROSCI.1666-03.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karatsoreos IN, Romeo RD, McEwen BS, Silver R. Diurnal regulation of the gastrin-releasing peptide receptor in the mouse circadian clock. Eur J Neurosci. 2006;23:1047–1053. doi: 10.1111/j.1460-9568.2006.04633.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klein DC, Moore RY, Reppert SM. Suprachiasmatic nucleus: the mind’s clock. Oxford University Press; New York: 1991. [Google Scholar]
- LeSauter J, Kriegsfeld LJ, Hon J, Silver R. Calbindin-D28k cells selectively contact intra-SCN neurons. Neuroscience. 2002;111:575–585. doi: 10.1016/s0306-4522(01)00604-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li JD, Burton KJ, Zhang C, Hu SB, Zhou QY. Vasopressin receptor V1a regulates circadian rhythms of locomotor activity and expression of clock-controlled genes in the suprachiasmatic nuclei. Am J Physiol Regul Integr Comp Physiol. 2009;296:R824–R830. doi: 10.1152/ajpregu.90463.2008. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Marchant EG, Watson NV, Mistlberger RE. Both neuropeptide Y and serotonin are necessary for entrainment of circadian rhythms in mice by daily treadmill running schedules. J Neurosci. 1997;17:7974–7987. doi: 10.1523/JNEUROSCI.17-20-07974.1997. [DOI] [PMC free article] [PubMed] [Google Scholar]
- McArthur A, Coogan A, Ajpru S, Sugden D, Biello S, Piggins H. Gastrin-releasing peptide phase-shifts suprachiasmatic nuclei neuronal rhythms in vitro. J Neurosci. 2000;20:5496–5502. doi: 10.1523/JNEUROSCI.20-14-05496.2000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Meyer-Bernstein EL, Morin LP. Differential serotonergic innervation of the suprachiasmatic nucleus and the intergeniculate leaflet and its role in circadian rhythm modulation. J Neurosci. 1996;16:2097–2111. doi: 10.1523/JNEUROSCI.16-06-02097.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mihai R, Juss TS, Ingram CD. Suppression of suprachiasmatic nucleus neurone activity with a vasopressin receptor antagonist: possible role for endogenous vasopressin in circadian activity cycles in vitro. Neurosci Lett. 1994;179:95–99. doi: 10.1016/0304-3940(94)90943-1. [DOI] [PubMed] [Google Scholar]
- Mikkelson JD, Larsen PJ, O’Hare MMT, Wiegand SJ. Gastrin releasing peptide in the rat suprachiasmatic nucleus: an immunohisotchemical, chromatographic and radioimmunological study. Neuroscience. 1991;40:55–66. doi: 10.1016/0306-4522(91)90174-m. [DOI] [PubMed] [Google Scholar]
- Moore RY, Lenn NJ. A retinohypothalamic projection in the rat. J Comp Neurol. 1972;146:1–14. doi: 10.1002/cne.901460102. [DOI] [PubMed] [Google Scholar]
- Moore RY. Organization and function of a central nervous system circadian oscillator: the suprachiasmatic hypothalamic nucleus. Fed Proc. 1983;42:2783–2789. [PubMed] [Google Scholar]
- Moore RY, Speh JC, Leak RK. Suprachiasmatic nucleus organization. Cell Tissue Res. 2002;309:89–98. doi: 10.1007/s00441-002-0575-2. [DOI] [PubMed] [Google Scholar]
- Morin LP, Shivers K-Y, Blanchard JH, Muscat L. Complex organization of mouse and rat suprachiasmatic nucleus. Neuroscience. 2006;137:1285–1297. doi: 10.1016/j.neuroscience.2005.10.030. [DOI] [PubMed] [Google Scholar]
- Nishino H, Koizumi K. Responses of neurons in the suprachiasmatic nuclei of the hypothalamus to putative transmitters. Brain Res. 1977;120:176–172. doi: 10.1016/0006-8993(77)90509-1. [DOI] [PubMed] [Google Scholar]
- Palm IF, van der Beek EM, Wiegant VM, Buijs RM, Kalsbeek A. Vasopressin induces an LH surge in ovariectomized, estradiol-treated rats with lesion of the suprachiasmatic nucleus. Neuroscience. 1999;93:659–666. doi: 10.1016/s0306-4522(99)00106-2. [DOI] [PubMed] [Google Scholar]
- Perlow MJ, Reppert SM, Artman HA, Fisher DA, Seif SM, Robinson AG. Oxytocin, vasopressin, and estrogen-stimulated neurophysin: daily patterns of concentration in cerebrospinal fluid. Science. 1982;216:1416–1418. doi: 10.1126/science.7201163. [DOI] [PubMed] [Google Scholar]
- Pickard GE. The afferent connections of the suprachiasmatic nucleus of the golden hamster with emphasis on the retinohypothalamic projection. J Comp Neurol. 1982;211:65–83. doi: 10.1002/cne.902110107. [DOI] [PubMed] [Google Scholar]
- Piggins HD, Cutler DJ, Rusak B. Effects of ionophoretically applied bombesin-like peptides on hamster suprachiasmatic nucleus neurons in vitro. Eur J Pharmacol. 1994;271:413–419. doi: 10.1016/0014-2999(94)90801-x. [DOI] [PubMed] [Google Scholar]
- Piggins HD, Antle MC, Rusak B. Neuropeptides phase shift the mammalian circadian pacemaker. J Neurosci. 1995;15:5612–5622. doi: 10.1523/JNEUROSCI.15-08-05612.1995. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Piggins HD, Goguen D, Rusak B. Gastrin-releasing peptide induces c-Fos in the hamster suprachiasmatic nucleus. Neurosci Lett. 2005;384:205–210. doi: 10.1016/j.neulet.2005.03.072. [DOI] [PubMed] [Google Scholar]
- Rea MA, Buckley B, Lutton LM. Local administration of EAA antagonists blocks light-induced phase shifts and c-fos expression in hamster SCN. J Physiol. 1993;265:R1191–R1198. doi: 10.1152/ajpregu.1993.265.5.R1191. [DOI] [PubMed] [Google Scholar]
- Rea MA, Glass JD, Colwell CS. Serotonin modulates photic responses in the hamster suprachiasmatic nuclei. J Neurosci. 1994;14:3635–3642. doi: 10.1523/JNEUROSCI.14-06-03635.1994. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rea MA, Barrera J, Glass JD, Gannon RL. Serotonergic potentiation of photic phase shifts of the circadian activity rhythm. Neuroreport. 1995;6:1417–1420. doi: 10.1097/00001756-199507100-00014. [DOI] [PubMed] [Google Scholar]
- Rea MA, Pickard GE. Serotonergic modulation of photic entrainment in the Syrian hamster. Biol Rhythm Res. 2000;31:284–314. [Google Scholar]
- Reghunandanan V, Reghunandanan R. Neurotransmitters of the suprachiasmatic nuclei. J Circadian Rhythms. 2006;4:2. doi: 10.1186/1740-3391-4-2. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rusak B, Zucker I. Neural regulation of circadian rhythms. Physiol Rev. 1979;59:449–526. doi: 10.1152/physrev.1979.59.3.449. [DOI] [PubMed] [Google Scholar]
- Schwartz WJ, Coleman RJ, Reppert SM. A daily vasopressin rhythm in rat cerebrospinal fluid. Brain Res. 1983;263:105–112. doi: 10.1016/0006-8993(83)91205-2. [DOI] [PubMed] [Google Scholar]
- Schwartz WJ, Reppert SM. Neural regulation of the circadian vasopressin rhythm in cerebrospinal fluid: a pre-eminent role for the suprachiasmatic nuclei. J Neurosci. 1985;5:2771–2778. doi: 10.1523/JNEUROSCI.05-10-02771.1985. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Selim M, Glass JD, Hauser UE, Rea MA. Serotonergic inhibition of light-induced Fos protein expression and extracellular glutamate in the suprachiasmatic nuclei. Brain Res. 1993;621:181–188. doi: 10.1016/0006-8993(93)90105-v. [DOI] [PubMed] [Google Scholar]
- Shibata S, Oomura Y, Hattori K, Hitoshi K. Responses of suprachiasmatic nucleus neurons to optic nerve stimulation in rat hypothalamic slice preparation. Brain Res. 1984;302:83–89. doi: 10.1016/0006-8993(84)91287-3. [DOI] [PubMed] [Google Scholar]
- Shinohara K, Tominaga K, Isobe Y, Inouye ST. Photic regulation of peptides located in the ventrolateral subdivision of the suprachiasmatic nucleus of the rat: daily variations of vasoactive intestinal polypeptide, gastrin-releasing peptide, and neuropeptide Y. J Neurosci. 1993;13:793–800. doi: 10.1523/JNEUROSCI.13-02-00793.1993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinohara K, Honma S, Katsuno Y, Abe H, Honma K. Two distinct oscillators in the rat suprachiasmatic nucleus in vitro. Proc Natl Acad Sci USA. 1995;92:7396–7400. doi: 10.1073/pnas.92.16.7396. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shinohara K, Hiruma H, Funabashi T, Kimura F. GABAergic modulation of gap junction communication in slice cultures of the rat suprachiasmatic mucleus. Neuroscience. 2000;96:591–596. doi: 10.1016/s0306-4522(99)00556-4. [DOI] [PubMed] [Google Scholar]
- Shirakawa T, Honma S, Honma K. Multiple oscillators in the suprachiasmatic nucleus. Chronobiol Int. 2001;18:371–387. doi: 10.1081/cbi-100103962. [DOI] [PubMed] [Google Scholar]
- Sterniczuk R, Stepkowski A, Jones M, Antle MC. Enhancement of photic shifts with the 5-HT1A mixed agonist/antagonist NAN-190: intra-suprachiasmatic nucleus pathway. Neuroscience. 2008;153:571–580. doi: 10.1016/j.neuroscience.2008.02.062. [DOI] [PubMed] [Google Scholar]
- Tanaka M, Hayashi S, Tamada Y, Ikeda T, Hisa Y, Takamatsu T, Ibata Y. Direct retinal projections to GRP neurons in the suprachiasmatic nucleus of the rat. Neuroreport. 1997;8:2187–2191. doi: 10.1097/00001756-199707070-00020. [DOI] [PubMed] [Google Scholar]
- Tominaga K, Shinohara K, Otori Y, Fukuhara C, Inouye ST. Circadian rhythms of vasopressin content in the suprachiasmatic nucleus of the rat. Neuroreport. 1992;3:809–812. doi: 10.1097/00001756-199209000-00022. [DOI] [PubMed] [Google Scholar]
- Van de Kar LD. Neuroendocrine pharmacology of serotonergic (5-HT) neurons. Annu Rev Pharmacol Toxicol. 1991;31:289–320. doi: 10.1146/annurev.pa.31.040191.001445. [DOI] [PubMed] [Google Scholar]
- Watanabe K, Koibuchi N, Ohtake H, Yamaoka S. Circadian rhythms of vasopressin release in primary cultures of rat suprachiasmatic nucleus. Brain Res. 1993;624:115–120. doi: 10.1016/0006-8993(93)90067-w. [DOI] [PubMed] [Google Scholar]
- Yambe Y, Arima H, Kakiya S, Murase T, Oiso Y. Diurnal changes in arginine vasopressin gene transcription in the rat suprachiasmatic nucleus. Mol Brain Res. 2002;104:132–136. doi: 10.1016/s0169-328x(02)00327-3. [DOI] [PubMed] [Google Scholar]
- Young WSI, Kovács K, Lolait SJ. The diurnal rhythm in vasopressin V1a receptor expression in the suprachiasmatic nucleus is not dependent on vasopressin. Endocrinology. 1993;133:585–590. doi: 10.1210/endo.133.2.8344200. [DOI] [PubMed] [Google Scholar]
- Zar JH. Biostatistical analysis. Prentice Hall; Englewood Cliffs: 1983. [Google Scholar]