Abstract
The fibrillins are a large family of chloroplast proteins that have been linked with stress tolerance and disease resistance. FIBRILLIN4 (FIB4) is found associated with the photosystem II light-harvesting complex, thylakoids, and plastoglobules, which are chloroplast compartments rich in lipophilic antioxidants. For this study, FIB4 expression was knocked down in apple (Malus 3 domestica) using RNA interference. Plastoglobule osmiophilicity was decreased in fib4 knockdown (fib4 KD) tree chloroplasts compared with the wild type, while total plastoglobule number was unchanged. Compared with the wild type, net photosynthetic CO2 fixation in fib4 KD trees was decreased at high light intensity but was increased at low light intensity. Furthermore, fib4 KD trees produced more anthocyanins than the wild type when transferred from low to high light intensity, indicating greater sensitivity to high light stress. Relative to the wild type, fib4 KD apples were more sensitive to methyl viologen and had higher superoxide levels during methyl viologen treatment. Arabidopsis (Arabidopsis thaliana) fib4 mutants and fib4 KD apples were more susceptible than their wild-type counterparts to the bacterial pathogens Pseudomonas syringae pathovar tomato and Erwinia amylovora, respectively, and were more sensitive to ozone-induced tissue damage. Following ozone stress, plastoglobule osmiophilicity decreased in wild-type apple and remained low in fib4 KD trees; total plastoglobule number increased in fib4 KD apples but not in the wild type. These results indicate that FIB4 is required for plastoglobule development and resistance to multiple stresses. This study suggests that FIB4 is involved in regulating plastoglobule content and that defective regulation of plastoglobule content leads to broad stress sensitivity and altered photosynthetic activity.
Increased production of reactive oxygen species (ROS) is among the first biochemical responses of plants when challenged by pathogens and harsh environmental conditions (Mehdy, 1994; Lamb and Dixon, 1997; Joo et al., 2005). ROS are implicated in tissue damage during environmental stress and in the promotion of disease development by necrotrophic and hemibiotrophic pathogens (Venisse et al., 2001; Apel and Hirt, 2004; Shetty et al., 2008). For example, ROS production is critical for host colonization and pathogenesis by the bacterium Erwinia amylovora, which causes fire blight disease in rosaceous plants such as apple (Malus 3 domestica) and pear (Pyrus communis; Venisse et al., 2001).
The chloroplast is a site of ROS production during biotic and abiotic stress (Joo et al., 2005; Liu et al., 2007). The chloroplast has a battery of enzymes such as superoxide dismutase and ascorbate peroxidase, and antioxidants such as ascorbate, glutathione, and tocopherols, for protection against ROS (Noctor and Foyer, 1998; Asada, 2006). Plastoglobules are lipoprotein bodies attached to the thylakoids (Austin et al., 2006) that store lipids, including antioxidants such as tocopherols, carotenes, and plastoquinones (Steinmüller and Tevini, 1985; Tevini and Steinmüller, 1985). In addition to antioxidants, plastoglobules contain tocopherol cyclase, which is involved in γ-tocopherol synthesis (Austin et al., 2006; Vidi et al., 2006). The antioxidant content of plastoglobules and their apparent involvement in tocopherol biosynthesis imply that they could play a role in plant responses to oxidative stress.
Plastoglobules contain fibrillins, which were initially described as protein components of chromoplast fibrils with a molecular mass of approximately 30 kD (Winkenbach et al., 1976; Knoth et al., 1986; Emter et al., 1990; Deruère et al., 1994). Fibrillins are ubiquitous proteins present from cyanobacteria to plants (Laizet et al., 2004). Fibrillins maintain plastoglobule structural integrity (Deruère et al., 1994; Pozueta-Romero et al., 1997; Langenkämper et al., 2001; Vidi et al., 2006; Bréhélin et al., 2007) and stabilize the photosynthetic apparatus during photooxidative stress (Gillet et al., 1998; Yang et al., 2006; Youssef et al., 2010), osmotic stress (Gillet et al., 1998), drought (Pruvot et al., 1996; Rey et al., 2000), and low temperature (Rorat et al., 2001). Fibrillins are involved in abscisic acid-mediated protection from photoinhibition (Yang et al., 2006), and a subfamily of Arabidopsis (Arabidopsis thaliana) fibrillins (FIB1a, -1b, and -2) conditions jasmonate production during low-temperature, photooxidative stress (Youssef et al., 2010). Arabidopsis plants lacking one fibrillin (At4g22240) and tomato (Solanum lycopersicum) plants with suppressed expression of a fibrillin (LeCHRC) are susceptible to Pseudomonas syringae and Botrytis cinerea, respectively (Cooper et al., 2003; Leitner-Dagan et al., 2006), indicating that fibrillins play a role in disease resistance.
The Arabidopsis fibrillin encoded by At3g23400 has received various appellations, including FIBRILLIN4 (FIB4; Laizet et al., 2004), Harpin-Binding Protein1 (Song et al., 2002), AtPGL 30.4 (Vidi et al., 2006), and Fibrillin6 (Galetskiy et al., 2008); here, it will be referred to by its earliest published name, FIB4. FIB4 is found associated with the PSII light-harvesting complex (Galetskiy et al., 2008). FIB4 has also been detected in plastoglobules (Vidi et al., 2006; Ytterberg et al., 2006) and thylakoids (Friso et al., 2004; Peltier et al., 2004). However, the specific function of FIB4 is unknown. Several lines of evidence suggest that FIB4 may be involved in plant disease resistance responses: pathogen-associated molecular patterns trigger its phosphorylation (Jones et al., 2006); pathogen-associated molecular patterns stimulate the expression of its ortholog in tobacco (Nicotiana tabacum; Jones et al., 2006; Sanabria and Dubery, 2006); and it can physically interact with the HrpN (harpin) virulence protein of the fire blight pathogen E. amylovora in a yeast two-hybrid assay, suggesting that it could be a receptor or target of HrpN (Song et al., 2002). In addition, it is thought that FIB4 may be involved in the transport of small, hydrophobic molecules because it contains a conserved lipocalin signature (Jones et al., 2006). Here, we report a genetic analysis of FIB4 function in apple and Arabidopsis in terms of its role in plastoglobule development and plant resistance to biotic and abiotic stresses.
RESULTS
Knockdown of FIB4 Expression in Apple
We generated fib4 knockdown (fib4 KD) apple trees using the RNA interference (RNAi) technique in the cv Royal Gala (Supplemental Figs. S1 and S2A; Maximova et al., 1998). Transgenic, vector-only control (VC) plants carrying the transformation marker genes Neomycin Phosphotransferase II and Enhanced Green Fluorescent Protein (EGFP), but not the FIB4 RNAi cassette, were also generated (Supplemental Figs. S1 and S2A). The level of FIB4 transcript in fib4 KD plantlets was less than 10% of the wild-type level, indicating successful silencing (Supplemental Fig. S2B). Wild-type, VC, and fib4 KD apple trees were phenotypically indistinguishable when grown under normal laboratory conditions (Supplemental Fig. S2C).
Plastoglobule Ultrastructure
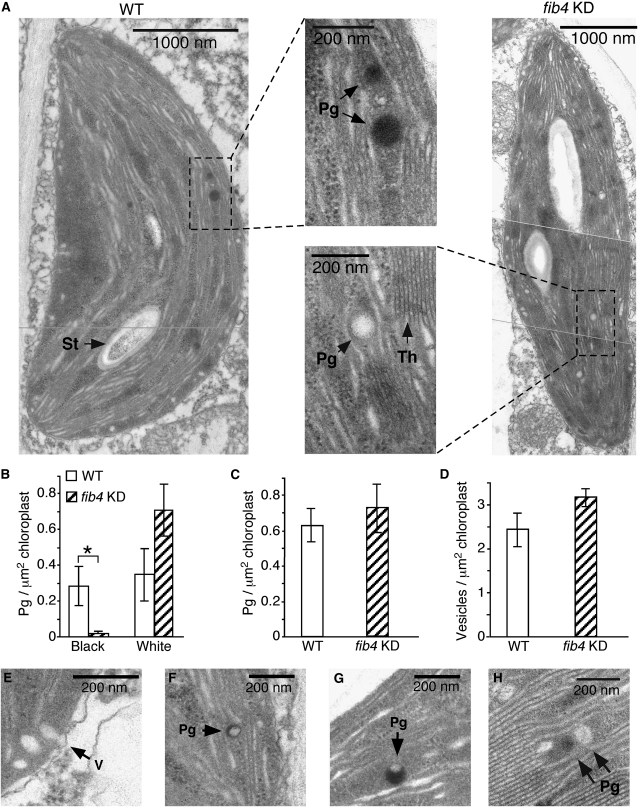
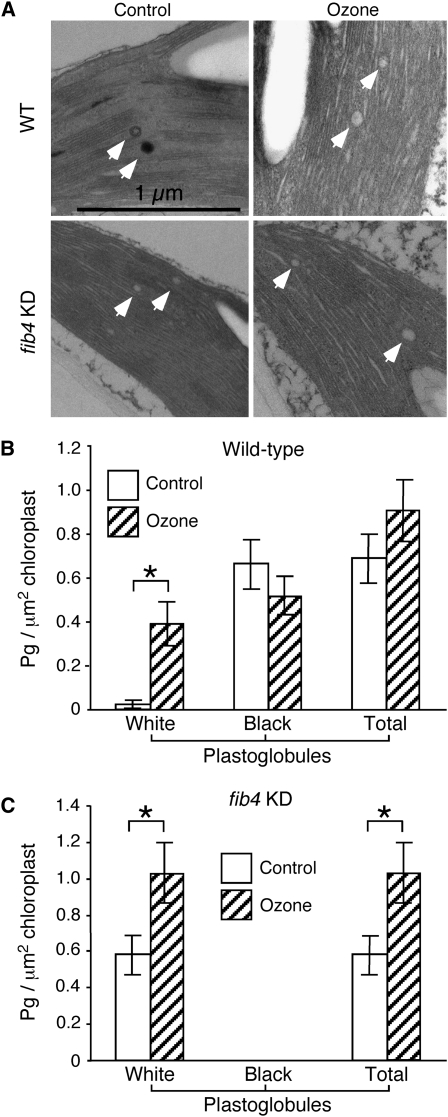
Since FIB4 is a component of the plastoglobule and is associated with the PSII light-harvesting complex, we investigated the effect of knockdown of FIB4 expression on chloroplast and plastoglobule ultrastructure in apple. Leaf sections from fib4 KD and wild-type apple trees were stained with osmium tetroxide (OsO4) and observed by transmission electron microscopy (TEM). Overall chloroplast morphology appeared normal in fib4 KD palisade mesophyll cells (Fig. 1A). Strikingly, however, fib4 KD chloroplasts contained almost exclusively electron-transparent (nonosmiophilic, or white) plastoglobules, while wild-type chloroplasts contained many electron-opaque (osmiophilic, or black) plastoglobules (Fig. 1, A and B). Plastoglobules are thought to be osmiophilic because the unsaturated lipids they contain reduce OsO4 (Padham et al., 2007). There were more white plastoglobules in fib4 KD chloroplasts than wild-type chloroplasts, although the difference was not statistically significant (Fig. 1B). The total number of plastoglobules (electron transparent + electron opaque) was similar in fib4 KD and wild-type chloroplasts (Fig. 1C). Peripheral vesicles were similar in number in fib4 KD and wild-type chloroplasts (Fig. 1D). The peripheral vesicles were electron transparent, irregularly shaped, located adjacent to the chloroplast inner envelope membrane, and clearly distinguishable from the round, white plastoglobules (Fig. 1E). Black plastoglobules had varying patterns of electron opacity ranging from partially to fully electron opaque (Fig. 1, A, F, and G). Black and white plastoglobules were often observed in the same chloroplast, sometimes in juxtaposition (Fig. 1H).
Figure 1.
Effects of FIB4 knockdown on plastoglobule ultrastructure. Trees sampled were 4 to 5 months old and grown under 90 μE m−2 s−1 light intensity with a 12-h photoperiod. A, Transmission electron micrographs of representative wild-type (WT) and fib4 KD palisade mesophyll cell chloroplasts stained with OsO4. Pg, Plastoglobule; St, starch body; Th, thylakoid membrane. For B, C, and D, 80 chloroplasts from six leaves (two leaves from each of three plants) were observed for each genotype; data are means ± se; * P < 0.05 using Student’s t test. B, Wild-type chloroplasts had more electron-opaque (black) plastoglobules than fib4 KD chloroplasts; the numbers of electron-transparent (white) plastoglobules were not significantly different between wild-type and fib4 KD chloroplasts. Partially and completely black plastoglobules were counted as electron opaque; plastoglobules scored as electron transparent had no electron-opaque sectors. C, Wild-type and fib4 KD chloroplasts had similar total numbers of plastoglobules (electron opaque + electron transparent). D, Wild-type and fib4 KD chloroplasts had similar numbers of peripheral vesicles. E, Transmission electron micrograph of peripheral vesicles (V). F, Electron-opaque plastoglobule in a wild-type chloroplast with less than 50% of its area appearing black. G, Electron-opaque plastoglobule in a wild-type chloroplast with more than 50% of its area appearing black. H, Adjacent electron-opaque and electron-transparent plastoglobules in a fib4 KD chloroplast.
Photosynthetic CO2 Fixation
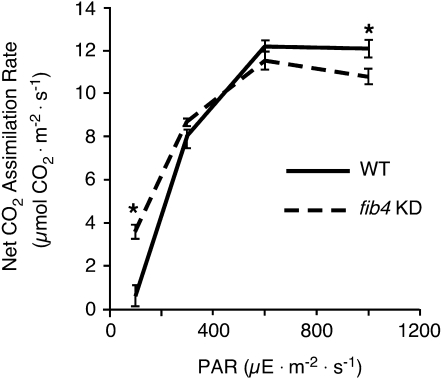
Since fib4 KD apple tree plastoglobules exhibited altered ultrastructure, we decided to determine whether knockdown of FIB4 affected photosynthesis rates. Net photosynthetic CO2 assimilation rates in wild-type and fib4 KD apple trees were compared under various levels of incident photosynthetically active radiation (PAR; Fig. 2). At midrange levels of PAR (300 and 600 μE m−2 s−1), no significant differences were detected between the net CO2 fixation rates of wild-type and fib4 KD trees. Under high levels of PAR (1,000 μE m−2 s−1), however, wild-type trees had a significantly higher net CO2 fixation rate than fib4 KD trees. Surprisingly, under low levels of PAR (100 μE m−2 s−1), the situation was reversed, with fib4 KD trees having a significantly higher net CO2 fixation rate than wild-type trees. No significant differences in stomatal conductance were observed between wild-type and fib4 KD trees; however, differences in internal [CO2] consistent with differences in net CO2 assimilation rates were observed in fib4 KD compared with wild-type trees (Supplemental Fig. S3).
Figure 2.
Effects of FIB4 knockdown on apple tree net photosynthetic CO2 assimilation rates. Net CO2 assimilation rates (μmol CO2 m−2 s−1) in wild-type (WT) and fib4 KD apple tree leaves at various levels of PAR are shown. For each genotype, measurements were taken from three leaves from each of five plants growing in a greenhouse with natural photoperiod and light intensity. Data are means ± se; * P < 0.05 using Student’s t test.
Sensitivity to Increases in Light Intensity
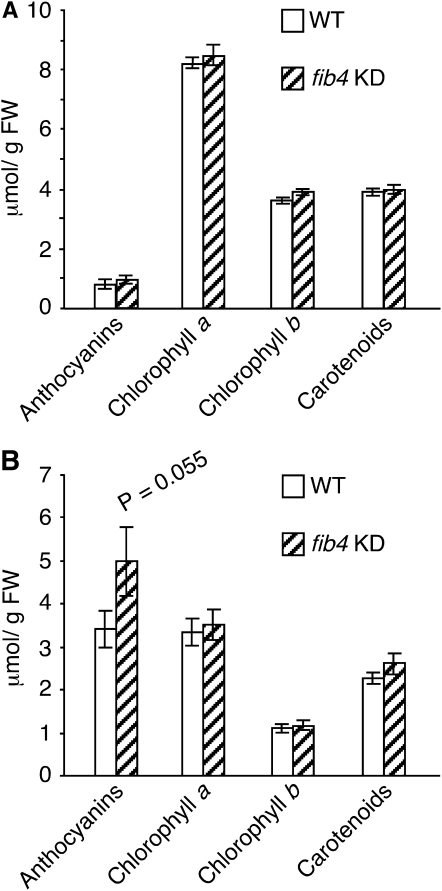
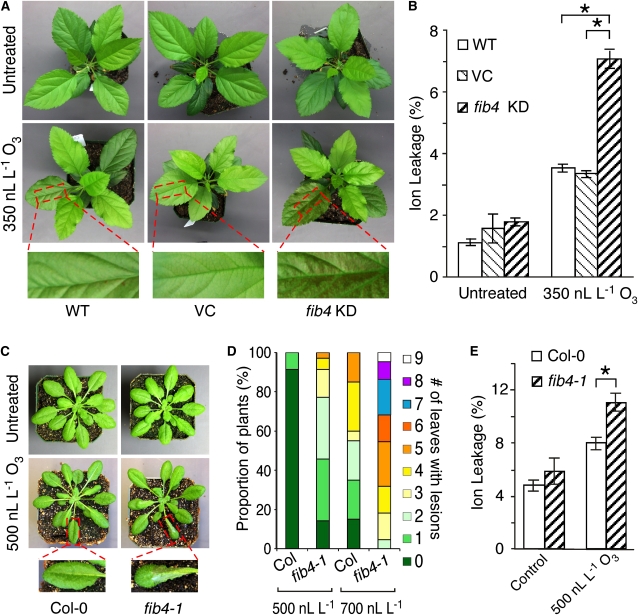
The results of the photosynthesis measurements suggested that fib4 KD apple trees were sensitive to high light. Plants typically accumulate anthocyanins in response to high light stress as photoprotectants (for review, see Steyn et al., 2002). Under a light intensity of 90 μE m−2 s−1 in plant growth chambers with a 12-h photoperiod, the leaves of fib4 KD and wild-type apple trees were green and had similar levels of anthocyanins, chlorophyll a, chlorophyll b, and carotenoids (Fig. 3A; Supplemental Fig. S4). However, following transfer to 600 μE m−2 s−1 light intensity conditions with a 12-h photoperiod for 6 d, fib4 KD tree leaves were visibly darker in color than leaves of wild-type trees (Supplemental Fig. S4) and accumulated higher amounts of anthocyanins than wild-type tree leaves, while levels of other pigments were similar (Fig. 3B).
Figure 3.
Effects of FIB4 knockdown on apple tree leaf major pigment levels. A, Major pigment levels in leaves of wild-type (WT) and fib4 KD plants grown in a growth chamber with 90 μE m−2 s−1 light intensity and a 12-h photoperiod. Data are from one representative experiment; data are means ± se; n = 6 per genotype/experiment. No statistically significant differences at P < 0.05 between wild-type and fib4 KD plants were observed for any pigment under these conditions using Student’s t test. B, Major pigment levels in leaves of wild-type and fib4 KD plants at 6 d after transfer from a growth chamber with 90 μE m−2 s−1 light intensity and a 12-h photoperiod to a growth room having 600 μE m−2 s−1 light intensity and a 12-h photoperiod. Data are means ± se; n = 6 per genotype. FW, Fresh weight.
Methyl Viologen Sensitivity
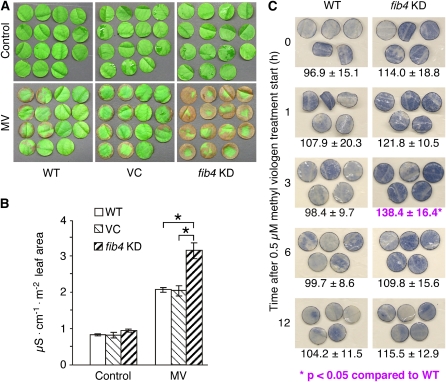
The sensitivity of fib4 KD apple trees to high light and increases in light intensity led us to suspect that they might be more sensitive to ROS generation in the chloroplast. Therefore, we tested the sensitivity of fib4 KD apple trees to methyl viologen (MV). MV generates ROS in chloroplasts by preferential electron acceptance by MV, rather than by ferredoxin, from the reaction center of PSI, producing MV radical, whose oxidation by oxygen produces the superoxide radical O2.– (Izawa, 1980; Ashton and Crafts, 1981; Taiz and Zeiger, 2006), thereby inducing oxidative damage. When floated on 0.5 μm MV solution for 24 h, leaf discs from fib4 KD apple trees developed obviously larger areas of brown pigmentation compared with leaf discs from wild-type and VC trees (Fig. 4A). Little or no brown pigmentation was observed on most of the wild-type and VC leaf discs after MV treatment; in contrast, all fib4 KD leaf discs developed extensive brown coloration. When observed, brown pigmentation in wild-type and VC leaf discs was generally restricted to limited areas near the disc margins. In addition, tissue damage as measured by electrolyte leakage was greater in fib4 KD leaf discs than wild-type or VC leaf discs at 24 h after the start of MV treatment (Fig. 4B). The greater sensitivity to MV was accompanied by higher levels of superoxide during the course of MV treatment (Fig. 4C), as detected by in situ staining of leaf discs with nitroblue tetrazolium (NBT) using the method described by Garmier et al. (2007). At 3 h after the start of MV treatment, leaf discs from fib4 KD trees exhibited measurably darker NBT staining than leaf discs from wild-type trees (Fig. 4C). This indicates that fib4 KD trees had higher levels of superoxide during MV treatment than wild-type trees. Apple showed a high, stable background signal in the superoxide assay; however, the signal increase in fib4 KD was clear. These results suggest that fib4 KD trees were more sensitive to oxidative stress.
Figure 4.
Knockdown of FIB4 increases apple sensitivity to MV. Leaf discs for each experiment were taken from plants growing in a growth chamber with 90 μE m−2 s−1 light intensity and a 12-h photoperiod. A, Wild-type (WT), VC, and fib4 KD apple tree leaf discs after exposure to 0.5 μm MV under continuous 50 μE m−2 s−1 light for 24 h. Photographs are from one representative experiment; the experiment was repeated three times with similar results; n = 15 discs per genotype, two source plants per genotype. B, Electrolyte leakage from wild-type and fib4 KD leaf discs after exposure to 0.5 μm MV for 24 h under continuous 50 μE m−2 s−1 light. Electrolyte leakage was measured at 24 h after the beginning of treatment. Data are means ± se of six replicates; * P < 0.05 using Student’s t test. C, Superoxide radical detection in wild-type and fib4 KD leaf discs floated on 0.5 μm MV under continuous 50 μE m−2 s−1 light. Leaf discs were floated on 0.5 μm MV for the indicated times, stained with NBT, and then cleared with ethanol (Garmier et al., 2007). NBT staining intensity was measured using ImageJ software (Abramoff et al., 2004). The experiment was performed using leaf discs from five trees per genotype. Statistical significance was determined using Student’s t test.
Susceptibility to Bacterial Pathogens
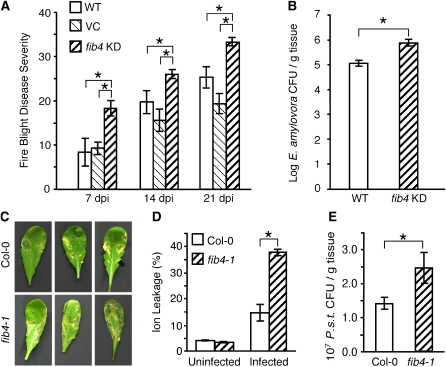
Oxidative damage also plays a role in disease processes, such as fire blight (Venisse et al., 2001). Therefore, the susceptibility of fib4 KD apple trees to E. amylovora was tested. One of the major symptoms of fire blight is a zone of darkened, shriveled, necrotic tissue that develops and spreads systemically from a point of initial infection. Necrotic region length as a percentage of total shoot length is a standard, quantitative measure of fire blight disease severity (Lee et al., 2010). Wild-type, VC, and fib4 KD apple trees growing in soil in a growth chamber were inoculated with 108 colony-forming units (cfu) mL−1 E. amylovora strain Ea581a at wounded shoot tips. Fire blight disease severity was measured once each week for 3 weeks following inoculation. At each time point, fire blight disease severity was significantly higher in fib4 KD apple trees than in either wild-type or VC trees (Fig. 5A). In addition, fib4 KD apple tree shoots supported significantly larger E. amylovora population sizes than wild-type shoots (Fig. 5B). These results indicated that fib4 KD apple trees were more susceptible to E. amylovora than control trees.
Figure 5.
Knockdown or mutation of FIB4 causes increased susceptibility to bacterial pathogens in apple and Arabidopsis. A and B represent experiments done in apple; C, D, and E represent experiments done in Arabidopsis. All plants were maintained under 90 μE m−2 s−1 light intensity, with a 12- and 10-h photoperiod for apple and Arabidopsis, respectively, for the duration of the experiments. A, Fire blight disease severity (necrotic zone size) in wild-type (WT), VC, and fib4 KD trees at 7, 14, and 21 dpi with 108 cfu mL−1 E. amylovora. Data are from one representative experiment; the experiment was repeated four times with similar results. Data are means ± se; n = 8 per genotype/experiment; * P < 0.05 using Student’s t test. B, E. amylovora population levels in wild-type and fib4 KD tree stems at 7 dpi. Data are means ± se; n = 4 or more per genotype; * P < 0.05 using Student’s t test. C, Symptom development on Col-0 and fib4-1 plant leaves at 6 dpi with 104 cfu mL−1 P. s. t. D, Ion leakage from Col-0 and fib4-1 leaves at 6 dpi with 104 cfu mL−1 P. s. t. Data are from one representative experiment; the experiment was repeated three times with similar results. Data are means ± se; n = 5 per genotype/experiment; * P < 0.05 using Student’s t test. E, P. s. t. population levels in Col-0 and fib4-1 mutant plant leaves at 4 dpi with 104 cfu mL−1 bacterium. Data are from one representative experiment; the experiment was repeated three times with similar results. Data are means ± se; n = 5 per genotype/experiment; * P < 0.05 using Student’s t test.
To determine whether plant immune responses were altered in fib4 KD apple trees during fire blight infection, we analyzed the expression of Pathogenesis-Related (PR) genes in E. amylovora-infected apple trees. PR gene expression is induced in apple trees during E. amylovora infection (Venisse et al., 2002). Our results indicated that expression of PR2 and PR8 was induced in control and fib4 KD plants; however, induction was significantly less in fib4 KD apple trees compared with wild-type trees (Supplemental Fig. S5).
To further study the role of FIB4 in disease resistance, effects of mutations in the orthologous Arabidopsis FIB4 gene (At3g23400) on bacterial disease resistance were determined. For these experiments, two transgenic Arabidopsis lines homozygous for T-DNA mutations in FIB4 (fib4-1 and fib4-2) were obtained and characterized (Supplemental Fig. S6). Pseudomonas syringae pv tomato (P. s. t.) causes bacterial speck disease in Arabidopsis (Whalen et al., 1991). Symptoms observed on the leaves of susceptible Arabidopsis plants include tissue necrosis, water-soaked lesion development, and chlorosis. Wild-type ecotype Columbia-0 (Col-0) and fib4-1 mutant plants were challenged with the virulent P. s. t. strain DC3000 by leaf infiltration, and symptom development and bacterial growth were monitored. Bacterial speck disease symptoms were visibly more severe on fib4-1 than Col-0 at 6 d post inoculation (dpi) with 104 cfu mL−1 P. s. t. (Fig. 5C). Significantly greater electrolyte leakage was recorded for fib4-1 than Col-0 at 6 dpi with 104 cfu mL−1 P. s. t. (Fig. 5D), indicating more extensive tissue damage in the mutant. In addition, the P. s. t. population size in fib4-1 plant leaves was double that in Col-0 leaves at 4 dpi with 104 cfu mL−1 bacterium (Fig. 5E). These results show that the Arabidopsis fib4-1 mutant was more susceptible to bacterial speck disease than wild-type control plants, although the change in bacterial growth was relatively modest compared with some other Arabidopsis mutants that support increased P. s. t. growth, such as sid2 (Block et al., 2005), eds1, and pad4 (Xing and Chen, 2006).
Ozone Sensitivity
The sensitivity of fib4 KD trees to MV, high light intensity, and bacterial pathogens prompted us to test whether fib4 KD trees were sensitive to ozone (O3), another oxidative stress. Exposure to O3 triggers tissue necrosis and cell death in sensitive plants (Runeckles and Chevonne, 1992). Wild-type and fib4 KD apple trees were exposed to 350 nL L−1 O3 for 6 h. At 6 h after the start of the O3 treatment, none of the apple trees had necrotic lesions. However, at 24 h after the start of O3 treatment, fib4 KD apple trees developed extensive necrosis, especially in the older leaves, while wild-type and VC trees displayed little or no necrosis (Fig. 6A). Tissue damage as measured by electrolyte leakage was significantly greater in fib4 KD apple trees than in wild-type and VC apple trees at 24 h after the start of the O3 treatment (Fig. 6B). These results indicate that fib4 KD apple trees were much more sensitive to O3 than control wild-type and VC trees.
Figure 6.
Knockdown and mutation of FIB4 increases sensitivity to O3 in apple and Arabidopsis. A and B represent experiments done in apple; C, D, and E represent experiments done in Arabidopsis. A, Effects of exposure to 350 ± 50 nL L−1 O3 for 6 h under 90 μE m−2 s−1 light intensity on wild-type (WT), VC, and fib4 KD plants. Plants were returned to their normal light conditions (90 μE m−2 s−1, 10-h photoperiod) after O3 treatment; photographs were taken 24 h after the start of O3 treatment. B, Ion leakage from wild-type, VC, and fib4 KD leaves caused by exposure to 350 ± 50 nL L−1 O3 for 6 h under 90 μE m−2 s−1 light intensity. Plants were returned to their normal light conditions (90 μE m−2 s−1, 10-h photoperiod) after O3 treatment; measurements were taken at 24 h after the start of O3 treatment. Data are from one representative experiment; the experiment was repeated three times with similar results. Data are means ± se; n = 5 per genotype/experiment; * P < 0.05 using Student’s t test. C, Effects of exposure to 500 ± 50 nL L−1 O3 for 6 h under 90 μE m−2 s−1 light intensity on Col-0 and fib4-1 plants. Plants were returned to their normal light conditions (90 μE m−2 s−1, 10-h photoperiod) after O3 treatment; photographs were taken 24 h after the start of O3 treatment. D, Amount of lesion formation in Col-0 and fib4-1 plants following exposure to 500 ± 50 nL L−1 O3 for 6 h or 700 ± 50 nL L−1 O3 for 3 h under 90 μE m−2 s−1 light intensity. Plants were returned to their normal light conditions (90 μE m−2 s−1, 10-h photoperiod) after O3 treatment; measurements were taken at 24 h after the start of O3 treatment. The percentage of plants with the indicated numbers of lesion-bearing leaves is shown. The number of lesion-bearing leaves per plant ranged from zero to nine. n = 35 plants per genotype for plants exposed to 500 ± 50 nL L−1 O3; n > 20 plants per genotype for plants exposed to 700 ± 50 nL L−1 O3. E, Ion leakage from Col-0 and fib4-1 plant leaves after exposure to 500 ± 50 nL L−1 O3 for 3 h under 90 μE m−2 s−1 light intensity. Plants were returned to their normal light conditions (90 μE m−2 s−1, 10-h photoperiod) after O3 treatment; measurements were taken at 6 h after the start of O3 treatment. Data are means ± se; n = 5 per genotype; * P < 0.05 using Student’s t test.
Arabidopsis fib4 mutants were also more sensitive to O3. Exposure of homozygous Arabidopsis fib4-1 mutants and Col-0 control plants to 500 nL L−1 O3 for 6 h or 700 nL L−1 O3 for 3 h resulted in higher numbers of leaves with visible lesions per plant in fib4-1 than in Col-0 at 24 h after the start of O3 exposure (Fig. 6, C and D). Tissue damage as measured by electrolyte leakage was greater in fib4-1 and fib4-2 mutant plants than in Col-0 plants at 6 h after the start of a 3-h, 500 nL L−1 O3 treatment (Fig. 6E; Supplemental Fig. S7). These results indicate that Arabidopsis fib4-1 and fib4-2 mutant plants were more sensitive to O3 than Col-0, which was consistent with the increased O3 sensitivity of fib4 KD apple trees.
Plastoglobule Ultrastructure during O3 Stress
In order to determine whether O3 stress caused any changes in plastoglobule ultrastructure, wild-type and fib4 KD apple trees were exposed to 350 nL L−1 O3 for 6 h, leaf tissue was collected and fixed at 2 d after the beginning of O3 exposure, and plastoglobules in palisade mesophyll cells were examined by TEM. In wild-type plants, the proportion of electron-transparent plastoglobules increased from 4% of total plastoglobules before O3 treatment to 43% of total plastoglobules after O3 treatment (Fig. 7, A and B). There were fewer electron-opaque plastoglobules and more total plastoglobules in wild-type plants after O3 treatment, although the differences were not statistically significant (Fig. 7B). In fib4 KD plants, electron-opaque plastoglobules were virtually absent both before and after O3 treatment, as expected; however, the total number of plastoglobules in fib4 KD increased by 56% after O3 treatment due to an increase in the number of electron-transparent plastoglobules (Fig. 7, A and C).
Figure 7.
Plastoglobule ultrastructure changes in wild-type and fib4 KD following O3 exposure. Wild-type (WT) and fib4 KD apple trees were exposed to 350 nL L−1 O3 for 6 h under 90 μE m−2 s−1 light intensity and then returned to their normal growth conditions (90 μE m−2 s−1, 10-h photoperiod). Leaf tissues were collected at 2 d after the beginning of the O3 exposure and examined by TEM. Data shown are from one experiment; the experiment was performed twice with similar results each time. A, Transmission electron micrographs of representative plastoglobules (arrows) in control and O3-treated wild-type and fib4 KD leaf palisade mesophyll cell chloroplasts. The scale is the same in all four micrographs. B, Numbers of electron-transparent plastoglobules (white), electron-opaque plastoglobules (black), and total plastoglobules (black + white) per μm2 in wild-type chloroplasts before and after O3 exposure. Plastoglobules were counted in 14 chloroplasts from a control plant and 11 chloroplasts from an O3-exposed plant. Data are means ± se; * P < 0.05 using Student’s t test. C, Numbers of black and white plastoglobules and total plastoglobules per μm2 in fib4 KD chloroplasts before and after O3 exposure. Plastoglobules were counted in 15 chloroplasts from a control plant and 15 chloroplasts from an O3-exposed plant. Data are means ± se; * P < 0.05 using Student’s t test.
DISCUSSION
This study indicates that FIB4 is required for plant resistance to several abiotic stresses and for resistance to bacterial pathogens. This finding is consistent with the growing body of evidence linking fibrillins with stress tolerance and disease resistance (Pruvot et al., 1996; Gillet et al., 1998; Rey et al., 2000; Rorat et al., 2001; Cooper et al., 2003; Leitner-Dagan et al., 2006; Yang et al., 2006; Youssef et al., 2010). It is interesting that FIB4 was found to be required for resistance to P. s. t., as FIB4 is one of a handful of proteins that is phosphorylated early on during defense responses against this bacterium, suggesting a role for FIB4 in basal defenses (Jones et al., 2006). The phenotypic similarities between Arabidopsis fib4 mutants and fib4 KD apple trees indicate that the function of FIB4 is conserved between these two very different species. This implies that FIB4 may play a basic, conserved role in plant physiology, perhaps as a contributor to plant stress tolerance and/or photosynthetic acclimatization.
This study also indicates that FIB4 is involved in plastoglobule development. While FIB4 does not appear to be required for the production of plastoglobules, the dramatic decrease in plastoglobule osmiophilicity in fib4 KD apple tree leaf chloroplasts compared with the wild type under benign conditions suggests that FIB4 is required for the development of plastoglobule osmiophilic content. The reactivity of osmium oxides with certain biological materials was noted upon the discovery of osmium (Tennant, 1804). Some major plastoglobule constituents such as plastoquinone, carotenoids (Bahr, 1954), and triacylglycerides are expected to contribute to plastoglobule osmiophilicity because their ethylenic double bonds reduce OsO4, probably producing stable diester adducts (Wigglesworth, 1957) that scatter electrons due to the high mass density and atomic number of osmium (Valentine, 1958). In addition, the reducing power of tocopherols and plastoquinone may contribute to plastoglobule osmiophilicity (Dahlin and Ryberg, 1986). Under mild conditions, FIB4 appears to play a role in the accumulation of osmiophilic material within plastoglobules. However, FIB4 cannot be the sole factor required for plastoglobule osmiophilicity, since osmiophilic plastoglobules were observed in fib4 KD trees, albeit much fewer than in wild-type apples.
Our finding that plastoglobule osmiophilicity decreased in wild-type apple chloroplasts following acute O3 stress was consistent with the reduced plastoglobule osmiophilicity observed in earlier studies of chronic O3 exposure in radish (Raphanus sativus; Miyake et al., 1989) and Norway spruce (Picea abies; Kivimäenpää et al., 2003). Decreased plastoglobule osmiophilicity could reflect oxidation (Miyake et al., 1989) or egress of plastoglobule osmiophilic content during O3 stress. This suggests that plastoglobules may be dynamic reservoirs of antioxidants that dispense their contents as needed during O3 stress; fib4 KD plastoglobules, which contain very little osmiophilic material to begin with, may be unable to perform this function. This could account for the increased O3 sensitivity and oxidative stress sensitivity of fib4 KD trees compared with wild-type trees. In addition, the increase in total number of plastoglobules in fib4 KD during O3 stress may represent a physiological compensation for a dearth of osmiophilic plastoglobules.
The increased sensitivity of fib4 KD plants to MV implies that they are more sensitive to oxidative stress, particularly in the chloroplast. The higher photosynthesis rates of fib4 KD trees compared with wild-type trees under low-light conditions, which were used for the MV treatments, would likely have exacerbated oxidative stress due to MV treatment in fib4 KD trees. The higher level of superoxide in MV-treated fib4 KD apple leaves than in wild-type apple leaves is consistent with this scenario. Increases or decreases in chloroplast ROS sensitivity are known to affect MV sensitivity. For example, when chloroplastic ROS-scavenging capacity is inhibited by decreasing the expression of thylakoid ascorbate peroxidase (APX), MV sensitivity increases (Tarantino et al., 2005). Furthermore, increasing chloroplastic ROS-scavenging capacity by overexpressing chloroplastic APX (Murgia et al., 2004) or overexpressing cytosolic APX in the chloroplast (Badawi et al., 2004) reduces sensitivity to MV.
Higher anthocyanin accumulation in the leaves of fib4 KD apple trees than in wild-type apple trees upon shifting to higher light intensity further supports the notion that fib4 KD plants are more sensitive to oxidative stress in the chloroplast. Anthocyanins protect photosynthetic systems from high light by sunscreen and antioxidant activities (Smillie and Hetherington, 1999; Hughes et al., 2005; Kytridis and Manetas, 2006). Higher anthocyanin accumulation in the leaves of fib4 KD apple trees upon shifting to higher light intensity might partially compensate for a lack of FIB4-mediated control of oxidative stress in the chloroplast. In addition, increased photoinhibition in fib4 KD apple leaves compared with the wild type at high light intensity suggests a reduced ability to control photooxidative damage of the photosynthetic system in fib4 KD trees. The increased O3 sensitivity of fib4 KD apple and fib4 Arabidopsis plants is also consistent with FIB4 contributing to chloroplast oxidative damage control, since chloroplastic ROS production is an early response of plants to O3 exposure (Joo et al., 2005).
Similarly, the increased susceptibility of Arabidopsis fib4-1 mutant plants to bacterial speck disease is consistent with increased sensitivity to oxidative damage. P. s. t. induces light-dependent ROS accumulation in susceptible host tissues via the phytotoxic virulence factor coronatine (Ishiga et al., 2009). Furthermore, the chloroplast electron transport inhibitor 3-(3,4-dichlorophenyl)-1,1-dimethylurea, which suppresses chloroplast ROS production (Joo et al., 2005), blocks bacterial speck disease development in tomato cotyledons (Ishiga et al., 2009). This suggests that ROS production in chloroplasts is required for bacterial speck disease development (Ishiga et al., 2009). Thus, FIB4 could contribute to bacterial speck disease resistance by mediating oxidative damage control in the chloroplast.
The increased susceptibility of fib4 KD apple trees to E. amylovora is consistent with an increased sensitivity to oxidative damage, although there is currently no clear link between the chloroplast and fire blight. E. amylovora induces superoxide production in host plants during infection, resulting in membrane damage and necrosis (Venisse et al., 2001). The production of superoxide appears to play a role in fire blight disease development, possibly by inducing cell death processes in the host. In addition, PR gene expression was partially suppressed in fib4 KD apple trees during E. amylovora infection. PR gene expression is normally induced in apple trees during E. amylovora infection (Venisse et al., 2002), and some of these genes could play a role in reducing apple susceptibility to E. amylovora (Maxson-Stein et al., 2002; Malnoy et al., 2007). These results indicate that fib4 KD apple trees had weakened resistance to E. amylovora. In light of these findings, it is interesting that the E. amylovora-secreted virulence protein HrpN (Wei et al., 1992) can physically associate with FIB4 (Song et al., 2002). Although this interaction has not been confirmed in vivo, it might make sense if FIB4 were a target of bacterial effector proteins, since reduction of FIB4 expression triggered disease susceptibility in this study.
The presence of FIB4 in the plastoglobule, the PSII light-harvesting complex, and the thylakoid membrane system, coupled with the presence of a conserved lipocalin signature in FIB4 (Jones et al., 2006), presents a possible mechanism for FIB4 function in plastoglobule development and oxidative stress. Based on its structural similarity to lipocalins (for review, see Flower et al., 2000), FIB4 may be hypothesized to interact with one or more plastoglobule lipid components, helping to transport lipophilic antioxidants in and out of the plastoglobule. In addition, FIB4 could be involved in the delivery of lipophilic antioxidants to particular locations within the photosynthetic membrane system, such as the PSII light-harvesting complex. When the amount of FIB4 is greatly reduced, such as in the fib4 KD apple trees, then lipophilic antioxidants may not be properly loaded into plastoglobules under benign conditions and may not be dispensed from plastoglobules to locations where they are needed during stress. This hypothesis would explain the increased sensitivity to oxidative stress and alterations in plastoglobule osmiophilicity observed in the fib4 KD apple trees.
MATERIALS AND METHODS
Plant Material
Arabidopsis (Arabidopsis thaliana) plants were grown as described previously (Jambunathan et al., 2001). Arabidopsis T-DNA insertion mutants were obtained from the Arabidopsis Biological Resource Center, a publicly accessible collection of T-DNA insertion lines (http://www.biosci.ohio-state.edu/pcmb/Facilities/abrc/abrchome.htm; Alonso et al., 2003). Transgenic RNAi and control pGH00.0131-EGFP expressing apple (Malus 3 domestica) plants were generated as described previously (Maximova et al., 1998). For details of vector construction and transformation to generate fib4 KD apple plants, see Supplemental Figure S1 and its legend. In vitro rooted apple plants were transferred to potting mix (Redi-Earth) and acclimated in a growth chamber under the same environmental conditions as described for Arabidopsis (Jambunathan et al., 2001).
RNA Extraction, DNA Extraction, and PCR
Total RNA from apple leaves was isolated using an established protocol (Jensen et al., 2010). Total RNA from Arabidopsis leaves was isolated as described previously (Jambunathan et al., 2001). DNA was eliminated from RNA samples using the DNA-Free kit (Ambion). Conventional reverse transcription-PCR was performed as described previously (Liu et al., 2005; for primers, see Supplemental Table S1). Quantitative real-time PCR analyses were performed at the Pennsylvania State University Genomics Core Facility as described previously (Jensen et al., 2010; for primers and probes, see Supplemental Table S2). For PR gene expression analyses, apple inoculations with Erwinia amylovora and tissue collection were done as described by Norelli et al. (2009) using gene-specific primers for each PR gene (for primers, see Supplemental Table S2). Genomic DNA was isolated from young leaves using standard methods (Kasajima et al., 2004). PCR was performed using the Advantage 2 PCR enzyme system kit according to the manufacturer’s instructions (Clontech). The LBb1.3 primer sequence was obtained from http://signal.salk.edu/tdnaprimers.html.
TEM
For the TEM experiments presented in Figure 1, leaf discs were collected from fully expanded leaves of 4- to 5-month-old apple plants acclimated in the growth chamber with 90 μE m−2 s−1 light intensity and a 12-h photoperiod. Tissues were fixed with glutaraldehyde in HEPES buffer, postfixed in OsO4 in the same buffer, and embedded in Spurr’s resin, according to the method of Owen and Makaroff (1995), essentially as described by Li et al. (2004). Ultrathin sections (80 nm thick) were cut using a Reichart-Jung Ultracut E Microtome and stained with uranyl acetate and lead citrate. Observations of chloroplasts were made with a JEOL 1200 EX II transmission electron microscope fitted with a high-resolution Tietz F224 digital camera. Chloroplast areas were measured using ImageJ software (http://rsbweb.nih.gov/ij/). Plastoglobules were counted independently by two people, and one count was done as a blind experiment. For the O3 TEM experiments presented in Figure 7, 1- to 2-month-old apple trees growing in a growth room with 90 μE m−2 s−1 light intensity and a 10-h photoperiod were exposed to 350 nL L−1 O3 for 6 h. Leaf tissues were collected at 2 d after the beginning of the O3 exposure and examined by TEM, and plastoglobules were counted.
Photosynthesis Measurements
Photosynthesis measurements were carried out on potted fib4 KD and wild-type trees maintained at 22°C ± 3°C in a greenhouse. The second, third, and fourth fully expanded leaves on each of five replicate trees were used for measurements, and the readings were repeated three times for each of the experiments. Measurements were taken on the center of each leaf. For both light intensity and intercellular [CO2] experiments, measurements were taken on consecutive days between 10:00 am and 2:00 pm on each day. Gas-exchange measurements were taken with a CIRAS-I infrared gas analyzer equipped with a 2.5-cm2 Parkinson (PLC 6U) leaf cuvette with light-emitting diode lights (PP Systems). Light at the indicated intensity was applied to the leaves, and net CO2 assimilation, stomatal conductance, and intercellular [CO2] were measured.
Pigment Analysis
Anthocyanins, carotenoids, chlorophyll a, and chlorophyll b were extracted from leaves and measured as described by Sims and Gamon (2002).
Light, O3, and Herbicide Treatments
O3 treatments were performed in a tissue culture chamber (model CU-32L; Percival Scientific) attached to an O3 generator (model 2000; Jelight Company) and an O3 monitor (model 450; Advanced Pollution Instrumentation). High-light treatment consisted of exposure to 600 μE m−2 s−1 for 6 d at 21°C with a 12-h photoperiod and 35% to 45% relative humidity. For herbicide treatments, apple tree leaf discs were cut with a 1-cm-diameter cork borer and floated on 20 mL of water or 0.5 μm MV (Sigma) in a petri dish for 24 h. Fifteen leaf discs representing two individual plants (seven or eight discs per plant) were used per sample, and six biological replicates were done for each plant genotype for each treatment.
Disease Assays
Pseudomonas syringae infection and population analysis in Arabidopsis was done using a published protocol (Jambunathan et al., 2001). Apple infection with E. amylovora and disease severity measurement was done as described previously (Jensen et al., 2003). Bacterial populations in apple tissue were determined by tissue extraction and serial dilution plating.
Electrolyte Leakage Assays
For the MV experiments, the conductivity of the MV treatment and control solutions (in μS cm−1) was measured at 24 h after the beginning of the experiment using a Corning 316 conductivity meter and divided by the leaf area used in the sample (in m2). Relative ion leakage (Joo et al., 2005) was used for the bacterial speck disease and O3 experiments. Apple or Arabidopsis leaves were collected, rinsed with deionized water, and immersed in 25 mL of deionized water for 4 h with shaking at 100 rpm, and the conductivity (μS cm−1) of the resulting solution was measured. The samples were then autoclaved, and conductivity was measured again. Relative ion leakage was calculated by dividing the conductivity of the electrolyte leakage solution before autoclaving by the conductivity of the electrolyte solution after autoclaving and multiplying by 100.
Detection of Superoxide
In situ O2.– was detected using a NBT staining method essentially as described by Garmier et al. (2007). In brief, MV-treated leaf discs were vacuum infiltrated with a solution of 0.5 mg mL−1 NBT (Sigma) in 10 mm potassium phosphate buffer (pH 7.8) and kept in dark for 1 h, and then chlorophyll was cleared by immersion in ethanol at 70°C. Leaf discs were photographed using a FinePix S9000 digital camera (FujiFilm), and NBT staining intensity was measured using ImageJ software (Abramoff et al., 2004). Each sample consisted of five leaf discs, each disc taken from a different tree.
Supplemental Data
The following materials are available in the online version of this article.
Supplemental Figure S1. Construction of the apple FIB4 RNAi binary vector used for Agrobacterium tumefaciens-mediated genetic transformation of apple.
Supplemental Figure S2. Confirmation of reduced expression of the FIB4 gene in fib4 KD apple trees, and phenotype of wild-type and transgenic trees.
Supplemental Figure S3. Internal [CO2] and stomatal conductance of fib4 KD and wild-type apple trees.
Supplemental Figure S4. Effects of transfer from 90 μE m−2 s−1 PAR to 600 μE m−2 s−1 PAR for 6 d on wild-type and fib4 KD plants.
Supplemental Figure S5. Expression of PR genes was induced in wild-type (WT) and fib4 KD apple tree during E. amylovora infection.
Supplemental Figure S6. Confirmation of T-DNA insertion in the Arabidopsis FIB4 gene.
Supplemental Figure S7. The fib4-1 and fib4-2 mutants are more sensitive to O3 than wild-type Col-0 plants.
Supplemental Table S1. PCR primer sequences.
Supplemental Table S2. Real-time PCR primer and probe sequences.
Supplementary Material
Acknowledgments
We thank Dr. Mark Guiltinan (The Pennsylvania State University) for providing tissue culture laboratory and plant growth facilities for the generation and maintenance of the transgenic apple plants, Ann Young (The Pennsylvania State University) for help with the construction of pGY06.0228, and Sharon Pishak (The Pennsylvania State University) for her support in generating the transgenic apple plants. We thank Dr. Herb S. Aldwinckle (Cornell University) for providing the cv Royal Gala tissue culture lines used for transformation. We thank Ruth Haldeman, Missy Hazen, and Greg Ning at the Pennsylvania State University Electron Microscopy Facility at University Park for providing training and support needed to use the transmission electron microscope. We thank Sherry Coven (The Pennsylvania State University) for assistance with plastoglobule counts.
References
- Abramoff MD, Magelhaes PJ, Ram SJ. (2004) Image processing with ImageJ. Biophotonics Int 11: 36–42 [Google Scholar]
- Alonso JM, Stepanova AN, Leisse TJ, Kim CJ, Chen H, Shinn P, Stevenson DK, Zimmerman J, Barajas P, Cheuk R, et al. (2003) Genome-wide insertional mutagenesis of Arabidopsis thaliana. Science 301: 653–657 [DOI] [PubMed] [Google Scholar]
- Apel K, Hirt H. (2004) Reactive oxygen species: metabolism, oxidative stress, and signal transduction. Annu Rev Plant Biol 55: 373–399 [DOI] [PubMed] [Google Scholar]
- Asada K. (2006) Production and scavenging of reactive oxygen species in chloroplasts and their functions. Plant Physiol 141: 391–396 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ashton FM, Crafts AS. (1981) Mode of Action of Herbicides, Ed 2. Wiley, New York [Google Scholar]
- Austin JR, II, Frost E, Vidi P-A, Kessler F, Staehelin LA. (2006) Plastoglobules are lipoprotein subcompartments of the chloroplast that are permanently coupled to thylakoid membranes and contain biosynthetic enzymes. Plant Cell 18: 1693–1703 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Badawi GH, Kawano N, Yamauchi Y, Shimada E, Sasaki R, Kubo A, Tanaka K. (2004) Over-expression of ascorbate peroxidase in tobacco chloroplasts enhances the tolerance to salt stress and water deficit. Physiol Plant 121: 231–238 [DOI] [PubMed] [Google Scholar]
- Bahr GF. (1954) Osmium tetroxide and ruthenium tetroxide and their reactions with biologically important substances: electron stains III. Exp Cell Res 7: 457–479 [DOI] [PubMed] [Google Scholar]
- Block A, Schmelz E, Jones JB, Klee HJ. (2005) Coronatine and salicylic acid: the battle between Arabidopsis and Pseudomonas for phytohormone control. Mol Plant Pathol 6: 79–83 [DOI] [PubMed] [Google Scholar]
- Bréhélin C, Kessler F, van Wijk KJ. (2007) Plastoglobules: versatile lipoprotein particles in plastids. Trends Plant Sci 12: 260–266 [DOI] [PubMed] [Google Scholar]
- Cooper B, Clarke JD, Budworth P, Kreps J, Hutchison D, Park S, Guimil S, Dunn M, Luginbühl P, Ellero C, et al. (2003) A network of rice genes associated with stress response and seed development. Proc Natl Acad Sci USA 100: 4945–4950 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dahlin C, Ryberg H. (1986) Accumulation of phytoene in plastoglobuli of SAN-9789 (norflurazon)-treated dark-grown wheat. Physiol Plant 68: 39–45 [Google Scholar]
- Deruère J, Römer S, d’Harlingue A, Backhaus RA, Kuntz M, Camara B. (1994) Fibril assembly and carotenoid overaccumulation in chromoplasts: a model for supramolecular lipoprotein structures. Plant Cell 6: 119–133 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Emter O, Falk H, Sitte P. (1990) Specific carotenoids and proteins as prerequisites for chromoplast tubule formation. Protoplasma 157: 128–135 [Google Scholar]
- Flower DR, North ACT, Sansom CE. (2000) The lipocalin protein family: structural and sequence overview. Biochim Biophys Acta 1482: 9–24 [DOI] [PubMed] [Google Scholar]
- Friso G, Giacomelli L, Ytterberg AJ, Peltier J-B, Rudella A, Sun Q, van Wijk KJ. (2004) In-depth analysis of the thylakoid membrane proteome of Arabidopsis thaliana chloroplasts: new proteins, new functions, and a plastid proteome database. Plant Cell 16: 478–499 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Galetskiy D, Susnea I, Reiser V, Adamska I, Przybylski M. (2008) Structure and dynamics of photosystem II light-harvesting complex revealed by high-resolution FTICR mass spectrometric proteome analysis. J Am Soc Mass Spectrom 19: 1004–1013 [DOI] [PubMed] [Google Scholar]
- Garmier M, Priault P, Vidal G, Driscoll S, Djebbar R, Boccara M, Mathieu C, Foyer CH, De Paepe R. (2007) Light and oxygen are not required for harpin-induced cell death. J Biol Chem 282: 37556–37566 [DOI] [PubMed] [Google Scholar]
- Gillet B, Beyly A, Peltier G, Rey P. (1998) Molecular characterization of CDSP 34, a chloroplastic protein induced by water deficit in Solanum tuberosum L. plants, and regulation of CDSP 34 expression by ABA and high illumination. Plant J 16: 257–262 [DOI] [PubMed] [Google Scholar]
- Hughes NM, Neufeld HS, Burkey KO. (2005) Functional role of anthocyanins in high-light winter leaves of the evergreen herb Galax urceolata. New Phytol 168: 575–587 [DOI] [PubMed] [Google Scholar]
- Ishiga Y, Uppalapati SR, Ishiga T, Elavarthi S, Martin B, Bender CL. (2009) The phytotoxin coronatine induces light-dependent reactive oxygen species in tomato seedlings. New Phytol 181: 147–160 [DOI] [PubMed] [Google Scholar]
- Izawa S. (1980) Acceptors and donors for chloroplast electron transport. Methods Enzymol 69: 413–434 [Google Scholar]
- Jambunathan N, Siani JM, McNellis TW. (2001) A humidity-sensitive Arabidopsis copine mutant exhibits precocious cell death and increased disease resistance. Plant Cell 13: 2225–2240 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jensen PJ, Makalowska I, Altman N, Fazio G, Praul C, Maximova SN, Crassweller RM, Travis JW, McNellis TW. (2010) Rootstock-regulated gene expression patterns in apple tree scions. Tree Genet Genomes 6: 57–72 [Google Scholar]
- Jensen PJ, Rytter J, Detwiler EA, Travis JW, McNellis TW. (2003) Rootstock effects on gene expression patterns in apple tree scions. Plant Mol Biol 53: 493–511 [DOI] [PubMed] [Google Scholar]
- Jones AME, Bennett MH, Mansfield JW, Grant M. (2006) Analysis of the defence phosphoproteome of Arabidopsis thaliana using differential mass tagging. Proteomics 6: 4155–4165 [DOI] [PubMed] [Google Scholar]
- Joo JH, Wang S, Chen JG, Jones AM, Fedoroff NV. (2005) Different signaling and cell death roles of heterotrimeric G protein α and β subunits in the Arabidopsis oxidative stress response to ozone. Plant Cell 17: 957–970 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kasajima I, Ide Y, Ohkama-Ohtsu N, Hayashi H, Yoneyama T, Fujiwara T. (2004) A protocol for rapid DNA extraction from Arabidopsis thaliana for PCR analysis. Plant Mol Biol Rep 22: 49–52 [Google Scholar]
- Kivimäenpää M, Sutinen S, Karlsson PE, Selldén G. (2003) Cell structural changes in the needles of Norway spruce exposed to long-term ozone and drought. Ann Bot (Lond) 92: 779–793 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Knoth R, Hansmann P, Sitte P. (1986) Chromoplasts of Palisota barteri, and the molecular structure of chromoplast tubules. Planta 168: 167–174 [DOI] [PubMed] [Google Scholar]
- Kytridis VP, Manetas Y. (2006) Mesophyll versus epidermal anthocyanins as potential in vivo antioxidants: evidence linking the putative antioxidant role to the proximity of oxy-radical source. J Exp Bot 57: 2203–2210 [DOI] [PubMed] [Google Scholar]
- Laizet Y, Pontier D, Mache R, Kuntz M. (2004) Subfamily organization and phylogenetic origin of genes encoding plastid lipid-associated proteins of the fibrillin type. J Genome Sci Technol 3: 19–28 [Google Scholar]
- Lamb C, Dixon RA. (1997) The oxidative burst in plant disease resistance. Annu Rev Plant Physiol Plant Mol Biol 48: 251–275 [DOI] [PubMed] [Google Scholar]
- Langenkämper G, Manac’h N, Broin M, Cuiné S, Becuwe N, Kuntz M, Rey P. (2001) Accumulation of plastid lipid-associated proteins (fibrillin/CDSP34) upon oxidative stress, ageing and biotic stress in Solanaceae and in response to drought in other species. J Exp Bot 52: 1545–1554 [DOI] [PubMed] [Google Scholar]
- Lee SA, Ngugi HK, Halbrendt NO, O’Keefe G, Lehman B, Travis JW, Sinn JP, McNellis TW. (2010) Virulence characteristics accounting for fire blight disease severity in apple trees and seedlings. Phytopathology 100: 539–550 [DOI] [PubMed] [Google Scholar]
- Leitner-Dagan Y, Ovadis M, Shklarman E, Elad Y, Rav David D, Vainstein A. (2006) Expression and functional analyses of the plastid lipid-associated protein CHRC suggest its role in chromoplastogenesis and stress. Plant Physiol 142: 233–244 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li W, Chen C, Markmann-Mulisch U, Timofejeva L, Schmelzer E, Ma H, Reiss B. (2004) The Arabidopsis AtRAD51 gene is dispensable for vegetative development but required for meiosis. Proc Natl Acad Sci USA 101: 10596–10601 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Liu J, Jambunathan N, McNellis TW. (2005) Transgenic expression of the von Willebrand A domain of the BONZAI 1/COPINE 1 protein triggers a lesion-mimic phenotype in Arabidopsis. Planta 221: 85–94 [DOI] [PubMed] [Google Scholar]
- Liu Y, Ren D, Pike S, Pallardy S, Gassmann W, Zhang S. (2007) Chloroplast-generated reactive oxygen species are involved in hypersensitive response-like cell death mediated by a mitogen-activated protein kinase cascade. Plant J 51: 941–954 [DOI] [PubMed] [Google Scholar]
- Malnoy M, Jin Q, Borejsza-Wysocka EE, He SY, Aldwinckle HS. (2007) Overexpression of the apple MpNPR1 gene confers increased disease resistance in Malus × domestica. Mol Plant Microbe Interact 20: 1568–1580 [DOI] [PubMed] [Google Scholar]
- Maximova SN, Dandekar AM, Guiltinan MJ. (1998) Investigation of Agrobacterium-mediated transformation of apple using green fluorescent protein: high transient expression and low stable transformation suggest that factors other than T-DNA transfer are rate-limiting. Plant Mol Biol 37: 549–559 [DOI] [PubMed] [Google Scholar]
- Maxson-Stein K, He SY, Hammerschmidt R, Jones AL. (2002) Effect of treating apple trees with acibenzolar-S-methyl on fire blight and expression of pathogenesis-related protein genes. Plant Dis 86: 785–790 [DOI] [PubMed] [Google Scholar]
- Mehdy MC. (1994) Active oxygen species in plant defense against pathogens. Plant Physiol 105: 467–472 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Miyake H, Matsumura H, Fujinuma Y, Totsuka T. (1989) Effects of low concentrations of ozone on the fine structure of radish leaves. New Phytol 111: 187–195 [DOI] [PubMed] [Google Scholar]
- Murgia I, Tarantino D, Vannini C, Bracale M, Carravieri S, Soave C. (2004) Arabidopsis thaliana plants overexpressing thylakoidal ascorbate peroxidase show increased resistance to paraquat-induced photooxidative stress and to nitric oxide-induced cell death. Plant J 38: 940–953 [DOI] [PubMed] [Google Scholar]
- Noctor G, Foyer CH. (1998) Ascorbate and glutathione: keeping active oxygen under control. Annu Rev Plant Physiol Plant Mol Biol 49: 249–279 [DOI] [PubMed] [Google Scholar]
- Norelli JL, Farrell RE, Jr, Bassett CL, Baldo AM, Lalli DA, Aldwinckle HS, Wisniewski ME. (2009) Rapid transcriptional response of apple to fire blight disease revealed by cDNA suppression subtractive hybridization analysis. Tree Genet Genomes 5: 27–40 [Google Scholar]
- Owen HA, Makaroff CA. (1995) Ultrastructure of microsporogenesis and microgametogenesis in Arabidopsis thaliana (L.) Heynh. ecotype Wassilewskija (Brassicaceae). Protoplasma 185: 7–21 [Google Scholar]
- Padham AK, Hopkins MT, Wang T-W, McNamara LM, Lo M, Richardson LGL, Smith MD, Taylor CA, Thompson JE. (2007) Characterization of a plastid triacylglycerol lipase from Arabidopsis. Plant Physiol 143: 1372–1384 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peltier J-B, Ytterberg AJ, Sun Q, van Wijk KJ. (2004) New functions of the thylakoid membrane proteome of Arabidopsis thaliana revealed by a simple, fast, and versatile fractionation strategy. J Biol Chem 279: 49367–49383 [DOI] [PubMed] [Google Scholar]
- Pozueta-Romero J, Rafia F, Houlné G, Cheniclet C, Carde JP, Schantz ML, Schantz R. (1997) A ubiquitous plant housekeeping gene, PAP, encodes a major protein component of bell pepper chromoplasts. Plant Physiol 115: 1185–1194 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pruvot G, Cuiné S, Peltier G, Rey P. (1996) Characterization of a novel drought-induced 34-kDa protein located in the thylakoids of Solanum tuberosum L. plants. Planta 198: 471–479 [DOI] [PubMed] [Google Scholar]
- Rey P, Gillet B, Römer S, Eymery F, Massimino J, Peltier G, Kuntz M. (2000) Over-expression of a pepper plastid lipid-associated protein in tobacco leads to changes in plastid ultrastructure and plant development upon stress. Plant J 21: 483–494 [DOI] [PubMed] [Google Scholar]
- Rorat T, Havaux M, Irzykowski W, Cuiné S, Becuwe N, Rey P. (2001) PSII-S gene expression, photosynthetic activity and abundance of plastid thioredoxin-related and lipid-associated proteins during chilling stress in Solanum species differing in freezing resistance. Physiol Plant 113: 72–78 [Google Scholar]
- Runeckles VC, Chevonne BI. (1992) Crop responses to ozone. Lefohn AS, , Surface Level Ozone Exposures and Their Effects on Vegetation. Lewis Publishers, Chelsea, MI, pp 189–270 [Google Scholar]
- Sanabria NM, Dubery IA. (2006) Differential display profiling of the Nicotiana response to LPS reveals elements of plant basal resistance. Biochem Biophys Res Commun 344: 1001–1007 [DOI] [PubMed] [Google Scholar]
- Shetty NP, Jørgensen HJL, Jensen JD, Collinge DB, Shetty HS. (2008) Roles of reactive oxygen species in interactions between plants and pathogens. Eur J Plant Pathol 121: 267–280 [Google Scholar]
- Sims DA, Gamon JA. (2002) Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens Environ 81: 337–354 [Google Scholar]
- Smillie RM, Hetherington SE. (1999) Photoabatement by anthocyanin shields photosynthetic systems from light stress. Photosynthetica 36: 451–463 [Google Scholar]
- Song X, Fan H, Wei Z-M, inventors. January 17, 2002 Receptors for hypersensitive response elicitors and uses thereof. U.S. Patent Application No. 20020007501 [Google Scholar]
- Steinmüller D, Tevini M. (1985) Composition and function of plastoglobuli. Planta 163: 201–207 [DOI] [PubMed] [Google Scholar]
- Steyn WJ, Wand SJE, Holcroft DM, Jacobs G. (2002) Anthocyanins in vegetative tissues: a proposed unified function in photoprotection. New Phytol 155: 349–361 [DOI] [PubMed] [Google Scholar]
- Taiz L, Zeiger E. (2006) Mode of action of some herbicides. Plant Physiology. http://4e.plantphys.net/index2.php (August 5, 2010)
- Tarantino D, Vannini C, Bracale M, Campa M, Soave C, Murgia I. (2005) Antisense reduction of thylakoidal ascorbate peroxidase in Arabidopsis enhances paraquat-induced photooxidative stress and nitric oxide-induced cell death. Planta 221: 757–765 [DOI] [PubMed] [Google Scholar]
- Tennant S. (1804) On two metals, found in the black powder remaining after the solution of platina. Philos Trans R Soc Lond 94: 411–418 [Google Scholar]
- Tevini M, Steinmüller D. (1985) Composition and function of plastoglobuli. Planta 163: 91–96 [DOI] [PubMed] [Google Scholar]
- Valentine RC. (1958) Contrast in the electron microscope image. Nature 181: 832–833 [DOI] [PubMed] [Google Scholar]
- Venisse JS, Gullner G, Brisset MN. (2001) Evidence for the involvement of an oxidative stress in the initiation of infection of pear by Erwinia amylovora. Plant Physiol 125: 2164–2172 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Venisse JS, Malnoy M, Faize M, Paulin JP, Brisset MN. (2002) Modulation of defense responses of Malus spp. during compatible and incompatible interactions with Erwinia amylovora. Mol Plant Microbe Interact 15: 1204–1212 [DOI] [PubMed] [Google Scholar]
- Vidi PA, Kanwischer M, Baginsky S, Austin JR, Csucs G, Dörmann P, Kessler F, Bréhélin C. (2006) Tocopherol cyclase (VTE1) localization and vitamin E accumulation in chloroplast plastoglobule lipoprotein particles. J Biol Chem 281: 11225–11234 [DOI] [PubMed] [Google Scholar]
- Wei ZM, Laby RJ, Zumoff CH, Bauer DW, He SY, Collmer A, Beer SV. (1992) Harpin, elicitor of the hypersensitive response produced by the plant pathogen Erwinia amylovora. Science 257: 85–88 [DOI] [PubMed] [Google Scholar]
- Whalen MC, Innes RW, Bent AF, Staskawicz BJ. (1991) Identification of Pseudomonas syringae pathogens of Arabidopsis and a bacterial locus determining avirulence on both Arabidopsis and soybean. Plant Cell 3: 49–59 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wigglesworth VB. (1957) The use of osmium in the fixation and staining of tissues. Proc R Soc Lond B Biol Sci 147: 185–199 [DOI] [PubMed] [Google Scholar]
- Winkenbach F, Falk H, Liedvogel B, Sitte P. (1976) Chromoplasts of Tropaeolum majus L.: isolation and characterization of lipoprotein elements. Planta 128: 23–28 [DOI] [PubMed] [Google Scholar]
- Xing D, Chen Z. (2006) Effects of mutations and constitutive overexpression of EDS1 and PAD4 on plant resistance to different types of microbial pathogens. Plant Sci 171: 251–262 [Google Scholar]
- Yang Y, Sulpice R, Himmelbach A, Meinhard M, Christmann A, Grill E. (2006) Fibrillin expression is regulated by abscisic acid response regulators and is involved in abscisic acid-mediated photoprotection. Proc Natl Acad Sci USA 103: 6061–6066 [DOI] [PMC free article] [PubMed] [Google Scholar]
- Youssef A, Laizet Y, Block MA, Maréchal E, Alcaraz JP, Larson TR, Pontier D, Gaffé J, Kuntz M. (2010) Plant lipid-associated fibrillin proteins condition jasmonate production under photosynthetic stress. Plant J 61: 436–445 [DOI] [PubMed] [Google Scholar]
- Ytterberg AJ, Peltier JB, van Wijk KJ. (2006) Protein profiling of plastoglobules in chloroplasts and chromoplasts: a surprising site for differential accumulation of metabolic enzymes. Plant Physiol 140: 984–997 [DOI] [PMC free article] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.