Abstract
In the title compound, C19H16O2S, the sulfur-bound phenyl group is approximately parallel to one of the two phenyl rings of the benzhydryl group, making a dihedral angle of 12.53 (10)°, and forms a dihedral angle of 41.25 (9)° with the other phenyl ring. In the crystal, weak C—H⋯O interactions form a two-dimensional network propagating along the bc plane.
Related literature
For background to the sulfone anion, see: da Silva Corrêa et al. (1968 ▶); Mayr et al. (2001 ▶, 2008 ▶). For a related structure, see: Li et al. (2005 ▶). For graph-set analysis of hydrogen-bond networks, see: Bernstein et al. (1995 ▶); Etter et al. (1990 ▶).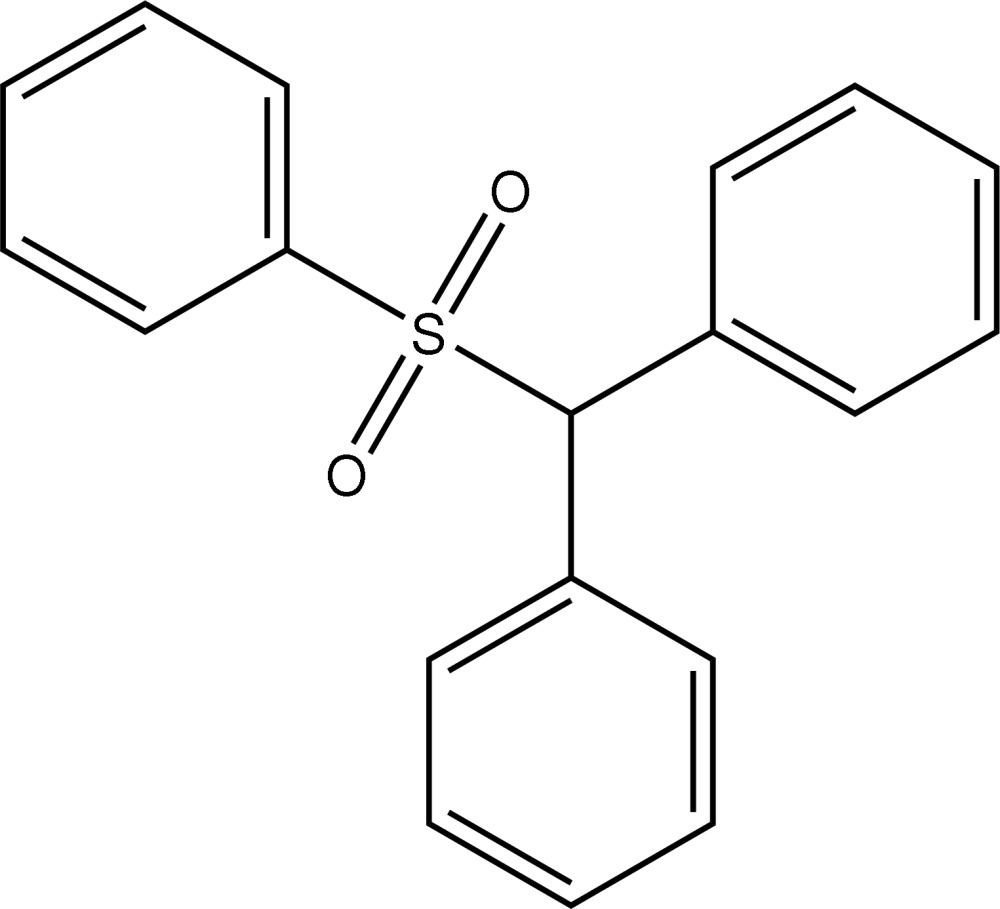
Experimental
Crystal data
C19H16O2S
M r = 308.40
Orthorhombic,

a = 16.3250 (4) Å
b = 5.7979 (1) Å
c = 16.4983 (4) Å
V = 1561.58 (6) Å3
Z = 4
Mo Kα radiation
μ = 0.21 mm−1
T = 200 K
0.20 × 0.10 × 0.09 mm
Data collection
Nonius KappaCCD diffractometer
Absorption correction: none
11675 measured reflections
3499 independent reflections
3136 reflections with I > 2σ(I)
R int = 0.027
Refinement
R[F 2 > 2σ(F 2)] = 0.031
wR(F 2) = 0.075
S = 1.04
3499 reflections
199 parameters
1 restraint
H-atom parameters constrained
Δρmax = 0.14 e Å−3
Δρmin = −0.28 e Å−3
Absolute structure: Flack (1983 ▶), 1646 Friedel pairs
Flack parameter: −0.03 (6)
Data collection: COLLECT (Hooft, 2004 ▶); cell refinement: SCALEPACK (Otwinowski & Minor, 1997 ▶); data reduction: DENZO (Otwinowski & Minor, 1997 ▶) and SCALEPACK; program(s) used to solve structure: SIR97 (Altomare et al., 1999 ▶); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008 ▶); molecular graphics: ORTEP-3 (Farrugia, 1997 ▶) and Mercury (Macrae et al., 2006 ▶); software used to prepare material for publication: PLATON (Spek, 2009 ▶).
Supplementary Material
Crystal structure: contains datablocks I, global. DOI: 10.1107/S1600536809050363/ds2011sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536809050363/ds2011Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Table 1. Hydrogen-bond geometry (Å, °).
| D—H⋯A | D—H | H⋯A | D⋯A | D—H⋯A |
|---|---|---|---|---|
| C1—H1⋯O2i | 1.00 | 2.46 | 3.449 (2) | 171 |
| C4—H4⋯O1ii | 0.95 | 2.66 | 3.390 (2) | 134 |
| C7—H7⋯O2i | 0.95 | 2.68 | 3.543 (2) | 152 |
Symmetry codes: (i) 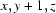 ; (ii)
; (ii)  .
.
Acknowledgments
The authors thank Professor Peter Klüfers for generous allocation of diffractometer time.
supplementary crystallographic information
Comment
During our studies on the ambident reactivity of the phenylsulfinate anion we used diarylcarbenium ions (Ar2CH+) as reference electrophiles [Mayr et al. (2001, 2008)] and, hence, obtained the title compound from a reaction of sodium benzenesulfinate with benzhydryl chloride (Ph2CHCl) in dimethyl sulfoxide.
The asymmetric unit of the title compound contains one complete molecule, which is shown in Figure 1. The sulfur-bound phenyl group is approximately parallel to one of the two phenyl rings of the benzhydryl group with an dihedral angle of 12.53 (10)°. The other one forms a dihedral angle of 41.25 (9)° with the phenyl group bound to the sulfur atom.
Three weak C–H···O interactions are found (Table 1) which lead to the formation of a two-dimensional network that propagates along the bc plane (Fig. 2). Contacts of this type have been described for a structure of a related sulfone [Li et al. (2005)]. In terms of graph-set analysis [Bernstein et al. (1995), Etter et al. (1990)], the descriptors on the unitary level are C11(4) for the H1···O2 interaction, C11(6) for the H7···O2 interaction, and C11(7) for the H4···O1 interaction.
Experimental
Benzhydryl Phenyl Sulfone was obtained by heating a mixture of sodium benzenesulfinate (0.21 g, 1.3 mmol) and benzhydryl chloride (0.26 g, 1.3 mmol) in DMSO at 70 °C. After completion of the reaction (4 h), the reaction mixture was cooled to room temperature, diluted with water, and extracted with ethyl acetate. The organic phase was washed several times with water and dried (MgSO4). A viscous oil was obtained after evaporation of the solvent under reduced pressure that solidified on standing. After column chromatography (silica gel, isohexane/EtOAc = 9/1), benzhydryl phenyl sulfone was isolated as colorless solid (0.33 g, 82%). A small amount of the title compound was dissolved in ethyl acetate. The solvent was allowed to evaporate slowly at room temperature. After 2 days crystals had formed that were suitable for X-ray analysis. mp 189 °C (186–187 °C [da Silva Corrêa et al. (1968)]).
Refinement
All H atoms were found in difference maps. C-bonded H atoms were positioned geometrically (C—H = 1.00 Å for aliphatic, 0.95 Å for aromatic H) and treated as riding on their parent atoms [Uiso(H) = 1.2Ueq(C)].
Figures
Fig. 1.
The molecular structure of the title compound, with atom labels and anisotropic displacement ellipsoids (drawn at 50% probability level) for non-H atoms.
Fig. 2.
Weak intermolecular hydrogen bonds of the type C–H···O leading to a two-dimensional network that propagates along the bc plane (viewing direction approximately along [110]). Color scheme for dashed lines: blue: H1···O2 contacts, red: H7···O2 contacts, green: H4···O1 contacts.
Crystal data
| C19H16O2S | F(000) = 648 |
| Mr = 308.40 | Dx = 1.312 (1) Mg m−3 |
| Orthorhombic, Pca21 | Mo Kα radiation, λ = 0.71073 Å |
| Hall symbol: P 2c -2ac | Cell parameters from 6504 reflections |
| a = 16.3250 (4) Å | θ = 3.1–27.5° |
| b = 5.7979 (1) Å | µ = 0.21 mm−1 |
| c = 16.4983 (4) Å | T = 200 K |
| V = 1561.58 (6) Å3 | Rod, colourless |
| Z = 4 | 0.20 × 0.10 × 0.09 mm |
Data collection
| Nonius KappaCCD diffractometer | 3136 reflections with I > 2σ(I) |
| Radiation source: rotating anode | Rint = 0.027 |
| MONTEL, graded multilayered X-ray optics | θmax = 27.5°, θmin = 3.5° |
| Detector resolution: 9 pixels mm-1 | h = −21→21 |
| CCD; rotation images; thick slices, phi/ω–scan | k = −7→6 |
| 11675 measured reflections | l = −21→20 |
| 3499 independent reflections |
Refinement
| Refinement on F2 | Secondary atom site location: difference Fourier map |
| Least-squares matrix: full | Hydrogen site location: inferred from neighbouring sites |
| R[F2 > 2σ(F2)] = 0.031 | H-atom parameters constrained |
| wR(F2) = 0.075 | w = 1/[σ2(Fo2) + (0.0396P)2 + 0.2163P] where P = (Fo2 + 2Fc2)/3 |
| S = 1.04 | (Δ/σ)max < 0.001 |
| 3499 reflections | Δρmax = 0.14 e Å−3 |
| 199 parameters | Δρmin = −0.28 e Å−3 |
| 1 restraint | Absolute structure: Flack (1983), 1646 Friedel pairs |
| Primary atom site location: structure-invariant direct methods | Flack parameter: −0.03 (6) |
Special details
| Geometry. All e.s.d.'s (except the e.s.d. in the dihedral angle between two l.s. planes) are estimated using the full covariance matrix. The cell e.s.d.'s are taken into account individually in the estimation of e.s.d.'s in distances, angles and torsion angles; correlations between e.s.d.'s in cell parameters are only used when they are defined by crystal symmetry. An approximate (isotropic) treatment of cell e.s.d.'s is used for estimating e.s.d.'s involving l.s. planes. |
| Refinement. Refinement of F2 against ALL reflections. The weighted R-factor wR and goodness of fit S are based on F2, conventional R-factors R are based on F, with F set to zero for negative F2. The threshold expression of F2 > 2σ(F2) is used only for calculating R-factors(gt) etc. and is not relevant to the choice of reflections for refinement. R-factors based on F2 are statistically about twice as large as those based on F, and R-factors based on all data will be even larger. |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| S1 | 0.06340 (2) | 0.12898 (6) | 0.26302 (3) | 0.02887 (10) | |
| O1 | 0.08669 (8) | 0.1945 (2) | 0.34378 (7) | 0.0408 (3) | |
| O2 | 0.03981 (7) | −0.10715 (19) | 0.24877 (8) | 0.0384 (3) | |
| C1 | −0.01788 (10) | 0.3229 (3) | 0.23092 (10) | 0.0276 (3) | |
| H1 | 0.0034 | 0.4820 | 0.2409 | 0.033* | |
| C2 | −0.03155 (9) | 0.3073 (3) | 0.14009 (10) | 0.0272 (3) | |
| C3 | −0.06491 (11) | 0.1160 (3) | 0.10143 (11) | 0.0347 (4) | |
| H3 | −0.0816 | −0.0139 | 0.1324 | 0.042* | |
| C4 | −0.07391 (11) | 0.1141 (3) | 0.01781 (12) | 0.0405 (4) | |
| H4 | −0.0964 | −0.0180 | −0.0080 | 0.049* | |
| C5 | −0.05082 (12) | 0.2999 (4) | −0.02807 (12) | 0.0428 (4) | |
| H5 | −0.0576 | 0.2970 | −0.0853 | 0.051* | |
| C6 | −0.01740 (12) | 0.4927 (4) | 0.00955 (13) | 0.0451 (5) | |
| H6 | −0.0008 | 0.6218 | −0.0218 | 0.054* | |
| C7 | −0.00847 (11) | 0.4955 (3) | 0.09276 (11) | 0.0363 (4) | |
| H7 | 0.0138 | 0.6283 | 0.1182 | 0.044* | |
| C8 | −0.09145 (10) | 0.2969 (3) | 0.28610 (9) | 0.0304 (4) | |
| C9 | −0.14154 (12) | 0.1021 (3) | 0.28814 (12) | 0.0431 (4) | |
| H9 | −0.1290 | −0.0274 | 0.2551 | 0.052* | |
| C10 | −0.20970 (13) | 0.0971 (4) | 0.33832 (13) | 0.0500 (5) | |
| H10 | −0.2444 | −0.0343 | 0.3383 | 0.060* | |
| C11 | −0.22748 (13) | 0.2805 (4) | 0.38811 (13) | 0.0531 (5) | |
| H11 | −0.2740 | 0.2751 | 0.4227 | 0.064* | |
| C12 | −0.17763 (14) | 0.4719 (4) | 0.38771 (14) | 0.0547 (5) | |
| H12 | −0.1895 | 0.5985 | 0.4223 | 0.066* | |
| C13 | −0.10994 (12) | 0.4802 (3) | 0.33668 (11) | 0.0406 (4) | |
| H13 | −0.0760 | 0.6133 | 0.3365 | 0.049* | |
| C14 | 0.14411 (10) | 0.2005 (3) | 0.19613 (10) | 0.0292 (3) | |
| C15 | 0.18653 (11) | 0.4044 (3) | 0.20890 (12) | 0.0407 (4) | |
| H15 | 0.1739 | 0.5002 | 0.2539 | 0.049* | |
| C16 | 0.24784 (12) | 0.4662 (3) | 0.15468 (14) | 0.0479 (5) | |
| H16 | 0.2775 | 0.6055 | 0.1623 | 0.057* | |
| C17 | 0.26549 (12) | 0.3259 (4) | 0.09010 (13) | 0.0503 (5) | |
| H17 | 0.3075 | 0.3691 | 0.0532 | 0.060* | |
| C18 | 0.22315 (13) | 0.1231 (4) | 0.07798 (14) | 0.0517 (5) | |
| H18 | 0.2364 | 0.0267 | 0.0333 | 0.062* | |
| C19 | 0.16122 (12) | 0.0598 (3) | 0.13093 (12) | 0.0403 (4) | |
| H19 | 0.1311 | −0.0782 | 0.1224 | 0.048* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| S1 | 0.03180 (18) | 0.02898 (18) | 0.02584 (18) | 0.00027 (14) | −0.00044 (18) | 0.00176 (17) |
| O1 | 0.0447 (7) | 0.0530 (8) | 0.0247 (6) | 0.0025 (6) | −0.0042 (5) | 0.0005 (5) |
| O2 | 0.0408 (6) | 0.0273 (6) | 0.0471 (9) | −0.0006 (4) | 0.0029 (6) | 0.0047 (5) |
| C1 | 0.0315 (8) | 0.0251 (8) | 0.0263 (8) | −0.0019 (6) | 0.0000 (7) | −0.0014 (6) |
| C2 | 0.0257 (7) | 0.0301 (8) | 0.0258 (8) | 0.0028 (6) | 0.0008 (6) | 0.0011 (6) |
| C3 | 0.0384 (9) | 0.0357 (9) | 0.0301 (9) | −0.0062 (7) | −0.0017 (7) | −0.0001 (7) |
| C4 | 0.0394 (9) | 0.0507 (11) | 0.0312 (10) | −0.0053 (8) | −0.0032 (8) | −0.0060 (8) |
| C5 | 0.0423 (10) | 0.0597 (12) | 0.0262 (9) | 0.0046 (9) | −0.0005 (8) | 0.0006 (8) |
| C6 | 0.0561 (13) | 0.0471 (11) | 0.0321 (9) | 0.0014 (9) | 0.0063 (9) | 0.0098 (7) |
| C7 | 0.0440 (10) | 0.0316 (9) | 0.0334 (9) | −0.0002 (7) | 0.0045 (7) | 0.0013 (7) |
| C8 | 0.0327 (8) | 0.0324 (8) | 0.0261 (9) | 0.0015 (6) | −0.0017 (6) | 0.0028 (6) |
| C9 | 0.0433 (10) | 0.0450 (11) | 0.0410 (11) | −0.0093 (8) | 0.0040 (8) | −0.0017 (8) |
| C10 | 0.0403 (10) | 0.0638 (13) | 0.0460 (12) | −0.0138 (9) | 0.0040 (9) | 0.0146 (10) |
| C11 | 0.0409 (10) | 0.0738 (14) | 0.0445 (12) | 0.0098 (10) | 0.0133 (9) | 0.0159 (11) |
| C12 | 0.0575 (13) | 0.0597 (13) | 0.0470 (12) | 0.0113 (10) | 0.0174 (10) | −0.0022 (10) |
| C13 | 0.0465 (10) | 0.0412 (10) | 0.0340 (10) | 0.0044 (8) | 0.0048 (8) | −0.0049 (8) |
| C14 | 0.0270 (8) | 0.0333 (8) | 0.0273 (8) | 0.0008 (6) | −0.0026 (7) | 0.0016 (7) |
| C15 | 0.0384 (10) | 0.0429 (10) | 0.0409 (11) | −0.0064 (8) | −0.0044 (8) | −0.0039 (8) |
| C16 | 0.0340 (9) | 0.0507 (11) | 0.0589 (13) | −0.0111 (9) | −0.0065 (9) | 0.0088 (10) |
| C17 | 0.0320 (9) | 0.0683 (13) | 0.0505 (12) | 0.0027 (9) | 0.0089 (9) | 0.0144 (10) |
| C18 | 0.0466 (11) | 0.0625 (13) | 0.0459 (12) | 0.0048 (9) | 0.0128 (10) | −0.0074 (10) |
| C19 | 0.0402 (9) | 0.0394 (9) | 0.0414 (11) | 0.0016 (8) | 0.0045 (8) | −0.0056 (8) |
Geometric parameters (Å, °)
| S1—O1 | 1.4366 (13) | C9—C10 | 1.387 (3) |
| S1—O2 | 1.4415 (12) | C9—H9 | 0.9500 |
| S1—C14 | 1.7681 (17) | C10—C11 | 1.374 (3) |
| S1—C1 | 1.8180 (16) | C10—H10 | 0.9500 |
| C1—C8 | 1.514 (2) | C11—C12 | 1.376 (3) |
| C1—C2 | 1.518 (2) | C11—H11 | 0.9500 |
| C1—H1 | 1.0000 | C12—C13 | 1.390 (3) |
| C2—C3 | 1.390 (2) | C12—H12 | 0.9500 |
| C2—C7 | 1.394 (2) | C13—H13 | 0.9500 |
| C3—C4 | 1.387 (3) | C14—C19 | 1.379 (2) |
| C3—H3 | 0.9500 | C14—C15 | 1.386 (2) |
| C4—C5 | 1.369 (3) | C15—C16 | 1.389 (3) |
| C4—H4 | 0.9500 | C15—H15 | 0.9500 |
| C5—C6 | 1.390 (3) | C16—C17 | 1.371 (3) |
| C5—H5 | 0.9500 | C16—H16 | 0.9500 |
| C6—C7 | 1.381 (3) | C17—C18 | 1.378 (3) |
| C6—H6 | 0.9500 | C17—H17 | 0.9500 |
| C7—H7 | 0.9500 | C18—C19 | 1.386 (3) |
| C8—C13 | 1.385 (2) | C18—H18 | 0.9500 |
| C8—C9 | 1.395 (3) | C19—H19 | 0.9500 |
| O1—S1—O2 | 118.23 (7) | C10—C9—C8 | 120.08 (19) |
| O1—S1—C14 | 108.64 (8) | C10—C9—H9 | 120.0 |
| O2—S1—C14 | 108.67 (8) | C8—C9—H9 | 120.0 |
| O1—S1—C1 | 107.45 (8) | C11—C10—C9 | 120.67 (19) |
| O2—S1—C1 | 110.16 (7) | C11—C10—H10 | 119.7 |
| C14—S1—C1 | 102.53 (8) | C9—C10—H10 | 119.7 |
| C8—C1—C2 | 118.10 (14) | C10—C11—C12 | 119.74 (19) |
| C8—C1—S1 | 110.02 (11) | C10—C11—H11 | 120.1 |
| C2—C1—S1 | 110.99 (11) | C12—C11—H11 | 120.1 |
| C8—C1—H1 | 105.6 | C11—C12—C13 | 120.05 (19) |
| C2—C1—H1 | 105.6 | C11—C12—H12 | 120.0 |
| S1—C1—H1 | 105.6 | C13—C12—H12 | 120.0 |
| C3—C2—C7 | 118.25 (15) | C8—C13—C12 | 120.79 (18) |
| C3—C2—C1 | 123.92 (15) | C8—C13—H13 | 119.6 |
| C7—C2—C1 | 117.83 (14) | C12—C13—H13 | 119.6 |
| C4—C3—C2 | 120.27 (16) | C19—C14—C15 | 121.46 (17) |
| C4—C3—H3 | 119.9 | C19—C14—S1 | 119.94 (13) |
| C2—C3—H3 | 119.9 | C15—C14—S1 | 118.53 (13) |
| C5—C4—C3 | 120.94 (18) | C14—C15—C16 | 118.82 (18) |
| C5—C4—H4 | 119.5 | C14—C15—H15 | 120.6 |
| C3—C4—H4 | 119.5 | C16—C15—H15 | 120.6 |
| C4—C5—C6 | 119.59 (18) | C17—C16—C15 | 119.91 (18) |
| C4—C5—H5 | 120.2 | C17—C16—H16 | 120.0 |
| C6—C5—H5 | 120.2 | C15—C16—H16 | 120.0 |
| C7—C6—C5 | 119.67 (18) | C16—C17—C18 | 120.89 (19) |
| C7—C6—H6 | 120.2 | C16—C17—H17 | 119.6 |
| C5—C6—H6 | 120.2 | C18—C17—H17 | 119.6 |
| C6—C7—C2 | 121.27 (17) | C17—C18—C19 | 120.03 (19) |
| C6—C7—H7 | 119.4 | C17—C18—H18 | 120.0 |
| C2—C7—H7 | 119.4 | C19—C18—H18 | 120.0 |
| C13—C8—C9 | 118.64 (17) | C14—C19—C18 | 118.88 (18) |
| C13—C8—C1 | 117.33 (15) | C14—C19—H19 | 120.6 |
| C9—C8—C1 | 124.03 (15) | C18—C19—H19 | 120.6 |
| O1—S1—C1—C8 | −61.76 (13) | C13—C8—C9—C10 | 1.9 (3) |
| O2—S1—C1—C8 | 68.31 (13) | C1—C8—C9—C10 | −177.35 (17) |
| C14—S1—C1—C8 | −176.16 (11) | C8—C9—C10—C11 | −1.8 (3) |
| O1—S1—C1—C2 | 165.65 (11) | C9—C10—C11—C12 | 0.6 (3) |
| O2—S1—C1—C2 | −64.28 (13) | C10—C11—C12—C13 | 0.5 (3) |
| C14—S1—C1—C2 | 51.25 (12) | C9—C8—C13—C12 | −0.9 (3) |
| C8—C1—C2—C3 | −59.0 (2) | C1—C8—C13—C12 | 178.46 (18) |
| S1—C1—C2—C3 | 69.33 (18) | C11—C12—C13—C8 | −0.3 (3) |
| C8—C1—C2—C7 | 121.61 (17) | O1—S1—C14—C19 | 144.32 (14) |
| S1—C1—C2—C7 | −110.03 (15) | O2—S1—C14—C19 | 14.45 (16) |
| C7—C2—C3—C4 | 0.6 (3) | C1—S1—C14—C19 | −102.16 (15) |
| C1—C2—C3—C4 | −178.76 (17) | O1—S1—C14—C15 | −38.52 (16) |
| C2—C3—C4—C5 | −0.5 (3) | O2—S1—C14—C15 | −168.39 (13) |
| C3—C4—C5—C6 | 0.4 (3) | C1—S1—C14—C15 | 75.01 (15) |
| C4—C5—C6—C7 | −0.5 (3) | C19—C14—C15—C16 | −0.3 (3) |
| C5—C6—C7—C2 | 0.7 (3) | S1—C14—C15—C16 | −177.45 (15) |
| C3—C2—C7—C6 | −0.7 (3) | C14—C15—C16—C17 | −0.2 (3) |
| C1—C2—C7—C6 | 178.68 (17) | C15—C16—C17—C18 | 0.0 (3) |
| C2—C1—C8—C13 | −120.05 (16) | C16—C17—C18—C19 | 0.7 (3) |
| S1—C1—C8—C13 | 111.14 (15) | C15—C14—C19—C18 | 1.0 (3) |
| C2—C1—C8—C9 | 59.2 (2) | S1—C14—C19—C18 | 178.09 (15) |
| S1—C1—C8—C9 | −69.59 (19) | C17—C18—C19—C14 | −1.2 (3) |
Hydrogen-bond geometry (Å, °)
| D—H···A | D—H | H···A | D···A | D—H···A |
| C1—H1···O2i | 1.00 | 2.46 | 3.449 (2) | 171 |
| C4—H4···O1ii | 0.95 | 2.66 | 3.390 (2) | 134 |
| C7—H7···O2i | 0.95 | 2.68 | 3.543 (2) | 152 |
Symmetry codes: (i) x, y+1, z; (ii) −x, −y, z−1/2.
Footnotes
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: DS2011).
References
- Altomare, A., Burla, M. C., Camalli, M., Cascarano, G. L., Giacovazzo, C., Guagliardi, A., Moliterni, A. G. G., Polidori, G. & Spagna, R. (1999). J. Appl. Cryst. 32, 115–119.
- Bernstein, J., Davis, R. E., Shimoni, L. & Chang, N.-L. (1995). Angew. Chem. Int. Ed. Engl. 34, 1555–1573.
- Etter, M. C., MacDonald, J. C. & Bernstein, J. (1990). Acta Cryst. B46, 256–262. [DOI] [PubMed]
- Farrugia, L. J. (1997). J. Appl. Cryst. 30, 565.
- Flack, H. D. (1983). Acta Cryst. A39, 876–881.
- Hooft, R. W. W. (2004). COLLECT. Bruker–Nonius BV, Delft, The Netherlands.
- Li, Y.-S. & Su, W.-K. (2005). Acta Cryst. E61, o2450–o2451.
- Macrae, C. F., Edgington, P. R., McCabe, P., Pidcock, E., Shields, G. P., Taylor, R., Towler, M. & van de Streek, J. (2006). J. Appl. Cryst. 39, 453–457.
- Mayr, H., Bug, T., Gotta, M. F., Hering, N., Irrgang, B., Janker, B., Kempf, B., Loos, R., Ofial, A. R., Remennikov, G. & Schimmel, H. (2001). J. Am. Chem. Soc. 123, 9500–9512. [DOI] [PubMed]
- Mayr, H. & Ofial, A. R. (2008). J. Phys. Org. Chem. 21, 584–595.
- Otwinowski, Z. & Minor, W. (1997). Methods in Enzymology, Vol. 276, Macromolecular Crystallography, Part A, edited by C. W. Carter Jr & R. M. Sweet, pp. 307–326. New York: Academic Press.
- Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. [DOI] [PubMed]
- Silva Corrêa, C. M. M. da, Lindsay, A. S. & Waters, W. A. (1968). J. Chem. Soc C., pp. 1872–1874.
- Spek, A. L. (2009). Acta Cryst. D65, 148–155. [DOI] [PMC free article] [PubMed]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablocks I, global. DOI: 10.1107/S1600536809050363/ds2011sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536809050363/ds2011Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report




