Abstract
The greatest therapeutic promise of human embryonic stem cells (hESC) is to generate specialized cells to replace damaged tissue in patients suffering from various degenerative diseases. However, the signaling mechanisms involved in lineage restriction of ESC to adopt various cellular phenotypes are still under investigation. Furthermore, for progression of hESC-based therapies towards clinical applications, appropriate culture conditions must be developed to generate genetically stable homogenous populations of cells, to hinder possible adverse effects following transplantation. Other critical challenges that must be addressed for successful cell implantation include problems related to survival and functional efficacy of the grafted cells. This review initially describes the derivation of hESC and focuses on recent advances in generation, characterization, and maintenance of these cells. We also give an overview of original and emerging differentiation strategies used to convert hESC to different cell types. Finally, we will discuss transplantation studies of hESC-derived cells with respect to safety and functional recovery.
Keywords: Human, embryonic, cells, progenitors, differentiation, ectoderm, endoderm, mesoderm, karyotype, feeder-free, synthetic scaffolds, hydrogels, hyaluronic acid, hepatocyte, embryoid bodies, mesenchymal, osteocytes, chondrocytes, stromal, hematopoietic, cardiomyocytes, endothelial, epithelial, neural, neurons, motor neurons, dopaminergic neurons, oligodendrocytes, myelination, degenerative disorders, transplantation
1. Introduction
Embryonic stem cells (ESC) are pluripotent cells which give rise to all somatic cell types in the embryo. ESC can be a valuable tool for understanding the complex mechanisms involved in development of specialized cells and establishment of organ structures. Moreover, the indefinite self-renewal ability and plasticity of ESC allows for in vitro generation of an unlimited number of distinct cell types, and has opened new avenues for regenerative medicine. The greatest therapeutic promise of human ESC (hESC) is to generate specialized cells to replace damaged tissue in patients suffering from various degenerative diseases. However, the signaling mechanisms involved in lineage restriction of ESC to adopt various cellular phenotypes are still under investigation. Furthermore, for progression of hESC-based therapies towards clinical applications, appropriate culture conditions must be developed to generate genetically stable homogenous populations of cells, to avoid possible adverse effects following transplantation. Other critical challenges that must be addressed for successful cell implantation include problems related to survival and functional efficacy of the grafted cells. This review initially describes the derivation of hESC and focuses on recent advances in generation, characterization, and maintenance of these cells. We also give an overview of differentiation strategies used to convert hESC to different cell types. Finally, we will discuss transplantation studies of hESC-derived cells with respect to safety and functional recovery.
2. Derivation of ESC
Following fertilization of an egg and formation of a diploid zygote, a structure referred to as a blastocyst is generated by multiple mitotic cell divisions during early embryogenesis. The blastocyst consists of an inner layer of cells called the embryoblast and an outer layer of cells called the trophoblast. The trophectoderm, also referred to as the outer cell mass, forms the extra-embryonic tissue, which eventually gives rise to the placenta, chorion, and the umbilical cord. The embryoblast, also known as the inner cell mass (ICM), develops into the embryo (Gilbert, 2006). Early studies of development of mouse blastocysts by Sherman et al. (1975) examined the growth and differentiation of trophoblast cells as well as the proliferation of the inner cell mass in long-term cultures. Four cell lines were obtained and maintained for more than a year. However, these lines contained cell types other than undifferentiated ESC, were not able to differentiate to all the three germ layers in vivo and eventually developed chromosomal abnormalities. Subsequently, established cultures of embryonal carcinoma stem cells were used to develop appropriate culture conditions and determine the optimal stage of isolation of pluripotent embryonic stem cells, leading to the successful derivation of the first stable mouse embryonic stem cell lines in 1981 (Evans and Kaufman, 1981; Martin, 1980; Martin, 1981).
The pioneering work on mouse ESC, and later advances in culturing techniques that were developed to culture nonhuman primate ESC lines (Thomson et al., 1995; Thomson et al., 1996) eventually led to the first successful generation of hESC lines by Thompson and coworkers (1998) and Reubinoff and coworkers (2000). These hESC were derived from human embryos that were produced by in vitro fertilization for clinical purposes. Human ESC lines described by Thompson and coworkers retained their pluripotency, were karyotypically normal when grown on mouse embryonic fibroblast (MEF) feeders, and fulfilled all the criteria for ESC including having the capability to generate large germ cell tumors that containing several different types of tissue (teratomas) when grafted to severe combined immunodeficient (SCID) mice (Bosma et al., 1983). As the SCID mouse lacks both B and T cells, these animals can be used to study the behavior of transplanted hESC in vivo without the need for immunosuppressant drugs.
To date, hundreds of hESC lines have been generated from donated embryos. Isolation of the ICM from the trophectoderm at the blastocyst stage has, for the most part, been achieved by immunosurgery or mechanical dissection. The first hESC lines were established using the immunosurgical method, which requires the use of animal-derived products including anti-human serum antibodies and guinea pig complement (Bosma et al., 1983; Cowan et al., 2004; Ellerström et al., 2006; Reubinoff et al., 2000). Exposure to animal-derived products would prevent the later use of hESC for transplantation therapies, due to possible transfer of pathogens which would potentially initiate the patient’s innate immune mechanisms leading to an increased risk of graft rejection. Therefore, mechanical or enzymatic isolation of the ICM from the trophectoderm in a manner that avoids contact between the ICM and animal products during the derivation procedure would be advantageous for future clinical applications (Amit and Itskovitz-Eldor, 2002; Genbacev et al., 2005; Strom et al., 2007). In addition, laser beams have been used to derive hESC lines by creating a small opening at the zona pellucida that encapsulates the blastocyst, followed by laser-assisted isolation of the ICM (Turetsky et al., 2008).
Generation of hESC lines from the inner cell mass at the blastocyst stage has thus far obligated the destruction of the embryo, which has raised ethical and political concerns. In order to address this issue, much work has been devoted to isolating cells from earlier stages of embryonic development without destruction of the embryo. Initial attempts at removal of one cell at the 8-cell or morula stage resulted in variable success rates and required co-culture of isolated blastomeres with established hESC lines (Klimanskaya et al., 2006; Klimanskaya et al., 2007; Strelchenko et al., 2004). Blastomere differentiation to ICM was highly inefficient because the blastomere-derived aggregates mostly gave rise to trophectoderm-like vesicles. To circumvent this problem and increase the efficiency of hESC derivation, a modified approach using culture media supplemented with laminin was employed (Chung et al., 2008). This strategy was almost as efficient as conventional methods used to derive hESC lines from whole blastocysts. The rationale behind this essential effect of laminin was suggested to be simulation of the natural ICM niche, which prevented polarization of the blastomeres into ICM and trophectoderm. In addition, optimization of culture conditions for this new procedure allowed successful generation of blastomere-derived hESC in feeder-free conditions, eliminating the need for co-cultures with animal-derived feeder layers or previously established hESC lines.
3. In vitro growth and maintenance of hESC
Indefinite self-renewal is a fundamental hallmark of successful hESC culture. When the first hESC lines were derived, MEF feeder layers were used to support the propagation of hESC in the primitive undifferentiated state (Bosma et al., 1983; Reubinoff et al., 2000). Ever since, in order to move toward xeno-free hESC culturing systems, various approaches using human-derived cell types including fibroblast feeder cells derived from fallopian tube epithelium (Bongso et al., 1994), fetal foreskin, muscle (Amit et al., 2003; Richards et al., 2002), bone marrow (Cheng et al., 2003), or amniotic epithelium (Miyamoto et al., 2004), have been established. Alternatively, hESC can be maintained in feeder-free environments in the presence of extracellular matrices such as matrigel and fibronectin. Nevertheless, media conditioned by feeder fibroblast cells and supplementation with basic fibroblast growth factor (bFGF) were initially used to maintain hESC in an undifferentiated state in such feeder-free conditions (Van Hoof et al., 2008; Xu et al., 2001).
In feeder-free culture systems, hESC often give rise to fibroblast or stromal-like cells that may serve as supportive cells in maintaining the undifferentiated growth of hESC. Studies examining the nature of these feeder cells provided evidence that feeder cells derived from hESC can be used to support their own growth (Stojkovic et al., 2005; Wang et al., 2005). Although these cells fulfill the growth requirements of hESCs, they are not immortal and will senesce after several passages, thereby limiting their continual use. Derivation of new feeder cells can be cumbersome and may result in highly variable culture systems.
Thus, additional efforts are required to completely eliminate the need of feeder cells and establish a defined environment for hESC growth. Studies focused on secreted factors released from MEF feeder layers, that have the capacity to maintain self-renewal of hESC, have identified a number of factors responsible for maintenance of hESC pluripotency (Cai et al., 2006; Chin et al., 2007; Lim and Bodnar, 2002; Xu et al., 2005). In addition, high concentrations of bFGF and repression of bone morphogenetic protein (BMP) signaling by noggin have been suggested to sustain undifferentiated proliferation of hESC in serum-free media (Beattie et al., 2005; Pera et al., 2004; Wang et al., 2005; Xu et al., 2005). Other multifaceted exogenous treatments of hESC with cocktails of human recombinant proteins and signaling molecules including activin A and transforming growth factor-beta 1 (TGF-β1) have also been employed for hESC culture (Beattie et al., 2005; James et al., 2005; Lu et al., 2006). Although there is some evidence that maintaining hESC in feeder-free culture systems can decrease their stability and predispose them to developing genetic abnormalities (Draper et al., 2004), whether this applies to all feeder-free culture systems is unknown.
Feeder-free culture systems using medium that contains only human-sourced recombinant proteins have been developed for culture of hESC and are commercially available; however, these conditions may not be optimal for a wide range of hESC lines (Rajala et al., 2007). Therefore, even though feeder-free and serum-free defined conditions for maintenance of hESC have been developed, further investigations are needed to determine the factors responsible for maintenance of the pluripotent phenotype and stability of hESC lines in general.
3.1. Methods for expansion of hESC
hESC have a high capacity for self renewal and need to be frequently passaged and transferred to freshly-prepared cultures upon confluency. Typically, hESC in co-culture with various murine or human cell types are isolated from their supportive feeder cell layer by enzymatic treatment or mechanical dissection. Commonly used animal-derived enzymes include collagenase IV, dispase, and trypsin (Hoffman and Carpenter, 2005). In view of potential clinical applications, recombinant animal protein-free enzymes and human collagenase have also been used for hESC culture (Crook et al., 2007). Enzymatic passaging as compared to mechanical isolation is less labor intensive and can easily be applied on a large scale, providing a more defined and reproducible culture system. However, enzymatic methods used in propagation of hESC have been strongly linked to occurrence of genetic abnormalities (Brimble et al., 2004; Draper et al., 2004; Mitalipova et al., 2005). To address concerns associated with enzymatic passaging, a number of groups have established enzymatic techniques involving bulk passaging and single cell dissociation without compromising pluripotency or genetic stability over extended culture periods (more that 100 passages) (Ellerström et al., 2007; Suemori et al., 2006). Furthermore an automated method has also been developed for robust mechanical large-scale expansion of undifferentiated hESC (Joannides et al., 2006).
3.2. Three-dimensional culture of hESC
Ideal culture conditions for hESC should recapitulate the niche and physical environment in which hESC naturally reside. Generally, hESC are grown in colonies which must remain within a narrow size range during passaging procedures to prevent differentiation. Thus, it is clear that cell-cell interactions as well as paracrine or autocrine signals within colonies play essential roles in maintaining hESC in an undifferentiated state.
There is a profound difference in cell signaling, gene expression and organization of cells in two dimensional cultures (2D) as compared the three dimensional (3D) culture systems, which more closely resemble the in vivo hESC environment (Lee et al., 2007; Postovit et al., 2006). A study by Nur-E-Kamal et al. (2006) showed that physical and mechanical cues are also crucial in emulating the native microenvironment of mouse ESC, as 3D culture greatly enhanced cell proliferation and self-renewal as compared to growth on 2D tissue culture surfaces. The authors used synthetic polyamide matrix scaffolds to mimic the fibrillar network of the basement membrane. The resultant gain in proliferative ability was correlated with the cellular mechanotransduction by activation of the small GTPase Rac, the activation of phosphoinositide 3-kinase (PI3K) pathway and enhanced expression of Nanog (Nur-E-Kamal et al., 2006).
Another type of polymeric fibrous scaffold is made of cellulose acetate, which that creates a meshwork permitting surface immobilization of various extra cellular matrix (ECM) molecules and growth factors. This type 3D matrix has been used in the study of uncommitted human embryonic germ cell derivatives, which share some properties with hESC. The enhanced interaction of cells in 3D cultures has been reported to lead to improved maintenance of, multipotency, proliferation, and cell survival (Yim and Leong, 2005).
Encapsulation of hESC has also been carried out in biocompatible poly-glycerolco-sebacate-acrylate (PGSA) elastomers, which are photo-polymerized to form porous scaffolds. Although hESC continue to proliferate under these conditions, the cells were found to have formed differentiating EB structures within the pores after seven days of culture and did not remain in an undifferentiated state (Gerecht et al., 2007a). Thus far, the most physiologically relevant matrix developed for culture of undifferentiated hESC is composed of hyaluronic acid (HA) hydrogel. The HA hydrogel replicates major components of the ECM that are abundant in embryos and stem cell niches, and can maintain hESC in undifferentiated state while preserving their full differentiation capacity (Gerecht et al., 2007b).
Similar to PGSA scaffolds, structure and chemistry of synthetic hydrogels can be modified by external stimuli to manipulate the temporal and spatial availability of bioactive molecules. Addition of components such as growth factors and ECM proteins can also be used in inducing differentiation. Development of methods capable of controlling cell-cell interactions in scalable culture by encapsulation of hESC in size-specified scaffolds is indeed highly desirable; however, the release of cells from these 3D structures requires enzymatic digestion. For example, release of hESC from HA hydrogel is achieved by addition of hyaluronidase into the growth medium, and it is uncertain how the enzymatic treatment affects hESC in long-term cultures.
4. Characterization of hESC
hESC grow in tightly packed colonies and maintain defined borders at the periphery of colonies. High nucleus to cytoplasm ratio and prominent nucleoli are typical features of individual hESC within colonies. hESC are also characterized by expression of a number of cell surface markers and transcription factors including stage-specific embryonic antigen-4 (SSEA-4), SSEA-3, TRA antigens, Oct3/4, Nanog, and absence of hESC negative markers, such as SSEA-1 (Carpenter, Rosler, and Rao, 2003; Chambers et al., 2003; Draper et al., 2004; Heins et al., 2004; Nichols et al., 1998).
Analysis of self-renewal capacity of hESC is often evaluated by telomere length and telomerase activity measurement after prolonged in vitro cultivation (Brandenberger et al.,2004; Carpenter et al., 2004). In general, human somatic cells have a limited lifespan and can only escape replicative senescence upon acquisition of genetic alterations (Hayflick and Moorhead, 1961). In contrast, it has been established that the intrinsic mechanisms in hESC responsible for their immortality are not caused by genetic alterations or inactivation of the well-known p53 and Rb tumor suppressor pathways (Bradenberger et al., 2004; Rosler et al., 2004).
It is, however, evident that hESC can acquire karyotypic abnormalities during prolonged maintenance in vitro, which may be correlated to tumorigenic events that occur in vivo (Baker et al., 2007; Draper et al., 2004; Mitalipova et al., 2005). Chromosomal aberrations are believed to be a result of in vitro adaptation and selection, because abnormal rapidly-dividing cells gradually predominate and eventually take over the entire diploid culture (Baker et al., 2007). Also, the most recurrent chromosomal changes in hESC in vitro reflect over-expression of genes associated with pluripotency, cell proliferation, and anti-apoptosis, which are highly comparable to genetic changes seen in common human cancers (Baker et al., 2007). Consequently, for therapeutic use of hESC, in addition to traditional Giemsa-stained karyotyping that detects large chromosomal changes, methods such as fluorescent in situ hybridization, DNA microarray, whole-genome single nucleotide polymorphism, and short tandem repeat analysis would be require to ensure maintenance of a normal genetic signature at a higher level of resolution (Brimble et al., 2004; Draper et al., 2004; Josephson et al., 2006; Maitra et al., 2005; Mitalipova et al., 2005). How such data would be interpreted, and what level of genetic abnormality would be required to disqualify a cell line for clinical use are major unanswered questions. Some level of genetic alteration would almost inevitably be incurred by the process of maintaining cells, and it would probably be necessary to determine for the amount and type of genetic alterations that would be permissible.
Functional confirmation of the multipotent nature of hESC is generally achieved by examining their potential to differentiate into all three germ layers (ectoderm, mesoderm and endoderm) in vitro as well as in vivo. In vitro, hESC are allowed to randomly differentiate as embryoid bodies (EBs), which are aggregates of cells grown in suspension culture, followed by immunocytochemical analysis, or measurement of expression of genes associated with the three germ layers by RT-PCR. The in vivo test for pluripotency of hESCs is teratoma formation in SCID mice (Bosma et al., 1983; Reubinoff et al., 2000).
5. Directed in vitro differentiation of hESC
Theoretically, hESC have the capability to give rise to all somatic cell types present in an embryo. Although hESC exhibit this potential, for development of new therapeutic strategies the challenge still remains in understanding the intricate mechanisms that govern stem cell growth, cell fate decisions, tissue generation and organogenesis. Systematic studies of the developmental biology of various cell types are leading to increasing success in lineage-specific differentiation of hESC.
Aggregation of hESC into 3-dimensional EB structures has a general inductive influence, and is frequently used as a first step for producing in vitro differentiation of many cell lineages. Spontaneous differentiation produced by EB formation or allowing increased confluency of hESC cultures generally results in a small fraction of cells with any particular phenotype. Therefore, considerable efforts are being made in identifying exogenousbiochemical supplementation that can direct cell-specific differentiation, and results in enrichment of desired cell types.
5.1. Cell-specific endodermal differentiation
Examples of major cell types that have been derived from the endoderm using hESC include hepatocytes (Agarwal et al., 2008; Cai et al., 2007; Lavon and Benvenisty, 2005), insulin-producing β cells (Assady et al., 2001; Brolén et al., 2005; Edlund, 1999; Segev et al., 2004), and lung epithelium (Samadikuchaksaraei et al., 2006; Van Vranken et al., 2005; Wang et al., 2007) for development of potential cell replacement therapy in acute liver failure/hepatitis, type I diabetes, and pulmonary diseases.
The isolation of hepatocytes from randomly-differentiated hESC is relatively difficult, because many markers characteristic of liver cells are also expressed in the extraembryonic endoderm, which contributes to the formation of the yolk sac (Lavon and Benvenisty, 2005). Selective induction and isolation of definitive endoderm can be achieved by activin A treatment in combination with fluorescence-activated cell sorting (FACS) of cells that express lineage-specific cell-surface proteins including CXCR4 (Agarwal et al., 2008; Cai et al., 2007).
Hepatocytic induction is then accomplished by sequential addition of the inducing factors FGF4, BMP2, and hepatocyte growth factor (HGF). The highest reported yield of albumin-secreting hepatocytes was about 70% (Agarwal et al., 2008). Additionally, transplantation of hepatocyte precursors or mature hepatocytes to animals with tetrachloride-induced liver injury has resulted in successful integration and participation in liver repair.
The signaling pathways in the developing pancreas are very complex and include the TGF-β, notch, hedgehog, FGF, and epidermal growth factor (EGF) pathways (Edlund, 1999). A study by Assady et al. (2001) suggested that pancreatic β-cells could be produced by random differentiation of hESC as EBs. However, only 1–3% of cells within EBs were insulin-positive β-cells. Segev et al. (2004) used a more efficient methodology for derivation of immature pancreatic cells involving growth in insulin-transferrin-selenium-fibronectin medium, followed by supplementation with N2, B27, and bFGF. Nicotinamide and decreased glucose concentrations in the media were also used to improve insulin secretion in response to glucose. Recently, co-transplantation of randomly differentiated hESC with mouse embryonic dorsal pancreas has resulted in differentiation of β-cell–like cell clusters capable of insulin synthesis (Assady et al., 2001).
Another target of hESC differentiation is the alveolar epithelial type II (ATII) cells that constitute the primary lung-lining tissue. These cells might possibly be used for treatment of lung chronic respiratory disease as well as pulmonary genetic disorders. Generation of mature ATII cells from hESC has been carried out via EB formation in small airway growth medium (Samadikuchaksaraei et al., 2006), or by co-culture with pulmonary mesenchyme (Van Vranken et al., 2005). Both of these strategies gave rise to small percentages of cells with the pulmonary epithelial/ATII phenotype. Wang and colleagues (Wang et al., 2007) have recently achieved remarkable success in deriving an almost pure population of ATII cells from hESC by genetic selection. ATII cells are characterized by their unique ability to synthesize and secrete surfactant protein Wang et al. (2007) used nonviral transfection of hESC with a neomycin transgene under the control of a surfactant protein C promoter, and were thereby able to produce an almost homogenous population of surfactant protein C-expressing ATII cells under neomycin selection.
5.2. Cell-specific mesodermal differentiation
Bone and cartilage cells are of therapeutic interest because of their potential use for the treatment of osteoporosis and osteoarthritis. Mesodermal differentiation of mesenchymal precursors has been achieved by co-culturing hESC with murine OP9 stromal cells, followed by FACS using antibodies against the lineage-specific cell-surface protein CD73. In this paradigm, mesenchymal precursors gave rise to chondrocytes, osteocytes, and skeletal myoblasts under defined conditions (Barberi et al., 2005). A more selective system for osteogenic derivation from hESC was reported by Sottile et al. (2003), where mineralization of EB-derived cells was induced by an osteogenic supplement consisting of ascorbic acid phosphate, β-glycerophosphate and dexamethasone.
Major advances have been made in the treatment of ischemic heart disease and cardiomyopathy by heart transplantation. The number of patients waiting for cardiac transplantation continues, however, to rise, and the possibility of using cardiomyocytes derived from hESC for transplantation is a promising future alternative. Kehat et al. (2001) demonstrated that hESC differentiation as EBs can commit hESC to form mesodermal cells, and eventually contracting cardiac tissue. Recently, a technique was reported for enhancing the derivation and purity of cardiomyocytes directly from hESC, by treatment of high-density undifferentiated monolayer cultures with activin A and BMP4. Most importantly, the authors demonstrated that transplantation of these cells into damaged rat heart in combination with insulin-like growth factor 1 (IGF-1) and a multi-component prosurvival cocktail results in improvement in heart function (Laflamme et al., 2007).
Generation of two additional cell types, blood-forming hematopoietic cells, and endothelial cells from hESC- derived mesoderm precursors has also been established. These cell types could potentially be used for blood transfusion, analyzing hematopoietic and vascular defects, or for cell transplantation in the treatment of immunodeficiency disorders. It has been suggested that the cytokines interleukin 3 and interleukin 6, in combination with BMP4, promote differentiation of hESC into hematopoietic cells subsequent to EB formation (Chadwick et al., 2003). The use of this combination of factors resulted in conversion of the majority of treated EBs to hematopoietic cells, as indicated by the expression of the hematopoietic progenitor marker CD45. Alternatively, a co-culturing system with the mouse stromal cell lines S17 and OP9, or the yolk sac endothelial cell line C166, can be used to derive hematopoietic-like cells expressing CD34 (Kauman et al., 2001; Vodyanik et al., 2005).
Isolation of endothelial cells from randomly-differentiated EBs has been carried out by flow cytometry cell sorting using antibodies against platelet endothelial cell-adhesion molecule-1 (Levenberg et al., 2002). In this study, endothelial cell transplantation experiments using biodegradable scaffolds showed formation of functional vessel-like structures that were integrated with the animal’s vascular network (Levenberg et al., 2002).
5.3. Cell-specific ectodermal differentiation
The ectoderm derivatives include external ectoderm, neural crest, and neural tube. These structures give rise to cells of the epidermis, external sense organs, and the peripheral and central nervous system (Gilbert, 2006).
5.3.1. Non-neuronal derivatives of the ectoderm
Retinoic acid (RA) and BMP signaling have been found to act synergistically to enhance the induction of hESC to keratinocytes, which are the major component of skin. Addition of low concentrations (1 μM) of RA strongly upregulated expression of factors involved in epidermal morphogenesis and ectodermal specification, while repressing early neural induction (Metallo et al., 2007). Production of keratinocyte precursors from hESC can also be increased by several mesenchymal inducers, including fixed PA6 stromal feeder cells, BMP4, and collagen (Aberdam et al., 2008). In terms of evaluation of these cells for of suitability for cell transplantation therapy, hESC-derived keratinocytes have been expanded while retaining a normal karyotype, and transplanted into mice without generating teratomas.
Age-related macular degeneration is one of the leading causes of human blindness, and is mainly caused by loss and dysfunction of the retinal pigment epithelium. Transplantation studies indicate that donor retinal pigment epithelium can support photoreceptor survival and restore visual function in animals with degenerated retinal pigment epithelium (Coffey et al., 2002). The first retinal pigment epithelium cells to be isolated from hESC were generated by randomly differentiating colonies of hESC followed by mechanical isolation of epithelial-like clusters, or by transferring differentiated EBs to gelatin-coated substrates (Klimanskaya et al., 2004). Enhanced conversion of hESC to retinal pigment epithelial cells has been achieved by co-culturing hESC with a human retinal pigment epithelial cell line, or with human basement membranes of the chorio-capillaris endothelium and retinal pigment epithelium (Gong et al., 2008).
5.3.2. Differentiation of the neuroectoderm
Generating functional neurons from hESC with the aim of treating neurodegenerative diseases is the subject of intensive investigation. Shortly after derivation of the first hESC lines, Reubinoff et al. (2000) described isolation of expandable neural progenitor cells from hESC that had been cultivated for four to seven weeks at a high density in vitro. The authors illustrated that the neuroepithelium contained areas of differentiating hESC colonies, identified by expression of the embryonic polysialylated neural cell adhesion molecule (PSA-NCAM), and had distinct morphological features. These areas were mechanically dissected and expanded as neural aggregates or spheres in serum-free media. Neural induction was achieved by plating the spheres on coverslips coated with poly-D-lysine and laminin, which resulted in emergence of cells expressing the neuronal markers β-tubulin and microtubule-associated protein 2 (MAP2). They also identified a subset of the neuronal cells as being glutaminergic and GABAergic neurons, as shown by the expression of glutamate and glutamic acid decarboxylase (GAD).
Reubinoff and coworkers (2001) also optimized the expansion of hESC-derived NPC by addition of B27 supplement, human recombinant epidermal factor EGF and the mitogen bFGF. The expanded NPC were capable of differentiating into all three major neural lineages (neurons, astrocytes, oligodendrocytes) in vitro as well as in vivo. Lineage tracing studies showed that NPC grafted to the ventricles of newborn mice differentiated in a region-specific manner, according to normal developmental patterning signals. For example, neuronal differentiation was specifically detected in the olfactory bulb, where postnatal neurogenesis occurs (Reubinoff et al., 2001).
At about the same time, Zhang et al. (2001) used a different protocol with similar success in generating enriched populations of NPC from hESC. In that study, differentiating EBs were treated with insulin, transferrin, progesterone, heparin, and bFGF. Continuous exposure to bFGF led to formation of monolayers of neural tube-like rosettes that were isolated by dispase enzymatic treatment. Similar to the study by Reubinoff et al. (2001), the generated NPC were capable of generating oligodendrocytes, astrocytes, and mature neurons both in vitro and after transplantation into mice.
These observations confirming the multi-lineage differentiation potential of hESC-derived NPC, and promising indications of survival and integration of these cells in vivo, set the stage for future developments of methods for selective differentiation of different neuronal phenotypes that could potentially be used to treat several central nervous system disorders. Indeed, studies examining the signals and factors that govern the proliferation and cell fate specification of neural progenitors are accumulating rapidly.
Generation of transplantable motor neurons from hESC could have potential for treating victims of spinal cord injuries, or degenerative diseases such as amyotrophic lateral sclerosis. The first functional motor neurons originating from hESC were described by Li et al. (2005). In that study, Li and colleagues used the previously described method (Zhang et al., 2001) to generate NPC cells, which were subsequently induced to become motor neurons by addition of RA to the culture medium. Further maturation to postmitotic motorneurons was induced by the ventralizing morphogenic protein SHH. An interesting aspect of this study was the specific temporal effect of RA on motor neuron induction, in that RA could induce only early, but not late neuroectodermal cells, to differentiate into motorneurons (Li et al., 2005). This occurred through upregulation of expression of HOX genes that are involved in assigning the rostrocaudal positional identity of spinal motor neurons. The functionality of the generated motor neurons was confirmed by electrophysiological experiments and establishment of neuromuscular transmission in motorneuron-myotube co-cultures (Li et al., 2005).
In another study, directed differentiation of motor neurons was achieved by addition of RA/SHH extrinsic cues to cultures of differentiating NPC generated from hESC (Lee et al., 2007). With respect to clinical potential, transplantation of hESC-derived motor neurons in developing chick embryo spinal cord showed that these cells were capable of survival and directed axonal growth over relatively long distances (Lee et al., 2007). Nevertheless, transplantation in adult rats did not result in axonal growth to outside the CNS. It should be mentioned that, although caudal and ventral patterning was achieved by RA and SHH, the initial neural induction of hESC was obtained by co-culture with MS5 mouse stromal cells, which would preclude the use of motorneurons generated by this particular strategy for any type of human transplantation therapy (Lee et al., 2007).
In a later study by Li et al. (2008), neural induction medium containing heparin and cyclic adenosine monophosphate (cAMP) in addition to RA and SHH was successful in generating a nearly homogenous population of ventral spinal progenitor cells, with highly efficient generation of motor neurons.
Promoting remyelination for treatment of neurologic disorders caused by demyelination of motor neurons is another potential application of hESC-derived cells. One of the strategies used to promote remyelination involves transplantation of oligodendrocytes, which produce the myelin sheath of motor neurons and are essential for normal signal conduction. In 2005, Keirstead and his associates demonstrated that transplantation of hESC-derived oligodendrocyte progenitor cells (OPC) produced by glial restriction media, can lead to remyelination of motorneurons, and recovery of motor function after spinal cord injury in rats (Keirstead et al., 2005). Following further assessment of the safety concerns associated with OPC transplantation in animal models (Cloutier et al., 2006), Geron Corporation obtained FDA clearance in January 2009 to begin the first human clinical trials of hESC-derived cells in the United States (see Alper 2009). OPC were generated from the H1 hESC line under current good manufacturing practices without the use of feeder cells, in defined media containing only human recombinant proteins. The Phase I trial is designed to assess the safety of transplantation of OPC in patients with acute thoracic spinal cord injuries and will be carried out at multiple medical centers.
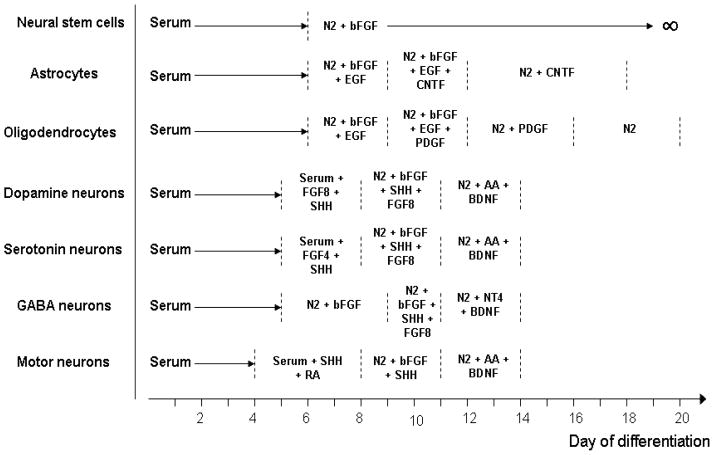
A number of additional studies have reported the production of multiple neuronal subtypes, including cholinergic, serotonergic, GABAergic, and dopaminergic (DA) neurons, from hESC (Erceg et al., 2008; Gerrard et al., 2005; Perrier et al., 2004; Yan et al., 2005). As previously mentioned, protocols used for neural conversion of hESC generally give rise to a mixture of neuronal phenotypes. Previous studies of neural differentiation of mouse ESC have established protocols for growth factor-mediated lineage selection and survival-promoting factors of neuronal cells (Barberi et al., 2003; Lee et al., 2000; Okabe et al., 1996). As a general strategy for obtaining selective neuronal differentiation, factors with effects on the anteroposterior (AP) or the dorsoventral (DV) neuronal patterning in combination with specific neurotrophins are used at specific stages during in vitro ESC differentiation. In a comprehensive study of neural development of mouse ESC, Barberi et al. used a stromal feeder-based differentiation system to generate early ectodermal cells (6 days co-culture) and identified various combinations of factors that govern neural and neuronal subtype specification (Fig. 1) (Barberi et al., 2003). It should be noted that these differentiation strategies for mouse ESC cannot be directly applied to hESC without some modifications.
Fig. 1.
Neural subtype specification from neural progenitors derived from mouse ESC using various combinations of inducing factors. AA, ascorbic acid; bFGF, basic fibroblast growth factor; BDNF, brain-derived neurotrophic factor; CNTF, ciliary neurotrophic factor; EGF, epidermal growth factor; FGF4, fibroblast growth factor 4; FGF8, fibroblast growth factor 8; NT4, neurotrophin-4; PDGF, platelet-derived growth factor; RA, retinoic acid; SHH, sonic hedgehog. {Adapted from Barberi et al. (2003), [100]}.
To date, the majority of studies on neural differentiation of hESC have been focused on generation of dopamine producing neurons of the midbrain subtype, due to their potential application in cell replacement therapy for Parkinson’s disease. Established protocols used to generate DA neurons include allowing spontaneous differentiation of hESC, followed by addition of DA inducing molecules, SHH and FGF8, and later neurotrophic factors, or by culturing hESC on feeder cells from animal or human origin that have the ability to direct hESC to become DA neurons.
Kawasaki and coworkers in Japan discovered in 2000 that certain mouse stromal cell lines had a neural and DA promoting effect on mouse ESC (Kawasaki et al., 2000). The authors showed that the activity of the stromal cells was not mimicked by FGF8/SHH, or Wnt signaling, previously known to be key factors in development and patterning of midbrain DA neurons. Thus, this strategy was established as a new approach to generate DA neurons and was termed “stromal-derived inducing activity” (SDIA).
Our group as well as others have adapted this approach to generate DA neurons from hESC. When the hESC line BG01 was cultured on the mouse stromal cells for three weeks, approximately 87% of colonies contained large numbers of TH+ cells (Zeng et al., 2004). The TH+ neurons generated by SDIA had midbrain characteristics, as determined by expression of Nurr1 and Pitx3 transcription factors that are strongly associated with midbrain DA neurons. The DA neurons were functional in vitro as confirmed by electrophysiological assessments and release of dopamine. However, the survival of TH+ neurons grafted into the striatum of parkinsonian rats was very limited. A parallel study of DA induction of hESC by Perrier and colleagues (2004), combined SDIA with SHH and FGF8 patterning molecules, ascorbic acid, and various neurotrophic factors including BDNF, GDNF, TGF-β3, dcAMP and demonstrated that the yield and functional properties of TH+ neurons were highly dependent on exposure to SHH and FGF8.
Other feeder cells that possess DA-inducing activity, and that have been used to generate DA neurons from ESC, include testis-derived sertoli cells (Yue et al., 2006), meningeal cells (Hayashi 2008), and striatal or mesencepahlic astrocytes (Buytaert-Hoefen et al., 2004; Roy et al., 2006). Secreted factors produced by astrocytes have also been reported to promote neurogenesis and induction of DA neurons (Nakayama et al., 2003).
Yan and coworkers (2005) demonstrated neural and DA induction of hESC in the absence of any type of feeder cells by addition of SHH and FGF8 to EB-derived neural rosettes which were manually isolated from mixed cultures. The resulting TH+ neurons comprised 50%–60% of the total neuronal population and were electrophysiologically active. Other differentiation paradigms have included addition of an NPC expansion step to this protocol in order to generate a more pure population of DA neurons (Cho et al., 2008).
Although mouse stromal cells that possess SDIA activity are considered as one of the most efficient tools for converting hESC to DA neurons, the use of animal cells would preclude any downstream clinical application due to possible transfer of xenogeneic material. To understand the molecular activity of SDIA, we further assessed the activity of stromal cells and found that stromal cell surface activity promoted hESC survival and was able to enhance overall neurogenesis, whereas soluble secreted factors provided DA lineage-specific instructions (Vazin et al., 2008). We then examined the gene expression profile of potent PA6 stromal cells as compared to that of cell lines lacking the DA-inducing effect (Vazin et al., 2009). Several soluble factors and growth-inducing proteins potentially responsible for the DA phenotype-promoting component of SDIA were identified, based on high levels of expression in potent DA-inducing PA6 cells. Testing of these factors showed that a combination of four factors, stromal cell-derived factor 1, pleiotrophin, insulin-like growth factor 2, and ephrin-B1, termed “SPIE” was sufficient to induce DA neuronal differentiation from hESC. The combination of these four factors mimicked SDIA activity, providing an approach for differentiating DA neurons from hESC in a culture system that is potentially suitable for clinical applications (Vazin et al., 2009).
Transplantation of DA precursors or neuronal cells is still at the stage where survival and integration needs to be optimized, as the majority of studies focusing on neural transplantation have reported limited or no survival of DA neurons. A few studies have, however, reported more encouraging results. A study by Roy et al. (2006) transplanted hESC-derived DA progenitors induced with immortalized human fetal midbrain astrocytes in the presence of SHH and FGF8, and illustrated that about 21% of the total number of transplanted cells (5 × 105 cells) were TH+. Long-lasting behavioral recovery was found in animals that received cell implants. The enhanced viability of the TH+ neurons post-transplantation may have been caused by the influence of fetal midbrain astrocytes during development or specification of these neurons.
A more recent study by Chiba and colleagues (2008) has indicated that SDIA-induced DA differentiation of hESC can be improved by addition of the BMP inhibitor noggin. Importantly, the number of TH+ cells found in animals transplanted with hESC treated with noggin was five times more (average of about 500 cells/animal) than the animals that received hESC induced by SDIA alone. The enhanced in vivo viability of TH+ cells was also reflected in animal behavioral recovery.
As previously discussed, the patterning of the neural tube along its DV and AP axis is determined by specific concentrations of morphogens including SHH, BMP, FGF and RA. Other important aspects involved in regional specification of NPC are the temporal effect of these factors, as well as the duration of signaling. There is evidence indicating that NPC progressively lose their differentiation potential and can no longer be regionally specified in response to instructive patterning cues after extended in vitro culturing (Machon et al., 2005; Santa-Olalla et al., 2003).
A recent study by Elkabetz and colleagues (2008) has identified a novel population of hESC-derived neural stem cells with a unique gene expression profile, termed neural rosette cells (R-NSC), which are isolated at an earlier stage of differentiation, as compared to the previously described NPC. Forse1 was used as a marker to isolate these early rosette stage cells, which adopted an anterior forebrain characteristic in the absence of extrinsic patterning factors. In contrast to NSC, the R-NSC could be re-specified toward caudal neuronal fates including motor neurons and midbrain DA neurons by SHH/RA and SHH/FGF8 treatment, respectively. This study also illustrated the in vivo survival and phenotype maintenance of these two rosette stage-derived neuronal phenotypes.
These findings provide evidence that neuronal plasticity of NSC is highly dependent on the developmental stage and restricted to a specific time window. Selective expansion of neural stem cells that retain their ability to differentiate towards specified neurons is of great potential value. Moreover, generation of restricted NSC has clinical relevance, as such cells have been reported to have a tendency to migrate towards the site of injury and rescue degenerating neurons following implantation in animal models (Bjugstad et al., 2008; Ourednik et al., 2002).
Characterization studies of SDIA-mediated neural induction have also suggested that midbrain regional identity can only be established during early stages of ESC differentiation (Parmar and Li, 2007). In addition, it has been suggested that early exposure of FGF8, before the onset of the neural stem cell transcription factor Sox1, is necessary for generation of DA neurons with a midbrain phenotype (Yan et al., 2005). Signaling duration is also known to affect the mechanisms that underlie the patterning role of factors. For example cells are known to respond analogously to varying concentrations of SHH, or to varying duration of exposure to this factor (Dessaud et al., 2007).
6. Conclusions
Lineage restriction and induction of differentiation of hESC to produce specific cell types requires a complex interplay between graded concentrations of several patterning cues under temporal constraints, which presents a challenge in experimental control of directed differentiation of hESC. Moreover, the extracellular environment plays an important role in differentiation, which must be assessed to completely understand the signaling mechanisms that dictate specification of hESC to distinct cellular identities.
Recent studies have shown remarkable success in unveiling the key events that occur during hESC differentiation and cell fate decisions. Additionally, identification of genetic profiles and novel molecular markers characteristic of specific cellular phenotypes is likely to assist in better defining the stages of progression from unspecialized cells to cell types of interest. Yet, translation of these promising basic science discoveries into successful cell replacement therapy in human subjects is likely to require sophisticated methods for assessing genetic and phenotypic stability.
The potential for uncontrolled growth of hESC-derived cell progeny will need to be carefully examined, and the in vivo survival and functional efficacy of these cells will need to be improved. Finally, development of more efficient methods for detection and elimination of residual undifferentiated hESC that may potentially cause tumors is imperative to assure safety of hESC-based therapies.
Acknowledgments
Research was supported by the IRP of NIDA, NIH, DHHS.
References
- Aberdam E, Barak E, Rouleau M, de LaForest S, Berrih-Aknin S, Suter DM, et al. A pure population of ectodermal cells derived from human embryonic stem cells. Stem Cells. 2008;26(2):440–444. doi: 10.1634/stemcells.2007-0588. [DOI] [PubMed] [Google Scholar]
- Agarwal S, Holton KL, Lanza R. Efficient differentiation of functional hepatocytes from human embryonic stem cells. Stem Cells. 2008;26(5):1117–1127. doi: 10.1634/stemcells.2007-1102. [DOI] [PubMed] [Google Scholar]
- Alper J. Geron gets green light for human trial of ES cell-derived product. Nat Biotechnol. 2009;27(3):213–214. doi: 10.1038/nbt0309-213a. [DOI] [PubMed] [Google Scholar]
- Amit M, Itskovitz-Eldor J. Derivation and spontaneous differentiation of human embryonic stem cells. J Anat. 2002;200(Pt 3):225–232. doi: 10.1046/j.1469-7580.2002.00032.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Amit M, Margulets V, Segev H, Shariki K, Laevsky I, Coleman R, et al. Human feeder layers for human embryonic stem cells. Biol Reprod. 2003;68(6):2150–2156. doi: 10.1095/biolreprod.102.012583. [DOI] [PubMed] [Google Scholar]
- Assady S, Maor G, Amit M, Itskovitz-Eldor J, Skorecki KL, Tzukerman M. Insulin production by human embryonic stem cells. Diabetes. 2001;50(8):1691–1697. doi: 10.2337/diabetes.50.8.1691. [DOI] [PubMed] [Google Scholar]
- Baker DE, Harrison NJ, Maltby E, Smith K, Moore HD, Shaw PJ, et al. Adaptation to culture of human embryonic stem cells and oncogenesis in vivo. Nat Biotechnol. 2007;25(2):207–215. doi: 10.1038/nbt1285. [DOI] [PubMed] [Google Scholar]
- Barberi T, Klivenyi P, Calingasan NY, Lee H, Kawamata H, Loonam K, et al. Neural subtype specification of fertilization and nuclear transfer embryonic stem cells and application in parkinsonian mice. Nat Biotechnol. 2003;21(1):1200–1207. doi: 10.1038/nbt870. [DOI] [PubMed] [Google Scholar]
- Barberi T, Willis LM, Socci ND, Studer L. Derivation of multipotent mesenchymal precursors from human embryonic stem cells. PLoS Med. 2005;2(6):e161. doi: 10.1371/journal.pmed.0020161. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Beattie GM, Lopez AD, Bucay N, Hinton A, Firpo MT, King CC, et al. Activin A maintains pluripotency of human embryonic stem cells in the absence of feeder layers. Stem Cells. 2005;23(4):489–495. doi: 10.1634/stemcells.2004-0279. [DOI] [PubMed] [Google Scholar]
- Bjugstad KB, Teng YD, Redmond DE, Jr, Elsworth JD, Roth RH, Cornelius SK, et al. Human neural stem cells migrate along the nigrostriatal pathway in a primate model of Parkinson’s disease. Exp Neurol. 2008;211(2):362–369. doi: 10.1016/j.expneurol.2008.01.025. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bongso A, Fong CY, Ng SC, Ratnam SS. Isolation and culture of inner cell mass cells from human blastocysts. Hum Reprod. 1994;9(11):2110–2117. doi: 10.1093/oxfordjournals.humrep.a138401. [DOI] [PubMed] [Google Scholar]
- Bosma GC, Custer RP, Bosma MJ. A severe combined immunodeficiency mutation in the mouse. Nature. 1983;301(5900):527–530. doi: 10.1038/301527a0. [DOI] [PubMed] [Google Scholar]
- Brandenberger R, Khrebtukova I, Thies RS, Miura T, Jingli C, Puri R, et al. MPSS profiling of human embryonic stem cells. BMC Dev Biol. 2004;4:10. doi: 10.1186/1471-213X-4-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Brimble SN, Zeng X, Weiler DA, Luo Y, Liu Y, Lyons IG, et al. Karyotypic stability, genotyping, differentiation, feeder-free maintenance, and gene expression sampling in three human embryonic stem cell lines derived prior to August 9, 2001. Stem Cells Dev. 2004;13(6):585–597. doi: 10.1089/scd.2004.13.585. [DOI] [PubMed] [Google Scholar]
- Brolén GK, Heins N, Edsbagge J, Semb H. Signals from the embryonic mouse pancreas induce differentiation of human embryonic stem cells into insulin-producing beta-cell-like cells. Diabetes. 2005;54(1):2867–2874. doi: 10.2337/diabetes.54.10.2867. [DOI] [PubMed] [Google Scholar]
- Buytaert-Hoefen KA, Alvarez E, Freed CR. Generation of tyrosine hydroxylase positive neurons from human embryonic stem cells after coculture with cellular substrates and exposure to GDNF. Stem Cells. 2004;22(5):669–674. doi: 10.1634/stemcells.22-5-669. [DOI] [PubMed] [Google Scholar]
- Cai J, Chen J, Liu Y, Miura T, Luo Y, Loring JF, et al. Assessing self-renewal and differentiation in human embryonic stem cell lines. Stem Cells. 2006;24(3):516–530. doi: 10.1634/stemcells.2005-0143. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Cai J, Zhao Y, Liu Y, Ye F, Song Z, Qin H, et al. Directed differentiation of human embryonic stem cells into functional hepatic cells. Hepatology. 2007;45(5):1229–1239. doi: 10.1002/hep.21582. [DOI] [PubMed] [Google Scholar]
- Carpenter MK, Rosler E, Rao MS. Characterization and differentiation of human embryonic stem cells. Cloning Stem Cells. 2003;5(1):79–88. doi: 10.1089/153623003321512193. [DOI] [PubMed] [Google Scholar]
- Carpenter MK, Rosler ES, Fisk GJ, Brandenberger R, Ares X, Miura T, et al. Properties of four human embryonic stem cell lines maintained in a feeder-free culture system. Dev Dyn. 2004;229(2):243–258. doi: 10.1002/dvdy.10431. [DOI] [PubMed] [Google Scholar]
- Chadwick K, Wang L, Li L, Menendez P, Murdoch B, Rouleau A, et al. Cytokines and BMP-4 promote hematopoietic differentiation of human embryonic stem cells. Blood. 2003;102(3):906–915. doi: 10.1182/blood-2003-03-0832. [DOI] [PubMed] [Google Scholar]
- Chambers I, Colby D, Robertson M, Nichols J, Lee S, Tweedie S, et al. Functional expression cloning of Nanog, a pluripotency sustaining factor in embryonic stem cells. Cell. 2003;113(5):643–655. doi: 10.1016/s0092-8674(03)00392-1. [DOI] [PubMed] [Google Scholar]
- Cheng L, Hammond H, Ye Z, Zhan X, Dravid G. Human adult marrow cells support prolonged expansion of human embryonic stem cells in culture. Stem Cells. 2003;21(2):131–142. doi: 10.1634/stemcells.21-2-131. [DOI] [PubMed] [Google Scholar]
- Chiba S, Lee YM, Zhou W, Curt R, Freed CR. Noggin enhances dopamine neuron production from human embryonic stem cells and improves behavioral outcome after transplantation into parkinsonian rats. Stem Cells. 2008;26(11):2810–2820. doi: 10.1634/stemcells.2008-0085. [DOI] [PubMed] [Google Scholar]
- Chin AC, Fong WJ, Goh LT, Philp R, Oh SK, Choo AB. Identification of proteins from feeder conditioned medium that support human embryonic stem cells. J Biotechnol. 2007;130(3):320–328. doi: 10.1016/j.jbiotec.2007.04.013. [DOI] [PubMed] [Google Scholar]
- Cho MS, Lee YE, Kim JY, Chung S, Cho YH, Kim DS, et al. Highly efficient and large-scale generation of functional dopamine neurons from human embryonic stem cells. Proc Natl Acad Sci U S A. 2008;105(9):3392–3397. doi: 10.1073/pnas.0712359105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chung Y, Klimanskaya I, Becker S, Li T, Maserati M, Lu SJ, et al. Human embryonic stem cell lines generated without embryo destruction. Cell Stem Cell. 2008;2(2):113–117. doi: 10.1016/j.stem.2007.12.013. [DOI] [PubMed] [Google Scholar]
- Cloutier F, Siegenthaler MM, Nistor G, Keirstead HS. Transplantation of human embryonic stem cell-derived oligodendrocyte progenitors into rat spinal cord injuries does not cause harm. Regen Med. 2006;1(4):469–479. doi: 10.2217/17460751.1.4.469. [DOI] [PubMed] [Google Scholar]
- Coffey PJ, Girman S, Wang SM, Hetherington L, Keegan DJ, Adamson P, et al. Long-term preservation of cortically dependent visual function in RCS rats by transplantation. Nat Neurosci. 2002;5(1):53–56. doi: 10.1038/nn782. [DOI] [PubMed] [Google Scholar]
- Cowan CA, Klimanskaya I, McMahon J, Atienza J, Witmyer J, Zucker JP, et al. Derivation of embryonic stem-cell lines from human blastocysts. N Engl J Med. 2004;350(13):1353–1356. doi: 10.1056/NEJMsr040330. [DOI] [PubMed] [Google Scholar]
- Crook JM, Peura TT, Kravets L, Bosman AG, Buzzard JJ, Horne R, et al. The generation of six clinical-grade human embryonic stem cell lines. Cell Stem Cell. 2007;1(5):490–494. doi: 10.1016/j.stem.2007.10.004. [DOI] [PubMed] [Google Scholar]
- Dessaud E, Yang LL, Hill K, Cox B, Ulloa F, Ribeiro A, et al. Interpretation of the sonic hedgehog morphogen gradient by a temporal adaptation mechanism. Nature. 2007;450(7170):717–720. doi: 10.1038/nature06347. [DOI] [PubMed] [Google Scholar]
- Draper JS, Pigott C, Thomson JA, Andrews PW. Surface antigens of human embryonic stem cells: changes upon differentiation in culture. J Anat. 2002;200(Part 3):249–258. doi: 10.1046/j.1469-7580.2002.00030.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Draper JS, Smith K, Gokhale P, Moore HD, Maltby E, Johnson J, et al. Recurrent gain of chromosomes 17q and 12 in cultured human embryonic stem cells. Nat Biotechnol. 2004;22(1):53–54. doi: 10.1038/nbt922. [DOI] [PubMed] [Google Scholar]
- Edlund H. Pancreas: How to get there from the gut? Curr Opin Cell Biol. 1999;11(6):663–668. doi: 10.1016/s0955-0674(99)00033-2. [DOI] [PubMed] [Google Scholar]
- Elkabetz Y, Panagiotakos G, Al Shamy G, Socci ND, Tabar V, Studer L. Human ES cell-derived neural rosettes reveal a functionally distinct early neural stem cell stage. Genes Dev. 2008;22(2):152–165. doi: 10.1101/gad.1616208. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ellerström C, Strehl R, Moya K, Andersson K, Bergh C, Lundin K, et al. Derivation of a xeno-free human embryonic stem cell line. Stem Cells. 2006;24(1):2170–2176. doi: 10.1634/stemcells.2006-0130. [DOI] [PubMed] [Google Scholar]
- Ellerström C, Strehl R, Noaksson K, Hyllner J, Semb H. Facilitated expansion of human embryonic stem cells by single-cell enzymatic dissociation. Stem Cells. 2007;25(7):1690–1696. doi: 10.1634/stemcells.2006-0607. [DOI] [PubMed] [Google Scholar]
- Erceg S, Laínez S, Ronaghi M, Stojkovic P, Pérez-Aragó MA, Moreno-Manzano V, et al. Differentiation of human embryonic stem cells to regional specific neural precursors in chemically defined medium conditions. PLoS ONE. 2008;3(5):e2122. doi: 10.1371/journal.pone.0002122. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Evans MJ, Kaufman M. Establishment in culture of pluripotential stem cells from mouse embryos. Nature. 1981;292(5819):151–156. doi: 10.1038/292154a0. [DOI] [PubMed] [Google Scholar]
- Genbacev O, Krtolica A, Zdravkovic T, Brunette E, Powell S, Nath A, et al. Serum-free derivation of human embryonic stem cell lines on human placental fibroblast feeders. Fertil Steril. 2005;83(5):1517–1529. doi: 10.1016/j.fertnstert.2005.01.086. [DOI] [PubMed] [Google Scholar]
- Gerecht S, Townsend SA, Pressler H, Zhu H, Nijst CL, Bruggeman JP, et al. A porous photocurable elastomer for cell encapsulation and culture. Biomaterials. 2007;28(32):4826–4835. doi: 10.1016/j.biomaterials.2007.07.039. [DOI] [PubMed] [Google Scholar]
- Gerecht S, Burdick JA, Ferreira LS, Townsend SA, Langer R, Vunjak-Novakovic G. Hyaluronic acid hydrogel for controlled self-renewal and differentiation of human embryonic stem cells. Proc Natl Acad Sci U S A. 2007;104(27):11298–11303. doi: 10.1073/pnas.0703723104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gerrard L, Rodgers L, Cui W. Differentiation of human embryonic stem cells to neural lineages in adherent culture by blocking bone morphogenetic protein signaling. Stem Cells. 2005;23(9):1234–1241. doi: 10.1634/stemcells.2005-0110. [DOI] [PubMed] [Google Scholar]
- Gilbert SF. Developmental Biology. 8. Sunderland, MA: Sinauer Associates, Inc. Publishers; 2006. [Google Scholar]
- Gong J, Sagiv O, Cai H, Tsang SH, Del Priore LV. Effects of extracellular matrix and neighboring cells on induction of human embryonic stem cells into retinal or retinal pigment epithelial progenitors. Exp Eye Res. 2008;86(6):957–965. doi: 10.1016/j.exer.2008.03.014. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hayashi H, Morizane A, Koyanagi M, Ono Y, Sasai Y, Hashimoto N, et al. Meningeal cells induce dopaminergic neurons from embryonic stem cells. Eur J Neurosci. 2008;27(2):261–268. doi: 10.1111/j.1460-9568.2008.06027.x. [DOI] [PubMed] [Google Scholar]
- Hayflick L, Moorhead PS. The serial cultivation of human diploid cell strains. Exp Cell Res. 1961;25:585–621. doi: 10.1016/0014-4827(61)90192-6. [DOI] [PubMed] [Google Scholar]
- Heins N, Englund MC, Sjöblom C, Dahl U, Tonning A, Bergh C, Lindahl, et al. Derivation, characterization, and differentiation of human embryonic stem cells. Stem Cells. 2004;22(3):367–376. doi: 10.1634/stemcells.22-3-367. [DOI] [PubMed] [Google Scholar]
- Hoffman LM, Carpenter MK. Characterization and culture of human embryonic stem cells. Nat Biotechnol. 2005;23:699–708. doi: 10.1038/nbt1102. [DOI] [PubMed] [Google Scholar]
- James D, Levine AJ, Besser D, Hemmati-Brivanlou A. TGFbeta/activin/nodal signaling is necessary for the maintenance of pluripotency in human embryonic stem cells. Development. 2005;132(6):1273–1282. doi: 10.1242/dev.01706. [DOI] [PubMed] [Google Scholar]
- Joannides A, Fiore-Hériché C, Westmore K, Caldwell M, Compston A, Allen N, et al. Automated mechanical passaging: a novel and efficient method for human embryonic stem cell expansion. Stem Cells. 2006;24(2):230–235. doi: 10.1634/stemcells.2005-0243. [DOI] [PubMed] [Google Scholar]
- Josephson R, Sykes G, Liu Y, Ording C, Xu W, Zeng X, et al. A molecular scheme for improved characterization of human embryonic stem cell lines. BMC Biol. 2006;4:28. doi: 10.1186/1741-7007-4-28. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kaufman DS, Hanson ET, Lewis RL, Auerbach R, Thomson JA. Hematopoietic colony-forming cells derived from human embryonic stem cells. Proc Natl Acad Sci U S A. 2001;98(19):10716–10721. doi: 10.1073/pnas.191362598. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kawasaki H, Mizuseki K, Nishikawa S, Kaneko S, Kuwana Y, Nakanishi S, et al. Induction of midbrain dopaminergic neurons from ES cells by stromal cell-derived inducing activity. Neuron. 2000;28(1):31–40. doi: 10.1016/s0896-6273(00)00083-0. [DOI] [PubMed] [Google Scholar]
- Kehat I, Kenyagin-Karsenti D, Snir M, Segev H, Amit M, Gepstein A, et al. Human embryonic stem cells can differentiate into myocytes with structural and functional properties of cardiomyocytes. J Clin Invest. 2001;108(3):407–414. doi: 10.1172/JCI12131. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Keirstead HS, Nistor G, Bernal G, Totoiu M, Cloutier F, Sharp K, et al. Human embryonic stem cell-derived oligodendrocyte progenitor cell transplants remyelinate and restore locomotion after spinal cord injury. J Neurosci. 2005;25(19):4694–4705. doi: 10.1523/JNEUROSCI.0311-05.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Klimanskaya I, Chung Y, Becker S, Lu SJ, Lanza R. Human embryonic stem cell lines derived from single blastomeres. Nature. 2006;444(7118):481–485. doi: 10.1038/nature05142. [DOI] [PubMed] [Google Scholar]
- Klimanskaya I, Chung Y, Becker S, Lu SJ, Lanza R. Derivation of human embryonic stem cells from single blastomeres. Nat Protoc. 2007;2(8):1963–1972. doi: 10.1038/nprot.2007.274. [DOI] [PubMed] [Google Scholar]
- Klimanskaya I, Hipp J, Rezai KA, West M, Atala A, Lanza R. Derivation and comparative assessment of retinal pigment epithelium from human embryonic stem cells using transcriptomics. Cloning Stem Cells. 2004;6(3):217–245. doi: 10.1089/clo.2004.6.217. [DOI] [PubMed] [Google Scholar]
- Kornack DR, Rakic P. The generation, migration, and differentiation of olfactory neurons in the adult primate brain. Proc Natl Acad Sci U S A. 2001;98(8):4752–4757. doi: 10.1073/pnas.081074998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laflamme MA, Chen KY, Naumova AV, Muskheli V, Fugate JA, Dupras SK, et al. Cardiomyocytes derived from human embryonic stem cells in pro-survival factors enhance function of infarcted rat hearts. Nat Biotechnol. 2007;25(9):1015–1024. doi: 10.1038/nbt1327. [DOI] [PubMed] [Google Scholar]
- Lavon N, Benvenisty N. Study of hepatocyte differentiation using embryonic stem cells. J Cell Biochem. 2005;96(6):1193–1202. doi: 10.1002/jcb.20590. [DOI] [PubMed] [Google Scholar]
- Lee H, Shamy GA, Elkabetz Y, Schofield CM, Harrsion NL, Panagiotakos G, et al. Directed differentiation and transplantation of human embryonic stem cell-derived motoneurons. Stem Cells. 2007;25(8):1931–1939. doi: 10.1634/stemcells.2007-0097. [DOI] [PubMed] [Google Scholar]
- Lee GY, Kenny PA, Lee EH, Bissell MJ. Three-dimensional culture models of normal and malignant breast epithelial cells. Nat Methods. 2007;4(4):359–365. doi: 10.1038/nmeth1015. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lee SH, Lumelsky N, Studer L, Auerbach JM, McKay RD. Efficient generation of midbrain and hindbrain neurons from mouse embryonic stem cells. Nat Biotechnol. 2000;18(6):675–679. doi: 10.1038/76536. [DOI] [PubMed] [Google Scholar]
- Levenberg S, Golub JS, Amit M, Itskovitz-Eldor J, Langer R. Endothelial cells derived from human embryonic stem cells. Proc Natl Acad Sci U S A. 2002;99(7):4391–4396. doi: 10.1073/pnas.032074999. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Li XJ, Du ZW, Zarnowska ED, Pankratz M, Hansen LO, Pearce RA, et al. Specification of motoneurons from human embryonic stem cells. Nat Biotechnol. 2005;23(2):215–221. doi: 10.1038/nbt1063. [DOI] [PubMed] [Google Scholar]
- Li XJ, Hu BY, Jones SA, Zhang YS, Lavaute T, Du ZW, et al. Directed differentiation of ventral spinal progenitors and motor neurons from human embryonic stem cells by small molecules. Stem Cells. 2008;26(4):886–893. doi: 10.1634/stemcells.2007-0620. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Lim JW, Bodnar A. Proteome analysis of conditioned medium from mouse embryonic fibroblast feeder layers which support the growth of human embryonic stem cells. Proteomics. 2002;2(9):1187–1203. doi: 10.1002/1615-9861(200209)2:9<1187::AID-PROT1187>3.0.CO;2-T. [DOI] [PubMed] [Google Scholar]
- Lu J, Hou R, Booth CJ, Yang SH, Snyder M. Defined culture conditions of human embryonic stem cells. Proc Natl Acad Sci U S A. 2006;103(15):5688–5693. doi: 10.1073/pnas.0601383103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Machon O, Backman M, Krauss S, Kozmik Z. The cellular fate of cortical progenitors is not maintained in neurosphere cultures. Mol Cell Neurosci. 2005;30(3):388–397. doi: 10.1016/j.mcn.2005.08.003. [DOI] [PubMed] [Google Scholar]
- Maitra A, Arking DE, Shivapurkar N, Ikeda M, Stastny V, Kassauei K, et al. Genomic alterations in cultured human embryonic stem cells. Nat Genet. 2005;37(10):1099–1103. doi: 10.1038/ng1631. [DOI] [PubMed] [Google Scholar]
- Martin GR. Teratocarcinomas and mammalian embryogenesis. Science. 1980;209(4458):768–776. doi: 10.1126/science.6250214. [DOI] [PubMed] [Google Scholar]
- Martin GR. Isolation of a pluripotent cell line from early mouse embryos cultured in medium conditioned by teratocarcinoma stem cells. Proc Natl Acad Sci U S A. 1981;78(12):7634–7638. doi: 10.1073/pnas.78.12.7634. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Metallo CM, Ji L, de Pablo JJ, Palecek SP. Retinoic acid and bone morphogenetic protein signaling synergize to efficiently direct epithelial differentiation of human embryonic stem cells. Stem Cells. 2007;26(2):372–380. doi: 10.1634/stemcells.2007-0501. [DOI] [PubMed] [Google Scholar]
- Mitalipova MM, Rao RR, Hoyer DM, Johnson JA, Meisner LF, Jones KL, et al. Preserving the genetic integrity of human embryonic stem cells. Nat Biotechnol. 2005;23(1):19–20. doi: 10.1038/nbt0105-19. [DOI] [PubMed] [Google Scholar]
- Miyamoto K, Hayashi K, Suzuki T, Ichihara S, Yamada T, Kano Y, et al. Human placenta feeder layers support undifferentiated growth of primate embryonic stem cells. Stem Cells. 2004;22(4):433–440. doi: 10.1634/stemcells.22-4-433. [DOI] [PubMed] [Google Scholar]
- Nakayama T Momoki-Soga, Inoue N. Astrocyte-derived factors instruct differentiation of embryonic stem cells into neurons. Neurosci Res. 2003;46(2):241–249. doi: 10.1016/s0168-0102(03)00063-4. [DOI] [PubMed] [Google Scholar]
- Nichols J, Zevnik B, Anastassiadis K, Niwa H, Klewe-Nebenius D, Chambers I, et al. Formation of pluripotent stem cells in the mammalian embryo depends on the POU transcription factor Oct4. Cell. 1998;95(3):379–391. doi: 10.1016/s0092-8674(00)81769-9. [DOI] [PubMed] [Google Scholar]
- Nur-E-Kamal A, Ahmed I, Kamal J, Schindler M, Meiners S. Three-dimensional nanofibrillar surfaces promote self-renewal in mouse embryonic stem cells. Stem Cells. 2006;24(2):426–433. doi: 10.1634/stemcells.2005-0170. [DOI] [PubMed] [Google Scholar]
- Okabe S, Forsberg-Nilsson K, Spiro AC, Segal M, McKay RD. Development of neuronal precursor cells and functional postmitotic neurons from embryonic stem cells in vitro. Mech Dev. 1996;59(1):89–102. doi: 10.1016/0925-4773(96)00572-2. [DOI] [PubMed] [Google Scholar]
- Ourednik J, Ourednik V, Lynch WP, Schachner M, Snyder EY. Neural stem cells display an inherent mechanism for rescuing dysfunctional neurons. Nat Biotechnol. 2002;20(11):1103–1110. doi: 10.1038/nbt750. [DOI] [PubMed] [Google Scholar]
- Parmar M, Li M. Early specification of dopaminergic phenotype during ES cell differentiation. BMC Dev Biol. 2007;7:86. doi: 10.1186/1471-213X-7-86. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pera MF, Andrade J, Houssami S, Reubinoff B, Trounson A, Stanley EG, et al. Regulation of human embryonic stem cell differentiation by BMP-2 and its antagonist noggin. J Cell Sci. 2004;117(Pt 7):1269–1280. doi: 10.1242/jcs.00970. [DOI] [PubMed] [Google Scholar]
- Perrier AL, Tabar V, Barberi T, Rubio ME, Bruses J, Topf N, et al. Derivation of midbrain dopamine neurons from human embryonic stem cells. Proc Natl Acad Sci U S A. 2004;101(34):12543–12548. doi: 10.1073/pnas.0404700101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Postovit LM, Seftor EA, Seftor RE, Hendrix MJ. A three-dimensional model to study the epigenetic effects induced by the microenvironment of human embryonic stem cells. Stem Cells. 2006;24(3):501–505. doi: 10.1634/stemcells.2005-0459. [DOI] [PubMed] [Google Scholar]
- Rajala K, Hakala H, Panula S, Aivio S, Pihlajamäki H, Suuronen R, et al. Testing of nine different xeno-free culture media for human embryonic stem cell cultures. Hum Reprod. 2007;22(5):1231–1238. doi: 10.1093/humrep/del523. [DOI] [PubMed] [Google Scholar]
- Reubinoff BE, Itsykson P, Turetsky T, Pera MF, Reinhartz E, Itzik A, et al. Neural progenitors from human embryonic stem cells. Nat Biotechnol. 2001;19(12):1134–1140. doi: 10.1038/nbt1201-1134. [DOI] [PubMed] [Google Scholar]
- Reubinoff BE, Pera MF, Fong CY, Trounson A, Bongso A. Embryonic stem cell lines from human blastocysts: somatic differentiation in vitro. Nat Biotechnol. 2000;18(4):399–404. doi: 10.1038/74447. [DOI] [PubMed] [Google Scholar]
- Richards M, Fong CY, Chan WK, Wong PC, Bongso A. Human feeders support prolonged undifferentiated growth of human inner cell masses and embryonic stem cells. Nat Biotechnol. 2002;20(9):933–936. doi: 10.1038/nbt726. [DOI] [PubMed] [Google Scholar]
- Rosler ES, Fisk GJ, Ares X, Irving J, Miura T, Rao MS, et al. Long-term culture of human embryonic stem cells in feeder-free conditions. Dev Dyn. 2004;229(2):259–274. doi: 10.1002/dvdy.10430. [DOI] [PubMed] [Google Scholar]
- Roy NS, Cleren C, Singh SK, Yang L, Beal MF, Goldman SA. Functional engraftment of human ES cell-derived dopaminergic neurons enriched by coculture with telomerase-immortalized midbrain astrocytes. Nat Med. 2006;12(11):1259–1268. doi: 10.1038/nm1495. [DOI] [PubMed] [Google Scholar]
- Samadikuchaksaraei A, Cohen S, Isaac K, Rippon HJ, Polak JM, Bielby RC, et al. Derivation of distal airway epithelium from human embryonic stem cells. Tissue Eng. 2006;12(4):867–875. doi: 10.1089/ten.2006.12.867. [DOI] [PubMed] [Google Scholar]
- Santa-Olalla J, Baizabal JM, Fregoso M, del Carmen Cardenas M, Covarrubias L. The in vivo positional identity gene expression code is not preserved in neural stem cells grown in culture. Eur J Neurosci. 2003;18(5):1073–1084. doi: 10.1046/j.1460-9568.2003.02824.x. [DOI] [PubMed] [Google Scholar]
- Segev H, Fishman B, Ziskind A, Shulman M, Itskovitz-Eldor J. Differentiation of human embryonic stem cells into insulin-producing clusters. Stem Cells. 2004;22(3):265–274. doi: 10.1634/stemcells.22-3-265. [DOI] [PubMed] [Google Scholar]
- Sherman MI. Long term culture of cells derived from mouse blastocysts. Differentiation. 1975;3(1–3):51–67. doi: 10.1111/j.1432-0436.1975.tb00845.x. [DOI] [PubMed] [Google Scholar]
- Sottile V, Thomson A, McWhir J. In vitro osteogenic differentiation of human ES cells. Cloning Stem Cells. 2003;5(2):149–155. doi: 10.1089/153623003322234759. [DOI] [PubMed] [Google Scholar]
- Stojkovic P, Lako M, Stewart R, Przyborski S, Armstrong L, Evans J, et al. An autogeneic feeder cell system that efficiently supports growth of undifferentiated human embryonic stem cells. Stem Cells. 2005;23(3):306–314. doi: 10.1634/stemcells.2004-0137. [DOI] [PubMed] [Google Scholar]
- Strelchenko N, Verlinsky O, Kukharenko V, Verlinsky Y. Morula-derived human embryonic stem cells. Reprod Biomed Online. 2004;9(6):623–629. doi: 10.1016/s1472-6483(10)61772-5. [DOI] [PubMed] [Google Scholar]
- Strom S, Inzunza J, Grinnemo KH, Holmberg K, Matilainen E, Stromberg AM, et al. Mechanical isolation of the inner cell mass is effective in derivation of new human embryonic stem cell lines. Hum Reprod. 2007;22(12):3051–3058. doi: 10.1093/humrep/dem335. [DOI] [PubMed] [Google Scholar]
- Suemori H, Tada T, Torii R, Hosoi Y, Kobayashi K, Imahie H, et al. Establishment of embryonic stem cell lines from cynomolgus monkey blastocysts produced by IVF or ICSI. Dev Dyn. 2001;222(2):273–279. doi: 10.1002/dvdy.1191. [DOI] [PubMed] [Google Scholar]
- Suemori H, Yasuchika K, Hasegawa K, Fujioka T, Tsuneyoshi N, Nakatsuji N. Efficient establishment of human embryonic stem cell lines and long-term maintenance with stable karyotype by enzymatic bulk passage. Biochem Biophys Res Commun. 2006;345(3):926–932. doi: 10.1016/j.bbrc.2006.04.135. [DOI] [PubMed] [Google Scholar]
- Thomson JA, Itskovitz-Eldor J, Shapiro SS, Waknitz MA, Swiergiel JJ, Marshall VS, et al. Embryonic stem cell lines derived from human blastocysts. Science. 1998;282(5391):1145–1147. doi: 10.1126/science.282.5391.1145. [DOI] [PubMed] [Google Scholar]
- Thomson JA, Kalishman J, Golos TG, Durning M, Harris CP, Becker RA, et al. Isolation of a primate embryonic stem cell line. Proc Natl Acad Sci U S A. 1995;92(17):7844–7848. doi: 10.1073/pnas.92.17.7844. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Thomson JA, Kalishman J, Golos TG, Durning M, Harris CP, Hearn JP. Pluripotent cell lines derived from common marmoset (Callithrix jacchus) blastocysts. Biol Reprod. 1996;55(2):254–259. doi: 10.1095/biolreprod55.2.254. [DOI] [PubMed] [Google Scholar]
- Turetsky T, Aizenman E, Gil Y, Weinberg N, Shufaro Y, Revel A, et al. Laser-assisted derivation of human embryonic stem cell lines from IVF embryos after preimplantation genetic diagnosis. Hum Reprod. 2008;23(1):46–53. doi: 10.1093/humrep/dem351. [DOI] [PubMed] [Google Scholar]
- Van Hoof D, Braam SR, Dormeyers W, Wardvan Oostwaard D, Heck AJ, Krijgsveld J, et al. Feeder-free monolayer cultures of human embryonic stem cells express an epithelial plasma membrane protein profile. Stem Cells. 2008;26(11):2777–2781. doi: 10.1634/stemcells.2008-0365. [DOI] [PubMed] [Google Scholar]
- Van Vranken BE, Romanska HM, Polak JM, Rippon HJ, Shannon JM, Bishop AE. Coculture of embryonic stem cells with pulmonary mesenchyme: a microenvironment that promotes differentiation of pulmonary epithelium. Tissue Eng. 2005;11(7–8):1177–1187. doi: 10.1089/ten.2005.11.1177. [DOI] [PubMed] [Google Scholar]
- Vazin T, Becker KG, Chen J, Spivak CE, Lupica CR, Zhang Y, et al. A novel combination of factors, termed SPIE, which promotes dopaminergic neuron differentiation from human embryonic stem cells. PLoS ONE. 2009;4(8):e6606. doi: 10.1371/journal.pone.0006606. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vazin T, Chen J, Lee CT, Amable R, Freed WJ. Assessment of stromal-derived inducing activity in the generation of dopaminergic neurons from human embryonic stem cells. Stem Cells. 2008;26(6):1517–1525. doi: 10.1634/stemcells.2008-0039. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vodyanik MA, Bork JA, Thomson JA, Slukvin II. Human embryonic stem cell-derived CD34+ cells: efficient production in the coculture with OP9 stromal cells and analysis of lymphohematopoietic potential. Blood. 2005;105(2):617–626. doi: 10.1182/blood-2004-04-1649. [DOI] [PubMed] [Google Scholar]
- Wang D, Haviland DL, Burns AR, Zsigmond E, Wetsel RA. A pure population of lung alveolar epithelial type II cells derived from human embryonic stem cells. Proc Natl Acad Sci U S A. 2007;104(11):4449–4454. doi: 10.1073/pnas.0700052104. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang G, Zhang H, Zhao Y, Li J, Cai J, Wang P, et al. Noggin and bFGF cooperate to maintain the pluripotency of human embryonic stem cells in the absence of feeder layers. Biochem Biophys Res Commun. 2005;330(3):934–942. doi: 10.1016/j.bbrc.2005.03.058. [DOI] [PubMed] [Google Scholar]
- Wang Q, Fang ZF, Jin F, Lu Y, Gai H, Sheng HZ. Derivation and growing human embryonic stem cells on feeders derived from themselves. Stem Cells. 2005;23(9):1221–1227. doi: 10.1634/stemcells.2004-0347. [DOI] [PubMed] [Google Scholar]
- Xu C, Inokuma MS, Denham J, Golds K, Kundu P, Gold JD, et al. Feeder-free growth of undifferentiated human embryonic stem cells. Nat Biotechnol. 2001;19(10):971–974. doi: 10.1038/nbt1001-971. [DOI] [PubMed] [Google Scholar]
- Xu RH, Peck RM, Li DS, Feng X, Ludwig T, Thomson JA. Basic FGF and suppression of BMP signaling sustain undifferentiated proliferation of human ES cells. Nat Methods. 2005;2(3):185–190. doi: 10.1038/nmeth744. [DOI] [PubMed] [Google Scholar]
- Yan Y, Yang D, Zarnowska ED, Du Z, Werbel B, Valliere C, et al. Directed differentiation of dopaminergic neuronal subtypes from human embryonic stem cells. Stem Cells. 2005;23(6):781–790. doi: 10.1634/stemcells.2004-0365. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yim EK, Leong KW. Proliferation and differentiation of human embryonic germ cell derivatives in bioactive polymeric fibrous scaffold. J Biomater Sci Polym Ed. 2005;16(10):1193–1217. doi: 10.1163/156856205774269485. [DOI] [PubMed] [Google Scholar]
- Yue F, Cui L, Johkura K, Ogiwara N, Sasaki K. Induction of midbrain dopaminergic neurons from primate embryonic stem cells by coculture with sertoli cells. Stem Cells. 2006;24(7):1695–1706. doi: 10.1634/stemcells.2005-0409. [DOI] [PubMed] [Google Scholar]
- Zeng X, Cai J, Chen J, Luo Y, You ZB, Fotter E, et al. Dopaminergic differentiation of human embryonic stem cells. Stem Cells. 2004;22(6):925–940. doi: 10.1634/stemcells.22-6-925. [DOI] [PubMed] [Google Scholar]
- Zhang SC, Wernig M, Duncan ID, Brüstle O, Thomson JA. In vitro differentiation of transplantable neural precursors from human embryonic stem cells. Nat Biotechnol. 2001;19(12):1129–1133. doi: 10.1038/nbt1201-1129. [DOI] [PubMed] [Google Scholar]