Abstract
Reduced functional bladder capacity and concomitant increased micturition frequency (pollakisuria) are common lower urinary tract symptoms associated with conditions such as cystitis, prostatic hyperplasia, neurological disease, and overactive bladder syndrome. These symptoms can profoundly affect the quality of life of afflicted individuals, but available pharmacological treatments are often unsatisfactory. Recent work has demonstrated that the cation channel TRPV4 is highly expressed in urothelial cells and plays a role in sensing the normal filling state of the bladder. In this article, we show that the development of cystitis-induced bladder dysfunction is strongly impaired in Trpv4−/− mice. Moreover, we describe HC-067047, a previously uncharacterized, potent, and selective TRPV4 antagonist that increases functional bladder capacity and reduces micturition frequency in WT mice and rats with cystitis. HC-067047 did not affect bladder function in Trpv4−/− mice, demonstrating that its in vivo effects are on target. These results indicate that TRPV4 antagonists may provide a promising means of treating bladder dysfunction.
Keywords: transient receptor potential channels, urothelium
The urinary bladder functions to store urine and to expel it at a socially convenient moment (1). The storage function of the bladder is disturbed in several pathological conditions, including cystitis, bladder outlet obstruction (e.g., such as caused by benign prostatic hyperplasia), multiple sclerosis, and spinal cord injury, as well as in patients with overactive bladder syndrome (2, 3). A reduction of the functional bladder capacity induces bothersome lower urinary tracts symptoms such as pollakisuria (frequent micturition), nocturia, urgency, and urge incontinence, which strongly affect the quality of life of afflicted patients and impose high costs on Western healthcare systems (4–6). Antimuscarinic agents, which act, at least in part, by reducing acetylcholine-induced contraction of the detrusor muscle, are widely used in the treatment of bladder storage dysfunction (1, 2). However, their therapeutic efficacy is often limited, and treatment can be associated with adverse effects such as severe dry mouth, constipation, headache, blurred vision, and even cognitive deterioration, which frequently lead to discontinuation of treatment (2, 7–9). Thus, there is need for alternative pharmaco-logical therapy for bladder storage disorders (3, 8).
Recent research has shown that TRPV4, a cation channel of the transient receptor potential (TRP) superfamily (10, 11), is highly expressed in the urothelial cell layer of the bladder, where it is implicated in sensing the filling state of the bladder (12–16). Importantly, Trpv4−/− mice exhibited a lower voiding frequency and larger voided volume than wild type (WT) mice did. In this study, we evaluated TRPV4 as a potential pharmacological target for the treatment of bladder storage dysfunction. We discovered HC-067047, a previously undescribed, potent, and selective TRPV4 antagonist. When applied systemically, HC-067047 was efficacious in reducing micturition frequency and increasing functional bladder capacity in mice and rats with cystitis caused by pretreatment with the chemotherapeutic agent cyclophosphamide. TRPV4-deficient mice treated with cyclophosphamide developed the same degree of cystitis as WT animals did, but they did not develop clear pollakisuria and were insensitive to application of HC-067047. Our results provide a proof of principle for the use of TRPV4 as a target for the treatment of bladder dysfunction.
Results
Development of Cyclophosphamide-Induced Cystitis in WT and Trpv4−/− Mice.
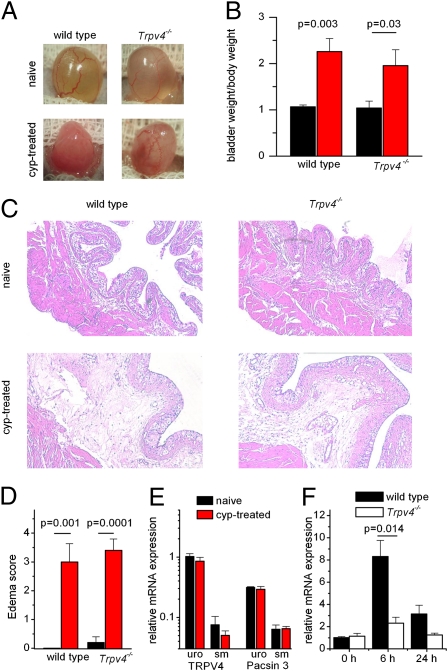
Mice and rats suffering from cyclophosphamide-induced cystitis have been widely used as animal models of bladder dysfunction (17). Upon systemic application, the chemotherapeutic agent cyclophosphamide is partially metabolized to acrolein, which accumulates in the bladder, where it evokes strong hemorrhagic cystitis (18). To investigate the role of TRPV4 in this process, we first compared the severity of cystitis in cyclophosphamide-treated WT and Trpv4−/− mice. Macroscopically, equal signs of bladder inflammation were present in WT and Trpv4−/− mice 24 h after i.p. cyclophosphamide administration (300 mg/kg). Both genotypes displayed macroscopic edema of the bladder wall (Fig. 1A) and inflamed bladders that weighed two times more than bladders from nontreated (naive) animals (Fig. 1B). In bladder sections, cyclophosphamide-treated WT and Trpv4−/− mice revealed clear signs of lamina propria edema (Fig. 1C), and the severity of edema, as quantified by the histological edema score, was similar in both genotypes (Fig. 1D). At the behavioral level, cyclophosphamide treatment evoked clear signs of pain/discomfort in all treated WT and Trpv4−/− mice animals, as evidenced by rounded-back posture and strongly reduced mobility. Furthermore, quantitative PCR on urothelium and bladder smooth muscle of control and cyclophosphamide-pretreated WT mice (Fig. 1E) revealed no changes in mRNA expression of TRPV4 or the associated protein Pacsin 3 (19, 20). Because early stages of bladder inflammation are associated with a transient increase in expression of the immediate early gene c-fos (21), we also analyzed the expression of this gene in WT and Trpv4−/− bladders before as well as 6 and 24 h after treatment with cyclophosphamide. Although we found a transient increase in c-fos expression in both WT and Trpv4−/− mice, the increase was significantly reduced in the latter (Fig. 1F).
Fig. 1.
Cyclophosphamide induces cystitis in WT and Trpv4−/− mice. (A) Images of WT and Trpv4−/− mice pretreated with cyclophosphamide or vehicle (saline). (B) Bladder weight/body weight ratio in vehicle- and cyclophosphamide-pretreated WT (n = 4) and Trpv4−/− (n = 4) mice expressed in mg/g. (C) Histological bladder sections of vehicle-pretreated (naive) or cyclophosphamide-pretreated WT and Trpv4−/− mice. (D) Histological edema score showing the severity of edema in WT (n = 4) and Trpv4−/− (n = 4) mice pretreated with cyclophosphamide but not in vehicle-pretreated mice. Relative expression of TRPV4 and Pacsin3 mRNA in urothelium and smooth muscle from vehicle-pretreated (n = 4) or cyclophosphamide-pretreated (n = 4) mice. (E) Levels of mRNA were normalized to the TRPV4 mRNA level in urothelium from vehicle-pretreated mice. (F) Changes in c-fos mRNA after cyclophosphamide treatment in WT and Trpv4−/− mice.
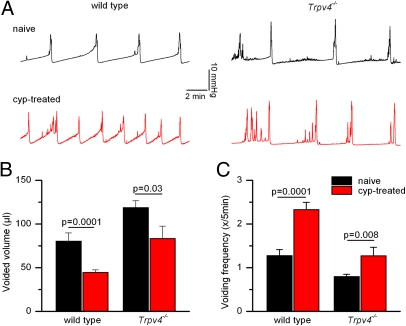
To investigate the impact of TRPV4 on the development of bladder dysfunction associated with cystitis, we performed cystometric recordings in WT and Trpv4−/− mice. In these experiments, intravesical pressure and voided volume were recorded while the bladder was instilled with saline at a constant rate. In this assay, the anesthetized mice have no access to water for 2 h before the actual recording; thus, potential effects of genotype or pharmacological interventions on voluntary fluid intake are not expected influence the urodynamic parameters. In naive WT mice, we measured a regular pattern of pressure build-up and voiding (Fig. 2A), as described previously (15). In comparison, cyclophosphamide-pretreated WT mice exhibited clear signs of pollakisuria, characterized by a doubling of the voiding frequency and an ∼50% reduction of the voided volume (Fig. 2 B and C). In line with previous work (15), naive Trpv4−/− mice exhibited significantly lower voiding frequencies and larger voided volumes than WT animals did (Fig. 2 A–C). Interestingly, we found that the effects of cyclophosphamide treatment on voiding frequency and voided volume were less pronounced in Trpv4−/− mice than in WT mice (Fig. 2 B and C). Notably, the average voiding frequency and voided volume of cyclophosphamide-pretreated Trpv4−/− mice were identical to those of naive WT mice (Fig. 2 B and C). Together, these data indicate that TRPV4 is not directly involved in the development of cyclophosphamide-induced hemorrhagic cystitis but influences the expression of c-fos and the development of pollakisuria and reduced functional bladder capacity during cystitis.
Fig. 2.
The development of cystitis-induced detrusor overactivity is impaired in Trpv4−/− mice. (A) Cystometry patterns in WT and Trpv4−/− mice pretreated with vehicle or cyclophosphamide (cyp). (B and C) Voided volume and voiding frequency in naive WT (n = 12), cyp-treated WT (n = 21), naive Trpv4−/− (n = 11), and cyp-treated Trpv4−/− (n = 6) mice.
Discovery of HC-067047 as a Potent and Selective TRPV4 Antagonist.
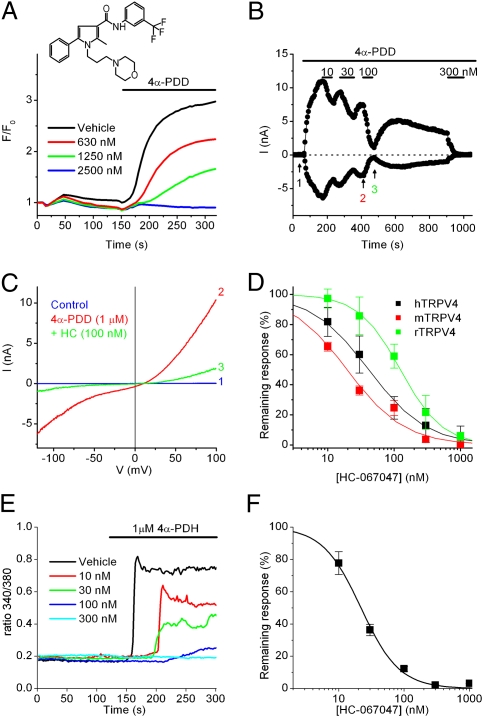
To investigate whether TRPV4 can be used as a target for the pharmacological treatment of bladder dysfunction, we performed a high-throughput screen for small-molecule antagonists of TRPV4 on a cell line expressing recombinant hTRPV4. HC-067047, a compound that reduced 4α-phorbol 12,13-didecanoate (4α-PDD)-induced Ca2+ responses, was chosen for further study (Fig. 3A). To determine whether the observed antagonism was a result of TRPV4 current inhibition, we examined whole-cell currents activated by 4α-PDD in HEK cells expressing hTRPV4 (Fig. 3 B and C). HC-067047 reversibly inhibited currents through the human, rat, and mouse TRPV4 orthologs with IC50 values of 48 ± 6 nM, 133 ± 25 nM, and 17 ± 3 nM, respectively (Fig. 3D). In Ca2+ imaging experiments, 1 μM HC-067047 caused a complete inhibition of mouse TRPV4 activated by various known stimuli, including heat, hyposmotic solution, arachidonic acid, 4α-PDD, 4α-phorbol 12,13-dihexanoate (4α-PDH), and N-((1S)-1-[4-((2S)-2-[(2,4-dichlorophenyl)sulfonyl]amino-3-hydroxypropanoyl)-1-piperazinyl]carbonyl-3-methylbutyl)-1-benzothiophene-2-carboxamide (GSK1016790A) (Table S1). Finally, Ca2+ imaging experiments in cultured mouse urothelial cells revealed that HC-067047 inhibits endogenous TRPV4-mediated response to 4α-PDH with an IC50 of 22 ± 1 nM (Fig. 3 E and F).
Fig. 3.
Characterization of the TRPV4 antagonist HC-067047. (A) Representative Fluo-4 fluorescence traces showing the effect of HC-067047 (Inset; MW = 472) on TRPV4-mediated Ca2+ responses in T-REx-HEK-293 cells expressing hTRPV4 stimulated with 4α-PDD (1 μM). (B) Representative whole-cell patch-clamp recording showing the dose-dependent effect of HC-067047 on TRPV4-mediated currents at +80 (red) and −80 mV (black) in T-REx-HEK-293 cells expressing hTRPV4 stimulated with 4α-PDD (1 μM). (C) Current–voltage relations of TRPV4-expressing cells obtained at the time points indicated in B. (D) Dose–response curves for the inhibition of inward TRPV4 currents at −80 mV for human, mouse and rat TRPV4. (E) Inhibition by HC-067047 of Ca2+ responses to 4α-PDH (1 μM) in Fura-2–loaded mouse urothelial cells. (F) Dose–response curve of the inhibition of TRPV4-mediated Ca2+ responses in mouse urothelial cells.
We explored the selectivity of HC-067047 for a variety of ion-channel targets, including representative channels of the TRPV, TRPC, TRPA, and TRPM families, voltage-gated sodium and potassium channels, and hERG, as well as endogenous TRPM7-like and TRPV2-like channels in isolated mouse urothelial cells (Fig. S1 and Table S1). Apart from TRPV4, the only channels observed to be significantly affected by HC-067047 at submicromolar concentrations were TRPM8 and hERG.
Because of its promising selectivity profile, we endeavored to use HC-067047 to probe the in vivo function of TRPV4. First, we investigated the pharmacokinetic properties of HC-067047 after i.v. or i.p. injection into rats. When administered i.p. at 10 mg/kg, HC-067047 levels remained significantly above the IC50 for rTRPV4 for more than 2 h, suggesting that, at this dose, a sufficient window would be available for the study of TRPV4 in vivo (Table S2 and Fig. S2). Although a longer time window was observed with administration of a higher dose of HC-067047 (100 mg/kg), this dose also produced some obvious side effects, including hunched posture and piloerection, which were not present at the lower dose. Given that TRPV4 is broadly expressed and has been implicated in a variety of (patho)physiological processes, we performed a series of assays to test for possible effects of HC-067047 (i.p., 10 mg/kg), outside the urinary tract. Previous work has demonstrated that TRPV4-deficient mice, when housed solitarily, drink less than WT mice do, leading to hyperosmolarity (22). However, when testing water consumption during the first 6 or 24 h after i.p. injection, we found no significant differences between vehicle- and HC-067047-injected mice (Fig. S3). TRPV4 expression has also been implicated in the detection of warm temperatures and in thermal selection behavior (23). However, when placed on a thermal gradient for 2 h after i.p. injection, we did not find significant differences in thermal preference between vehicle- and HC-067047-injected mice (Fig. S4). In addition, application of HC-067047 in mice did not affect core body temperature (vehicle: 37.3 ± 0.4 °C; HC-067047: 37.6 ± 0.3 °C; n = 3; P = 0.6), heart rate [vehicle: 597 ± 17 beats per minute (bpm); HC-067047: 610 ± 9 bpm; n = 3; P = 0.65], or voluntary locomotion (Fig. S4). Finally, testing motor coordination in rats by using the accelerating rotarod assay, HC-067047 had no effect on the delay to fall (vehicle: 78 ± 12 s; HC-067047: 78 ± 8 s; P = 0.5). Overall, these data indicate that a single 10 mg/kg dose of HC-067047 is well tolerated and does not cause obvious unwanted side effects.
HC-067047 Reduces Pollakisuria and Increases Functional Bladder Capacity.
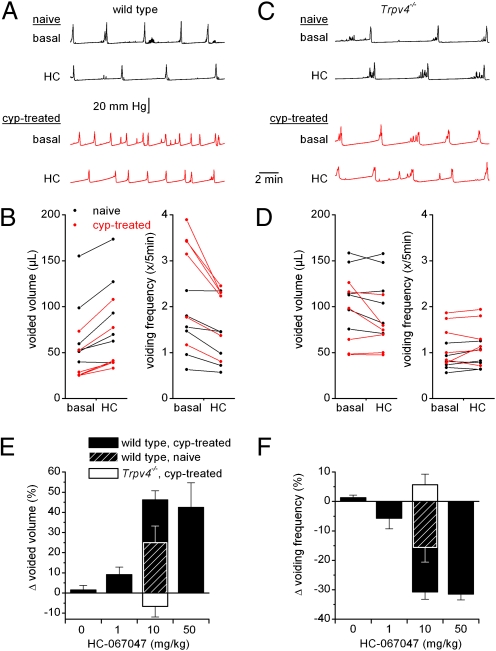
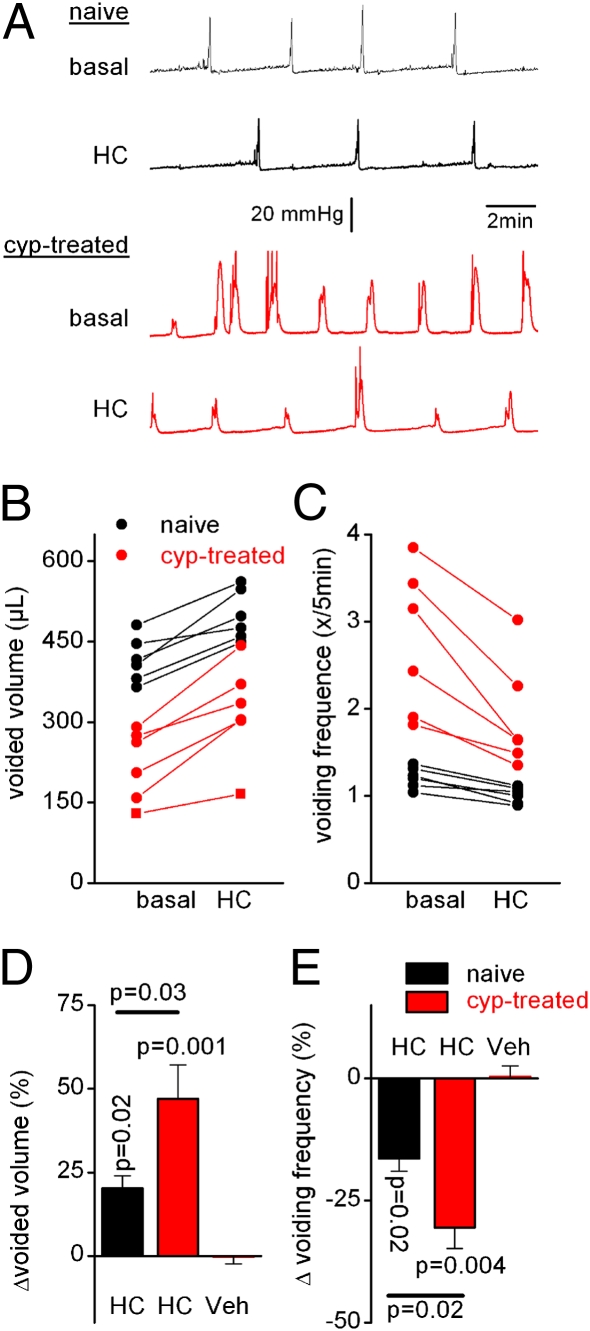
Next, we examined the in vivo effect of HC-067047 on bladder function in healthy mice and animals suffering from cyclophosphamide-induced cystitis. Cystometric parameters were monitored over 30-min recording periods before and after i.p. administration of HC-067047. Administration of vehicle had no significant effect on the various cystometric parameters (Fig. 4 E and F and Table S3). In cyclophosphamide-treated WT mice, application of HC-067047 resulted in a dose-dependent decrease in voiding frequency and an increase in voided volume (Fig. 4 A, B, E, and F and Table S3). At 10 mg/kg, HC-067047 also decreased the voiding frequency and increased the voided volume of naive WT animals, but the relative effect was significantly lower (P = 0.04) than in cyclophosphamide-pretreated mice (Fig. 4 E and F). Analysis of blood samples taken at the end of the cystometry experiments from mice treated with the highest dose of HC-067047 (50 mg/kg) yielded an average plasma concentration of 5.16 ± 0.85 μM (i.e., more than 200-fold higher than the IC50 of mouse TRPV4), suggesting that, at this dose, channel block is maximal. Even at this highest dose, we always observed a regular pattern of pressure buildup and voiding, in contrast with recent results obtained with a specific inhibitor of the related capsaicin receptor TRPV1 (GRC-6211), which reduces bladder voiding frequency at low doses and completely blocks bladder contractions at higher doses (24). Importantly, systemic application of HC-067047 did not have any significant effect on the cystometric parameters of either naive or cyclophosphamide-pretreated Trpv4−/− mice (Fig. 4 C–F and Table S3). Together, our data indicate that systemic application of HC-067047 improves the storage function of the bladder via a specific inhibition of TRPV4.
Fig. 4.
HC-067047 decreases detrusor overactivity in anesthetized WT mice. (A) Cystometry recordings in naive and cyclophosphamide-pretreated WT mice before (basal) and after (HC) i.p. administration of 10 mg/kg HC-067047. (B) Effects of HC-067047 on voided volume and voiding frequency in naive and cyclophosphamide-pretreated WT mice. Each line represents the value for a single animal before and 30 min after the i.p. administration of 10 mg/kg HC-067047. (C and D) Same experiments as in A and B with Trpv4−/− mice. (E and F) Summary of the relative changes in voided volume and voiding frequency after the administration of different doses of HC-067047 in cyclophosphamide-treated WT and Trpv4−/− mice, and in naive WT mice. Additional cystometric values and n numbers are provided in Table S3.
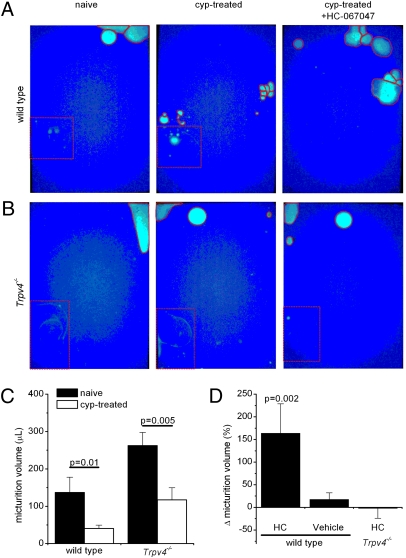
In addition, we studied the effect of HC-067047 on the voiding behavior of freely moving naive and cyclophosphamide-pretreated mice. For this purpose, we determined the size and corresponding volume of individual urine spots on filter paper at the bottom of a metabolic cage (Fig. 5 A and B). We observed a strong decrease in micturition volume (to ∼25% of pretreatment control) in WT mice treated with cyclophosphamide (Fig. 5 A and C). Subsequent administration of HC-067047 (i.p., 10 mg/kg), but not vehicle, caused a more than twofold increase in micturition volume (Fig. 5 A and D). In agreement with previous results (15), we observed that naive Trpv4−/− mice exhibit a micturition volume significantly greater than that of WT mice (Fig. 5 B and C). Cyclophosphamide treatment of the Trpv4−/− animals reduced their micturition volume to a level comparable with that of untreated WT mice (Fig. 5C), an effect that was not significantly affected by treatment with HC-067047 (Fig. 5 B and D). These results demonstrate that HC-067047 increases the functional bladder capacity of awake mice because of selective inhibition of TRPV4. Finally, we investigated the effects of HC-067047 on healthy rats and rats with cyclophosphamide-induced cystitis. Rats offer the advantage that cystometry can be performed in conscious animals, because rats are more tranquil than mice and therefore evoke fewer movement-induced intravesical pressure artifacts. We found that systemic administration of HC-067047 (i.p., 10 mg/kg) in awake rats resulted in an increased voided volume and a decreased voiding frequency and that these effects were significantly more pronounced in rats with pollakisuria caused by cyclophosphamide treatment (Fig. 6 A–E). In addition, we found that HC-067047 caused a significant reduction of the maximal micturition pressure in rats, which might be caused by an effect on TRPV4 in smooth muscle (Table S4). Although a reduction in micturition pressure could potentially cause urinary retention, this effect was never observed.
Fig. 5.
HC-067047 increases micturition volume in freely moving mice suffering from cyclophosphamide-induced cystitis. (A and B) Representative micturition pattern of a single WT (A) or Trpv4−/− (B) mouse before (Left) and after (Center) cyclophosphamide pretreatment and after subsequent i.p. injection with HC-067047 (10 mg/kg; Right). Solid red lines delineate individual urine spots. Dotted red lines delineate an area that was discarded from analysis because of spilling of drinking water. (C) Comparison of the micturition volumes in WT (n = 10) and Trpv4−/− (n = 13) mice before and after cyclophosphamide treatment. (D) Summary of relative changes in micturition volume after the administration of HC-067047 (n = 6 for WT; n = 5 for Trpv4−/−) or vehicle (n = 4).
Fig. 6.
HC-067047 decreases detrusor overactivity in conscious rats. (A) Cystometry recordings in naive and cyclophosphamide-pretreated rats before (basal) and after (HC) i.p. administration of 10 mg/kg HC-067047. (B and C) Effects of HC-067047 on voided volume and voiding frequency in naive and cyclophosphamide-pretreated rats. Each line represents the value for a single animal before and 30 min after the i.p. administration of HC-067047. (D and E) Summary of the relative changes in voided volume and voiding frequency after the administration of HC-067047 or vehicle. Parameter values and n numbers are provided in Table S4.
Discussion
Recent research has identified TRPV4 as an important regulator of normal bladder function (12–16, 25), but its involvement in the development of cystitis and the associated changes in bladder function was unknown. In this article, we show that the development of reduced functional bladder capacity and pollakisuria after cyclophosphamide treatment is strongly impaired in Trpv4−/− mice, despite clear signs of severe hemorrhagic cystitis. The discovery of HC-067047, a potent and selective TRPV4 antagonist, allowed us to investigate the acute effect of TRPV4 inhibition on the voiding behavior of healthy and inflamed bladders. Our results show that HC-067047 causes a significant increase in functional bladder capacity in mice and rats suffering from cystitis, thus providing proof of principle that pharmacological inhibition of TRPV4 can be used to treat bladder dysfunction.
HC-067047 is a previously uncharacterized, potent, and selective TRPV4 antagonist for which in vivo activity has been reported. We found that HC-067047 rapidly and reversibly inhibited heterologously expressed human, mouse, and rat TRPV4 channels as well as endogenous TRPV4 channels in mouse urothelial cells, with IC50 values ranging between 20 and 140 nM. Inhibition of HC-067047 was observed with different TRPV4-activating stimuli (heat, cell swelling, arachidonic acid, synthetic ligands, etc.), which are known to act on the channel via different pathways (26), suggesting that HC-067047–mediated channel antagonism is noncompetitive. HC-067047 exhibits high selectivity toward TRPV4: IC50 values were at least 100-fold higher for the closely related channels TRPV1, TRPV2, and TRPV3, and ∼10-fold higher for TRPM8 and hERG. HC-067047 concentrations up to 3.2 μM did not significantly agonize or antagonize any other channels tested. Our finding that HC-067047 had no effect on the cystometric parameters of Trpv4−/− mice demonstrates that the observed in vivo effects are specific.
Previous work had shown that healthy Trpv4−/− mice have lower voiding frequency and larger bladder capacity than WT controls do (15). Our current work extends these findings by showing that Trpv4−/− mice are much less prone to develop pollakisuria during cystitis. Although the exact mechanism whereby TRPV4 regulates bladder function is not yet fully understood, current evidence suggests that TRPV4 mediates stretch-evoked ATP release from the urothelium (14, 15). Released ATP can activate purinergic receptors on the afferent nerve fibers, which transduce information on the filling state of the bladder to the central nervous system (3, 27). Inhibition of this mechanism represents a straightforward explanation for the reduced micturition frequency and larger functional bladder capacity that we observed upon pharmacological inhibition or genetic deletion of TRPV4. In line herewith, the changes in bladder function induced by HC-067047 were highly similar to those observed with the P2X3 and P2X2/3 receptor antagonist A-317491 (28), which is thought to act by inhibiting ATP-induced activation of afferent fibers. At this point, we cannot exclude that inhibition of TRPV4 in other cell types contributes to the observed effects of HC-067047 on bladder function. In the bladder, TRPV4 is also expressed in the detrusor muscle layer, albeit at an ∼20-fold lower expression level than in urothelium (13), and in endothelial cells surrounding blood vessels. Moreover, TRPV4 expression has been documented in dorsal root ganglion neurons innervating the viscera and has been implicated in tonicity-induced neurogenic inflammation and release of substance P and calcitonin gene-related peptide (CGRP) from bladder tissue (29, 30). Clearly, further research using tissue-specific (genetic) approaches to influence TRPV4 function will be required to establish the exact mode of action of TRPV4 antagonism on bladder function.
TRPV4 is broadly expressed, and its functions in various tissues outside the bladder are incompletely understood (11). Whereas Trpv4−/− mice exhibit normal viability and have no obvious behavioral abnormalities, several studies have reported various milder phenotypes in TRPV4 knockout mice, including deficits in osmotic homeostasis, alterations in mechanosensation and thermosensation, increased bone mass caused by impaired osteoclast maturation and aging-related hearing impairment (22, 23, 31–34). Moreover, agonists of TRPV4 were shown to cause profound circulatory collapse and death in rodents (35). These findings raise the important question of whether pharmacologic inhibition of TRPV4 may have adverse effects, which would preclude further clinical development. Our first results in this respect are encouraging, showing no significant effects of systemic application of HC-067047 on various parameters, including core body temperature, heart rate, locomotion, motor coordination, fluid intake, or thermal selection behavior. It should be noted that the effects of HC-067047 on the urodynamic parameters of mice were smaller than the alterations observed in the Trpv4−/− mice. Moreover, administration of HC-067047 did not reproduce the deficits in thermal selection behavior or water intake that have been reported for Trpv4−/− mice. This finding may indicate that the concentration of HC-067047 in the relevant tissues was insufficient to cause full inhibition of TRPV4, or, alternatively, that compensatory and/or developmental processes contribute to the phenotypes in the Trpv4−/− mice.
Mutations in the human TRPV4 gene have been identified as the direct cause of divergent hereditary diseases, including skeletal dysplasias, spinal muscular atrophy, and Charcot–Marie–Tooth disease type 2C (36–41). Although the exact role of TRPV4 in the etiology of these clinically diverse diseases is unknown, it appears that many disease-causing mutations lead to gain of TRPV4 function (41). Interestingly, one study documented that 9 of 17 patients with Charcot–Marie–Tooth disease type 2C caused by gain of function mutations in TRPV4 complained of bladder urgency and incontinence (40). The availability of potent and selective TRPV4 modulators such as HC-067047 may greatly facilitate research aimed at understanding the pathophysiology of TRPV4-related diseases, which may ultimately lead to the development of pharmaceutical therapies to prevent or impede the development of disease symptoms in patients carrying TRPV4 mutations.
Available evidence indicates that genetic disruption or pharmacological inhibition of TRPV4 increases functional bladder capacity by reducing the urothelium-mediated transduction of intravesical pressure. Thus, in contrast to the widely used antimuscarinics, which mainly target the contractile apparatus of the bladder, TRPV4 antagonists may provide a unique means to treat bladder dysfunction at the level of sensory signal initiation.
Materials and Methods
In Vivo Experiments.
Experiments were conducted on 12- to 14-wk-old C57BL/6J mice and 10- to 12-wk-old Wistar rats. In some experiments, we used Trpv4−/− mice backcrossed into the C57BL/6J background, as described previously (22). All animal experiments were carried out in accordance with the European Union Community Council guidelines and were approved by the local ethics committee. Details about cystometry, micturition pattern analysis, telemetric heart rate and core body temperature measurements, thermal preference assay, and rotarod are described in SI Materials and Methods.
Bladder Histology and mRNA Expression.
After euthanasia, bladders were quickly removed, weighed, fixed in formalin, embedded in paraffin, and cut into 5-μm sections, which were stained with hematoxylin and eosin for morphological analysis. An edema index was established to reflect the severity of cystitis: 0 = no edema, 1 = mild edema expanding the lamina propria to less than double the normal size, 2 = moderate edema doubling the size of the lamina propria compared with normal, 3 = moderate edema tripling the size of the lamina propria compared with normal, and 4 = severe edema of the lamina propria and detrusor expanding the lamina propria more than three times the normal size. For each bladder, six sections were cut and scored. Quantitative PCR analysis was performed as previously described (16).
Cell Culture and Cellular Recordings.
Isolation and culture of urothelial cells and culture of HEK293 cells was performed as previously described (16). Cells were transiently transfected with the indicated ion channel cloned in the bicistronic pCAGGS-IRES-GFP vector using TransIT-293 transfection reagent (Mirus). Alternatively, indicated channels were subcloned in the pCDNA5/TO vector (Invitrogen), and stable clonal 293T-REx cell lines were grown and induced according to the manufacturer's instructions. Channel activity was assayed by using Ca2+ imaging and patch-clamp recordings, as described in SI Materials and Methods.
Reagents.
Cyclophosphamide, allyl isothiocyanate (AITC), Δ9-tetrahydrocannabinol (Δ9-THC), 4α-PDD, and 2-aminoethoxydiphenyl borate (2-APB) were purchased from Sigma. 4α-PDH was kindly provided by G. Appendino (L’Università degli Studi del Piemonte Orientale “Amedeo Avogadro,” Novara, Italy). GSK1016790A was a gift from GlaxoSmithKline.
Statistical Analyses.
Values are expressed as means ± SEM from n mice or n cells. Statistical comparisons between groups were made with Student's unpaired, two-tailed t test. To assess the in vivo effects of drug treatment, we determined the relevant parameters in the same animal before and after i.p injection of drug (or vehicle) and expressed the effects as percentage change (Δ) relative to the preinjection parameters. Student's paired, two-tailed t test was used to evaluate the statistical significance of the treatment-induced changes.
Supplementary Material
Acknowledgments
We thank Dr. W. Liedtke (Duke University, Durham, NC) for the TRPV4 knockout mice; Melissa Benoit, Sara Kerselaers, and JoAnn Witek for technical assistance; Valeri Agueev for early selectivity work; Marc D'Amours for the pharmacokinetic work and interpretation; and David Kimball, Christine Bellon, and all the members of the Laboratory of Ion Channel Research for helpful suggestions. This work was supported by Belgian Federal Government Grant IUAP P6/28, Research Foundation-Flanders (F.W.O.) Grants G.0565.07 and G.0686.09, Astellas European Foundation Award 2009, and Research Council of Katholieke Universiteit Leuven Grants GOA 2009/07 and EF/95/010. W.E. is a doctoral fellow, J.V. a postdoctoral fellow, and D.D.R. is a fundamental-clinical fellow of the F.W.O.
Footnotes
Conflict of interest statement: All authors from Hydra Biosciences, except for C.R.M., are current employees, and all are granted stock options in that company.
This article is a PNAS direct submission.
This article contains supporting information online at www.pnas.org/lookup/suppl/doi:10.1073/pnas.1005333107/-/DCSupplemental.
References
- 1.Andersson KE, Arner A. Urinary bladder contraction and relaxation: physiology and pathophysiology. Physiol Rev. 2004;84:935–986. doi: 10.1152/physrev.00038.2003. [DOI] [PubMed] [Google Scholar]
- 2.Ouslander JG. Management of overactive bladder. N Engl J Med. 2004;350:786–799. doi: 10.1056/NEJMra032662. [DOI] [PubMed] [Google Scholar]
- 3.Fowler CJ, Griffiths D, de Groat WC. The neural control of micturition. Nat Rev Neurosci. 2008;9:453–466. doi: 10.1038/nrn2401. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Abrams P, et al. The standardisation of terminology of lower urinary tract function: Report from the Standardisation Sub-committee of the International Continence Society. Am J Obstet Gynecol. 2002;187:116–126. doi: 10.1067/mob.2002.125704. [DOI] [PubMed] [Google Scholar]
- 5.Kannan H, Radican L, Turpin RS, Bolge SC. Burden of illness associated with lower urinary tract symptoms including overactive bladder/urinary incontinence. Urology. 2009;74:34–38. doi: 10.1016/j.urology.2008.12.077. [DOI] [PubMed] [Google Scholar]
- 6.Irwin DE, et al. Prevalence, severity, and symptom bother of lower urinary tract symptoms among men in the EPIC study: Impact of overactive bladder. Eur Urol. 2009;56:14–20. doi: 10.1016/j.eururo.2009.02.026. [DOI] [PubMed] [Google Scholar]
- 7.Chapple CR, et al. The effects of antimuscarinic treatments in overactive bladder: an update of a systematic review and meta-analysis. Eur Urol. 2008;54:543–562. doi: 10.1016/j.eururo.2008.06.047. [DOI] [PubMed] [Google Scholar]
- 8.Andersson KE, et al. Pharmacological treatment of overactive bladder: report from the International Consultation on Incontinence. Curr Opin Urol. 2009;19:380–394. doi: 10.1097/MOU.0b013e32832ce8a4. [DOI] [PubMed] [Google Scholar]
- 9.Hegde SS. Muscarinic receptors in the bladder: From basic research to therapeutics. Br J Pharmacol. 2006;147(Suppl 2):S80–S87. doi: 10.1038/sj.bjp.0706560. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Nilius B, Owsianik G, Voets T, Peters JA. Transient receptor potential cation channels in disease. Physiol Rev. 2007;87:165–217. doi: 10.1152/physrev.00021.2006. [DOI] [PubMed] [Google Scholar]
- 11.Everaerts W, Nilius B, Owsianik G. The vallinoid transient receptor potential channel TRPV4: From structure to disease. Prog Biophys Mol Biol. 2010;103:2–17. doi: 10.1016/j.pbiomolbio.2009.10.002. [DOI] [PubMed] [Google Scholar]
- 12.Yamada T, et al. Differential localizations of the transient receptor potential channels TRPV4 and TRPV1 in the mouse urinary bladder. J Histochem Cytochem. 2009;57:277–287. doi: 10.1369/jhc.2008.951962. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Thorneloe KS, et al. N-((1S)-1-[4-((2S)-2-[(2,4-dichlorophenyl)sulfonyl]amino-3-hydroxypropanoyl)-1-piperazinyl]carbonyl-3-methylbutyl)-1-benzothiophene-2-carboxamide (GSK1016790A), a novel and potent transient receptor potential vanilloid 4 channel agonist induces urinary bladder contraction and hyperactivity: Part I. J Pharmacol Exp Ther. 2008;326:432–442. doi: 10.1124/jpet.108.139295. [DOI] [PubMed] [Google Scholar]
- 14.Mochizuki T, et al. The TRPV4 cation channel mediates stretch-evoked Ca2+ influx and ATP release in primary urothelial cell cultures. J Biol Chem. 2009;284:21257–21264. doi: 10.1074/jbc.M109.020206. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Gevaert T, et al. Deletion of the transient receptor potential cation channel TRPV4 impairs murine bladder voiding. J Clin Invest. 2007;117:3453–3462. doi: 10.1172/JCI31766. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 16.Everaerts W, et al. Functional characterization of transient receptor potential channels in mouse urothelial cells. Am J Physiol Renal Physiol. 2010;298:F692–F701. doi: 10.1152/ajprenal.00599.2009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Geppetti P, Nassini R, Materazzi S, Benemei S. The concept of neurogenic inflammation. BJU Int. 2008;101(Suppl 3):2–6. doi: 10.1111/j.1464-410X.2008.07493.x. [DOI] [PubMed] [Google Scholar]
- 18.Cox PJ. Cyclophosphamide cystitis—Identification of acrolein as the causative agent. Biochem Pharmacol. 1979;28:2045–2049. doi: 10.1016/0006-2952(79)90222-3. [DOI] [PubMed] [Google Scholar]
- 19.D'hoedt D, et al. Stimulus-specific modulation of the cation channel TRPV4 by PACSIN 3. J Biol Chem. 2008;283:6272–6280. doi: 10.1074/jbc.M706386200. [DOI] [PubMed] [Google Scholar]
- 20.Cuajungco MP, et al. PACSINs bind to the TRPV4 cation channel. PACSIN 3 modulates the subcellular localization of TRPV4. J Biol Chem. 2006;281:18753–18762. doi: 10.1074/jbc.M602452200. [DOI] [PubMed] [Google Scholar]
- 21.Saban MR, Nguyen NB, Hammond TG, Saban R. Gene expression profiling of mouse bladder inflammatory responses to LPS, substance P, and antigen-stimulation. Am J Pathol. 2002;160:2095–2110. doi: 10.1016/S0002-9440(10)61159-5. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Liedtke W, Friedman JM. Abnormal osmotic regulation in trpv4−/− mice. Proc Natl Acad Sci USA. 2003;100:13698–13703. doi: 10.1073/pnas.1735416100. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 23.Lee H, Iida T, Mizuno A, Suzuki M, Caterina MJ. Altered thermal selection behavior in mice lacking transient receptor potential vanilloid 4. J Neurosci. 2005;25:1304–1310. doi: 10.1523/JNEUROSCI.4745.04.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 24.Charrua A, et al. GRC-6211, a new oral specific TRPV1 antagonist, decreases bladder overactivity and noxious bladder input in cystitis animal models. J Urol. 2009;181:379–386. doi: 10.1016/j.juro.2008.08.121. [DOI] [PubMed] [Google Scholar]
- 25.Birder L, et al. Activation of urothelial transient receptor potential vanilloid 4 by 4α-phorbol 12,13-didecanoate contributes to altered bladder reflexes in the rat. J Pharmacol Exp Ther. 2007;323:227–235. doi: 10.1124/jpet.107.125435. [DOI] [PubMed] [Google Scholar]
- 26.Vriens J, et al. Cell swelling, heat, and chemical agonists use distinct pathways for the activation of the cation channel TRPV4. Proc Natl Acad Sci USA. 2004;101:396–401. doi: 10.1073/pnas.0303329101. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 27.Cockayne DA, et al. Urinary bladder hyporeflexia and reduced pain-related behaviour in P2X3-deficient mice. Nature. 2000;407:1011–1015. doi: 10.1038/35039519. [DOI] [PubMed] [Google Scholar]
- 28.Ito K, Iwami A, Katsura H, Ikeda M. Therapeutic effects of the putative P2X3/P2X2/3 antagonist A-317491 on cyclophosphamide-induced cystitis in rats. Naunyn Schmiedebergs Arch Pharmacol. 2008;377:483–490. doi: 10.1007/s00210-007-0197-z. [DOI] [PubMed] [Google Scholar]
- 29.Cenac N, Altier C, Chapman K, Liedtke W, Zamponi G, Vergnolle N. Transient receptor potential vanilloid-4 has a major role in visceral hypersensitivity symptoms. Gastroenterology. 2008;135:937–946. doi: 10.1053/j.gastro.2008.05.024. [DOI] [PubMed] [Google Scholar]
- 30.Vergnolle N, et al. A role for transient receptor potential vanilloid 4 in tonicity-induced neurogenic inflammation. Br J Pharmacol. 2010;159:1161–1173. doi: 10.1111/j.1476-5381.2009.00590.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 31.Masuyama R, et al. TRPV4-mediated calcium influx regulates terminal differentiation of osteoclasts. Cell Metab. 2008;8:257–265. doi: 10.1016/j.cmet.2008.08.002. [DOI] [PubMed] [Google Scholar]
- 32.Mizuno A, Matsumoto N, Imai M, Suzuki M. Impaired osmotic sensation in mice lacking TRPV4. Am J Physiol Cell Physiol. 2003;285:C96–C101. doi: 10.1152/ajpcell.00559.2002. [DOI] [PubMed] [Google Scholar]
- 33.Suzuki M, Mizuno A, Kodaira K, Imai M. Impaired pressure sensation in mice lacking TRPV4. J Biol Chem. 2003;278:22664–22668. doi: 10.1074/jbc.M302561200. [DOI] [PubMed] [Google Scholar]
- 34.Tabuchi K, Suzuki M, Mizuno A, Hara A. Hearing impairment in TRPV4 knockout mice. Neurosci Lett. 2005;382:304–308. doi: 10.1016/j.neulet.2005.03.035. [DOI] [PubMed] [Google Scholar]
- 35.Willette RN, et al. Systemic activation of the transient receptor potential vanilloid subtype 4 channel causes endothelial failure and circulatory collapse: Part 2. J Pharmacol Exp Ther. 2008;326:443–452. doi: 10.1124/jpet.107.134551. [DOI] [PubMed] [Google Scholar]
- 36.Rock MJ, et al. Gain-of-function mutations in TRPV4 cause autosomal dominant brachyolmia. Nat Genet. 2008;40:999–1003. doi: 10.1038/ng.166. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Krakow D, et al. Mutations in the gene encoding the calcium-permeable ion channel TRPV4 produce spondylometaphyseal dysplasia, Kozlowski type and metatropic dysplasia. Am J Hum Genet. 2009;84:307–315. doi: 10.1016/j.ajhg.2009.01.021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 38.Auer-Grumbach M, et al. Alterations in the ankyrin domain of TRPV4 cause congenital distal SMA, scapuloperoneal SMA and HMSN2C. Nat Genet. 2010;42:160–164. doi: 10.1038/ng.508. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 39.Deng HX, et al. Scapuloperoneal spinal muscular atrophy and CMT2C are allelic disorders caused by alterations in TRPV4. Nat Genet. 2010;42:165–169. doi: 10.1038/ng.509. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 40.Landouré G, et al. Mutations in TRPV4 cause Charcot-Marie-Tooth disease type 2C. Nat Genet. 2010;42:170–174. doi: 10.1038/ng.512. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Nilius B, Owsianik G. Channelopathies converge on TRPV4. Nat Genet. 2010;42:98–100. doi: 10.1038/ng0210-98. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.