Abstract
Current influenza virus vaccines protect mostly against homologous virus strains; thus, regular immunization with updated vaccine formulations is necessary to guard against the virus' hallmark remodeling of regions that mediate neutralization. Development of a broadly protective influenza vaccine would mark a significant advance in human infectious diseases research. Antibodies with broad neutralizing activity (nAbs) against multiple influenza virus strains or subtypes have been reported to bind the stalk of the viral hemagglutinin, suggesting that a vaccine based on this region could elicit a broadly protective immune response. Here we describe a hemagglutinin subunit 2 protein (HA2)-based synthetic peptide vaccine that provides protection in mice against influenza viruses of the structurally divergent subtypes H3N2, H1N1, and H5N1. The immunogen is based on the binding site of the recently described nAb 12D1, which neutralizes H3 subtype viruses, demonstrates protective activity in vivo, and, in contrast to a majority of described nAbs, appears to bind to residues within a single α-helical portion of the HA2 protein. Our data further demonstrate that the specific design of our immunogen is integral in the induction of broadly active anti-hemagglutinin antibodies. These results provide proof of concept for an HA2-based influenza vaccine that could diminish the threat of pandemic influenza disease and generally reduce the significance of influenza viruses as human pathogens.
Keywords: mice, pandemic, synthetic peptide, vaccine, HA2
Vaccines designed to elicit antibodies against the stalk of the hemagglutinin can protect against influenza virus infection (1, 2), and serum from animals immunized with stalk constructs can have significantly enhanced heterosubtypic binding activity over serum from infected animals (2). Interestingly, immunization with DNA coding for the hemagglutinin has been observed to generate a predominantly stalk-specific antibody response (3). This phenomenon was also observed in a subsequent report demonstrating that priming with hemagglutinin DNA before seasonal vaccination can boost the anti-stalk antibody titer (4). Heterosubtypic protection provided by vaccination with stalk vaccine constructs or with DNA coding for hemagglutinin has not yet been described.
In this report, we demonstrate a synthetic peptide-based vaccine construct that elicits antibodies against the stalk of the influenza virus hemagglutinin protein. This construct has protective activity against antigenically divergent virus subtypes that currently cause seasonal and pandemic disease in humans. Additionally, the vaccine has protective activity against an avian H5N1 virus, a subtype with potential to cause pandemic influenza disease in humans. As a peptide-based construct, this vaccine would be inexpensive and uncomplicated to manufacture.
The influenza virus hemagglutinin is a polypeptide made up of the HA1 and HA2 proteins, with the stalk of the molecule comprising the N- and C-terminal portions of HA1 along with the HA2 molecule (5). Seasonal influenza vaccines act primarily by generating neutralizing antibodies against the viral hemagglutinin; these antibodies act by preventing either of two steps involved in virus entry: binding of the virus to the host cell or fusion of the viral and host cell membranes (6). Antibodies described to date with broad neutralizing activity against an array of virus subtypes or strains (nAbs) act by preventing the fusion step of virus entry (3, 7–9).
Influenza hemagglutinin proteins are divided into 16 subtypes which are further divided into two major phylogenetic groups: group 1 (subtypes H1, H2, H5, H6, H8, H9, H11, H12, H13, and H16) and group 2 (subtypes H3, H4, H7, H10, H14, and H15). This division of the 16 subtypes correlates with two distinct basic structures taken by the stalk of the hemagglutinin, so that group 1 proteins have similar stalk structures, as do group 2 proteins (9). The vaccine construct described here shows activity against viruses expressing hemagglutinin proteins from both group 1 and group 2.
Results
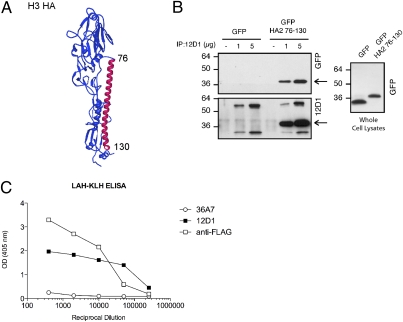
Mouse mAb 12D1 is known to bind a continuous portion of the HA2 molecule and has broad neutralizing activity against influenza viruses of the H3 subtype. By generating constructs designed to express short regions of the HA2 molecule, we found that nAb 12D1 binds amino acids within the highly conserved long α-helix (LAH) region of the protein. The portion of the hemagglutinin that interacts with nAb 12D1 was identified originally by interpretation of binding data using multiple HA2 truncations of varying lengths. Based on the cumulative truncation data, it was determined that nAb 12D1 binds within amino acids 76–106 of HA2 (3). Subsequent work, however, revealed that a peptide representing the entire LAH (amino acids 76–130) of the H3 virus A/Hong Kong/1/1968 provided the necessary structural elements for maximal binding by nAb 12D1 (Fig. 1A). This region, amino acids 76–130 of HA2, was expressed in 293T cells and could be pulled down by nAb 12D1 (Fig. 1B). We therefore hypothesized that immunization with the amino acid 76–130 region of HA2 might elicit an antibody repertoire with functional similarity to that of nAb 12D1 and provide protection against influenza viruses of the H3 subtype or of multiple subtypes.
Fig. 1.
mAb 12D1 reacts with the LAH of HA2. (A) Hemagglutinin monomer. Amino acids 76–130 of HA2 are highlighted in red. (B) Lysates from 293T cells were transfected with pCAGGs-GFP or pCAGGs-GFP HA2 76–130, incubated with mAb 12D1, and pulled down with protein G beads. Pulled-down fractions were blotted with mAb 12D1 or rabbit anti-GFP. Arrows indicate location of the amino acid 76–130 region–GFP fusion protein. Anti-mouse HRP used to detect 12D1-binding reacts with the mouse Ig heavy and light chains within the pulled-down fraction. (C) Structural integrity of the 12D1 epitope within the LAH-KLH conjugate was confirmed by direct-binding ELISA. Hemagglutinin-specific mAb 36A7 does not bind within the amino acid 76–130 region of HA2.
To enhance antigenicity of the amino acid 76–130 polypeptide (LAH), we designed a conjugate vaccine consisting of the LAH plus a C-terminal spacer domain of eight amino acids (Flag tag) followed by a cysteine residue which facilitated coupling to the carrier protein keyhole limpet hemocyanin (KLH). To extend serum half-life, the LAH peptide was acetylated at the N terminus (10). The structural integrity of the nAb 12D1-binding region within the conjugate was confirmed by direct-binding ELISA (Fig. 1C).
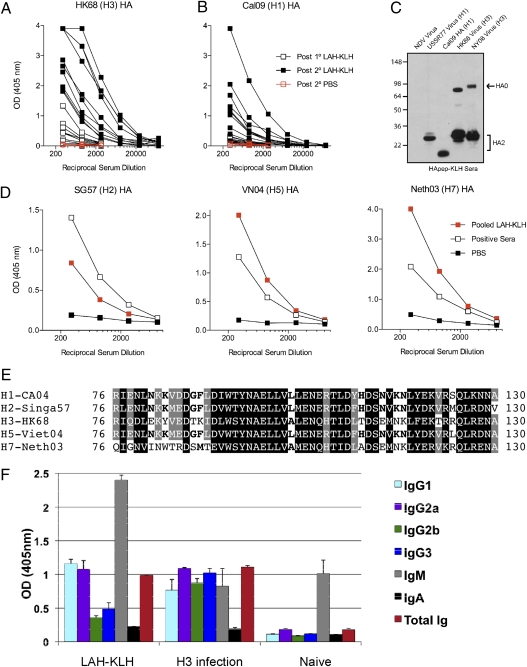
To test the construct in vivo, mice were immunized with the LAH-KLH conjugate in a prime-boost schedule with 3 wk lapsing between immunizations. Sera were taken from mice 10 d after primary and secondary immunizations. To evaluate the ability of the conjugate to elicit the production of antibodies of relevant specificity, antisera were tested for reactivity with purified hemagglutinin protein of different subtypes. First, we noted that the LAH antiserum did react with hemagglutinin protein by both ELISA and by Western blot (Fig. 2 A–C). Second, a marked increase in anti-hemagglutinin titer following secondary immunization demonstrated that the construct acted as a productive immunogen in mice (Fig. 2 A and B). Finally, we were intrigued to find that sera from immunized mice had substantial heterosubtypic binding activity. Anti-LAH sera demonstrated activity by ELISA with hemagglutinins from the 1968 pandemic H3 virus A/Hong Kong/1/1968 and the 2009 pandemic H1 virus A/California/04/09 as well as hemagglutinins of H2, H5, and H7 subtypes (Fig. 2D). Indeed, alignment of the 76–130 amino acid region of hemagglutinins from these subtypes demonstrates a high degree of conservation in amino acid sequence and amino acid type (Fig. 2E). Further serologic analysis demonstrated that antibody generated in LAH-KLH vaccination boosts serum IgM and IgG subtypes specific for the viral hemagglutinin. The significant boost in IgG subtypes indicates T cell-dependent antibody production and suggests an affinity matured anti-hemagglutinin response (Fig. 2F) (11).
Fig. 2.
LAH-KLH vaccine acts as a robust immunogen, and the elicited serum antibody reacts with multiple hemagglutinin subtypes. (A and B) Sera from individual mice were taken 10 d after primary and secondary immunization and were tested for binding to H3 or H1 hemagglutinin by ELISA. (C) Pooled sera from 20 mice taken after secondary immunization with LAH-KLH were tested by Western blot for reactivity with purified influenza virus subtypes A/Hong Kong/1/1968 (HK68; H3), A/New York/1/2008 (NY08; H3) or A/USSR/90/1977 (USSR77; H1), purified A/California/04/09 (transmembrane domain absent) (Cal09 HA; H1); or Newcastle disease virus (NVD). (D) Pooled sera were tested for binding activity against H2, H5, or H7 hemagglutinin by ELISA. LAH-KLH antiserum has considerable binding activity against all three hemagglutinin subtypes. Positive sera used were from mice infected with either a group 1 influenza virus [A/Singapore/1/57 (SG57); H2 ELISA and A/Viet Nam/1203/2004 (VN04); H5 ELISA] or a group 2 virus [A/Netherlands/219/2003 (Neth03); H7 ELISA] (2). (E) LAH amino acids 76–130 from the HA2 of different hemagglutinin subtypes. White letters on a black background indicate a residue conserved in all five HAs. Conserved residues fall into one of four groups: (i) D/N/E/Q, (ii) I/L/V/M, (iii) K/R, or (iv) S/T. White letters on a gray background indicate a residue conserved in four of five HAs or less stringent conservation (R vs. H near middle conserves charge, but there is a change in size). Bold text on a white background indicates partial conservation (three of five HAs) or less stringent (L vs. A at middle or F vs. M toward left). (F) Isotype profile of hemagglutinin-specific antibody in serum pools from normal (Naïve) mice, mice infected with A/Hong Kong/1/1968 (H3), or mice immunized with LAH-KLH. Recombinant hemagglutinin from A/Hong Kong/1/1968 was used to coat plates for ELISA.
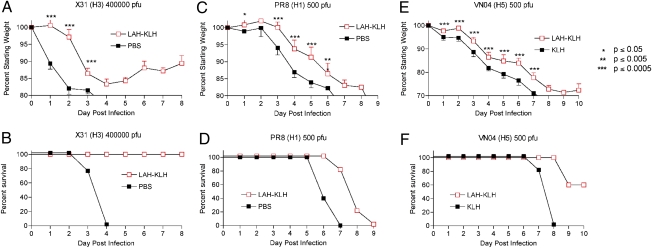
Mice were challenged 2 to 3 wk following secondary immunization by intranasal administration with 4 × 105 pfu of X31, a mouse-adapted virus expressing the hemagglutinin and neuraminidase of the 1968 pandemic H3 influenza virus. Mice immunized with the LAH-KLH construct lost significantly less weight at all time points than did mice that received PBS with adjuvant. In addition, all immunized mice survived challenge, whereas control mice succumbed to infection by day 4 (Fig. 3 A and B).
Fig. 3.
Immunization with LAH-KLH protects mice in vivo. Two weeks after secondary immunization, mice were challenged with 4 × 105 pfu of X31, a mouse-adapted H3 influenza virus (A and B), 500 pfu (10–15 MLD50) of the mouse-adapted H1 virus PR8 (C and D), or 500 pfu of an H5 highly pathogenic avian influenza virus modified to remove the polybasic cleavage site in the viral hemagglutinin (HAlo virus) (12) (E and F). n = five BALB/c mice per group. Because of differences in pathogenicity, survival was defined as 20% weight loss for X31 (H3) and PR8 (H1) viruses and 30% weight loss for VN/2004 (H5) virus.
Next, immunized mice were challenged with other virus subtypes that cause human influenza disease but that belong to a phylogenetic class distinct from H3 subtype viruses (6). Mice were infected with 500 pfu [10–15 times the dose lethal to 50% of mice (MLD50)] of PR8 (a mouse-adapted H1 virus) or with 500 pfu of an H5 highly pathogenic avian influenza virus modified to remove the polybasic cleavage site in the viral hemagglutinin (12). Vaccination with the LAH-KLH conjugate was protective against weight loss caused by H1 and H5 influenza disease to a highly significant degree on virtually every day during infection. Vaccinated mice infected with PR8 showed a significant delay in kinetics of weight loss, and 60% of vaccinated mice infected with the H5 avian virus survived lethal challenge to 10 d postinfection (Fig. 3 C–F).
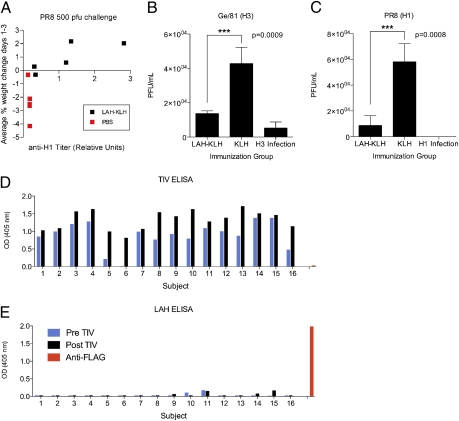
An effective vaccine against seasonal or pandemic influenza disease must act to prime the humoral arm of the immune system for a response capable of significantly diminishing virus replication. In the days immediately after infection, serum antibody can provide a critical role in curbing viral replication because the cell-mediated and innate arms of the immune system are not yet fully engaged (13, 14). Analysis of prechallenge sera from mice that subsequently were infected with PR8 revealed a positive correlation between hemagglutinin-specific antibody titer and increase in body weight in days following infection (Fig. 4A). Animals productively immunized (with anti-H1 serum antibody) gained weight during days 1–3 postinfection, whereas animals without H1-specific antibody lost weight during this critical period. These data suggested that antibody induced by LAH-KLH vaccination was a requisite component in protection of mice against disease.
Fig. 4.
Antibody mediates protection afforded by immunization with LAH-KLH. (A) Analysis of prechallenge serum from mice infected with PR8 reveals a positive correlation between hemagglutinin-specific antibody titer and increase in body weight on days 1–3 after infection. (B and C) Pooled sera from mice immunized with LAH-KLH, mice infected with H1 or H3 virus, or mice immunized with KLH alone were transferred to mice 2 h before infection with A/Georgia/81, a seasonal human H3 virus, or with the H1 virus PR8. Lung titers were evaluated on day 2 postinfection (***P < 0.001). (D and E) Human sera taken pre- or postimmunization with the TIV were evaluated for binding activity with the LAH polypeptide. Data shown are from serum samples diluted 1:3,000. (D) Subjects responded variably to seasonal vaccination, and (E) serum demonstrates minimal binding activity against the LAH peptide.
To investigate further the role of anti-LAH antibody in protection, we performed in vivo passive transfer experiments. Two hours before infection, recipient mice were given 200 uL of serum by i.p. administration from donor mice that had been infected with H1 or H3 virus and vaccinated with KLH alone or with the LAH-KLH vaccine. Recipient mice then were infected with a human seasonal H3 virus, A/Georgia/81, or with the H1 virus PR8. Lung titers were evaluated 2 d after infection. The transfer of LAH-KLH antiserum was found to reduce lung titers significantly in animals infected with either the human seasonal H3 virus (P = 0.0009) or the H1 virus (P = 0.0008) (Fig. 4 B and C). This transfer experiment suggests that the LAH construct induces neutralizing antibodies in the vaccinated mouse.
Next, we investigated whether seasonal influenza vaccination in humans induces antibody specific for the LAH region of the hemagglutinin. To explore this possibility, we evaluated binding activity in human sera taken pre- and postimmunization with the 2008–2009 trivalent inactivated influenza virus vaccine (TIV). This seasonal vaccine composition contained an A/Brisbane/59/2007 (H1N1)-like virus, an A/Brisbane/10/2007 (H3N2)-like virus, and a B/Florida/4/2006-like virus (15). Serum samples from human patients were evaluated for a postvaccination boost in IgG antibody titer against the seasonal TIV composition as a measure of vaccine response. Minimal serum antibody specific for the LAH peptide was detected even in subjects demonstrating the highest response to seasonal vaccination (Fig. 4 D and E).
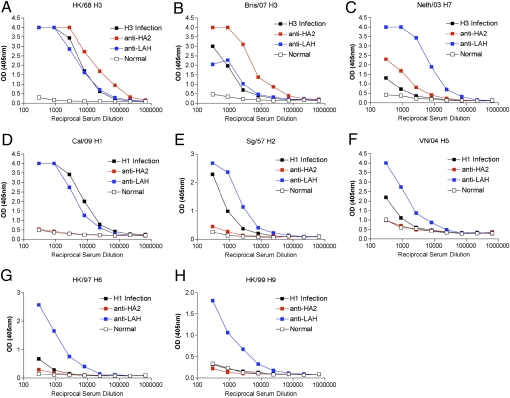
As demonstrated in Fig. 2 A–D, the breadth of reactivity seen in the LAH-KLH antiserum is greater than has been described previously in studies of hemagglutinin stalk vaccine constructs (1, 2). To probe the importance of the design of the conjugate complex in eliciting this broad response, we compared the serum activity elicited by vaccination with the LAH-KLH construct with that elicited by vaccination with the intact HA2 molecule. The ectodomain of the A/Hong Kong/1/1968 HA2 protein was recombinantly expressed, as previously described (16). Mice were vaccinated with pure, uncoupled HA2 protein by the same methods used to vaccinate mice with LAH-KLH. Pooled antisera from 20 mice taken 10 d after secondary vaccination with either LAH-KLH or HA2 protein were evaluated for binding activity against a panel of recombinantly expressed hemagglutinins. Anti-HA2, anti-LAH, and positive control sera demonstrated approximately equivalent activity against the homologous HK/68 hemagglutinin. Although the LAH-KLH antiserum reacted with all hemagglutinin subtypes tested, the HA2 antiserum contained antibody reactive only with group 2 hemagglutinin proteins (Fig. 5 A–H and Table 1). Because the LAH structure is present in the HA2 protein, the broad reactivity seen in the LAH-KLH antiserum must be a consequence of the manner in which the LAH is presented as an antigen within the conjugate complex. Elimination of immunodominant regions of the HA2 protein may cause the LAH-KLH vaccine to induce a more focused anti-LAH immune response that mediates broad reactivity between hemagglutinin subtypes. Alternately, the induction of broadly reactive antibody may be a consequence of anchoring the LAH at the C terminus to a carrier protein, thus rendering immunogenic regions of the LAH that otherwise are antigenically silent in the context of the intact HA2 protein.
Fig. 5.
LAH-KLH antiserum reacts with both group 1 and group 2 hemagglutinin proteins, whereas HA2 antiserum reacts only with group 2 hemagglutinin proteins. (A–C) Activity of antisera against group 2 hemagglutinin proteins. HA2 and LAH-KLH serum pools demonstrate comparable binding activity against the A/Hong Kong/68 H3 hemagglutinin but have different binding activities against other group 2 hemagglutinins. (D–H) Activity of antisera against group 1 hemagglutinin proteins. The LAH-KLH serum pool reacts with all group 1 hemagglutinin proteins tested, but the HA2 antiserum does not. Positive control serum used in this figure and Table 1 was from mice infected with either the group 2 H3 subtype X31 virus or the group 1 H1 subtype PR8 virus.
Table 1.
Summary of ELISA data in Fig. 5
| Hemagglutinin | Anti-LAH-KLH | Anti-HA2 |
| HK/68 H3* | + | + |
| Bris/07 H3* | + | + |
| Neth/03 H7* | + | + |
| Cal/09 H1† | + | − |
| Sing/57 H2† | + | − |
| Viet/04 H5† | + | − |
| HK/97 H6† | + | − |
| HK/99 H9† | + | − |
*Group II.
†Group I.
Discussion
That immunization with the TIV in humans does not generate significant anti-LAH antibody is not surprising, because the stem of the intact viral hemagglutinin is known to be poorly immunogenic in the presence of the immunodominant head of the hemagglutinin molecule (17). Importantly, however, human survivors of H5N1 virus infection do have serum antibody specific for a peptide comprising amino acids 79–134 of the HA2 protein (18). This result suggests that the human antibody repertoire includes specificities that would be elicited or boosted by vaccination with an LAH-based vaccine. In general, however, after infection or vaccination with conventional vaccines, immune responses are made against the immunodominant portion of the hemagglutinin.
Development of a human vaccine analog of the LAH-KLH construct would be relatively straightforward. One of several carrier proteins used in human vaccines (e.g., CRM197, tetanus toxoid, diphtheria toxoid, or meningococcal outer membrane protein) might be used in place of KLH. A human LAH peptide conjugate vaccine could be administered in aluminum salts or in an oil-in-water adjuvant to enhance the anamnestic response and to reduce the amount of antigen required for productive immunization (19). As seen in our animal studies, such a construct may act to diminish disease caused by a variety of influenza virus subtypes.
The fact that the LAH-KLH construct is based on the H3 long α-helix sequence but elicits an immune response that is broadly reactive with other hemagglutinin subtypes is intriguing. The absolute percent identity between subtypes for the amino acid 76–130 region of HA2 can be less than 50%; however, amino acids accessible to an antibody-combining site in the context of the helical structure may be more conserved. It also is possible that the significant conservation of amino acid properties at each position enables the H3 helix to act as an antigenic mimetic for helices of other subtypes. We are particularly encouraged by the activity of this construct, because the LAH region of the HA2 is highly resistant to mutations that allow escape from neutralization by the 12D1 antibody (3). Incorporation of other polypeptides mediating broad neutralization of influenza viruses into the LAH conjugate would reduce further the likelihood of inducing an immune response that would select for viral escape variants.
Because in vivo efficacy of the LAH vaccine was determined in challenge experiments using different infectious doses of a variety of influenza strains, the relative degree of protection afforded against the various subtypes cannot be determined. Additionally, it is not possible to infer from the mouse protection data presented here which strains would be more or less targeted by vaccination of humans with the LAH conjugate. We would hypothesize, however, that the LAH vaccine would be most efficacious in protecting against H3 virus subtypes and provide moderate to substantial protection against other virus subtypes. Subbarao et al. (20) have suggested that, realistically, an appropriate vaccine for pandemic influenza will act to diminish morbidity and mortality without obviating all disease symptoms caused by infection. A synthetic peptide vaccine such as the LAH-KLH virus would be straightforward and inexpensive to generate and may be ideal for reducing the burden of seasonal and pandemic influenza disease.
Materials and Methods
Viruses and Purified Hemagglutinins.
Viruses used were X31 virus (A/Hong Kong/1/1968 hemagglutinin and neuraminidase with remaining six segments from PR8), A/Puerto Rico/8/34 (PR8) virus, A/USSR/90/1977 virus, A/Georgia/81 virus, and HAlo virus (A/Viet Nam/4/2005 virus with hemagglutinin modified to remove the polybasic cleavage site). Purified hemagglutinin for A/Hong Kong/1/1968 and A/Brisbane/10/2007 were generated by I.W. and D.C.E.; purified hemagglutinin for A/Viet Nam/1203/2004 (H5), A/Singapore/1/57 (H2), A/Teal/HK/312/97 (H6), A/Netherlands/219/2003 (H7), A/Hong Kong/1073/99 (H9), and A/California/04/2009 (H1) were obtained from the Biodefense and Emerging Infections Research Resources Repository (BEI Resources).
Western Blot.
Blots were produced by methods previously described (21). Samples were boiled for 5 min at 100 °C in loading buffer containing SDS and 0.6 M DTT. Immunoprecipitated complexes, cell lysates, or purified viruses were resolved in a 4–20% Tris-HCl SDS/PAGE gel (Bio-Rad, Inc.), and samples were blotted onto a Protran nitrocellulose membrane (Whatman). GFP and fusion GFP-HA truncated peptides were detected using rabbit anti-GFP (Santa Cruz Biotechnology, Inc.) and/or mAb 12D1. Secondary antibodies were anti-rabbit IgG HRP (Dako) and anti-mouse Ig-HRP (GE Healthcare, Inc.).
Immunoprecipitation.
The LAH region (amino acids 76–130) of the A/HK/1/68 HA2 was generated by PCR amplification of viral RNA and was subcloned into a pCAGG-GFP plasmid (22). GFP was present at the N-terminal of the HA2 truncation. We transfected 293T cells with the GFP-LAH construct using Lipofectamine 2000 (Invitrogen, Inc.). At 24 h posttransfection, cells were lysed with radioimmunoprecipitation assay buffer, and the GFP-LAH fusion protein was immunoprecipitated overnight at 4 °C with 1–5 μg of mAb 12D1 bound to protein G-Agarose (Roche, Inc.).
LAH-KLH Vaccine.
The polypeptide sequence is Ac-RIQDLEKYVEDTKIDLWSYNAELLVALENQHTIDLTDSEMNKLFEKTRRQLRENADYKDDDDKC.
The construct is acetylated at the N terminus and consists of amino acids 76–130 of the H3 A/Hong Kong/1/1968 HA2 molecule followed by a Flag-tag (DYKDDDDK), followed by a cysteine. The polypeptide is coupled to the carrier protein KLH by thiol to primary amine coupling. This conjugate was produced by CHI Scientific, Inc.
ELISAs.
Ninety-six–well plates (Immulon 2; Nunc) were coated with 2 μg/mL LAH-KLH conjugate (Fig. 1B), purified hemagglutinin (Fig. 2 A and C), or influenza virus vaccine [FLUVIRON (R), obtained from BEI Resources] purified surface antigen (Novartis Vaccines) in PBS overnight at 4 °C. Plates were blocked for 30 min at room temperature with 1% BSA/PBS and were washed twice with PBS/0.025% Tween. Antibodies, antiserum, or serum from individuals vaccinated with the 2008–2009 TIV were serially diluted in 1% BSA/PBS and added to the plate followed by 3-h incubation at 37 °C. An anti-Flag antibody (Sigma) was used as a positive control in wells coated with the LAH-KLH conjugate. Plates were washed three times, and anti-mouse alkaline phosphatase (AP) (Southern Biotech) diluted 1:2,000 was added to wells followed by 3-h incubation at 37 °C. For human sera, anti-human IgG (Fc spscific)-AP (Sigma) antibody was used at 1:500 dilution. Anti-rabbit Ig-AP (Southern Biotech) at 1:500 dilution was used as the secondary for the anti-Flag antibody. P-nitrophenyl phosphate substrate then was added to the wells and allowed to develop for 20–30 min at room temperature. Optical density measurements were taken at 405 nm.
Mouse Immunizations and Challenge Experiments.
All animal procedures were performed in accordance with Institutional Animal Care and Use Committee (IACUC) guidelines and have been approved by the IACUC of Mount Sinai School of Medicine. BALB/C mice (6- to 8-wk-old) (Jackson Laboratories) were immunized with 25 μg LAH-KLH, HA2, KLH alone, or PBS in Complete Freund's adjuvant (Sigma) by s.c. administration. Three weeks after primary immunization, mice were boosted with 25 μg of the same immunogen or with PBS in Incomplete Freund's adjuvant. Two to three weeks after the boost, mice were challenged with virus. Before virus infection, mice were anesthetized by i.p. administration of a mixture of ketamine (75 mg/kg body weight)/xylazine (15 mg/kg body weight). Virus was administered intranasally in 50 μL total PBS; challenge doses consisted of 4 × 105 pfu X31 or 500 pfu PR8 or HAlo virus. Body weights were monitored daily. For passive transfer experiments, mice were bled 2 wk after the last immunization with KLH or LAH-KLH or 3 wk after infection with PR8 virus or A/Hong Kong/1/1968 virus. Sera from mice were pooled according to vaccination antigen or virus infection, and 200 μL of serum was transferred to each recipient mouse by i.p. administration 2 h before infection with either 50 pfu PR8 virus or 3,700 pfu A/Georgia/81 virus. Lung titers were assessed by plaque assay 2 d postinfection.
Acknowledgments
We thank Lauren Peters for technical assistance. This research was supported by National Institutes of Health (NIH) Grants UO1 AI070469 (Live Attenuated Vaccines for Epidemic and Pandemic Flu), HHSN2662000700010C (Center for Research on Influenza Pathogenesis), 1RC1 AI086061-01 (Development of A Universal Influenza Virus Vaccine), and U54 AI057158-04 (Northeast Biodefense Center) and by NIH/National Institute of Allergy and Infectious Diseases Grants R01 AI0401111 (TH1 and TH2 Responses in Viral Immunity) and HHSN266200500028C (Immune Response to Virus Infection During Pregnancy). T.T.W. was supported by NIH Training Grant T32 AI007647 and by Mount Sinai Medical Scientists Training Grant T32 GM007280. This work also was supported in part by NIH Grant AI058113 (to I.A.W.), by Predoctoral Fellowships from the Achievement Rewards for College Scientists Foundation, by Grant GM080209 from the NIH Molecular Evolution Training Program (to D.C.E.), and by the Skaggs Institute for Chemical Biology.
Footnotes
Conflict of interest statement: Mount Sinai School of Medicine has filed a provisional patent covering some of the results described in this paper.
References
- 1.Bommakanti G, et al. Design of an HA2-based Escherichia coli expressed influenza immunogen that protects mice from pathogenic challenge. Proc Natl Acad Sci USA. 2010;107:13563–13564. doi: 10.1073/pnas.1007465107. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Steel J, et al. Influenza virus vaccine based on the conserved hemagglutinin stalk domain. MBio. 2010;1(1):1–9. doi: 10.1128/mBio.00018-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 3.Wang TT, et al. Broadly protective monoclonal antibodies against H3 influenza viruses following sequential immunization with different hemagglutinins. PLoS Pathog. 2010;6:e1000796. doi: 10.1371/journal.ppat.1000796. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Wei CJ, et al. Induction of broadly neutralizing H1N1 influenza antibodies by vaccination. Science. 2010;329(5995):1060–1064. doi: 10.1126/science.1192517. [DOI] [PubMed] [Google Scholar]
- 5.Wilson IA, Skehel JJ, Wiley DC. Structure of the haemagglutinin membrane glycoprotein of influenza virus at 3 A resolution. Nature. 1981;289:366–373. doi: 10.1038/289366a0. [DOI] [PubMed] [Google Scholar]
- 6.Palese P, Shaw M. Fields' Virology. 5th Ed. Philadelphia: Lippincott Williams & Wilkins; 2007. [Google Scholar]
- 7.Throsby M, et al. Heterosubtypic neutralizing monoclonal antibodies cross-protective against H5N1 and H1N1 recovered from human IgM+ memory B cells. PLoS ONE. 2008;3:e3942. doi: 10.1371/journal.pone.0003942. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Sui J, et al. Structural and functional bases for broad-spectrum neutralization of avian and human influenza A viruses. Nat Struct Mol Biol. 2009;16:265–273. doi: 10.1038/nsmb.1566. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Ekiert DC, et al. Antibody recognition of a highly conserved influenza virus epitope. Science. 2009;324:246–251. doi: 10.1126/science.1171491. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Werle M, Bernkop-Schnürch A. Strategies to improve plasma half life time of peptide and protein drugs. Amino Acids. 2006;30:351–367. doi: 10.1007/s00726-005-0289-3. [DOI] [PubMed] [Google Scholar]
- 11.Jumper MD, Splawski JB, Lipsky PE, Meek K. Ligation of CD40 induces sterile transcripts of multiple Ig H chain isotypes in human B cells. J Immunol. 1994;152:438–445. [PubMed] [Google Scholar]
- 12.Steel J, et al. Live attenuated influenza viruses containing NS1 truncations as vaccine candidates against H5N1 highly pathogenic avian influenza. J Virol. 2009;83:1742–1753. doi: 10.1128/JVI.01920-08. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Moltedo B, et al. Cutting edge: Stealth influenza virus replication precedes the initiation of adaptive immunity. J Immunol. 2009;183:3569–3573. doi: 10.4049/jimmunol.0900091. [DOI] [PubMed] [Google Scholar]
- 14.Hermesh T, Moltedo B, Moran TM, López CB. Antiviral instruction of bone marrow leukocytes during respiratory viral infections. Cell Host Microbe. 2010;7:343–353. doi: 10.1016/j.chom.2010.04.006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Centers for Disease Control and Prevention. Update: influenza activity–United States, September 30, 2007–April 5, 2008, and composition of the 2008–09 influenza vaccine. MMWR Morb Mortal Wkly Rep. 2008;57(15):404–409. [PubMed] [Google Scholar]
- 16.Chen J, Skehel JJ, Wiley DC. N- and C-terminal residues combine in the fusion-pH influenza hemagglutinin HA(2) subunit to form an N cap that terminates the triple-stranded coiled coil. Proc Natl Acad Sci USA. 1999;96:8967–8972. doi: 10.1073/pnas.96.16.8967. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 17.Kwong PD, Wilson IA. HIV-1 and influenza antibodies: Seeing antigens in new ways. Nat Immunol. 2009;10:573–578. doi: 10.1038/ni.1746. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Khurana S, et al. Antigenic fingerprinting of H5N1 avian influenza using convalescent sera and monoclonal antibodies reveals potential vaccine and diagnostic targets. PLoS Med. 2009;6:e1000049. doi: 10.1371/journal.pmed.1000049. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Tritto E, Mosca F, De Gregorio E. Mechanism of action of licensed vaccine adjuvants. Vaccine. 2009;27:3331–3334. doi: 10.1016/j.vaccine.2009.01.084. [DOI] [PubMed] [Google Scholar]
- 20.Subbarao K, Murphy BR, Fauci AS. Development of effective vaccines against pandemic influenza. Immunity. 2006;24:5–9. doi: 10.1016/j.immuni.2005.12.005. [DOI] [PubMed] [Google Scholar]
- 21.Towbin H, Staehelin T, Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: Procedure and some applications. Proc Natl Acad Sci USA. 1979;76:4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 22.Basler CF, et al. Sequence of the 1918 pandemic influenza virus nonstructural gene (NS) segment and characterization of recombinant viruses bearing the 1918 NS genes. Proc Natl Acad Sci USA. 2001;98:2746–2751. doi: 10.1073/pnas.031575198. [DOI] [PMC free article] [PubMed] [Google Scholar]