Abstract
Background and objectives: Cardiovascular events are common in patients with ESRD. Whether sympathetic overactivity or vagal withdrawal contribute to cardiovascular events is unclear. We determined the general prevalence and clinical correlates of heart rate variability in patients on hemodialysis.
Design, setting, participants, & measurements: We collected baseline information on demographics, clinical conditions, laboratory values, medications, physical performance, left ventricular mass (LVM), and 24-hour Holter monitoring on 239 subjects enrolled in the Frequent Hemodialysis Network Daily Trial.
Results: The mean R-R interval was 812 ± 217 ms. The SD of R-R intervals was 79.1 ± 40.3 ms. Spectral power analyses showed low-frequency (sympathetic modulation of heart rate) and high-frequency power (HF; vagal modulation of heart rate) to be 106.0 (interquartile range, 48.0 to 204 ms2) and 42.4 ms2 (interquartile range, 29.4 to 56.3 ms2), respectively. LVM was inversely correlated with log HF (−0.02 [−0.0035; −0.0043]) and the R-R interval (−1.00 [−1.96; −0.032]). Physical performance was associated with mean R-R intervals (1.98 [0.09; 3.87]) and SD of R-R intervals (0.58 [0.049; 1.10]). After adjustment for age, race, ESRD vintage, diabetes, and physical performance, the relationship between log HF and LVM (per 10 g) remained significant (−0.025 [−0.042; −0.0085]).
Conclusions: Holter findings in patients on hemodialysis are characterized by sympathetic overactivity and vagal withdrawal and are associated with higher LVM and impaired physical performance. Understanding the spectrum of autonomic heart rate modulation and its determinants could help to guide preventive and therapeutic strategies.
Cardiovascular events in general, and sudden death in particular, are exceptionally common in patients with ESRD (1). Despite the potential role for autonomic dysfunction contributing to excess cardiovascular morbidity and mortality in uremia, few studies of cardiac autonomic function in patients with ESRD have been performed. Measurement of heart rate variability (HRV) is a noninvasive method to assess for autonomic modulation of sino-atrial discharge (2). Previous studies have shown attenuated HRV to be a potent marker of premature cardiac death in patients with high cardiovascular risk (3). Given accumulating evidence that sympathetic hyperactivity and/or vagal withdrawal may lead to left ventricular hypertrophy and poorer physical fitness, understanding associations among these factors and potential mechanisms explaining these associations merits further study.
The Frequent Hemodialysis Network (FHN) Daily Trial aims to examine the effects of frequent (six times per week) hemodialysis versus conventional thrice weekly hemodialysis on left ventricular mass (LVM) and physical function, among an array of intermediate outcome measures (4). The study objectives and protocol summary have been published previously (4). One of the study's secondary goals was to describe impairments of autonomic modulation of heart rate and to explore possible associations with structural cardiac abnormalities and/or other demographic, clinical, and laboratory factors or performance measures.
We conducted the present cross-sectional study to (1) describe the magnitude of autonomic dysfunction in HRV in a relatively large cohort of hemodialysis patients and (2) explore the potential relations among indices of HRV and possible clinical determinants.
Materials and Methods
Study Design
All subjects enrolled in the trial were asked to undergo 24-hour Holter testing at baseline. A description of the array of demographic, clinical, and laboratory factors and bioelectrical impedance analysis conducted as part of baseline evaluation in the FHN study is available elsewhere (4). In addition, we recorded the use of cardiovascular-related medications including beta blockers, calcium channel blockers, aspirin, and lipid-lowering medications. All patients received conventional hemodialysis of three sessions per week with a minimum eKt/V of 1.1 per session and a minimum treatment time of >2.5 hours per session (5). All patients provided written informed consent. All participating sites received institutional research board/ethics board approval. The FHN Daily Trial is registered at www.clinicaltrials.gov (study number NCT00264758). Comorbidity was summarized using the modified Charlson index (6).
HRV
Study staff measured continuous heart rate recordings using the KCI X5 digital Holter recorder (KCI Technology and Services). All study coordinators were trained and demonstrated competence in obtaining Holter tracings. Electrocardiographic data were stored and couriered to the core laboratory in Toronto, Canada. We analyzed the acquired data centrally in a blinded manner as previously reported (7). Only normal sinus recordings were used to assess HRV. We determined low-frequency (LF) harmonic (0.05 to 0.15 Hz; sympathetic modulation of heart rate) and high-frequency (HF) harmonic (0.15 to 0.5 Hz; parasympathetic modulation of heart rate) contributions to spectral power. LF/HF was used to denote sympathetic to parasympathetic ratio. R-R intervals and SD of R-R intervals (SDNN; a measure of beat to beat variability) were also computed.
Cardiac Magnetic Resonance Imaging
We measured left ventricular geometry and mass by cardiac magnetic resonance imaging (CMRI). As with Holter data, all CMRI images were analyzed centrally in a blinded manner. CMRI was performed on 1.5-T MRI systems (Avanto or Sonata; Siemens Medical Solutions, Erlangen, Germany, or GE Signa Systems) with eight-channel cardiac coils. We applied standard, breath-held, retrospectively electrocardiographic-gated gradient-echo sequences (steady-state free precession) in contiguous short-axis views (20 to 25 phases, 8-mm slice thickness). Using validated software (Argus; Siemens Medical Solutions), we measured volumes on end-diastolic frames by manually tracing endocardial and epicardial contours. We excluded papillary muscles from the calculation of myocardial mass. The formula of DuBois and DuBois was used to index LVM to body surface area (8).
Physical Performance
We used the Short Physical Performance Battery (SPPB) to capture the level of physical functioning. The SPPB (9) is a simple battery that can be performed at the bedside. It is based on (1) the time needed (in seconds) to walk 4 m at a normal pace; (2) the time needed to stand up and sit down five times as quickly as possible from a chair with the arms folded across the chest; and (3) a timed balance test that measures the ability to maintain balance with feet together in three positions (side by side, semitandem, and tandem). Each of the three performance measures are assigned a score ranging from 0 to 4, with 0 being the inability to complete the test and 4 indicating the highest level of performance. The total SPPB performance score ranges from 0 to 12.
Statistical Analyses
Descriptive data are presented as mean ± SD or median and interquartile range for normal and skewed population data, respectively. Potential associations among indices of HRV and various clinical determinants including LVM, SPPB, and laboratory measures were first examined graphically to determine the appropriate functional form and then tested using linear regression. Predictors that were significant at the 0.10 level on univariate testing were further examined with adjustment for age, race (black versus non-black), diabetes, ESRD vintage, and SPPB score.
Before performing multivariable testing, missing covariate values (<5%) were multiply imputed while incorporating auxiliary variables reflecting factors for baseline dropout. This was done to better accommodate the assumption that missing data would be associated with variables used for imputation. SEs for the multivariable models corrected for the multiple imputation.
All statistical tests were two-tailed, with P < 0.05 taken to indicate significance. All analyses were conducted using SAS v9.1 (SAS Institute, Cary, NC).
Results
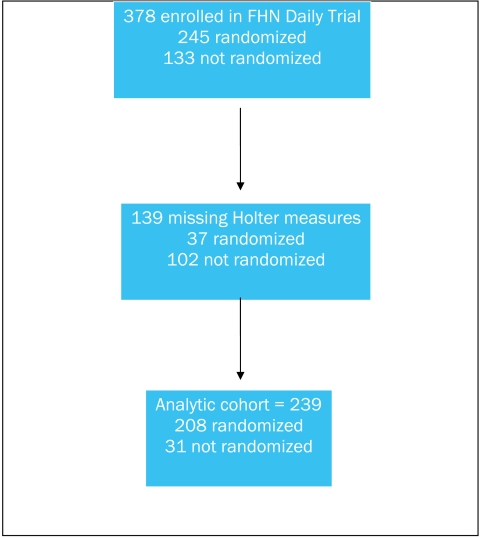
Our analytic cohort included 239 of the 378 (63%) enrolled subjects (Figure 1). Among the analytical cohort, 31 subjects did not qualify for randomization. Baseline characteristics of subjects who did and did not undergo randomization are summarized in Table 1. Some subjects who were excluded before randomization or otherwise voluntarily dropped from the study did not complete the Holter examination at baseline.
Figure 1.
Derivation of the analytic cohort.
Table 1.
Baseline characteristics
| Variable | Analytical Cohort (n = 239) | Randomized Patients (n = 208) | Patients Who Did Not Complete Baseline (n = 31) |
|---|---|---|---|
| Age (years) | 50.1 ± 13.6 | 50.1 ± 13.4 | 49.8 ± 15.0 |
| Female | 97 (41%) | 84 (40%) | 13 (42%) |
| Black race | 100 (42%) | 91 (44%) | 9 (29%) |
| ESRD vintage (years) | 3.16 (1.33; 6.48) | 3.63 (1.60; 6.93) | 1.68 (1.07; 2.54) |
| Diabetesa | 97 (42%) | 87 (42%) | 10 (40%) |
| Congestive heart failurea | 48 (21%) | 41 (20%) | 7 (28%) |
| COPDa | 13 (6%) | 10 (5%) | 3 (12%) |
| Past myocardial infarctiona | 30 (13%) | 25 (12%) | 5 (20%) |
| Peripheral vascular diseasea | 26 (11%) | 22 (11%) | 4 (16%) |
| Charlson comorbidity indexa | |||
| 0 | 93 (40%) | 83 (40%) | 10 (40%) |
| 1 | 40 (17%) | 37 (18%) | 3 (12%) |
| 2 | 40 (17%) | 38 (18%) | 2 (8%) |
| 3–4 | 42 (18%) | 36 (17%) | 6 (24%) |
| 5–6 | 14 (6%) | 12 (6%) | 2 (8%) |
| 7–12 | 4 (2%) | 2 (1%) | 2 (8%) |
| Beta blockera | 139 (60%) | 126 (61%) | 13 (50%) |
| Calcium channel blockera | 120 (51%) | 110 (53%) | 10 (38%) |
| Aspirina | 89 (38%) | 78 (38%) | 11 (42%) |
| Statins and/or other lipid lowering medicationsa | 83 (35%) | 76 (37%) | 7 (27%) |
Descriptive data are presented as mean ± SD or median and quartiles for normal and skewed population data, respectively. COPD, chronic obstructive pulmonary disease.
Missing data in five to six patients among the analytical cohort.
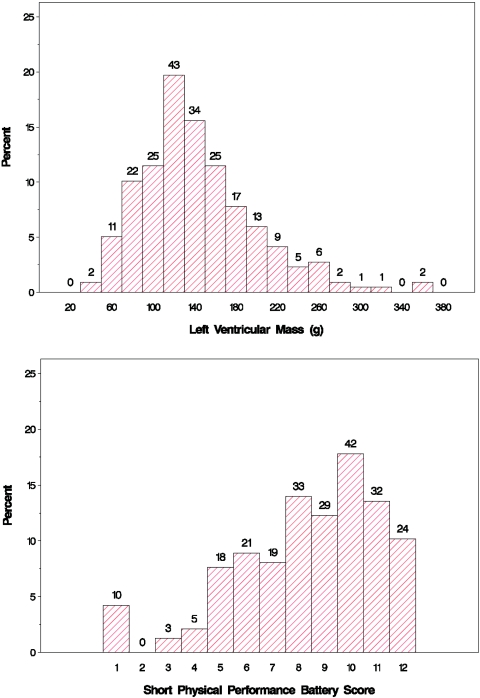
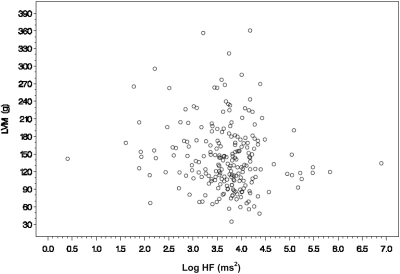
The mean LVM of the analytic cohort was 144 ± 56 g, and the mean SPPB score was 8.4 ± 2.7 (Table 2). The distribution of these outcome measures are shown in Figure 2. The mean R-R interval was 812 ± 217 ms, with the SDNN being 79.1 ± 40.3 ms. Spectral analysis showed that the median LF and HF harmonics were 106 (48.0; 204) and 42.4 (29.4; 56.3) ms2, respectively. LF/HF was 2.37 (1.43; 3.93). Diabetes, ESRD vintage, Charlson comorbidity index, left ventricular end-diastolic volume, plasma parathyroid hormone, phosphate, albumin, calcium, BP, and interdialytic weight gain were not associated with measures of heart rate variability. There were significant univariate associations between the mean R-R interval and age, race, LVM, hemoglobin concentration, SPPB, and the use of calcium channel blockers. Among the various indices of HRV, univariate associations were shown between log HF and LVM, log LF and the use of calcium channel blockers, SDNN and physical performance, and SDNN and age (Table 3). After adjusting for age, race, ESRD vintage, diabetes, and physical performance, the relation between LVM (per 10 g) and log HF remained statistically significant (−0.025 [−0.042; −0.0085]; P = 0.003; Figure 3). Similarly, after adjusting for the same base variables, the use of calcium channel blockers remained inversely related to log HF (−0.36 [−0.55; −0.18]; P = 0.001) and to log LF (−0.36 [−0.62, −0.11]; P = 0.0056).
Table 2.
Baseline clinical and laboratory values
| Variable | N Missing | Analytical Cohort (n = 239) | N Missing | Randomized Patients (n = 208) | N Missing | Patients Who Did Not Complete Baseline (n = 31) |
|---|---|---|---|---|---|---|
| Hemoglobin (g/dl) | 20 | 12.0 ± 1.3 | 9 | 12.0 ± 1.2 | 11 | 12.3 ± 1.7 |
| Plasma parathyroid hormone (pg/ml) | 14 | 316 (193; 562) | 3 | 313 (193; 565) | 11 | 359 (189; 559) |
| Plasma calcium (mEq/L) | 3 | 2.49 ± 0.25 | 2.50 ± 0.26 | 3 | 2.48 ± 0.09 | |
| Plasma phosphate (mg/dl) | 3 | 5.76 ± 1.62 | 5.79 ± 1.58 | 3 | 5.58 ± 1.88 | |
| Interdialytic weight gain (kg) | 7 | 3.41 ± 1.73 | 3.40 ± 1.66 | 7 | 3.49 ± 2.29 | |
| Albumin (g/dl) | 3 | 3.96 ± 0.41 | 3.96 ± 0.41 | 3 | 3.96 ± 0.35 | |
| Systolic BP (mmHg) | 3 | 148 ± 21 | 147 ± 21 | 3 | 152 ± 24 | |
| Diastolic BP (mmHg) | 3 | 80 ± 13 | 80 ± 13 | 3 | 79 ± 13 | |
| Left ventricular mass (g) | 21 | 144 ± 56 | 142 ± 55 | 21 | 175 ± 71 | |
| Left ventricular end diastolic volume (g) | 24 | 179 ± 61 | 178 ± 61 | 21 | 201 61 | |
| Short physical performance battery score | 3 | 8.4 ± 2.7 | 8.4 ± 2.7 | 3 | 8.4 ± 3.0 |
Descriptive data are presented as mean ± SD or median and quartiles for normal and skewed population data, respectively.
Figure 2.
Distributions of LVM and SPPB score.
Table 3.
Results of univariate associations
| A. Time domain analyses | ||||
|---|---|---|---|---|
| Variables | Mean RR interval (ms) Estimate (95% Confidence Interval) | P | SDNN (ms) Estimate (95% Confidence Interval) | P |
| Age (per decade) | 3.62 (−0.15, 7.38) | 0.059 | −2.05 (−3.07, −1.03) | 0.0001 |
| Black race (vs. non-black race) | −11.2 (−21.5, −0.85) | 0.034 | 2.29 (−0.6, 5.18) | 0.12 |
| Left ventricular mass (per 10 g) | −1.00 (−1.96, −0.032) | 0.043 | 0.09 (−0.18, 0.36) | 0.51 |
| Hemoglobin (g/dl) | 4.13 (0.059, 8.2) | 0.047 | −0.95 (−2.13, 0.23) | 0.11 |
| SPPB (per unit) | 1.98 (0.09, 3.87) | 0.041 | 0.58 (0.049, 1.10) | 0.032 |
| Calcium channel blocker | −12.9 (−23.2, −2.5) | 0.015 | 1.54 (−1.38, 4.45) | 0.30 |
| B. Frequency domain analyses | ||||||
|---|---|---|---|---|---|---|
| Variables | LogHF (95% Confidence Interval) | P | Log LF (95% Confidence Interval) | P | LF/HF (95% Confidence Interval) | P |
| Left ventricular mass (per 10 g) | −0.018 (−0.0035, −0.0043) | 0.045 | −0.017 (−0.041, 0.0067) | 0.16 | 0.00047 (−0.040, 0.041) | 0.98 |
| Calcium channel blocker | −0.35 (−0.53, −0.17) | 0.0002 | −0.37 (−0.62, −0.11) | 0.005 | 0.02 (−0.42, 0.46) | 0.93 |
SPPB, short physical performance battery score; SDNN, SD of RR intervals; HF, high-frequency harmonic.
Figure 3.
Scatterplot of LVM versus logHF.
Discussion
Left ventricular hypertrophy (LVH), poor physical performance, and sympathetic overactivity are known attributes of uremia (10–13). Our study adds to the literature by showing a potential mechanistic link among these three independent adverse clinical risk factors leading to excess cardiac morbidity and mortality in ESRD. In addition, our observations highlight that loss of vagal modulation of heart rate is an independent correlate of LVM after controlling for the effects of age, race, diabetes, ESRD vintage, and physical performance.
Sympathetic activation and vagal withdrawal have been associated with poor cardiovascular prognosis in patients with ESRD (14). Increased muscle sympathetic nerve activity has also been shown (15). Because patients undergoing hemodialysis have significantly higher sympathetic burst frequency than patients with bilateral nephrectomy, this finding has been attributed to an afferent signal arising from the failing kidney. There is now an emerging body of literature showing the independent contribution of elevated sympathetic activity and/or vagal withdrawal on LVH in ESRD (16). Norepinephrine is known to cause cardiomyocyte hypertrophy in vitro (17). Using an observational design, Zoccali et al. (10) examined the association between plasma norepinephrine and LVH in patients with ESRD. These investigators showed that 91 of 197 (46%) of their patients had plasma norepinephrine concentrations above the upper limit of the normal range. In addition, patients with the highest tertile in plasma norepinephrine concentrations were 3.8-fold more likely to have concentric LVH. Similarly, using Holter electrocardiography, Nishimura et al. (18) documented in a cohort of 154 patients with diabetes undergoing hemodialysis an inverse association between echocardiographically derived LVM index and HF harmonic (r2 = 0.08). These authors suggested that parasympathetic withdrawal in ESRD may be a complication of diabetic neuropathy leading to higher LVM. Zoccali et al. (19) also examined the clinical influence of parasympathetic control of the heart by studying respiratory sinus arrhythmia in 50 patients with ESRD. Once again, abnormal parasympathetic function was independently linked to concentric LVH as assessed by two-dimensional echocardiography. Our observations are consistent with the notion that cardiac autonomic dysfunction is associated with LVH in ESRD. In addition, given that CMRI was used to determine our cohort's LVM, it is reasonable to assume that our assessment of LVM was less likely to be confounded by various measurement errors such as excess extracellular fluid volume, a problem known to plague echocardiogram-derived estimates of LVM.
Studies of HRV and ESRD have consistently shown a decrement in HRV (2,20,21). Reduced HRV has been established as an independent risk factor in patients with high cardiac risk profile (22), which was exemplified by The Autonomic Tone and Reflexes After Myocardial Infarction (3) trial. This study enrolled 1284 patients with a recent (<28 day) myocardial infarction. Sympathetic overactivity was assessed by SDNN. During 21 months of follow-up, patients with SDNN <70 ms experienced a 3.2-fold increase in cardiac mortality. Consistent with this observation, other investigators examined the prognostic value of impaired HRV in ESRD patients. Oikawa et al. (23) found that SDNN of <75 ms was associated with a 3.64-fold increase in cardiovascular death among 383 patients on chronic hemodialysis. Nishimura et al. (24) found that 5-year survival was 98.1% in patients with LF/HF <1.9, whereas sudden cardiac death free survival rate was only 29% in patients with LF/HF >1.9.
Normal engagement of the sympathetic nervous system optimizes physical fitness and exercise performance. In health, the sympathetic nervous system is responsible for the increases in heart rate, stroke volume, and optimal distribution of blood flow to the working muscle (25–27). It is interesting to note that we showed a direct association between SDNN and physical performance in our analytical cohort, implying that patients who do not have a sustained increase in sympathetic modulation of heart rate have better physical performance scores. A tonic increase in cardiac sympathetic activity may lead to end-organ unresponsiveness. Indeed, previous investigators examined ß2-adrenoceptor densities and responsiveness in circulating lymphoctes among normotensive and hypotensive hemodialysis patients (28). As expected, these authors noted that there was an elevation in plasma norepinephrine concentrations in all patients with ESRD and in particular hypotensive patients on hemodialysis. However, the densities and responsiveness of adrenceptors were most reduced in those patients with the highest plasma norepinephrine concentrations. The authors suggested that a sustained increase in sympathetic activity may lead to downregulation of adrenceptors contributing to hemodynamic instability during hemodialysis. In this cohort, it is tempting to hypothesize that sustained elevation in cardiac sympathetic activity may lead to cardiac unresponsiveness, which may be associated with poor physical performance in ESRD.
This study has several important limitations. Although we hypothesized an association between structural cardiac abnormalities (by CMRI) and HRV, as well as other clinical factors with HRV, we tested multiple hypotheses because of the exploratory nature of the study. Therefore, it is possible that one or more of the significant associations we described may be “false positives.” We did not perform maneuvers other than Holter testing to assess for autonomic dysfunction. Although our sample was large relative to previous studies of HRV in ESRD, we had limited statistical power to detect associations, which are modest in magnitude, especially after multivariable adjustment. Because these analyses were cross-sectional, we could not determine whether the associations we found were causal. Longitudinal analyses of HRV in the FHN study subjects may provide additional insight into the factors associated with cardiac autonomic dysfunction and the extent to which HRV may be modifiable.
In summary, derived from a relatively large cohort of patients on hemodialysis, we showed associations among vagal modulation of heart rate, LVM, and physical performance. After controlling for various confounders, higher LVM remained significantly associated with the loss of vagal modulation of heart rate, as was impaired physical performance. Understanding the spectrum of autonomic heart rate modulation and its determinants may help to guide preventive and therapeutic strategies to reduce the burden of cardiovascular disease in this population.
Disclosures
None.
Acknowledgments
The authors were supported by grants from the National Institutes of Health, NIDDK, Centers for Medicare and Medicaid Services, Fresenius Medical Care Canada, and the Renal Research Institute. Listing of members of the FHN Daily Trial Group was previously published (4).
Footnotes
Published online ahead of print. Publication date available at www.cjasn.org.
References
- 1.Foley RN: Clinical epidemiology of cardiac disease in dialysis patients: Left ventricular hypertrophy, ischemic heart disease, and cardiac failure. Semin Dial 16: 111–117, 2003 [DOI] [PubMed] [Google Scholar]
- 2.Ranpuria R, Hall M, Chan CT, Unruh M: Heart rate variability (HRV) in kidney failure: Measurement and consequences of reduced HRV. Nephrol Dial Transplant 23: 444–449, 2008 [DOI] [PubMed] [Google Scholar]
- 3.La Rovere MT, Bigger JT, Jr, Marcus FI, Mortara A, Schwartz PJ: Baroreflex sensitivity and heart-rate variability in prediction of total cardiac mortality after myocardial infarction. ATRAMI (Autonomic Tone and Reflexes After Myocardial Infarction) Investigators. Lancet 351: 478–484, 1998 [DOI] [PubMed] [Google Scholar]
- 4.Suri RS, Garg AX, Chertow GM, Levin NW, Rocco MV, Greene T, Beck GJ, Gassman JJ, Eggers PW, Star RA, Ornt DB, Kliger AS: Frequent Hemodialysis Network (FHN) randomized trials: Study design. Kidney Int 71: 349–359, 2007 [DOI] [PubMed] [Google Scholar]
- 5.Greene T, Daugirdas JT, Depner TA, Gotch F, Kuhlman M: Solute clearances and fluid removal in the frequent hemodialysis network trials. Am J Kidney Dis 53: 835–844, 2009 [DOI] [PubMed] [Google Scholar]
- 6.Hemmelgarn BR, Manns BJ, Quan H, Ghali WA: Adapting the Charlson Comorbidity Index for use in patients with ESRD. Am J Kidney Dis 42: 125–132, 2003 [DOI] [PubMed] [Google Scholar]
- 7.Lorenzi-Filho G, Dajani HR, Leung RS, Floras JS, Bradley TD: Entrainment of blood pressure and heart rate oscillations by periodic breathing. Am J Respir Crit Care Med 159: 1147–1154, 1999 [DOI] [PubMed] [Google Scholar]
- 8.DuBois D, DuBois EF: Aformula to estimate the approximate surface area if height and weight be known. Arch Intern Med 17: 863–871, 1916 [Google Scholar]
- 9.Guralnik JM, Ferrucci L, Pieper CF, Leveille SG, Markides KS, Ostir GV, Studenski S, Berkman LF, Wallace RB: Lower extremity function and subsequent disability: consistency across studies, predictive models, and value of gait speed alone compared with the short physical performance battery. J Gerontol A Biol Sci Med Sci 55: M221–M231, 2000 [DOI] [PubMed] [Google Scholar]
- 10.Zoccali C, Mallamaci F, Tripepi G, Parlongo S, Cutrupi S, Benedetto FA, Cataliotti A, Malatino LS: Norepinephrine and concentric hypertrophy in patients with end-stage renal disease. Hypertension 40: 41–46, 2002 [DOI] [PubMed] [Google Scholar]
- 11.Zoccali C, Mallamaci F, Tripepi G: Traditional and emerging cardiovascular risk factors in end-stage renal disease. Kidney Int Suppl: 105–110, 2003 [DOI] [PubMed] [Google Scholar]
- 12.Johansen KL, Chertow GM, Ng AV, Mulligan K, Carey S, Schoenfeld PY, Kent-Braun JA: Physical activity levels in patients on hemodialysis and healthy sedentary controls. Kidney Int 57: 2564–2570, 2000 [DOI] [PubMed] [Google Scholar]
- 13.Johansen KL, Chertow GM, da Silva M, Carey S, Painter P: Determinants of physical performance in ambulatory patients on hemodialysis. Kidney Int 60: 1586–1591, 2001 [DOI] [PubMed] [Google Scholar]
- 14.Zoccali C, Mallamaci F, Parlongo S, Cutrupi S, Benedetto FA, Tripepi G, Bonanno G, Rapisarda F, Fatuzzo P, Seminara G, Cateliotti A, Stancanelli B, Malatino LS: Plasma norepinephrine predicts survival and incident cardiovascular events in patients with end-stage renal disease. Circulation 105: 1354–1359, 2002 [DOI] [PubMed] [Google Scholar]
- 15.Converse RL, Jr, Jacobsen TN, Toto RD, Jost CM, Cosentino F, Fouad-Tarazi F, Victor RG: Sympathetic overactivity in patients with chronic renal failure. N Engl J Med 327: 1912–1918, 1992 [DOI] [PubMed] [Google Scholar]
- 16.Schlaich MP, Socratous F, Hennebry S, Eikelis N, Lambert EA, Straznicky N, Esler MD, Lambert GW: Sympathetic activation in chronic renal failure. J Am Soc Nephrol 20: 933–939, 2009 [DOI] [PubMed] [Google Scholar]
- 17.Simpson P, McGrath A, Savion S: Myocyte hypertrophy in neonatal rat heart cultures and its regulation by serum and by catecholamines. Circ Res 51: 787–801, 1982 [DOI] [PubMed] [Google Scholar]
- 18.Nishimura M, Hashimoto T, Kobayashi H, Fukuda T, Okino K, Yamamoto N, Nakamura N, Yoshikawa T, Takahashi H, Ono T: Association between cardiovascular autonomic neuropathy and left ventricular hypertrophy in diabetic haemodialysis patients. Nephrol Dial Transplant 19: 2532–2538, 2004 [DOI] [PubMed] [Google Scholar]
- 19.Zoccali C, Mallamaci F, Tripepi G, Benedetto FA: Autonomic neuropathy is linked to nocturnal hypoxaemia and to concentric hypertrophy and remodelling in dialysis patients. Nephrol Dial Transplant 16: 70–77, 2001 [DOI] [PubMed] [Google Scholar]
- 20.Rubinger D, Revis N, Pollak A, Luria MH, Sapoznikov D: Predictors of haemodynamic instability and heart rate variability during haemodialysis. Nephrol Dial Transplant 19: 2053–2060, 2004 [DOI] [PubMed] [Google Scholar]
- 21.Rubinger D, Sapoznikov D, Pollak A, Popovtzer MM, Luria MH: Heart rate variability during chronic hemodialysis and after renal transplantation: Studies in patients without and with systemic amyloidosis. J Am Soc Nephrol 10: 1972–1981, 1999 [DOI] [PubMed] [Google Scholar]
- 22.La Rovere MT, Pinna GD, Hohnloser SH, Marcus FI, Mortara A, Nohara R, Bigger JT, Jr, Camm AJ, Schwartz PJ: Baroreflex sensitivity and heart rate variability in the identification of patients at risk for life-threatening arrhythmias: Implications for clinical trials. Circulation 103: 2072–2077, 2001 [DOI] [PubMed] [Google Scholar]
- 23.Oikawa K, Ishihara R, Maeda T, Yamaguchi K, Koike A, Kawaguchi H, Tabata Y, Murotani N, Itoh H: Prognostic value of heart rate variability in patients with renal failure on hemodialysis. Int J Cardiol 131: 370–377, 2009 [DOI] [PubMed] [Google Scholar]
- 24.Nishimura M, Tokoro T, Nishida M, Hashimoto T, Kobayashi H, Yamazaki S, Imai R, Okino K, Iwamoto N, Takahashi H, Ono T: Sympathetic overactivity and sudden cardiac death among hemodialysis patients with left ventricular hypertrophy. Int J Cardiol 142: 80–86, 2010 [DOI] [PubMed] [Google Scholar]
- 25.Seals DR, Monahan KD, Bell C, Tanaka H, Jones PP: The aging cardiovascular system: Changes in autonomic function at rest and in response to exercise. Int J Sport Nutr Exerc Metab 11[Suppl]: S189–S195, 2001 [DOI] [PubMed] [Google Scholar]
- 26.Seals DR, Taylor JA, Ng AV, Esler MD: Exercise and aging: autonomic control of the circulation. Med Sci Sports Exerc 26: 568–576, 1994 [PubMed] [Google Scholar]
- 27.Seals DR, Victor RG: Regulation of muscle sympathetic nerve activity during exercise in humans. Exerc Sport Sci Rev 19: 313–349, 1991 [PubMed] [Google Scholar]
- 28.Esforzado Armengol N, Cases Amenos A, Bono Illa M, Gaya Bertran J, Calls Ginesta J, Rivera Fillat F: Autonomic nervous system and adrenergic receptors in chronic hypotensive haemodialysis patients. Nephrol Dial Transplant 12: 939–944, 1997 [DOI] [PubMed] [Google Scholar]