Abstract
Respiratory syncytial virus (RSV), a negative-strand RNA virus, is the most common cause of epidemic respiratory disease in infants and young children. RSV infection of airway epithelial cells induces the expression of immune/inflammatory genes through the activation of a subset of transcription factors, including Nuclear Factor-κB (NF-κB). In this study we have investigated the role of the non canonical IκB kinase (IKK)ε in modulating RSV-induced NF-κB activation. Our results show that inhibition of IKKε activation results in significant impairment of viral-induced NF-κB-dependent gene expression, through a reduction in NF-κB transcriptional activity, without changes in nuclear translocation or DNA-binding activity. Absence of IKKε results in a significant decrease of RSV-induced NF-κB phosphorylation on serine 536, a post-translational modification important for RSV-induced NF-κB-dependent gene expression, known to regulate NF-κB transcriptional activity without affecting nuclear translocation. This study identifies a novel mechanism by which IKKε regulates viral-induced cellular signaling.
Keywords: RSV, airway epithelial cells, NF-κB, inflammation
INTRODUCTION
Respiratory syncytial virus (RSV) is an enveloped negative-sense single-strand RNA virus belonging to the Paramyxoviridae family. It is the leading cause of epidemic respiratory tract illness in children worldwide (Hall, 2001). RSV has also been shown to be a significant cause of respiratory illness among elderly and immunocompromised patients (Falsey, 2007;Falsey, Hennessey et al., 2005). RSV induces a broad spectrum of clinical diseases ranging from otitis media to mild upper respiratory infection, acute laryngeo-tracheo-bronchitis, or more severe lower respiratory tract infections, such as bronchiolitis and pneumonia. Several studies, both in vitro and in vivo, have suggested that the severity of RSV disease is the result of enhanced local inflammatory response and that pulmonary inflammation occurs in part through the ability of infected airway epithelial cells, the primary target of RSV infection, to synthesize chemokines, leading to the migration and activation of leucocytes at the site of infection [Reviewed in (Garofalo & Haeberle, 2000)].
NF-κB is a family of ubiquitous transcription factors involved in the inducible expression of viral, cytokine, adhesion molecules and acute-phase reactant genes (Osborn, Kunkel et al., 1989;Schreck, Rieber et al., 1991;Henkel, Machleidt et al., 1993;Karin, 1998). The Rel family proteins include NF-κB1 (p50), NF-κB2 (p52), Rel A (p65), Rel B and c-Rel. NF-κB activation is partly controlled by accessory inhibitory proteins called IκBs (Beg & Baldwin, 1993). NF-κB inducing stimuli control its cytoplasmic-nuclear abundance by a mechanism involving proteolytic degradation of the IκB inhibitor (Henkel, Machleidt et al., 1993;Karin, 1995). Once liberated, free cytoplasmic NF-κB passes through nuclear pores and enters the nucleus (translocates), to bind and activate target gene promoters. We have previously show that RSV is a potent activator of NF-κB in lung epithelial cells (Garofalo, Sabry et al., 1996; Jamaluddin, Casola et al., 1998; Casola, Garofalo et al., 2001). A number of RSV-inducible inflammatory and immunoregulatory genes require NF-κB for their transcription and/or are dependent on an intact NF-κB signaling pathway, as we have shown in vitro for IL-8 (Garofalo, Sabry et al., 1996), RANTES (Casola, Garofalo et al., 2001), as well as other chemokines, cytokine, secreted proteins and signaling molecules (Tian B, Zhang Y et al., 2002). RSV infection induces IκBα proteolysis (Jamaluddin, Casola et al., 1998), which leads to NF-κB nuclear translocation and DNA-binding, in part through activation of the canonical IKK complex (Casola A., unpublished data).
Optimal NF-κB activity also requires signal-induced phosphorylation. Several NF-κB family members, in particular p65, have been shown to be phosphorylated on specific serine residues, either constitutively or in an inducible manner, by stimuli like LPS, IL-1 and TNF (Zhong, Voll et al., 1998; Zhong, Suyang et al., 1997; Wang & Baldwin, Jr., 1998; Wang, Westerheide et al., 2000). This event is associated with an increase in p65 transcriptional activity, without modification of nuclear translocation or DNA-binding affinity (Zhong, Voll et al., 1998). Among the inducible serine phosphoacceptor sites that regulate NF-κB transcriptional activity, serines 276 (Ser276) and 536 (Ser536) have been shown to be potential targets for PKA and MSK1 (Zhong, Voll et al., 1998; Vermeulen, De Wilde et al., 2003) or casein kinase II and IKKβ kinases, respectively (Wang, Westerheide et al., 2000; Sakurai, Chiba et al., 1999).
The IKK-like molecule IKKε, together with TANK Binding Kinase (TBK)-1, has been identified as a critical component of the virus-activated kinas (VAK) complex responsible for IRF-3 activation (Fitzgerald, McWhirter et al., 2003; Sharma, tenOever et al., 2003). In addition to its role in IRF-3 activation, IKKε has also been shown to modulate NF-κB activation in response to LPS and PMA stimulation (Shimada, Kawai et al., 1999; Peters, Liao et al., 2000). Recently, IKKε has been shown to mediate p65 phosphorylation in response to IL-1 and TNF stimulation (Buss, Dorrie et al., 2004; Wietek, Cleaver et al., 2006), and to regulate constitutive NF-κB activity in cancer cells through a similar mechanism (Adli & Baldwin, 2006).
We have previously shown that RSV infection of airway epithelial cells induces IKKε expression and kinase activity, modulating IRF-3 activation and IRF-3-dependent gene expression (Indukuri, Castro et al., 2006). In this study, we investigated the role of IKKε in RSV-induced NF-κB activation and NF-κB-dependent gene expression. Our results show that IKKε regulates RSV-induced NF-κB gene transcription by affecting Ser536 p65 phosphorylation, which we show to be important for RSV-induced NF-κB-dependent gene expression, without modification of p65 nuclear translocation or DNA-binding activity, identifying a novel mechanism by which IKKε regulates viral-induced cellular signaling.
RESULTS
IKKε expression leads to enhanced NF-κB-dependent gene transcription
IKKε is a molecule initially cloned as an IKK-like kinase, which can activate NF-κB following certain inducers like LPS and PMA (Shimada, Kawai et al., 1999; Peters, Liao et al., 2000). To confirm that IKKε could modulate NF-κB-dependent gene transcription, we used 293 stable cell lines expressing wild type (WT) Flag-tagged IKKε, under the control of an inducible, tetracycline-regulated promoter. Cells transfected with the corresponding empty vector (EV) were used as a negative control. To induce IKKε expression, doxycycline was added to the culture medium and IKKε protein level was monitored by Western blot at different time points following tetracycline addition. IKKε expression was maximal 48 h after tetracycline treatment, shown in Fig.1A. There was no corresponding IKKε band in EV-transfected cells or in cells without tetracycline treatment (data not shown). Since NF-kB activation is absolutely required for viral- and cytokine-induced IL-8 gene expression (Garofalo, Sabry et al., 1996; Brasier, Jamaluddin et al., 1998), we compared IL-8 secretion in IKKε WT versus EV expressing cells. We found that IKKε WT expressing cells secreted significantly more IL-8 than EV cells (Fig. 1B), indicating that IKKε induces NF-κB-dependent gene expression. The role of IKKε in mediating IL-8 secretion was also confirmed at the transcription level. As shown in Fig. 1C, we found that luciferase activity driven by the human IL-8 promoter was significantly higher in IKKε-expressing cells than EV cells.
Fig. 1. Expression of IKKε induces IL-8 gene transcription.


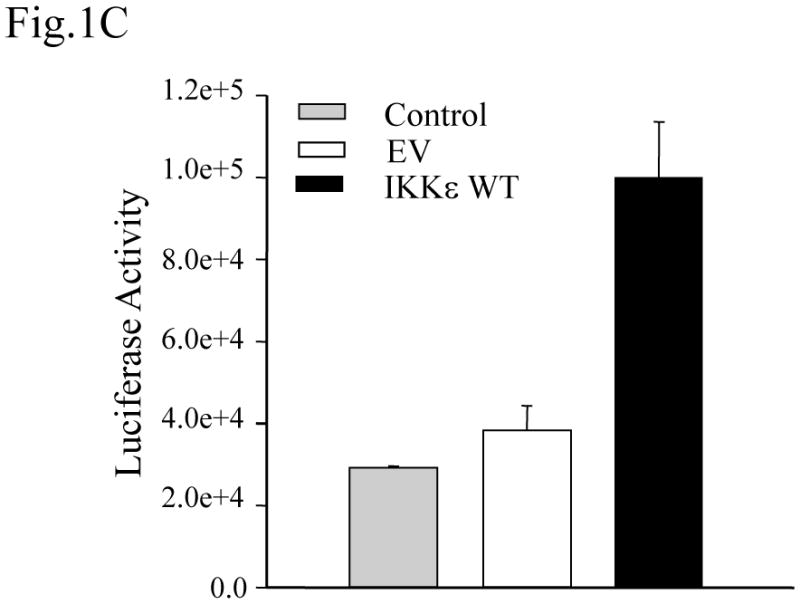
(A) Nuclear extracts were prepared from tetracycline-treated 293 cell lines expressing the empty vector (EV) or IKKε wild type (WT), fractionated on a 10% SDS-PAGE, transferred to PVDF membranes and probed with anti-Flag antibody. (B) 293 stable cell lines expressing EV or IKKε WT were treated with tetracycline for 48 h and infected with RSV for the indicated time points. Cell culture supernatants were assayed for IL-8 production by ELISA. Data are expressed as mean ± standard deviation of triplicate samples. (C) 293 stable cell lines expressing EV or IKKε WT were treated with tetracycline for 48 h and transfected with a plasmid containing the human IL-8 promoter linked to the luciferase gene. Cells were harvested 12 h after transfection to measure luciferase activity. For each plate luciferase was normalized to the β-galactosidase reporter activity. Data are expressed as mean ± standard deviation of normalized luciferase activity. Bars represent the mean value of triplicate samples of one representative experiment.
IKKε modulates RSV-induced NF-κB-dependent gene expression
To investigate the role of IKKε in RSV-induced NF-κB activation, we studied the effect of overexpressing catalytically inactive IKKε, also defines as dominant negative mutant (DN) (mutated on the lysine residue 38 of the catalytic domain by substitution with alanine) (Peters, Liao et al., 2000; Indukuri, Castro et al., 2006), on RSV-induced IL-8 gene expression. 293 stable cell lines expressing Flag-tagged IKKε DN, under the control of a tetracycline-regulated promoter, or a control cell line containing the empty vector (EV) were infected with RSV and harvested to measure IL-8 secretion. As shown in Fig. 2A, RSV infection induced significant amounts of IL-8 at 24 h post-infection (p.i) in EV cells, compared to mock infection (30 fold). However, the induction of IL-8 by RSV was much lower (10-fold) in IKKε DN expressing cells, suggesting that IKKε modulates RSV-induced NF- κB-dependent gene expression.
Fig. 2. IKKε modulates RSV-induced NF-κB transcriptional activity.



(A) 293 stable cell lines expressing the empty vector (EV) or a dominant negative (DN) mutant of IKKε were treated with tetracycline for 48 h and then infected with RSV. Culture supernatants, from uninfected and infected cells, were assayed 24 h later for IL-8 production by ELISA. Data are expressed as mean ± standard deviation of triplicate samples. *, P< 0.05 relative to RSV-infected EV-expressing cells. (B) 293 cells were transiently transfected with a plasmid containing the human IL-8 promoter linked to the luciferase gene and either a plasmid expressing IKKε DN or the empty vector (EV). Cells were infected with RSV and harvested at 12 h p.i. to measure luciferase activity. For each plate luciferase was normalized to the β-galactosidase reporter activity. Data are expressed as mean ± standard deviation of normalized luciferase activity. Bars represent the mean value of triplicate samples of one representative experiment. *, P< 0.05 relative to RSV-infected EV-expressing cells. (C) 293 cells were transiently transfected with a plasmid containing multimers of the IL-8 NF-κB sites and either the IKKε DN expressing plasmid or the empty vector (EV). Cells were infected with RSV and harvested at 12 h p.i. to measure luciferase activity, as described above. *, P< 0.05 relative to RSV-infected EV-expressing cells.
To determine whether the observed decrease in RSV-induced IL-8 production in IKKε DN expressing cells was due to inhibition of IL-8 gene transcription, 293 cells were co-transfected with a plasmid containing the human IL-8 promoter linked to the luciferase reporter gene and a plasmid encoding IKKε DN or its control vector. Cells were infected with RSV and harvested at 12 h p.i. to measure luciferase activity. Consistent with IL-8 secretion, RSV-induced IL-8 promoter activation was significantly lower in IKKε DN expressing cells, compared to EV cells (Fig. 2B). To determine whether IKKε affected RSV-induced IL-8 promoter activation through the NF-κB site, 293 cells were transiently co-transfected with a construct containing multiple copies of the IL-8 NF-κB site linked to luciferase reporter gene and either a plasmid expressing IKKε DN or the empty vector. Cells were infected with RSV and harvested at 12 h p.i. to measure luciferase activity. As shown in Fig.2C, RSV infection significantly enhanced NF-κB-dependent gene transcription, which was significantly inhibited by the expression of IKKε DN, but not the empty vector.
To confirm the role of f IKKε in RSV-induced NF-κB-dependent gene expression, we assessed IL-8 secretion in A549 cells transfected with either a scramble siRNA, as control, or one targeting IKKε and infected with RSV. Our results show that treatment of A549 cells with siRNA targeting IKKε effectively blocked IKKε gene and protein expression (both basal and inducible levels) (Fig.3A), as well as IL-8 production in response to RSV infection (Fig.3B). In addition to IL-8, secretion of several other cytokines and chemokines, whose expression is controlled by NF-κB (Schreck, Zorbas et al., 1990)(Martin, Cardarelli et al., 1997)(Zhang, Bryan et al., 2010), was significantly reduced in viral-infected cells treated with IKKε siRNA (Table I).
Fig. 3. Lack of IKKε affects RSV-induced NF-κB-dependent gene expression.




A549 cells were transfected with 100 nM siRNA targeting IKKε (siIKKε) or a scrambled control (siScr). A549 cells were mock infected or infected with RSV and harvested to prepare total RNA, total cell lysates or to collect cell supernatants. IKKε mRNA levels were measured at 24 h p.i. by Q-RT-PCR (A, left panel), IKKε protein levels were measured at 24 h p.i. by western blot (A, right panel), while IL-8 production was measured at various time points p.i. by ELISA (B). MEFs derived from wild type (WT) or IKKε−/− mice were infected with RSV and harvested at different times p.i. to measure KC production by ELISA. Data for ELISAs are expressed as mean ± standard deviation of triplicate samples (C). MEFs derived from WT or IKKε−/− mice were transiently transfected with a plasmid containing multimers of IL-8 NF-κB sites and either infected with RSV for 15 h or stimulated with 10 ng/ml of TNF-α for 6 h. Cells were harvested to measure luciferase activity (D).
Table 1.
Effect of IKKε silencing on RSV-induced chemokines and cytokines secretiona
| Gene symbol | SiScr mock | SiScr RSV | SiIKKε mock | SiIKKε RSV |
|---|---|---|---|---|
| TNF-α | 3.8±0.0 | 66±2.70 | 3.6±0.55 | 35±1.95 |
| MCP-1 | 95±5.21 | 528.79±13.15 | 72.51±3.30 | 185±5.55 |
| MIP-1β | 2.29±0.41 | 164±4.10 | 2.00±0.20 | 112.21±4.15 |
| MIP-1α | 1.31±1.31 | 80.57±4.13 | 1.49±1.50 | 50.17±4.05 |
| G-CSF | -- | 1441.41±26.20 | -- | 341.33±21.60 |
Supernatants from RSV-infected cells, treated with scramble (siScr) or IKKε (siIKKε) siRNA, were harvested at 24 h p.i. Chemokine and cytokine secretion was measured by Bio-Plex assay. Values are expressed as picograms/ml. Results are representative two separate experiments. Mock represent cells treated as the infected ones with the exception of viral inoculation.
Inhibition of IL-8 production was not due to inhibition of viral replication since viral replication was similar in scramble versus IKKε-targeting siRNA-treated cells (data not shown).
Similar results were found using cells obtained from an IKKε−/− mouse. MEFs were infected with RSV and harvested at various time p.i. to measure abundance of KC, a mouse homologue of human IL-8, whose expression is NF-κB dependent (Ohmori, Fukumoto et al., 1995). The results showed a significant reduction of RSV-induced KC in IKKε−/− MEFs compared to wild type (Fig.3C). To determine whether the observed decreased in RSV-induced KC production in IKKε−/− MEFS was due to inhibited NF-κB-dependent gene transcription, MEFS were transfected with a plasmid containing multimers of IL-8 promoter NF-κB site linked to the luciferase reporter gene and either infected with RSV or stimulated with TNF-α. Lack of IKKε expression blocked the induction of NF-κB-driven luciferase in response to both viral infection and cytokine stimulation (Fig.3D), confirming an important role of this kinase in NF- κB-dependent gene expression.
IKKε modulates NF-kB transcriptional activity by affecting p65 phosphorylation
Among the different members of the NF-κB family, we have previously shown that p65 is necessary for a variety of RSV-induced chemokine and cytokine gene expression (Tian B, Zhang Y et al., 2002). To investigate the mechanism(s) by which IKKε affects RSV-induced NF-κB-dependent gene transcription, RSV-induced p65 nuclear translocation was compared in A549 cells transfected with either a scramble siRNA, as control, or one targeting IKKε. Surprisingly, there was no difference in nuclear abundance of p65 between intact cells and cells in which IKKε expression was silenced (Fig. 4A). Similarly, there was no difference in RSV-induced DNA-binding to the IL-8 promoter NF-κB site between scramble and IKKε siRNA treated cells (data not shown). Interestingly, there was an increase in p65 nuclear levels in IKKe-treated cells versus the scramble-treated ones. A possible explanation of this finding could be a decrease in degradation, associated with a decrease in level of phosphorylation, as recently suggested for p65 undergoing phosphorylation on Ser276, which targets p65 for proteosomal degradation by ubiquitin-mediated proteolysis (Nihira, Ando et al., 2010).
Fig. 4. IKKε regulates RSV-induced p65 phosphorylation at Ser536.




A549 cells were transfected with 100 nM siRNA targeting IKKε (siIKKε) or a scrambled control (siScr). A549 cells were mock infected or infected with RSV and harvested to prepare nuclear extracts or total cell lysates. Nuclear translocation of p65 was assessed by Western blot. Membrane was stripped and reprobed with Lamin B to determine equal loading of the samples (A). Ser536 phosphorylation levels were detected by Western blot of total cell lysates. Membrane was stripped and reprobed for total p65 and β-actin to determine equal loading of the samples. Densitometric analysis of band intensity of Ser 536 p65 was performed using the histogram function of Adobe Photoshop. Results are shown after corrections to p65. Results are representative of two independent experiments (B). MEFs derived from wild type (WT) or IKKε−/− mice were infected with RSV and harvested to prepare nuclear extracts or total cell lysates. Nuclear translocation of p65 was assessed by Western blot. Membrane was stripped and reprobed with Lamin B to determine equal loading of the samples (C). Ser536 phosphorylation levels were detected by Western blot of total cell lysates. Membrane was stripped and reprobed for total p65 and β-actin. Densitometric analysis of band intensity of Ser536 p65 was performed using the histogram function of Adobe Photoshop. Results are shown after corrections to p65. Results are representative of two independent experiments (D).
NF-κB must undergo a variety of post-translational modification, including phosphorylation and acetylation, to achieve its full biological activity (Chen & Greene, 2004; Chen, Williams et al., 2005). Inducible phosphorylation on distinct serine residues, including Ser276 and Ser536, has been shown to regulate NF-κB transcriptional activity, without modification of nuclear translocation or DNA-binding activity (Zhong, Voll et al., 1998). To determine whether IKKε could modulate RSV-induced NF-κB-dependent gene transcription by affecting p65 post-translational modifications, we investigated p65 Ser536 and Ser276 phosphorylation in A549 cells in which IKKε expression was silenced. We observed significant less p65 Ser536 phosphorylation at 15 and 24 h p.i. in IKKε siRNA treated cells, compared to cells treated with scramble siRNA (Fig. 4B). On the other hand, there was no difference in Ser276 p65 phosphorylation in infected A549 cells treated with IKKε siRNA versus the scramble (data not shown), indicating that IKKε specifically affects RSV-induced p65 phosphorylation on Ser536.
Similar results were found using WT and IKKε−/− MEFs. As in airway epithelial cells, there was no reduction in RSV-induced p65 nuclear translocation in IKKε−/− MEF compared to WT cells (Fig.4C), however, there was a significant decrease in Ser536 p65 phosphorylation both at early and late time points of infection, although changes in phosphorylation between the WT and IKKε−/− cells showed a different kinetics compared to airway epithelial cells. Similar to airway epithelial cells, there was an increase in p65 nuclear levels in IKKε−/− MEF compared to WT cells.
The presence of residual phospho-Ser536 p65 levels in both siRNA-treated cells and MEFs suggested the presence of potential additional pathways involved in viral-induced Ser536 p65 phosphorylation. A very recent work from Yoboua et al. have shown that in airway epithelial cells initial RSV-induced p65 Ser536 phosphorylation is dependent on RIG-I, through a pathway involving MAVS, TRAF6 and the IKKβ kinase (Yoboua, Martel et al., 2010). To confirm whether IKKβ played a significant role in p65 Ser536 phosphorylation in our model of infection, A549 cells were transfected with either a scramble siRNA or one targeting IKKβ, infected with RSV and harvested at different times p.i. There was significant less RSV-induced p65 Ser536 phosphorylation in IKKβ siRNA treated cells, in particular at the basal level and at 6 h p.i., although there was some reduction also at 15 and 24 h p.i., compared to cells treated with scramble siRNA (supplementary Fig.S1). Overall, these data indicate that IKKε plays a significant role in p65 Ser536 phosphorylation, in particular at later time points of infection in the case of airway epithelial cells infected with RSV.
To test the functional consequences of p65 phosphorylation at Ser536 for NF-κB-dependent gene expression in the context of RSV infection, we compared the effect of overexpressing WT or a Ser536Ala mutant p65 on RSV-induced IL-8 reporter gene transcription. As shown in Fig.5A, IL-8 promoter activation in response to RSV infection was significantly enhanced by the expression of WT p65, but not by Ser536Ala p65.
Fig. 5. Ser536 is important for RSV-induced NF-κB-dependent gene expression.


A549 cells were transiently transfected with a plasmid containing the human IL-8 promoter linked to the luciferase gene and either a plasmid expressing WT p65 or the 536A mutant. Cells were infected with RSV and harvested at 12 h p.i. to measure luciferase activity. For each plate luciferase was normalized to the β-galactosidase reporter activity. Data are expressed as mean ± standard deviation of normalized luciferase activity*, P< 0.05 relative to RSV-infected cells (A). MEFs derived from p65−/− mice were transfected with a plasmid encoding p65 either wild type (WT) or mutated on serine 536 (Ser536Ala). Cells were either infected with RSV for 15 and 24 h or stimulated with TNF for 1 h and harvested to prepare total RNA. Gro-β gene expression was analyzed by Q-RT-PC (B).
To confirm a role of Ser536 in RSV-induced NF-κB-dependent gene expression, p65 −/− MEFs were stably transfected with either an empty vector or WT or Ser536Ala p65 expression plasmid and either infected with RSV or treated with TNF-α. Cells were harvested to extract total RNA and measure expression of Gro-β, a CXC chemokine we have shown to be NF-κB-dependent in the context of RSV infection and TNF-α treatment (Tian, Nowak et al., 2005; Tian B, Zhang Y et al., 2002). We found that expression of WT p65, but not Ser536Ala mutant, resulted in a 6.5 fold induction of basal Gro-β gene transcription in p65 −/− cells. In addition, Ser536Ala p65 transfected cells did not exhibit significantly enhanced Gro-β expression as WT p65 transfected cells did, both in the context of RSV infection and following treatment with TNF-α (Fig. 5), indicating that RSV-induced Ser536 p65 phosphorylation plays an important role in regulating NF-κB-dependent gene transcription. There was no basal or RSV-induced Gro-β gene transcription in cells transfected with the empty vector (data not shown).
DISCUSSION
RSV is the most common cause of epidemic respiratory infections in infants and young children and represents the prototype of mucosal-restricted viral pathogens with epithelial cell tropism and profound proinflammatory activity. RSV replication in airway epithelial cells results in the activation of multiple cellular signaling pathways involved in the expression of early response genes, which is coordinated by a small subset of transcription factors including NF-κB proteins. RSV infection induces NF-κB nuclear translocation by releasing it from the cytoplasmic inhibitors p100 and IκB-α (Jamaluddin, Casola et al., 1998; Liu, Li et al., 2008). However, this event is necessary but not sufficient for NF-κB activation, as several post-translational modifications, including phosphorylation and acetylation, are required for effective NF-κB transcriptional activity. We have recently shown that RSV infection of airway epithelial cells induces p65 phosphorylation on Ser276 and Ser536 and that mitogen- and stress-related kinase 1 (MSK1) is the kinase responsible for viral-induced p65 Ser276 phosphorylation (Jamaluddin, Tian et al., 2009). However, the pathway leading to RSV-induced p65 Ser536 phosphorylation has remained elusive.
Several kinases, including canonical and non-canonical IκB kinases and casein kinase II, have been shown to be able to phosphorylate p65. In this study, we show for the first time that IKKε, a kinase known to regulate IRF-3 activation (Sharma, tenOever et al., 2003; Fitzgerald, McWhirter et al., 2003), also regulates NF-κB transcriptional activity in response to RSV infection, by modulating p65 phosphorylation on Ser536. Expression of catalytically inactive IKKε significantly inhibited RSV-induced IL-8 secretion, promoter activation and NF-κB-driven gene transcription, indicating, for the first time, a fundamental role of this kinase in the pathway leading to RSV-induced NF-κB activation. This observation was confirmed using two independent approaches, the first was siRNA-mediated downregulation of IKKε and the second by using MEFs lacking IKKε expression. In both experimental settings, lack of IKKε significantly decreased NF-κB- dependent gene expression, without affecting p65 nuclear translocation and DNA-binding. On the other hand, the absence of IKKε greatly reduced p65 Ser536 phosphorylation, a post-translational modification important for RSV-induced NF-κB-dependent gene expression, as indicated by our reconstitution experiments using WT and Ser536Ala p65 mutant of p65−/− MEFs. In these cells, expression of WT p65, but not Ser536Ala p65 mutant, resulted in enhanced expression of both basal and RSV-inducible expression of Gro- β, an RSV-inducible NF-κB-dependent gene (Tian B, Zhang Y et al., 2002), indicating that phosphorylation of Ser536 plays an important role in RSV-induced NF-κB transcriptional activity. A similar result was recently reported by Fink et al., who showed that this p65 phosphorylation site is important in modulating NF-κB-dependent promoter activation in A549 cells infected with RSV (Fink, Duval et al., 2008).
Multiple functions of IKKε in controlling cellular signaling have been reported. Besides the well known role of IKKε, together with TANK-binding kinase -1 (TBK-1) in regulating IFN gene expression through direct phosphorylation of IRF-3(Fitzgerald, McWhirter et al., 2003; Sharma, tenOever et al., 2003), TenOever et al. have shown that IKKε phosphorylates signal transducer and activator of transcription 1 (STAT1), playing a critical role in specific IFN-inducible antiviral transcriptional responses (tenOever, Ng et al., 2007). Recent evidence has indicated that IKKε is involved in activation of different members of the NF-κB family. IKKε can control cRel nuclear accumulation via phosphorylation of its C-terminal domain (Harris, Oliere et al., 2006), and it promotes p52-dependent transactivation in TNF-stimulated cells (Wietek, Cleaver et al., 2006). In cancer cells, IKKε regulates constitutive NF-κB activity by phosphorylating p65 on Ser536. Similarly, Buss et al. have shown that IKKε is important for basal and IL-1-inducible phosphorylation of p65 on Ser536, and that this phosphorylation is necessary for p65 to be coupled to TATA-binding protein-associated factor II31 (Buss, Dorrie et al., 2004). This regulatory NF-κB activity is distinct from the canonical IKKα/β-mediated NF-κB activation, where IKKα/β phosphorylates IκBs, leading to degradation and subsequently release of NF-κB, which then migrates to the nucleus and activate gene transcription (Buss, Dorrie et al., 2004). Upon RSV infection, we have shown that p65 is released from the IκB complex and migrate into the nuclear compartment (Garofalo, Sabry et al., 1996; Jamaluddin, Casola et al., 1998), a process that is not IKKε-dependent, since in our study cells lacking IKKε expression showed a similar pattern of p65 nuclear translocation.
The presence of residual phospho-Ser536 p65 levels in both siRNA-treated cells and MEFs indicated the presence of potential additional pathways involved in viral-induced Ser536 p65 phosphorylation. Another kinase also involved in RSV-induced p65 Ser536 phosphorylation is IKKβ, as recently showed by Yoboua et al. and as confirmed in this study (Yoboua, Martel et al., 2010). At least in airway epithelial cells, IKKβ seems to play a bigger role in the initial viral-induced p65 phosphorylation, while IKKε is involved in later time points of infection.
In summary, this study highlights a new role of IKKε in modulating viral-induced NF-κB activation by affecting p65 phosphorylation and subsequent NF-κB-dependent gene expression. Identification of pathways controlling viral-induced NF-κB activation is important for the design of novel approaches to treat pulmonary inflammation in the context of RSV infection.
MATERIALS AND METHODS
RSV preparation
RSV A2 was grown in HEp-2 cells (American Type Culture Collection, Manassas, VA) and purified by polyethylene glycol precipitation, followed by centrifugation on 35 to 65% discontinuous sucrose gradients as described elsewhere (Olszewska-Pazdrak, Casola et al., 1998; Ueba, 1978). The virus titer was determined by methylcellulose plaque assay (Kisch & Johnson, 1963). Viral pools were aliquoted, quick-frozen on dry ice/alcohol and stored at −70°C until used.
Cell culture and infection of cells with RSV
A549, human alveolar type II-like epithelial cells, 293, a human embryonic kidney epithelial cell line (both from ATCC, Manassas, VA), and mouse embryonic fibroblasts (MEFs) were maintained in F12K, MEM and DMEM medium respectively, containing 10% (v/v) FBS, 10 mM glutamine, 100 IU/ml penicillin and 100 μg/ml streptomycin. Cell monolayers were infected with RSV at multiplicity of infection (MOI) of 3 (unless otherwise stated), as described (Garofalo, Sabry et al., 1996). An equivalent amount of a 30% sucrose solution was added to uninfected cells, as control.
Preparation of mouse embryonic fibroblasts (MEFs)
Mouse embryos from wild type and IKKε−/− mice (a generous gift from Millennium Pharmaceuticals Inc, Cambridge, Massachusetts) were harvested at 14–16 days gestation. After pulling each embryo from the uterus and removal of “excess” uterine tissue, embryos were pushed through a 1cc syringe with 18g needle attached and into a 15ml centrifuge tube for a number of times. Dissociated embryos were incubated with 0.5ml trypsin/embryo for 5–10 minutes at 37°C. After incubation, tissue larger clumps were broken by vigorous pipetting and plated at one embryo/100 mm dish, with DMEM containing 10% (v/v) FBS, 10 mM glutamine, 100 IU/ml penicillin and 100 μg/ml streptomycin (Hanada, Morishima et al., 1986). For all the experiments, cells were used at passage number 3 to 4.
Reporter gene assays
Logarithmically growing cells were co-transfected in triplicate with plasmids containing either the IL-8 promoter or multiple copies of the IL-8 NF-κB site, linked to the luciferase reporter gene, and a plasmid encoding IKKε either wild type or catalytically-inactive (K38A), using FuGene 6 (Roche), as previously described (Indukuri, Castro et al., 2006). Empty vector for IKKε was used as control. The next day, cells were infected with RSV and harvested at 12 or 24 h p.i. to measure luciferase and β-galactosidase reporter activity, as previously described (Casola, Burger et al., 2001). Luciferase was normalized to the internal control β-galactosidase activity. All experiments were performed in duplicate or triplicate.
RNA interference
Transfections of A549 cell with siRNA targeting IKKε or a scrambled negative control (Dharmacon, Lafayette, CO) were carried out at a final concentration of 100 nM by electroporation, using Nucleofector system (Amaxa, Gaithersburg, MD). 48 h later, A549 cells were mock infected or infected with RSV for various length of time and harvested to extract total RNA or to prepare nuclear extracts or total cell lysates.
Western blot
Nuclear extracts of uninfected and infected cells were prepared using hypotonic/nonionic detergent lysis, according to Schaffner protocol (Schreiber, Matthias et al., 1989). To prevent contamination with cytoplasmic proteins, isolated nuclei were purified by centrifugation through 1.7 M sucrose buffer for 30 minutes, at 12,000 rpm, before nuclear protein extraction, as previously described (Brasier, Spratt et al., 2004). Total cell lysates of uninfected and infected cells were prepared by adding ice-cold lysis buffer (50mM Tric-HCl, pH 7.4, 150mM NaCl, 1mM EGTA, 0.25% sodium deoxycholate, 1mM Na3VO4, 1mM NaF, 1% Triton X-100 and 1 μg/ml of aprotinin, leupeptin and pepstatin). After incubation on ice for 10 min, the lysates were collected and detergent insoluble materials were removed by centrifugation at 4°C at 14,000 g. After normalizing for protein content, using Bio-Rad Protein Assay kit (Bio-Rad, Hercules, CA), nuclear extracts or total cell lysates were fractionated by SDS-PAGE, and transferred to polyvinylidene difluoride membranes. Membranes were blocked with 5% milk in TBS-Tween and incubated with the proper primary antibodies according to manufacturer’s instruction. Antibodies anti-p65, anti-Ser536 or Ser276 p65, anti-IKKε and anti-IKKβ were purchased from Cell Signaling (Cell Signaling Technology, Inc, Danvers, MA). Following probing with HRP-secondary antibody, proteins were detected using an enhanced chemiluminescence system (Amersham Life Science, Piscataway, NJ) and visualized through autoradiography.
Quantitative Real Time Polymerase Chain Reaction (Q-RT-PCR)
Total RNA was extracted from A549 cells uninfected or infected with RSV for 24 h p.i. and either treated with siRNA targeting IKKε or a scrambled negative control, using the RNeasy Mini Kit (Qiagen, Valencia CA). RNA integrity was assessed by gel electrophoresis and quantitated with absorbance A260> 1.5. For gene transcript amplification by Q-RT-PCR, Taqman® Gene Expression Assay singleplex containing a 20 × mix of primers and FAM TaqMan® MGB probes for target genes and 18S rRNA VIC TaqMan® assay reagent were used (Applied Biosystems Foster City, CA). Separate tubes (singleplex) one-step RT-PCR was performed with 100ng RNA for both target genes and endogenous control. The cycling parameters using TaqMan® master mix one-step RT-PCR are as follows: reverse transcription 48°C for 30 min, AmpliTaq activation 95°C for 10 min, denaturation 95°C for 15 sec and annealing/extension 60°C for 1 min (repeat 30 times) on ABI7000. Duplicate CT values were analyzed in Microsoft Excel using the comparative CT (ΔΔCT) method as described by the manufacturer (Applied Biosystems Foster City, CA). The amount of target (2-Δ ΔCT) was obtained by normalizing to endogenous reference (18S) sample. Values are expressed as fold changes compared to uninfected, scrambled-treated cells.
ELISA
IL-8 and KC were quantitated by a double antibody ELISA kit (DuoSet, R&D Systems, Minneapolis, MN) following the manufacturer's instructions.
Bio-Plex
Chemokines and cytokines (IL-1RA, IL-1β, IL-6,, IL-9, IL-10, IL-12 p70, IL-13, IL-17, G-CSF, GM-CSF, IFN-γ, IP-10, EOTAXIN, MIP-1α, MIP-1β, GCSF, FGFB, PDGF, VEGF and TNF-α) were quantified by Luminex-based Bio-Plex system (Bio-Rad Laboratories, Hercules, CA) according to the manufacturer's instructions. The lower limit of detection for all cytokines measured by this assay is 3 pg/ml.
Statistical analysis
Statistical significance was analyzed by using analysis of variance (ANOVA). P value of less than 0.05 was considered significant. Results are shown as mean ± SEM.
Supplementary Material
Acknowledgments
This work was supported by grants NIEHS 06676 and NIAID P01 062885, American Heart Association (SDG 0835151N) and Parker Francis Foundation. The authors would like to thank Dr. Sha-Mei Liao (Millennium Pharmaceuticals Inc, Cambridge, Massachusetts) for the gift of the IKKε stable cell line and Cynthia Tribble for her assistance in the manuscript submission.
Footnotes
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Adli M, Baldwin AS. IKK-i/IKKepsilon controls constitutive, cancer cell-associated NF-kappaB activity via regulation of Ser-536 p65/RelA phosphorylation. Journal of Biological Chemistry. 2006;281:26976–26984. doi: 10.1074/jbc.M603133200. [DOI] [PubMed] [Google Scholar]
- Beg AA, Baldwin ASJ. The I kappa B proteins: multifunctional regulators of Rel/NF- kappa B transcription factors. Genes and Development. 1993;7:2064–2070. doi: 10.1101/gad.7.11.2064. [DOI] [PubMed] [Google Scholar]
- Brasier AR, Jamaluddin M, Casola A, Duan W, Shen Q, Garofalo RP. A promoter recruitment mechanism for tumor necrosis factor-alpha-induced interleukin-8 transcription in type II pulmonary epithelial cells. Dependence on nuclear abundance of Rel A, NF-kappaB1, and c-Rel transcription factors. Journal of Biological Chemistry. 1998;273:3551–3561. doi: 10.1074/jbc.273.6.3551. [DOI] [PubMed] [Google Scholar]
- Brasier AR, Spratt H, Wu Z, Boldogh I, Zhang Y, Garofal RP, Casola A, Pashmi J, Haag A, Luxon B, Kurosky A. Nuclear Heat Shock Response and Novel Nuclear Domain 10 Reogranization in Respiratory Syncytial Virus-Infected A549 Cells identified By High Resolution 2D Gel Electrophoresis. Journal of Virology. 2004;78:11461–11476. doi: 10.1128/JVI.78.21.11461-11476.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Buss H, Dorrie A, Schmitz ML, Hoffmann E, Resch K, Kracht M. Constitutive and interleukin-1-inducible phosphorylation of p65 NF-{kappa}B at serine 536 is mediated by multiple protein kinases including I{kappa}B kinase (IKK)-{alpha}, IKK{beta}, IKK{epsilon}, TRAF family member-associated (TANK)-binding kinase 1 (TBK1), and an unknown kinase and couples p65 to TATA-binding protein-associated factor II31-mediated interleukin-8 transcription. Journal of Biological Chemistry. 2004;279:55633–55643. doi: 10.1074/jbc.M409825200. [DOI] [PubMed] [Google Scholar]
- Casola A, Burger N, Liu T, Jamaluddin M, Brasier AR, Garofal RP. Oxidant tone regulates RANTES gene transcription in airway epithelial cells infected with Respiratory Syncytial Virus: role in viral-induced Interferon Regulatory Factor activation. J Biol Chem. 2001;276:19715–19722. doi: 10.1074/jbc.M101526200. [DOI] [PubMed] [Google Scholar]
- Casola A, Garofalo RP, Haeberle H, Elliott TF, Lin A, Jamaluddin M, Brasier AR. Multiple cis regulatory elements control RANTES promoter activity in alveolar epithelial cells infected with respiratory syncytial virus. Journal of Virology. 2001;75:6428–6439. doi: 10.1128/JVI.75.14.6428-6439.2001. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Chen LF, Greene WC. Shaping the nuclear action of NF-kappaB. Nat Rev Mol Cell Biol. 2004;5:392–401. doi: 10.1038/nrm1368. [DOI] [PubMed] [Google Scholar]
- Chen LF, Williams SA, Mu Y, Nakano H, Duerr JM, Buckbinder L, Greene WC. NF-kappaB RelA phosphorylation regulates RelA acetylation. Molecular & Cellular Biology. 2005;25:7966–7975. doi: 10.1128/MCB.25.18.7966-7975.2005. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Falsey AR. Respiratory syncytial virus infection in adults. Semin Respir Crit Care Med. 2007;28:171–181. doi: 10.1055/s-2007-976489. [DOI] [PubMed] [Google Scholar]
- Falsey AR, Hennessey PA, Formica MA, Cox C, Walsh EE. Respiratory syncytial virus infection in elderly and high-risk adults. N Engl J Med. 2005;352:1749–1759. doi: 10.1056/NEJMoa043951. [DOI] [PubMed] [Google Scholar]
- Fink K, Duval A, Martel A, Soucy-Faulkner A, Grandvaux N. Dual Role of NOX2 in Respiratory Syncytial Virus- and Sendai Virus-Induced Activation of NF-{kappa}B in Airway Epithelial Cells. Journal of Immunology. 2008;180:6911–6922. doi: 10.4049/jimmunol.180.10.6911. [DOI] [PubMed] [Google Scholar]
- Fitzgerald KA, McWhirter SM, Faia KL, Rowe DC, Latz E, Golenbock DT, Coyle AJ, Liao SM, Maniatis T. IKKepsilon and TBK1 are essential components of the IRF3 signaling pathway. Nat Immunol. 2003;4:491–496. doi: 10.1038/ni921. [DOI] [PubMed] [Google Scholar]
- Garofalo RP, Haeberle H. Epithelial regulation of innate immunity to respiratory syncytial virus. American Journal of Respiratory Cell and Molecular Biology. 2000;23:581–585. doi: 10.1165/ajrcmb.23.5.f204. [DOI] [PubMed] [Google Scholar]
- Garofalo RP, Sabry M, Jamaluddin M, Yu RK, Casola A, Ogra PL, Brasier AR. Transcriptional activation of the interleukin-8 gene by respiratory syncytial virus infection in alveolar epithelial cells: Nuclear translocation of the RelA transcription factor as a mechanism producing airway mucosal inflammation. Journal of Virology. 1996;70:8773–8781. doi: 10.1128/jvi.70.12.8773-8781.1996. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Hall CB. Respiratory syncytial virus and parainfluenza virus. N Engl J Med. 2001;344:1917–1928. doi: 10.1056/NEJM200106213442507. [DOI] [PubMed] [Google Scholar]
- Hanada N, Morishima T, Nishikawa K, Isomura S, Nagai Y. Interferon-mediated self-limiting growth of respiratory syncytial virus in mouse embryo cells. J Med Virol. 1986;20:363–370. doi: 10.1002/jmv.1890200409. [DOI] [PubMed] [Google Scholar]
- Harris J, Oliere S, Sharma S, Sun Q, Lin R, Hiscott J, Grandvaux N. Nuclear accumulation of cRel following C-terminal phosphorylation by TBK1/IKK epsilon. Journal of Immunology. 2006;177:2527–2535. doi: 10.4049/jimmunol.177.4.2527. [DOI] [PubMed] [Google Scholar]
- Henkel T, Machleidt T, Alkalay I, Kronke M, Ben-Neriah Y, Baeuerle PA. Rapid proteolysis of I kappa B-alpha is necessary for activation of transcription factor NF-kappa B. Nature (London) 1993;365:182–185. doi: 10.1038/365182a0. [DOI] [PubMed] [Google Scholar]
- Indukuri H, Castro SM, Liao SM, Feeney LA, Dorsch M, Coyle AJ, Garofalo RP, Brasier AR, Casola A. Ikkepsilon regulates viral-induced interferon regulatory factor-3 activation via a redox-sensitive pathway. Virology (New York NY) 2006;353:155–165. doi: 10.1016/j.virol.2006.05.022. [DOI] [PubMed] [Google Scholar]
- Jamaluddin M, Casola A, Garofalo RP, Han Y, Elliott T, Ogra PL, Brasier AR. The major component of IkBa proteolysis occurs independently of the proteasome pathway in Respiratory Syncytial Virus-infected pulmonary epithelial cells. Journal of Virology. 1998;72:4849–4857. doi: 10.1128/jvi.72.6.4849-4857.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jamaluddin M, Tian B, Boldogh I, Garofalo RP, Brasier AR. Respiratory syncytial virus infection induces a reactive oxygen species-MSK1-phospho-Ser-276 RelA pathway required for cytokine expression. J Virol. 2009;83:10605–10615. doi: 10.1128/JVI.01090-09. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Karin M. The regulation of AP-1 activity by mitogen-activated protein kinase. Journal of Biological Chemistry. 1995;270:16483–16486. doi: 10.1074/jbc.270.28.16483. [DOI] [PubMed] [Google Scholar]
- Karin M. The NF-kappa B activation pathway: its regulation and role in inflammation and cell survival. Cancer Journal From Scientific American. 1998;4(Suppl 1):S92–S99. [Review] [74 refs] [PubMed] [Google Scholar]
- Kisch AL, Johnson KM. A plaque assay for respiratory syncytial virus. Proceeding of Society for Experimental Biology and Medicine. 1963;112:583. doi: 10.3181/00379727-112-28111. [DOI] [PubMed] [Google Scholar]
- Liu P, Li K, Garofalo RP, Brasier AR. Respiratory syncytial virus induces RelA release from cytoplasmic 100-kDa NF-kappa B2 complexes via a novel retinoic acid-inducible gene-I{middle dot}NF- kappa B-inducing kinase signaling pathway. Journal of Biological Chemistry. 2008;283:23169–23178. doi: 10.1074/jbc.M802729200. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Martin T, Cardarelli PM, Parry GC, Felts KA, Cobb RR. Cytokine induction of monocyte chemoattractant protein-1 gene expression in human endothelial cells depends on the cooperative action of NF-kappa B and AP-1. Eur J Immunol. 1997;27:1091–1097. doi: 10.1002/eji.1830270508. [DOI] [PubMed] [Google Scholar]
- Nihira K, Ando Y, Yamaguchi T, Kagami Y, Miki Y, Yoshida K. Pim-1 controls NF-kappaB signalling by stabilizing RelA/p65. Cell Death Differ. 2010;17:689–698. doi: 10.1038/cdd.2009.174. [DOI] [PubMed] [Google Scholar]
- Ohmori Y, Fukumoto S, Hamilton TA. Two structurally distinct kappa B sequence motifs cooperatively control LPS-induced KC gene transcription in mouse macrophages. Journal of Immunology. 1995;155:3593–3600. [PubMed] [Google Scholar]
- Olszewska-Pazdrak B, Casola A, Saito T, Alam R, Crowe SE, Mei F, Ogra PL, Garofalo RP. Cell-specific expression of RANTES, MCP-1, and MIP-1alpha by lower airway epithelial cells and eosinophils infected with respiratory syncytial virus. Journal of Virology. 1998;72:4756–4764. doi: 10.1128/jvi.72.6.4756-4764.1998. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Osborn L, Kunkel S, Nabel GJ. Tumor necrosis factor alpha and interleukin 1 stimulate the human immunodeficiency virus enhancer by activation of the nuclear factor kappa B. Proceedings of National Academy Sciences, USA. 1989;86:2336–2340. doi: 10.1073/pnas.86.7.2336. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Peters RT, Liao SM, Maniatis T. IKKepsilon is part of a novel PMA-inducible IkappaB kinase complex. Mol Cell. 2000;5:513–522. doi: 10.1016/s1097-2765(00)80445-1. [DOI] [PubMed] [Google Scholar]
- Sakurai H, Chiba H, Miyoshi H, Sugita T, Toriumi W. IkappaB kinases phosphorylate NF-kappaB p65 subunit on serine 536 in the transactivation domain. J Biol Chem. 1999;274:30353–30356. doi: 10.1074/jbc.274.43.30353. [DOI] [PubMed] [Google Scholar]
- Schreck R, Rieber P, Baeuerle PA. Reactive oxygen intermediates as apparently widely used messengers in the activation of the NF-kappa B transcription factor and HIV-1. EMBO Journal. 1991;10:2247–2258. doi: 10.1002/j.1460-2075.1991.tb07761.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreck R, Zorbas H, Winnacker EL, Baeuerle PA. The NF-kappa B transcription factor induces DNA bending which is modulated by its 65-kD subunit. Nucleic Acids Research. 1990;18:6497–6502. doi: 10.1093/nar/18.22.6497. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Schreiber E, Matthias P, Muller MM, Schaffner W. Rapid detection of octamer binding proteins with 'mini-extracts', prepared from a small number of cells. Nucleic Acids Res. 1989;17:6419. doi: 10.1093/nar/17.15.6419. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sharma S, tenOever BR, Grandvaux N, Zhou GP, Lin R, Hiscott J. Triggering the interferon antiviral response through an IKK-related pathway. Science (Washington DC) 2003;300:1148–1151. doi: 10.1126/science.1081315. [DOI] [PubMed] [Google Scholar]
- Shimada T, Kawai T, Takeda K, Matsumoto M, Inoue J, Tatsumi Y, Kanamaru A, Akira S. IKK-i, a novel lipopolysaccharide-inducible kinase that is related to IkappaB kinases. International Immunology (Oxford) 1999;11:1357–1362. doi: 10.1093/intimm/11.8.1357. [DOI] [PubMed] [Google Scholar]
- tenOever BR, Ng SL, Chua MA, McWhirter SM, Garcia-Sastre A, Maniatis T. Multiple functions of the IKK-related kinase IKKepsilon in interferon-mediated antiviral immunity. Science (Washington DC) 2007;315:1274–1278. doi: 10.1126/science.1136567. [DOI] [PubMed] [Google Scholar]
- Tian B, Zhang Y, Luxon B, Garofalo RP, Casola A, Sinha M, Brasier AR. Identification of NF-kB dependent gene networks in respiratory syncytial virus-infected cells. Journal of Virology. 2002;76:6800–6814. doi: 10.1128/JVI.76.13.6800-6814.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tian B, Nowak DE, Jamaluddin M, Wang S, Brasier AR. Identification of direct genomic targets downstream of the nuclear factor-kappaB transcription factor mediating tumor necrosis factor signaling. Journal of Biological Chemistry. 2005;280:17435–17448. doi: 10.1074/jbc.M500437200. [DOI] [PubMed] [Google Scholar]
- Ueba O. Respiratory syncytial virus: I. concentration and purification of the infectious virus. Acta Medica Okayama. 1978;32:265–272. [PubMed] [Google Scholar]
- Vermeulen L, De Wilde G, Van Damme P, Vanden Berghe W, Haegeman G. Transcriptional activation of the NF-kappaB p65 subunit by mitogen- and stress-activated protein kinase-1 (MSK1) EMBO J. 2003;22:1313–1324. doi: 10.1093/emboj/cdg139. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Wang D, Baldwin AS., Jr Activation of nuclear factor-kappaB-dependent transcription by tumor necrosis factor-alpha is mediated through phosphorylation of RelA/p65 on serine 529. Journal of Biological Chemistry. 1998;273:29411–29416. doi: 10.1074/jbc.273.45.29411. [DOI] [PubMed] [Google Scholar]
- Wang D, Westerheide SD, Hanson JL, Baldwin AS., Jr Tumor necrosis factor alpha-induced phosphorylation of RelA/p65 on Ser529 is controlled by casein kinase II. J Biol Chem. 2000;275:32592–32597. doi: 10.1074/jbc.M001358200. [DOI] [PubMed] [Google Scholar]
- Wietek C, Cleaver CS, Ludbrook V, Wilde J, White J, Bell DJ, Lee M, Dickson M, Ray KP, O'Neill LA. IkappaB kinase epsilon interacts with p52 and promotes transactivation via p65. Journal of Biological Chemistry. 2006;281:34973–34981. doi: 10.1074/jbc.M607018200. [DOI] [PubMed] [Google Scholar]
- Yoboua F, Martel A, Duval A, Mukawera E, Grandvaux N. Respiratory-Syncytial Virus-mediated NF-kappaB p65 phosphorylation at Serine 536 is dependent on RIG-I, TRAF6 and IKKbeta. J Virol. 2010 doi: 10.1128/JVI.00142-10. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhang Z, Bryan JL, Delassus E, Chang LW, Liao W, Sandell LJ. CCAAT/enhancer-binding protein {beta} (C/EBP{beta}) and nuclear factor kappa B (NF-{kappa}B) mediate high level expression of chemokine genes CCL3 and CCL4 by human chondrocytes in response to interleukin-1{beta} (IL-1{beta}) Journal of Biological Chemistry. 2010 doi: 10.1074/jbc.M110.130377. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zhong H, Suyang H, Erdjument-Bromage H, Tempst P, Ghosh The transcriptional activity of NF-kappaB is regulated by the IkappaB-associated PKAc subunit through a cyclic AMP-independent mechanism. Cell (Cambridge MA) 1997;89:413–424. doi: 10.1016/s0092-8674(00)80222-6. [DOI] [PubMed] [Google Scholar]
- Zhong H, Voll RE, Ghosh S. Phosphorylation of NF-kappa B p65 by PKA stimulates transcriptional activity by promoting a novel bivalent interaction with the coactivator CBP/p300. Molecular Cell. 1998;1:661–671. doi: 10.1016/s1097-2765(00)80066-0. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.


