Abstract
Immunity against Campylobacter jejuni, a major food-borne pathogen causing diarrhea, is largely serotype specific. The major outer membrane protein (MOMP) of C. jejuni, PorA, is a common antigen with the potential to provide broad protection. Adult BALB/c mice were orally immunized with a recombinant glutathione S-transferase (GST) fused to PorA prepared from Campylobacter jejuni C31 (O:6,7) (GST-PorA) combined with a modified heat-labile enterotoxin of Escherichia coli as an adjuvant and later orally challenged with C31 strain or three heterologous strains: 48 (O:19), 75 (O:3), and 111 (O:1,44). Protection from colonization with the challenge organism was studied by fecal screening daily for 9 days. Serum and intestinal lavage fluid antibodies against the vaccine and Sarkosyl-purified MOMP from C31 were measured by using an enzyme-linked immunosorbent assay. The vaccine produced robust antibody responses against both antigens in serum and secretion. Since strain C31 was a poor colonizer, homologous protection could not be studied. The protective efficacies of heterologous strains were 43% (for strain 48, P < 0.001), 29% (for strain 75, P < 0.005), and 42% (for strain 111, P < 0.001) for the 9-day period compared to control mice given phosphate-buffered saline. Thus, PorA provided appreciable protection against colonization with heterologous serotypes.
Campylobacter jejuni is a food-borne pathogen and a leading cause of diarrhea worldwide (19, 33, 34). Campylobacteriosis is associated with a number of important sequelae, including Guillain-Barre syndrome, Reiter's syndrome, reactive arthritis, and irritable bowel syndrome (10, 37). C. jejuni also contributes to significant mortality of children in developing countries (18). In view of significant morbidity, mortality, and economic burden associated with C. jejuni infection, the control and prevention of campylobacteriosis are a priority. One possible tool for the prevention of campylobacteriosis is vaccination. It has been observed that C. jejuni infection results in the acquisition of immunity. However, immunity in humans seems to be serotype (Penner serotype) specific (13, 36). There are numerous serotypes of C. jejuni according to the Penner serotyping scheme, which is based on the lipopolysaccharide capsule (25). Therefore, vaccines based on common antigens that are shared by all serotypes seem attractive for broad protection. One such antigen is the major outer membrane protein (MOMP) of C. jejuni, PorA, which is present in abundant quantity in the organism. There are three conformational forms of MOMP: folded monomer (35 kDa), denatured monomer (45 kDa), and the native trimer (120 to 140 kDa). Only the folded monomer and the native trimer have pore-forming activities (44). The MOMP is involved in ion transport across the bacterial cell wall and adhesion of the bacterium to the intestinal mucosa (30, 41). It consists of 18 β-strands connected by short periplasmic turns and nine external loops. The external loops are antigenically variable (44). This is evident in the extreme genetic diversity of the porA gene encoding the MOMP (17, 44). However, the β-strands representing the conserved regions have common antigenic epitopes for different strains (16, 44). The sera of patients recovering from C. jejuni infection (8, 12, 35), as well as those of animals immunized with C. jejuni (21), possess antibodies to MOMP. Therefore, PorA seems to be an appropriate antigen that could be investigated as a possible subunit vaccine.
There are no easily handled, widely available, and inexpensive natural animal models of C. jejuni diarrhea. Laboratory mice are widely available, inexpensive, and easy to handle. Oral feeding of the adult mouse with C. jejuni does not result in diarrhea in the animal. However, the organism colonizes the intestine, with shedding of the organism for several days in the stool. The organism can be isolated from the blood as well (7, 9). Thus, the adult mouse can be used as a colonization model of C. jejuni infection.
Since the PorA of C. jejuni has never been investigated as a vaccine candidate, we studied a recombinant PorA in the form of a fusion protein in the adult mouse colonization model for broad protection against colonization. We studied fecal excretion of the challenge organisms and serum and intestinal antibody responses to the vaccine.
MATERIALS AND METHODS
Bacterial strains and growth conditions.
The C. jejuni C31 strain, isolated from a patient with bloody diarrhea in the United States (20), was donated by Richard L. Guerrant, University of Virginia School of Medicine, Charlottesville. The other three C. jejuni strains—48, 75, and 111—were isolated from diarrheal stools of patients treated at the Mubarak Al-Kabir Hospital, Kuwait, during 2000 to 2005. The organisms were grown on campylobacter-selective agar consisting of agar base (Oxoid, Basingstoke, Hampshire, England) supplemented with 7% defibrinated horse blood SR0050 (Oxoid), growth supplement SR0232 (Oxoid), and selective supplement SR0098E (Oxoid). The culture was incubated at 42°C for 48 h in an atmosphere of 85% N2, 10% CO2, and 5% O2 in a water-jacketed incubator (Nuaire, US Autoflow, Plymouth, MN). The organisms were confirmed by Gram staining, motility, and oxidase, catalase, and hippurate hydrolysis tests (31). Molecular confirmation of species was carried out by PCR as described previously (1). The molecular fingerprint of each C. jejuni strain was identified by flaA-restriction fragment length polymorphism (RFLP) described elsewhere (2). Each of the four strains had unique fingerprints. All strains were motile by spot inoculation by the Mueller-Hinton soft agar method (24).
Animals.
BALB/c female mice (6 to 8 weeks old) were obtained from the Animal Resources Centre, Health Sciences Centre, Kuwait University, and housed separately for 1 week prior to experimentation. The animals were fed a standard laboratory chow (Special Diet Food Services, Ltd., Essex, United Kingdom) and water ad libitum. Mice used in the present study were free from campylobacter infection. The studies were conducted according to institutional guidelines.
Serotyping.
The C. jejuni strains were serotyped by the Penner serotyping scheme by a passive hemagglutination test using a commercial kit (Denka Seiken Company, Ltd., Tokyo, Japan) according to the procedures recommended by the manufacturer.
Preparation of GST-PorA fusion protein of C. jejuni.
A recombinant clone, pGSTP7RIL, containing the porA gene, which encodes the MOMP, from the C31 strain was used (26). This clone expressed PorA as a fusion with glutathione S-transferase (GST). Urea-solubilized GST-PorA fusion protein was affinity purified by use of a glutathione-Sepharose B column. The identity of the protein was confirmed by immunoblotting with GST-specific antibody (26). The fusion protein was in denatured form (26). The protein concentration was estimated by using the Bio-Rad (Hercules, CA) protein assay.
Preparation of enriched MOMP (PorA).
The MOMP of C. jejuni strain C31 was enriched by the Sarkosyl method (11). Briefly, C. jejuni C31 was grown on blood agar at 42°C for 48 h in a microaerobic atmosphere as described previously. Bacterial cells from plates were harvested in 50 ml of sterile distilled water, centrifuged twice at 5,000 × g for 10 min at 25°C, and then suspended in 0.01 M Tris buffer (pH 7.4) (Sigma, St. Louis, MO). The cells were disrupted by sonication and centrifuged twice at 5,000 × g for 20 min to remove whole cells. The supernatant was centrifuged at 100,000 × g at 4°C for 1 h in an L8-70 ultracentrifuge (Beckman Instruments, Fullerton, CA). The pellet was suspended in 20 ml of 1% N-lauryl sarcocinate (Sigma) containing 7 mM EDTA (Sigma), incubated at 37°C for 20 min, and centrifuged at 100,000 × g for 2 h. The pellet was then suspended in 0.01 M Tris buffer (pH 7.4) (Sigma) and centrifuged at 100,000 × g for 2 h. The pellet produced was suspended in 1 ml of sterile distilled water and used as the Sarkosyl-enriched MOMP. The concentration of the protein was measured by the Bio-Rad method. The preparation was checked by immunoblotting as described previously (26) using rabbit antibody to GST-PorA fusion prepared previously (26) and then stored at −70°C.
Cross-reactivity of GST with C. jejuni.
The cross-reactivity of GST with C. jejuni proteins was checked by immunoblotting. The agar-grown C. jejuni was suspended in phosphate-buffered saline (PBS; pH 7.2) to a concentration of 109 CFU/ml and disrupted by sonication. The bacterial proteins were separated by discontinuous sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) with a 5% stacking gel and a 12.0% separating gel according to the method of Laemmli (27) and stained with Coomassie blue. For immunoblotting, the separated proteins were transferred electrophoretically onto nitrocellulose (Bio-Rad) and then blocked with 5% skim milk in PBS (pH 7.2). The membrane was reacted with goat GST antibody (Pharmacia Biotech, Piscataway, NJ) or normal goat serum control, as appropriate, both diluted to 1:5,000. The secondary antibody (peroxidase-conjugated anti-goat IgG raised in rabbit; Sigma) diluted to 1:5,000 was added, after which the gel was developed with enhanced chemiluminescence Western blotting detection reagents according to the instructions of the manufacturer (Amersham Pharmacia Biotech). GST-PorA fusion protein was used as a positive control.
Vaccination.
Vaccination of mice was carried out as described by Baqar et al. (7). Briefly, BALB/c mice were orally vaccinated twice at a weekly interval with the purified GST-PorA fusion protein combined with a modified heat-labile enterotoxin of Escherichia coli toxin, LT R192G, as an adjuvant (15) (provided by John D. Clements, Department of Microbiology and Immunology, Tulane University Medical Center, New Orleans, LA). Mice immunized with LT R192G or PBS (pH 7.2) alone were included as controls. Vaccination was done by using a stainless steel, curved-ball-tip feeding needle (20 gauge, 1.5-in. long; Popper and Sons, Inc., New Hyde Park, NY). Just prior to vaccination, gastric acidity was neutralized with two doses (0.5 ml each) of a 5% sodium bicarbonate (pH 8.5) solution given as an oral gavage at an interval of 15 min. An amount of 300 μg of the GST-PorA fusion protein mixed with 5 μg of LT R192G in a total volume of 300 μl in PBS (pH 7.2) was given to each of nine animals. Another group of mice was each fed with 5 μg of LT R192G and yet another group each fed with 1× PBS (pH 7.2). The mouse groups were immunized separately on different days for challenge with different C. jejuni strains.
Collection of intestinal lavage fluid.
Intestinal lavage fluid from individual mice was collected 7 days after the second vaccine dose according to the procedure described previously by Baqar et al. (7). Four doses of 0.5 ml of the lavage solution (25 mM NaCl, 40 mM Na2SO4, 10 mM KCl, 20 mM NaHCO3, 48.5 mM polyethylene glycol; Sigma) were given orally to the mice at 15-min intervals. At 30 min after administration of the last dose, the mice were given 0.1 ml of PBS (pH 7.2) containing 0.1 mg of pilocarpine (Sigma) intraperitoneally. The intestinal discharge was collected in a petri dish over the following 20 min. It was transferred to a 15-ml conical centrifuge tube, and the volume brought up to 5 ml with PBS (pH 7.2). The sample was vigorously vortexed and then centrifuged at 650 × g for 15 min. A total of 40 μl of phenylmethylsulfonyl fluoride (35 mg/ml in 95% ethanol; Sigma) and 200 μl of fetal bovine serum (Gibco/Invitrogen Life Technologies, Carlsbad, CA) were added to 4 ml of the supernatant. The protein concentration was determined by using the Bio-Rad method and adjusted to 1 mg/ml with PBS (pH 7.2) for samples from each mouse. The samples were stored at −70°C until used.
Collection of blood.
Blood was collected from individual mice 3 weeks after the second vaccine dose by snipping the tail as described previously (7). A 50-μl portion of blood was mixed with 950 μl of PBS (pH 7.2) containing 0.1% Tween 20 (PBS-T). After one cycle of freezing and thawing, the sample was centrifuged at 400 × g for 15 min, and the supernatant was stored at −70°C until used.
Challenge of vaccinated mice.
Mice were challenged with bacterial culture 3 weeks after the second vaccine dose, immediately after tail vein blood collection. A 48-h bacterial culture grown on campylobacter agar was suspended in PBS (pH 7.2) to a concentration of 2 × 109 CFU per ml, and 0.5 ml of the suspension was orally fed to the mice immediately after neutralization of the gastric acidity, as described above. Fecal shedding of the organism by each mouse was monitored for nine consecutive days according to the method described by Baqar et al. (7) with some modifications. Instead of a few fresh pellets of feces, all fecal pellets shed every day were collected by using a sterile stainless steel spoon and placed in a sterile 50-ml centrifuge tube. Feces was diluted to 10% with PBS (pH 7.2), followed by 5-fold serial dilutions. An aliquot of 50 μl of each dilution was spread onto campylobacter agar by using a sterile glass spreader. Plates were incubated at 42°C under microaerobic condition for 48 h and examined for the presence of C. jejuni colonies. C. jejuni colonies appeared as gray, moist colonies about 2 to 3 mm in diameter, which were easily distinguishable from normal fecal flora. The colonies were further confirmed by Gram staining, motility, and catalase and oxidase tests. The strain identity was confirmed by flaA-RFLP as described above.
Passage of C. jejuni strain C31 through mouse intestine.
To increase the colonization potential of C. jejuni C31 for mice, six BALB/c mice were orally inoculated with C. jejuni C31 as described in the preceding section. Feces was cultured on campylobacter agar every day for the next 4 days. Several colonies from each mouse were pooled and used as the seed culture for preparation of challenge inoculum for another 6 mice. This way, the strain was passaged four times. After each passage, the identity of the strain was monitored by flaA-RFLP (2).
Measurement of antibody responses.
C. jejuni-specific antibodies (IgA, IgG, and IgM isotypes combined) were measured in mouse intestinal lavage fluid and blood samples by enzyme-linked immunosorbent assay using the modified method of Baqar et al. (7). The wells of microtiter Maxisorp plates (Nunc, Rochester, NY) were coated with carbonate buffer (pH 9.6) containing either 1 μg of purified GST-PorA fusion protein or enriched PorA of C. jejuni C31 at 4°C for 24 h. The wells were washed four times with PBS (pH 7.2) containing 0.1% Tween 20 (PBS-T) and blocked with 5% milk powder (Regilait, Saint-Martin-Belle-Roche, France) in PBS (pH 7.2) at 37°C for 1 h. Serial doubling dilutions of lavage fluid or lysed blood samples in PBS-0.05% Tween 20 were added to the wells, followed by incubation at 37°C for 1 h. The wells were washed four times with PBS-T and then incubated with horseradish peroxidase-conjugated goat anti-mouse secondary antibodies (that recognize IgA, IgG, and IgM isotypes) (Kirkegaard & Perry Laboratories, Gaithersburg, MD) diluted 1:5,000 with 1× PBS (pH 7.2) at 37°C for 1 h. The wells were washed four times with PBS-T. The substrate 2,2′-azino-di-(3-ethyl-benzthiazoline sulfonate) (ABTS; Sigma) was added, and after 30 min of incubation at 37°C, the optical density at 405 nm was measured in a Powerwave Microplate spectrophotometer (Biotek Instruments, Inc., Bedfordshire, United Kingdom). Endpoint titers were expressed as the reciprocal of the dilution giving an absorbance value ≥2 standard deviations above the background absorbance in wells containing intestinal lavage fluid or lysed blood samples from normal mice (7).
Vaccine efficacy.
Vaccine efficacy was calculated as follows: ([rate of control mice − rate of vaccinated mice]/rate of control mice) × 100, where the rate is the percentage of mice free of C. jejuni colonization (5). Protection for the total duration of 9 days was calculated by taking into account the sum of vaccinated mice free of colonization for each of 9 days in comparison to similar data in the control group of mice.
Statistical analysis.
Statistical analysis was done by using SPSS version 17 (SPSS, Chicago, IL). Comparison of rates of mice free from colonization with C. jejuni was done by the Fisher exact test. The comparisons of endpoint antibody titers were done by Mann-Whitney test. Values differing with P ≤ 0.05 were considered statistically significant.
RESULTS
Serotypes of C. jejuni strains.
The Penner serotypes of C. jejuni strains were determined by passive hemagglutination test. Strain C31 belonged to serotype 6,7, strain 48 belonged to serotype 19, strain 75 belonged to serotype 3, and strain 111 belonged to serotype 1,44. Since the porA gene was cloned from strain C31 to make the GST-PorA fusion protein for vaccination, strain C31 was considered the homologous strain, and the other strains were considered heterologous strains.
GST-PorA fusion protein.
The recombinant porA gene clone pGSTP7RIL expressed a fusion protein of 76 kDa, as expected (26). The identity of the protein was confirmed by immunoblotting with GST-specific antibody (data not shown).
Sarkosyl-enriched MOMP (PorA).
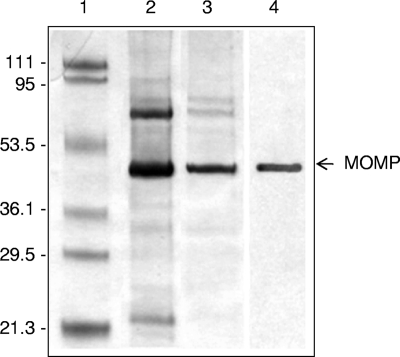
The Sarkosyl-enriched MOMP of strain C31 had a molecular mass of ∼48 kDa. Its identity was confirmed by immunoblotting with anti-GST-PorA antibody (Fig. 1).
FIG. 1.
Coomassie blue-stained SDS-12% PAGE gel after electrophoresis (lanes 1 to 3) and immunoblot gel of the purified MOMP from C. jejuni C31 (lane 4). Lane 1 shows molecular mass markers, lane 2 shows crude MOMP (∼48 kDa), and lane 3 shows Sarkosyl-purified MOMP. Lane 4 shows the Sarkosyl-purified MOMP detected using anti-GST-PorA antibody by immunoblotting. The picture of the Coomassie blue-stained gel containing lanes 1 to 3 and the picture of the immunoblot containing lane 4 were combined for the composite picture. The values on the left of the figure indicate the molecular masses (in kilodaltons) of standard markers, and the arrow on the right indicates the MOMP.
Cross-reactivity of GST with C. jejuni.
No cross-reactivity of GST with antigens from the four C. jejuni strains C31, 48, 75, and 111 was seen. However, the positive control produced a 76-kDa band, as expected (data not shown).
Colonization of C. jejuni in vaccinated and control mice and vaccine efficacy.
Reduced colonization in vaccinated mice on certain days, which achieved significance in comparison to the control mice, and colonization only for the 9-day period are shown below for each strain.
Strain C31.
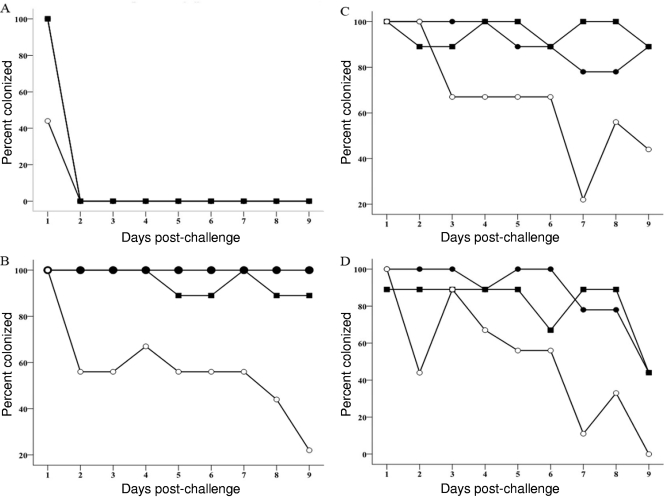
With the homologous strain C31, colonization was seen on day 1 in both the vaccinated and control groups of mice (Fig. 2A). Attempts at improving the colonization potential of the strain by repeated passage through mice did not improve colonization. The organism was not shed in the feces beyond the first day postinfection after each of the four passages. The percentage of mice colonized on day 1 in the vaccinated group was significantly reduced (P = 0.029 versus both PBS and LT R192G control groups), with a high vaccine efficacy (56%).
FIG. 2.
Percent colonization of mice which orally received PBS (•), LT R192G (▪), or GST-PorA + LT (○). Each group of mice (n = 9) was challenged with the homologous strain C. jejuni C31 (O:6,7) (A) or the heterologous strains C. jejuni 48 (O:19) (B), C. jejuni 75 (O:3) (C), and C. jejuni 111 (O:1,44) (D).
Strain 48.
This strain colonized vaccinated and control mice on all 9 days (Fig. 2B), but colonization was significantly reduced in the vaccinated group compared to the PBS group on days 8 (P = 0.029) and 9 (P = 0.009) and for the 9-day period (P = 0.001). Reduction was also significant against the LT control group on day 9 (P = 0.015) and for the 9-day period (P = 0.001). The vaccine efficacy was appreciable for days 8 (56%) and 9 (78%) and for the 9-day period (43%).
Strain 75.
This strain colonized the vaccinated and control mice throughout the 9 days, but a lower proportion of vaccinated mice experienced colonization (Fig. 2C). Reduction in colonization was significant for day 7 (P = 0.029 versus the PBS control group and P = 0.009 versus the LT control group) and for the 9-day period (P = 0.002 versus the PBS control group and P = 0.001 versus the LT control group). The vaccine efficacy for day 7 (72%) was greater than the efficacy for the 9-day period (29%).
Strain 111.
Vaccinated mice were colonized for 8 days and the control mice for all 9 days by this strain. A lower proportion of vaccinated mice than control mice was colonized (Fig. 2D). Reduction in colonization was significant on days 2 (P = 0.029 versus the PBS control group), 7 (P = 0.015 versus the PBS control group and P = 0.003 versus the LT control group), and 9 (P = 0.029 versus the PBS control group only) and for the 9-day period (P = 0.001 versus both the PBS and the LT control groups). The vaccine efficacies for these different days (56 to 100%) were higher than the efficacy for the 9-day period (42%).
Antibody response to vaccine.
Serum and intestinal lavage fluid samples from the PBS control group, the adjuvant control group, and the vaccinated group were used to estimate antibody responses. There were nine mice in each group that were randomly selected (Table 1). There was no antibody response in the PBS control group. Some antibody response in the LT control group was noticed. There was a higher antibody response in the vaccinated group. In general, the response in lavage fluid was higher than the response in serum. There was antibody response to both MOMP and GST-PorA, although the response to the latter was somewhat higher. The antibody response in the vaccinated group was in general significantly higher than the responses in the PBS and LT control groups.
TABLE 1.
Mean endpoint titers of antibody responses in sera and intestinal lavage fluid of vaccinated and control (PBS or LT R192G) mice (n = 9)
| Immunizing agent(s) | Endpoint titer (range)a |
|||
|---|---|---|---|---|
| Serum |
Intestinal lavage fluid |
|||
| Plates coated with MOMP | Plates coated with GST-PorA | Plates coated with MOMP | Plates coated with GST-PorA | |
| PBS | 0 (0-0) | 0 (0-0) | 0 (0-0) | 0 (0-0) |
| LT R192G | 0 (0-0) | 11 (0-100) | 20 (0-100) | 40 (0-100) |
| GST-PorA + LT R192G | 1,444 (200-3,200)* | 1,956 (800-3,200)* | 2,667 (800-3,200)†‡ | 3,200 (3,200-3,200)* |
*, P = 0.001 compared to the PBS and adjuvant control groups; †, P = 0.004 compared to the PBS control group; ‡, P = 0.024 compared to the adjuvant control group.
DISCUSSION
An effective vaccine could be based on antigens common to all strains, critical for bacterial metabolism or virulence and against which immunity can be elicited. Since PorA protein fulfills these criteria, we evaluated this antigen for its ability to provide homologous and heterologous protection. We calculated the protective efficacy of the vaccine by taking into consideration the ability of the vaccine to reduce colonization, as done by others (4, 6, 28, 29).
The PorA protein used as a vaccine in the present study was expressed from a previously cloned porA gene from strain C31 in fusion with GST protein (26). When we tested C31 in the current study, we found the strain to be a poor colonizer of the mouse intestine. Passage of the strain by oral inoculation of mice did not improve colonization. Motility is linked to colonization of chickens, the natural host of the organism (32). However, this may not correlate with the colonization of mouse intestine since C31 was motile and yet a mouse noncolonizer. It has been shown that the colonization potential of some strains increases by chicken passage (14, 24) but remains unchanged for others (39). We had to use C31 as the homologous strain since the vaccine was derived from this strain. Being a poor colonizer, the strain was excreted on the first day only, and the vaccine afforded significant protection against colonization on the first day. Protection on the first day was seen against the homologous challenge strain only and not against heterologous challenge strains. This shows that homologous protection is stronger than heterologous protection. Since there was no colonization beyond the first day for strain C31, we did not compute protection for the 9-day period for this homologous challenge strain. Had we used a better colonizing strain as the homologous strain, it is likely that we would have seen colonization of the strain for more days and higher protection for the 9-day period. However, to confirm homologous protection, studies have to be done with PorA derived from a better-colonizing strain.
With the heterologous strains, the vaccine afforded significant protection against colonization on certain days and for the total duration. The variability of protection on different days among the three strains can be attributed to their different colonizing abilities and/or differences in the amount of shared antigenic epitopes present in their PorAs.
We did not vaccinate mice with PorA alone, but vaccination was done with the mucosal adjuvant LT R192G, as was done with several candidate campylobacter vaccines to boost immune response (4, 6, 29). This mucosal adjuvant induces dendritic cell migration into the follicle-associated epithelium of Peyer's patches, thus promoting capture of the antigens by dendritic cells of the Peyer's patches (3). The adjuvant enhances Th1 responses and stimulates acquired immunity by activating the components of the innate immune system (40). However, control mice vaccinated with LT alone did not have protection.
Vaccinated mice responded with antibody production in the serum and intestinal secretion. We did not look for individual isotypes of antibodies. Since all three isotypes are present in the serum and the IgA isotype, along with small amounts of IgG and IgM isotypes, is present in the secretion, we assayed for the total antibodies representing all three isotypes. Since the antigen was delivered orally, the antibody response in the secretion was higher than in the serum. Since the vaccine is a combination of the PorA and the carrier protein, the antibody response would be expected to be directed against both components. To find out whether appreciable antibody production occurred against the PorA component, we tested the samples in parallel against the Sarkosyl-enriched MOMP as the antigen. This showed that most of the antibody was directed against MOMP.
In C. jejuni infection of chickens, the serum antibodies produced are predominantly against conformational epitopes and weakly against denatured (linear) epitopes of the MOMP. Also, rabbits immunized with conformational epitopes of MOMPs from different strains produced strain-specific antibodies, while antibodies induced by denatured MOMP were cross-reactive with MOMPs from different strains (21). At the same time, sera from naturally infected humans and from rabbits immunized with whole bacterial cells were found to have antibodies against a 45-kDa MOMP from C. jejuni strains, which is the denatured epitope of MOMP (12). Taken together, these data suggest that in natural C. jejuni infection, most of the antibodies produced are directed against conformational epitopes and some against linear epitopes of MOMP. Since the PorA vaccine used in our study is a denatured antigen, we believe that antibodies to this denatured antigen are providing heterologous protection. Boosting antibody response to linear epitopes (by use of denatured antigen) should improve heterologous protection. Comparative studies in which animals are immunized separately with the two different forms of antigen and then challenged with homologous and heterologous serotypes will clarify whether linear epitopes are involved in heterologous protection. With other organisms, both linear and conformational epitopes (45) or linear epitopes of OMPs (22) were found to impart protection.
We did not include control mice immunized with GST and measure antibodies to it in serum and intestinal lavage fluid. No such control group was needed since the four C. jejuni challenge strains did not cross-react with GST. Therefore, it can be concluded that antibodies to PorA, but not to GST, were responsible for the protection observed.
Different antigens have been evaluated as possible vaccines using different animal models for homologous and/or heterologous protection (reviewed in reference 23). However, vaccines evaluated in mice are pertinent to our own data. The vaccine candidates evaluated included killed whole cells (6), maltose-binding protein of Escherichia coli fused to flagellin protein (28), periplasmic binding protein (38), flagellum-secreted proteins (FlaC, FspA1, and FspA2) (4), conjugated capsular polysaccharides (29), and an adherence protein expressed on and delivered by an attenuated Salmonella enterica serovar Typhimurium strain (42). The mouse models included intranasal or intestinal colonization models. Vaccines were delivered intranasally, orally, or parenterally. The protection evaluated was homologous or both homologous and heterologous. The vaccines provided no or various degrees of protection. Further studies with some of these vaccines are required to assess their usefulness as serious candidates.
Ours is the first study in which the MOMP of C. jejuni in the form of a fusion protein has been evaluated as a vaccine in a mouse intestinal colonization model. Our data provide evidence that this vaccine can impart substantial heterologous protection. There is preliminary evidence to show that it could provide homologous protection as well. Our study was undertaken with a view to developing a vaccine for human use. There are several other issues that need to be addressed in the preparation of a PorA vaccine. For example, since bacterial porins can be posttranslationally modified, a recombinant protein needs to be compared to PorA enriched from bacteria to assess which constitutes a better vaccine.
It is pertinent to note that PorA antigen-based vaccines are in use against group B meningococcus. Since there is antigenic polymorphism in meningococcal PorA, many antigenic types need to be incorporated in a vaccine for optimal protection (43). Likewise, it is possible that protection can be enhanced by incorporating PorA from different strains of C. jejuni.
Acknowledgments
This study was funded by grant YM 06/07 from the College of Graduate Studies, Kuwait University, Kuwait. The use of some instruments in the Health Sciences Centre Core Facility (RCF grants GM01/01 and GM01/05), Kuwait University, is gratefully acknowledged.
We thank Sunny Ojoko of the Kuwait Animal Resource Centre, Kuwait University, for care of the animals.
Footnotes
Published ahead of print on 22 September 2010.
REFERENCES
- 1.Albert, M. J., S. Haridas, and B. Adler. 2008. Major outer membrane proteins from many Campylobacter species cross-react with cholera toxin. Clin. Vaccine Immunol. 15:859-862. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 2.Albert, M. J., E. Udo, B. T. Jose, S. Haridas, and V. O. Rotimi. 2009. Tetracycline resistance is frequent among Campylobacter jejuni isolates from Kuwait. Microb. Drug Res. 15:115-120. [DOI] [PubMed] [Google Scholar]
- 3.Anosova, N. G., S. Chabot, V. Shreedhar, J. A. Borawski, B. L. Dickinson, and M. R. Neutra. 2008. Cholera toxin, Escherichia coli heat-labile toxin, and non-toxic derivatives induce cell migration into the follicle-associated epithelium of Peyer's patches. Mucosal Immunol. 1:59-67. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 4.Baqar, S., L. A. Applebee, T. C. Gilliland, Jr., L. H. Lee, C. K. Porter, and P. Guerry. 2008. Immunogenicity and protective efficacy of recombinant Campylobacter jejuni flagellum-secreted proteins in mice. Infect. Immun. 76:3170-3175. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Baqar, S., A. L. Bourgeois, L. A. Applebee, A. S. Mourad, M. T. Kleinosky, Z. Mohran, and J. R. Murphy. 1996. Murine intranasal challenge model for the study of Campylobacter pathogenesis and immunity. Infect. Immun. 64:4933-4939. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Baqar, S., A. L. Bourgeois, P. J. Schultheiss, R. I. Walker, D. M. Rollins, R. L. Haberberger, and O. R. Pavlovskis. 1995. Safety and immunogenicity of a prototype oral whole-cell killed Campylobacter vaccine administered with a mucosal adjuvant in non-human primates. Vaccine 13:22-28. [DOI] [PubMed] [Google Scholar]
- 7.Baqar, S., N. D. Pacheco, and F. M. Rollwagen. 1993. Modulation of mucosal immunity against Campylobacter jejuni by orally administered cytokines. Antimicrob. Agents Chemother. 37:2688-2692. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Blaser, M. J., and D. J. Duncan. 1984. Human serum antibody response to Campylobacter jejuni infection as measured in an enzyme-linked immunosorbent assay. Infect. Immun. 44:292-298. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Blaser, M. J., D. J. Duncan, G. H. Warren, and W. L. Wang. 1983. Experimental Campylobacter jejuni infection of adult mice. Infect. Immun. 39:908-916. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 10.Blaser, M. J., and J. Engberg. 2008. Clinical aspects of Campylobacter jejuni and Campylobacter coli infections, p. 99-121. In I. Nachamkin, C. M. Szymanski, and M. J. Blaser (ed.), Campylobacter, 3rd ed. ASM Press, Washington, DC.
- 11.Blaser, M. J., J. A. Hopkins, R. M. Berka, M. L. Vasil, and W. L. Wang. 1983. Identification and characterization of Campylobacter jejuni outer membrane proteins. Infect. Immun. 42:276-284. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Blaser, M. J., J. A. Hopkins, and M. L. Vasil. 1984. Campylobacter jejuni outer membrane proteins are antigenic for humans. Infect. Immun. 43:986-993. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Burr, D. H., M. B. Caldwell, A. L. Bourgeois, H. R. Morgan, R. Wistar, Jr., and R. I. Walker. 1988. Mucosal and systemic immunity to Campylobacter jejuni in rabbits after gastric inoculation. Infect. Immun. 56:99-105. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 14.Cawthraw, S. A., T. M. Wassenaar, R. Ayling, and D. G. Newell. 1996. Increased colonization potential of Campylobacter jejuni strain 81116 after passage through chickens and its implication on the rate of transmission within flocks. Epidemiol. Infect. 117:213-215. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 15.Chong, C., M. Friberg, and J. D. Clements. 1998. LT(R192G), a non-toxic mutant of the heat-labile enterotoxin of Escherichia coli, elicits enhanced humoral and cellular immune responses associated with protection against lethal oral challenge with Salmonella spp. Vaccine 16:732-740. [DOI] [PubMed] [Google Scholar]
- 16.Clark, C. G., A. Beeston, L. Bryden, G. Wang, C. Barton, W. Cuff, M. W. Gilmour, and L. K. Ng. 2007. Phylogenetic relationships of Campylobacter jejuni based on porA sequences. Can. J. Microbiol. 53:27-38. [DOI] [PubMed] [Google Scholar]
- 17.Cody, A. J., M. J. C. Maiden, and K. E. Dingle. 2009. Genetic diversity and stability of the porA allele as a genetic marker in human Campylobacter infection. Microbiology 155:4145-4154. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Coker, A. O., R. D. Isokpehi, B. N. Thomas, K. O. Amisu, and C. L. Obi. 2002. Human campylobacteriosis in developing countries. Emerg. Infect. Dis. 8:237-243. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Friedman, C. R., J. Neimann, H. C. Wegener, and R. V. Tauxe. 2000. Epidemiology of Campylobacter jejuni infections in the United States and other industrialized nations, p. 121-138. In I. Nachamkin and M. J. Blaser (ed.), Campylobacter, 2nd ed. ASM Press, Washington, DC.
- 20.Guerrant, R. L., C. A. Wanke, R. A. Pennie, L. J. Barrett, A. A. Lima, and A. D. O'Brien. 1987. Production of a unique cytotoxin by Campylobacter jejuni. Infect. Immun. 55:2526-2530. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Huang, S., O. Sahin, and Q. Zhang. 2007. Infection-induced antibodies against the major outer membrane protein of Campylobacter jejuni mainly recognize conformational epitopes. FEMS Microbiol. Lett. 272:137-143. [DOI] [PubMed] [Google Scholar]
- 22.Hughes, E. E., and H. E. Gilleland, Jr. 1995. Ability of synthetic peptides representing epitopes of outer membrane protein F of Pseudomonas aeruginosa to afford protection against P. aeruginosa infection in a murine acute pneumonia model. Vaccine 13:1750-1753. [DOI] [PubMed] [Google Scholar]
- 23.Jagusztyn-Krynicka, E. K., P. Laniewski, and A. Wyszynska. 2009. Update on Campylobacter jejuni vaccine development for preventing human campylobacteriosis. Expert Rev. Vaccines 8:625-645. [DOI] [PubMed] [Google Scholar]
- 24.Jones, M. A., K. L. Marston, C. A. Woodall, D. J. Maskell, D. Linton, A. V. Karlyshev, N. Dorrell, B. W. Wren, and P. A. Barrow. 2004. Adaptation of Campylobacter jejuni NCTC11168 to high-level colonization of the avian gastrointestinal tract. Infect. Immun. 72:3769-3776. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 25.Karlyshev, A. V., D. Linton, N. A. Gregson, A. J. Lastovica, and B. W. Wren. 2000. Genetic and biochemical evidence of a Campylobacter jejuni capsular polysaccharide that accounts for Penner serotype specificity. Mol. Microbiol. 35:529-541. [DOI] [PubMed] [Google Scholar]
- 26.Khan, I., B. Adler, S. Haridas, and M. J. Albert. 2005. PorA protein of Campylobacter jejuni is not a cytotoxin mediating inflammatory diarrhoea. Microbes Infect. 7:853-859. [DOI] [PubMed] [Google Scholar]
- 27.Laemmli, U. K. 1970. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature 227:680-685. [DOI] [PubMed] [Google Scholar]
- 28.Lee, L. H., E. Burg III, S. Baqar, A. L. Bourgeois, D. H. Burr, C. P. Ewing, T. J. Trust, and P. Guerry. 1999. Evaluation of a truncated recombinant flagellin subunit vaccine against Campylobacter jejuni. Infect. Immun. 67:5799-5805. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 29.Monteiro, M. A., S. Baqar, E. R. Hall, Y. H. Chen, C. K. Porter, D. E. Bentzel, L. Applebee, and P. Guerry. 2009. Capsule polysaccharide conjugate vaccine against diarrheal disease caused by Campylobacter jejuni. Infect. Immun. 77:1128-1136. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 30.Moser, I., W. Schroeder, and J. Salnikow. 1997. Campylobacter jejuni major outer membrane protein and a 59-kDa protein are involved in binding to fibronectin and INT 407 cell membranes. FEMS Microbiol. Lett. 157:233-238. [DOI] [PubMed] [Google Scholar]
- 31.Murray, P. R., E. J. Baron, M. A. Pfaller, F. C. Tenover, and R. H. Yolken. 1999. Manual of clinical microbiology, 7th ed. ASM Press, Washington, DC.
- 32.Nachamkin, I., X. H. Yang, and N. J. Stern. 1993. Role of Campylobacter jejuni flagella as colonization factors for three-day-old chicks: analysis with flagellar mutants. Appl. Environ. Microbiol. 59:1269-1273. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 33.Newell, D. G., M. Koopmans, L. Verhoef, E. Duizer, A. Aidara-Kane, H. Sprong, M. Opsteegh, M. Langelaar, J. Threfall, F. Scheutz, J. van der Giessen, and H. Kruse. 2010. Food-borne diseases-The challenge of 20 years ago still persist while new ones continue to emerge. Int. J. Food Microbiol. 139(Suppl.):S3-S15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 34.Oberhelman, R. A., and D. N. Taylor. 2000. Campylobacter infections in developing countries, p. 139-154. In I. Nachamkin and M. J. Blaser (ed.), Campylobacter, 2nd ed. ASM Press, Washington, DC.
- 35.Panigrahi, P., G. Losonsky, L. J. DeTolla, and J. G. Morris, Jr. 1992. Human immune response to Campylobacter jejuni proteins expressed in vivo. Infect. Immun. 60:4938-4944. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 36.Pavlovskis, O. R., D. M. Rollins, R. L. Haberberger, Jr., A. E. Green, L. Habash, S. Strocko, and R. I. Walker. 1991. Significance of flagella in colonization resistance of rabbits immunized with Campylobacter spp. Infect. Immun. 59:2259-2264. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Peterson, M. C. 1994. Clinical aspects of Campylobacter jejuni infections in adults. West. J. Med. 161:148-152. [PMC free article] [PubMed] [Google Scholar]
- 38.Prokhorova, T. A., P. N. Nielsen, J. Petersen, T. Kofoed, J. S. Crawford, C. Morsczeck, A. Boysen, and P. Schrotz-King. 2006. Novel surface polypeptides of Campylobacter jejuni as traveller's diarrhoea vaccine candidates discovered by proteomics. Vaccine 24:6446-6455. [DOI] [PubMed] [Google Scholar]
- 39.Ringoir, D. D., and V. Korolik. 2003. Colonization phenotype and colonization potential differences in Campylobacter jejuni strains in chickens before and after passage in vivo. Vet. Microbiol. 92:225-235. [DOI] [PubMed] [Google Scholar]
- 40.Ryan, E. J., E. McNeela, M. Pizza, R. Rappouli, L. O'Neill, and K. H. G. Mills. 2000. Modulation of innate and acquired immune responses by Escherichia coli heat-labile toxin: distinct pro- and anti-inflammatory effects of the nontoxic AB complex and enzyme activity. J. Immunol. 165:5750-5759. [DOI] [PubMed] [Google Scholar]
- 41.Schroder, W., and I. Moser. 1997. Primary structure analysis and adhesion studies on the major outer membrane protein of Campylobacter jejuni. FEMS Microbiol. Lett. 150:141-147. [DOI] [PubMed] [Google Scholar]
- 42.Sizemore, D. R., B. Warner, J. Lawrence, A. Jones, and K. P. Killeen. 2006. Live, attenuated Salmonella typhimurium vectoring Campylobacter antigens. Vaccine 24:3793-3803. [DOI] [PubMed] [Google Scholar]
- 43.Taha, M. K., M. L. Zarantonelli, J. M. Alonso, L. M. Naess, J. Holst, B. Feiring, and E. Rosenqvist. 2007. Use of available outer membrane vesicles to control serogroup B meningococcal outbreaks. Vaccine 25:2537-2538. [DOI] [PubMed] [Google Scholar]
- 44.Zhang, Q., J. C. Meitzler, S. Huang, and T. Morishita. 2000. Sequence polymorphism, predicted secondary structures, and surface-exposed conformational epitopes of Campylobacter major outer membrane protein. Infect. Immun. 68:5679-5689. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 45.Zhang, Y. X., S. Stewart, T. Joseph, H. R. Taylor, and H. D. Caldwell. 1987. Protective monoclonal antibodies recognize epitopes located on the major outer membrane protein of Chlamydia trachomatis. J. Immunol. 138:575-581. [PubMed] [Google Scholar]