Abstract
The periaqueductal gray (PAG) consists of distinct columns that participate in the integrated control of autonomic function. We sought to determine whether the PAG is affected in multiple system atrophy (MSA), a disorder characterized by prominent autonomic failure. Brains were obtained at autopsy from 13 MSA patients (10 M, 3 F, age 61 ± 3 years) and 13 controls (8 M, 5 F, age 67 ± 4 years). Transverse formalin-fixed 50 μm sections were obtained throughout the PAG and immunostained for the vesicular transporter 2 (VGLUT-2), nitric oxide synthase (NOS), or α-synuclein and co-stained with thionin. Some sections were processed for myelin or astrocyte staining. Stereological quantitation was performed separately in the ventrolateral, lateral, dorsolateral, and dorsomedial columns of the PAG. In MSA cases, there was a decrease in the total estimated number of VGLUT-2 immunoreactive neurons in the ventrolateral, lateral, and dorsomedial and to a lesser extent dorsolateral PAG compared to controls (ventrolateral PAG: 16,299 ± 1,612 vs. 27,906 ± 2,480 respectively, p<0.01; lateral PAG: 11,004 ± 1,401 vs. 16,078 ± 1,140 respectively, p< 0.05; and dorsomedial PAG: 8,847 ± 1,052 vs. 15,412 ± 1,097 respectively, p< 0.001). The number of NOS immunoreactive neurons in the dorsolateral PAG was similar to controls. In all columns, the number of non-immunolabelled Nissl-stained cells was similar between groups. There was accumulation of glial cytoplasmic inclusions in all PAG columns in MSA. Our findings indicate involvement of the PAG columns in MSA, which may contribute to autonomic disturbances in this disorder.
Keywords: Central gray, VGLUT-2, nitric oxide synthase, glial cytoplasmic inclusions
Introduction
The periaqueductal gray (PAG) provides an anatomical and functional interface between the forebrain and the lower brain stem and has a major role in integrated autonomic responses to stress, pain modulation, and other adaptive functions. The PAG is subdivided into different longitudinal columns, ventrolateral, lateral, dorsolateral, and dorsomedial, which have different cortical and brainstem connections, and coordinate different types of behaviors. Different neuronal groups in the PAG participate in integrated cardiovascular responses associated with pain modulation, thermoregulation, respiration, airway control, micturition, arousal, and rapid eye movement (REM) sleep.
These functions involve extensive PAG interconnections with the prefrontal and anterior cingulate cortex, amygdala, hypothalamus, and brainstem nuclei involved in autonomic, motor, and pain modulatory responses. The PAG is affected in neurodegenerative disorders, such as Alzheimer disease and Parkinson disease.
Multiple system atrophy (MSA) is a neurodegenerative disorder characterized by prominent autonomic failure associated with parkinsonism, cerebellar ataxia, or both. Features of MSA include orthostatic hypotension, neurogenic bladder, respiratory disturbances, and disorders of arousal and REM sleep. The prominent role of the PAG in the control of cardiovascular, micturition, respiration and sleep, and its connections with brainstem areas that are neuropathologically affected in MSA, such as the rostral ventrolateral medulla and medullary raphe, suggest that PAG involvement may contribute to the clinical manifestations of this disorder. We have reported that there is loss of tyrosine hydroxylase (TH)-immunoreactive neurons in the ventrolateral PAG in MSA. Whereas TH cell loss may contribute to disorder of arousal, there is no clear evidence that these neurons are involved in cardiovascular, respiratory, or other functions affected in MSA. Many PAG neurons of the different columns projecting to brainstem autonomic and respiratory nuclei are glutamatergic, and neurons in the dorsolateral PAG, which do not project directly to brainstem autonomic regions, express nitric oxide synthase (NOS). The possible involvement of glutamatergic or nitroxidergic cell groups in the different PAG columns, and the relationship between neuronal involvement and the degree of accumulation of α-synuclein immunoreactive glial cytoplasmic inclusions (GCIs) and other neuropathological markers of MSA, such as astrocytic gliosis have not yet been explored. This information could provide insight into the pathodynamics and potential contributory anatomical substrates for the autonomic manifestations of MSA. To address these questions, we performed a stereological quantitative analysis of the glutamatergic and nitroxidergic cell groups as well as the burden of GCIs in the different functional columns of the PAG in patients with neuropathologically confirmed MSA.
Subjects and Methods
Brains were obtained at autopsy from 13 patients (10 men, 3 women; age 61 ± 3 years) with clinically probable MSA according to current criteria, and 13 controls with no history of neurologic disease (8 men and 5 women; age 67 ± 4years) (Table 1). All subjects had given informed consent for autopsy according to the guidelines of the Institutional Review Board. For the MSA cases, symptom duration was 7 ± 0.8 years; range (2–14); parkinsonism was the prominent finding in 8 (MSA-P) and cerebellar ataxia in 5 (MSA-C). All MSA cases had history of orthostatic hypotension and neurogenic bladder; and all men had erectile dysfunction. Seven cases had history of excessive diurnal somnolence; 7 had REM sleep behavior disorder; 3 had laryngeal stridor; 5 had documented sleep apnea; and 2 had periodic leg movements of sleep.
Table 1.
Patient Population
| Case | Age/Gender | PMD (hrs) | S×D (yrs) | Motor phenotype | Autonomic, manifestations | Stridor | Sleep disorder | Pathological diagnosis |
|---|---|---|---|---|---|---|---|---|
| C 1 | 51/M | 22 | N/A | None | None | N/A | None | CHF |
| C 2 | 57/F | 9 | N/A | None | None | N/A | None | Ovarian carcinoma |
| C 3 | 58/M | 9 | N/A | None | None | N/A | None | Lymphoma |
| C 4 | 64/F | 20 | N/A | None | None | N/A | None | COPD, Cardiac arrest |
| C 5 | 66/M | 22 | N/A | None | None | N/A | None | AAA |
| C 6 | 66/F | 16 | N/A | None | None | N/A | None | Cholestatic hepatitis |
| C 7 | 67/F | 15 | N/A | None | None | N/A | None | Gastrointestinal hemorraghe |
| C 8 | 67/M | 20 | N/A | None | None | N/A | None | Respiratory failure |
| C 9 | 68/M | 16 | N/A | None | None | N/A | None | Pulmonary fibrosis |
| C 10 | 73/M | 18 | N/A | None | Constipation | N/A | None | CHF |
| C 11 | 75/M | 18 | N/A | None | None | N/A | None | Renal disease |
| C 12 | 77/M | 11 | N/A | None | None | N/A | None | CHF |
| C 13 | 77/F | 22 | N/A | None | Constipation | N/A | None | Scleroderma, CAD |
| MSA 1 | 46/M | 17 | 5 | MSA-C | OH, NB, ED, Constipation, Anhidrosis | Yes* | EDS, OSA, PLMS, RBD * | MSA |
| MSA 2 | 46/M | 3 | 6 | MSA-C | OH, NB, ED, Dyspahgia, Anhidrosis | No | None | MSA |
| MSA 3 | 53/M | 10 | 6 | MSA-C | OH, NB, ED, Constipation, Anhidrosis | No | None | MSA |
| MSA 4 | 56/M | 10 | 12 | MSA-P | OH, NB, ED, Dysphagia, Anhidrosis | Yes* | EDS, OSA, RBD * | MSA |
| MSA 5 | 56/M | 12 | 14 | MSA-P | OH, NB, ED, Constipation, Anhidrosis | No | EDS | MSA |
| MSA 6 | 59/M | 26 | 5 | MSA-P | OH, NB, ED, Constipation, Anhidrosis | Yes* | EDS, OSA, RBD * | MSA |
| MSA 7 | 63/F | 11 | 6 | MSA-P | OH, NB, Dyspahgia, Anhidrosis | No | RBD | MSA |
| MSA 8 | 65/F | 10 | 7 | MSA-C | OH, NB, Constipation, Anhidrosis, | No | EDS, OSA, PLMS, RBD * | MSA |
| MSA 9 | 66/M | 5 | 4 | MSA-P | OH, NB, ED, Constipation, Anhidrosis, | No | None | MSA |
| MSA 10 | 67/M | 1 | 2 | MSA-P | OH, NB, Constipation | No | OSA | MSA |
| MSA 11 | 68/M | 23 | 14 | MSA-P | OH, NB, ED | No | RBD | MSA |
| MSA 12 | 70/M | 20 | 10 | MSA-C | OH, ED, Constipation, Anhidrosis | No | EDS, RBD | MSA |
| MSA 13 | 75/F | 8 | 4 | MSA-P | OH, NB, Dysphagia, Constipation | No | EDS | MSA |
AAA=abdominal aortic aneurysm, C=control, CAD=coronary artery disease, CHF= congestive heart failure, COPD= chronic obstructive pulmonary disorder, ED= erectile dysfunction, EDS=excessive diurnal somnolence, MSA=multiple system atrophy, MSA-C= multiple system atrophy cerebellar, MSA-P= multiple system atrophy parkinsonism, N/A=not applicable, NB=neurogenic bladder, OH=orthostatic hypotension, OSA=obstructive sleep apnea, PLMS=periodic leg movements sleep, PMD=post-mortem delay, RBD=REM behavior sleep disorder, S×D=symptom duration,
documented on polysomnogram
Tissue Processing and Immunocytochemistry
Post-mortem delay was similar in controls (17 ± 1 hour) and in MSA cases (12 ± 2 hours). One half of the brain was separated for routine neuropathological examination and the other half for our present study. All cases had neuropathological criteria for MSA. Of the 13 MSA cases, 10 had severe involvement of the putamen and substantia nigra pars compacta and 6 had moderate to severe involvement of the pons, inferior olivary nucleus, and cerebellum. All cases had moderate to severe GCI accumulation in these regions. One MSA case had mild to moderate and two had only rare associated Lewy body pathology. The presence of neurofibrillary tangles and senile plaques was classified in each case according to the stages of Alzheimer disease as described by Braak and Braak (B&B) and defined by the Consortium to Establish a Registry for Alzheimer ’s disease (CERAD). Twelve MSA cases had B&B staging 0-II, and one had stage III; and none fulfilled CERAD criteria for Alzheimer disease.
A block containing the midbrain from 33–47 mm rostral to the obex to include the whole extent of the PAG was separated, immersion fixed in 5% formalin for 24 hours at 4°C and cryoprotected in buffered 30% sucrose for 5 to 7 days prior to processing. Serial transverse 50 μm cryostat sections were obtained from one half of the midbrain and every eighth section was processed for immunoreactivity for vesicular glutamate transporter-2 (VGLUT-2, guinea pig polyclonal, 1:2000, Millipore, Temecula, CA.); nitric oxide synthase 1 (NOS1, rabbit polyclonal, 1:2000, Santa Cruz Biotechnologies, Santa Cruz, CA.); tyrosine hydroxylase (TH, mouse monoclonal, 1:3000, Immunostar, Hudson, WI.), or α-synuclein (goat polyclonal, 1:400, Santa Cruz Biotechnologies, Santa Cruz, CA). Diaminobenzidine/glucose oxidase solution with nickel enhancement (SIGMA, St. Louis, MO.) was used for the substrate reaction. Immunoreactive neurons were identified under bright-field illumination by a characteristic dark brown to black reaction product that densely fills the perikarya and their processes. Omission of the primary antibody, use of appropriate blocking peptide, or incubation with normal sera resulted in a lack of immunostaining. All sections were co-stained with thionin to identify surrounding structures and to determine whether loss of immunoreactivity reflected neuronal loss or lack of expression of the antigen. Selected paraffin embedded 12 um sections were processed for Luxol Fast Blue for myelin staining, or double immunofluorescence for α-synuclein (Santa Cruz Biotechnology Inc., Santa Cruz, CA) which incorporated a fluorescein-conjugated (donkey anti-rabbit IgG, 1:200, Millipore, Temecula, CA), and glial fibrillary acidic protein (GFAP) (rabbit polyclonal, 1:400, Dako, Glostrup, Denmark) followed by a Cy-3 conjugated (donkey anti rabbit IgG, 1:200, MIllipore, Temecula, CA).
Image analysis and quantitation
For each MSA and control case, we analyzed between 14–18 sections obtained 800 μm apart to span the length of the PAG. The sections were examined under bright-field microscopy using a modified light microscope (Zeiss Axioimager A-1; Zeiss, Thornwood, NY) equipped with a motorized specimen stage for automated sampling (Ludl Electronics; Hawthorne, NY, USA), CCD color video camera (Microfire; Optronics, Goleta, CA, USA) and stereology software (Stereo Investigator, v9.2; MBF Bioscience, Williston, VT, USA). Cell counts were performed separately in the ventrolateral, lateral, dorsolateral, and dorsomedial PAG defined according to the atlas of Paxinos and Huang. In each of the columns, we counted separately the VGLUT-2, TH, and NOS immunoreactive neurons as well as, and thionin stained cells that were not immunoreactive for any of the markers. We also counted separately the number of GCIs in each column. Stereological analysis of the total number of neurons or GCIs was performed using the optical fractionator method. All systematic unbiased random samples in each section were analyzed according to established criteria. In addition to the total estimated numbers, the density of neurons or GCIs in each PAG column was calculated using the Cavalieri volumetric method. The same investigator, blinded to the clinical and final neuropathological diagnoses, performed all counts.
Statistical analysis
All results are expressed as mean ± SEM. Statistical analysis was performed using the software SPSS 14.0 for Windows. Multivariate analysis of variance was used to determine if age, gender, disease phenotype, symptom duration, or post-mortem delay had any effect on cell counts. For cell counts, density and sizes in each PAG column, comparison of control vs. MSA cases was performed using unpaired t-test. Comparison of the density of GCIs among the different columns was made using univariate analysis of variance. Post-hoc analysis was then conducted using Dunnett’s and Bonferroni’s formulas. A p value of <0.05 was considered significant.
Results
Estimated total number of neurons in the different PAG columns in MSA compared to controls
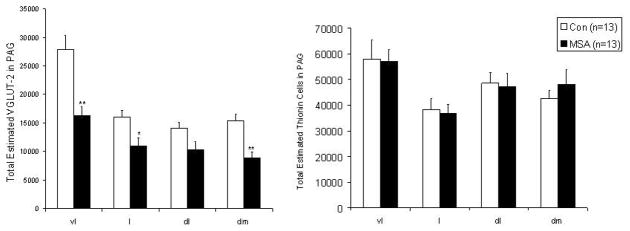
The distribution and morphology of VGLUT-2 and NOS immunoreactive neurons in the ventrolateral and dorsolateral PAG, respectively, was consistent with observations in experimental animals (Figure 1). Compared to controls, there was loss of VGLUT-2 immunoreactive neurons in the ventrolateral, lateral, and dorsomedial and to a lesser extent dorsolateral column of the PAG. Cell loss in the ventrolateral and dorsomedial PAG was relatively more severe than in the lateral PAG. There was a mild VGLUT-2 cell loss in the dorsolateral PAG compared to other columns (Table 2). For the ventrolateral PAG the total estimated cell number was 16,299 ± 1,612 in MSA and 27,906 ± 2,480 in controls (p<0.01); for the lateral PAG, it was 11,004 ± 1,401 in MSA and 16,078 ± 1,140 in controls (p< 0.05); and in the dorsomedial PAG it was 8,847 ± 1,052 in MSA and 15,412 ± 1,097 in controls (p< 0.001). The degree of VGLUT-2 cell loss did not correlate with symptom duration, age, sex, disease phenotype (MSA-P or MSA-C), presence or absence of laryngeal stridor, or post-mortem delay. In contrast, there was no significant difference in the number of NOS immunoreactive cells in the dorsolateral PAG compared to controls (Table 2) (Figure 1). Consistent with our previous study, there was also a significant reduction in the numbers of TH immunoreactive cells in the ventrolateral PAG in MSA compared to controls (Table 2). The loss of VGLUT-2 or TH-immunoreactivity could not be explained by down-regulation of the marker, as assessed using Nissl staining. The total estimated numbers and cell density of Nissl-stained, non VGLUT-2, non-NOS, non-TH immunoreactive neurons was similar between MSA and controls in each of the PAG columns (Figure 2). There was no difference in cell size between MSA and controls in any of the columns (Table 2).
Figure 1.
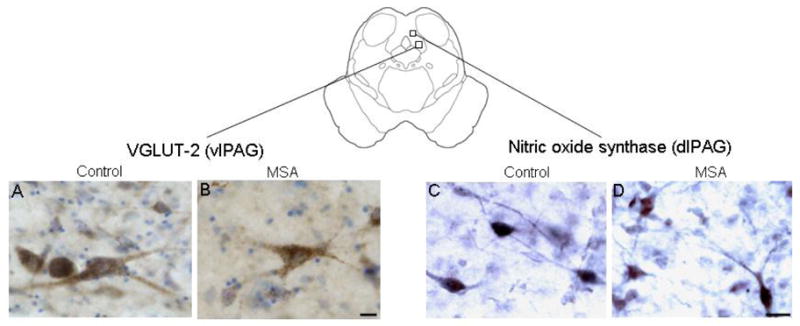
Representative 50 μm transverse hemisection at the level of the periaqueductal gray (PAG) showing the characteristics of vesicular glutamate transporter-2 (VGLUT-2) immunoreactive neurons in the ventrolateral (vl) column) (left panel), and nitric oxide synthase (NOS) immunoreactive neurons in the dorsolateral (dl) column in a 75 year-old man with no neurological history (A,C, post mortem delay 18 hrs), and in a 65 year-old woman with multiple system atrophy (B,D, post mortem delay 10 hrs). There was loss of VGLUT-2 immunoreactive neurons in the vlPAG but preservation of NOS neurons in the dlPAG in the MSA compared to the control case. Bar = 25μm.
Table 2.
Cell counts, density and sizes in the different columns of the PAG
| Column | Cell type | Variable | Control (n=13) (mean±SEM) | MSA (n=13) (mean±SEM) |
|---|---|---|---|---|
| Ventrolateral | VGLUT-2 | Total estimated number | 27,906 ± 2,480 | 16,299 ± 1,612 |
| Density/mm3 | 1,413 ± 59 | 836 ± 114 | ||
| Size (μm2) (n=100) | 349 ± 18 | 323 ± 19 | ||
| TH | Total estimated number | 21,895 ± 2,248 | 11,348 ± 1,634 | |
| Density/mm3 | 1,006 ± 147 | 523 ± 72 | ||
| Size (μm2) (n=100) | 617 ± 61 | 709 ± 87 | ||
| Nissl (non-VGLUT-2, non-TH) | Total estimated number | 58,073 ± 7,302 | 57,165 ± 4,612 | |
| Density/mm3 | 2,266 ± 135 | 2,168 ± 201 | ||
| Size (μm2) (n=100) | 195 ± 9 | 197± 8 | ||
| Lateral | VGLUT-2 | Total estimated number | 16,078 ± 1,140 | 11,004 ± 1,401 |
| Density/mm3 | 2,266 ± 135 | 1,081 ± 159 | ||
| Size (μm2) (n=100) | 218 ± 10 | 240 ± 19 | ||
| Nissl (non-VGLUT-2) | Total estimated number | 38,283 ± 4,481 | 36,743 ± 3,700 | |
| Density/mm3 | 2,760 ± 263 | 2,384 ± 344 | ||
| Size (μm2) (n=100) | 175 ± 13 | 177 ± 6 | ||
| Dorsolateral | VGLUT-2 | Total estimated number | 14,080 ± 1,107 | 10,306 ± 1,455 |
| Density/mm3 | 1,296 ± 63 | 1,007 ± 169 | ||
| Size (μm2) (n=100) | 187 ± 14 | 200 ± 9 | ||
| NOS | Total estimated number | 27,576 ± 1,653 | 27,484 ± 2,626 | |
| Density/mm3 | 1,627 ± 182 | 1,575 ± 204 | ||
| Size (μm2) (n=100) | 202 ± 8 | 192 ± 9 | ||
| Nissl (non-VGLUT-2, non-NOS) | Total estimated number | 48,852 ± 3,996 | 47,382 ± 5,101 | |
| Density/mm3 | 2,813 ± 98 | 2,753 ± 194 | ||
| Size (μm2) (n=100) | 197 ± 14 | 188 ± 9 | ||
| Dorsomedial | VGLUT-2 | Total estimated number | 15,412 ± 1,097 | 8,847 ± 1,052 |
| Density/mm3 | 1,277 ± 74 | 796 ± 101 | ||
| Size (μm2) (n=100) | 197 ± 11 | 212 ± 10 | ||
| Nissl (non-VGLUT-2) | Total estimated number | 42,611 ± 3,142 | 48,148 ± 5,714 | |
| Density/mm3 | 2,549 ± 141 | 2,468 ± 181 | ||
| Size (μm2) (n=100) | 169 ± 18 | 175 ± 9 |
NOS=nitric oxide synthase, TH=tyrosine hydroxylase, VGLUT-2=vesicular transporter 2
Figure 2.
Total estimated numbers of VGLUT-2 (left) and non immunoreactive Nissl stained neurons in the ventrolateral, (vl), lateral (l), dorsolateral (dl), and the dorsomedial (dm) columns of the PAG (right). In MSA cases, there was a reduction in the total estimated number of VGLUT-2 immunoreactive neurons in the ventrolateral, lateral and dorsomedial with a trend to decrease in the dorsolateral PAG. There was no significant difference between MSA and controls in the total estimated cell counts or cell density of Nissl-stained, non-VGLUT-2, non-TH, or non-NOS immunoreactive neurons in any of the PAG columns.
Alpha-Synuclein inclusions and Other Neuropathological Changes in the PAG
There were abundant GCIs in all columns in the PAG in the MSA cases (Figure 3). We found no convincing evidence of neuronal inclusions in our samples. Although there was large variability among cases, GCIs appeared to be more abundant in the ventrolateral than in the other columns (Figure 4). There was no correlation between the number of GCIs and the patients’ age or symptom duration and no differences between cases with clinical MSA-P or MSA-C. There was loss of myelin and astrocytic gliosis in the area of GCI accumulation, as assessed with double immunocytochemistry in all columns of the PAG (Figure 5).
Figure 3.
Glial cytoplasmic inclusion (GCI) distribution throughout the periaqueductal gray (PAG) in a 66 year-old man with multiple system atrophy (MSA). (Case MSA-9 in Table 1). Insert shows the approximate level of the sections.
Figure 4.
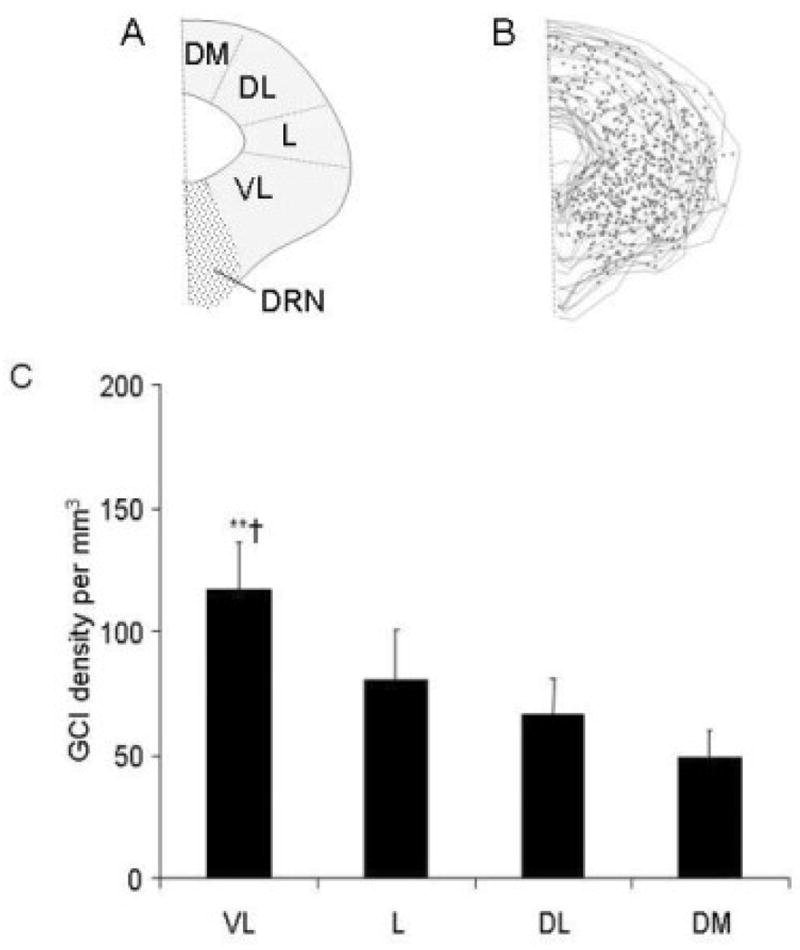
A: Schematic representation of the periaqueductal gray (PAG) showing the topography ventrolateral (vl), lateral (l), dorsolateral (dl), and the dorsomedial (dm) columns and B, composite of the thirteen sections (from Figure 3) showing the distribution of glial cytoplasmic inclusions (GCIs) in one MSA case. C. Density of GCIs in the different PAG columns in all MSA cases. GCIs appeared to be more abundant in the ventrolateral than in other columns. (** p<0.01 vl vs dm, † p<0.05, vl vs dl). DRN, dorsal raphe nucleus. Bar = 20μm.
Figure 5.
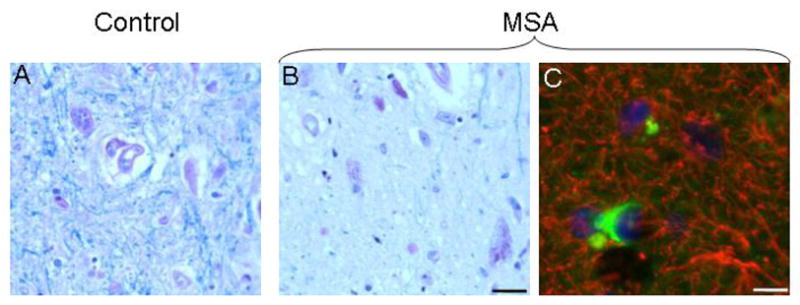
Representative 12 μm sections of the ventrolateral PAG stained for Luxol fast blue for myelin (A, B), and double-label immunocytochemistry for α-synuclein (FITC) and glial fibrillary acidic protein (GFAP) (Cy3). There was loss of myelin and astrocytic gliosis in the area of GCI accumulation. Bar = 25 μm.
Discussion
Our results indicate that the PAG is involved in MSA. Our main findings are that in MSA (1) there involvement of selected cell groups in the different columns of the PAG; and (2) there may be dissociation between GCI accumulation and cell loss. Loss of glutamatergic neurons was observed in the ventrolateral, lateral and dorsomedial, and to a lesser extent the dorsolateral PAG; in contrast, there was preservation of NOS immunoreactive neurons in the dorsolateral PAG. Involvement of the PAG may contribute to some of the manifestations of MSA. Experimental evidence indicates that the ventrolateral PAG has a major role in integrated cardiovascular and pain modulatory responses to visceral and somatic pain, control of micturition, respiration (Zhang et al., 2007) and upper airway resistance. The ventrolateral PAG projects to the nucleus of the solitary tract and may thus contribute to modulation of the baroreflex. The ventrolateral PAG is a critical relay nucleus in the micturition reflex and, via its connections with the nucleus retroambiguus, inhibits muscle tone in the upper airway. Therefore, loss of glutamatergic neurons in the ventrolateral PAG may contribute to impaired baroreflex, abnormal micturition, and laryngeal stridor in MSA. The lateral PAG column mediates sympathoexcitatory responses via its projections to the rostral ventrolateral medulla (Bandler et al., 2000) and cell loss in this column may contribute to impaired sympathetic cardiovascular control in MSA. The dorsomedial PAG has a major role in mediating anxiety responses in experimental animals, but the clinical significance of neuronal loss in this region in MSA is uncertain. The affected PAG columns receive inputs from the medial preoptic area, and their involvement may contribute to abnormal thermoregulation in MSA patients.
Our results also show abundant GCI accumulation in all columns of the PAG. The preservation of Nissl-stained non-VGLUT-2 immunoreactive neurons in all PAG columns and the preservation of NOS immunoreactive neurons in the dorsolateral PAG are consistent with previous studies showing a lack of a direct correlation between GCI density and severity of neuronal loss in MSA. This supports the hypothesis that oligodendrocyte pathology is an early feature preceding neuronal loss in MSA, and that the degeneration of cell bodies is not a prerequisite for the formation of GCIs. There was myelin loss and astrocytic gliosis in association with GCI accumulation in all columns of the PAG. This is also consistent with previous observations and suggests that functional disconnection, abnormal local microenvironment, or both may affect PAG neuronal function in MSA. Further studies are required to further address these critical issues.
Acknowledgments
This work was supported by a grant from the National Institutes of Health (NS32352-P2) and Mayo Funds.
Footnotes
The authors have no financial interests to disclose.
Publisher's Disclaimer: This is a PDF file of an unedited manuscript that has been accepted for publication. As a service to our customers we are providing this early version of the manuscript. The manuscript will undergo copyediting, typesetting, and review of the resulting proof before it is published in its final citable form. Please note that during the production process errors may be discovered which could affect the content, and all legal disclaimers that apply to the journal pertain.
References
- Bandler R, Keay KA, Floyd N, Price J. Central circuits mediating patterned autonomic activity during active vs. passive emotional coping. Brain Res Bull. 2000;53:95–104. doi: 10.1016/s0361-9230(00)00313-0. [DOI] [PubMed] [Google Scholar]
- Benarroch EE, Schmeichel AM, Dugger BN, Sandroni P, Parisi JE, Low PA. Dopamine cell loss in the periaqueductal gray in multiple system atrophy and Lewy body dementia. Neurology. 2009;73:106–112. doi: 10.1212/WNL.0b013e3181ad53e7. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Benarroch EE, Schmeichel AM, Low PA, Parisi JE. Involvement of medullary serotonergic groups in multiple system atrophy. Ann Neurol. 2004;55:418–422. doi: 10.1002/ana.20021. [DOI] [PubMed] [Google Scholar]
- Benarroch EE, Smithson IL, Low PA, Parisi JE. Depletion of catecholaminergic neurons of the rostral ventrolateral medulla in multiple systems atrophy with autonomic failure. Ann Neurol. 1998;43:156–163. doi: 10.1002/ana.410430205. [DOI] [PubMed] [Google Scholar]
- Borelli KG, Brandao ML. Effects of ovine CRF injections into the dorsomedial, dorsolateral and lateral columns of the periaqueductal gray: a functional role for the dorsomedial column. Horm Behav. 2008;53:40–50. doi: 10.1016/j.yhbeh.2007.08.013. [DOI] [PubMed] [Google Scholar]
- Braak H, Rub U, Sandmann-Keil D, et al. Parkinson’s disease: affection of brain stem nuclei controlling premotor and motor neurons of the somatomotor system. Acta Neuropathol. 2000;99:489–495. doi: 10.1007/s004010051150. [DOI] [PubMed] [Google Scholar]
- Carrive P. The periaqueductal gray and defensive behavior: functional representation and neuronal organization. Behav Brain Res. 1993;58:27–47. doi: 10.1016/0166-4328(93)90088-8. [DOI] [PubMed] [Google Scholar]
- de Menezes RC, Zaretsky DV, Fontes MA, DiMicco JA. Cardiovascular and thermal responses evoked from the periaqueductal grey require neuronal activity in the hypothalamus. J Physiol. 2009;587:1201–1215. doi: 10.1113/jphysiol.2008.161463. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Fuller PM, Saper CB, Lu J. The pontine REM switch: past and present. J Physiol. 2007;584:735–741. doi: 10.1113/jphysiol.2007.140160. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gilman S, Wenning GK, Low PA, et al. Second consensus statement on the diagnosis of multiple system atrophy. Neurology. 2008;71:670–676. doi: 10.1212/01.wnl.0000324625.00404.15. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Haxhiu MA, Yamamoto BK, Dreshaj IA, Ferguson DG. Activation of the midbrain periaqueductal gray induces airway smooth muscle relaxation. J Appl Physiol. 2002;93:440–449. doi: 10.1152/japplphysiol.00752.2001. [DOI] [PubMed] [Google Scholar]
- Heinricher MM, Tavares I, Leith JL, Lumb BM. Descending control of nociception: Specificity, recruitment and plasticity. Brain Res Rev. 2009;60:214–225. doi: 10.1016/j.brainresrev.2008.12.009. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Holstege G. Micturition and the soul. J Comp Neurol. 2005;493:15–20. doi: 10.1002/cne.20785. [DOI] [PubMed] [Google Scholar]
- Inui K, Murase S, Nosaka S. Facilitation of the arterial baroreflex by the ventrolateral part of the midbrain periaqueductal grey matter in rats. J Physiol. 1994;477:89–101. doi: 10.1113/jphysiol.1994.sp020174. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ishizawa K, Komori T, Arai N, Mizutani T, Hirose T. Glial cytoplasmic inclusions and tissue injury in multiple system atrophy: A quantitative study in white matter (olivopontocerebellar system) and gray matter (nigrostriatal system) Neuropathology. 2008;28:249–257. doi: 10.1111/j.1440-1789.2007.00855.x. [DOI] [PubMed] [Google Scholar]
- Kavia RB, Dasgupta R, Fowler CJ. Functional imaging and the central control of the bladder. J Comp Neurol. 2005;493:27–32. doi: 10.1002/cne.20753. [DOI] [PubMed] [Google Scholar]
- Lu J, Jhou TC, Saper CB. Identification of wake-active dopaminergic neurons in the ventral periaqueductal gray matter. Journal of Neuroscience. 2006;26:193–202. doi: 10.1523/JNEUROSCI.2244-05.2006. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Onstott D, Mayer B, Beitz AJ. Nitric oxide synthase immunoreactive neurons anatomically define a longitudinal dorsolateral column within the midbrain periaqueductal gray of the rat: analysis using laser confocal microscopy. Brain Res Bull. 1993;610:317–324. doi: 10.1016/0006-8993(93)91416-p. [DOI] [PubMed] [Google Scholar]
- Papp MI, Lantos PL. The distribution of oligodendroglial inclusions in multiple system atrophy and its relevance to clinical symptomatology. Brain. 1994;117:235–243. doi: 10.1093/brain/117.2.235. [DOI] [PubMed] [Google Scholar]
- Parvizi J, Van Hoesen GW, Damasio A. Selective pathological changes of the periaqueductal gray matter in Alzheimer’s disease. Ann Neurol. 2000;48:344–353. [PubMed] [Google Scholar]
- Paxinos G, Huang XF. Atlas of the Human Brainstem. Academic Press; San Diego: 1995. [Google Scholar]
- Rizvi TA, Ennis M, Shipley MT. Reciprocal connections between the medial preoptic area and the midbrain periaqueductal gray in rat: a WGA-HRP and PHA-L study. J Comp Neurol. 1992;315:1–15. doi: 10.1002/cne.903150102. [DOI] [PubMed] [Google Scholar]
- Schmitz C, Hof PR. Design-based stereology in neuroscience. Neuroscience. 2005;130:813–831. doi: 10.1016/j.neuroscience.2004.08.050. [DOI] [PubMed] [Google Scholar]
- Trojanowski JQ, Revesz T, MSA NWGo. Proposed neuropathological criteria for the post mortem diagnosis of multiple system atrophy. Neuropathol Appl Neurobiol. 2007;33:615–620. doi: 10.1111/j.1365-2990.2007.00907.x. [DOI] [PubMed] [Google Scholar]
- Wenning GK, Stefanova N, Jellinger KA, Poewe W, Schlossmacher MG. Multiple system atrophy: a primary oligodendrogliopathy. Ann Neurol. 2008;64:239–246. doi: 10.1002/ana.21465. [DOI] [PubMed] [Google Scholar]
- Zhang W, Hayward LF, Davenport PW. Respiratory responses elicited by rostral versus caudal dorsal periaqueductal gray stimulation in rats. Auton Neurosci. 2007;134:45–54. doi: 10.1016/j.autneu.2007.02.003. [DOI] [PubMed] [Google Scholar]