Abstract
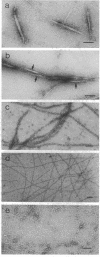
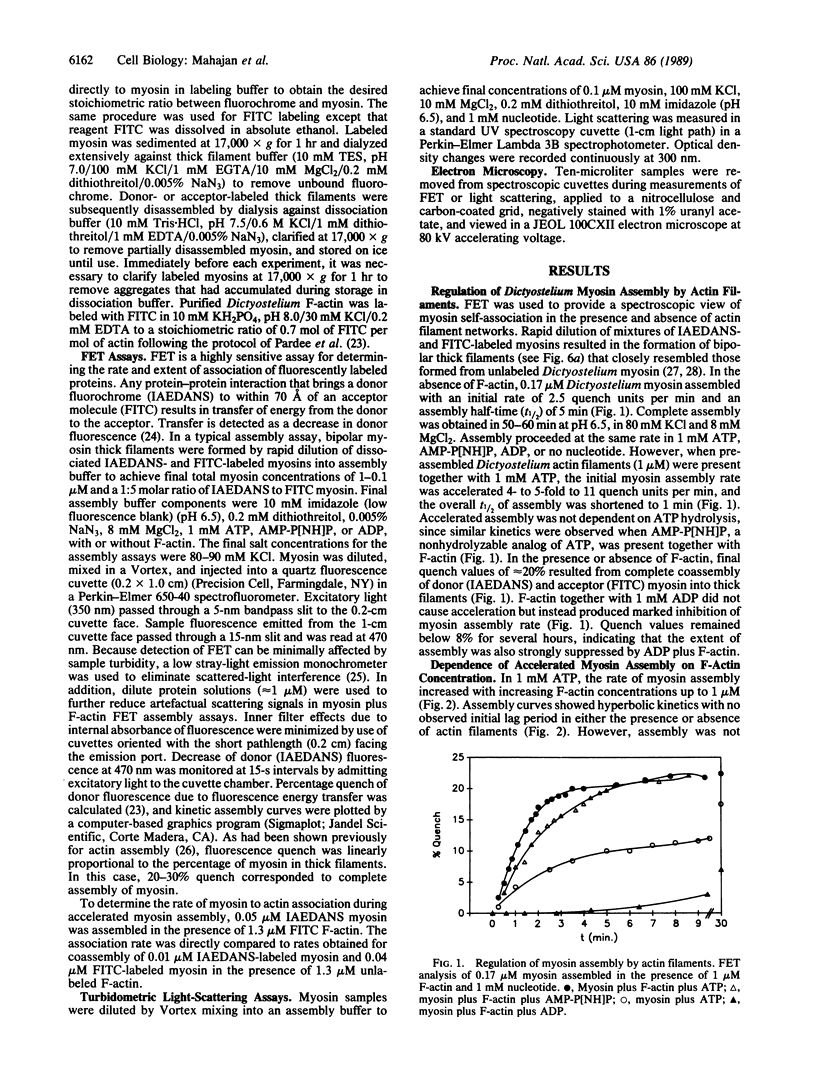
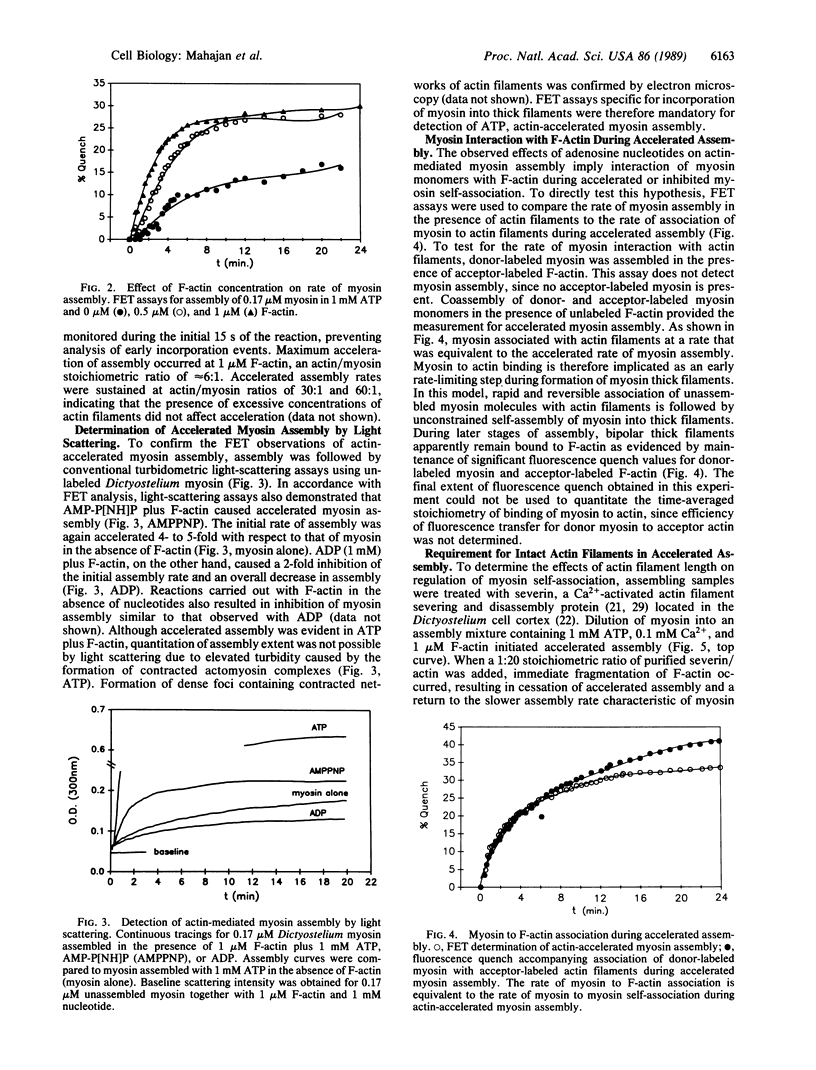
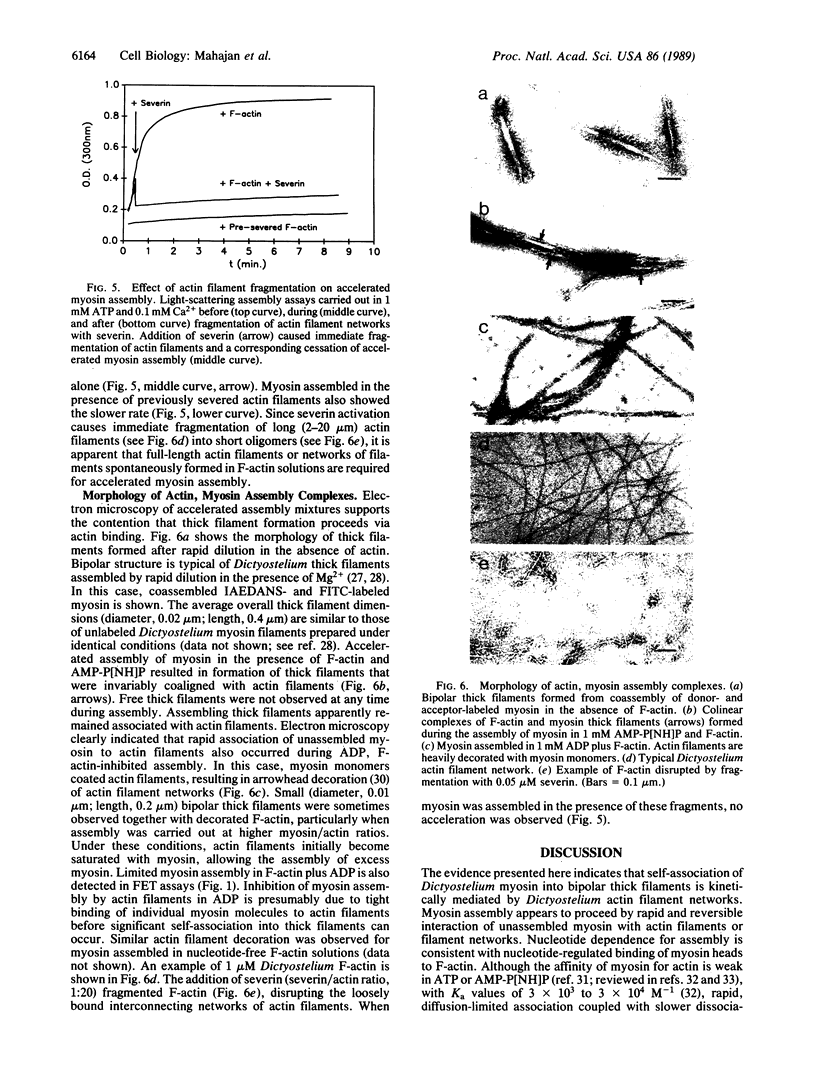
Because myosin thick filaments form in the actin-rich cortex of nonmuscle cells, we have examined the role of Dictyostelium actin filaments in the assembly of Dictyostelium myosin (type II). Fluorescence energy transfer and light-scattering assembly assays indicate that self-association of Dictyostelium myosin into bipolar thick filaments is kinetically regulated by actin filament networks. Regulation is nucleotide dependent but does not require ATP hydrolysis. Myosin assembly is accelerated approximately 5-fold by actin filaments when either 1 mM ATP or 1 mM adenosine 5'-[beta,gamma-imido]triphosphate (AMP-P[NH]P) is present. However, actin filaments together with 1 mM ADP abolish myosin assembly. Accelerated assembly appears to require transient binding of myosin molecules to actin filaments before incorporation into thick filaments. Fluorescence energy-transfer assays demonstrate that myosin associates with actin filaments at a rate that is equivalent to the accelerated myosin assembly rate, evidence that myosin to actin binding is a rate-limiting step in accelerated thick filament formation. Actin filament networks are also implicated in regulation of thick filament formation, since fragmentation of F-actin networks by severin causes immediate cessation of accelerated myosin assembly. Electron microscopic studies support a model of actin filament-mediated myosin assembly. In ADP, myosin monomers rapidly decorate F-actin, preventing extensive formation of thick filaments. In AMP-P[NH]P, myosin assembles along actin filaments, forming structures that resemble primitive stress fibers. Taken together, these data suggest a model in which site-directed assembly of thick filaments in Dictyostelium is mediated by the interaction of myosin monomers with cortical actin filament networks.
Full text
PDF

Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Adelstein R. S., Eisenberg E. Regulation and kinetics of the actin-myosin-ATP interaction. Annu Rev Biochem. 1980;49:921–956. doi: 10.1146/annurev.bi.49.070180.004421. [DOI] [PubMed] [Google Scholar]
- Atkinson M. A., Korn E. D. A model for the polymerization of Acanthamoeba myosin II and the regulation of its actin-activated Mg2+-ATPase activity. J Biol Chem. 1987 Nov 15;262(32):15809–15811. [PubMed] [Google Scholar]
- Berlot C. H., Devreotes P. N., Spudich J. A. Chemoattractant-elicited increases in Dictyostelium myosin phosphorylation are due to changes in myosin localization and increases in kinase activity. J Biol Chem. 1987 Mar 15;262(8):3918–3926. [PubMed] [Google Scholar]
- Berlot C. H., Spudich J. A., Devreotes P. N. Chemoattractant-elicited increases in myosin phosphorylation in Dictyostelium. Cell. 1985 Nov;43(1):307–314. doi: 10.1016/0092-8674(85)90036-4. [DOI] [PubMed] [Google Scholar]
- Biosca J. A., Greene L. E., Eisenberg E. Binding of ADP and ATP analogs to cross-linked and non-cross-linked acto X S-1. J Biol Chem. 1986 Jul 25;261(21):9793–9800. [PubMed] [Google Scholar]
- Brock A. M., Pardee J. D. Cytoimmunofluorescent localization of severin in Dictyostelium amoebae. Dev Biol. 1988 Jul;128(1):30–39. doi: 10.1016/0012-1606(88)90263-1. [DOI] [PubMed] [Google Scholar]
- Brown S. S., Yamamoto K., Spudich J. A. A 40,000-dalton protein from Dictyostelium discoideum affects assembly properties of actin in a Ca2+-dependent manner. J Cell Biol. 1982 Apr;93(1):205–210. doi: 10.1083/jcb.93.1.205. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Clarke M., Spudich J. A. Biochemical and structural studies of actomyosin-like proteins from non-muscle cells. Isolation and characterization of myosin from amoebae of Dictyostelium discoideum. J Mol Biol. 1974 Jun 25;86(2):209–222. doi: 10.1016/0022-2836(74)90013-8. [DOI] [PubMed] [Google Scholar]
- Cooke R. The mechanism of muscle contraction. CRC Crit Rev Biochem. 1986;21(1):53–118. doi: 10.3109/10409238609113609. [DOI] [PubMed] [Google Scholar]
- Craig R., Smith R., Kendrick-Jones J. Light-chain phosphorylation controls the conformation of vertebrate non-muscle and smooth muscle myosin molecules. 1983 Mar 31-Apr 6Nature. 302(5907):436–439. doi: 10.1038/302436a0. [DOI] [PubMed] [Google Scholar]
- Davis J. S. Pressure-jump studies on the length-regulation kinetics of the self-assembly of myosin from vertebrate skeletal muscle into thick filament. Biochem J. 1981 Aug 1;197(2):309–314. doi: 10.1042/bj1970309. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Korn E. D., Hammer J. A., 3rd Myosins of nonmuscle cells. Annu Rev Biophys Biophys Chem. 1988;17:23–45. doi: 10.1146/annurev.bb.17.060188.000323. [DOI] [PubMed] [Google Scholar]
- Kron S. J., Spudich J. A. Fluorescent actin filaments move on myosin fixed to a glass surface. Proc Natl Acad Sci U S A. 1986 Sep;83(17):6272–6276. doi: 10.1073/pnas.83.17.6272. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuczmarski E. R., Spudich J. A. Regulation of myosin self-assembly: phosphorylation of Dictyostelium heavy chain inhibits formation of thick filaments. Proc Natl Acad Sci U S A. 1980 Dec;77(12):7292–7296. doi: 10.1073/pnas.77.12.7292. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kuczmarski E. R., Tafuri S. R., Parysek L. M. Effect of heavy chain phosphorylation on the polymerization and structure of Dictyostelium myosin filaments. J Cell Biol. 1987 Dec;105(6 Pt 2):2989–2997. doi: 10.1083/jcb.105.6.2989. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mittal B., Sanger J. M., Sanger J. W. Visualization of myosin in living cells. J Cell Biol. 1987 Oct;105(4):1753–1760. doi: 10.1083/jcb.105.4.1753. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mockrin S. C., Spudich J. A. Calcium control of actin-activated myosin adenosine triphosphatase from Dictyostelium discoideum. Proc Natl Acad Sci U S A. 1976 Jul;73(7):2321–2325. doi: 10.1073/pnas.73.7.2321. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pardee J. D., Simpson P. A., Stryer L., Spudich J. A. Actin filaments undergo limited subunit exchange in physiological salt conditions. J Cell Biol. 1982 Aug;94(2):316–324. doi: 10.1083/jcb.94.2.316. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Pollard T. D. Structure and polymerization of Acanthamoeba myosin-II filaments. J Cell Biol. 1982 Dec;95(3):816–825. doi: 10.1083/jcb.95.3.816. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rahmsdorf H. J., Malchow D., Gerisch G. Cyclic AMP-induced phosphorylation in Dictyostelium of a polypeptide comigrating with myosin heavy chains. FEBS Lett. 1978 Apr 15;88(2):322–326. doi: 10.1016/0014-5793(78)80203-8. [DOI] [PubMed] [Google Scholar]
- Reines D., Clarke M. Immunochemical analysis of the supramolecular structure of myosin in contractile cytoskeletons of Dictyostelium amoebae. J Biol Chem. 1985 Nov 15;260(26):14248–14254. [PubMed] [Google Scholar]
- Reines D., Clarke M. Quantitative immunochemical studies of myosin in Dictyostelium discoideum. J Biol Chem. 1985 Jan 25;260(2):1133–1140. [PubMed] [Google Scholar]
- Rubino S., Fighetti M., Unger E., Cappuccinelli P. Location of actin, myosin, and microtubular structures during directed locomotion of Dictyostelium amebae. J Cell Biol. 1984 Feb;98(2):382–390. doi: 10.1083/jcb.98.2.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Rubino S., Fighetti M., Unger E., Cappuccinelli P. Location of actin, myosin, and microtubular structures during directed locomotion of Dictyostelium amebae. J Cell Biol. 1984 Feb;98(2):382–390. doi: 10.1083/jcb.98.2.382. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Saad A. D., Pardee J. D., Fischman D. A. Dynamic exchange of myosin molecules between thick filaments. Proc Natl Acad Sci U S A. 1986 Dec;83(24):9483–9487. doi: 10.1073/pnas.83.24.9483. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Sheetz M. P., Spudich J. A. Movement of myosin-coated fluorescent beads on actin cables in vitro. Nature. 1983 May 5;303(5912):31–35. doi: 10.1038/303031a0. [DOI] [PubMed] [Google Scholar]
- Stewart P. R., Spudich J. A. Structural states of dictyostelium myosin. J Supramol Struct. 1979;12(1):1–14. doi: 10.1002/jss.400120102. [DOI] [PubMed] [Google Scholar]
- Stryer L. Fluorescence energy transfer as a spectroscopic ruler. Annu Rev Biochem. 1978;47:819–846. doi: 10.1146/annurev.bi.47.070178.004131. [DOI] [PubMed] [Google Scholar]
- Taylor D. L., Reidler J., Spudich J. A., Stryer L. Detection of actin assembly by fluorescence energy transfer. J Cell Biol. 1981 May;89(2):362–367. doi: 10.1083/jcb.89.2.362. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Trybus K. M., Lowey S. Conformational states of smooth muscle myosin. Effects of light chain phosphorylation and ionic strength. J Biol Chem. 1984 Jul 10;259(13):8564–8571. [PubMed] [Google Scholar]
- Trybus K. M., Lowey S. Subunit exchange between smooth muscle myosin filaments. J Cell Biol. 1987 Dec;105(6 Pt 2):3021–3030. doi: 10.1083/jcb.105.6.3021. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Warrick H. M., Spudich J. A. Myosin structure and function in cell motility. Annu Rev Cell Biol. 1987;3:379–421. doi: 10.1146/annurev.cb.03.110187.002115. [DOI] [PubMed] [Google Scholar]
- White H. D., Taylor E. W. Energetics and mechanism of actomyosin adenosine triphosphatase. Biochemistry. 1976 Dec 28;15(26):5818–5826. doi: 10.1021/bi00671a020. [DOI] [PubMed] [Google Scholar]
- Yamamoto K., Pardee J. D., Reidler J., Stryer L., Spudich J. A. Mechanism of interaction of Dictyostelium severin with actin filaments. J Cell Biol. 1982 Dec;95(3):711–719. doi: 10.1083/jcb.95.3.711. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Yumura S., Fukui Y. Reversible cyclic AMP-dependent change in distribution of myosin thick filaments in Dictyostelium. Nature. 1985 Mar 14;314(6007):194–196. doi: 10.1038/314194a0. [DOI] [PubMed] [Google Scholar]