Abstract
Bis[2-(4,6-dimethylpyrimidin-2-ylsulfanyl)ethyl]amine under hydrothermal conditions has unexpectedly been transformed into the title compound, C32H44N10S4. In the title molecule, the zigzag 3,10-diaza-6,7-disulfanyldodecyl skeleton has two dimethylpyrimidinylsulfanyl groups at both ends, and the aza atoms each carry a dimethylpyrimidinyl unit. The N atoms in the skeleton show a planar coordination.
Related literature
For the crystal structures of ligands having two 4,6-dimethylpyridimin-2-ylsulfanyl units linked to a hydrocarbon chain, see: Chen et al. (2007 ▶); Wang et al. (2007 ▶); Wu et al. (2007a
▶,b
▶).
Experimental
Crystal data
C32H44N10S4
M r = 697.01
Triclinic,

a = 11.7626 (5) Å
b = 12.7672 (6) Å
c = 13.7444 (7) Å
α = 106.382 (4)°
β = 103.276 (4)°
γ = 102.294 (4)°
V = 1840.15 (17) Å3
Z = 2
Cu Kα radiation
μ = 2.67 mm−1
T = 293 K
0.30 × 0.25 × 0.20 mm
Data collection
Oxford Diffraction Xcalibur Sapphire 3 diffractometer
Absorption correction: multi-scan (CrysAlis RED; Oxford Diffraction, 2009 ▶) T min = 0.947, T max = 1.000
11434 measured reflections
7060 independent reflections
5177 reflections with I > 2σ(I)
R int = 0.020
Refinement
R[F 2 > 2σ(F 2)] = 0.081
wR(F 2) = 0.252
S = 1.07
7060 reflections
423 parameters
4 restraints
H-atom parameters constrained
Δρmax = 1.45 e Å−3
Δρmin = −0.75 e Å−3
Data collection: CrysAlis PRO (Oxford Diffraction, 2009 ▶); cell refinement: CrysAlis PRO; data reduction: CrysAlis PRO; program(s) used to solve structure: SHELXS97 (Sheldrick, 2008 ▶); program(s) used to refine structure: SHELXL97 (Sheldrick, 2008 ▶); molecular graphics: X-SEED (Barbour, 2001 ▶); software used to prepare material for publication: publCIF (Westrip, 2010 ▶).
Supplementary Material
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536810035245/xu5021sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536810035245/xu5021Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report
Acknowledgments
We thank the Special Foundation for Nano Technology of the Shanghai Committee for S&T (1052 nm00600), the Foundation of the S&T Program of Shanghai Maritime University (20100128), the State Key Laboratory of Pollution Control and Resource Reuse Foundation (PCRRF09001) and the University of Malaya for supporting this study.
supplementary crystallographic information
Comment
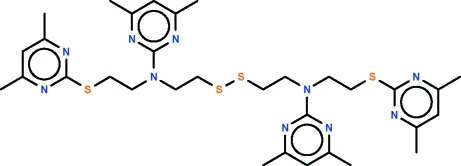
We are interested in synthesizing flexible ligands having two 4,6-dimethylpyridimin-2-ylsulfanyl units linked to a hydrocarbon chain; compounds such as 2,2'-bis(4,6-dimethylpyrimidn-2-ylsulfanyl)methane (Chen et al., 2007) and its ethane (Wu et al., 2007b), propane (Wu et al., 2007a) and butane (Wang et al., 2007) analogs have been synthesized for the purpose of studying its coordination chemistry in metal adducts. The coordination chemistry can be expanded in bis[2-(4,6-dimethylpyrimidin-2-ysulfanyl)ethyl]amine, whose synthesis has not been reported yet. However, the attempted complexation with copper ions under hydrothermal conditions yielded bis{2-[(4,6-dimethylpyrimidin-1-yl)(4,6-dimethylpyrimidin-1-ylsulfanyl-2-ethyl)amino]ethyl}disulfide (Scheme I, Fig. 1), a more interesting ligand whose exocyclic sulfur and endocyclic nitrogen sites offer many more sites for coordination.
Experimental
Bis[2-(4,6-dimethylpyrimidin-2-ylsulfanyl)ethyl]amine was synthesized from the reaction of bis(2-chloroethyl)ammonium hydrochloride (1.78 g, 0.01 mol) dissolved in ethanol (100 ml) and 4,6-dimethylpyrimidine-2-thiol (2.80 g, 0.02 mol)/sodium hydroxide (0.8 g, 0.02 mol) dissolved in ethanol (200 ml). The solution was heated at 353 K for 8 h. The solvent was removed and the residue was column chromatographed with ethly acetate/petroleum ether (1/1 v/v) as eluent to yield a white powder; yield 63%. The formulation was confirmed by 1H NMR (CDCl3, 400 MHz) spectroscopy: 1.36–1.402(m, 1H), 2.274 (d,6H), 2.403–2.426 (d,6H), 2.813–2.871(m, 2H), 3.343–3.380 (m, 2H), 6.279 (s, 1H), 6.715 (s, 1H). This compound has not been reported in the chemical literature yet.
The title compound was the unexpected product obtained in the reaction of bis(2-(4,6-dimethylpyrimidin-2-ylthio)ethyl)amine (0.175 g, 0.5 mmol), copper perchlorate (0.132 g, 0.5 mmol) and water (8 ml). The reactants were heated in a 23-ml Teflon-lined Parr reactor at 413 K for 3 days. The mixture was cooled to room temperature at a rate of 5 K h-1. The prismatic crystals were collected and washed with water; yield: 40%. MS (ESI) m/z(%): 698.2 (M+1). CH&N elemental analysis, calculated for C32H44N10S4: C 55.14, H 6.36, N 20.09%. Found: C 55.50, H 6.56, N 19.56%.
Refinement
Carbon-bound H-atoms were placed in calculated positions (C—H 0.93 to 0.96 Å) and were included in the refinement in the riding model approximation, with U(H) set to 1.2–1.5 times Ueq(C). The final difference Fourier map had a peak in the vicinity of N6.
For the ethyl portions, the carbon-carbon distance was restrained to 1.53±0.01 Å.
Figures
Fig. 1.
Thermal ellipsoid plot (Barbour, 2001) of C32H44N10S4 at the 50% probability level; hydrogen atoms are drawn as spheres of arbitrary radius.
Crystal data
| C32H44N10S4 | Z = 2 |
| Mr = 697.01 | F(000) = 740 |
| Triclinic, P1 | Dx = 1.258 Mg m−3 |
| Hall symbol: -P 1 | Cu Kα radiation, λ = 1.54184 Å |
| a = 11.7626 (5) Å | Cell parameters from 5344 reflections |
| b = 12.7672 (6) Å | θ = 4.6–74.5° |
| c = 13.7444 (7) Å | µ = 2.67 mm−1 |
| α = 106.382 (4)° | T = 293 K |
| β = 103.276 (4)° | Prism, colorless |
| γ = 102.294 (4)° | 0.30 × 0.25 × 0.20 mm |
| V = 1840.15 (17) Å3 |
Data collection
| Oxford Diffraction Xcalibur Sapphire 3 diffractometer | 7060 independent reflections |
| Radiation source: fine-focus sealed tube | 5177 reflections with I > 2σ(I) |
| graphite | Rint = 0.020 |
| Detector resolution: 16.0855 pixels mm-1 | θmax = 72.6°, θmin = 4.6° |
| ω scans | h = −14→11 |
| Absorption correction: multi-scan (CrysAlis RED; Oxford Diffraction, 2009) | k = −15→15 |
| Tmin = 0.947, Tmax = 1.000 | l = −16→16 |
| 11434 measured reflections |
Refinement
| Refinement on F2 | Primary atom site location: structure-invariant direct methods |
| Least-squares matrix: full | Secondary atom site location: difference Fourier map |
| R[F2 > 2σ(F2)] = 0.081 | Hydrogen site location: inferred from neighbouring sites |
| wR(F2) = 0.252 | H-atom parameters constrained |
| S = 1.07 | w = 1/[σ2(Fo2) + (0.1399P)2 + 1.4306P] where P = (Fo2 + 2Fc2)/3 |
| 7060 reflections | (Δ/σ)max = 0.001 |
| 423 parameters | Δρmax = 1.45 e Å−3 |
| 4 restraints | Δρmin = −0.75 e Å−3 |
Fractional atomic coordinates and isotropic or equivalent isotropic displacement parameters (Å2)
| x | y | z | Uiso*/Ueq | ||
| S1 | 0.74226 (10) | 0.71528 (10) | 0.39171 (9) | 0.0696 (3) | |
| S2 | 0.57455 (12) | 0.67895 (10) | 0.41240 (10) | 0.0812 (4) | |
| S3 | 0.96757 (10) | 0.65326 (9) | 0.03184 (10) | 0.0661 (3) | |
| S4 | 0.36046 (14) | 0.95980 (10) | 0.78051 (12) | 0.0903 (5) | |
| N1 | 0.6607 (3) | 0.6061 (3) | 0.1261 (3) | 0.0538 (7) | |
| N2 | 0.5256 (3) | 0.4838 (3) | 0.1734 (3) | 0.0527 (7) | |
| N3 | 0.6443 (3) | 0.4147 (3) | 0.0636 (2) | 0.0531 (7) | |
| N4 | 0.9874 (3) | 0.8593 (3) | 0.1672 (3) | 0.0582 (8) | |
| N5 | 1.0838 (3) | 0.8487 (3) | 0.0304 (3) | 0.0598 (8) | |
| N6 | 0.6477 (4) | 0.9727 (4) | 0.6648 (4) | 0.0905 (14) | |
| N7 | 0.7688 (3) | 1.0764 (3) | 0.5901 (3) | 0.0745 (11) | |
| N8 | 0.6908 (4) | 1.1703 (3) | 0.7257 (3) | 0.0685 (10) | |
| N9 | 0.3262 (4) | 0.7490 (3) | 0.6468 (3) | 0.0712 (10) | |
| N10 | 0.1976 (4) | 0.7779 (3) | 0.7571 (3) | 0.0731 (10) | |
| C1 | 0.7241 (4) | 0.7789 (3) | 0.2897 (3) | 0.0645 (11) | |
| H1A | 0.7028 | 0.8486 | 0.3162 | 0.077* | |
| H1B | 0.8021 | 0.8000 | 0.2770 | 0.077* | |
| C2 | 0.6275 (4) | 0.7027 (3) | 0.1837 (3) | 0.0590 (9) | |
| H2A | 0.5514 | 0.6746 | 0.1972 | 0.071* | |
| H2B | 0.6132 | 0.7485 | 0.1392 | 0.071* | |
| C3 | 0.6078 (3) | 0.4974 (3) | 0.1210 (3) | 0.0484 (8) | |
| C4 | 0.4770 (3) | 0.3770 (3) | 0.1680 (3) | 0.0554 (9) | |
| C5 | 0.3843 (5) | 0.3613 (4) | 0.2253 (4) | 0.0772 (13) | |
| H5A | 0.4117 | 0.4218 | 0.2932 | 0.116* | |
| H5B | 0.3743 | 0.2891 | 0.2358 | 0.116* | |
| H5C | 0.3075 | 0.3628 | 0.1837 | 0.116* | |
| C6 | 0.5088 (4) | 0.2862 (3) | 0.1110 (3) | 0.0588 (9) | |
| H6 | 0.4739 | 0.2122 | 0.1075 | 0.071* | |
| C7 | 0.5939 (3) | 0.3083 (3) | 0.0593 (3) | 0.0537 (9) | |
| C8 | 0.6332 (5) | 0.2155 (4) | −0.0056 (4) | 0.0706 (11) | |
| H8A | 0.7200 | 0.2308 | 0.0227 | 0.106* | |
| H8B | 0.6130 | 0.2129 | −0.0782 | 0.106* | |
| H8C | 0.5918 | 0.1434 | −0.0031 | 0.106* | |
| C9 | 0.7418 (3) | 0.6247 (3) | 0.0630 (3) | 0.0537 (9) | |
| H9A | 0.7489 | 0.6996 | 0.0571 | 0.064* | |
| H9B | 0.7049 | 0.5684 | −0.0084 | 0.064* | |
| C10 | 0.8697 (3) | 0.6174 (4) | 0.1083 (3) | 0.0591 (9) | |
| H10A | 0.8643 | 0.5404 | 0.1081 | 0.071* | |
| H10B | 0.9051 | 0.6693 | 0.1816 | 0.071* | |
| C11 | 1.0171 (3) | 0.8040 (3) | 0.0842 (3) | 0.0551 (9) | |
| C12 | 1.0309 (4) | 0.9735 (4) | 0.2019 (4) | 0.0638 (10) | |
| C13 | 0.9967 (5) | 1.0376 (4) | 0.2947 (5) | 0.0839 (14) | |
| H13A | 1.0156 | 1.0076 | 0.3511 | 0.126* | |
| H13B | 0.9106 | 1.0291 | 0.2729 | 0.126* | |
| H13C | 1.0418 | 1.1172 | 0.3196 | 0.126* | |
| C14 | 1.1014 (4) | 1.0279 (4) | 0.1525 (4) | 0.0691 (11) | |
| H14 | 1.1308 | 1.1073 | 0.1768 | 0.083* | |
| C15 | 1.1274 (4) | 0.9623 (4) | 0.0662 (4) | 0.0611 (10) | |
| C16 | 1.2025 (4) | 1.0147 (4) | 0.0081 (4) | 0.0761 (13) | |
| H16A | 1.1798 | 0.9639 | −0.0643 | 0.114* | |
| H16B | 1.2876 | 1.0277 | 0.0425 | 0.114* | |
| H16C | 1.1883 | 1.0862 | 0.0088 | 0.114* | |
| C17 | 0.5658 (5) | 0.8234 (6) | 0.4963 (4) | 0.112 (2) | |
| H17A | 0.5790 | 0.8783 | 0.4604 | 0.134* | |
| H17B | 0.4857 | 0.8149 | 0.5061 | 0.134* | |
| C18 | 0.6604 (5) | 0.8636 (5) | 0.5998 (4) | 0.0878 (15) | |
| H18A | 0.7409 | 0.8759 | 0.5906 | 0.105* | |
| H18B | 0.6495 | 0.8076 | 0.6347 | 0.105* | |
| C19 | 0.7040 (5) | 1.0778 (4) | 0.6585 (4) | 0.0789 (14) | |
| C20 | 0.8330 (4) | 1.1790 (4) | 0.5960 (3) | 0.0659 (11) | |
| C21 | 0.9099 (5) | 1.1770 (6) | 0.5229 (5) | 0.0984 (18) | |
| H21A | 0.8579 | 1.1487 | 0.4503 | 0.148* | |
| H21B | 0.9600 | 1.1280 | 0.5320 | 0.148* | |
| H21C | 0.9613 | 1.2530 | 0.5393 | 0.148* | |
| C22 | 0.8282 (4) | 1.2782 (4) | 0.6648 (4) | 0.0668 (11) | |
| H22 | 0.8742 | 1.3490 | 0.6685 | 0.080* | |
| C23 | 0.7540 (4) | 1.2708 (3) | 0.7285 (3) | 0.0590 (9) | |
| C24 | 0.7415 (5) | 1.3748 (4) | 0.8051 (4) | 0.0796 (14) | |
| H24A | 0.6594 | 1.3786 | 0.7827 | 0.119* | |
| H24B | 0.7977 | 1.4422 | 0.8062 | 0.119* | |
| H24C | 0.7595 | 1.3701 | 0.8752 | 0.119* | |
| C25 | 0.5709 (5) | 0.9658 (4) | 0.7391 (4) | 0.0811 (14) | |
| H25A | 0.5534 | 0.8904 | 0.7449 | 0.097* | |
| H25B | 0.6147 | 1.0219 | 0.8097 | 0.097* | |
| C26 | 0.4571 (4) | 0.9884 (4) | 0.6943 (5) | 0.0888 (16) | |
| H26A | 0.4172 | 0.9381 | 0.6208 | 0.107* | |
| H26B | 0.4727 | 1.0671 | 0.6971 | 0.107* | |
| C27 | 0.2882 (4) | 0.8113 (4) | 0.7202 (4) | 0.0642 (10) | |
| C28 | 0.2640 (5) | 0.6370 (4) | 0.6049 (4) | 0.0784 (13) | |
| C29 | 0.3050 (7) | 0.5657 (5) | 0.5205 (5) | 0.106 (2) | |
| H29A | 0.3261 | 0.6081 | 0.4762 | 0.159* | |
| H29B | 0.2399 | 0.4968 | 0.4776 | 0.159* | |
| H29C | 0.3751 | 0.5468 | 0.5536 | 0.159* | |
| C30 | 0.1711 (6) | 0.5938 (4) | 0.6403 (5) | 0.0937 (17) | |
| H30 | 0.1300 | 0.5159 | 0.6127 | 0.112* | |
| C31 | 0.1387 (5) | 0.6670 (5) | 0.7175 (5) | 0.0860 (15) | |
| C32 | 0.0362 (7) | 0.6270 (6) | 0.7581 (7) | 0.132 (3) | |
| H32A | 0.0588 | 0.6671 | 0.8335 | 0.198* | |
| H32B | 0.0197 | 0.5464 | 0.7445 | 0.198* | |
| H32C | −0.0357 | 0.6419 | 0.7225 | 0.198* |
Atomic displacement parameters (Å2)
| U11 | U22 | U33 | U12 | U13 | U23 | |
| S1 | 0.0649 (6) | 0.0682 (7) | 0.0666 (7) | 0.0186 (5) | 0.0204 (5) | 0.0108 (5) |
| S2 | 0.0860 (8) | 0.0554 (6) | 0.0813 (8) | −0.0002 (5) | 0.0436 (7) | −0.0057 (5) |
| S3 | 0.0617 (6) | 0.0514 (5) | 0.0835 (7) | 0.0110 (4) | 0.0360 (5) | 0.0143 (5) |
| S4 | 0.0995 (9) | 0.0567 (6) | 0.1033 (10) | −0.0047 (6) | 0.0636 (8) | 0.0056 (6) |
| N1 | 0.0538 (17) | 0.0440 (16) | 0.0608 (18) | 0.0087 (13) | 0.0228 (15) | 0.0141 (14) |
| N2 | 0.0541 (17) | 0.0459 (16) | 0.0562 (18) | 0.0106 (13) | 0.0209 (14) | 0.0152 (13) |
| N3 | 0.0549 (17) | 0.0450 (16) | 0.0532 (17) | 0.0090 (13) | 0.0192 (14) | 0.0104 (13) |
| N4 | 0.0518 (17) | 0.0540 (18) | 0.068 (2) | 0.0145 (14) | 0.0229 (15) | 0.0174 (15) |
| N5 | 0.0507 (17) | 0.0581 (19) | 0.070 (2) | 0.0102 (15) | 0.0252 (16) | 0.0213 (16) |
| N6 | 0.100 (3) | 0.061 (2) | 0.101 (3) | 0.008 (2) | 0.057 (3) | 0.005 (2) |
| N7 | 0.066 (2) | 0.075 (2) | 0.067 (2) | 0.0054 (18) | 0.0332 (18) | 0.0032 (18) |
| N8 | 0.079 (2) | 0.0455 (17) | 0.073 (2) | 0.0055 (16) | 0.0401 (19) | 0.0054 (16) |
| N9 | 0.074 (2) | 0.054 (2) | 0.077 (2) | 0.0105 (17) | 0.0210 (19) | 0.0160 (18) |
| N10 | 0.072 (2) | 0.064 (2) | 0.078 (2) | 0.0025 (18) | 0.0258 (19) | 0.0287 (19) |
| C1 | 0.067 (2) | 0.0390 (19) | 0.082 (3) | 0.0097 (17) | 0.031 (2) | 0.0108 (18) |
| C2 | 0.061 (2) | 0.049 (2) | 0.071 (2) | 0.0184 (17) | 0.0242 (19) | 0.0219 (18) |
| C3 | 0.0443 (17) | 0.0452 (18) | 0.0489 (19) | 0.0073 (14) | 0.0111 (14) | 0.0135 (15) |
| C4 | 0.051 (2) | 0.055 (2) | 0.056 (2) | 0.0070 (16) | 0.0172 (17) | 0.0192 (17) |
| C5 | 0.080 (3) | 0.067 (3) | 0.096 (3) | 0.015 (2) | 0.048 (3) | 0.033 (3) |
| C6 | 0.063 (2) | 0.0431 (19) | 0.066 (2) | 0.0081 (17) | 0.0184 (19) | 0.0184 (17) |
| C7 | 0.054 (2) | 0.0458 (19) | 0.051 (2) | 0.0107 (16) | 0.0103 (16) | 0.0107 (15) |
| C8 | 0.081 (3) | 0.050 (2) | 0.073 (3) | 0.018 (2) | 0.026 (2) | 0.010 (2) |
| C9 | 0.054 (2) | 0.051 (2) | 0.057 (2) | 0.0110 (16) | 0.0187 (17) | 0.0218 (17) |
| C10 | 0.056 (2) | 0.055 (2) | 0.068 (2) | 0.0122 (17) | 0.0220 (19) | 0.0270 (19) |
| C11 | 0.0441 (18) | 0.054 (2) | 0.065 (2) | 0.0122 (16) | 0.0185 (17) | 0.0168 (18) |
| C12 | 0.054 (2) | 0.056 (2) | 0.072 (3) | 0.0145 (18) | 0.0188 (19) | 0.012 (2) |
| C13 | 0.083 (3) | 0.068 (3) | 0.095 (4) | 0.020 (2) | 0.041 (3) | 0.011 (3) |
| C14 | 0.061 (2) | 0.047 (2) | 0.087 (3) | 0.0054 (18) | 0.020 (2) | 0.016 (2) |
| C15 | 0.048 (2) | 0.062 (2) | 0.073 (3) | 0.0114 (17) | 0.0171 (18) | 0.027 (2) |
| C16 | 0.064 (3) | 0.078 (3) | 0.089 (3) | 0.007 (2) | 0.028 (2) | 0.040 (3) |
| C17 | 0.074 (3) | 0.142 (6) | 0.083 (4) | 0.004 (3) | 0.042 (3) | −0.008 (4) |
| C18 | 0.099 (4) | 0.084 (3) | 0.088 (4) | 0.034 (3) | 0.035 (3) | 0.031 (3) |
| C19 | 0.082 (3) | 0.056 (2) | 0.085 (3) | 0.000 (2) | 0.047 (3) | 0.002 (2) |
| C20 | 0.046 (2) | 0.090 (3) | 0.061 (2) | 0.012 (2) | 0.0162 (18) | 0.032 (2) |
| C21 | 0.078 (3) | 0.140 (5) | 0.105 (4) | 0.038 (3) | 0.053 (3) | 0.059 (4) |
| C22 | 0.054 (2) | 0.064 (3) | 0.078 (3) | 0.0054 (19) | 0.015 (2) | 0.031 (2) |
| C23 | 0.055 (2) | 0.050 (2) | 0.061 (2) | 0.0077 (17) | 0.0109 (18) | 0.0150 (17) |
| C24 | 0.075 (3) | 0.044 (2) | 0.100 (4) | 0.008 (2) | 0.020 (3) | 0.009 (2) |
| C25 | 0.122 (4) | 0.055 (2) | 0.060 (3) | 0.028 (3) | 0.016 (3) | 0.018 (2) |
| C26 | 0.071 (3) | 0.058 (3) | 0.124 (5) | 0.009 (2) | 0.011 (3) | 0.036 (3) |
| C27 | 0.064 (2) | 0.054 (2) | 0.069 (3) | 0.0066 (19) | 0.021 (2) | 0.022 (2) |
| C28 | 0.082 (3) | 0.053 (2) | 0.088 (3) | 0.014 (2) | 0.013 (3) | 0.022 (2) |
| C29 | 0.125 (5) | 0.063 (3) | 0.107 (4) | 0.026 (3) | 0.023 (4) | 0.008 (3) |
| C30 | 0.104 (4) | 0.050 (3) | 0.104 (4) | −0.004 (3) | 0.019 (3) | 0.024 (3) |
| C31 | 0.088 (3) | 0.065 (3) | 0.091 (4) | −0.005 (3) | 0.022 (3) | 0.032 (3) |
| C32 | 0.127 (6) | 0.103 (5) | 0.149 (6) | −0.024 (4) | 0.059 (5) | 0.048 (5) |
Geometric parameters (Å, °)
| S1—C1 | 1.802 (5) | C9—H9B | 0.9700 |
| S1—S2 | 2.0323 (17) | C10—H10A | 0.9700 |
| S2—C17 | 1.918 (7) | C10—H10B | 0.9700 |
| S3—C11 | 1.764 (4) | C12—C14 | 1.382 (6) |
| S3—C10 | 1.801 (4) | C12—C13 | 1.499 (6) |
| S4—C27 | 1.763 (4) | C13—H13A | 0.9600 |
| S4—C26 | 1.874 (6) | C13—H13B | 0.9600 |
| N1—C3 | 1.369 (5) | C13—H13C | 0.9600 |
| N1—C2 | 1.449 (5) | C14—C15 | 1.385 (6) |
| N1—C9 | 1.456 (5) | C14—H14 | 0.9300 |
| N2—C4 | 1.335 (5) | C15—C16 | 1.497 (6) |
| N2—C3 | 1.343 (5) | C16—H16A | 0.9600 |
| N3—C7 | 1.339 (5) | C16—H16B | 0.9600 |
| N3—C3 | 1.342 (5) | C16—H16C | 0.9600 |
| N4—C11 | 1.324 (5) | C17—C18 | 1.464 (6) |
| N4—C12 | 1.339 (5) | C17—H17A | 0.9700 |
| N5—C15 | 1.331 (5) | C17—H17B | 0.9700 |
| N5—C11 | 1.343 (5) | C18—H18A | 0.9700 |
| N6—C19 | 1.399 (6) | C18—H18B | 0.9700 |
| N6—C18 | 1.486 (7) | C20—C22 | 1.371 (6) |
| N6—C25 | 1.520 (7) | C20—C21 | 1.497 (6) |
| N7—C20 | 1.336 (6) | C21—H21A | 0.9600 |
| N7—C19 | 1.338 (6) | C21—H21B | 0.9600 |
| N8—C23 | 1.325 (5) | C21—H21C | 0.9600 |
| N8—C19 | 1.342 (5) | C22—C23 | 1.378 (6) |
| N9—C27 | 1.324 (6) | C22—H22 | 0.9300 |
| N9—C28 | 1.345 (6) | C23—C24 | 1.503 (6) |
| N10—C27 | 1.324 (6) | C24—H24A | 0.9600 |
| N10—C31 | 1.325 (6) | C24—H24B | 0.9600 |
| C1—C2 | 1.526 (5) | C24—H24C | 0.9600 |
| C1—H1A | 0.9700 | C25—C26 | 1.464 (6) |
| C1—H1B | 0.9700 | C25—H25A | 0.9700 |
| C2—H2A | 0.9700 | C25—H25B | 0.9700 |
| C2—H2B | 0.9700 | C26—H26A | 0.9700 |
| C4—C6 | 1.380 (6) | C26—H26B | 0.9700 |
| C4—C5 | 1.498 (6) | C28—C30 | 1.369 (8) |
| C5—H5A | 0.9600 | C28—C29 | 1.501 (8) |
| C5—H5B | 0.9600 | C29—H29A | 0.9600 |
| C5—H5C | 0.9600 | C29—H29B | 0.9600 |
| C6—C7 | 1.380 (6) | C29—H29C | 0.9600 |
| C6—H6 | 0.9300 | C30—C31 | 1.387 (8) |
| C7—C8 | 1.497 (5) | C30—H30 | 0.9300 |
| C8—H8A | 0.9600 | C31—C32 | 1.495 (8) |
| C8—H8B | 0.9600 | C32—H32A | 0.9600 |
| C8—H8C | 0.9600 | C32—H32B | 0.9600 |
| C9—C10 | 1.524 (5) | C32—H32C | 0.9600 |
| C9—H9A | 0.9700 | ||
| C1—S1—S2 | 104.28 (16) | N5—C15—C14 | 120.6 (4) |
| C17—S2—S1 | 104.44 (18) | N5—C15—C16 | 117.3 (4) |
| C11—S3—C10 | 102.6 (2) | C14—C15—C16 | 122.1 (4) |
| C27—S4—C26 | 102.5 (2) | C15—C16—H16A | 109.5 |
| C3—N1—C2 | 121.5 (3) | C15—C16—H16B | 109.5 |
| C3—N1—C9 | 119.8 (3) | H16A—C16—H16B | 109.5 |
| C2—N1—C9 | 118.3 (3) | C15—C16—H16C | 109.5 |
| C4—N2—C3 | 116.0 (3) | H16A—C16—H16C | 109.5 |
| C7—N3—C3 | 116.3 (3) | H16B—C16—H16C | 109.5 |
| C11—N4—C12 | 115.6 (4) | C18—C17—S2 | 108.2 (5) |
| C15—N5—C11 | 116.0 (4) | C18—C17—H17A | 110.0 |
| C19—N6—C18 | 121.8 (4) | S2—C17—H17A | 110.0 |
| C19—N6—C25 | 121.0 (4) | C18—C17—H17B | 110.0 |
| C18—N6—C25 | 117.2 (4) | S2—C17—H17B | 110.0 |
| C20—N7—C19 | 115.3 (4) | H17A—C17—H17B | 108.4 |
| C23—N8—C19 | 116.3 (4) | C17—C18—N6 | 107.1 (5) |
| C27—N9—C28 | 115.1 (4) | C17—C18—H18A | 110.3 |
| C27—N10—C31 | 115.9 (5) | N6—C18—H18A | 110.3 |
| C2—C1—S1 | 114.9 (3) | C17—C18—H18B | 110.3 |
| C2—C1—H1A | 108.5 | N6—C18—H18B | 110.3 |
| S1—C1—H1A | 108.5 | H18A—C18—H18B | 108.6 |
| C2—C1—H1B | 108.5 | N7—C19—N8 | 126.8 (4) |
| S1—C1—H1B | 108.5 | N7—C19—N6 | 117.4 (4) |
| H1A—C1—H1B | 107.5 | N8—C19—N6 | 115.7 (4) |
| N1—C2—C1 | 113.8 (3) | N7—C20—C22 | 121.7 (4) |
| N1—C2—H2A | 108.8 | N7—C20—C21 | 115.1 (5) |
| C1—C2—H2A | 108.8 | C22—C20—C21 | 123.2 (5) |
| N1—C2—H2B | 108.8 | C20—C21—H21A | 109.5 |
| C1—C2—H2B | 108.8 | C20—C21—H21B | 109.5 |
| H2A—C2—H2B | 107.7 | H21A—C21—H21B | 109.5 |
| N3—C3—N2 | 126.5 (3) | C20—C21—H21C | 109.5 |
| N3—C3—N1 | 116.0 (3) | H21A—C21—H21C | 109.5 |
| N2—C3—N1 | 117.5 (3) | H21B—C21—H21C | 109.5 |
| N2—C4—C6 | 121.8 (4) | C20—C22—C23 | 118.6 (4) |
| N2—C4—C5 | 116.2 (4) | C20—C22—H22 | 120.7 |
| C6—C4—C5 | 122.0 (4) | C23—C22—H22 | 120.7 |
| C4—C5—H5A | 109.5 | N8—C23—C22 | 121.0 (4) |
| C4—C5—H5B | 109.5 | N8—C23—C24 | 116.6 (4) |
| H5A—C5—H5B | 109.5 | C22—C23—C24 | 122.4 (4) |
| C4—C5—H5C | 109.5 | C23—C24—H24A | 109.5 |
| H5A—C5—H5C | 109.5 | C23—C24—H24B | 109.5 |
| H5B—C5—H5C | 109.5 | H24A—C24—H24B | 109.5 |
| C4—C6—C7 | 118.2 (4) | C23—C24—H24C | 109.5 |
| C4—C6—H6 | 120.9 | H24A—C24—H24C | 109.5 |
| C7—C6—H6 | 120.9 | H24B—C24—H24C | 109.5 |
| N3—C7—C6 | 121.3 (3) | C26—C25—N6 | 107.6 (4) |
| N3—C7—C8 | 116.5 (4) | C26—C25—H25A | 110.2 |
| C6—C7—C8 | 122.2 (4) | N6—C25—H25A | 110.2 |
| C7—C8—H8A | 109.5 | C26—C25—H25B | 110.2 |
| C7—C8—H8B | 109.5 | N6—C25—H25B | 110.2 |
| H8A—C8—H8B | 109.5 | H25A—C25—H25B | 108.5 |
| C7—C8—H8C | 109.5 | C25—C26—S4 | 104.7 (4) |
| H8A—C8—H8C | 109.5 | C25—C26—H26A | 110.8 |
| H8B—C8—H8C | 109.5 | S4—C26—H26A | 110.8 |
| N1—C9—C10 | 114.3 (3) | C25—C26—H26B | 110.8 |
| N1—C9—H9A | 108.7 | S4—C26—H26B | 110.8 |
| C10—C9—H9A | 108.7 | H26A—C26—H26B | 108.9 |
| N1—C9—H9B | 108.7 | N10—C27—N9 | 128.7 (4) |
| C10—C9—H9B | 108.7 | N10—C27—S4 | 111.4 (3) |
| H9A—C9—H9B | 107.6 | N9—C27—S4 | 119.9 (3) |
| C9—C10—S3 | 111.4 (3) | N9—C28—C30 | 120.5 (5) |
| C9—C10—H10A | 109.4 | N9—C28—C29 | 115.7 (5) |
| S3—C10—H10A | 109.4 | C30—C28—C29 | 123.8 (5) |
| C9—C10—H10B | 109.4 | C28—C29—H29A | 109.5 |
| S3—C10—H10B | 109.4 | C28—C29—H29B | 109.5 |
| H10A—C10—H10B | 108.0 | H29A—C29—H29B | 109.5 |
| N4—C11—N5 | 127.8 (4) | C28—C29—H29C | 109.5 |
| N4—C11—S3 | 120.2 (3) | H29A—C29—H29C | 109.5 |
| N5—C11—S3 | 111.9 (3) | H29B—C29—H29C | 109.5 |
| N4—C12—C14 | 121.1 (4) | C28—C30—C31 | 119.5 (5) |
| N4—C12—C13 | 116.3 (4) | C28—C30—H30 | 120.2 |
| C14—C12—C13 | 122.6 (4) | C31—C30—H30 | 120.2 |
| C12—C13—H13A | 109.5 | N10—C31—C30 | 120.3 (5) |
| C12—C13—H13B | 109.5 | N10—C31—C32 | 117.0 (6) |
| H13A—C13—H13B | 109.5 | C30—C31—C32 | 122.8 (5) |
| C12—C13—H13C | 109.5 | C31—C32—H32A | 109.5 |
| H13A—C13—H13C | 109.5 | C31—C32—H32B | 109.5 |
| H13B—C13—H13C | 109.5 | H32A—C32—H32B | 109.5 |
| C12—C14—C15 | 118.9 (4) | C31—C32—H32C | 109.5 |
| C12—C14—H14 | 120.6 | H32A—C32—H32C | 109.5 |
| C15—C14—H14 | 120.6 | H32B—C32—H32C | 109.5 |
| C1—S1—S2—C17 | −77.4 (3) | S1—S2—C17—C18 | −63.6 (5) |
| S2—S1—C1—C2 | −59.5 (3) | S2—C17—C18—N6 | −177.4 (4) |
| C3—N1—C2—C1 | 105.5 (4) | C19—N6—C18—C17 | −85.7 (7) |
| C9—N1—C2—C1 | −81.3 (4) | C25—N6—C18—C17 | 94.5 (6) |
| S1—C1—C2—N1 | −69.4 (4) | C20—N7—C19—N8 | 5.0 (8) |
| C7—N3—C3—N2 | −0.5 (6) | C20—N7—C19—N6 | −172.1 (5) |
| C7—N3—C3—N1 | 179.3 (3) | C23—N8—C19—N7 | −4.0 (8) |
| C4—N2—C3—N3 | 0.7 (6) | C23—N8—C19—N6 | 173.2 (5) |
| C4—N2—C3—N1 | −179.1 (3) | C18—N6—C19—N7 | 1.0 (8) |
| C2—N1—C3—N3 | 178.8 (3) | C25—N6—C19—N7 | −179.2 (5) |
| C9—N1—C3—N3 | 5.8 (5) | C18—N6—C19—N8 | −176.5 (5) |
| C2—N1—C3—N2 | −1.3 (5) | C25—N6—C19—N8 | 3.4 (8) |
| C9—N1—C3—N2 | −174.3 (3) | C19—N7—C20—C22 | −2.4 (7) |
| C3—N2—C4—C6 | −0.6 (6) | C19—N7—C20—C21 | 177.2 (5) |
| C3—N2—C4—C5 | −179.4 (4) | N7—C20—C22—C23 | −0.7 (7) |
| N2—C4—C6—C7 | 0.4 (6) | C21—C20—C22—C23 | 179.7 (4) |
| C5—C4—C6—C7 | 179.1 (4) | C19—N8—C23—C22 | 0.4 (7) |
| C3—N3—C7—C6 | 0.3 (5) | C19—N8—C23—C24 | −178.9 (4) |
| C3—N3—C7—C8 | 179.3 (3) | C20—C22—C23—N8 | 1.8 (7) |
| C4—C6—C7—N3 | −0.2 (6) | C20—C22—C23—C24 | −179.1 (4) |
| C4—C6—C7—C8 | −179.2 (4) | C19—N6—C25—C26 | 71.8 (6) |
| C3—N1—C9—C10 | −77.2 (4) | C18—N6—C25—C26 | −108.3 (5) |
| C2—N1—C9—C10 | 109.6 (4) | N6—C25—C26—S4 | 173.0 (3) |
| N1—C9—C10—S3 | −175.1 (3) | C27—S4—C26—C25 | −86.1 (4) |
| C11—S3—C10—C9 | 82.3 (3) | C31—N10—C27—N9 | 1.8 (8) |
| C12—N4—C11—N5 | −0.9 (6) | C31—N10—C27—S4 | −179.1 (4) |
| C12—N4—C11—S3 | 179.1 (3) | C28—N9—C27—N10 | −0.2 (7) |
| C15—N5—C11—N4 | 1.5 (6) | C28—N9—C27—S4 | −179.3 (4) |
| C15—N5—C11—S3 | −178.5 (3) | C26—S4—C27—N10 | −167.7 (3) |
| C10—S3—C11—N4 | 5.5 (4) | C26—S4—C27—N9 | 11.5 (4) |
| C10—S3—C11—N5 | −174.5 (3) | C27—N9—C28—C30 | −1.7 (7) |
| C11—N4—C12—C14 | 0.4 (6) | C27—N9—C28—C29 | 179.1 (5) |
| C11—N4—C12—C13 | 179.1 (4) | N9—C28—C30—C31 | 2.0 (9) |
| N4—C12—C14—C15 | −0.4 (7) | C29—C28—C30—C31 | −178.9 (5) |
| C13—C12—C14—C15 | −179.1 (4) | C27—N10—C31—C30 | −1.4 (8) |
| C11—N5—C15—C14 | −1.4 (6) | C27—N10—C31—C32 | 179.8 (6) |
| C11—N5—C15—C16 | 179.8 (4) | C28—C30—C31—N10 | −0.3 (9) |
| C12—C14—C15—N5 | 1.0 (7) | C28—C30—C31—C32 | 178.4 (6) |
| C12—C14—C15—C16 | 179.7 (4) |
Footnotes
Supplementary data and figures for this paper are available from the IUCr electronic archives (Reference: XU5021).
References
- Barbour, L. J. (2001). J. Supramol. Chem.1, 189–191.
- Chen, S.-K., Li, L., Tian, L. & Wu, J.-Y. (2007). Acta Cryst. E63, o1126–o1127.
- Oxford Diffraction (2009). CrysAlis PRO and CrysAlis RED Oxford Diffraction Ltd, Yarnton, England.
- Sheldrick, G. M. (2008). Acta Cryst. A64, 112–122. [DOI] [PubMed]
- Wang, M., Cheng, L.-H. & Wang, A.-M. (2007). Acta Cryst. E63, o3296.
- Westrip, S. P. (2010). J. Appl. Cryst.43, 920–925.
- Wu, G.-H., Liu, T.-B., Peng, Y.-F. & Wu, G.-Z. (2007a). Acta Cryst. E63, o4349.
- Wu, G.-H., Wu, X.-M., Zhang, J.-P. & Liu, T.-B. (2007b). Acta Cryst. E63, o3776.
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.
Supplementary Materials
Crystal structure: contains datablocks global, I. DOI: 10.1107/S1600536810035245/xu5021sup1.cif
Structure factors: contains datablocks I. DOI: 10.1107/S1600536810035245/xu5021Isup2.hkl
Additional supplementary materials: crystallographic information; 3D view; checkCIF report



