Abstract
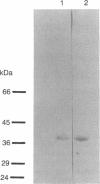
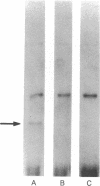
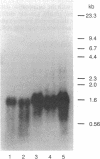
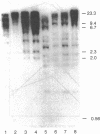
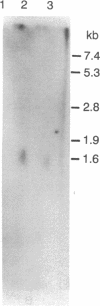
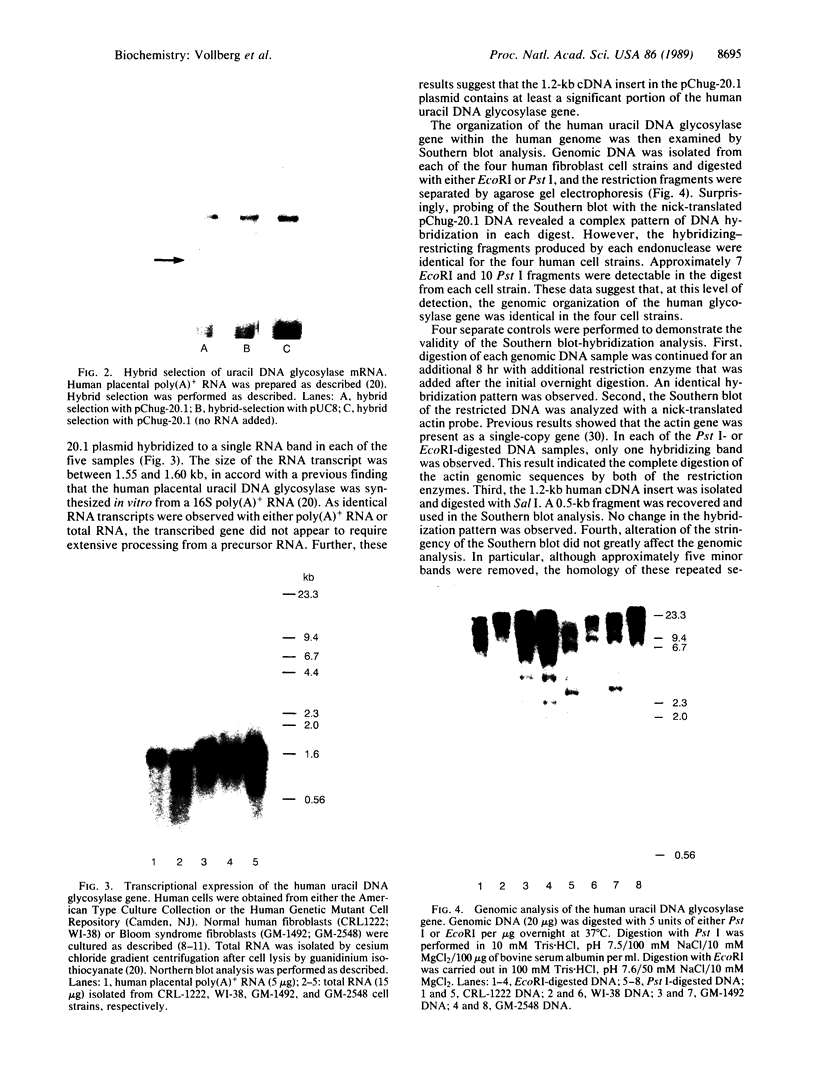
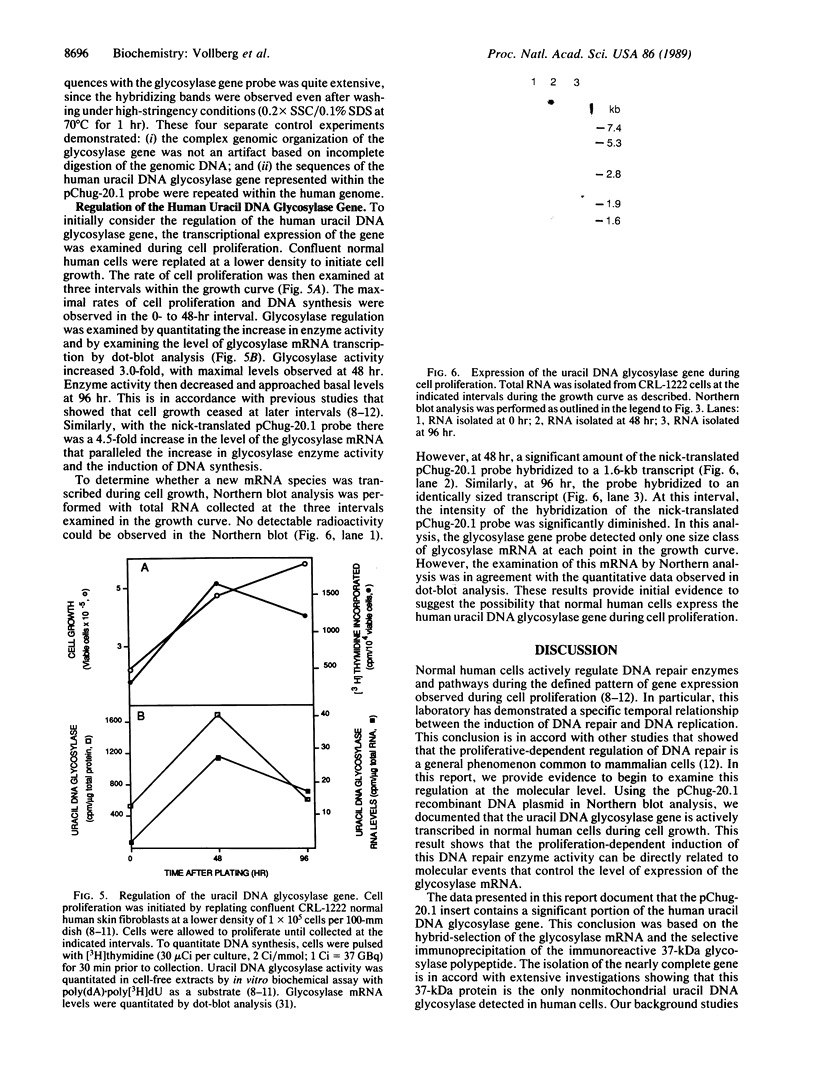
A series of anti-human placental uracil DNA glycosylase monoclonal antibodies was used to screen a human placental cDNA library in phage lambda gt11. Twenty-seven immunopositive plaques were detected and purified. One clone containing a 1.2-kilobase (kb) human cDNA insert was chosen for further study by insertion into pUC8. The resultant recombinant plasmid selected by hybridization a human placental mRNA that encoded a 37-kDa polypeptide. This protein was immunoprecipitated specifically by an anti-human placental uracil DNA glycosylase monoclonal antibody. RNA blot-hybridization (Northern) analysis using placental poly(A)+ RNA or total RNA from four different human fibroblast cell strains revealed a single 1.6-kb transcript. Genomic blots using DNA from each cell strain digested with either EcoRI or Pst I revealed a complex pattern of cDNA-hybridizing restriction fragments. The genomic analysis for each enzyme was highly similar in all four human cell strains. In contrast, a single band was observed when genomic analysis was performed with the identical DNA digests with an actin gene probe. During cell proliferation there was an increase in the level of glycosylase mRNA that paralleled the increase in uracil DNA glycosylase enzyme activity. The isolation of the human uracil DNA glycosylase gene permits an examination of the structure, organization, and expression of a human DNA repair gene.
Full text
PDF


Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Arenaz P., Sirover M. A. Isolation and characterization of monoclonal antibodies directed against the DNA repair enzyme uracil DNA glycosylase from human placenta. Proc Natl Acad Sci U S A. 1983 Oct;80(19):5822–5826. doi: 10.1073/pnas.80.19.5822. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bessman M. J., Lehman I. R., Adler J., Zimmerman S. B., Simms E. S., Kornberg A. ENZYMATIC SYNTHESIS OF DEOXYRIBONUCLEIC ACID. III. THE INCORPORATION OF PYRIMIDINE AND PURINE ANALOGUES INTO DEOXYRIBONUCLEIC ACID. Proc Natl Acad Sci U S A. 1958 Jul 15;44(7):633–640. doi: 10.1073/pnas.44.7.633. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohr V. A., Okumoto D. S., Hanawalt P. C. Survival of UV-irradiated mammalian cells correlates with efficient DNA repair in an essential gene. Proc Natl Acad Sci U S A. 1986 Jun;83(11):3830–3833. doi: 10.1073/pnas.83.11.3830. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Bohr V. A., Smith C. A., Okumoto D. S., Hanawalt P. C. DNA repair in an active gene: removal of pyrimidine dimers from the DHFR gene of CHO cells is much more efficient than in the genome overall. Cell. 1985 Feb;40(2):359–369. doi: 10.1016/0092-8674(85)90150-3. [DOI] [PubMed] [Google Scholar]
- Church G. M., Gilbert W. Genomic sequencing. Proc Natl Acad Sci U S A. 1984 Apr;81(7):1991–1995. doi: 10.1073/pnas.81.7.1991. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Dehazya P., Bell J., Sirover M. A. Proliferation-dependent regulation of base excision repair in normal and SV-40 transformed human cells. Carcinogenesis. 1986 Apr;7(4):621–625. doi: 10.1093/carcin/7.4.621. [DOI] [PubMed] [Google Scholar]
- DiLella A. G., Woo S. L. Cloning large segments of genomic DNA using cosmid vectors. Methods Enzymol. 1987;152:199–212. doi: 10.1016/0076-6879(87)52021-3. [DOI] [PubMed] [Google Scholar]
- Domena J. D., Mosbaugh D. W. Purification of nuclear and mitochondrial uracil-DNA glycosylase from rat liver. Identification of two distinct subcellular forms. Biochemistry. 1985 Dec 3;24(25):7320–7328. doi: 10.1021/bi00346a045. [DOI] [PubMed] [Google Scholar]
- Gunning P., Ponte P., Kedes L., Eddy R., Shows T. Chromosomal location of the co-expressed human skeletal and cardiac actin genes. Proc Natl Acad Sci U S A. 1984 Mar;81(6):1813–1817. doi: 10.1073/pnas.81.6.1813. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gunning P., Ponte P., Kedes L., Hickey R. J., Skoultchi A. I. Expression of human cardiac actin in mouse L cells: a sarcomeric actin associates with a nonmuscle cytoskeleton. Cell. 1984 Mar;36(3):709–715. doi: 10.1016/0092-8674(84)90351-9. [DOI] [PubMed] [Google Scholar]
- Gupta P. K., Sirover M. A. Cell cycle regulation of DNA repair in normal and repair deficient human cells. Chem Biol Interact. 1981 Jul;36(1):19–31. doi: 10.1016/0009-2797(81)90026-0. [DOI] [PubMed] [Google Scholar]
- Gupta P. K., Sirover M. A. Sequential stimulation of DNA repair and DNA replication in normal human cells. Mutat Res. 1980 Sep;72(2):273–284. doi: 10.1016/0027-5107(80)90042-1. [DOI] [PubMed] [Google Scholar]
- Gupta P. K., Sirover M. A. Stimulation of the nuclear uracil DNA glycosylase in proliferating human fibroblasts. Cancer Res. 1981 Aug;41(8):3133–3136. [PubMed] [Google Scholar]
- Hayatsu H. Co-operative mutagenic actions of bisulfite and nitrogen nucleophiles. J Mol Biol. 1977 Sep;115(1):19–31. doi: 10.1016/0022-2836(77)90243-1. [DOI] [PubMed] [Google Scholar]
- Henner W. D., Kiker N. P., Jorgensen T. J., Munck J. N. Purification and amino-terminal amino acid sequence of an apurinic/apyrimidinic endonuclease from calf thymus. Nucleic Acids Res. 1987 Jul 24;15(14):5529–5544. doi: 10.1093/nar/15.14.5529. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Kane C. M., Linn S. Purification and characterization of an apurinic/apyrimidinic endonuclease from HeLa cells. J Biol Chem. 1981 Apr 10;256(7):3405–3414. [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Leblanc J. P., Laval J. Comparison at the molecular level of uracil-DNA glycosylases from different origins. Biochimie. 1982 Aug-Sep;64(8-9):735–738. doi: 10.1016/s0300-9084(82)80120-x. [DOI] [PubMed] [Google Scholar]
- Madhani H. D., Bohr V. A., Hanawalt P. C. Differential DNA repair in transcriptionally active and inactive proto-oncogenes: c-abl and c-mos. Cell. 1986 May 9;45(3):417–423. doi: 10.1016/0092-8674(86)90327-2. [DOI] [PubMed] [Google Scholar]
- Ploegh H. L., Orr H. T., Strominger J. L. Molecular cloning of a human histocompatibility antigen cDNA fragment. Proc Natl Acad Sci U S A. 1980 Oct;77(10):6081–6085. doi: 10.1073/pnas.77.10.6081. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Seal G., Arenaz P., Sirover M. A. Purification and properties of the human placental uracil DNA glycosylase. Biochim Biophys Acta. 1987 Aug 13;925(2):226–233. doi: 10.1016/0304-4165(87)90113-9. [DOI] [PubMed] [Google Scholar]
- Seal G., Brech K., Karp S. J., Cool B. L., Sirover M. A. Immunological lesions in human uracil DNA glycosylase: association with Bloom syndrome. Proc Natl Acad Sci U S A. 1988 Apr;85(7):2339–2343. doi: 10.1073/pnas.85.7.2339. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Shaper N. L., Grafstrom R. H., Grossman L. Human placental apurinic/apyrimidinic endonuclease. Its isolation and characterization. J Biol Chem. 1982 Nov 25;257(22):13455–13458. [PubMed] [Google Scholar]
- Shapiro R., Klein R. S. The deamination of cytidine and cytosine by acidic buffer solutions. Mutagenic implications. Biochemistry. 1966 Jul;5(7):2358–2362. doi: 10.1021/bi00871a026. [DOI] [PubMed] [Google Scholar]
- Strauss B. S. Cellular aspects of DNA repair. Adv Cancer Res. 1985;45:45–105. doi: 10.1016/s0065-230x(08)60266-3. [DOI] [PubMed] [Google Scholar]
- Teebor G. W., Frenkel K. The initiation of DNA excision-repair. Adv Cancer Res. 1983;38:23–59. doi: 10.1016/s0065-230x(08)60186-4. [DOI] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tye B. K., Chien J., Lehman I. R., Duncan B. K., Warner H. R. Uracil incorporation: a source of pulse-labeled DNA fragments in the replication of the Escherichia coli chromosome. Proc Natl Acad Sci U S A. 1978 Jan;75(1):233–237. doi: 10.1073/pnas.75.1.233. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vollberg T. M., Cool B. L., Sirover M. A. Biosynthesis of the human base excision repair enzyme uracil-DNA glycosylase. Cancer Res. 1987 Jan 1;47(1):123–128. [PubMed] [Google Scholar]
- Vollberg T. M., Seal G., Sirover M. A. Monoclonal antibodies detect conformational abnormality of uracil DNA glycosylase in Bloom's syndrome cells. Carcinogenesis. 1987 Nov;8(11):1725–1729. doi: 10.1093/carcin/8.11.1725. [DOI] [PubMed] [Google Scholar]
- Vos J. M., Hanawalt P. C. Processing of psoralen adducts in an active human gene: repair and replication of DNA containing monoadducts and interstrand cross-links. Cell. 1987 Aug 28;50(5):789–799. doi: 10.1016/0092-8674(87)90337-0. [DOI] [PubMed] [Google Scholar]
- White B. A., Bancroft F. C. Cytoplasmic dot hybridization. Simple analysis of relative mRNA levels in multiple small cell or tissue samples. J Biol Chem. 1982 Aug 10;257(15):8569–8572. [PubMed] [Google Scholar]
- Wittwer C. U., Bauw G., Krokan H. E. Purification and determination of the NH2-terminal amino acid sequence of uracil-DNA glycosylase from human placenta. Biochemistry. 1989 Jan 24;28(2):780–784. doi: 10.1021/bi00428a055. [DOI] [PubMed] [Google Scholar]