Abstract
We studied a family in which the same 10 Mb inverted duplication of 2p25.3–p25.1 segregates in two children and their father, all showing a trisomy phenotype. As FISH analysis demonstrated that the duplication was inverted, we suspected that a contiguous terminal deletion was also present, according to the classical inv dup del type of rearrangements. Although FISH with 2p and 2q subtelomeric probes gave normal results, 100 kb resolution array-CGH (aCGH) showed that, beside the duplication, a 273 kb deletion was also present. The presence of a single-copy region between the deleted and duplicated regions was further suspected through high-resolution aCGH analysis (∼20 kb), although only one informative spot having a normal log ratio was detected. The precise structure of the rearrangement was re-defined by real-time PCR and breakpoint cloning, demonstrating the presence of a 2680 bp single-copy sequence between deleted and duplicated regions and the involvement of a simple repeat with the potential for forming a non-B DNA structure. The rearrangement was not mediated by segmental duplications or short inverted repeats, and the double-strand break might have been repaired by non-homologous end joining or microhomology-mediated intrastrand repair. These data highlight the fact that concomitant deletions associated with inverted duplications are very likely to be more frequent than classical cytogenetic methods alone have been able to demonstrate. The phenotypic effects of the trisomy and of the terminal 2p deletion are discussed.
Keywords: inverted deletion duplication, 2p trisomy, non-homologous end joining
Introduction
Unbalanced structural chromosome abnormalities detectable with moderate levels of banding are present in approximately 0.04% of unselected newborns.1 Among these, chromosome duplications have an approximate frequency of 1:4000 in the general population 2 and are found in about 2% of subjects with a typical chromosomal phenotype.3 According to the orientation of the duplicated segment, duplications may be either in tandem or inverted.4 Segmental duplications have a primary function in both types of rearrangement by causing non-allelic homologous recombination (NAHR).5 Unlike direct duplications, inverted duplications are usually associated with deletion of the distal region of the duplicated chromosome. Between duplicated and deleted region, a single-copy region flanked by homologous segmental duplications may be present. The inv dup(8p) is the paradigmatic example of this type of rearrangement.6, 7
Here, we report a family where two children and their affected father carry the same dup(2)(p25.3p25.1) detected by karyotype analysis. Molecular characterization by FISH and array-CGH (aCGH) analyses demonstrated that the rearranged chromosome was an inverted duplication of 2p spanning no less then 10 Mb, associated with a 270 kb deletion of 2pter. The presence of a single-copy region located between the duplicated and deleted regions was demonstrated by different molecular techniques. Breakpoint cloning demonstrated the involvement of a tetraplex-forming low complexity repeat in the rearrangement. A fine genotype/phenotype correlation was also carried out.
Patients and methods
Clinical report
Two children (Case1 and Case 2) and their father (Case 3) had mental retardation.
The oldest child (Case 1) is a male born at term by cesarean section delivery. His mother suffered from threatened abortion during the first 6 months of pregnancy and was treated with bed rest.
His birth weight was 3200 g (50th centile); head circumference and Apgar scores were not available. Psychomotor development revealed slight postural, motor and speech delay, with severe learning difficulties and behavioral problems (introversion, lack of initiative, passivity, negativism). He came for the first time to our examination when he was 11 years old. He showed dysmorphic features including hypertelorism, ogival palate, abnormal ear shape (big and irregular), high forehead with prominence of the frontal eminences, thin upper lip, flat philtrum. When examined at the age of 12 years, he had thoracolumbar kyphosis. Oculistic examination showed myopia.
At last evaluation at the age of 15 years, he had moderate MR (overall IQ: 54 at WISC-R scale). He shows difficulties in both verbal and non-verbal skills; he had slightly dysarthric, slow, contract, lexically poor speech (verbal IQ: 55; performance IQ: 61). Difficulties in fine motor coordination and praxic organization, lack of memory skills in all fields examined and difficulties in planning were also noted.
His younger sister (Case 2) was born at term by cesarean section after a complicated pregnancy (gestosis at the end). Her birth weight was 2800 g (10th centile); head circumference and Apgar scores were not available. Psychomotor development revealed slight postural and motor delay, and severe speech delay. On last evaluation at the age of 12 years, she had severe MR (overall IQ: <30). She was able to speak only a few words. She had a low level of personal autonomy and did not completely reach sphincter control. Behavioral and relational problems were present, with autistic-like conduct. Facial dysmorphisms, including hypertelorism, sharpened nose, ogival palate, low-set, big and irregular ears, narrow forehead, thin upper lip and short philtrum were noted. Oculistic examination revealed esotropia and myopia; kyphosis-scoliosis and chronic, catarrhal rinopathy were also present.
Their father (Case 3), on examination at the age of 37 years, was affected by moderate MR and showed dysmorphic features including hypertelorism, high forehead, low-set ears, thin upper lip, flat philtrum. Oculistic examination revealed esotropia. Their mother was 38 years old; she suffered from epilepsy and was taking daily drug therapy (carbamazepine, 1200 mg/day).
Cytogenetic analysis
High-resolution G-banded chromosomes were prepared from peripheral blood lymphocytes according to the standard protocols.
aCGH analysis
DNA from the children and their father was extracted from venous blood with the standard protocols.
Molecular karyotyping was performed using the aCGH kits (Agilent Technologies, Santa Clara, CA, USA). These platforms are 60-mer oligonucleotide-based microarrays that allow genome-wide survey and molecular profiling of genomic aberrations with a resolution of ∼100 kb (kit 44B) and ∼20 kb (kit 244A). DNAs (500 ng) from the patients and a reference of the same sex (Promega, Madison, WI, USA) were double-digested with RsaI and AluI for 2 h at 37°C. After heat inactivation of the enzymes at 65°C for 20 min, each digested sample was labeled by random priming (Agilent) for 2 h using Cy5-dUTP for patient DNAs and Cy3-dUTP for reference DNAs. Labeled products were column purified. After probe denaturation and pre-annealing with 20 μg of Cot-1 DNA, hybridization was performed at 65°C with rotation for 24 h (44B) or 40 h (244A). After two washing steps, the array was analyzed through the Agilent scanner with Feature Extraction software (v9.1).
FISH analysis
FISH was performed on metaphase cells according to the standard procedures.
BAC clones were selected from the human RPCI-11 library according to the UCSC Human Genome May 2004 assembly (http://genome.ucsc.edu/cgi-bin/hgGateway) and provided by the BACPAC Resource Center (BPRC) at the Children's Hospital Oakland Research Institute in Oakland, CA, USA (http://bacpac.chori.org/). BAC DNAs were labeled either by biotin-16-dUTP or by digoxigenin using a nick translation kit (Roche). The chromosomes were counterstained with DAPI (Sigma Aldrich, St Louis, MO, USA). Double-color FISH was performed with RP11-90H11 (2p25.3 at 2.3 Mb) and RP11-1B18 (2p25.1 at 8.7 Mb) labeled with biotin and digoxigenin, respectively. The labeled probes were visualized with FITC-avidin (Vector, Burlingame, CA, USA) or Rhodamin-conjugated antidigoxigenin (Sigma). Hybridizations were analyzed using an Olympus BX71 epifluorescence microscope and images captured with the Power Gene FISH System (PSI, Newcastle upon Tyne, UK). For chromosome 2, whole chromosome painting with a chromosome 2-specific library (Vysis, Abbott Molecular, Abbott Park, IL, USA) and the commercial probe TelVysion probe specific for the subtelomeric 2p region (Vysis) was used according to the manufacturer's protocols.
Real-time PCR
On the basis of the coordinates from the aCGH experiment, primer pairs for quantitative PCR analysis were selected within non-repeated portions of the chromosome using Primer Express software (Applied Biosystems, Foster City, CA, USA); a control amplicon was selected with the same parameters in the MAPK1 gene on 22q11.2; size (approximately 60 nt) and Tm (58°C) were the same for all amplicons. Primer sequences are available from the authors. Amplification and detection were performed on a ABI PRISM 7700 Sequence Detection System (Applied Biosystems) using SYBR Green PCR Master Mix (Applied Biosystems); thermal cycling conditions were 50°C for 2 min and 95°C for 10 min, followed by 40 cycles of 95°C for 15 s and 60°C for 1 min; all samples were amplified in duplicate. Validation experiments demonstrated that amplification efficiencies of the control and all target amplicons were approximately equal; accordingly, relative quantification of the amount of DNA was obtained using the comparative CT method (described in Applied Biosystems User Bulletin no. 2, 11 December 1997: ABI PRISM 7700 Sequence Detection System).
Breakpoint amplification and cloning
Long-range PCRs were performed with JumpStart Red ACCUTaq LA DNA polymerase (Sigma) and the following protocol: 30 s at 96°C, 35 cycles of 15 s at 94°C/20 s at 58°C/5 min at 68°C, 5 min final elongation time. Sequencing reactions were performed with a Big Dye Terminator Cycle Sequencing kit (Applied Biosystems) and carried out on an ABI Prism 3130xl Genetic Analyzer.
DNA sequence analysis
Quadruplex-forming sequences prediction was performed using the QGRS mapper software (http://bioinformatics.ramapo.edu/QGRS/analyze.php).
Results
Cytogenetic analysis
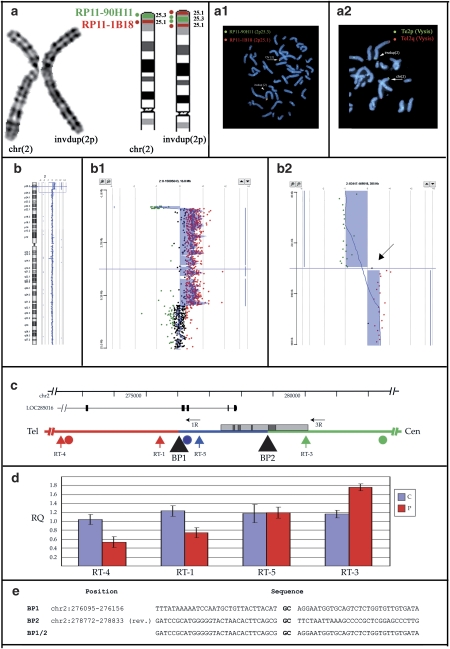
Cytogenetic investigations showed supernumerary bands on the distal p-arm of one chromosome 2 in the father and both children (Figure 1a).
Figure 1.
Overview of molecular data from Case 2. Identical results were obtained on Case 1 and Case 3. (a) Cytogenetics and FISH results. (from left) cut-out of the normal and abnormal chromosomes 2 in G-banding at a resolution of 550 bands. Ideogram of normal and inverted duplicated chromosomes 2p: th 2p25.3 and 2p25.1 bands are depicted in green and red, respectively. The dots represent BAC clones RP11-90H11 (2p25.3, green dot) and RP11-1B18 (2p25.1, red dot). Double-colour FISH with BACs RP11-90H11 (green signal) and RP11-1B18 (red signal): the normal chromosome 2 (arrow) has two signals. The order of red and green signals in the dup(2) (arrowhead) demonstrates that the duplication is inverted (a.1). FISH with subtelomeric 2p probe (green signal) and 2q (red signal) (Vysis) showed hybridization signals on both normal (arrow) and invdup (arrowhed) chromosome 2 (a.2). (b) Array-CGH (aCGH) analysis. aCGH profile of chromosome 2 showing the deletion/duplication at 2p25.3–25.1. Enlargement of the deleted and duplicated regions (b.2). On the right, detailed view of deletion/duplication boundary. The arrow points to a single probe with a log ratio of +0.2 located between the deleted and duplicated regions (b.3). (c) Detail of the breakpoint region on chromosome 2p. The deletion is shown in red, the duplication in green, the single-copy region in blue. The locations of the breakpoints (black triangles), aCGH (circles) and real-time (vertical arrows) probes, and long-range PCR primers (horizontal arrows) are indicated. The positions of the LOC285016 gene and of a CpG island (gray box) containing several repetitive elements (dark gray boxes) are also shown. (d) Real-time PCR results. Average Relative Quantification (RQ) by real-time PCR of Cases 1–3 (P) and three normal controls (c). (e) Sequence of the breakpoints. The CG dinucleotide representing the only homology between the two breakpoints is highlighted in bold.
FISH analysis
FISH investigations were performed on the two children and their affected father. Chromosome 2 painting (Vysis) showed homogeneous staining along normal and abnormal chromosomes 2. To determine how the duplication was organized, BAC clones from the human RPCI-11 library were hybridized on metaphase spread of patients' lymphocytes, and double-color FISH revealed that the duplication was inverted (Figure 1a.1).
The subtelomeric TelVysion 2p (Vysis) probe showed hybridization signals of equal intensity on both chromosome 2 homologs (Figure 1a.2).
Thus, the whole of the molecular data led to the final interpretation of the chromosome rearrangement consisting of add(2)(p25.3).ish invdup(2)(qter−>p25.3∷p25.3−>p25.1)(wcp2+, tel2p+, RP11−1B18++, RP11−90H11++).
aCGH analysis
To characterize the rearranged chromosome region, aCGH was performed on DNA from all affected subjects in the family. The 44k oligo chip, with a resolution of 100 kb, showed a terminal 2p deletion of about 300 kb (first spotted deleted oligomer at 29.193 kb; last deleted oligomer at 0.264 Mb) concurrent to a duplicated region of about 10 Mb (first duplicated oligomer at 0.469 Mb, last duplicated oligomer at 10.405 Mb; first normal oligomer at 10.440 Mb) (not shown). The high-resolution 244k platform (Agilent, Figure 1b.1) allowed a finer definition of the breakpoints and the identification of a single probe with a log ratio of +0.2 (Figure 1b.2), possibly indicating a single-copy region, located between the deleted and duplicated regions. The deleted region was shown to span the oligomers at 20.341 kb (first spotted and deleted) and 273.154 kb (last deleted); the oligomer at 276.329 kb resulted normal (log ratio: +0.2); the duplicated region started from the oligomer at 282.206 kb and the distal duplication breakpoint lay between oligomers at 10.404 Mb (last duplicated) and 10.417 Mb (first normal).
Real-time PCR analysis, breakpoint cloning and sequencing
To confirm the arrangement identified by aCGH analysis, we performed a quantitative analysis by real-time PCR on the three carriers and three normal controls. We selected four primer pairs along the chromosome 2 deletion-duplication boundary: one (RT-4) distal to the last deleted aCGH probe; one (RT-1) between the last deleted and normal probes; two (RT-5 and RT-3) between the normal and first duplicated probes (Figure 1c). The analysis confirmed decreased copy number for RT-4 and RT-1, consistent with the presence of a deletion; RT-5 showed normal copy number, whereas RT-3 had increased copy number, consistent with a duplication (Figure 1d).
We amplified the breakpoint region by long-range PCR with primers dup2-1R and dup2-3R (Figure 1c), and obtained a 1.5 kb band from both affected children and their father (not shown). No bands were obtained from normal control subjects. Sequencing of the amplified band revealed the location of both the deleted/normal and normal/duplicated region breakpoints (Figure 1e). Although the breakpoint between normal and deleted sequences is in a region without any apparent features, the breakpoint between normal and duplicated sequence is located within a large CpG island, at a distance of 41 bp from a 250 bp C-rich repetitive element (chr2:278844–279081). The (−) strand of this repeat is recognized as a potential quadruplex-forming G-rich sequence (QGRS) with a predictive score of 136.
Discussion
The rearrangement
Array CGH analysis has markedly enhanced the resolution of chromosome studies and enabled high-resolution genome analysis, thus proving a more accurate method for the identification and delineation of chromosomal rearrangements.8 As a result, precise definitions of complex chromosome rearrangements and their true complexity can now be better established.9
The multifaceted demonstration of the real complexity of a rearrangement is well illustrated by the analysis of our family. The karyotype revealed an abnormal chromosome 2 in both children and their father (Figure 1a). FISH analysis with BAC clones allowed us to establish the orientation of the 2p duplicated region and to demonstrate that it was an inverted duplication in all affected family members (Figure 1a.1).
Considering that inverted duplications are usually associated with the deletion of the contiguous distal region,6, 10, 11 FISH analysis with a 2p subtelomeric probe was performed, but no deletion was detected: hybridization signals of equal intensity were found on the rearranged 2p chromosome and its normal homolog (Figure 1a.2). The commercial 2ptel probe (Vysis) is obtained by Alu-PCR from the 340 kb half-YAC clone yRM2052, presumably containing the terminal 320 kb of chromosome 2p, as one of its ends (U32389) maps at chr2:322536–322595.12
As telomeric FISH markers can be located even at 300–400 kb proximal to the telomeric end, we postulated that the preservation of tel2p probe signals could represent either a single-copy region between the duplicated portion and a more distal deleted region or the start point of the duplicated region. Further experiments demonstrated that the second alternative was correct: the probe overlaps both the deleted segment and a portion of the duplicated sequence. aCGH analysis using oligo chips with a resolution of about 100 kb allowed the detection of a terminal deletion of ∼273 kb. The presence of a single-copy region between the deleted and duplicated regions was further suspected through high-resolution aCGH analysis at a resolution of ∼20 kb, although only one informative spot was detected having a normal log ratio (Figure 1b.2). Quantitative PCR analysis and breakpoint sequencing confirmed the presence of a 2680 bp single-copy sequence (chr2:276125–278802, Figure 1c–e) between the deleted and duplicated regions; within the single-copy sequence lies the single-informative probe (chr2:000276329–000276388) detected by high-resolution aCGH analysis (Figure 1b). As recently described in a study by Smith et al,13 applying a two-stage high-resolution aCGH approach on a cohort of 50 healthy subjects, the detection of just one deleted/normal/duplicated spot has 80% probability to indicate the correct status of that region.13
Duplications contiguous to terminal deletions are known to originate from the breakage of dicentric chromosomes. Dicentric chromosomes can arise in multiple ways (summarized by Narayanan and Lobachev14). Segmental duplications with opposite orientation have a primary function in causing non-allelic homologous recombination leading to a dicentric chromosome.5 Between duplicated and deleted regions, a single-copy region is usually present, flanked by homologous segmental duplications. A typical example of this type of rearrangement is the invdupdel (8p).6, 7 Alternatively, illegitimate repair of a double-strand break (DSB) by non-homologous end joining (NHEJ)15 or intrastrand annealing 16 can also form dicentrics. The rearrangement we analyzed was not mediated by segmental duplications, because none exists in the region. Neither it was facilitated by the presence of short inverted repeats (IR), as no IRs are present, whereas microhomology between the two breakpoint regions is limited to a CG dinucleotide (Figure 1e). The non-duplicated region starts shortly distal to a 250 bp C-rich low-complexity repeat that on the (−) strand is a G-rich sequence with a high-tetraplex-forming score. As tetraplex-forming sequences form stable strand-specific structures and can arrest DNA synthesis,17 we propose that the DSB was probably repaired after 5′-to-3′ resection stalled at the repeat, allowing NHEJ or intrastrand repair by microhomology.
The whole of the molecular data demonstrate that the rearranged chromosome 2p is a classical inverted/duplicated chromosome rearrangement. These data highlight the fact that FISH analysis can improve the study of ‘simple' chromosome rearrangement by providing genomic positional and orientation information of imbalances, but high-resolution aCGH, compared to conventional molecular cytogenetic analysis, reveals the true nature of the rearrangement. In the cases described here, in fact, it was possible to detect the terminal deletion only through aCGH analysis.
Thus, concomitant deletions associated with inverted duplications, both on chromosome 2p and on other chromosomes, are very likely to be more frequent than classical cytogenetic methods alone have been able to demonstrate.
Genotype/phenotype correlation
Until recently, only three cases with pure partial 2p trisomy have been reported in association with a contiguous terminal deletion; all were detected by FISH analysis with the commercially available subtelomeric 2p probe.18, 19, 20 Kochilas et al21 reported a case with a inverted duplication of 2p22–p25. All patients share several features with the 2p23-pter trisomy phenotype, including severe mental retardation, typical facial appearance, ocular anomalies and congenital heart defects.20 Two subjects with pure subtelomeric 2p deletion were recently reported by Ravnan et al22, but the size of the 2p deleted region was not defined at the molecular level (cases 30 and 31). The two patients showed developmental delay, dysmorphic features and a phenotype similar to that of Angelman syndrome.22
A de novo subtelomeric 2p deletion of ∼1.7–1.9 Mb and a partial duplication of 16q were recently described in a subject with MR and behavioral problems but no facial dysmorphisms or physical abnormalities. The authors concluded that either the 2pter deletion or the 16qter duplication, or both, could be contributing to the phenotype.23
In our patients, the size of the 2p inverted duplicated segment was demonstrated to be 10 Mb, spanning from 2p25.3 to 2p25.1. Our patients' phenotypes share some common features with the case described by Gruchy et al consisting of MR, abnormal ear shape, scoliosis (Table 1).
Table 1. Main clinical features in patients with partial trisomy 2p syndrome.
| Main clinical features in 2p23-pter trisomy | Kochilas et al21 | Gruchy et al20 | This paper | ||
|---|---|---|---|---|---|
| Case 1 | Case 2 | Case 3 | |||
| Mental retardation | + (Severe) | + (Severe) | + (Moderate) | + (Severe) | + (Moderate) |
| High forehead with frontal upsweep of hair | + | + | + | − | + |
| Hypertelorism | + | + | + | + | + |
| Short nose | + | + | − | − | − |
| Abnormal ear shape or position | + | + | + | + | + |
| Scoliosis/Kyphosis | +/− | −/+ | +/+ | −/+ | |
| Ocular abnormalities | Optical nerve atrophy | Myopia | Myopia esotropia | Esotropia | |
| Heart defects | + | + | − | − | − |
| Hypoplastic external genitalia | − | − | − | − | − |
| Diaphragmatic hernia | − | − | − | − | − |
| Neural tube defects | − | − | − | − | − |
| Neuroblastoma | − | − | − | − | − |
Our patients did not have the cardiac anomalies reported by Gruchy et al20 and Kochilas et al.21 Thus, a gene responsible for cardiac defects could be located distal to the region duplicated in our cases, unless the gene has reduced penetrance.
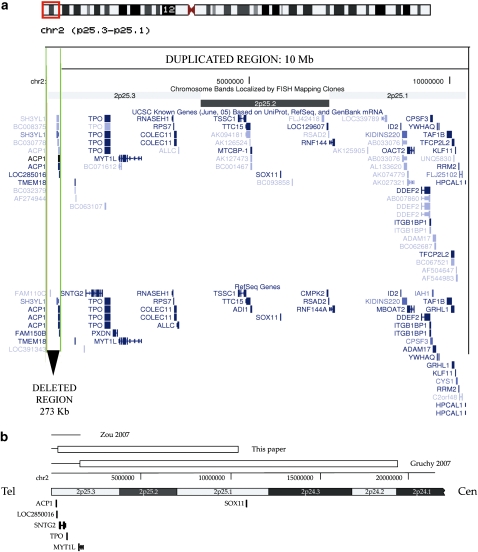
The 2p deletion region of overlap between the cases described here and those recently reported by Gruchy et al20 (del2p of 1.6 Mb) and Zou et al23 (del2p of 1.7–1.9 Mb) encompasses the terminal 273 kb, where three genes lie: SH3YL1, ACP1 and LOC285016 (Figure 2a).
Figure 2.
(a) Schematic representation of the deleted/duplicated region in our patients. Screenshot obtained from http://genome.ucsc.edu, UCSC Genome Browser on Human May 2004 Assembly. The upper part of the figure shows the idiogram of chromosome 2, in which the aberrant region is indicated by a grey square*. The deletion of 273 kb is indicated by the two vertical grey lines** and pointed by a black arrowhead. The duplicated region of 10 Mb is shown by a black square. (b) Structure of the rearrangement and comparison with other recently described 2p rearrangements. Deleted regions are indicated by lines, duplicated regions by boxes. *Red square in the html version, **green lines in the html version.
The human red-cell acid phosphatase encoded by the ACP1 gene (MIM:171500) belongs to the phosphotyrosine protein phosphatase family of proteins. The genes SH3YL1 and LOC285016 code for hypothetical proteins of unknown function. Thus, we cannot exclude that haploinsufficiency of these genes could contribute to the phenotype of our cases.
In our cases the TPO and SNTG2 genes are duplicated, whereas in Gruchy's and Zou's cases they were deleted. The TPO gene codes for an enzyme that has a central function in thyroid gland function. Mutations in TPO are associated with several disorders of thyroid hormonogenesis and are recessively inherited.24 Because no thyroid disorders have been described in the patients with deletion and/or duplication of this region, most likely deletion/duplication of the TPO gene did not contribute to the patients' phenotype.
In the 2p25.3 distal region there are two other genes, both expressed in human neuronal cells: syntrophin-γ2 (SYTG2) and myelin transcription factor 1 (MYT1L). SNTG2 is a dystrophin-binding protein prominently expressed in both rat and human central nervous system (CNS). The gene was duplicated in our cases, deleted in Gruchy's and most likely partially deleted in Zou's cases (Figure 2b). The MYT1L gene is expressed in developing neurons in the mammalian CNS.25 Its developmental expression and localization suggest that it may have a function in the development of neurons and oligodendroglia. Thus, the deletion/duplication of these genes may contribute to psychomotor development delay in these patients.
Moderate-to-severe mental retardation was present both in our patients and in Gruchy's and Aviram–Goldring's patients. Our patients showed the smallest duplicated 2p region (10 Mb) reported so far. Thus, the common duplicated region in all invdup(2p) patients encompasses 8.6 Mb and several genes. Among these, the human SOX11 gene is predominantly expressed in the developing nervous system in both glial and neuronal lineages 26, 27 and may contribute to mental retardation.
Gruchy et al20 hypothesized a possible involvement of the ALK gene, a tyrosine-kinase receptor expressed mainly in the nervous system, in his patient's phenotype. Because this gene was not duplicated in our patients, we can rule out its involvement in the developmental delay of patients with terminal invdup2p.
In conclusion, our results further confirm the usefulness of high-resolution aCGH analysis both in the delineation of chromosome rearrangements and in clinical diagnostic.
Our data also highlight the fact that concomitant deletions associated with inverted duplications, both on chromosome 2p and on other chromosomes, are very likely to be more frequent than classical cytogenetic methods alone have been able to demonstrate.
According to the mechanism we propose, all non-recurrent inverted duplications are mediated by a process involving NHEJ or microhomology-mediated intrastrand repair, which favors intrachromatid misalignment and recombination.
Both in the deleted and duplicated region lie genes whose imbalance could contribute to the patients' phenotype.
Acknowledgments
This study was supported by Telethon Grant GGP06208A to MCB and RG, PRIN 2005 and 2006 to OZ and ER and Fondazione Mariani and Fondazione CARIPLO (all to OZ).
References
- Jacobs PA, Browne C, Gregson N, Joyce C, White H. Estimates of the frequency of chromosome abnormalities detectable in unselected newborns using moderate levels of banding. J Med Genet. 1992;29:103–108. doi: 10.1136/jmg.29.2.103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Phelan MC, Crawford EC, Bealer DM. Mental retardation in South Carolina III. Chromosome aberrations. Proc Greenwood Genet Center. 1996;15:45–60. [Google Scholar]
- Shaffer LG, Lupski JR. Molecular mechanisms for constitutional chromosomal rearrangements in humans. Annu Rev Genet. 2000;34:297–329. doi: 10.1146/annurev.genet.34.1.297. [DOI] [PubMed] [Google Scholar]
- Van Dyke DL, Miller MJ, Weiss L. The origin of inverted tandem duplications, and phenotypic effects of tandem duplication of the X chromosome long arm. Am J Med Genet. 1983;15:441–450. doi: 10.1002/ajmg.1320150309. [DOI] [PubMed] [Google Scholar]
- Shaw CJ, Lupski JR. Implications of human genome architecture for rearrangement-based disorders: the genomic basis of disease. Hum Mol Genet. 2004;13:R57–R64. doi: 10.1093/hmg/ddh073. [DOI] [PubMed] [Google Scholar]
- Floridia G, Piantanida M, Minelli A, et al. The same molecular mechanism at the maternal meiosis I produces mono and dicentric 8p duplications. Am J Hum Genet. 1996;58:785–796. [PMC free article] [PubMed] [Google Scholar]
- Giglio S, Broman KW, Matsumoto N, et al. Olfactory receptor-gene clusters, genomic-inversion polymorphisms, and common chromosome rearrangements. Am J Hum Genet. 2001;68:874–883. doi: 10.1086/319506. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Vissers LE, de Vries BB, Osoegawa K, et al. Array-based comparative genomic hybridization for the genomewide detection of submicroscopic chromosomal abnormalities. Am J Hum Genet. 2003;73:1261–1270. doi: 10.1086/379977. [DOI] [PMC free article] [PubMed] [Google Scholar]
- De Gregori M, Ciccone R, Magini P, et al. Cryptic deletions are a common finding in ‘balanced' reciprocal and complex chromosome rearrangements: a study of 59 patients. J Med Genet. 2007;44:750–762. doi: 10.1136/jmg.2007.052787. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Jenderny J, Poetsch M, Hoeltzenbein M, Friedrich U, Jauch A. Detection of a concomitant distal deletion in an inverted duplication of chromosome 3. Is there an overall mechanism for the origin of such duplications/deficiencies. Eur J Hum Genet. 1998;6:439–444. doi: 10.1038/sj.ejhg.5200217. [DOI] [PubMed] [Google Scholar]
- Bonaglia MC, Giorda R, Poggi G, et al. Inverted duplications are recurrent rearrangements always associated with a distal deletion: description of a new case involving 2q. Eur J Hum Genet. 2000;8:597–603. doi: 10.1038/sj.ejhg.5200509. [DOI] [PubMed] [Google Scholar]
- Macina RA, Morii K, Hu XL, et al. Molecular cloning and RARE cleavage mapping of human 2p, 6q, 8q, 12q, and 18q telomeres. Genome Res. 1995;5:225–232. doi: 10.1101/gr.5.3.225. [DOI] [PubMed] [Google Scholar]
- de Smith AJ, Tsalenko A, Sampas N, et al. Array CGH analysis of copy number variation identifies 1284 new genes variant in healthy white males: implications for association studies of complex diseases. Hum Mol Genet. 2007;16:2783–2794. doi: 10.1093/hmg/ddm208. [DOI] [PubMed] [Google Scholar]
- Narayanan V, Lobachev KS. Intrachromosomal gene amplification triggered by hairpin-capped breaks requires homologous recombination and is independent of nonhomologous end-joining. Cell Cycle. 2007;6:1814–1818. doi: 10.4161/cc.6.15.4522. [DOI] [PubMed] [Google Scholar]
- Okuno Y, Hahn PJ, Gilbert DM. Structure of a palindromic amplicon junction implicates microhomology-mediated end joining as a mechanism of sister chromatid fusion during gene amplification. Nucleic Acids Res. 2004;32:749–756. doi: 10.1093/nar/gkh244. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Tanaka H, Cao Y, Bergstrom DA, Kooperberg C, Tapscott SJ, Yao MC. Intrastrand annealing leads to the formation of a large DNA palindrome and determines the boundaries of genomic amplification in human cancer. Mol Cell Biol. 2007;27:1993–2002. doi: 10.1128/MCB.01313-06. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Woodford KJ, Howell RM, Usdin K. A novel K(+)-dependent DNA synthesis arrest site in a commonly occurring sequence motif in eukaryotes. J Biol Chem. 1994;269:27029–27035. [PubMed] [Google Scholar]
- Aviram-Goldring A, Fritz B, Bartsch C, et al. Molecular cytogenetic studies in three patients with partial trisomy 2p, including CGH from paraffin-embedded tissue. Am J Med Genet. 2000;91:74–82. doi: 10.1002/(sici)1096-8628(20000306)91:1<74::aid-ajmg14>3.0.co;2-o. [DOI] [PubMed] [Google Scholar]
- Thangavelu M, Frolich G, Rogers D. Partial duplication 2p as the sole abnormality in two cases with anencephaly. Am J Med Genet Part A. 2004;124A:170–172. doi: 10.1002/ajmg.a.20322. [DOI] [PubMed] [Google Scholar]
- Gruchy N, Jacquemont ML, Lyonnet S, et al. Recurrent inverted duplication of 2p with terminal deletion in a patient with the classical phenotype of trisomy 2p23-pter. Am J Med Genet. 2007;143:2417–2422. doi: 10.1002/ajmg.a.31931. [DOI] [PubMed] [Google Scholar]
- Kochilas LK, Abuelo DN, Tantravahi U. Bilateral semilunar valve dysplasia in a patient with inverted duplication 2p25-22. Pediatr Cardiol. 2008;29:172–175. doi: 10.1007/s00246-007-9013-2. [DOI] [PubMed] [Google Scholar]
- Ravnan JB, Tepperberg JH, Papenhausen P, et al. Subtelomere FISH analysis of 11 688 cases: an evaluation of the frequency and pattern of subtelomere rearrangements in individuals with developmental disabilities. J Med Genet. 2006;43:478–489. doi: 10.1136/jmg.2005.036350. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Zou YS, Van Dyke DL, Ellison JW. Microarray comparative genomic hybridization and FISH studies of an unbalanced cryptic telomeric 2p deletion/16q duplication in a patient with mental retardation and behavioral problems. Am J Med Genet A. 2007;143:746–751. doi: 10.1002/ajmg.a.31645. [DOI] [PubMed] [Google Scholar]
- Bakker B, Bikker H, Vulsma T, De Randamie JSE, Wiedijk BM, De Vijlder JJM. Two decades of screening for congenital hypothyroidism in the Netherlands: TPO gene mutations in total iodide organification defects (an update) J Clin Endocr Metab. 2000;85:3708–3712. doi: 10.1210/jcem.85.10.6878. [DOI] [PubMed] [Google Scholar]
- Kim JG, Hudson LD. Novel member of the zinc finger superfamily: a C(2)-HC finger that recognizes a glia-specific gene. Molec Cell Biol. 1992;12:5632–5639. doi: 10.1128/mcb.12.12.5632. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Azuma T, Ao S, Saito Y, et al. Human SOX11, an upregulated gene during the neural differentiation, has a long 30 untranslated region. DNA Res. 1999;6:357–360. doi: 10.1093/dnares/6.5.357. [DOI] [PubMed] [Google Scholar]
- Sock E, Rettig SD, Enderich J, Bosl MR, Tamm ER, Wegner M. Gene targeting reveals a widespread role for the highmobility- group transcription factor Sox11 in tissue remodeling. Mol Cell Biol. 2004;24:6635–6644. doi: 10.1128/MCB.24.15.6635-6644.2004. [DOI] [PMC free article] [PubMed] [Google Scholar]