Abstract
In a 2 and a half-year-old girl with onset of puberty before the age of 5 months, short stature, hand anomalies and severe mental retardation, an 8.9 Mb interstitial 19p13 duplication containing 215 predicted genes was detected. It was initially assumed that the duplication involved the kisspeptin receptor gene, GPR54, known to stimulate induction of puberty, but more refined duplication mapping excluded this possibility. In an attempt to further understand the genotype–phenotype correlation, global gene expression was measured in skin fibroblasts. The overall expression pattern was quite similar to controls, and only about 25% of the duplicated genes had an expression level that was increased by more than 1.3-fold, with no obvious changes that could explain the precocious puberty. The proband's mother carried a balanced between-arm insertion of the duplicated segment that resembled a pericentric inversion. The same insertion was found in several other family members, including one who had lost a daughter with severe mental retardation and menarche at the age of 10 years. Another close relative was severely mentally retarded, but neither dysmorphic nor microcephalic. His phenotype was initially ascribed to a presumed cryptic chromosome 19 imbalance caused by the 19p-into19q insertion, but subsequent array-CGH detected a 3.9-Mb deletion of 2q23.3q24.1. This novel microdeletion involves seven genes, of which FMNL2, a suggested regulator of Rho-GTPases, and NR4A2, an essential gene for differentiation of dopaminergic neurons, may be critical genes for the proposed 2q23q24 microdeletion syndrome.
Keywords: intrachromosomal insertion, 19p13 duplication, 2q23.3 deletion, 2q24.1 deletion, precocious puberty, NR4A2, FMNL2
Introduction
Intrachromosomal insertions are difficult to detect cytogenetically and are often mistaken for inversions or duplications.1, 2, 3, 4 Only around 30 families with such insertions have so far been described.2 For carriers of between-arm (pericentric) insertions, the risk of having an abnormal child with chromosomal imbalances may be high with estimates that vary from 15 to 50%.2, 4, 5 Here, we describe diagnostic problems and unexpected findings when studying a three generation family with a between-arm insertion of a subtelomeric segment of 19p13-into-19q; ins(19)(q13.3p13.2p13.3).
Patients and methods
Index patient and family history
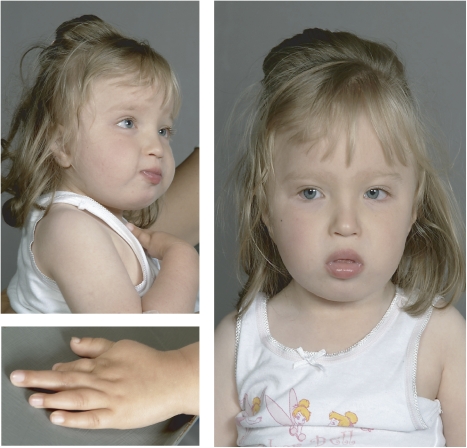
The index patient is a young girl with growth retardation of prenatal onset who was delivered by emergency caesarian section at weeks 35 of gestation for foetal distress. Her birth weight was 1790 g (3rd centile), length 42.5 cm (3rd centile), and head circumference was 30.5 cm (10th centile). She has dysmorphic facial features including a small mouth and short philtrum. Her left hand is smaller than her right, and the second left finger is short and malformed (Figure 1). She has small feet and long toes. Severe eating problems necessitated gastrostomy tube insertion, and funduplication was performed for gastroesophageal reflux. Bilateral congenital hip dysplasia was treated surgically. A small muscular VSD closed spontaneously. Within the first 5 months of life she developed signs of puberty including pubic hair, breast enlargement and high serum oestrogen levels. At the age of 2 and a half years, she had severe psychomotor retardation with no verbal language, and she could not walk independently.
Figure 1.
The proband at the age of 2 and a half years. There is mild facial dysmorphism as described in the manuscript text and finger malformations.
The index patient's mother has had four additional recognised pregnancies. Two resulted in healthy girls, one ended in early spontaneous abortion, and one was complicated by severe preeclampsia culminating in intrauterine death of a male child who weighed 750 g at ∼32 weeks of gestation and had no obvious malformations.
The mother also has a 54-year-old brother with severe mental retardation who is not dysmorphic and who has no known organ malformations. During the last 5 years, increasing gait unsteadiness and shaking on voluntary use of the right arm has greatly interfered with activities of daily living. The reason for his regression in motor skills and coordination is unknown. Upon examination, neither tremor at rest, rigidity and difficulty initiating voluntary movements nor drooling were noted. He previously had an expressive vocabulary of a few single words and short phrases, but during the last 5 years he had not spoken. However, his non-verbal communication skills had improved during the same period, and he seemed unchanged cognitively. He is 155 cm tall with an occipitofrontal head circumference of 56.5 cm (∼50 centile). He has always avoided prolonged eye contact. A tendency to insert objects into his ears, and especially his nostrils, was quite problematic in childhood and in early adulthood. He walked at age 18 months. Epilepsy was also diagnosed at this age. He has had general tonic–clonic, myoclonic–atonic and complex partial seizures. Seizure control was intermittently difficult to achieve in childhood. He now uses lamotrigine 50 mg twice daily, valproic acid 1500 mg twice daily and perphenazine 2 mg twice daily. His last generalised tonic–clonic seizure was 2 years ago, and since then he has only had complex partial seizures lasting ∼10 s. Several cerebral CTs, including one with contrast, were reported as normal when he was investigated in his 30s.
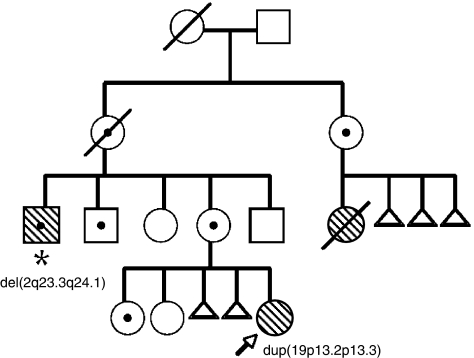
The proband's mother has a maternal aunt who had three spontaneous abortions. She had a cognitively impaired microcephalic and thin daughter who died at the age of 20 years (Figure 2). She did not learn to walk independently. She lacked speech, had bilateral congenital hip dysplasia, scoliosis and menarche at the age of 10 years.
Figure 2.
Family pedigree: The proband is marked with an arrow, and the proband's maternal uncle is marked with an asterix. Mentally retarded individuals have hatched symbols. Balanced between-arm insertion carriers (ins(19)(q13.3p13.2p13.3)) are indicated by a black dot inside their pedigree symbol, and the imbalances detected in the proband and her maternal uncle are written below their pedigree symbols.
Chromosome investigations
Standard G-banded chromosome analysis was performed on good quality metaphases from blood T-lymphocyte or fibroblast cultures at a band level of 400–550. Fluorescence in situ hybridisation (FISH) analysis using commercial TelVysion 19p and 19q subtelomeric probes (Abbott Molecular Inc., Des Plaines, IL, USA) or the TCF3 FISH Probe (DakoCytomation, Glostrup, Denmark) was performed according to the instructions from the suppliers. FISH analysis using in-house generated bacterial artificial chromosome (BAC) probes was performed as described previously.6 BAC clones were from human genomic libraries prepared by the California Institute of Technology Cancer Institute (CTD) or the Roswell Park Cancer Institute (RP11), all delivered by Research Genetics/Invitrogen (Invitrogen Inc., Carlsbad, CA, USA). The genomic location of the BAC clones used is based on information found in the Ensembl database (release 49, NCBI build 36.2).
High-resolution chromosome-based comparative genomic hybridisation (HR-CGH) was performed as described previously.7 Microarray-based CGH (array-CGH) was performed using three different platforms: The 1 Mb CytoChip™ BAC-array (BlueGnome, Cambridge, UK), the 244 K human genome CGH microarray (Agilent Technologies Inc., Santa Clara, CA, USA) and the 250 K Nsp1 GeneChip® human mapping SNP array (Affymetrix Inc., Santa Clara, CA, USA). The following array-CGH analyses were carried out according to the suppliers' instructions: the CytoChip Reference Manual Version 0.10 (BlueGnome), the Oligonucleotide Array-based CGH for Genomic DNA Analysis Protocol v5.0. (Agilent Technologies) and the GeneChip Human Mapping 500 K Assay Manual (Affymetix). The Affymetrix SNP arrays were scanned in an Affymetrix GeneChip scanner 3000, whereas BlueGnome or Agilent arrays were scanned in an Agilent G2565B scanner. Array-CGH by the CytoChip was analysed by the BlueFuse for Microarrays Software 3.4 (BlueGnome) with thresholds for normal DNA copy numbers set at a log2 scale of 0.29 and −0.29. Mapping of imbalances was based on BAC information found in the Ensembl database (release 49, NCBI build 36.2). Array-CGH performed on the 244 K Agilent oligoarray was analysed by the Agilent Feature Extraction Software (v9.5) and the Agilent CGH Analytics Software 3.5. Aberrations of statistical significance were determined by the ADM-2 algorithm, and the boundaries of the imbalances within these regions were manually determined from the ratio signal intensities of the individual oligos. Mapping of imbalances was based on the genomic location of the oligos specified in the NCBI build 36.1 (UCSC hg18, March 2006). Copy number studies carried out on the 250 K SNP array were analysed by the Genotyping consol software v2.1 (Affymetrix). Aberrations of statistical significance were determined by the CN4 algorithm, and the boundaries of the imbalances within these regions were manually determined from the ratio signal intensities of the individual SNPs. Mapping of imbalances was based on the genomic location of the SNPs as specified in NCBI build 36.1.
Gene expression profiling
The gene expression profile of a skin fibroblast culture prepared from the index patient with severe mental retardation and extreme pubertas praecox was determined on the Applied Biosystems Human Genome Survey Microarray v. 2.0 (Applied Biosystems, Foster City, CA, USA) and compared to skin fibroblast expression profiles from three age-matched female subjects without mental retardation. Total RNA was extracted from ∼1 × 106 cells using the RNeasy mini kit (Qiagen Ltd, Sussex, UK). The quality and integrity of the RNA was evaluated on the Agilent 2100 Bioanalyzer (Agilent Technologies) and the concentration was measured using a NanoDrop® ND-1000 Spectrophotometer (NanoDrop, Wilmington, DE). cRNA labelling, hybridisation and detection were carried out as described previously.8 The array contains 31 878 probes covering a total of 29 098 predicted human genes. Data from the four gene expression experiments were interarray normalised by quantile normalisation across the microarrays to achieve the same distribution of signal intensities for each array using the J-Express Pro v.2.7 software (http://www.molmine.com). Quality filtering of unreliable spots showing a low signal-to-noise ratio (S/N<3) was performed before further data analysis. Genes located in the duplicated area were compiled into an expression profile data matrix and annotated using Applied Biosystem's Microarray Annotation files (http://www.pantherdb.org). Further data analysis was performed as follows: for each probe, the absolute mean and the geometric mean of the three controls were calculated. Only probes having a difference of <10% between absolute and geometric means of the controls were accepted for further analysis. For each probe, the average of the absolute and geometric means (A/G means) of the controls were calculated, and this was compared to the expression value found in the patient. A gene was considered upregulated when the difference between the patient value and the A/G means was ≥33%, and downregulated when ≥−33%.
Results and discussion
The index patient's combination of short stature, dysmorphic facies, extreme pubertas praecox and limb malformations was not compatible with any known syndrome. The patient's family history with two close relatives with severe mental retardation separated from the index patient by healthy individuals was highly suggestive of a familial chromosomal rearrangement (Figure 2). However, only normal findings were reported after standard G-banded chromosome analysis of blood lymphocytes and skin fibroblast cultures (data not shown). HR-CGH analysis was also normal, whereas a 1 Mb resolution BAC-based array-CGH detected a terminal duplication of 19p (Figure 3a). When added to the family history, this finding suggested the presence of a cryptic translocation. However, the 19p termini appeared normal on subtelomere 19p and 19q FISH investigations (Figure 4b), ruling out the presence of an unbalanced translocation. Subsequent G-banding chromosome investigations of both parents revealed what appeared to be a pericentric inversion on one of the mother's chromosome 19 homologues (Figure 4a). Usually, familial pericentric inversions are low-risk rearrangements, but an important exception is large pericentric inversions involving the major part of both chromosome arms.2 To search for the p-terminal breakpoint of the presumed pericentric inversion, we performed FISH analysis with a TCF3 (E2A) split probe that hybridises centromeric (probe labelled red) and telomeric (probe labelled green) to the TCF3 gene (Figure 4d). A split signal was seen (Figure 4c), apparently indicating that the p-terminal breakpoint was in the TCF3 gene. However, the signal from the centromeric TCF3 probe was dislocated more distal on 19q than expected from a pericentric inversion (Figure 4c and d). Furthermore, the chromosomal imbalances caused by large pericentric inversions are usually a combination of a deletion of one chromosomal terminus and a duplication of the other,2 which was not the case here. Instead, the findings were consistent with the presence of a balanced between-arm insertion in the mother, described by the karyotype 46,XX,ins(19)(q13.3p13.2p13.3). On the basis of the location of the centromeric TCF3 probe signal, the insertion was most likely to be a direct insertion (Figure 4d). A meiotic cross-over on proximal 19p would produce a normal 19p-arm, and in the absence of a compensatory cross-over on proximal 19q, the observed chromosomal imbalance would result (Figure 4a and d).
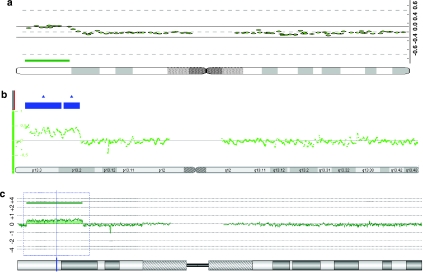
Figure 3.
Array-CGH and SNP-chip-based copy number analyses of the proband. A subtelomeric 19p duplication was detected by 1 Mb CytoChip BAC array-CGH analysis, as indicated by the green line (a). The 19p duplication was subsequently fine mapped by Affymetrix 250 K SNP-chip copy number analysis (marked by blue boxes; b), and Agilent 244 K oligo array-CGH analysis (marked by a green line; c), both placing the 19p-duplication distal to the GPR54 gene. In all, 850-band chromosome ideograms are shown below the array profiles.
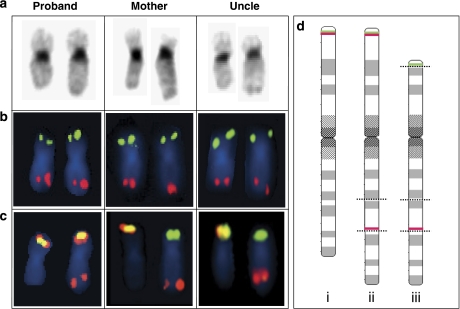
Figure 4.
G-banding ( a) and FISH analyses (b and c) of the proband, her mother and the maternal uncle. In all panels, the normal chromosome 19 is on the left. The proband has a 19q arm that is too long, reflecting her inheritance of a recombinant chromosome 19 from her mother with a p13.2p13.3 insertion into 19q13 combined with a normal 19p arm. The mother and uncle both have a balanced pericentric insertion. In panel b, the subtelomere chromosome 19 FISH results are shown, giving normal signals in all the examined individuals. In panel c, the TCF3 FISH result is shown. The TCF3 FISH split signal probe normally produces a yellow fusion signal at 19p. In the proband, this signal is found in addition to a red signal on 19q, representing the centromeric part of the TCF3 probe. In the mother and uncle, the presence of TCF3 at the distal duplication breakpoint results in the absence of a normal yellow fusion signal. (d) The three different types of chromosome 19 found in this family are shown, illustrated by 850-band level ideograms: (i) a normal chromosome 19; (ii) chromosome 19 with an unbalanced pericentric insertion, as detected in the proband; (iii) chromosome 19 with a balanced 19p13.2p13.3-into-19q13.3 pericentric insertion, as detected in the mother and uncle. The positions of the breakpoints are marked with dotted lines. Also, the positions of the detected TCF3 FISH probe signals are indicated by green lines (the telomeric TCF3 probe) and red lines (the centromeric TCF3 probe).
The observed chromosomal aberration in this family was likely to be related to the proband's phenotypic features, including the extreme precocious puberty. Early puberty (<2.5th centile) was also seen in her mentally retarded first cousin once removed (Figure 2). The 1 Mb array-CGH result suggested that the kisspeptin receptor GPR54 was included in the duplication (Figure 3a). Haploinsufficiency of this gene may lead to hypogonadotropic hypogonadism, and activating mutations can cause precocious puberty.9, 10 However, refined mapping on Agilent 244 K oligoarrays and Affymetrix 250 K SNP arrays excluded duplication of GPR54, placing the gene 0.6 Mb telomeric to the distal breakpoint according the NCBI build 36.3 (Figure 3b and c). The duplication size was finally mapped to be ∼8.9 Mb (range: 8.87–8.94 Mb) with breakpoints 1.41–1.44 and 10.31–10.35 Mb from 19pter (Figure 3). To exclude involvement of GPR54 in a more complex chromosome 19 rearrangement, FISH with the BAC clones CTD-2563K04 covering the GRP54 gene and with the flanking clones CTD-3195E18 and RP11-75H06 was carried out with normal findings (data not shown).
Having excluded an obvious candidate gene for precocious puberty, we explored the possibility that a skin fibroblast gene expression analysis could provide insight into the cause of the hormonal disturbance. However, in our pilot study (a single experiment without real-time PCR validation of individual gene expression levels), the global gene expression pattern on Applied Biosystems expression array appeared to be quite similar in the patient fibroblasts and in fibroblasts from three age-matched control female subjects. A more focused examination of the 215 genes located in the duplicated segment (covered by 229 probes) was then performed. Only 104 of these genes were rated as expressed above background level, of which nearly 50% and 30% had mRNA levels that were more than 1.3- and 1.5-fold increased, respectively, compared with controls (Supplementary Table 1). This is 3–4 times more than would be expected from random ‘overexpression'. Our findings are in line with what others have found when studying trisomy 21 where only 22% of the involved genes had an ∼1.5-fold increase in expression level.11 Among the overexpressed genes, no genes that might explain the precocious puberty were found.
It was initially assumed that the proband's severely retarded maternal uncle also had a chromosomal imbalance related to the familial between-arm insertion. Although he appeared to have inherited the same derivative chromosome 19 as the proband's healthy mother (Figure 4a–c), his G-banded karyotype was otherwise normal. To search for subtle chromosome 19 imbalances that might be related to the 19p-into-19q insertion, a 1 Mb BAC array-CGH was performed. No chromosome 19 imbalances were found, but unexpectedly, a deletion in the middle of the long arm of chromosome 2 was detected (data not shown) and later fine mapped by 250 K SNP-array analysis (Supplementary Figure 5a). No imbalances were detected on chromosome 19 by the 250 K SNP analysis (Supplementary Figure 5b). We have no reason to believe that the familiar between-arm insertion on chromosome 19 predisposes for deletions or duplications on other chromosomes, leaving the chromosome 2 deletion as an apparently unrelated random event. The deletion affected subdomains 2q23.3q24.1 with breakpoints between 153.10–153.15 and 156.98–157.02 Mb from the 2pter, that is, a deletion of ∼3.9 Mb (range: 3.83–3.92 Mb).
According to the NCBI Human Genome Browser Resources database (NCBI build 36.3) there are seven genes in the deleted interval: FMNL2, PRPF40A, ARL6IP6, RPRM, GALNT13, KCNJ3 and NR4A2 (Supplementary Table 2). FMNL2 is located in the 2q23.3 breakpoint with the majority of the gene deleted. Of the seven genes, only deletion of the GALNT13 gene is reported as a copy number variant in the Toronto Database of Genomic Variants (http://projects.tcag.ca/variation/). The most obvious candidate genes for causing mental retardation when haploinsufficient are the formin-like 2 gene FMNL2 and the nuclear receptor subfamily 4 group A member 2 gene NR4A2, a member of the steroid–thyroid hormone and retinoid receptor superfamily. FMNL2 is an alternatively spliced gene with 26 exons and high expression predominantly in the brain (see GNF expression atlas 2 data on the UCSC genome browser; http://genome.ucsc.edu/). The gene's function is unknown, but it has similarity to genes involved in Rho-dependent signal transduction.12 Rho-dependent GTPases may regulate the actin and microtubule cytoskeletons, and thus, the morphology of dendritic spines; their possible involvement in mental retardation is well known.13, 14 NR4A2, also called Nurr1, is a transcription factor essential for the differentiation of dopaminergic neurons in substantia nigra. Mutations in the leader sequence (5′UTR) of NR4A2 mRNA that cause reduced gene expression have been linked to familial parkinsonism,15 and mice heterozygous for NR4A2 null mutations have been suggested as animal models for schizophrenia.16 Although our patient has developed balance problems with shaking upon voluntary use of his right arm since 50 years of age, he has no signs of parkinsonism. We have no reason to believe that he has ever experienced psychotic episodes. In brain gene expression databases available to us, we found that NR4A2 is highly expressed in the parieto–occipital cortex of the adult rat brain with only negligible expression in the frontal cortex. High expression was also seen in mice hippocampal projection neurons (data not shown). There are earlier reports of patients with much larger deletions that include the same chromosomal subdomains as in our patient (see the Decipher database (https://decipher.sanger.ac.uk/), and Maas et al17 suggest the presence of a 2q23q24 microdeletion syndrome and provide an overview of nine patients with deletions that includes 2q23.3q24.1.17 The phenotypes as well as the deletion size are quite variable. However, microcephaly, epilepsy and mental retardation are recurrent features.
In conclusion, this family serves to re-emphasise that pericentric or between-arm insertions are high-risk chromosomal rearrangements that can easily be misinterpreted as pericentric inversions, and that duplications can be difficult to detect. We were not able to determine a cause for the extreme precocious puberty in one individual, and this patient underscores the need for careful mapping of chromosomal aberrations before making assumptions about genotype–phenotype correlations. Furthermore, the family reminds us that the aetiology of severe mental retardation is not necessarily the same in closely related individuals. The usefulness of array-CGH for detecting alternate explanations for mental retardation phenotypes in the same family is shown. Finally, a potential critical region for the proposed 2q23q24 microdeletion syndrome has been narrowed down, and we propose that haploinsufficiency for FMNL2 and/or NR4A2 may compromise normal brain development and function.
Acknowledgments
All the routine laboratory works were carried out at the cytogenetic diagnostic laboratory under the skilful direction of Kjetil Solland. We are most grateful to the family of the index patient for its collaboration. This study was supported by Helse Vest RHF. We acknowledge the infrastructure provided by the Norwegian Microarray Consortium (NMC), a national FUGE microarray technology platform, funded by the Research Council of Norway.
Footnotes
Supplementary Information accompanies the paper on European Journal of Human Genetics website (http://www.nature.com/ejhg)
Supplementary Material
References
- Ardalan A, Prieur M, Choiset A, Turleau C, Goutieres F, Girard-Orgeolet S. Intrachromosomal insertion mimicking a pericentric inversion: molecular cytogenetic characterization of a three break rearrangement of chromosome 20. Am J Med Genet A. 2005;138A:288–293. doi: 10.1002/ajmg.a.30966. [DOI] [PubMed] [Google Scholar]
- Gardner R, Sutherland G. Chromosome Abnormalities and Genetic Counseling. New York: Oxford University Press; 2004. [Google Scholar]
- Henry I, Hoovers J, Barichard F, et al. Pericentric intrachromosomal insertion responsible for recurrence of del(11)(p13p14) in a family. Genes Chromosomes Cancer. 1993;7:57–62. doi: 10.1002/gcc.2870070110. [DOI] [PubMed] [Google Scholar]
- Madan K, Menko FH. Intrachromosomal insertions: a case report and a review. Hum Genet. 1992;89:1–9. doi: 10.1007/BF00207032. [DOI] [PubMed] [Google Scholar]
- Friedrich U, Houman M, Sandgaard J, Rosgaard A, Sunde L. Microdissection of chromosome 2 – between-arm intrachromosomal insertion. Eur J Hum Genet. 2000;8:393–395. doi: 10.1038/sj.ejhg.5200421. [DOI] [PubMed] [Google Scholar]
- Lybaek H, Øyen N, Fauske L, Houge G. A 2.1 Mb deletion adjacent but disyal to a 14q21q23 paracentric inversion in a family with spherocytosis and severe learning difficulties. Clin Genet. 2008;74:553–559. doi: 10.1111/j.1399-0004.2008.01072.x. [DOI] [PubMed] [Google Scholar]
- Lybaek H, Meza-Zepeda LA, Kresse SH, Hoysaeter T, Steen VM, Houge G. Array-CGH fine mapping of minor and cryptic HR-CGH detected genomic imbalances in 80 out of 590 patients with abnormal development. Eur J Hum Genet. 2008;16:1318–1328. doi: 10.1038/ejhg.2008.78. [DOI] [PubMed] [Google Scholar]
- Stansberg C, Vik-Mo AO, Holdhus R, et al. Gene expression profiles in rat brain disclose CNS signature genes and regional patterns of functional specialisation. BMC Genomics. 2007;8:94. doi: 10.1186/1471-2164-8-94. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Semple RK, Achermann JC, Ellery J, et al. Two novel missense mutations in g protein-coupled receptor 54 in a patient with hypogonadotropic hypogonadism. J Clin Endocrinol Metab. 2005;90:1849–1855. doi: 10.1210/jc.2004-1418. [DOI] [PubMed] [Google Scholar]
- Teles MG, Bianco SD, Brito VN, et al. A GPR54-activating mutation in a patient with central precocious puberty. N Engl J Med. 2008;358:709–715. doi: 10.1056/NEJMoa073443. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Ait Yahya-Graison E, Aubert J, Dauphinot L, et al. Classification of human chromosome 21 gene-expression variations in Down syndrome: impact on disease phenotypes. Am J Hum Genet. 2007;81:475–491. doi: 10.1086/520000. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Katoh M, Katoh M. Identification and characterization of human FMNL1, FMNL2 and FMNL3 genes in silico. Int J Oncol. 2003;22:1161–1168. [PubMed] [Google Scholar]
- Nadif Kasri N, Van Aelst L. Rho-linked genes and neurological disorders. Pflugers Arch. 2008;455:787–797. doi: 10.1007/s00424-007-0385-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Newey SE, Velamoor V, Govek EE, Van Aelst L. Rho GTPases, dendritic structure, and mental retardation. J Neurobiol. 2005;64:58–74. doi: 10.1002/neu.20153. [DOI] [PubMed] [Google Scholar]
- Le WD, Xu P, Jankovic J, et al. Mutations in NR4A2 associated with familial Parkinson disease. Nat Genet. 2003;33:85–89. doi: 10.1038/ng1066. [DOI] [PubMed] [Google Scholar]
- Rojas P, Joodmardi E, Hong Y, Perlmann T, Ogren SO. Adult mice with reduced Nurr1 expression: an animal model for schizophrenia. Mol Psychiatry. 2007;12:756–766. doi: 10.1038/sj.mp.4001993. [DOI] [PubMed] [Google Scholar]
- Maas SM, Hoovers JM, van Seggelen ME, Menzel DM, Hennekam RC. Interstitial deletion of the long arm of chromosome 2: a clinically recognizable microdeletion syndrome. Clin Dysmorphol. 2000;9:47–53. doi: 10.1097/00019605-200009010-00010. [DOI] [PubMed] [Google Scholar]
Associated Data
This section collects any data citations, data availability statements, or supplementary materials included in this article.