Abstract
Adult bone marrow stem cells seem to differentiate into muscle, skin, liver, lung, and neuronal cells in rodents and have been shown to regenerate myocardium, hepatocytes, and skin and gastrointestinal epithelium in humans. Because we have demonstrated previously that transplanted bone marrow cells can enter the brain of mice and differentiate into neurons there, we decided to examine postmortem brain samples from females who had received bone marrow transplants from male donors. The underlying diseases of the patients were lymphocytic leukemia and genetic deficiency of the immune system, and they survived between 1 and 9 months after transplant. We used a combination of immunocytochemistry (utilizing neuron-specific antibodies) and fluorescent in situ hybridization histochemistry to search for Y chromosome-positive cells. In all four patients studied we found cells containing Y chromosomes in several brain regions. Most of them were nonneuronal (endothelial cells and cells in the white matter), but neurons were certainly labeled, especially in the hippocampus and cerebral cortex. The youngest patient (2 years old), who also lived the longest time after transplantation, had the greatest number of donor-derived neurons (7 in 10,000). The distribution of the labeled cells was not homogeneous. There were clusters of Y-positive cells, suggesting that single progenitor cells underwent clonal expansion and differentiation. We conclude that adult human bone marrow cells can enter the brain and generate neurons just as rodent cells do. Perhaps this phenomenon could be exploited to prevent the development or progression of neurodegenerative diseases or to repair tissue damaged by infarction or trauma.
Neurogenesis used to be thought to be completed during embryonic life in rodents as well as humans. During the last decade, however, numerous studies have suggested that neurogenesis continues in adult animals and humans, at least to a certain extent in a few privileged areas of the brain (1–4). Most of these studies have focused on endogenous neural progenitor cells (neural stem cells) localized in the subventricular zone of the lateral ventricle and in the dentate gyrus in the hippocampus in rodents (4). In the monkeys these cells are present in the hippocampus and neocortex (5, 6). Likewise, Eriksson et al. (7) found that new neurons are generated continuously in the human dentate gyrus throughout life.
It is also conceivable that stem cells from other sources might enter the brain and form neurons there. Uchida et al. (8) isolated CNS stem cells from human fetal tissue and transplanted them into the brains of mice, where they subsequently proliferated and differentiated into neuronal cells. One source of such cells in the brain could be the bone marrow. Adult bone marrow stem cells seem able to differentiate into muscle, skin, liver, lung, and neural cells in rodents (9–18). Furthermore, transplanted bone marrow cells in humans have also been shown to form myocardial cells (19, 20), hepatocytes (21, 22), and epithelium of the skin and gastrointestinal tract (20). Because we have demonstrated previously that transplanted bone marrow cells migrate into the brains of mice and give rise to neurons there (15), we hypothesized that the same thing might occur in the human CNS after bone marrow transplantation. We tested this hypothesis by looking for Y chromosome-positive neuron-like cells in postmortem brain samples from females who had received bone marrow transplants from male donors.
Methods
Four female patients who had had bone marrow transplants from male donors were selected from the autopsy files of The Johns Hopkins Hospital. Patient 1 had Omenn's syndrome, was transplanted at 9 months of age, and died 10 months later. Patient 2 had Hodgkin's disease and was transplanted at 34 years of age. Patient 3 had acute lymphocytic leukemia and was transplanted at 10 years of age. Patient 4 had acute lymphocytic leukemia and was transplanted at 20 years of age. Patients 2–4 died within ≈2 months of receiving their transplants.
Formalin-fixed, paraffin-embedded sections (6 μm) from the following brain areas were examined in each case: neocortex, striatum including the lateral ventricular wall, hippocampus with adjacent mesial temporal lobe structures, and cerebellum. Sections from three nontransplanted female patients were used as negative controls for Y-chromosomal staining. Sections from four male patients were used as positive controls. One tissue sample each from a male and a control female brain were reembedded together into one paraffin block and serially sectioned at a thickness of 6 μm; sections from this block were used as controls in all experimental series.
After deparaffinization in Citrisolv (Fisher Scientific) the sections were rehydrated, and heat-induced antigen retrieval was performed in a histology microwave oven by using a citrate buffer (Citra-plus, Innogenex, San Ramon, CA) for 5 min at 600 W. Next, immunostaining was performed by using primary antibodies that were detected by the Sternberger peroxidase antiperoxidase (PAP) method (23) followed by either biotinylated tyramide (for Kv2.1) or FITC-tyramide plus [(for neuronal nuclear antigen (NeuN)] (Perkin–Elmer). The primary antibodies used were directed against two neuronal proteins: NeuN (24), a neuron-specific nuclear protein, and Kv2.1, a neuron-specific voltage-gated potassium channel antibody (25, 26). Kv2.1 was recognized as a neuron-specific potassium channel that was first described in the principal neurons of the hippocampus and cortex and later shown to be present in the vast majority of interneurons as well (26). The NeuN antibody was a mouse monoclonal (used at 1:1,000) (Chemicon), and the Kv2.1 antibody was a rabbit polyclonal (used at 1:500, Alomone, Jerusalem). After immunostaining, the in situ hybridization was carried out as described (15) by using a digoxigenin-labeled riboprobe complementary to the satellite region of the human Y chromosome. We prepared the template from which we made probe from human genomic DNA by using primers that amplified a 1.3-kb-long DNA of the human Y chromosome. The probe was visualized by using peroxidase-conjugated antidigoxigenin antibody (Roche, Indianapolis) followed by a tyramide-CY3 fluorochrome plus (Perkin–Elmer) amplification. At the end, all sections were stained with a 1% Sudan-black solution to mask lipofuscin-induced autofluorescence, which is a common problem when working with human brain tissue (27). All sections were stained with 4′,6-diamidino-2-phenylindole (DAPI, Sigma), a chromosomal stain to label nuclei, and mounted with 80% glycerol/20% Tris. A separate series of sections was hybridized by using a radiolabeled Y chromosome probe, which is easily visualized at low magnification. This enabled us to count Y-positive cells and examine their overall distribution. All sections were viewed with conventional Leitz and confocal Zeiss microscopes.
In addition to the nontransplanted male and female brains, to make sure of the specificity of the techniques we ran as controls immunostaining with amplification without primary and/or without the secondary antibodies.
Analysis of the distribution of Y chromosome-positive cells was done as follows. The Y chromosome probe was radiolabeled with [35S]UTP and hybridized to brain sections. After emulsion coating and autoradiographic development of the sections, the Y-positive nuclei were counted at low magnification (×10). The counts in individual 1.2-mm2 microsquares (visual field) of a grid that covered the entire section were determined. The mean and variance of the counts and the variance-to-mean ratio were calculated. In a random (Poisson) distribution, the variance-to-mean ratio is 1. Therefore, the nearness to 1 of this ratio is a measure of the randomness of the distribution (28).
Results
In the control male brain sections we detected the Y chromosome with both the fluorescent and autoradiographic techniques in >90% of the nuclei, whereas no labeling was observed in the brain sections of female patients who have not received transplants. In each of the transplanted patients examined we readily detected Y-positive cells by means of autoradiography. In all patients using conventional fluorescent microscopy we observed double-labeled cells that were positive for both the Y chromosome and one of the neuronal markers as shown in an example in Fig. 1. Most of the cells that were double-labeled with the Y chromosome and the neuronal markers were detected in the hippocampus and the neocortex of patients (Fig. 2). The antibody that binds to Kv2.1 gave a very convincing staining of neuronal somata and dendrites in all cortical areas and in the hippocampus (as described in the literature; see ref. 26), and also a few axons seemed to show a patchy immunostaining after signal amplification.
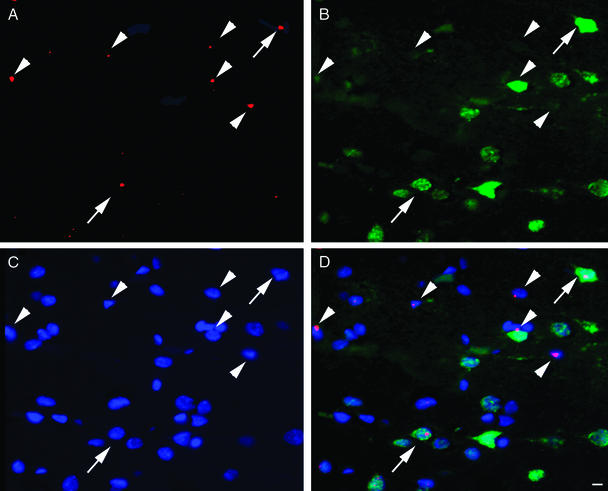
Figure 1.
(A) A 6-μm-thin section from somatosensory cortex of patient 2 demonstrates the presence of the Y chromosome depicted as red dots and viewed through a rhodamine filter. The same field as in A is shown when viewed through the FITC filter to demonstrate the immunostaining for the neuronal marker NeuN in green (B), and the UV filter shows all cell nuclei in blue after staining with 4′,6-diamidino-2-phenylindole, a chromosomal stain (C). (D) The overlay of the three filters, where arrows point to cells that carry all markers, indicating that they derived from the donor bone marrow (Y chromosome-positive) and bear the specific neuronal marker NeuN. Arrowheads point at nonneuronal donor-derived cells. (Scale bars, 10 μm.)
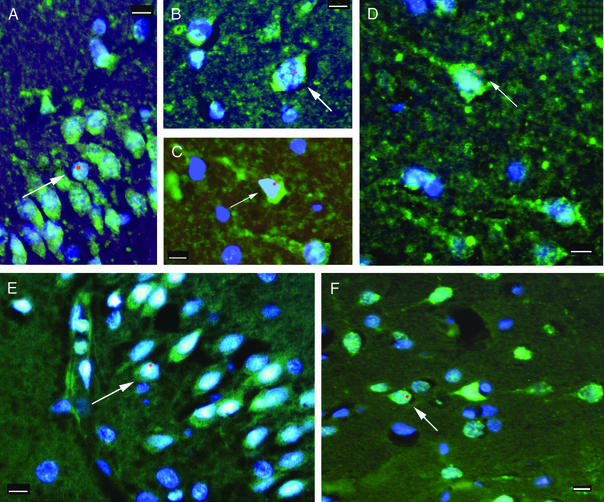
Figure 2.
Neuronal markers colocalized with the Y chromosome. Fluorescent microscopic images of neocortex from patients 2 (A–C) and 1 (E) and hippocampus from patients 1 (D) and 3 (F) are shown. The green color represents the immunostaining for neuronal markers Kv2.1 (A–D) and NeuN (E and F), and the Y chromosome is represented by the red fluorescent dots. All cell nuclei are stained with 4′,6-diamidino-2-phenylindole, a chromosomal marker that shows up as blue fluorescence. All images are overlays of the images seen through the three separate filters to show all colors. Arrows point to cells that are labeled with neuronal markers and are also Y chromosome-positive. In the Kv2.1 immunostaining the initial axons of some neurons can also be visualized. (Scale bars, 10 μm.)
Confocal z series confirmed the presence of Y chromosomes in the nuclei of the same cells that were immunopositive for the neuronal markers. Examples from two patients are shown in Fig. 3. In the patient with the greatest number of double-labeled cells (patient 1), the distribution of the Y-positive cells suggested that clonal expansion had occurred: We detected no labeled cells in many visual fields but typically saw clusters of positive cells when such cells were detected. This pattern was easier to recognize in the autoradiographic sections, which could be examined at low magnification. In several clusters, the Y-positive cells were both neuronal and nonneuronal. In the cortex, based on morphology and location, the Y-positive neurons were small pyramidal cells, whereas in the hippocampus the Y-positive cells seemed to be granule cells. We also saw Y-positive cells in the white matter, some of which looked like oligodendrocytes based on the shape, size, and arrangement of their nuclei. We saw many Y-positive cells inside vessels (in cross section) and also some Y-positive endothelial cells in the vascular wall.
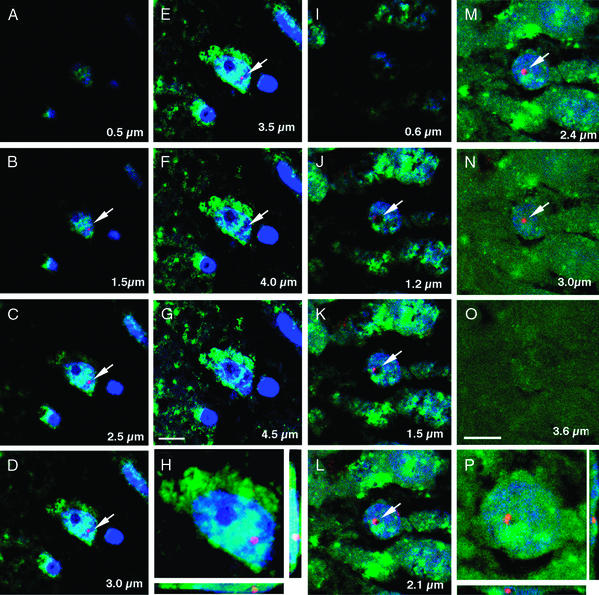
Figure 3.
Two confocal z series are shown. (A–D) Optical sections (1-μm-thin) of a neocortical neuron from patient 2. (E–F) Optical 1-μm-thin slices of a hippocampal granule cell from patient 1. Both cells are immunostained with the neuronal marker Kv2.1 (green); the Y chromosome is red (CY3-plus), and the nucleus is blue (4′,6-diamidino-2-phenylindole). The arrowheads point to the double-labeled cells. Note that the cell nucleus and the Y chromosome are consistently in the same plane.
We counted all the Y chromosome-positive cells in two entire sections of striatum/cortex and hippocampus/cortex blocks from two patients, analyzing 281 and 247 microscopic fields from patients 1 and 3, respectively (Table 1). In patient 1 we found 519 Y-positive cell nuclei among 182,000 nuclei (stained with ethidium bromide), and in patient 3 we found 1,842 Y-positive nuclei among 196,700. In the same sections we found 19 and 5 Y chromosome-containing nuclei, respectively, that also colocalized with neuronal antigens. Based on these observations and conservatively assuming that 25% of all nuclei in the human brain are neurons [this number in the cortex of primates varies between 27% and 60% (29, 30)], one in every 2,000–4,000 neurons might derive from the bone marrow.
Table 1.
Number of Y chromosome-positive (Y+) cells in two patient samples
| Patient | No. of visual fields (1 field = 0.94 mm2) | No. of Y+ nuclei | No. of all nuclei | No. of Y+ nuclei in cells with neuronal markers |
|---|---|---|---|---|
| 1 | 281 | 519 | 182,000 | 19 |
| 3 | 247 | 1,842 | 196,700 | 5 |
When we evaluated the distribution of Y chromosome-positive cells in patients 1 and 3 we found that it was not random. The variance-to-mean ratios of all counts in microsquares overlaying the sections studied were calculated to be 10.2 (patient 1) and 4.7 (patient 3).
Discussion
Several studies have shown that there is neurogenesis in the adult brain, even though it may be limited. Cells with neuron-specific markers can be formed by neural stem cells in vivo (1, 3, 31) as well as by bone marrow cells in vitro (32, 33) and in vivo (9, 15). Recently, Priller et al. (34) published a picture of a well differentiated, enhanced GFP-positive Purkinje cell in the cerebellum of a mouse transplanted with enhanced GFP-tagged bone marrow from a second animal. Along with earlier studies, this finding was a significant step toward showing that fully functional neurons can be generated from bone marrow cells. Based on our present study, we have no way of knowing which population of bone marrow cells entered the CNS and differentiated into cells expressing neuronal morphology and neuronal markers. Mesenchymal stem cells have been shown to differentiate into many different cell lineages (35). All of our patients received total bone marrow transplants containing both hematopoietic and mesenchymal stem cells.
Recently, two studies (36, 37) suggested the possibility that Y-positive cells of bone marrow origin might simply fuse with embryonic stem cells instead of transdifferentiating into cells characteristic of various tissues. Although this idea is interesting, no data support its relevance in vivo (see ref. 38). In fact, the fusion process is very inefficient in vitro: 1 in a million, which is a figure much lower than the number of double-labeled cells that we see in the brain. Recently, Castro et al. (39) failed to detect blue cells in the brains of lethally irradiated mice that received bone marrow from Rosa26 mice with a β-galactosidase-containing transgene. The use of protein products of transgenes as markers to follow graft fate is plagued with problems, but to date these may not have been emphasized enough. It is practically impossible to achieve ubiquitous transgene expression. Transgenes, including those driven by the Rosa26 promoter, suffer from instability in several tissues. Furthermore, to detect low levels of lacZ is difficult, and the detection is sensitive to fixation and staining conditions (40). Because one would have expected to see at least labeled microglia in the brain, one must assume that the results reported were due to technical problems.
In the present cases it is difficult to make general statements about the extent of CNS colonization or to conclude whether cells enter the brain continuously or just at the time of the transplant. All of our patients received irradiation to eliminate the underlying disease. We do not know how much effect the irradiation might have on the migration of circulating cells into the brain by either injuring the blood–brain barrier and/or releasing possible recruiting factors due to the injury caused by the radiation. It is known, however, that circulating blood cells are able to enter the brain parenchyma freely in healthy subjects (41). We studied a limited number of patients with different diseases, ages, survival times after transplant, and treatments of postmortem tissue. Because of the technical difficulties of double-labeling cells in paraffin-embedded postmortem tissue and the fact that we had to reduce the sensitivity of the method to eliminate background, we believe that our numbers are in fact lower that the real number of differentiating cells in vivo. Although we found donor (i.e., Y chromosome-positive) cells exhibiting two specific neuronal markers (NeuN and Kv2.1) in the hippocampus and neocortex in all three of the patients with technically satisfactory samples, the numbers of double-labeled cells were much lower than those reported in rodents (9, 15). We found 2–5 Y-positive neurons per 10,000 human neurons vs. 50 per 10,000 rodent neurons. Whether this is a species difference is unclear. The sections with the highest number of newly formed neurons (7 per 10,000 neurons) were from patient 1, the youngest studied, who had her transplant in infancy and also had the longest posttransplant survival time. All the other patients lived only weeks after the transplant, and there was no significant difference in the number of donor-positive cells in their brains.
It should also be noted that among the Y-positive cells, neurons were consistently in the minority. Based on their nuclear morphology, size, and location, we feel that the nonneuronal cells bearing the Y chromosome were a mixture of oligodendrocytes, astrocytes, and possibly microglia. We also detected endothelial, meningeal, and ependymal cells that were Y-positive. Finally, many circulating white blood cells within the vascular lumen were also Y-positive, as one would expect.
Our analysis showed that the distribution of Y-positive cells within the brain is not random. Instead, the cells appeared in clusters, a spatial distribution that suggests nondifferentiated cells may enter an area and then further propagate there. In these areas we see different kinds of Y-positive cells (neuronal and nonneuronal). One possibility is that one undifferentiated cell migrates into an “area of need” and then goes through asymmetrical divisions to produce different lineages of cells. Another possibility is that many progenitor cells are “called in,” and they differentiate into different lineages of cells. Whichever is the case, we speculate that areas in need of new cells (because of physiological turnover or pathological loss of cells) may be able to signal to potential stem cells to coax them into the region, and then clonal expansion occurs to help restore the number of cells to normal. Therefore, it will be very important to try to find the factors responsible for inducing stem cells to migrate into lesioned or sick areas of the brain. Discovering these factors could aid the attempt to use bone marrow cells to repair the brain.
Acknowledgments
We thank Ted Usdin for help in preparing the human Y chromosome probe; Carolyn Smith for help in confocal microscopy; and Michael Brownstein and Harold Gainer for helpful suggestions and for editing the manuscript.
Abbreviations
- NeuN
neuronal nuclear antigen
- Kv2.1
neuron-specific potassium channel
Footnotes
This paper was submitted directly (Track II) to the PNAS office.
References
- 1.Gage F H. Science. 2000;287:1433–1438. doi: 10.1126/science.287.5457.1433. [DOI] [PubMed] [Google Scholar]
- 2.Gage F H, Kempermann G, Palmer T D, Peterson D A, Ray J. J Neurobiol. 1998;36:249–266. doi: 10.1002/(sici)1097-4695(199808)36:2<249::aid-neu11>3.0.co;2-9. [DOI] [PubMed] [Google Scholar]
- 3.McKay R. Science. 1997;276:66–71. doi: 10.1126/science.276.5309.66. [DOI] [PubMed] [Google Scholar]
- 4.Weissman I L, Anderson D J, Gage F. Annu Rev Cell Dev Biol. 2001;17:387–403. doi: 10.1146/annurev.cellbio.17.1.387. [DOI] [PubMed] [Google Scholar]
- 5.Gould E, Vail N, Wagers M, Gross C G. Proc Natl Acad Sci USA. 2001;98:10910–10917. doi: 10.1073/pnas.181354698. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 6.Gould E, Gross C G. J Neurosci. 2002;22:619–623. doi: 10.1523/JNEUROSCI.22-03-00619.2002. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 7.Eriksson P S, Perfilieva E, Bjork-Eriksson T, Alborn A M, Nordborg C, Peterson D A, Gage F H. Nat Med. 1998;4:1313–1317. doi: 10.1038/3305. [DOI] [PubMed] [Google Scholar]
- 8.Uchida N, Buck D W, He D, Reitsma M J, Masek M, Phan T V, Tsukamoto A S, Gage F H, Weissman I L. Proc Natl Acad Sci USA. 2000;97:14720–14725. doi: 10.1073/pnas.97.26.14720. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 9.Brazelton T R, Rossi F M, Keshet G I, Blau H M. Science. 2000;290:1775–1779. doi: 10.1126/science.290.5497.1775. [DOI] [PubMed] [Google Scholar]
- 10.Bittner R E, Schofer C, Weipoltshammer K, Ivanova S, Streubel B, Hauser E, Freilinger M, Hoger H, Elbe-Burger A, Wachtler F. Anat Embryol. 1999;199:391–396. doi: 10.1007/s004290050237. [DOI] [PubMed] [Google Scholar]
- 11.Eglitis M A, Mezey E. Proc Natl Acad Sci USA. 1997;94:4080–4085. doi: 10.1073/pnas.94.8.4080. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Ferrari G, Cusella-De Angelis G, Coletta M, Paolucci E, Stornaiuolo A, Cossu G, Mavilio F. Science. 1998;279:1528–1530. doi: 10.1126/science.279.5356.1528. [DOI] [PubMed] [Google Scholar]
- 13.Gussoni E, Soneoka Y, Strickland C D, Buzney E A, Khan M K, Flint A F, Kunkel L M, Mulligan R C. Nature. 1999;401:390–394. doi: 10.1038/43919. [DOI] [PubMed] [Google Scholar]
- 14.Mahmood A, Lu D, Wang L, Li Y, Lu M, Chopp M. Neurosurgery. 2001;49:1196–1203. [PubMed] [Google Scholar]
- 15.Mezey E, Chandross K J, Harta G, Maki R A, McKercher S R. Science. 2000;290:1779–1782. doi: 10.1126/science.290.5497.1779. [DOI] [PubMed] [Google Scholar]
- 16.Orlic D, Kajstura J, Chimenti S, Jakoniuk I, Anderson S M, Li B S, Pickel J, McKay R, Nadal-Ginard B, Bodine D M, et al. Nature. 2001;410:701–705. doi: 10.1038/35070587. [DOI] [PubMed] [Google Scholar]
- 17.Theise N D, Badve S, Saxena R, Henegariu O, Sell S, Crawford J M, Krause D S. Hepatology. 2000;31:235–240. doi: 10.1002/hep.510310135. [DOI] [PubMed] [Google Scholar]
- 18.Theise N D, Nimmakayalu M, Gardner R, Illei P B, Morgan G, Teperman L, Henegariu O, Krause D S. Hepatology. 2000;32:11–16. doi: 10.1053/jhep.2000.9124. [DOI] [PubMed] [Google Scholar]
- 19.Quaini F, Urbanek K, Beltrami A P, Finato N, Beltrami C A, Nadal-Ginard B, Kajstura J, Leri A, Anversa P. N Engl J Med. 2002;346:5–15. doi: 10.1056/NEJMoa012081. [DOI] [PubMed] [Google Scholar]
- 20.Korbling M, Katz R L, Khanna A, Ruifrok A C, Rondon G, Albitar M, Champlin R E, Estrov Z. N Engl J Med. 2002;346:738–746. doi: 10.1056/NEJMoa3461002. [DOI] [PubMed] [Google Scholar]
- 21.Lagasse E, Connors H, Al-Dhalimy M, Reitsma M, Dohse M, Osborne L, Wang X, Finegold M, Weissman I L, Grompe M. Nat Med. 2000;6:1229–1234. doi: 10.1038/81326. [DOI] [PubMed] [Google Scholar]
- 22.Alison M R, Poulsom R, Jeffery R, Dhillon A P, Quaglia A, Jacob J, Novelli M, Prentice G, Williamson J, Wright N A. Nature. 2000;406:257. doi: 10.1038/35018642. [DOI] [PubMed] [Google Scholar]
- 23.Sternberger L A, Petrali J P. J Histochem Cytochem. 1977;25:1036–1042. doi: 10.1177/25.9.333018. [DOI] [PubMed] [Google Scholar]
- 24.Mullen R J, Buck C R, Smith A M. Development (Cambridge, UK) 1992;116:201–211. doi: 10.1242/dev.116.1.201. [DOI] [PubMed] [Google Scholar]
- 25.Betancourt L, Colom L V. J Neurosci Res. 2000;61:646–651. doi: 10.1002/1097-4547(20000915)61:6<646::AID-JNR8>3.0.CO;2-B. [DOI] [PubMed] [Google Scholar]
- 26.Du J, Tao-Cheng J H, Zerfas P, McBain C J. Neuroscience. 1998;84:37–48. doi: 10.1016/s0306-4522(97)00519-8. [DOI] [PubMed] [Google Scholar]
- 27.Schnell S A, Staines W A, Wessendorf M W. J Histochem Cytochem. 1999;47:719–730. doi: 10.1177/002215549904700601. [DOI] [PubMed] [Google Scholar]
- 28.Pielou E C. Mathematical Ecology. New York: Wiley Interscience; 1976. [Google Scholar]
- 29.Leuba G, Garey L J. Exp Brain Res. 1989;77:31–38. doi: 10.1007/BF00250564. [DOI] [PubMed] [Google Scholar]
- 30.O'Kusky J, Colonnier M. J Comp Neurol. 1982;210:278–290. doi: 10.1002/cne.902100307. [DOI] [PubMed] [Google Scholar]
- 31.Gage F H. Curr Opin Neurobiol. 1998;8:671–676. doi: 10.1016/s0959-4388(98)80098-6. [DOI] [PubMed] [Google Scholar]
- 32.Woodbury D, Schwarz E J, Prockop D J, Black I B. J Neurosci Res. 2000;61:364–370. doi: 10.1002/1097-4547(20000815)61:4<364::AID-JNR2>3.0.CO;2-C. [DOI] [PubMed] [Google Scholar]
- 33.Sanchez-Ramos J, Song S, Cardozo-Pelaez F, Hazzi C, Stedeford T, Willing A, Freeman T B, Saporta S, Janssen W, Patel N, et al. Exp Neurol. 2000;164:247–256. doi: 10.1006/exnr.2000.7389. [DOI] [PubMed] [Google Scholar]
- 34.Priller J, Persons D A, Klett F F, Kempermann G, Kreutzberg G W, Dirnagl U. J Cell Biol. 2001;155:733–738. doi: 10.1083/jcb.200105103. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 35.Jiang Y, Jahagirdar B N, Reinhardt R L, Schwartz R E, Keene C D, Ortiz-Gonzalez X R, Reyes M, Lenvik T, Lund T, Blackstad M, et al. Nature. 2002;418:41–49. doi: 10.1038/nature00870. [DOI] [PubMed] [Google Scholar]
- 36.Ying Q L, Nichols J, Evans E P, Smith A G. Nature. 2002;416:545–548. doi: 10.1038/nature729. [DOI] [PubMed] [Google Scholar]
- 37.Terada N, Hamazaki T, Oka M, Hoki M, Mastalerz D M, Nakano Y, Meyer E M, Morel L, Petersen B E, Scott E W. Nature. 2002;416:542–545. doi: 10.1038/nature730. [DOI] [PubMed] [Google Scholar]
- 38.McKay R. Nat Biotechnol. 2002;20:426–427. doi: 10.1038/nbt0502-426. [DOI] [PubMed] [Google Scholar]
- 39.Castro R F, Jackson K A, Goodell M A, Robertson C S, Liu H, Shine H D. Science. 2002;297:1299. doi: 10.1126/science.297.5585.1299. [DOI] [PubMed] [Google Scholar]
- 40.Trainor P A, Zhou S X, Parameswaran M, Quinlan G A, Gordon M A, Sturm K, Tam P P L. Molecular Embryology: Methods and Protocols. Vol. 97. Totowa, NJ: Humana; 1999. pp. 183–200. [DOI] [PubMed] [Google Scholar]
- 41.Hickey W F. Brain Pathol. 1991;1:97–105. doi: 10.1111/j.1750-3639.1991.tb00646.x. [DOI] [PubMed] [Google Scholar]