Abstract
A case of hepatocellular carcinoma (HCC) with pulmonary recurrence after liver transplantation for HCC is presented in this report. The patient showed disease progression on sorafenib therapy demonstrated by computed tomography scans as well as serial serum α-fetoprotein (AFP) elevation. After his immunosuppression therapy was successfully transitioned to sirolimus and a continuation of sorafenib, he achieved partial remission based on RECIST criteria and normalization of AFP. Mammalian target of rapamycin inhibitors including sirolimus alone or in conjunction with sorafenib may be useful in the treatment of post transplant HCC.
Keywords: Hepatocellular carcinoma, Liver transplantation, Mammalian target of rapamycin inhibitor, Sirolimus, Sorafenib
INTRODUCTION
Hepatocellular carcinoma (HCC) is a major health problem with a rising incidence in Western countries[1,2]. It is the third leading cause of cancer-related deaths worldwide, the seventh in the United States, and also the most common cause of death in patients with liver cirrhosis. The trend parallels an increase in advanced hepatitis C virus (HCV) related liver disease. The prevalence is much higher in developing countries and is associated with a significant morbidity and mortality. The treatment of HCC remains challenging due to the comorbidities with hepatitis B or C virus infection combined with decompensated liver cirrhosis. Only about 30% of patients with HCC are eligible for potentially curative treatment modalities such as liver resection, liver transplantation, or local ablation[3]. Conventional cytotoxic chemotherapeutic agents have been proven ineffective for patients with HCC[4]. Based on the SHARP trial[5], the Raf/vascular endothelial growth factor receptor (VEGF)/platelet derived growth factor receptor inhibitor, sorafenib, has increased the overall survival rate in patients with advanced HCC. The study was carried out in a highly selected population of HCC patients with liver function classified as Child-Pugh A and a small percentage of patients classified as Child-Pugh B. Orthotopic liver transplantation (OLT) for HCC has been highly successful with a low tumor recurrence rate in selected patient populations[6]. Tumor recurrence post OLT is the major obstacle in preventing successful liver transplantation in patients with HCC. Therefore, it is important to develop strategies for preventing HCC recurrence post OLT. Here, we present a case of HCC recurrence post OLT that was treated with sorafenib in addition to switching from tacrolimus to sirolimus as an immunosuppressant.
CASE REPORT
A 60-year-old male with a history of hepatitis C virus infection and liver cirrhosis was found to have several liver lesions consistent with HCC with an elevated α-fetoprotein (AFP) level on routine surveillance in December 2006. Based on his typical images and serum AFP level, the patient was diagnosed as HCC. He underwent an uneventful OLT at our liver transplant center in January 2007. Pathology showed multi-focal HCC with 4 lesions (3 lesions preoperatively), the largest measuring 5.0 cm × 3.0 cm × 2.9 cm. The tumors were moderately differentiated with vascular invasion and the pathologic staging was pT2N0Mx. The patient was given tacrolimus 4 mg twice a day for immunosuppression with serum tacrolimus levels at a range of 5-15 ng/mL after liver transplantation. HCV hepatitis recurrence was diagnosed by liver biopsy and serology in February 2008 when the patient had laboratory evidence of transaminase elevation. He continued on tacrolimus with no anti-HCV therapy and was followed up by the liver transplant team. In August 2008, he was admitted to the hospital for renal failure and hyperkalemia suspicious for tacrolimus toxicity with an elevated tacrolimus level at 16 ng/mL. His creatine level was 1.7 mg/dL. His tacrolimus dose was reduced to 2 mg twice a day orally and his tacrolimus target levels were at a low normal therapeutic range. During hospitalization, he was found to have new pulmonary nodules on a computed tomography (CT) scan. A biopsy confirmed those lesions to be metastatic HCC with its morphology similar to his original HCC. Due to bilateral pulmonary metastasis, the patient was not a candidate for surgery. He was referred to the medical oncology clinic in October 2008 for treatment. The recommendation was sorafenib therapy for his recurrent metastatic HCC after liver transplantation. Four weeks after he started on oral sorafenib at a dose of 400 mg twice a day from November 20, 2008, he developed the adverse effects of painful hand/foot syndrome. The dose of sorafenib was reduced to 200 mg orally twice a day on December 18, 2008. He tolerated sorafenib at 200 mg orally twice daily very well with less chest wall pain and resolution of hand/foot syndrome. His CT scan on January 26, 2008 showed progression of the disease based on RECIST criteria. Since the patient had improvement in symptoms, he wanted to continue taking sorafenib at 200 mg orally twice daily. Unfortunately, his AFP continued rising and reached a peak level of 19 817 ng/mL on March 26, 2009 (Figure 1). The metastatic tumors in his lungs were also increasing in size based on a CT scan performed on January 26, 2009. He was advised to consider systemic chemotherapy due to his excellent performance status and good graft liver function, but there was no other evidence based treatment available for him. He refused systemic chemotherapy after the literature of chemotherapy in HCC was reviewed with him. He was not eligible for an experimental trial since he was on an immunosuppressant and had active hepatitis C virus infection.
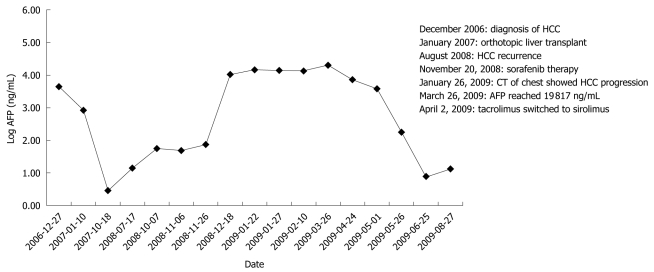
Figure 1.
Serum α-fetoprotein level measured as related to the treatment history. Serum α-fetoprotein (AFP) level was trending up after sorafenib therapy but normalized after sirolimus was initiated. Events are outlined in the Figure. CT: Computed tomography; HCC: Hepatocellular carcinoma.
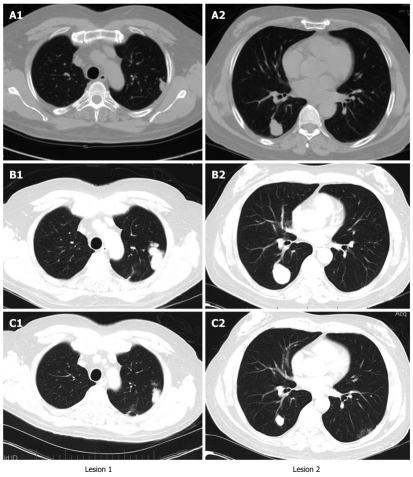
Given the positive preclinical data on mammalian target of rapamycin (mTOR) inhibitors in HCC as well as the potential benefit of sirolimus for HCC patients requiring immunosuppression, his immunosuppressant was switched from tacrolimus to sirolimus at 2 mg orally once daily for a week after the visit on March 26, 2009. He continued taking sorafenib at 200 mg orally twice a day against advice from his physician because he believed that it helped him symptomatically. He tolerated sirolimus at 2 mg orally daily with no obvious toxicity, and a desirable serum level (4-12 ng/mL) of sirolimus was achieved shortly after sirolimus therapy was initiated. His AFP dropped to normal limits and the CT showed a partial response (Figure 2). The dates for the representative images shown in Figure 2 were August 6, 2008, January 26, 2009 and August 26, 2009. There was no evidence of biochemical or radiographical progression when he was seen in the clinic on February 4, 2010.
Figure 2.
Computed tomographys of chest at different time points of the treatment. The lung lesions were increased in size after sorafenib therapy but significantly reduced in size after sirolimus was introduced in combination with sorafenib. The dates were August 6, 2008 (A), January 26, 2009 (B) and August 26, 2009 (C).
DISCUSSION
The treatment algorithm for HCC is based on functional status, liver functional reserve and stage of the tumor[3]. Surgical resection of HCC can be achieved in patients with a tumor that is amenable to surgical resection and has a good liver functional reserve. For smaller tumors, local therapy with radiofrequency ablation is used. OLT in selected patients is the most desirable modality because it cures both liver cirrhosis and HCC. Tumor recurrence is a cause of transplant failure in this population of patients. Adjuvant therapy for HCC post liver transplantation is an active research area but currently there is no evidence that supports the practice.
The treatment of recurrent HCC after liver transplantation is a great challenge in clinical practice. The use of immunosuppressant and liver transplantation are usually exclusion criteria for participation in clinical trials. The use of sorafenib in this setting is an extrapolation of data and by no means evidence-based care.
The most commonly used immunosuppressant after liver transplantation is a calcineurin inhibitor. In contrast, sirolimus, a mTOR inhibitor has also been used successfully for immunosuppression after liver transplantation[7]. In comparison to tacrolimus as an immunosuppressant, the advantages of sirolimus include less nephrotoxicity and potential antineoplastic properties. This makes it suitable for patients with renal insufficiency or HCC. The drug development for mTOR inhibitors has generated several novel anti-cancer mTOR inhibitors which have been approved by the FDA for the treatment of renal cell carcinoma[8,9]. The use of sirolimus has been limited to immunosuppression in organ transplantation patients. There is an ongoing phase 3 clinical trial (SiLVER trial) studying the use of sirolimus as an immunosuppressant in liver transplantation for high risk HCC to reduce tumor recurrence (ClinicalTrials.gov Identifier: NCT00355862). Currently, there is no standard therapy for HCC recurrence post OLT. Sorafenib has been used empirically for relapsing HCC after OLT, but there are no data supporting the use of sorafenib in this setting. Our own experience with sorafenib for recurrent HCC post liver transplantation is disappointed. Preclinical and clinical data indicate that mTOR inhibitors have significant anti-tumor activity for HCC[10-13]. There was a case report of complete remission of lung metastases from HCC post liver transplantation under therapy with sirolimus and mycophenolate mofetil[14], and experience with the use of sirolimus as an immunosuppressant for patients with HCC post OLT.
The use of sirolimus as an immunosuppressant is a reasonable approach for patients with HCC post liver transplantation for HCC or for patients with relapsing HCC post liver transplantation. The PI3K/Akt/mTOR signal pathway is involved in multiple cellular functions including proliferation, differentiation, tumorigenesis, and apoptosis. In about 15%-41% HCC patients, activation of the mTOR pathway is reported[15-17] and implicated in metastasis, invasion and prediction of poor prognosis[10,18]. Blocking of the mTOR pathway confers combined anticancer and immunosuppressive properties. Therefore, mTOR inhibitor treatment for HCC is directed by the molecular biology of HCC. Multiple clinical trials have been carried out using an mTOR inhibitor as an anticancer agent for HCC. Experimental data indicate that tumor inhibition can be accomplished by mTOR inhibitors while protecting allografts against rejection. The role of mTOR inhibitors as an immunosuppressant in HCC patients post OLT can be further determined depending on data from the SiLIVER trial. The case presented in this report clearly suggests that an mTOR inhibitor, sirolimus, may have antitumor activity in this patient with advanced metastatic relapsing HCC post OLT. This conclusion is also supported by published preclinical data[12]. The patient was at a high risk of tumor recurrence based on the presence of multifocal liver lesions and vascular invasion. Molecular evaluation of tumor samples from patients with HCC may give us an insight about post surgical or post transplantation adjuvant therapy using either sorafenib, sirolimus or their combination. The hypothesis that sirolimus synergizes with the anti-tumor activity of sorafenib is supported by pre-clinical data[12], but whether this applies to the patient reported here is unknown. The tumor sample is being analyzed for Erk/MAPK pathway and PI3/Akt/mTOR pathway abnormalities. The fact that the patient did not derive any apparent benefit from more than 4 mo of sorafenib therapy alone after his tumor recurrence indicated that his HCC had inherent resistance to the Raf/VEGF/platelet derived growth factor receptor inhibitor, sorafenib. No reports are available on the delayed anti-tumor effects of sorafenib. Biomarkers that predict the effectiveness of mTOR inhibitor and/or sorafenib therapy and clinical trials to explore the effectiveness of sirolimus alone and in combination with sorafenib with molecular pathway correlation analysis are justified.
In conclusion, mTOR inhibitor, sirolimus, may have anti-tumor activity in HCC. Use of sirolimus as an immunosuppressant for HCC post OLT is being investigated, and its use in HCC alone or in combination with sorafenib warrants further investigation with biomarker correlation.
Footnotes
Peer reviewer: Taku Aoki, MD, Division of Hepato-Biliary-Pancreatic and Transplantation Surgery, Department of Surgery, Graduate School of Medicine, University of Tokyo, 7-3-1 Hongo, Bunkyo-ku, Tokyo, 113-8655, Japan
S- Editor Tian L L- Editor Wang XL E- Editor Zheng XM
References
- 1.El-Serag HB, Lau M, Eschbach K, Davila J, Goodwin J. Epidemiology of hepatocellular carcinoma in Hispanics in the United States. Arch Intern Med. 2007;167:1983–1989. doi: 10.1001/archinte.167.18.1983. [DOI] [PubMed] [Google Scholar]
- 2.Sangiovanni A, Del Ninno E, Fasani P, De Fazio C, Ronchi G, Romeo R, Morabito A, De Franchis R, Colombo M. Increased survival of cirrhotic patients with a hepatocellular carcinoma detected during surveillance. Gastroenterology. 2004;126:1005–1014. doi: 10.1053/j.gastro.2003.12.049. [DOI] [PubMed] [Google Scholar]
- 3.Llovet JM, Burroughs A, Bruix J. Hepatocellular carcinoma. Lancet. 2003;362:1907–1917. doi: 10.1016/S0140-6736(03)14964-1. [DOI] [PubMed] [Google Scholar]
- 4.Thomas MB. Systemic therapy for hepatocellular carcinoma. Cancer J. 2008;14:123–127. doi: 10.1097/PPO.0b013e31816a6058. [DOI] [PubMed] [Google Scholar]
- 5.Llovet JM, Ricci S, Mazzaferro V, Hilgard P, Gane E, Blanc JF, de Oliveira AC, Santoro A, Raoul JL, Forner A, et al. Sorafenib in advanced hepatocellular carcinoma. N Engl J Med. 2008;359:378–390. doi: 10.1056/NEJMoa0708857. [DOI] [PubMed] [Google Scholar]
- 6.Takada Y, Uemoto S. Liver transplantation for hepatocellular carcinoma: the Kyoto experience. J Hepatobiliary Pancreat Sci. 2010;17:527–532. doi: 10.1007/s00534-009-0162-y. [DOI] [PubMed] [Google Scholar]
- 7.Nocera A, Andorno E, Tagliamacco A, Morelli N, Bottino G, Ravazzoni F, Casaccia M, Barocci S, Alice S, Santori G, et al. Sirolimus therapy in liver transplant patients: an initial experience at a single center. Transplant Proc. 2008;40:1950–1952. doi: 10.1016/j.transproceed.2008.05.005. [DOI] [PubMed] [Google Scholar]
- 8.Atkins MB, Hidalgo M, Stadler WM, Logan TF, Dutcher JP, Hudes GR, Park Y, Liou SH, Marshall B, Boni JP, et al. Randomized phase II study of multiple dose levels of CCI-779, a novel mammalian target of rapamycin kinase inhibitor, in patients with advanced refractory renal cell carcinoma. J Clin Oncol. 2004;22:909–918. doi: 10.1200/JCO.2004.08.185. [DOI] [PubMed] [Google Scholar]
- 9.Staehler M, Rohrmann K, Haseke N, Stief CG, Siebels M. Targeted agents for the treatment of advanced renal cell carcinoma. Curr Drug Targets. 2005;6:835–846. doi: 10.2174/138945005774574498. [DOI] [PubMed] [Google Scholar]
- 10.Chen JS, Wang Q, Fu XH, Huang XH, Chen XL, Cao LQ, Chen LZ, Tan HX, Li W, Bi J, et al. Involvement of PI3K/PTEN/AKT/mTOR pathway in invasion and metastasis in hepatocellular carcinoma: Association with MMP-9. Hepatol Res. 2009;39:177–186. doi: 10.1111/j.1872-034X.2008.00449.x. [DOI] [PubMed] [Google Scholar]
- 11.Huynh H, Chow KH, Soo KC, Toh HC, Choo SP, Foo KF, Poon D, Ngo VC, Tran E. RAD001 (everolimus) inhibits tumour growth in xenograft models of human hepatocellular carcinoma. J Cell Mol Med. 2009;13:1371–1380. doi: 10.1111/j.1582-4934.2008.00364.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 12.Huynh H, Ngo VC, Koong HN, Poon D, Choo SP, Thng CH, Chow P, Ong HS, Chung A, Soo KC. Sorafenib and rapamycin induce growth suppression in mouse models of hepatocellular carcinoma. J Cell Mol Med. 2009;13:2673–2683. doi: 10.1111/j.1582-4934.2009.00692.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 13.Rizell M, Andersson M, Cahlin C, Hafström L, Olausson M, Lindnér P. Effects of the mTOR inhibitor sirolimus in patients with hepatocellular and cholangiocellular cancer. Int J Clin Oncol. 2008;13:66–70. doi: 10.1007/s10147-007-0733-3. [DOI] [PubMed] [Google Scholar]
- 14.Elsharkawi M, Staib L, Henne-Bruns D, Mayer J. Complete remission of postransplant lung metastases from hepatocellular carcinoma under therapy with sirolimus and mycophenolate mofetil. Transplantation. 2005;79:855–857. doi: 10.1097/01.tp.0000154913.88193.ff. [DOI] [PubMed] [Google Scholar]
- 15.Llovet JM, Chen Y, Wurmbach E, Roayaie S, Fiel MI, Schwartz M, Thung SN, Khitrov G, Zhang W, Villanueva A, et al. A molecular signature to discriminate dysplastic nodules from early hepatocellular carcinoma in HCV cirrhosis. Gastroenterology. 2006;131:1758–1767. doi: 10.1053/j.gastro.2006.09.014. [DOI] [PubMed] [Google Scholar]
- 16.Semela D, Piguet AC, Kolev M, Schmitter K, Hlushchuk R, Djonov V, Stoupis C, Dufour JF. Vascular remodeling and antitumoral effects of mTOR inhibition in a rat model of hepatocellular carcinoma. J Hepatol. 2007;46:840–848. doi: 10.1016/j.jhep.2006.11.021. [DOI] [PubMed] [Google Scholar]
- 17.Sieghart W, Fuereder T, Schmid K, Cejka D, Werzowa J, Wrba F, Wang X, Gruber D, Rasoul-Rockenschaub S, Peck-Radosavljevic M, et al. Mammalian target of rapamycin pathway activity in hepatocellular carcinomas of patients undergoing liver transplantation. Transplantation. 2007;83:425–432. doi: 10.1097/01.tp.0000252780.42104.95. [DOI] [PubMed] [Google Scholar]
- 18.Zhou L, Huang Y, Li J, Wang Z. The mTOR pathway is associated with the poor prognosis of human hepatocellular carcinoma. Med Oncol. 2010;27:255–261. doi: 10.1007/s12032-009-9201-4. [DOI] [PubMed] [Google Scholar]