Abstract
The aim of this study was to evaluate the efficacy of multiple platelet-rich plasma (PRP) injections on the healing of chronic refractory patellar tendinopathy after previous classical treatments have failed. We treated 15 patients affected by chronic jumper’s knee, who had failed previous nonsurgical or surgical treatments, with multiple PRP injections and physiotherapy. We also compared the clinical outcome with a homogeneous group of 16 patients primarily treated exclusively with the physiotherapy approach. Multiple PRP injections were performed on three occasions two weeks apart into the site of patellar tendinopathy. Tegner, EQ VAS and pain level were used for clinical evaluation before, at the end of the treatment and at six months follow-up. Complications, functional recovery and patient satisfaction were also recorded. A statistically significant improvement in all scores was observed at the end of the PRP injections in patients with chronic refractory patellar tendinopathy and a further improvement was noted at six months, after physiotherapy was added. Moreover, comparable results were obtained with respect to the less severe cases in the EQ VAS score and pain level evaluation, as in time to recover and patient satisfaction, with an even higher improvement in the sport activity level achieved in the PRP group. The clinical results are encouraging, indicating that PRP injections have the potential to promote the achievement of a satisfactory clinical outcome, even in difficult cases with chronic refractory tendinopathy after previous classical treatments have failed.
Introduction
Jumper’s knee is a disabling and often chronic condition that affects athletes in many sports, particularly elite athletes engaged in explosive jumping sports. Epidemiological evaluations showed that the average duration of substantial pain problems and reduced function is nearly three years [1] and that at 15 years’ follow-up 53% of the subjects reported quitting their sports career due to the damaged knee [2].
Various therapeutic approaches have been adopted for the treatment of patellar tendinopathy. The initial treatment of jumper’s knee typically includes rest, ice, electrotherapy, massage, taping, anti-inflammatory medication or corticosteroid injections [3]. Also, physiotherapy, including eccentric training protocols [4], extra-corporeal shock-wave therapy [5] and sclerosant injections [6] have been proposed, but in the difficult cases, where results are poor, surgical treatment may be considered. Surgery, with decompression, resection and open stimulation of the affected tendon, is recommended for athletes whose sports careers seem to be in danger, and good results have been reported [7]. However, for the most part, treatment options are poorly researched and outcomes after both conservative and surgical treatment may offer incomplete recovery and difficulties returning to sport [8].
Recent knowledge regarding tissue biology highlights a complex regulation of growth factors in the normal tissue structure and in the reaction to tissue damage and shows an important role and effectiveness for growth factor application in the healing of damaged tissue. Studies showed the role of TGF-β in increasing the expression of procollagen type I and III and improving tendon mechanical properties [9, 10]. PDGF-BB, IGF-1,VEGF and BFGF promote tendon cell proliferation and also tendon healing [11]. It has been demonstrated that the healing tendon is responsive to the local application of growth factors and the fact that platelets secrete growth factors and active metabolites means that their applied use can have a positive influence on damaged tissues with low healing potential. Platelet-rich plasma (PRP) is a method which provides many growth factors in a simple, low cost and minimally invasive way.
The purpose of our study was to evaluate the therapeutic effects of multiple PRP injections to promote the healing of chronic refractory patellar tendinopathy after previous classical treatments have failed. We also compared the results obtained in this group of patients with a homogeneous group of patients, primarily treated exclusively with the physiotherapy protocol, to understand better the actual role of PRP in increasing the tendon healing potential.
Materials and methods
Patient selection
Clinical experimentation was approved by the Hospital Ethics Committee and informed consent of all patients was obtained. Two groups of patients were enrolled and treated either with PRP and physiotherapy or with the physiotherapy approach alone.
In the PRP group, the inclusion criteria used were history (>3 months) of exercise-associated pain, pain or tenderness on palpation and imaging findings of degenerative changes. For this group we enrolled only patients who had failed previous nonsurgical or surgical treatments, including all or a combination of rest for at least two months, conventional stretching exercises and strengthening, and were classified as grade III-B according to the criteria originally described by Blanzina and later modified by Lian et al. [12] (patients unable to participate in sports at the same level as before the onset of symptoms). Exclusion criteria were: systemic disorders such as diabetes, rheumatoid arthritis, coagulopathies, cardiovascular diseases, infections, immunodepression, patients in therapy with anticoagulants–antiaggregants, use of NSAIDs in the five days before blood donation, Hb values of <11 and platelets values of <150,000/mmc. Fifteen consecutive patients with refractory jumper’s knee were treated and prospectively evaluated at a minimum of six months’ follow-up.
In the control group, the same inclusion and exclusion criteria were used for this group of patients affected by jumper’s knee. The only difference was related to previous treatments, i.e. for this group we selected patients who had not undergone any treatment (for at least two months) and were primarily treated with the physiotherapy protocol alone. A group of 16 consecutive patients, treated during the same period by the same surgeons, was enrolled.
The groups were matched for sex, age and sport activity level; the only differences were regarding previous treatment (due to the study design) and duration of symptoms, which was markedly longer in the PRP group (p < 0.0005) (Table 1).
Table 1.
Comparison of the characteristics of the two groups of patients evaluated and treated
| Characteristic | Platelet-rich plasma (PRP) group | Control group |
|---|---|---|
| Number of patients | 15 | 16 |
| Age | 28.8 ± 8.5 | 25.5 ± 9.2 |
| Sex | Male | Male |
| Bilateral | 5 | 1 |
| Sports activity level | 7.5 ± 1.6 | 7.8 ± 1.3 |
| Duration of symptoms (mo) | 24.1 ± 19.9 | 8.4 ± 4.1 |
| Previous treatments | 15 | 7 |
| Rest (at least 2 mo) | 15 | 0 |
| Stretching exercises | 15 | 0 |
| Strengthening exercises | 15 | 2 |
| Ultrasound therapy | 10 | 1 |
| Shock wave therapy | 7 | 0 |
| Laser therapy | 7 | 7 |
| Deep tendon massage | 6 | 1 |
| Corticosteroids injections | 3 | 0 |
| Sclerotherapy | 1 | 0 |
| Surgery | 5 | 0 |
| Pain level | 6.6 ± 1.4 | 6.7 ± 1.5 |
| EQ VAS score | 52.7 ± 22.2 | 50.6 ± 22.3 |
Treatment procedure and follow-up
The procedure described by Kon et al. [13] was used for the preparation and injection of the platelet concentrate and for the postinjection phase. The procedure consisted of two centrifugations (the first at 1800 rpm for 15 minutes and the second at 3500 rpm for 10 minutes) of a 150-ml venous blood sample in order to concentrate platelets and produce 20 ml of PRP. The unit of PRP was divided into four small units of 5 ml each, whereby one unit was sent to the laboratory for analysis, one unit was used for the first injection within two hours, and the other two units were stored at −30°C. The total number of platelets per milliliter presented a concentration 6.1 ± 1.6 higher compared with whole blood values, and an average of 6.5 ± 1.5 million platelets were administered at every injection. Injections were performed every 15 days. For the second and third treatments the samples were thawed in a dry-thermostat at 37°C for 30’ just before application. Before every injection, 10% of Ca-chloride (Ca++ = 0.22 mEq x dose) was added to the PRP concentrate to activate platelets.
After the injection, the patients were sent home with instructions to limit the use of the leg for at least 24 hours and to use cold therapy for pain. Between the first and the second injection rest was indicated. After the second injection stretching exercises and mild activities (such as bicycle exercise and mild exercises in a pool) were allowed, whereas after the third application patients were recommended to continue with stretching and light activities, and to begin a strengthening program and proceed gradually after one month with normal sports or recreational activities as tolerated. The same physiotherapy protocol was used for the other group of patients.
Patients were clinically prospectively evaluated before, at the end of the treatment and at six months follow-up. Participants with bilateral jumper’s knee were asked to select the most symptomatic knee. Tegner, EQ VAS and pain level were used for clinical evaluation. Complications, functional recovery and patient satisfaction were also recorded.
Statistical methods
All continuous data are expressed in terms of mean ± SD. Categorical variables are expressed as proportions or percentages. The Kolmogorov-Smirnov test was performed to test normality of continuous variables. For the non-normal distribution of evaluated variables non-parametric tests were used. The Wilcoxon test was used for paired comparisons and the Mann Whitney test was used to investigate data differences between the two groups. Pearson’s Chi square test was performed to investigate the relationships between grouping variables. For all tests p < 0.05 was considered significant.
Results
No complications related to the injections or severe adverse events were observed during the treatment and follow-up period.
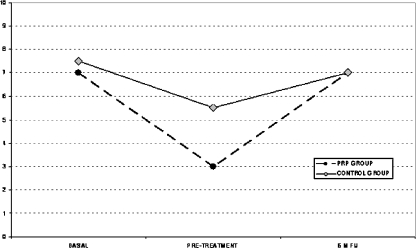
PRP group Statistical analysis showed a significant improvement in the EQ VAS (p = 0.004) from basal evaluation (52.7 ± 22.0) to the end of the injection cycle (68.0 ± 13.9), and a further improvement was present at six months’ follow-up (78.3 ± 13.3) (p = 0.007) (Fig. 1). Similarly, a significant decrease in the pain level (p = 0.002) was achieved from basal evaluation (6.6 ± 1.4) to the end of the PRP injections (4.3 ± 1.7), and a further improvement was present at six months’ follow-up (3.1 ± 1.2) (p = 0.02) (Fig. 2). Sport activity, which was evaluated using the Tegner score, showed an improvement (p = 0.001) from pre-treatment level (3.7 ± 1.8) to six months’ follow-up (6.6 ± 2.4); most of the patients returned to their sport, with a lower score, which was not significantly different compared to the previous activity level (7.5 ± 1.6) (Fig. 3). Evaluation of the functional recovery showed five patients with a complete recovery, six with a marked improvement, and two with a mild improvement. There was a mean return-to-sport time of 3.0 ± 1.1 months, whereas in two cases we did not obtain any improvement, thus the patient satisfaction was 86.7% (13/15).
Fig. 1.
Comparison of the health status, evaluated with EQ-VAS, in the platelet-rich plasma (PRP) and control groups
Fig. 2.
Comparison of the pain level, evaluated on a 1–10 scale, in the platelet-rich plasma (PRP) and control groups
Fig. 3.
Comparison of the sports activity level, evaluated with the Tegner score, in the platelet-rich plasma (PRP) and control groups
Control group Statistical analysis showed a significant improvement in the EQ VAS (p = 0.006) from basal evaluation (50.6 ± 22.3) to the end of the therapy (72.5 ± 24.7), and the results remained stable at six months’ follow-up (73.5 ± 26.3) (Fig. 1). Similarly, a significant decrease in the pain level (p = 0.001) was achieved from basal evaluation (6.7 ± 1.5) to the end of therapy (3.2 ± 2.4) and six months’ follow-up (3.7 ± 2.8) (Fig. 2). The sport activity showed an improvement (p < 0.0005) from pre-treatment level (5.3 ± 2.0) to six months’ follow-up (6.8 ± 1.6); most of the patients returned to their sport, but with a mean lower score with respect to the previous activity level (7.8 ± 1.3) (p = 0.01) (Fig. 3). The evaluation of the functional recovery showed four patients with a complete recovery, four with a marked improvement, and five with a mild improvement. There was a mean return-to-sport time of 3.2 ± 1.6 months, whereas in three cases we did not obtain any improvement. The patient satisfaction was 68.8% (11/16).
When comparing the two groups, no statistically significant differences were obtained with the EQ VAS and pain level evaluation, as with time-to-recover and patient satisfaction, whereas a greater improvement in the sport activity level was achieved in the PRP group (percentage improvement, 39 ± 22%) with respect to the control group (20 ± 27%) (p = 0.048) (Fig. 4).
Fig. 4.
A 22-year-old soccer player with a three year history of refractory jumper’s knee who failed both non-operative treatments and surgery. The patient returned to previous activity level after four months; MRI shows the patellar tendon before treatment (a) and the improvement of the tendon structure after the platelet-rich plasma (PRP) injections at six months’ follow-up (b)
Discussion
Jumper’s knee is a common overuse injury in athletes that occurs because of the high chronic repetitive loading that surpasses the adaptive abilities of the tendon and causes micro tears and degeneration in the tendon substance.
Many factors have been suspected to predispose patients to this condition [2] by increasing the patellar tendon overload. The high chronic repetitive loading stimulates the local release of cytokines, with an autocrine and paracrine modulation of cell activity [14], that fails to adapt to continued abusive load and irritation and leads to intratendinous damage. The poor regeneration capacity of tendons, explained by the poor vascularity, oxygenation and nutrition of this tissue, cannot cope with the applied forces and explains the low healing potential and the difficulties in the treatment of this chronic tendon disease [14].
Recently, some authors [15, 16] have postulated that autologous blood injections might provide the necessary cellular and humoral mediators to induce the healing cascade and promote tendon repair. The rationale was based on the mitomorphogenic activity of blood growth factors and the inflammatory response induced in an otherwise degenerative process, leading to strengthening of the tendon through the formation of granulation tissue and tissue regeneration, in a tendon with an increased healing potential. The effect of autologous blood injections into the tendon have been evaluated in vitro and in vivo, whereby an increase in rabbit patellar tendon strength has been assessed [15]. Clinical applications for the treatment of chronic tendinopathies have also showed good results [16–19].
The benefits are the result of some kind of bleeding, which stimulates the healing response through chemical modifiers of cellular activity contained in the blood. In cases of injury, platelets are the first cells carried to the lesion site and, in fact, they play a key role in mediating healing of the damaged tissue because of the capacity to release growth factors from their α-granules [20]. Platelets contain storage pools of growth factors including PDGF, TGF-β, VEGF, IGF-1, FGF, and cytokines, chemokines and metabolites [20]. Platelet-rich plasma is a concentrate of platelets and therefore autologous growth factors. Additionally, their administration in the form of platelet gel provides an adhesive support that can confine secretion to a chosen site.
As in the earlier described technique based on the injection of autologous blood, PRP injections combine the dry needling stimulus, which may induce internal haemorrhage and consequently an inflammatory response and a repair process, with the biological stimulus of the injected platelet growth factors. Releasate from platelet-rich plasma has been seen to activate circulation-derived cells [21] that play a crucial role in the tissue healing process and stimulate gene expression of the matrix molecules, collagen production and tendon cell proliferation [22]. In vitro studies demonstrated this mitogenic activity and that the stimulated tenocytes synthesise VEGF and HGF, thus suggesting a beneficial effect for the treatment of tendon injuries by inducing cell proliferation and promoting the synthesis of angiogenic factors during the healing process. An animal model has also confirmed the usefulness of platelet concentrate for the treatment of tendon damage [23], with an increased tendon callus strength and stiffness after percutaneous injections in transacted tendons; a more rapid recovery in surgically repaired tendons has also been seen in a human study [24]. Currently, PRP is widely used experimentally in different fields of medicine, but the evidence base for the clinical use of PRP is still in its infancy. Only a few articles specifically address treatment applications in the orthopaedic field and, to our knowledge, only one study has been published regarding the treatment of tendinopathy through PRP injections. Mishra treated patients affected by severe chronic tennis elbow and reported promising results [25], with improvement in pain and function and no complications. The purpose of our study was to investigate this novel approach for the treatment of severe chronic patellar tendinopathy. We recruited and evaluated prospectively athletes who failed classical therapies. The rationale of the study was to test the use of PRP only in failed cases and compare the results achieved by treating recalcitrant tendinopathy through PRP injections and physiotherapy with those obtained in a homogeneous group of patients with jumper’s knee primarily treated only with the physiotherapy protocol. It was difficult to have a control group for this study, because it was impossible and unethical to have a group of patients who had just failed previous physiotherapy, to be treated once again with classical therapies for control. However, the non-homogeneous control group represents one of the main biases of this study. The fact that patients were subjected to two therapies simultaneously represents another limitation of our study, since we do not know the relative importance and the real contributions of PRP and physiotherapy to the therapeutic outcome. However, all the patients treated with PRP had previously failed the physiotherapy treatments, and therefore we believe that the PRP injection may have increased the tissue reparative capability and may have played an important role in the tendon reparative process. Other weak points of this study are represented by the lack of randomisation (not usable in this case due to the pre-determined different selection criteria) and the small number of patients treated; nonetheless, this study shows interesting results that deserve to be elaborated and confirmed in wider randomised studies.
After evaluating the safety and feasibility of this procedure in a preliminary study [13], we used this biological approach to treat 15 patients affected by pain and tenderness at the inferior pole of the patella (in most of the cases the proximal–central part of the tendon also presented degenerative changes and was symptomatic) for more than three months despite rest and classical therapies. In our previous study we underlined the necessity to combine PRP treatment with physiotherapy, i.e. after injections, the patients followed a standardised physiotherapy protocol to aid rehabilitation before returning to full sporting activity. Stretching and strengthening therapies play a key role after the PRP injections, due to the interplay between biological and mechanical stimulation, as shown in an animal model [23]. On the other hand, the better function of the extensor apparatus may reduce the overload, thus aiding healing of the tendon and maintaining the results over time.
We observed a statistically significant improvement in all scores evaluated at the end of the therapy and at six months’ follow-up. Knee function and quality of life markedly improved, and the majority of the patients had a good recovery, returned to their previous sporting activity level and were satisfied with the results of the treatment. The analysis showed comparable results with the control group, with an even higher improvement in sports activity level in the PRP group. In other words, patients with a long history (much longer with respect to that of the control group) of chronic refractory jumper’s knee, who had previous failed nonsurgical or even surgical treatments, were able, through the combination of multiple PRP injections and physiotherapy, to achieve the same results obtainable in less severe cases. This was probably due to an increased tendon healing potential, but needs to be verified through imaging and histological evaluation.
Conclusions
This report investigates the use of autologous growth factors through platelet-rich plasma injections as a treatment for jumper’s knee. The clinical results of our study are encouraging and suggest this method can be useful for the treatment of chronic patellar tendinopathy, even in difficult cases with refractory tendinopathy where the only physiotherapy approach had failed.
Acknowledgments
Thanks go to M. Lo Presti, M. Delcogliano, S. Zaffagnini, A. Bondi, A. Di Martino, and S. Bassini from the IX Division – Biomechanics Lab, Rizzoli Orthopedic Institute, Bologna, Italy; to A. Cenacchi, F. Pieretti, A. Gabriele, M. Vaccari, A.M. Del Vento, M. Zagarella, V. Roverini, I. Brognara, L. D’Amato and S. Ardone from the Immunohematology and Transfusion Medicine Service, Rizzoli Orthopedic Institute, Bologna, Italy; to E. Pignotti, and K. Smith from Task Force, Rizzoli Orthopedic Institute, Bologna, Italy; and to G. Nanni from Isokinetic, Education Research Department, Bologna, Italy.
Contributor Information
Giuseppe Filardo, Phone: +39-51-6366567, FAX: +39-51-583789, Email: g.filardo@biomec.ior.it.
Elizaveta Kon, Email: e.kon@biomec.ior.it.
Stefano Della Villa, Email: s.dellavilla@isokinetic.com.
Ferruccio Vincentelli, Email: f.vincentelli@isokinetic.com.
Pier Maria Fornasari, Email: piermaria.fornasari@ior.it.
Maurilio Marcacci, Email: m.marcacci@biomec.ior.it.
References
- 1.Lian OB, Engebretsen L, Bahr R. Prevalence of jumper’s knee among elite athletes from different sports: a cross-sectional study. Am J Sport Med. 2005;33:561–567. doi: 10.1177/0363546504270454. [DOI] [PubMed] [Google Scholar]
- 2.Kettunen JA, Kvist M, Alanen E, et al. Long term prognosis for jumper’s knee in male athletes. A prospective follow-up study. Am J Sport Med. 2002;30(5):689–692. doi: 10.1177/03635465020300051001. [DOI] [PubMed] [Google Scholar]
- 3.Peers KH, Lysens RJ. Patellar tendinopathy in athletes: current diagnostic and therapeutic recommendations. Sports Med. 2005;35:71–87. doi: 10.2165/00007256-200535010-00006. [DOI] [PubMed] [Google Scholar]
- 4.Young MA, Cook JL, Purdam CR, et al. Eccentric decline squat protocol offers superior results at 12 months compared with traditional eccentric protocol for patellar tendinopathy in volleyball players. Br J Sports Med. 2005;39(2):102–105. doi: 10.1136/bjsm.2003.010587. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 5.Peers KH, Lysens RJ, Brys P, Bellemans J. Cross-sectional outcome analysis of athletes with chronic patellar tendinopathy treated surgically and by extracorporeal shock wave therapy. Clin J Sport Med. 2003;13(2):79–83. doi: 10.1097/00042752-200303000-00003. [DOI] [PubMed] [Google Scholar]
- 6.Alfredson H, Ohberg L. Neovascularisation in chronic painful patellar tendinosis. Promising results after sclerosing neovessels outside the tendon challenge the need for surgery. Knee Surg Sports Traumatol Arthrosc. 2005;13(2):74–80. doi: 10.1007/s00167-004-0549-x. [DOI] [PubMed] [Google Scholar]
- 7.Ferretti A, Puddu G, Mariani PP, Neri M. The natural history of jumper's knee. Patellar or quadriceps tendonitis. Int Orthop. 1985;8(4):239–242. doi: 10.1007/BF00266866. [DOI] [PubMed] [Google Scholar]
- 8.Almekinders L, Temple J. Etiology, diagnosis and treatment of tendonitis: an analysis of the literature. Med Sci Sport Exerc. 1998;30(8):1183–1190. doi: 10.1097/00005768-199808000-00001. [DOI] [PubMed] [Google Scholar]
- 9.Kashiwagi K, Mochizuki Y, Yasunaga Y, et al. Effects of transforming growth factor-beta 1 on the early stages of healing of the Achilles tendon in a rat model. Scand J Plast Reconstr Surg Hand Surg. 2004;38(4):193–197. doi: 10.1080/02844310410029110. [DOI] [PubMed] [Google Scholar]
- 10.Katsura T, Tohyama H, Kondo E, et al. Effects of administration of transforming growth factor (TGF)-beta1 and anti-TGF-beta1 antibody on the mechanical properties of the stress-shielded patellar tendon. J Biomech. 2006;39(14):2566–2572. doi: 10.1016/j.jbiomech.2005.09.001. [DOI] [PubMed] [Google Scholar]
- 11.Costa MA, Wu C, Pham BV, et al. Tissue engineering of flexor tendons: optimization of tenocyte proliferation using growth factor supplementation. Tissue Eng. 2006;12(7):1937–1943. doi: 10.1089/ten.2006.12.1937. [DOI] [PubMed] [Google Scholar]
- 12.Lian O, Holen KJ, Engebretsen L, et al. Relationship between symptoms of jumper’s knee and the ultrasound characteristics of the patella tendon among high level male volleyball players. Scand J Med Sci Sports. 1996;6:291–296. doi: 10.1111/j.1600-0838.1996.tb00473.x. [DOI] [PubMed] [Google Scholar]
- 13.Kon E, Filardo G, Delcogliano M, et al. Platelet-rich plasma: new clinical application: a pilot study for treatment of jumper's knee. Injury. 2009;40(6):598–603. doi: 10.1016/j.injury.2008.11.026. [DOI] [PubMed] [Google Scholar]
- 14.Josza LG, Kannus P (1997) Human tendons. Anatomy, physiology, and pathology. Human Kinetics Books pp 178–184
- 15.Taylor MA, Norman TL, Clovis NB, et al. The response of rabbit patellar tendons after autologous blood injection. Med Sci Sports Exerc. 2002;34(1):70–73. doi: 10.1097/00005768-200201000-00012. [DOI] [PubMed] [Google Scholar]
- 16.Edwards SG, Calandruccio JH. Autologous blood injections for refractory lateral epicondylitis. J Hand Surg [Am] 2003;28(2):272–278. doi: 10.1053/jhsu.2003.50041. [DOI] [PubMed] [Google Scholar]
- 17.Suresh SP, Ali KE, Jones H, et al. Medial epicondylitis: is ultrasound guided autologous blood injection an effective treatment? Br J Sports Med. 2006;40(11):935–939. doi: 10.1136/bjsm.2006.029983. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 18.Connell DA, Ali KE, Ahmad M, et al. Ultrasound-guided autologous blood injection for tennis elbow. Skeletal Radiol. 2006;35(6):371–377. doi: 10.1007/s00256-006-0081-9. [DOI] [PubMed] [Google Scholar]
- 19.James SL, Ali K, Pocock C, et al. Ultrasound guided dry needling and autologous blood injection for patellar tendinosis. Br J Sports Med. 2007;41(8):518–521. doi: 10.1136/bjsm.2006.034686. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 20.Sanchez A, Sheridan P, Kupp L. Is platelet-rich plasma the perfect enhancement factor? A current review. Int J Oral Maxillofac Implants. 2003;18(1):93–103. [PubMed] [Google Scholar]
- 21.Kajikawa Y, Morihara T, Sakamoto H, et al. Platelet-rich plasma enhances the initial mobilization of circulation-derived cells for tendon healing. J Cell Physiol. 2008;215(3):837–845. doi: 10.1002/jcp.21368. [DOI] [PubMed] [Google Scholar]
- 22.Mos M, Windt AE, Jahr H, et al. Can platelet-rich plasma enhance tendon repair? A cell culture study. Am J Sports Med. 2008;36(6):1171–1178. doi: 10.1177/0363546508314430. [DOI] [PubMed] [Google Scholar]
- 23.Virchenko O, Aspenberg P. How can one platelet injection after tendon injury lead to a stronger tendon after 4 weeks? Interplay between early regeneration and mechanical stimulation. Acta Orthop. 2006;77(5):806–812. doi: 10.1080/17453670610013033. [DOI] [PubMed] [Google Scholar]
- 24.Sanchez M, Anitua E, Azofra J, et al. Comparison of surgically repaired Achilles tendon tears using platelet-rich fibrin matrices. Am J Sports Med. 2007;35(2):245–251. doi: 10.1177/0363546506294078. [DOI] [PubMed] [Google Scholar]
- 25.Mishra A, Pavelko T. Treatment of chronic elbow tendinosis with buffered platelet-rich plasma. Am J Sport Med. 2006;34(11):1774–1778. doi: 10.1177/0363546506288850. [DOI] [PubMed] [Google Scholar]