Abstract
The bone marrow is an invaluable source of adult pluripotent stem cells, as it gives rise to hematopoietic stem cells, endothelial progenitor cells, and mesenchymal cells, amongst others. The use of bone marrow-derived stem cell (BMC) transplantation (BMT) may be of assistance in achieving tissue repair and regeneration, as well as in modulating immune responses in the context of autoimmunity and transplantation. Ongoing clinical trials are evaluating the effects of BMC to preserve functional beta-cell mass in subjects with type 1 and type 2 diabetes, and to favor engraftment and survival of transplanted islets. Additional trials are evaluating the impact of BMT (i.e., mesenchymal stem cells) on the progression of diabetes complications. This article reviews the progress in the field of BMC for the treatment of subjects with insulin-dependent diabetes, and summarizes clinical data of pilot studies performed over the last two decades at our research center by combining allogeneic islet transplantation with donor-specific BMC.
Keywords: bone marrow-derived stem cell, diabetes, mesenchymal stem cell, transplant, islet transplantation, beta-cell replacement, chimerism, clinical trial, tolerance
Abbreviations: ALG - anti-lymphocyte globulin; ATG - anti-thymocyte globulin; AZA - azathioprine; BID - twice daily; BM - bone marrow; BMSC - bone marrow-derived stem cell; BMT - bone marrow-derived stem cell transplantation; C1H - Campath-1H; CD3 - cluster of differentiation 3 (surface glycoproteins associated with the T cell receptor to activate T cells); CD34 - cluster of differentiation 34 (surface glycoprotein, cell-cell adhesion factor, mediates attachment of stem cells to bone marrow); CyA - cyclosporine A; DAC - daclizumab; EPC - endothelial progenitor cell; ETC - etanercept; GAD - glutamic acid decarboxylase; G-CSF - granulocyte colony-stimulating factor; GM-CSF - granulocyte-macrophage colony-stimulating factor; GvHD - graft-versus-host disease; HbA1c - glycosylated hemoglobin A1c; HOMA-B - homeostasis model assessment beta-cell (score for assessment of beta-bell function); HOMA-IR - homeostasis model assessment insulin resistance (score for assessment of insulin resistance); HOT - hyperbaric oxygen therapy; hrG-CSF - human recombinant granulocyte colony-stimulating factor; HSC - hematopoietic stem cell; IAK - islet after kidney; IEQ - islet equivalent; IFN - interferon; INF - infliximab; IRDM - insulin-requiring diabetes mellitus; ITA - islet transplantation alone; MHC - major histocompatibility complex; MMF - mycophenolate mofetil; MP - methylprednisolone; MSC - mesenchymal stem cell; mTOR - mammalian target of rapamycin (regulates cell growth, proliferation, motility, and survival); NCT - national clinical trial; NIDDK - National Institute of Diabetes and Digestive and Kidney Diseases; OKT3 - muromonab-CD3 (trade name orthoclone OKT3; monoclonal antibody targeted against the CD3 receptor); SIK - simultaneous islet-kidney (transplantation); SIL - simultaneous islet-liver (transplantation); SIR - sirolimus; TAC - tacrolimus; T1D - type 1 diabetes; T2D - type 2 diabetes; TNF - tumor necrosis factor
Introduction
Cellular therapies for the treatment of diabetes may enable the restoration of glucose-sensing and -secreting machinery to attain physiologic metabolic control. Restoration of beta-cell function is an important therapeutic goal for the treatment of patients with insulin-dependent diabetes [1]. Pancreatic islets are highly specialized glucose sensors that finely regulate glucose metabolism in normal conditions. Functional islet mass becomes lost in an autoimmune process that selectively targets insulin-producing cells in type 1 diabetes (T1D). In type 2 diabetes (T2D), the loss is due to metabolic exhaustion. In these patients, metabolic control throughout the day is very difficult to achieve by current medical therapy using exogenous insulin supply.
Encouraging results from clinical trials have demonstrated the ability of allogeneic islet transplants to impact positively on glycemic control in T1D patients. Transplantation benefits include a significant reduction of mean glycemic aberrances, normalization of glycosylated hemoglobin A1c (HbA1c) values, and the abrogation of severe hypoglycemia. These advances are retained even when exogenous insulin is required after transplantation of an inadequate islet mass, or after graft dysfunction [2]. The metabolic effects are paralleled by a significant improvement in the patients' quality of life [3-8]. Also, a positive impact of islet transplantation on the progression of diabetes complications has been reported. Although, most of the studies showing reduced complications were small-scale and nonrandomized [9-16].
Current limitations of islet transplantation include the limited number of available cadaveric pancreata, which is too small for the number of potential recipients who could benefit from the treatment. Also, after islet infusion in the hepatic portal system of the recipients, a conspicuous mass of functional islets is lost due to poor engraftment. Therefore, a relatively large islet mass (usually more than one donor pancreas) is necessary to attain adequate metabolic control after transplantation. Finally, the need for life-long immunosuppression currently limits the indication of islet transplantation to adults with a brittle form of diabetes associated with recurrent severe hypoglycemia, and hypoglycemia unawareness.
The bone marrow is an invaluable source of adult, pluripotent stem cells. Among others, it gives rise to hematopoietic stem cells (HSC), endothelial progenitor cells (EPC), and mesenchymal stem cells (MSC). Bone marrow cell-derived stem cell (BMSC) transplantation (BMT) can assist in achieving tissue repair and regeneration. Also, this therapy can modulate the immune response in the context of autoimmunity and transplantation (Figure 1).
Figure 1. Schematics of the different cell subsets present in the bone marrow and their potential therapeutic application in the context of diabetes.
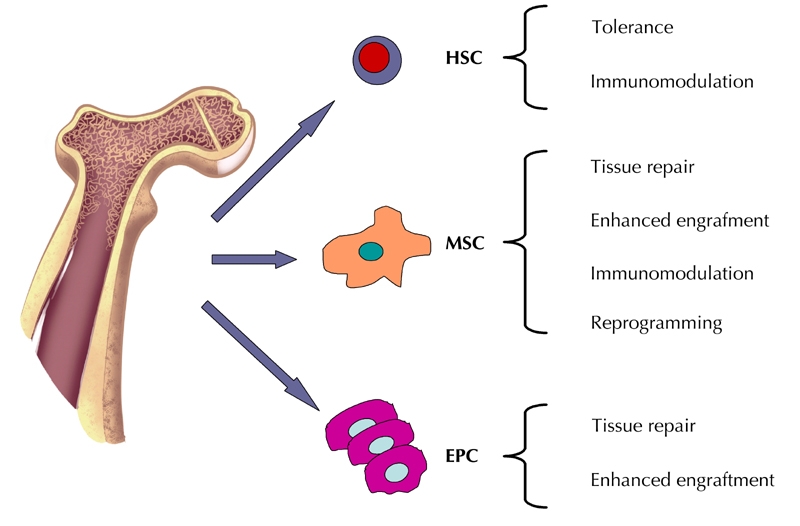
Hematopoietic stem cells (HSC) give rise to hematopoietic lineages. Autologous HSC may be used in combination with lymphodepletion preconditioning to restore self-tolerance in the context of autoimmune diabetes. Whereas, allogeneic HSC may be used to achieve mixed chimerism in recipients of donor-specific insulin-producing cells. The high plasticity of mesenchymal stem cells (MSC) may be exploited to induce tissue repair and modulation of inflammation, leading to the recovery of functional islet cell competence, and to the improvement of diabetes complications. MSC may also contribute to the engraftment of insulin-producing cells by providing trophic factors and/or stimuli in the local microenvironment. Also, it may be possible to reprogram MSC into insulin-producing cells ex vivo from a patient's own cells or from allogeneic donors. Endothelial progenitor cells (EPC) are present in the bone marrow and may be mobilized, or obtained from aspirates, and used to improve tissue repair in islets or diabetic complications. They may also be used to enhance the engraftment of insulin-producing cells.
Bone marrow-derived HSC contribute to the maintenance of hematopoietic cell homeostasis. Transplantation of autologous HSC has been attempted in combination with lymphodepleting protocols, as a means to restore self-tolerance, and halt the progression of autoimmunity in T1D subjects. It is also a means of providing precursor cells for tissue repair in chronic complications of diabetes (discussed in detail below). The use of allogeneic HSC obtained from the same donor of islet cells has been attempted to enhance engraftment, and possibly induce donor-specific acceptance of transplanted tissues. The great plasticity of BMSC includes tissue repair and increases the immune modulation potential of the MSC and EPC components. These advantages offer benefits towards the development of cellular therapies for diabetic complications, and for the preservation of functional beta-cell mass.
An exponential rise in recent publications and clinical trials, reflects the growing interest within the scientific community, in cellular therapies utilizing bone marrow-derived cell products for the treatment of diabetes and its complications. Currently, more than twenty clinical trials on the use of bone BMSC for the treatment of subjects with diabetes are listed in the ClinicalTrials.gov registry (Tables 1-3). This list includes clinical aimed at treating T1D (Table 1), T2D (Table 2), and some of the complications associated with diabetes (Table 3), in the United States and around the world. This manuscript reviews some of the recent research and clinical applications of bone marrow-derived stem cells used for the treatment of insulin-dependent diabetes.
Table 1. Clinical trials using bone marrow-derived stem cells for the treatment of T1D.

Legend: Source: ClinicalTrials.gov (updated August 2010).
Table 3. Clinical trials using bone marrow-derived stem cells for the treatment of diabetes complications.

Legend: Source: ClinicalTrials.gov (updated August 2010).
Table 2. Clinical trials using bone marrow-derived stem cells for the treatment of T2D.

Legend: Source: ClinicalTrials.gov (updated August 2010).
Bone marrow stem cell transplantation to induce hematopoietic chimerism in islet transplant recipients
Clinical and experimental data on the use of donor BMT following myelo- or lymphoablative conditioning, have shown the ability to induce hematopoietic chimerism and graft tolerance in recipients of solid organ transplantation. This enabled the reduction of and complete weaning from immunosuppression [17-20]. Recent trials using high doses of donor CD34+ HSC with minimal or non-ablative recipient conditioning showed successful engraftment, reduced adverse events, immunomodulation, and increased allograft survival [17, 18, 21-26]. During the last two decades, our research center has actively explored the possibility of long-term acceptance of solid organs (multi-visceral, liver, intestine, kidney, and heart) [27-34], and islet grafts, by combining BM-derived HSCs in the clinical setting [2, 35].
Achieving immune tolerance in islet allograft recipients is an appealing prospect, as it may enable durable function of transplanted islets, without life-long adverse side effects from immunosuppression. Currently, almost all classical anti-rejection drugs used in the transplant setting (including, calcineurin-inhibitors, mTOR inhibitors, and steroids) are toxic to islet cells [36-40]. Their continuous use can lead to loss of islet graft function; post-transplant diabetes is a common outcome, even in subjects not prone to diabetes [41]. The induction of hematopoietic chimerism has additional advantages in patients with T1D, as it can enable restoration of self-tolerance, thereby treating the underlying autoimmunity [20].
Several clinical pilot trials using allogeneic islets with donor-specific BMT have been performed at our center over the recent years (Table 4). The BMSC inoculum consisted of non-fractionated or enriched CD34+ BMSC obtained from the vertebral bodies of the same islet donor. The protocols included non-myeloablative immunosuppression. Primary objective of these trials was to induce hematopoietic chimerism and to promote acceptance of islet allografts.
Table 4. Clinical trials combining allogeneic islets and donor bone marrow stem cell transplant performed at the Diabetes Research Institute, University of Miami.

Legend: ALG: anti-lymphocyte globulins. ATG: anti-thymocyte globulin. AZA: azathioprine. BMSC: bone marrow-derived stem cells. C1H: campath-1H. CYA: cyclosporin A. DAC: daclizumab (humanized monoclonal antibody to the alpha subunit of the IL-2 receptor of T cells). ETC: etanercept (TNF inhibitor, functioning as a decoy receptor that binds to TNF). INF: infliximab (monoclonal antibody against TNFα). IAK: islet after kidney. IRDM: insulin-requiring diabetes mellitus. ITA: islet transplantation alone. MMF: mycophenolate mofetil. MP: methylprednisolone. SIR: sirolimus. OKT3: muromonab (murine monoclonal IgG2a antibody directed to the human CD3). SLI: simultaneous islet-liver transplantation. T1D: type 1 diabetes. TAC: tacrolimus.
The first trial (1994-1996) enrolled eight patients who received simultaneous islet-kidney (SIK, n = 7), or islet after kidney (IAK, n = 1) transplantation. The mean islet mass transplanted was 14,800 ± 7,152 IEQ/kg. Two patients received a combined islet preparation from 2 donors. Additionally, 109 non-fractionated donor-specific BMSC per kg body weight were injected on days 5 and 11 after islet transplantation. The immunosuppression protocol consisted of an induction with either the anti-CD3 T cell antibody OKT3 (n = 5), or anti-lymphocyte globulin (ALG, n = 3), followed by maintenance treatment with triple immunosuppression (tacrolimus (TAC), methylprednisolone (MP), and azathioprine (AZA) or mycophenolate mofetil (MMF)). Chimerism levels were monitored in peripheral blood by polymerase chain reaction and flow cytometry [42]. It showed that 7.1 ± 1.4% of donor cells at one month, and ~1% at 6 and 12 months, persisted in the recipients. Overall loss of islet graft function was observed at a mean time of 142 ± 53 days after transplantation (Figure 2). In one patient, rejection of both kidney and islet grafts occurred after treatment with interferon (IFN)-α for acute hepatitis C virus infection. Subsequently, the patient died of complications secondary to the viral infection. Also, an IAK recipient experienced a kidney rejection episode (creatinine 2.5 mg/dl), 3 months after islet transplantation (4 years after kidney transplant). The remaining patients maintained kidney function without rejection episodes during the follow-up period. Development of cytotoxic anti-donor MHC antibodies occurred in six patients, but it was not associated with loss of kidney graft function [43].
Figure 2.
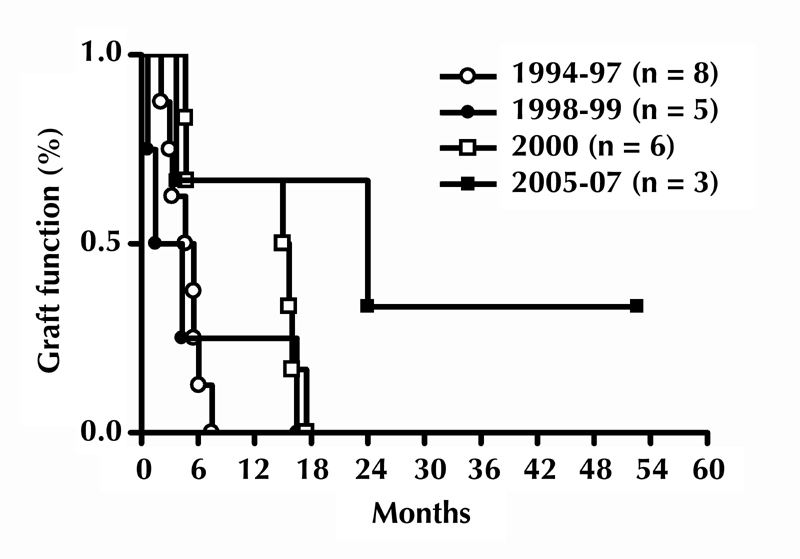
Cumulative graft survival of allogeneic islet grafts combined with donor-specific BMSC transplantation in single, center pilot clinical trials performed at the Diabetes Research Institute of Miami, USA. Graft function was defined as measurable C-peptide.
A subsequent ITA trial (year 1998) in patients with T1D (n = 5) involved intra-hepatic transplantation of allogeneic islets, combined with intravenous infusion of enriched CD34+ BMSC on days 5 and 11 after islet transplantation. In three patients, immunosuppression treatment consisted initially of daclizumab (biweekly for the first 5 doses and monthly for the first year). Maintenance therapy was carried out with TAC, MMF, and MP. Stable islet graft function for over one year was achieved in one patient who received 6,922 IEQ/kg. This patient had measurable C-peptide (0.63 ± 0.14 at one year vs. 0.2 ng/ml pre-transplantation), ~40% reduction of insulin requirements, and improved HbA1c levels (6.9 ± 0.6% post-transplantation vs. 8.1 ± 0.6% pre-transplantation). Chimerism levels declined over time in this patient (7.7%, 3.6%, and 1% at 1, 6, and 12 months, respectively). By protocol design, this subject was weaned from immunosuppression, and subsequently lost graft function (Figure 2). Another patient in this group received 10,536 IEQ/kg, and showed good function (C-peptide: 0.72 ± 0.21 ng/ml). Due to poor tolerance to immunosuppressive drugs, the subject developed graft loss by day 45 post-transplant. A third patient showed primary non-function with no measurable C-peptide after transplantation of 5,774 IEQ/kg. Notably, this subject displayed high basal serum TNF-α levels, which may have contributed to the failure of the islets to engraft. Two additional patients received induction with thymoglobulin (rabbit anti-human thymocyte globulin, ATG), and maintenance with cyclosporine A (CyA) and MMF. One of them showed normalization of HbA1c to 6.4%, and good islet graft function after transplantation of 10,669 IEQ/kg (C-peptide: 0.92 ± 0.26 ng/ml) for approximately 130 days. Then, loss of graft function occurred, possibly due to failure to achieve adequate trough levels of the immunosuppressive drugs. The other patient showed rapid reduction of insulin requirements and good graft function (C-peptide: 0.52 ± 0.46 ng/ml) after receiving 7,981 IEQ/kg, but developed serum sickness syndrome secondary to ATG treatment following the second BMSC infusion. This event required hospitalization. Immunosuppression was stopped, resulting in full recovery from symptoms, but loss of graft function by day 21. Microchimerism was detected in all patients in this study. No episodes of graft-versus-host disease (GvHD) were observed. All patients returned to pre-transplant insulin requirements after stopping immunosuppression.
In the year 2000, we performed an ITA trial (NCT00315614) with Edmonton style immunosuppression and donor-specific BMSC, in five patients with brittle T1D and hypoglycemia unawareness. All subjects received a single intra-hepatic islet infusion on day 0, followed by two intravenous donor-specific CD34+ BMSC treatments on days 5 and 11, post-transplant. One patient received a single infusion of non-fractionated BMSC. Induction treatment consisted of daclizumab (DAC), and a single dose of the anti-tumor necrosis factor (TNF)-α monoclonal antibody infliximab (INF), followed by tacrolimus and sirolimus maintenance [2]. Endpoints of the study included (i) induction of hematopoietic chimerism, and (ii) acceptance of donor-specific islets after stopping immunosuppressive drugs. The latter were tapered gradually in patients with a functional islet graft at one year, as per protocol design. Measurable islet graft function was observed in all study subjects who received 8,629 ± 2,102 IEQ/kg. Loss of graft function was observed in two patients (at 141 and 143 days post-transplantation, respectively), possibly due to failure to achieve therapeutic trough levels of sirolimus. Four patients who received 9,029 ± 2,589 IEQ/kg sustained islet graft function for up to one year, showing maximal mean C-peptide levels at 3 months (1.06 ± 0.23ng/ml), and 68 ± 26% reduction in insulin requirements from baseline. Transient insulin independence was achieved in three patients. Hematopoietic chimerism in peripheral blood was transient in these patients (4.6 ± 0.5% at one month, 0.14 ± 0.05% at one year). Patients with sustained graft function at one year underwent weaning from immunosuppression, as per protocol design. After immunosuppression was stopped, loss of islet graft function invariably occurred at a mean time of 95 ± 25 days (range, 74-131 days) (Figure 2).
The most recent trial (NCT00021801) performed in the years 2005-2007, consisted of ITA in three patients with brittle T1D and donor-specific CD34+ BMSC transplantation on days 5 and 11 after islet transplantation. Lymphodepletion at induction was achieved with Campath-1H (C1H; 20 mg on days -1 and 0). Anti-inflammatory treatment peri-transplant was based on TNF-inhibitor etanercept (ETC) given intravenously before islet infusion (50 mg). Thereafter, it was given subcutaneously, twice weekly for 2 weeks (25 mg). TAC was given continuously from the day before ITA, and adjusted to achieve trough levels of 4-6 ng/ml for three months. Then, patients were switched to MMF maintenance (250 mg a maximum of 1 g BID, calcineurin-sparing protocol). Sirolimus (Rapamune) was given orally from the day before ITA to achieve trough levels of 12-15 ng/ml for 4 months and 7-10 ng/ml thereafter. The mean islet mass transplanted in this small cohort of patients was 6,553 ± 1,272 IEQ/kg. All subjects displayed graft function, with measurable levels of C-peptide, and decreased insulin requirements after transplantation. One patient achieved insulin independence that was sustained for approximately 1.7 years. Then, the patient developed graft dysfunction with complete loss of C-peptide at 2 years. The other two patients displayed sustained graft function for 0.3 and 4.3 years, respectively (Figure 2). Chimerism levels were not detectable at any assessed time point. No major adverse side effects or GvHD were observed in the study subjects.
Collectively, our experience suggests that the use of BMSC with nonmyeloablative immunosuppression is feasible. It is not associated with adverse side effects or with dreadful GvHD (a frequent clinical issue in patients who receive BMT for hematological conditions). Therefore, it may be speculated that lack of harsh treatment to favor stable engraftment of the BMSC inoculum may have contributed to preventing the occurrence of GvHD. The most recent trials showed that overall better outcomes may be ascribed to the steady improvements recorded in the islet transplantation field over the years i.e., islet quality, immunosuppression, and patient management. Possible limitations in our studies include the use of conventional immunosuppression and/or the lack of myeloablation. Our protocols might have precluded adequate engraftment and/or survival of the BMSC inoculum. Indeed, it has been proposed that it is necessary to 'make space' in the recipient's marrow to allow for donor bone marrow cells to engraft [24]. In the two trials performed in 1998 and 2000, immunosuppression was weaned one year after islet transplant, by protocol design. Subsequently, all subjects lost graft function. At that time, there were no sufficiently sensitive immune monitoring methods, nor objective criteria, to guide the decision to stop, or to continue, immunosuppression in the study subjects. More recent studies are trying to identify specific markers and/or molecular signatures of a tolerogenic status in organ transplant recipients [44-48]. Such knowledge may assist the design of stringent rules for future trials.
Potential of bone marrow-derived stem cells to improve islet transplant outcomes
Increasing the number of BMSC in the peripheral blood of intrahepatic islet graft recipients may benefit islet engraftment. In a rodent experimental model of intrahepatic islet transplantation, mobilization of BMSC by administration of granulocyte-macrophage colony-stimulating factor (GM-CSF) resulted in enhanced vascularization and improved function [49]. This phenomenon was associated with increased peripheral blood angioblasts and higher intra-insular vascular density [49].
Tissue engineering approaches may enable manipulation of islet grafts prior to transplantation. The use of marrow-derived MSC and endothelial cells in culture, to coat pancreatic islets, has been proposed as a means to improve islet graft neovascularization and engraftment. This strategy was tested in vitro. It showed that a significantly higher neovasculogenesis in a three-dimensional fibrin gel assay can be achieved, with preservation of islet functional potency [50]. Transfer of this approach to in vivo transplantation settings may contribute to tissue remodeling in the early post-transplant period, and enhance islet engraft and survival.
Another approach to enhance engraftment and survival of allogeneic islet cells is the use of BM-derived MSC. Several properties have been attributed to MSC, including tissue repair and immune modulation. Recently, a trial was performed in a nonhuman primate model of allogeneic islet transplantation. Donor MSC were co-transplanted intraportally with islets, and subsequently, intravenously with donor marrow on postoperative days 5 and 11. It showed that islet engraftment and function assessed at 1 month post-transplant were significantly enhanced compared with control animals that received islets without MSC [51]. Interestingly, additional infusions of donor-specific, or third-party, MSC resulted in reversal of rejection episodes and prolonged islet function in some animals. Stable islet allograft function was associated with increased numbers of T regulatory cells in peripheral blood. These data suggest that MSC may assist in enhancing islet engraftment, and that MSC may represent a viable adjuvant anti-rejection therapy.
Recently, another approach of combined treatment with islets and BMSC has been proposed. It consists of islet graft implantation directly into the recipient's bone marrow [52]. This approach has proven effective in a murine model of syngeneic islet transplantation. Long-term reversal of diabetes and sustained euglycemia were achieved.
Bone marrow-derived stem cell transplantation to preserve/restore beta-cell function in type 1 diabetes
T1D is the consequence of beta-cell destruction by an autoimmune process. Restoration of self-tolerance may preserve functional beta-cell mass, and improve long-term clinical outcome in patients with T1D. In experimental models of T1D, allogeneic BMT has been shown to contribute to the prevention of islet destruction, and restoration of self-tolerance [20]. This has been achieved by induction of lymphodepletion with physical or chemical means, followed by BMT. The use of cellular therapies for the treatment of T1D is gaining momentum, as demonstrated by the increasing number of clinical trials using BMSC that have been registered at ClinicalTrials.gov in recent years (Table 1).
An increasing amount of data documents significant improvements in clinical outcomes following BMT for severe autoimmune diseases [53-55]. A recent trial of autologous BMT in recent onset T1D patients showed encouraging results, in terms of preservation of beta-cell function [56, 57]. Twenty three study subjects with new onset T1D received mobilization of BMSC by cyclophosphamide and granulocyte colony-stimulating factor (G-CSF). Mobilized BMSC were collected by leukapheresis and cryopreserved. The patients underwent a nonmyeloablative, lymphodepleting conditioning protocol consisting of rabbit anti-thymocyte globulin (ATG) and cyclophosphamide, followed by intravenous stem cell infusion (≥3 x 106 CD34+ cells/kg body weight), and subcutaneous treatment with G-CSF. Twenty of the 23 study subjects achieved insulin independence. Twelve subjects were insulin-free for continuous periods (in one case up to 4 years), while 8 patients showed transient insulin independence. Insulin requirements were reduced in most of the subjects, who also showed increased C-peptide production during follow-up. Two subjects developed major complications, consisting of bilateral pneumonia. Several subjects developed fever, urticaria, and/or rash. Three subjects developed endocrine dysfunction (i.e., Graves disease, hypothyroidism, and transient hypogonadism, respectively).
Collectively, this trial is developing quite encouraging. It demonstrates that autoimmune diabetes can be halted. However, the need for harsh preconditioning of the recipients increases the risk of severe morbidity. Therefore, the approach raises important concerns regarding its application in young T1D patients. Also, the long-term effects of this protocol need to be ascertained before it can be considered for large-scale clinical use. Nonetheless, the information gathered from this trial may assist in developing safe immune protocols to restore self-tolerance in T1D.
Recently, a study evaluated the impact of autologous, unfractionated BM-derived mononuclear cells obtained from iliac crest aspirate after mobilization with G-CSF, and injection via superselective cannulation of a pancreatic artery in three subjects who had T1D for more than 5 years [58]. The study showed no effect in terms of C-peptide levels after treatment (both basal and stimulated). The study aimed at enrolling 10 study subjects, but was halted by the local research ethic committee for lack of efficacy [58]. Possible limitations of this trial were lack of immune interventions to favor restoration of self-tolerance, and selection of a target population of diabetics who had undetectable C-peptide levels at baseline.
Bone marrow stem cell transplant in type 2 diabetes
T2D is a metabolic disorder characterized by a combination of insulin resistance and pancreatic beta-cell dysfunction. The latter is the major defect, causing beta-cells to secrete reduced insulin amounts in response to increased glycemic values. The common denominator of beta-cell dysfunction in T1D and T2D is inflammation leading to loss of functional pancreatic endocrine mass and a need for exogenous insulin therapy. Recent data reported in the medical literature suggest that autologous BMT may assist in improving metabolic control in subjects with T2D. In recent years, several trials have been initiated to study the impact of BMSC for the treatment of patients with T2D (Table 2).
A recent prospective phase 1 study of 25 patients, evaluated the impact of autologous BMT on the metabolic control of subjects with T2D [59]. The mononuclear fraction of a buffy coat, obtained from BMSC (from iliac crest aspirates) and peripheral blood, was injected via selective cannulation of the dorsal pancreatic artery, under angiographic guidance. The study subjects underwent a daily 1-h session of hyperbaric oxygen therapy (HOT, 100% at 2.3-2.5 atm), 5 days before and 5 days after BMT. Reductions in nonfasting and postprandial plasma glucose, and HbA1c levels, were observed in these subjects. This result was paralleled by an increase in C-peptide levels over the 1-year follow-up, and by a decrease in the number and dose of oral hypoglycemic drugs or insulin (Figure 3).
Figure 3.
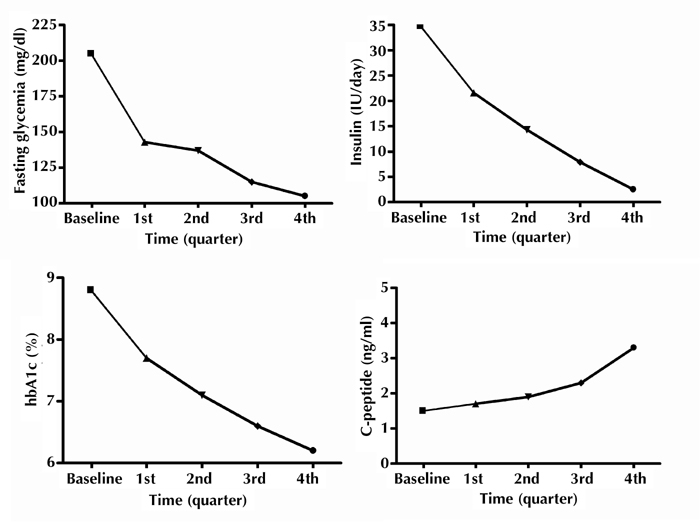
Metabolic control and insulin requirements in a clinical trial of autologous bone marrow stem cell transplantation and hyperbaric oxygen therapy in 25 subjects with T2D (adapted from [59]).
Another clinical trial of autologous BMT was performed in ten human subjects who were negative for glutamic acid decarboxylase (GAD) antibody [60]. The patients were treated with triple oral antidiabetic drugs and insulin (≥0.7 U/kg/day) for ≥1 year. However, the patients failed to respond to this treatment. A mean of 3.5 ± 1.4 x 108 mononuclear cells obtained through posterior iliac spine aspirate were injected via the gastroduodenal artery by transfemoral cannulation. Seven subjects showed reduced insulin requirements (~75% of baseline) by 48 days. Three subjects achieved insulin independence for some time. Also, reduced HbA1c values were recorded in these subjects. In the three responders, this reduction was more pronounced (~7% from baseline). The improved metabolic control after BMT was paralleled by amelioration of fasting and glucagon-stimulated C-peptide levels, and increased HOMA-B. Whereas, no changes in HOMA-IR were observed. Importantly, no serious adverse effects were recorded in this trial.
Overall, both trials showed encouraging results. Numerous studies have shown the beneficial effects of BMT in preventing the progression of the autoimmune process and/or restoring self-tolerance in T1D, both in clinical and experimental settings (see above). However, the underlying mechanisms are currently unknown. It has been hypothesized that BMSC transplant might hamper inflammation and preserve functional beta-cell mass. Also, it might provide precursors and/or signals to favor tissue repair and regeneration. Prospective, controlled, randomized studies on large cohorts of subjects are needed in the clinical setting to characterize this interesting phenomenon.
Conclusions
Cellular therapies offer new opportunities for the treatment of human diseases. The research endeavor is developing rapidly in the areas of regenerative medicine and immunity. However, there are still many challenges ahead. One of which is the regulatory aspect of handling cellular products for transplantation. This regulation requires a great investment burden to implement cell processing methods within a current good manufacturing practice (cGMP) framework. Simultaneously, it is necessary to adhere to standard protocols, establish strict product release criteria, and secure dedicated infrastructures and personnel. In the case of cellular products derived by enrichment and/or by in vitro expansion, all reagents and their sources must be tested and certified for sterility, and for lack of potentially adventitious agents (i.e., endotoxin, etc.). Also, definition of potency, cellular identity, viability, and phenotypical characteristics should be part of the standard assessment performed on each cellular product before transplantation.
Monitoring of clinical trials is essential. This is achievable through institutional review boards (IRB), ethical committees, and independent clinical research organizations. Monitoring protocol implementation and appropriate stopping of trials is necessary to warrant the validity of data generated in clinical trials. Only data from clinical trials that adhere to international ethical standards for human research should be published in peer-reviewed journals. Development of a transplant registry for the collection of clinical trials data may help to interpret results and to assess safety and efficacy. Also, this action may accelerate progress in the field.
Present and future trials will help to assess and optimize safety and efficacy of clinical protocols. They will help to identify the optimal cell type or combination for a specific condition, the optimal cell markers for characterization and product release criteria, and the optimal treatment for a cellular product recipient. Also, further studies will help to find out which primary endpoints enable optimal assessment of efficacy, and how the results of different pilot clinical trials performed at different centers should be compared. The development of multi-center randomized trials may be desirable, but require standardization of cellular processing, and protocols based on the results emerging from pilot clinical studies.
The presence of pluripotent stem cells in the bone marrow makes this compartment an appealing source of cell subsets. Their application in transplantation regimens will hopefully one day be useful in curing diabetes and its complications. Currently, an increasing body of evidence points to the invaluable potential of bone marrow-derived stem cells for diabetes treatment. Furthermore, the encouraging preliminary data from clinical studies recently reported justify cautious optimism for the near future.
Disclosures (conflict of interests statement): The authors report no conflict of interests.
Acknowledgments
This work was supported by National Institutes of Health, National Center for Research Resources, Islet Cell Resources (5U42RR016603-08S1; M01RR16587); National Institute of Diabetes and Digestive & Kidney Diseases (1DP2DK083096-01, 5R01DK059993-04, 1R21DK076098-01, 1U01 DK70460-02, 5R01DK25802-24, 5R01DK56953-05, 5R01DK55347, 5R01DK056953, R01DK025802, 1R21HD060195-01, 1R43DK083832); National Institute of Biomedical Imaging and Bioengineering (1R01 EB008009-02); Cooperative Study Group for Autoimmune Disease Prevention Formation and History; Juvenile Diabetes Research Foundation International (4-2000-946, 4-2000-947, 4-2004-361, 4-2008-811, 17-2010-5); American Diabetes Association; State of Florida; the University of Miami Interdisciplinary Research Development Initiative; a contract for support of this research, sponsored by Congressman Bill Young and funded by a special congressional out of the Navy Bureau of Medicine and Surgery, is currently managed by the Naval Health Research Center, San Diego, CA; Helmsley Charitable Trust; Converge Biotech, Inc.; Biorep Technologies, Inc.; and the continuous support of the Diabetes Research Institute Foundation (www.DiabetesResearch.org). The authors alone are responsible for reporting and interpreting these data; the views expressed herein are those of the authors and not necessarily those of the United States government.
References
- 1.Ricordi C. Islet transplantation: a brave new world. Diabetes. 2003;52:1595–1603. doi: 10.2337/diabetes.52.7.1595. [DOI] [PubMed] [Google Scholar]
- 2.Mineo D, Ricordi C, Xu X, Pileggi A, Garcia-Morales R, Khan A, Baidal DA, Han D, Monroy K, Miller J. et al. Combined islet and hematopoietic stem cell allotransplantation: a clinical pilot trial to induce chimerism and graft tolerance. Am J Transplant. 2008;8:1262–1274. doi: 10.1111/j.1600-6143.2008.02230.x. [DOI] [PubMed] [Google Scholar]
- 3.Benhamou PY, Milliat-Guittard L, Wojtusciszyn A, Kessler L, Toso C, Baertschiger R, Debaty I, Badet L, Penfornis A, Thivolet C. et al. Quality of life after islet transplantation: data from the GRAGIL 1 and 2 trials. Diabet Med. 2009;26:617–621. doi: 10.1111/j.1464-5491.2009.02731.x. [DOI] [PubMed] [Google Scholar]
- 4.Cure P, Pileggi A, Froud T, Messinger S, Faradji RN, Baidal DA, Cardani R, Curry A, Poggioli R, Pugliese A. et al. Improved metabolic control and quality of life in seven patients with type 1 diabetes following islet after kidney transplantation. Transplantation. 2008;85:801–812. doi: 10.1097/TP.0b013e318166a27b. [DOI] [PubMed] [Google Scholar]
- 5.Poggioli R, Faradji RN, Ponte G, Betancourt A, Messinger S, Baidal DA, Froud T, Ricordi C, Alejandro R. Quality of life after islet transplantation. Am J Transplant. 2006;6:371–378. doi: 10.1111/j.1600-6143.2005.01174.x. [DOI] [PubMed] [Google Scholar]
- 6.Speight J, Reaney MD, Woodcock AJ, Smith RM, Shaw JA. Patient-reported outcomes following islet cell or pancreas transplantation (alone or after kidney) in type 1 diabetes: a systematic review. Diabet Med. 2010;27:812–822. doi: 10.1111/j.1464-5491.2010.03029.x. [DOI] [PubMed] [Google Scholar]
- 7.Tharavanij T, Betancourt A, Messinger S, Cure P, Leitao CB, Baidal DA, Froud T, Ricordi C, Alejandro R. Improved long-term health-related quality of life after islet transplantation. Transplantation. 2008;86:1161–1167. doi: 10.1097/TP.0b013e31818a7f45. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 8.Toso C, Shapiro AM, Bowker S, Dinyari P, Paty B, Ryan EA, Senior P, Johnson JA. Quality of life after islet transplant: impact of the number of islet infusions and metabolic outcome. Transplantation. 2007;84:664–666. doi: 10.1097/01.tp.0000280550.01028.89. [DOI] [PubMed] [Google Scholar]
- 9.Lee TC, Barshes NR, O'Mahony CA, Nguyen L, Brunicardi FC, Ricordi C, Alejandro R, Schock AP, Mote A, Goss JA. The effect of pancreatic islet transplantation on progression of diabetic retinopathy and neuropathy. Transplant Proc. 2005;37:2263–2265. doi: 10.1016/j.transproceed.2005.03.011. [DOI] [PubMed] [Google Scholar]
- 10.Venturini M, Fiorina P, Maffi P, Losio C, Vergani A, Secchi A, Del Maschio A. Early increase of retinal arterial and venous blood flow velocities at color Doppler imaging in brittle type 1 diabetes after islet transplant alone. Transplantation. 2006;81:1274–1277. doi: 10.1097/01.tp.0000208631.63235.6a. [DOI] [PubMed] [Google Scholar]
- 11.Del Carro U, Fiorina P, Amadio S, De Toni Franceschini L, Petrelli A, Menini S, Boneschi FM, Ferrari S, Pugliese G, Maffi P, Comi G, Secchi A. Evaluation of polyneuropathy markers in type 1 diabetic kidney transplant patients and effects of islet transplantation: neurophysiological and skin biopsy longitudinal analysis. Diabetes Care. 2007;30:3063–3069. doi: 10.2337/dc07-0206. [DOI] [PubMed] [Google Scholar]
- 12.Thompson DM, Begg IS, Harris C, Ao Z, Fung MA, Meloche RM, Keown P, Meneilly GS, Shapiro RJ, Ho S. et al. Reduced progression of diabetic retinopathy after islet cell transplantation compared with intensive medical therapy. Transplantation. 2008;85:1400–1405. doi: 10.1097/TP.0b013e318172ca07. [DOI] [PubMed] [Google Scholar]
- 13.Fiorina P, Folli F, Bertuzzi F, Maffi P, Finzi G, Venturini M, Socci C, Davalli A, Orsenigo E, Monti L. et al. Long-term beneficial effect of islet transplantation on diabetic macro-/microangiopathy in type 1 diabetic kidney-transplanted patients. Diabetes Care. 2003;26:1129–1136. doi: 10.2337/diacare.26.4.1129. [DOI] [PubMed] [Google Scholar]
- 14.Fiorina P, Folli F, Maffi P, Placidi C, Venturini M, Finzi G, Bertuzzi F, Davalli A, D'Angelo A, Socci C. et al. Islet transplantation improves vascular diabetic complications in patients with diabetes who underwent kidney transplantation: a comparison between kidney-pancreas and kidney-alone transplantation. Transplantation. 2003;75:1296–1301. doi: 10.1097/01.TP.0000061788.32639.D9. [DOI] [PubMed] [Google Scholar]
- 15.Fiorina P, Gremizzi C, Maffi P, Caldara R, Tavano D, Monti L, Socci C, Folli F, Fazio F, Astorri E, Del Maschio A, Secchi A. Islet transplantation is associated with an improvement of cardiovascular function in type 1 diabetic kidney transplant patients. Diabetes Care. 2005;28:1358–1365. doi: 10.2337/diacare.28.6.1358. [DOI] [PubMed] [Google Scholar]
- 16.Fiorina P, Venturini M, Folli F, Losio C, Maffi P, Placidi C, La Rosa S, Orsenigo E, Socci C, Capella C, Del Maschio A, Secchi A. Natural history of kidney graft survival, hypertrophy, and vascular function in end-stage renal disease type 1 diabetic kidney-transplanted patients: beneficial impact of pancreas and successful islet cotransplantation. Diabetes Care. 2005;28:1303–1310. doi: 10.2337/diacare.28.6.1303. [DOI] [PubMed] [Google Scholar]
- 17.Scandling JD, Busque S, Dejbakhsh-Jones S, Benike C, Millan MT, Shizuru JA, Hoppe RT, Lowsky R, Engleman EG, Strober S. Tolerance and chimerism after renal and hematopoietic-cell transplantation. N Engl J Med. 2008;358:362–368. doi: 10.1056/NEJMoa074191. [DOI] [PubMed] [Google Scholar]
- 18.Kawai T, Cosimi AB, Spitzer TR, Tolkoff-Rubin N, Suthanthiran M, Saidman SL, Shaffer J, Preffer FI, Ding R, Sharma V. et al. HLA-mismatched renal transplantation without maintenance immunosuppression. N Engl J Med. 2008;358:353–361. doi: 10.1056/NEJMoa071074. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 19.Sykes M. Immune tolerance: mechanisms and application in clinical transplantation. J Intern Med. 2007;262:288–310. doi: 10.1111/j.1365-2796.2007.01855.x. [DOI] [PubMed] [Google Scholar]
- 20.Inverardi L, Ricordi C. Tolerance and pancreatic islet transplantation. Philos Trans R Soc Lond B Biol Sci. 2001;356:759–765. doi: 10.1098/rstb.2001.0849. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 21.Wu DD, Keating A. Hematopoietic stem cells engraft in untreated transplant recipients. Exp Hematol. 1993;21:251–256. [PubMed] [Google Scholar]
- 22.Ramshaw HS, Crittenden RB, Dooner M, Peters SO, Rao SS, Quesenberry PJ. High levels of engraftment with a single infusion of bone marrow cells into normal unprepared mice. Biol Blood Marrow Transplant. 1995;1:74–80. [PubMed] [Google Scholar]
- 23.Reisner Y, Martelli MF. Bone marrow transplantation across HLA barriers by increasing the number of transplanted cells. Immunol Today. 1995;16:437–440. doi: 10.1016/0167-5699(95)80021-2. [DOI] [PubMed] [Google Scholar]
- 24.Quesenberry PJ, Stewart MF, Peters S, Nillson S, Ramshaw H, Rao S, Tiarks C, Zhong S, Frimberger A, Reilly J. Engraftment of hematopoietic stem cells in nonmyeloablated and myeloablated hosts. Stem Cells. 1997;15(Suppl 1):167–169. doi: 10.1002/stem.5530150821. [DOI] [PubMed] [Google Scholar]
- 25.Rao SS, Peters SO, Crittenden RB, Stewart FM, Ramshaw HS, Quesenberry PJ. Stem cell transplantation in the normal nonmyeloablated host: relationship between cell dose, schedule, and engraftment. Exp Hematol. 1997;25:114–121. [PubMed] [Google Scholar]
- 26.Ricordi C, Karatzas T, Nery J, Webb M, Selvaggi G, Fernandez L, Khan FA, Ruiz P, Schiff E, Olson L. et al. High-dose donor bone marrow infusions to enhance allograft survival: the effect of timing. Transplantation. 1997;63:7–11. doi: 10.1097/00007890-199701150-00003. [DOI] [PubMed] [Google Scholar]
- 27.Fontes P, Rao AS, Demetris AJ, Zeevi A, Trucco M, Carroll P, Rybka W, Rudert WA, Ricordi C, Dodson F. et al. Bone marrow augmentation of donor-cell chimerism in kidney, liver, heart, and pancreas islet transplantation. Lancet. 1994;344:151–155. doi: 10.1016/s0140-6736(94)92756-1. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 28.Fernandez LA, Romaguera R, Viciana AL, Ruiz P, Tzakis AG, Ricordi C. Pulmonary embolism with bone fragments following vertebral body marrow infusion for tolerance induction. Cell Transplant. 1996;5:513–516. doi: 10.1177/096368979600500411. [DOI] [PubMed] [Google Scholar]
- 29.Burke GW, Ricordi C, Karatzas T, Carreno M, Markou M, Cirocco R, Ciancio G, Qian T, Selvaggi G, Alejandro R. et al. Donor bone marrow infusion in simultaneous pancreas/kidney transplantation with OKT3 induction: evidence for augmentation of chimerism. Transplant Proc. 1997;29:1207–1208. doi: 10.1016/s0041-1345(96)00553-2. [DOI] [PubMed] [Google Scholar]
- 30.Garcia-Morales R, Carreno M, Mathew J, Cirocco R, Zucker K, Ciancio G, Burke G, Roth D, Temple D, Fuller L. et al. Continuing observations on the regulatory effects of donor-specific bone marrow cell infusions and chimerism in kidney transplant recipients. Transplantation. 1998;65:956–965. doi: 10.1097/00007890-199804150-00016. [DOI] [PubMed] [Google Scholar]
- 31.Chatzipetrou MA, Mathew JM, Kenyon NS, Esquenazi V, Miller J, Ricordi C, Tzakis AG. Analysis of post-transplant immune status in recipients of liver/bone marrow allografts. Hum Immunol. 1999;60:1281–1288. doi: 10.1016/s0198-8859(99)00115-9. [DOI] [PubMed] [Google Scholar]
- 32.Ciancio G, Miller J, Garcia-Morales RO, Carreno M, Burke GW 3rd, Roth D, Kupin W, Tzakis AG, Ricordi C, Rosen A, Fuller L, Esquenazi V. Six-year clinical effect of donor bone marrow infusions in renal transplant patients. Transplantation. 2001;71:827–835. doi: 10.1097/00007890-200104150-00002. [DOI] [PubMed] [Google Scholar]
- 33.Ciancio G, Burke GW, Garcia-Morales R, Suzart K, Rosen A, Ricordi C, Kenyon NS, Mathew JM, Tzakis AG, Esquenazi V, Miller J. Effect of living-related donor bone marrow infusion on chimerism and in vitro immunoregulatory activity in kidney transplant recipients. Transplantation. 2002;74:488–496. doi: 10.1097/00007890-200208270-00010. [DOI] [PubMed] [Google Scholar]
- 34.Tryphonopoulos P, Tzakis AG, Weppler D, Garcia-Morales R, Kato T, Madariaga JR, Levi DM, Nishida S, Moon J, Selvaggi G. et al. The role of donor bone marrow infusions in withdrawal of immunosuppression in adult liver allotransplantation. Am J Transplant. 2005;5:608–613. doi: 10.1111/j.1600-6143.2004.00743.x. [DOI] [PubMed] [Google Scholar]
- 35.Pileggi A, Ricordi C, Kenyon NS, Froud T, Baidal DA, Kahn A, Selvaggi G, Alejandro R. Twenty years of clinical islet transplantation at the Diabetes Research Institute--University of Miami. Clin Transpl. 2004;2004:177–204. [PubMed] [Google Scholar]
- 36.Ricordi C, Zeng YJ, Alejandro R, Tzakis A, Venkataramanan R, Fung J, Bereiter D, Mintz DH, Starzl TE. In vivo effect of FK506 on human pancreatic islets. Transplantation. 1991;52:519–522. doi: 10.1097/00007890-199109000-00026. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 37.Fernandez LA, Lehmann R, Luzi L, Battezzati A, Angelico MC, Ricordi C, Tzakis A, Alejandro R. The effects of maintenance doses of FK506 versus cyclosporin A on glucose and lipid metabolism after orthotopic liver transplantation. Transplantation. 1999;68:1532–1541. doi: 10.1097/00007890-199911270-00017. [DOI] [PubMed] [Google Scholar]
- 38.Hafiz MM, Faradji RN, Froud T, Pileggi A, Baidal DA, Cure P, Ponte G, Poggioli R, Cornejo A, Messinger S, Ricordi C, Alejandro R. Immunosuppression and procedure-related complications in 26 patients with type 1 diabetes mellitus receiving allogeneic islet cell transplantation. Transplantation. 2005;80:1718–1728. doi: 10.1097/01.tp.0000187881.97068.77. [DOI] [PubMed] [Google Scholar]
- 39.Zahr E, Molano RD, Pileggi A, Ichii H, Jose SS, Bocca N, An W, Gonzalez-Quintana J, Fraker C, Ricordi C, Inverardi L. Rapamycin impairs in vivo proliferation of islet beta-cells. Transplantation. 2007;84:1576–1583. doi: 10.1097/01.tp.0000296035.48728.28. [DOI] [PubMed] [Google Scholar]
- 40.Nir T, Melton DA, Dor Y. Recovery from diabetes in mice by beta cell regeneration. J Clin Invest. 2007;117:2553–2561. doi: 10.1172/JCI32959. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 41.Pham PT, Pham PC, Lipshutz GS, Wilkinson AH. New onset diabetes mellitus after solid organ transplantation. Endocrinol Metab Clin North Am. 2007;36:873–890. doi: 10.1016/j.ecl.2007.07.007. [DOI] [PubMed] [Google Scholar]
- 42.Garcia-Morales R, Esquenazi V, Zucker K, Gomez CI, Fuller L, Carreno M, Cirocco R, Alamo A, Karatzas T, Burke GW 3rd. et al. An assessment of the effects of cadaver donor bone marrow on kidney allograft recipient blood cell chimerism by a novel technique combining PCR and flow cytometry. Transplantation. 1996;62:1149–1160. doi: 10.1097/00007890-199610270-00021. [DOI] [PubMed] [Google Scholar]
- 43.Cardani R, Pileggi A, Ricordi C, Gomez C, Baidal DA, Ponte GG, Mineo D, Faradji RN, Froud T, Ciancio G. et al. Allosensitization of islet allograft recipients. Transplantation. 2007;84:1413–1427. doi: 10.1097/01.tp.0000290388.70019.6e. [DOI] [PubMed] [Google Scholar]
- 44.Koshiba T, Li Y, Takemura M, Wu Y, Sakaguchi S, Minato N, Wood KJ, Haga H, Ueda M, Uemoto S. Clinical, immunological, and pathological aspects of operational tolerance after pediatric living-donor liver transplantation. Transpl Immunol. 2007;17:94–97. doi: 10.1016/j.trim.2006.10.004. [DOI] [PubMed] [Google Scholar]
- 45.Martinez-Llordella M, Puig-Pey I, Orlando G, Ramoni M, Tisone G, Rimola A, Lerut J, Latinne D, Margarit C, Bilbao I. et al. Multiparameter immune profiling of operational tolerance in liver transplantation. Am J Transplant. 2007;7:309–319. doi: 10.1111/j.1600-6143.2006.01621.x. [DOI] [PubMed] [Google Scholar]
- 46.Newell KA, Asare A, Kirk AD, Gisler TD, Bourcier K, Suthanthiran M, Burlingham WJ, Marks WH, Sanz I, Lechler RI. et al. Identification of a B cell signature associated with renal transplant tolerance in humans. J Clin Invest. 2010;120:1836–1847. doi: 10.1172/JCI39933. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 47.Castellaneta A, Thomson AW, Nayyar N, de Vera M, Mazariegos GV. Monitoring the operationally tolerant liver allograft recipient. Curr Opin Organ Transplant. 2010;15:28–34. doi: 10.1097/MOT.0b013e328334269a. [DOI] [PubMed] [Google Scholar]
- 48.Han D, Xu X, Baidal D, Leith J, Ricordi C, Alejandro R, Kenyon NS. Assessment of cytotoxic lymphocyte gene expression in the peripheral blood of human islet allograft recipients: elevation precedes clinical evidence of rejection. Diabetes. 2004;53:2281–2290. doi: 10.2337/diabetes.53.9.2281. [DOI] [PubMed] [Google Scholar]
- 49.Contreras JL, Smyth CA, Eckstein C, Bilbao G, Thompson JA, Young CJ, Eckhoff DE. Peripheral mobilization of recipient bone marrow-derived endothelial progenitor cells enhances pancreatic islet revascularization and engraftment after intraportal transplantation. Surgery. 2003;134:390–398. doi: 10.1067/msy.2003.250. [DOI] [PubMed] [Google Scholar]
- 50.Johansson U, Rasmusson I, Niclou SP, Forslund N, Gustavsson L, Nilsson B, Korsgren O, Magnusson PU. Formation of composite endothelial cell-mesenchymal stem cell islets: a novel approach to promote islet revascularization. Diabetes. 2008;57:2393–2401. doi: 10.2337/db07-0981. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 51.Berman DM, Willman MA, Han D, Kleiner G, Kenyon NM, Cabrera O, Karl JA, Wiseman RW, O'Connor DH, Bartholomew AM, Kenyon NS. Mesenchymal Stem Cells Enhance Allogeneic Islet Engraftment in Nonhuman Primates. Diabetes. 2010 doi: 10.2337/db10-0136. In press. [DOI] [PMC free article] [PubMed] [Google Scholar]
- 52.Cantarelli E, Melzi R, Mercalli A, Sordi V, Ferrari G, Lederer CW, Mrak E, Rubinacci A, Ponzoni M, Sitia G, Guidotti LG, Bonifacio E, Piemonti L. Bone marrow as an alternative site for islet transplantation. Blood. 2009;114:4566–4574. doi: 10.1182/blood-2009-03-209973. [DOI] [PubMed] [Google Scholar]
- 53.Martino G, Franklin RJ, Van Evercooren AB, Kerr DA. Stem cell transplantation in multiple sclerosis: current status and future prospects. Nat Rev Neurol. 2010;6(5):247–255. doi: 10.1038/nrneurol.2010.35. [DOI] [PubMed] [Google Scholar]
- 54.Krauss AC, Kamani NR. Hematopoietic stem cell transplantation for pediatric autoimmune disease: where we stand and where we need to go. Bone Marrow Transplant. 2009;44:137–143. doi: 10.1038/bmt.2009.147. [DOI] [PubMed] [Google Scholar]
- 55.Ikehara S. Stem cell transplantation for autoimmune diseases: what can we learn from experimental models? Autoimmunity. 2008;41:563–569. doi: 10.1080/08916930802197909. [DOI] [PubMed] [Google Scholar]
- 56.Couri CE, Oliveira MC, Stracieri AB, Moraes DA, Pieroni F, Barros GM, Madeira MI, Malmegrim KC, Foss-Freitas MC, Simoes BP. et al. C-peptide levels and insulin independence following autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA. 2009;301:1573–1579. doi: 10.1001/jama.2009.470. [DOI] [PubMed] [Google Scholar]
- 57.Voltarelli JC, Couri CE, Stracieri AB, Oliveira MC, Moraes DA, Pieroni F, Coutinho M, Malmegrim KC, Foss-Freitas MC, Simoes BP, Foss MC, Squiers E, Burt RK. Autologous nonmyeloablative hematopoietic stem cell transplantation in newly diagnosed type 1 diabetes mellitus. JAMA. 2007;297:1568–1576. doi: 10.1001/jama.297.14.1568. [DOI] [PubMed] [Google Scholar]
- 58.Esmatjes E, Montana X, Real MI, Blanco J, Conget I, Casamitjana R, Rovira M, Gomis R, Marin P. Regeneration of insulin production by autologous bone marrow blood autotransplantation in patients with type 1 diabetes. Diabetologia. 2010;53:786–789. doi: 10.1007/s00125-010-1660-9. [DOI] [PubMed] [Google Scholar]
- 59.Estrada EJ, Valacchi F, Nicora E, Brieva S, Esteve C, Echevarria L, Froud T, Bernetti K, Cayetano SM, Velazquez O, Alejandro R, Ricordi C. Combined treatment of intrapancreatic autologous bone marrow stem cells and hyperbaric oxygen in type 2 diabetes mellitus. Cell Transplant. 2008;17:1295–1304. doi: 10.3727/096368908787648119. [DOI] [PubMed] [Google Scholar]
- 60.Bhansali A, Upreti V, Khandelwal N, Marwaha N, Gupta V, Sachdeva N, Sharma RR, Saluja K, Dutta P, Walia R. et al. Efficacy of autologous bone marrow-derived stem cell transplantation in patients with type 2 diabetes mellitus. Stem Cells Dev. 2009;18(10):1407–1416. doi: 10.1089/scd.2009.0164. [DOI] [PubMed] [Google Scholar]


