Abstract
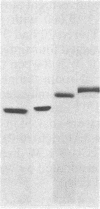
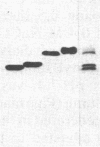
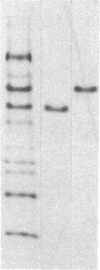
Four endochitinases (poly[1,4-(N-acetyl-β-D-glucosaminide)] glycanohydrolase, EC 3.2.1.14) have been purified from leaves of Nicotiana tabacum cv. Samsun NN reacting hypersensitively to tobacco mosaic virus. Two of them are acidic proteins of molecular weights 27,500 and 28,500 and have been identified as 2 of the 10 pathogenesis-related proteins that are known to accumulate in tobacco in response to stress or pathogen attack. These two pathogenesis-related proteins, named “P” and “Q” when their biological function was unknown, account for one-third of tobacco mosaic virus-induced chitinase activity of tobacco leaves. They are serologically closely related to the two other chitinases, which can be considered as new basic pathogenesis-related proteins. These two basic chitinases exhibit higher molecular weights (32,000 and 34,000) and higher specific enzyme activity than the two acidic isoforms.
Keywords: antimicrobial enzymes, purification, specific activities, serological relationships
Full text
PDF




Images in this article
Selected References
These references are in PubMed. This may not be the complete list of references from this article.
- Blake M. S., Johnston K. H., Russell-Jones G. J., Gotschlich E. C. A rapid, sensitive method for detection of alkaline phosphatase-conjugated anti-antibody on Western blots. Anal Biochem. 1984 Jan;136(1):175–179. doi: 10.1016/0003-2697(84)90320-8. [DOI] [PubMed] [Google Scholar]
- Bradford M. M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal Biochem. 1976 May 7;72:248–254. doi: 10.1016/0003-2697(76)90527-3. [DOI] [PubMed] [Google Scholar]
- Broglie K. E., Gaynor J. J., Broglie R. M. Ethylene-regulated gene expression: molecular cloning of the genes encoding an endochitinase from Phaseolus vulgaris. Proc Natl Acad Sci U S A. 1986 Sep;83(18):6820–6824. doi: 10.1073/pnas.83.18.6820. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Camacho Henriquez A., Sänger H. L. Analysis of acid-extractable tomato leaf proteins after infection with a viroid, two viruses and a fungus and partial purification of the "pathogenesis-related" protein p 14. Arch Virol. 1982;74(2-3):181–196. doi: 10.1007/BF01314711. [DOI] [PubMed] [Google Scholar]
- Conejero V., Semancik J. S. Exocortis viroid: alteration in the proteins of Gynura aurantiaca accompanying viroid infection. Virology. 1977 Mar;77(1):221–232. doi: 10.1016/0042-6822(77)90420-2. [DOI] [PubMed] [Google Scholar]
- Cornelissen B. J., Hooft van Huijsduijnen R. A., Van Loon L. C., Bol J. F. Molecular characterization of messenger RNAs for 'pathogenesis related' proteins la, lb and lc, induced by TMV infection of tobacco. EMBO J. 1986 Jan;5(1):37–40. doi: 10.1002/j.1460-2075.1986.tb04174.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Gianinazzi S., Martin C., Vallée J. C. Hypersensibilité aux virus, température et protéines soubles chez le Nicotiana Xanthi n.c. Apparition de nouvelles macromolécules lors de la répression de la synthèse virale. C R Acad Sci Hebd Seances Acad Sci D. 1970 May 11;270(19):2383–2386. [PubMed] [Google Scholar]
- Hooft van Huijsduijnen R. A., Cornelissen B. J., van Loon L. C., van Boom J. H., Tromp M., Bol J. F. Virus-induced synthesis of messenger RNAs for precursors of pathogenesis-related proteins in tobacco. EMBO J. 1985 Sep;4(9):2167–2171. doi: 10.1002/j.1460-2075.1985.tb03911.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Laemmli U. K. Cleavage of structural proteins during the assembly of the head of bacteriophage T4. Nature. 1970 Aug 15;227(5259):680–685. doi: 10.1038/227680a0. [DOI] [PubMed] [Google Scholar]
- Lucas J., Henriquez A. C., Lottspeich F., Henschen A., Sänger H. L. Amino acid sequence of the ;pathogenesis-related' leaf protein p14 from viroid-infected tomato reveals a new type of structurally unfamiliar proteins. EMBO J. 1985 Nov;4(11):2745–2749. doi: 10.1002/j.1460-2075.1985.tb03998.x. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Mauch F., Hadwiger L. A., Boller T. Ethylene: Symptom, Not Signal for the Induction of Chitinase and beta-1,3-Glucanase in Pea Pods by Pathogens and Elicitors. Plant Physiol. 1984 Nov;76(3):607–611. doi: 10.1104/pp.76.3.607. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Molano J., Polacheck I., Duran A., Cabib E. An endochitinase from wheat germ. Activity on nascent and preformed chitin. J Biol Chem. 1979 Jun 10;254(11):4901–4907. [PubMed] [Google Scholar]
- Morrissey J. H. Silver stain for proteins in polyacrylamide gels: a modified procedure with enhanced uniform sensitivity. Anal Biochem. 1981 Nov 1;117(2):307–310. doi: 10.1016/0003-2697(81)90783-1. [DOI] [PubMed] [Google Scholar]
- REISSIG J. L., STORMINGER J. L., LELOIR L. F. A modified colorimetric method for the estimation of N-acetylamino sugars. J Biol Chem. 1955 Dec;217(2):959–966. [PubMed] [Google Scholar]
- Shinshi H., Mohnen D., Meins F. Regulation of a plant pathogenesis-related enzyme: Inhibition of chitinase and chitinase mRNA accumulation in cultured tobacco tissues by auxin and cytokinin. Proc Natl Acad Sci U S A. 1987 Jan;84(1):89–93. doi: 10.1073/pnas.84.1.89. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Somssich I. E., Schmelzer E., Bollmann J., Hahlbrock K. Rapid activation by fungal elicitor of genes encoding "pathogenesis-related" proteins in cultured parsley cells. Proc Natl Acad Sci U S A. 1986 Apr;83(8):2427–2430. doi: 10.1073/pnas.83.8.2427. [DOI] [PMC free article] [PubMed] [Google Scholar]
- Towbin H., Staehelin T., Gordon J. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedure and some applications. Proc Natl Acad Sci U S A. 1979 Sep;76(9):4350–4354. doi: 10.1073/pnas.76.9.4350. [DOI] [PMC free article] [PubMed] [Google Scholar]